Structure–Activity Relationships of the Tetrapeptide Ac-His-Arg-(pI)DPhe-Tic-NH2 at the Mouse Melanocortin Receptors: Modification at the (pI)DPhe Position Leads to mMC3R Versus mMC4R Selective Ligands
Abstract
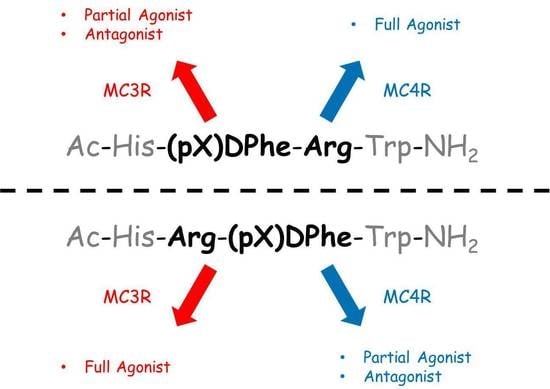
:1. Introduction
2. Results
Peptide Synthesis and Pharmacological Evaluation
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Peptide Synthesis
4.3. AlphaScreen Bioassay
4.4. β-Galactosidase Assay
4.5. Data Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chhajlani, V.; Muceniece, R.; Wikberg, J.E. Molecular cloning of a novel human melanocortin receptor. Biochem. Biophys. Res. Commun. 1993, 195, 866–873. [Google Scholar] [CrossRef]
- Chhajlani, V.; Wikberg, J.E. Molecular cloning and expression of the human melanocyte stimulating hormone receptor cDNA. FEBS Lett. 1992, 309, 417–420. [Google Scholar] [CrossRef] [Green Version]
- Mountjoy, K.G.; Robbins, L.S.; Mortrud, M.T.; Cone, R.D. The cloning of a family of genes that encode the melanocortin receptors. Science 1992, 257, 1248–1251. [Google Scholar] [CrossRef] [PubMed]
- Roselli-Rehfuss, L.; Mountjoy, K.G.; Robbins, L.S.; Mortrud, M.T.; Low, M.J.; Tatro, J.B.; Entwistle, M.L.; Simerly, R.B.; Cone, R.D. Identification of a receptor for γ melanotropin and other proopiomelanocortin peptides in the hypothalamus and limbic system. Proc. Natl. Acad. Sci. USA 1993, 90, 8856–8860. [Google Scholar] [CrossRef] [PubMed]
- Gantz, I.; Konda, Y.; Tashiro, T.; Shimoto, Y.; Miwa, H.; Munzert, G.; Watson, S.J.; DelValle, J.; Yamada, T. Molecular cloning of a novel melanocortin receptor. J. Biol. Chem. 1993, 268, 8246–8250. [Google Scholar] [PubMed]
- Gantz, I.; Miwa, H.; Konda, Y.; Shimoto, Y.; Tashiro, T.; Watson, S.J.; DelValle, J.; Yamada, T. Molecular cloning, expression, and gene localization of a fourth melanocortin receptor. J. Biol. Chem. 1993, 268, 15174–15179. [Google Scholar] [PubMed]
- Gantz, I.; Shimoto, Y.; Konda, Y.; Miwa, H.; Dickinson, C.J.; Yamada, T. Molecular cloning, expression, and characterization of a fifth melanocortin receptor. Biochem. Biophys. Res. Commun. 1994, 200, 1214–1220. [Google Scholar] [CrossRef] [PubMed]
- Griffon, N.; Mignon, V.; Facchinetti, P.; Diaz, J.; Schwartz, J.C.; Sokoloff, P. Molecular cloning and characterization of the rat fifth melanocortin receptor. Biochem. Biophys. Res. Commun. 1994, 200, 1007–1014. [Google Scholar] [CrossRef]
- Haynes, R.C. The activation of adrenal phosphorylase by the adreno-corticotropic hormone. J. Biol. Chem. 1958, 233, 1220–1222. [Google Scholar] [PubMed]
- Schioth, H.B.; Chhajlani, V.; Muceniece, R.; Klusa, V.; Wikberg, J.E. Major pharmacological distinction of the ACTH receptor from other melanocortin receptors. Life Sci. 1996, 59, 797–801. [Google Scholar] [CrossRef]
- Butler, A.A.; Kesterson, R.A.; Khong, K.; Cullen, M.J.; Pelleymounter, M.A.; Dekoning, J.; Baetscher, M.; Cone, R.D. A unique metabolic syndrome causes obesity in the melanocortin-3 receptor-deficient mouse. Endocrinology 2000, 141, 3518–3521. [Google Scholar] [CrossRef]
- Chen, A.S.; Marsh, D.J.; Trumbauer, M.E.; Frazier, E.G.; Guan, X.M.; Yu, H.; Rosenblum, C.I.; Vongs, A.; Feng, Y.; Cao, L.H.; et al. Inactivation of the mouse melanocortin-3 receptor results in increased fat mass and reduced lean body mass. Nat. Genet. 2000, 26, 97–102. [Google Scholar] [CrossRef]
- Huszar, D.; Lynch, C.A.; Fairchild-Huntress, V.; Dunmore, J.H.; Fang, Q.; Berkemeier, L.R.; Gu, W.; Kesterson, R.A.; Boston, B.A.; Cone, R.D.; et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 1997, 88, 131–141. [Google Scholar] [CrossRef]
- Fan, W.; Boston, B.A.; Kesterson, R.A.; Hruby, V.J.; Cone, R.D. Role of melanocortinergic neurons in feeding and the agouti obesity syndrome. Nature 1997, 385, 165–168. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Kelly, M.A.; Opitz-Araya, X.; Thomas, R.E.; Low, M.J.; Cone, R.D. Exocrine gland dysfunction in MC5-R-deficient mice: Evidence for coordinated regulation of exocrine gland function by melanocortin peptides. Cell 1997, 91, 789–798. [Google Scholar] [CrossRef]
- Nakanishi, S.; Inoue, A.; Kita, T.; Nakamura, M.; Chang, A.C.; Cohen, S.N.; Numa, S. Nucleotide sequence of cloned cDNA for bovine corticotropin-β-lipotropin precursor. Nature 1979, 278, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Eipper, B.A.; Mains, R.E. Structure and biosynthesis of pro-adrenocorticotropin/endorphin and related peptides. Endocr. Rev. 1980, 1, 1–27. [Google Scholar] [CrossRef]
- Smith, A.I.; Funder, J.W. Proopiomelanocortin processing in the pituitary, central nervous system, and peripheral tissues. Endocr. Rev. 1988, 9, 159–179. [Google Scholar] [CrossRef]
- Hruby, V.J.; Wilkes, B.C.; Hadley, M.E.; Al-Obeidi, F.; Sawyer, T.K.; Staples, D.J.; Devaux, A.E.; Dym, O.; Castrucci, A.M.D.; Hintz, M.F.; et al. α-Melanotropin: The minimal active sequence in the frog-skin bioassay. J. Med. Chem. 1987, 30, 2126–2130. [Google Scholar] [CrossRef] [PubMed]
- Castrucci, A.M.; Hadley, M.E.; Sawyer, T.K.; Wilkes, B.C.; Al-Obeidi, F.; Staples, D.J.; de Vaux, A.E.; Dym, O.; Hintz, M.F.; Riehm, J.P.; et al. α-Melanotropin: The minimal active sequence in the lizard skin bioassay. Gen. Comp. Endocrinol. 1989, 73, 157–163. [Google Scholar] [CrossRef]
- Kiefer, L.L.; Veal, J.M.; Mountjoy, K.G.; Wilkinson, W.O. Melanocortin receptor binding determinants in the agouti protein. Biochemistry 1998, 37, 991–997. [Google Scholar] [CrossRef]
- Tota, M.R.; Smith, T.S.; Mao, C.; MacNeil, T.; Mosley, R.T.; Van der Ploeg, L.H.T.; Fong, T.M. Molecular interaction of agouti protein and agouti-related protein with human melanocortin receptors. Biochemistry 1999, 38, 897–904. [Google Scholar] [CrossRef]
- Irani, B.G.; Xiang, Z.M.; Yarandi, H.N.; Holder, J.R.; Moore, M.C.; Bauzo, R.M.; Proneth, B.; Shaw, A.M.; Millard, W.J.; Chambers, J.B.; et al. Implication of the melanocortin-3 receptor in the regulation of food intake. Eur. J. Pharmacol. 2011, 660, 80–87. [Google Scholar] [CrossRef] [Green Version]
- Atalayer, D.; Robertson, K.L.; Haskell-Luevano, C.; Andreasen, A.; Rowland, N.E. Food demand and meal size in mice with single or combined disruption of melanocortin type 3 and 4 receptors. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R1667–R1674. [Google Scholar] [CrossRef] [Green Version]
- Rowland, N.E.; Fakhar, K.J.; Robertson, K.L.; Haskell-Luevano, C. Effect of serotonergic anorectics on food intake and induction of Fos in brain of mice with disruption of melanocortin 3 and/or 4 receptors. Pharmacol. Biochem. Behav. 2010, 97, 107–111. [Google Scholar] [CrossRef] [Green Version]
- Rowland, N.E.; Schaub, J.W.; Robertson, K.L.; Andreasen, A.; Haskell-Luevano, C. Effect of MTII on food intake and brain c-Fos in melanocortin-3, melanocortin-4, and double MC3 and MC4 receptor knockout mice. Peptides 2010, 31, 2314–2317. [Google Scholar] [CrossRef]
- Brown, K.S.; Gentry, R.M.; Rowland, N.E. Central injection in rats of α-melanocyte-stimulating hormone analog: Effects on food intake and brain Fos. Regul. Pept. 1998, 78, 89–94. [Google Scholar] [CrossRef]
- Ebihara, K.; Ogawa, Y.; Katsuura, G.; Numata, Y.; Masuzaki, H.; Satoh, N.; Tamaki, M.; Yoshioka, T.; Hayase, M.; Matsuoka, N.; et al. Involvement of agouti-related protein, an endogenous antagonist of hypothalamic melanocortin receptor, in leptin action. Diabetes 1999, 48, 2028–2033. [Google Scholar] [CrossRef]
- Hinney, A.; Volckmar, A.L.; Knoll, N. Melanocortin-4 receptor in energy homeostasis and obesity pathogenesis. Prog. Mol. Biol. Transl. Sci. 2013, 114, 147–191. [Google Scholar]
- Greenfield, J.R.; Miller, J.W.; Keogh, J.M.; Henning, E.; Satterwhite, J.H.; Cameron, G.S.; Astruc, B.; Mayer, J.P.; Brage, S.; See, T.C.; et al. Modulation of blood pressure by central melanocortinergic pathways. N. Engl. J. Med. 2009, 360, 44–52. [Google Scholar] [CrossRef]
- Dorr, R.T.; Lines, R.; Levine, N.; Brooks, C.; Xiang, L.; Hruby, V.J.; Hadley, M.E. Evaluation of melanotan-II, a superpotent cyclic melanotropic peptide in a pilot phase-I clinical study. Life Sci. 1996, 58, 1777–1784. [Google Scholar] [CrossRef]
- Lansdell, M.I.; Hepworth, D.; Calabrese, A.; Brown, A.D.; Blagg, J.; Burring, D.J.; Wilson, P.; Fradet, D.; Brown, T.B.; Quinton, F.; et al. Discovery of a Selective Small-Molecule Melanocortin-4 Receptor Agonist with Efficacy in a Pilot Study of Sexual Dysfunction in Humans. J. Med. Chem. 2010, 53, 3183–3197. [Google Scholar] [CrossRef] [PubMed]
- Hadley, M.E. Discovery that a melanocortin regulates sexual functions in male and female humans. Peptides 2005, 26, 1687–1689. [Google Scholar] [CrossRef]
- Kuhnen, P.; Clement, K.; Wiegand, S.; Blankenstein, O.; Gottesdiener, K.; Martini, L.L.; Mai, K.; Blume-Peytavi, U.; Gruters, A.; Krude, H. Proopiomelanocortin deficiency treated with a melanocortin-4 receptor agonist. N. Engl. J. Med. 2016, 375, 240–246. [Google Scholar] [CrossRef]
- Clement, K.; Biebermann, H.; Farooqi, I.S.; Van der Ploeg, L.; Wolters, B.; Poitou, C.; Puder, L.; Fiedorek, F.; Gottesdiener, K.; Kleinau, G.; et al. MC4R agonism promotes durable weight loss in patients with leptin receptor deficiency. Nat. Med. 2018, 24, 551–555. [Google Scholar] [CrossRef]
- Ni, X.P.; Butler, A.A.; Cone, R.D.; Humphreys, M.H. Central receptors mediating the cardiovascular actions of melanocyte stimulating hormones. J. Hypertens. 2006, 24, 2239–2246. [Google Scholar] [CrossRef]
- Martin, W.J.; McGowan, E.; Cashen, D.E.; Gantert, L.T.; Drisko, J.E.; Hom, G.J.; Nargund, R.; Sebhat, I.; Howard, A.D.; Van der Ploeg, L.H.T.; et al. Activation of melanocortin MC4 receptors increases erectile activity in rats ex copula. Eur. J. Pharmacol. 2002, 454, 71–79. [Google Scholar] [CrossRef]
- Van der Ploeg, L.H.T.; Martin, W.J.; Howard, A.D.; Nargund, R.P.; Austin, C.P.; Guan, X.M.; Drisko, J.; Cashen, D.; Sebhat, I.; Patchett, A.A.; et al. A role for the melanocortin 4 receptor in sexual function. Proc. Natl. Acad. Sci. USA 2002, 99, 11381–11386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Z.; Tao, Y.X. Mutations in melanocortin-3 receptor gene and human obesity. Prog. Mol. Biol. Transl. Sci. 2016, 140, 97–129. [Google Scholar] [PubMed]
- Doering, S.R.; Freeman, K.T.; Schnell, S.M.; Haslach, E.M.; Dirain, M.; Debevec, G.; Geer, P.; Santos, R.G.; Giulianotti, M.A.; Pinilla, C.; et al. Discovery of mixed pharmacology melanocortin-3 agonists and melanocortin-4 receptor tetrapeptide antagonist compounds (TACOs) based on the sequence Ac-Xaa(1)-Arg-(pI)DPhe-Xaa(4)-NH2. J. Med. Chem. 2017, 60, 4342–4357. [Google Scholar] [CrossRef]
- Fleming, K.A.; Freeman, K.T.; Powers, M.D.; Santos, R.G.; Debevec, G.; Giulianotti, M.A.; Houghten, R.A.; Doering, S.R.; Pinilla, C.; Haskell-Luevano, C. Discovery of polypharmacological melanocortin-3 and -4 receptor probes and identification of a 100-fold selective nM MC3R agonist versus a µM MC4R partial agonist. J. Med. Chem. 2019, 62, 2738–2749. [Google Scholar] [CrossRef]
- Proneth, B.; Pogozheva, I.D.; Portillo, F.P.; Mosberg, H.I.; Haskell-Luevano, C. Melanocortin tetrapeptide Ac-His-DPhe-Arg-Trp-NH(2) modified at the para position of the benzyl side chain (DPhe): Importance for mouse melanocortin-3 receptor agonist versus antagonist activity. J. Med. Chem. 2008, 51, 5585–5593. [Google Scholar] [CrossRef] [PubMed]
- Holder, J.R.; Bauzo, R.M.; Xiang, Z.M.; Haskell-Luevano, C. Structure-activity relationships of the melanocortin tetrapeptide Ac-His-DPhe-Arg-Trp-NH2 at the mouse melanocortin receptors: Part 2 modifications at the Phe position. J. Med. Chem. 2002, 45, 3073–3081. [Google Scholar] [CrossRef] [PubMed]
- Carpino, L.A.; Han, G.Y. 9-Fluorenylmethoxycarbonyl function, a new base-sensitive amino-protecting group. J. Am. Chem. Soc. 1970, 92, 5748–5749. [Google Scholar] [CrossRef]
- Carpino, L.A.; Han, G.Y. The 9-fluorenylmethoxycarbonyl amino-protecting group. J. Org. Chem. 1972, 37, 3404–3409. [Google Scholar] [CrossRef]
- Chen, W.B.; Shields, T.S.; Stork, P.J.S.; Cone, R.D. A colorimetric assay for measuring activation of Gs- and Gq-coupled signaling pathways. Anal. Biochem. 1995, 226, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Ericson, M.D.; Schnell, S.M.; Freeman, K.T.; Haskell-Luevano, C. A fragment of the Escherichia coli ClpB heat-shock protein is a micromolar melanocortin 1 receptor agonist. Bioorg. Med. Chem. Lett. 2015, 25, 5306–5308. [Google Scholar] [CrossRef] [PubMed]
- Tala, S.R.; Schnell, S.M.; Haskell-Luevano, C. Microwave-assisted solid-phase synthesis of side-chain to side-chain lactam-bridge cyclic peptides. Bioorg. Med. Chem. Lett. 2015, 25, 5708–5711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, A.; Tala, S.R.; Flores, V.; Freeman, K.; Haskell-Luevano, C. Synthesis and pharmacology of α/β3-peptides based on the melanocortin agonist Ac-His-DPhe-Arg-Trp-NH2 sequence. ACS Med. Chem. Lett. 2015, 6, 568–572. [Google Scholar] [CrossRef]
- Sawyer, T.K.; Sanfilippo, P.J.; Hruby, V.J.; Engel, M.H.; Heward, C.B.; Burnett, J.B.; Hadley, M.E. 4-Norleucine, 7-D-phenylalanine-α-melanocyte-stimulating hormone—A highly potent α-melanotropin with ultralong biological-activity. Proc. Natl. Acad. Sci. USA 1980, 77, 5754–5758. [Google Scholar] [CrossRef]
- Haskell-Luevano, C.; Holder, J.R.; Monck, E.K.; Bauzo, R.M. Characterization of melanocortin NDP-MSH agonist peptide fragments at the mouse central and peripheral melanocortin receptors. J. Med. Chem. 2001, 44, 2247–2252. [Google Scholar] [CrossRef]
- Schild, H.O. pA, a new scale for the measurement of drug antagonism. Br. J. Pharmacol. Chemother. 1947, 2, 189–206. [Google Scholar] [CrossRef]
- Haghighi, S.M.; Zhou, Y.; Dai, J.X.; Sawyer, J.R.; Hruby, V.J.; Cai, M.Y. Replacement of Arg with Nle and modified D-Phe in the core sequence of MSHs, Ac-His-D-Phe-Arg-Trp-NH2, leads to hMC1R selectivity and pigmentation. Eur. J. Med. Chem. 2018, 151, 815–823. [Google Scholar] [CrossRef]
- Kaiser, E.; Colescott, R.L.; Bossinger, C.D.; Cook, P.I. Color test for detection of free terminal amino groups in the solid-phase synthesis of peptides. Anal. Biochem. 1970, 34, 595–598. [Google Scholar] [CrossRef]
- Christensen, T. Qualitative test for monitoring coupling completeness in solid-phase peptide-synthesis using chloranil. Acta Chem. Scand. Ser. B 1979, 33, 763–766. [Google Scholar] [CrossRef]
- Lensing, C.J.; Freeman, K.T.; Schnell, S.M.; Adank, D.N.; Speth, R.C.; Haskell-Luevano, C. An in vitro and in vivo investigation of bivalent ligands that display preferential binding and functional activity for different melanocortin receptor homodimers. J. Med. Chem. 2016, 59, 3112–3128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Sample Availability: Samples of the compounds are available from the authors. |



| Peptide | Sequence | Retention Time (min) | M (Calculated) | M + H (Observed) | Purity (%) | |
|---|---|---|---|---|---|---|
| System 1 | System 2 | |||||
| KNS2-153 | Ac-His-DPhe-Arg-Trp-NH2 | 10.1 | 15.6 | 685.3 | 686.4 | >98 |
| KNS2-22-4 | Ac-His-Arg-(pI)DPhe-Tic-NH2 | 14.9 | 23.6 | 784.2 | 785.3 | >97 |
| KNS2-22-3 | Ac-His-Arg-(pBr)DPhe-Tic-NH2 | 14.9 | 23.2 | 736.3, 738.3 b | 737.3, 739.3 b | >98 |
| KNS2-22-1 | Ac-His-Arg-(pCl)DPhe-Tic-NH2 | 14.6 | 22.7 | 692.3 | 693.5 | >97 |
| KNS2-22-2 | Ac-His-Arg-(pF)DPhe-Tic-NH2 | 13.5 | 20.8 | 676.3 | 677.5 | >95 |
| KNS3-10 | Ac-His-Arg-DPhe-Tic-NH2 | 12.8 | 20.1 | 658.3 | 659.5 | >99 |
| KNS2-23-4 | Ac-His-Arg-(3,4-diCl)DPhe-Tic-NH2 | 15.6 | 24.2 | 726.3 | 727.4 | >97 |
| KNS2-23-7 | Ac-His-Arg-(pMe)DPhe-Tic-NH2 | 14.3 | 22.3 | 672.4 | 673.5 | >97 |
| KNS2-23-6 | Ac-His-Arg-(pCF3)DPhe-Tic-NH2 | 15.0 | 23.4 | 726.7 | 727.5 | >98 |
| KNS2-23-3 | Ac-His-Arg-(ptBu)DPhe-Tic-NH2 | 17.5 | 26.5 | 714.4 | 715.4 | >95 |
| KNS2-23-1 | Ac-His-Arg-DBip-Tic-NH2 | 16.7 | 25.9 | 734.3 | 735.5 | >96 |
| KNS2-23-9 | Ac-His-Arg-DTyr-Tic-NH2 | 10.5 | 15.0 | 674.3 | 675.4 | >96 |
| KNS2-23-8 | Ac-His-Arg-(pCN)DPhe-Tic-NH2 | 11.5 | 17.2 | 683.3 | 684.3 | >97 |
| Peptide | Sequence | mMC1R EC50 (nM) |
|---|---|---|
| NDP-MSH | Ac-Ser-Tyr-Ser-Nle-Glu-His-DPhe-Arg-Trp-Gly-Lys-Pro-Val-NH2 | 0.009 ± 0.002 |
| KNS2-153 | Ac-His-DPhe-Arg-Trp-NH2 | 10 ± 3 |
| KNS2-22-4 | Ac-His-Arg-(pI)DPhe-Tic-NH2 | 0.7 ± 0.2 |
| KNS2-22-3 | Ac-His-Arg-(pBr)DPhe-Tic-NH2 | 0.7 ± 0.3 |
| KNS2-22-1 | Ac-His-Arg-(pCl)DPhe-Tic-NH2 | 0.8 ± 0.2 |
| KNS2-22-2 | Ac-His-Arg-(pF)DPhe-Tic-NH2 | 1.8 ± 0.7 |
| KNS3-10 | Ac-His-Arg-DPhe-Tic-NH2 | 4.6 ± 0.4 |
| KNS2-23-4 | Ac-His-Arg-(3,4-diCl)DPhe-Tic-NH2 | 5 ± 2 |
| KNS2-23-7 | Ac-His-Arg-(pMe)DPhe-Tic-NH2 | 1.0 ± 0.3 |
| KNS2-23-6 | Ac-His-Arg-(pCF3)DPhe-Tic-NH2 | 5 ± 1 |
| KNS2-23-3 | Ac-His-Arg-(ptBu)DPhe-Tic-NH2 | 9 ± 3 |
| KNS2-23-1 | Ac-His-Arg-DBip-Tic-NH2 | 0.6 ± 0.1 |
| KNS2-23-9 | Ac-His-Arg-DTyr-Tic-NH2 | 40 ± 10 |
| KNS-2-23-8 | Ac-His-Arg-(pCN)DPhe-Tic-NH2 | 27 ± 6 |
| Peptide | Sequence | mMC3R EC50 (nM) | mMC4R | mMC5R EC50 (nM) | |
|---|---|---|---|---|---|
| EC50 (nM) | pA2 | ||||
| NDP-MSH | Ac-Ser-Tyr-Ser-Nle-Glu-His-DPhe-Arg-Trp-Gly-Lys-Pro-Val-NH2 | 0.52 ± 0.05 | 0.32 ± 0.02 | - | 3.4 ± 0.7 |
| KNS2-153 | Ac-His-DPhe-Arg-Trp-NH2 | 190 ± 40 | 12 ± 3 | - | 5 ± 2 |
| KNS2-22-4 | Ac-His-Arg-(pI)DPhe-Tic-NH2 | 13 ± 2 | Partial Agonist 150 ± 40 (40% NDP) | 7.3 ± 0.8 | 5 ± 1 |
| KNS2-22-3 | Ac-His-Arg-(pBr)DPhe-Tic-NH2 | 90 ± 20 | Partial Agonist 290 ± 50 (55% NDP) | - | 9.7 ± 0.5 |
| KNS2-22-1 | Ac-His-Arg-(pCl)DPhe-Tic-NH2 | 120 ± 20 | Partial Agonist 280 ± 60 (70% NDP) | - | 18 ± 6 |
| KNS2-22-2 | Ac-His-Arg-(pF)DPhe-Tic-NH2 | 450 ± 70 | Partial Agonist 560 ± 60 (70% NDP) | - | 70 ± 40 |
| KNS3-10 | Ac-His-Arg-DPhe-Tic-NH2 | Partial Agonist 900 ± 200 (85% NDP) | 3000 ± 2000 | - | Partial Agonist 200 ± 30 (65% NDP) |
| KNS2-23-4 | Ac-His-Arg-(3,4-diCl)DPhe-Tic-NH2 | 400 ± 100 | >100,000 | 6.15 ± 0.05 | 70 ± 7 |
| KNS2-23-7 | Ac-His-Arg-(pMe)DPhe-Tic-NH2 | 110 ± 20 | Partial Agonist 700 ± 200 (50% NDP) | - | 17 ± 4 |
| KNS2-23-6 | Ac-His-Arg-(pCF3)DPhe-Tic-NH2 | 90 ± 30 | Partial Agonist 600 ± 300 (20% NDP) | 6.5 ± 0.2 | 13 ± 4 |
| KNS2-23-3 | Ac-His-Arg-(ptBu)DPhe-Tic-NH2 | Partial Agonist 13 ± 4 (85% NDP) | >100,000 | 6.8 ± 0.3 | 3.4 ± 0.3 |
| KNS2-23-1 | Ac-His-Arg-DBip-Tic-NH2 | 14 ± 2 | Partial Agonist 1400 ± 700 (45% NDP) | 5.9 ± 0.2 | 7.6 ± 0.7 |
| KNS2-23-9 | Ac-His-Arg-DTyr-Tic-NH2 | Partial Agonist 4200 ± 800 (85% NDP) | >100,000 | <5.5 | 1000 ± 500 |
| KNS2-23-8 | Ac-His-Arg-(pCN)DPhe-Tic-NH2 | Partial Agonist 4000 ± 1000 (75% NDP) | 40% @ 100 µM | <5.5 | 500 ± 100 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schlasner, K.N.; Ericson, M.D.; Doering, S.R.; Freeman, K.T.; Weinrich, M.; Haskell-Luevano, C. Structure–Activity Relationships of the Tetrapeptide Ac-His-Arg-(pI)DPhe-Tic-NH2 at the Mouse Melanocortin Receptors: Modification at the (pI)DPhe Position Leads to mMC3R Versus mMC4R Selective Ligands. Molecules 2019, 24, 1463. https://doi.org/10.3390/molecules24081463
Schlasner KN, Ericson MD, Doering SR, Freeman KT, Weinrich M, Haskell-Luevano C. Structure–Activity Relationships of the Tetrapeptide Ac-His-Arg-(pI)DPhe-Tic-NH2 at the Mouse Melanocortin Receptors: Modification at the (pI)DPhe Position Leads to mMC3R Versus mMC4R Selective Ligands. Molecules. 2019; 24(8):1463. https://doi.org/10.3390/molecules24081463
Chicago/Turabian StyleSchlasner, Katherine N., Mark D. Ericson, Skye R. Doering, Katie T. Freeman, Mary Weinrich, and Carrie Haskell-Luevano. 2019. "Structure–Activity Relationships of the Tetrapeptide Ac-His-Arg-(pI)DPhe-Tic-NH2 at the Mouse Melanocortin Receptors: Modification at the (pI)DPhe Position Leads to mMC3R Versus mMC4R Selective Ligands" Molecules 24, no. 8: 1463. https://doi.org/10.3390/molecules24081463