Ethnopharmacology of Fruit Plants: A Literature Review on the Toxicological, Phytochemical, Cultural Aspects, and a Mechanistic Approach to the Pharmacological Effects of Four Widely Used Species
Abstract
:1. Introduction
2. Fruit Plants from the Brazilian Cerrado
3. Sapindaceae Family
3.1. Genus Talisia and Species Talisia esculenta
3.1.1. Geographic Distribution and Popular Use
3.1.2. Botanical Aspects
3.1.3. Phytochemical Aspects
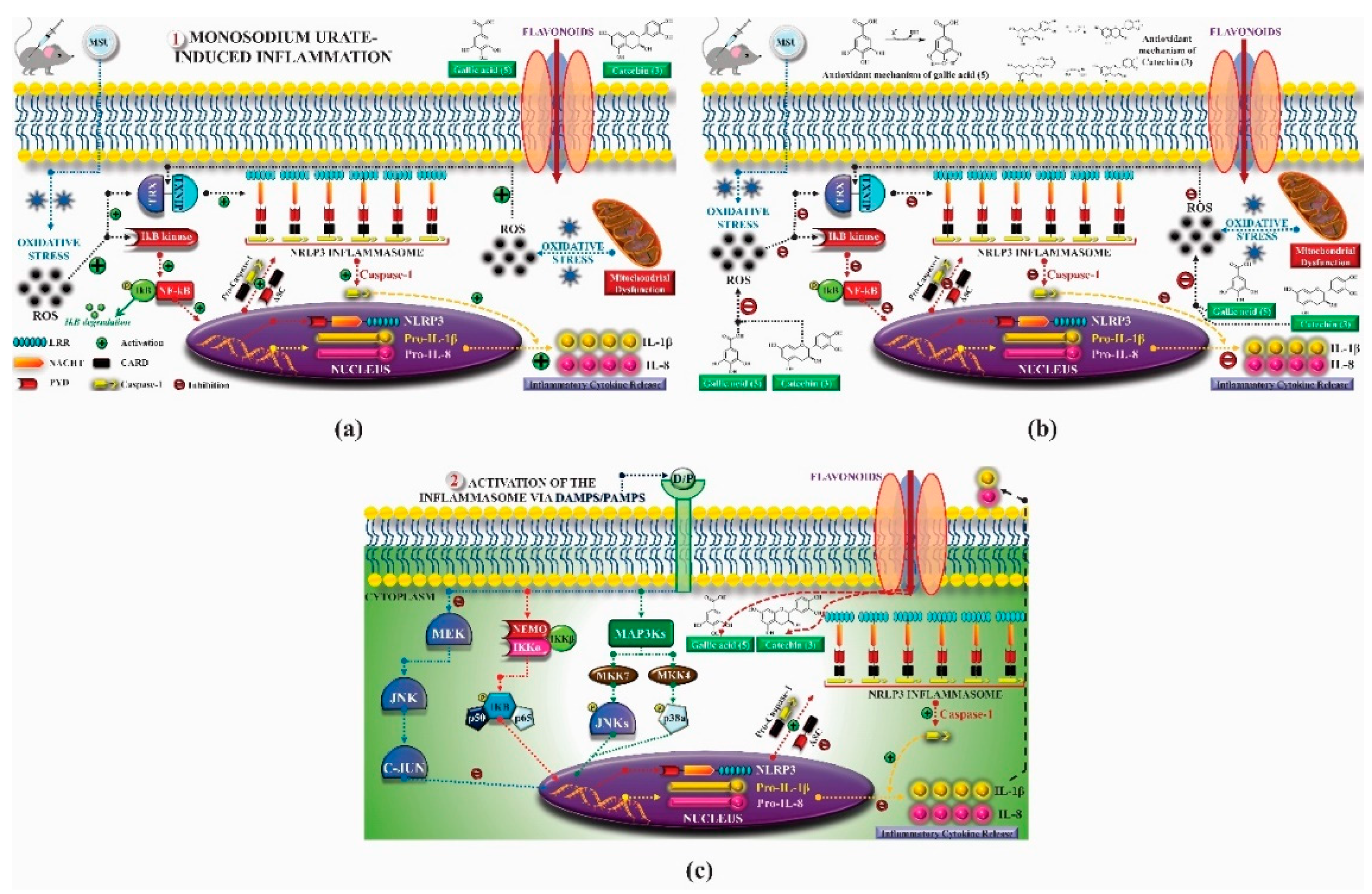
3.1.4. Pharmacological Studies
3.1.5. Toxicity Studies
4. Moraceae Family
4.1. Genus Brosimum and Species Brosimum gaudichaudii
4.1.1. Geographical Distribution and Popular Use
4.1.2. Botanical Aspects
4.1.3. Phytochemical Aspects
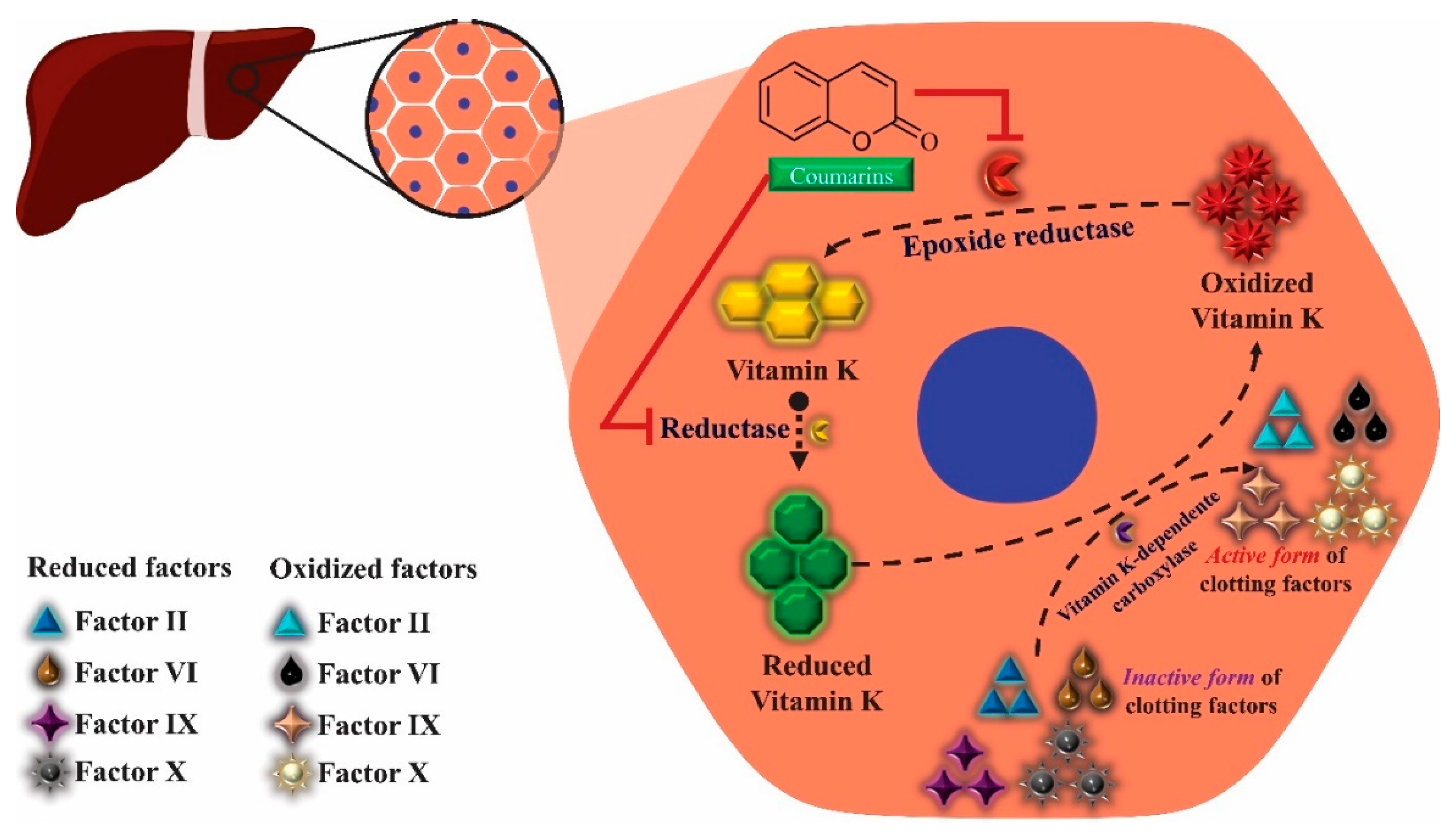
4.1.4. Pharmacological Studies
4.1.5. Toxicity Studies
5. Rubiaceae Family
5.1. Genus Genipa L. and Species Genipa americana L.
5.1.1. Geographical Distribution and Popular Use
5.1.2. Botanical Aspects
5.1.3. Phytochemical Aspects
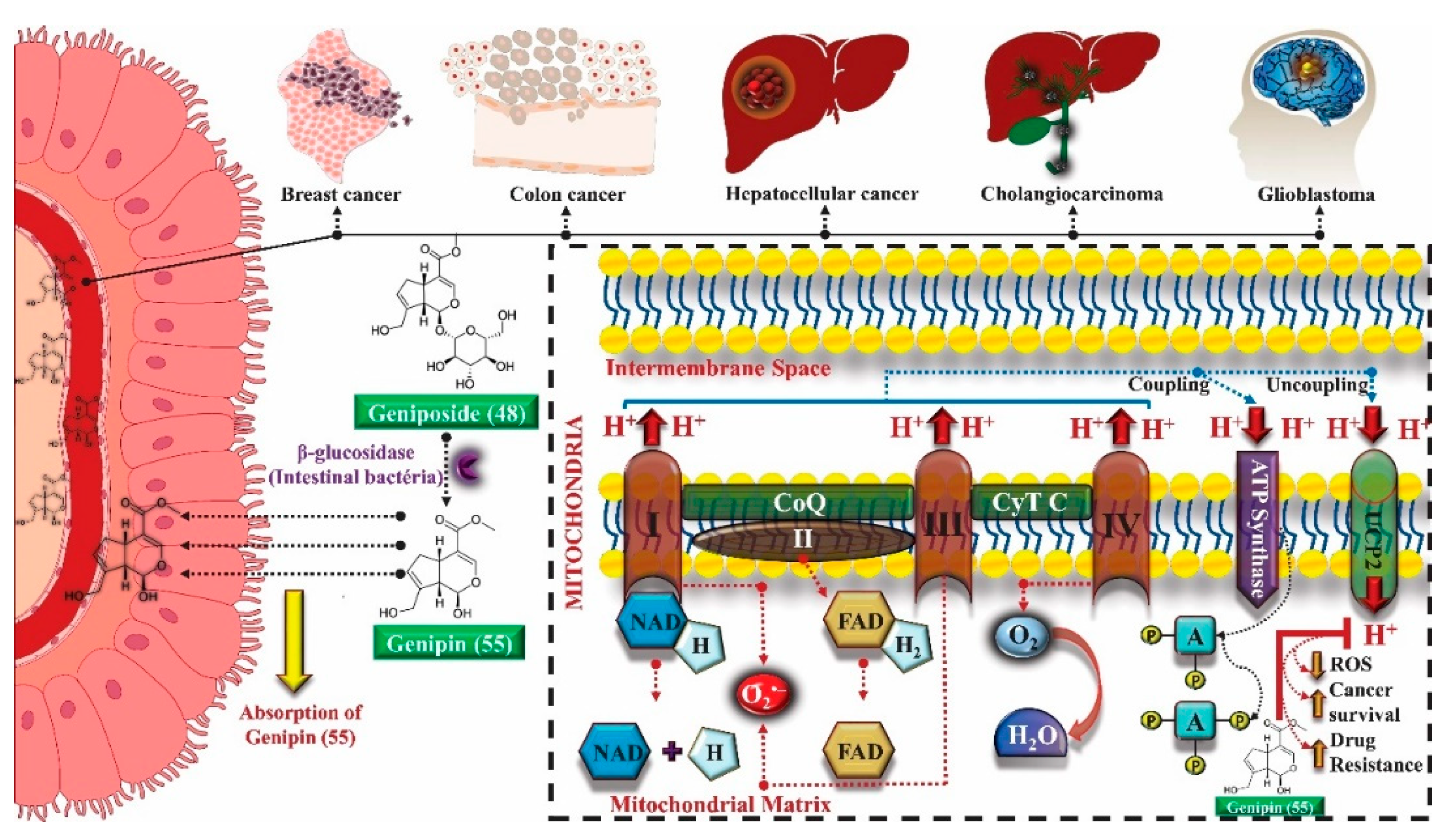
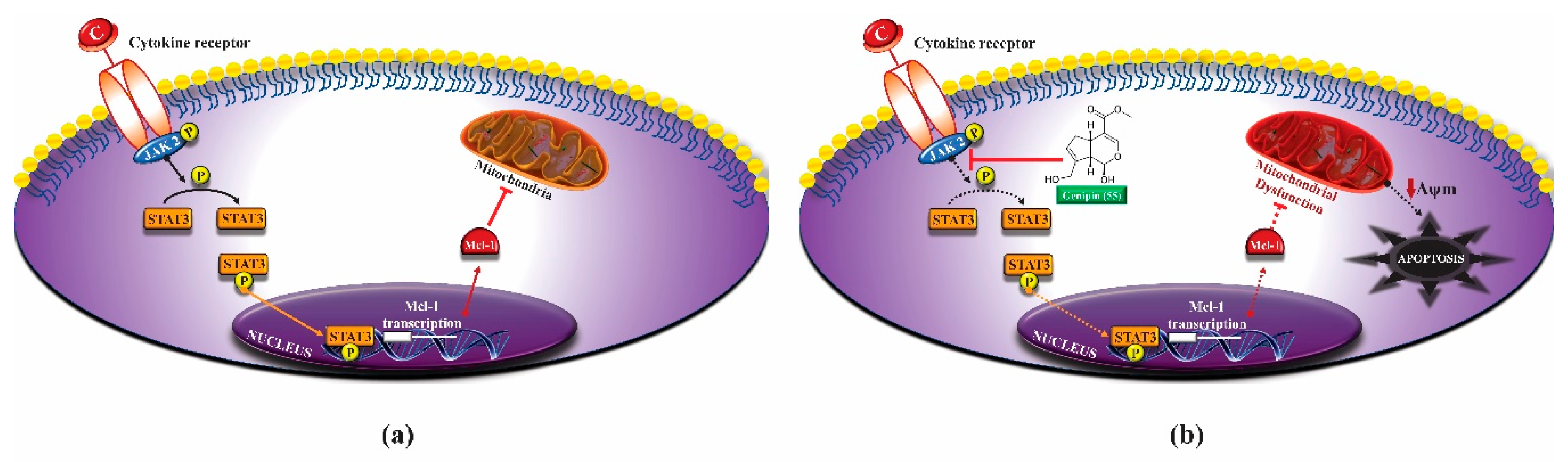
5.1.4. Toxicity Studies
6. Bromeliaceae Family
6.1. Genus Bromelia and Species Bromelia antiacantha
6.1.1. Geographical Distribution and Popular Use
6.1.2. Botanical Aspects
6.1.3. Phytochemical Aspects
6.1.4. Pharmacological Studies
6.1.5. Toxicity Studies
7. Clinical Trials
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Reis, A.F.; Schmiele, M. Características e potencialidades dos frutos do Cerrado na indústria de alimentos. Brazilian J. Food Technol. 2019, 22, e2017150. [Google Scholar] [CrossRef]
- Mohanraj, K.; Karthikeyan, B.S.; Vivek-Ananth, R.P.; Chand, R.P.B.; Aparna, S.R.; Mangalapandi, P.; Samal, A. IMPPAT: A curated database of Indian Medicinal Plants, Phytochemistry And Therapeutics. Sci. Rep. 2018, 8, 4329. [Google Scholar] [CrossRef] [Green Version]
- Souza, M.C.; Franco, A.C.; Haridasan, M.; Rossato, D.R.; de Araújo, J.F.; Morellato, L.P.C.; Habermann, G. The length of the dry season may be associated with leaf scleromorphism in cerrado plants. Ann. Braz. Acad. Sci. 2015, 87, 1691–1699. [Google Scholar] [CrossRef] [Green Version]
- Petropoulos, S.A.; Karkanis, A.; Martins, N.; Ferreira, I.C.F.R. Halophytic herbs of the Mediterranean basin: An alternative approach to health. Food Chem. Toxicol. 2018, 114, 155–169. [Google Scholar] [CrossRef] [Green Version]
- Bailão, E.; Devilla, I.; da Conceição, E.; Borges, L. Bioactive Compounds Found in Brazilian Cerrado Fruits. Int. J. Mol. Sci. 2015, 16, 23760–23783. [Google Scholar] [CrossRef]
- Roa, F.; de Telles, M.P.C. The Cerrado (Brazil) plant cytogenetics database. Comp. Cytogenet. 2017, 11, 285–297. [Google Scholar] [CrossRef]
- Klink, C.A.; Machado, R.B. Conservation of the Brazilian Cerrado. Conserv. Biol. 2005, 19, 707–713. [Google Scholar] [CrossRef]
- Villas-Boas, G.R.; dos Santos, A.C.; Souza, R.I.C.; Araújo, F.H.S.; Traesel, G.K.; Marcelino, J.M.; Silveira, A.P.S.; Farinelli, B.C.F.; Cardoso, C.A.L.; de Lacerda, R.B.; et al. Preclinical safety evaluation of the ethanolic extract from guavira fruits (Campomanesia pubescens (D.C.) O. BERG) in experimental models of acute and short-term toxicity in rats. Food Chem. Toxicol. 2018, 118, 1–12. [Google Scholar] [CrossRef]
- Zappi, D.C.; Filardi, F.L.R.; Leitman, P.; Souza, V.C.; Walter, B.M.T.; Pirani, J.R.; Morim, M.P.; Queiroz, L.P.; Cavalcanti, T.B.; Mansano, V.F.; et al. Growing knowledge: An overview of Seed Plant diversity in Brazil. Rodriguésia 2015, 66, 1085–1113. [Google Scholar] [CrossRef]
- Schiassi, M.C.E.V.; de Souza, V.R.; Lago, A.M.T.; Campos, L.G.; Queiroz, F. Fruits from the Brazilian Cerrado region: Physico-chemical characterization, bioactive compounds, antioxidant activities, and sensory evaluation. Food Chem. 2018, 245, 305–311. [Google Scholar] [CrossRef]
- Judd, W.S.; Campbell, C.S.; Kellogg, E.A.; Ste, P.F. Sistemática Vegetal: Um Enfoque Filogenético, 3rd ed.; Artmed: Porto Alegre, Brazil, 2009. [Google Scholar]
- Turland, N.J.; Wiersema, J.H.; Barrie, F.R.; Greuter, W.; Knapp, S.; Kusber, W.-H.; Li, D.-Z.; Marhold, K.; May, T.W.; McNeill, J.; et al. International Code of Nomenclature for Algae, fuNgi, and Plants; Turland, N., Wiersema, J., Barrie, F., Greuter, W., Hawksworth, D., Herendeen, P., Knapp, S., Kusber, W.-H., Li, D.-Z., Marhold, K., et al., Eds.; Regnum Vegetabile; Koeltz Botanical Books: Glashütten, Germany, 2018; Volume 159, ISBN 9783946583165. [Google Scholar]
- Miloski, J.; Somner, G.V.; Salimena, F.R.G.; Menini Neto, L. Sapindaceae na Serra Negra, Minas Gerais, Brasil. Rodriguésia 2017, 68, 671–690. [Google Scholar] [CrossRef]
- Guarim, G.N.; Santana, S.R.; Silva, J.V.B. Repertório botânico da “Pitombeira” (Talisia esculenta (A. ST.-HIL.) Radlk. - Sapindaceae). Acta Amaz. 2003, 33, 237–242. [Google Scholar] [CrossRef]
- Coutinho, D.J.G.; Ishiguro, M.A.; da Silva, T.G.; Mendes, L.C.A.; de Oliveira, A.F.M. Levantamento de espécies de Sapindaceae ocorrentes no estado de Pernambuco. JEPEX 2013. Available online: http://www.eventosufrpe.com.br/2013/cd/resumos/R1350-3.pdf (accessed on 21 July 2020).
- de Pereira, L.A.; Amorim, B.S.; Alves, M.; Somner, G.V.; de Barbosa, M.R.V. Flora da Usina São José, Igarassu, Pernambuco: Sapindaceae. Rodriguésia 2016, 67, 1047–1059. [Google Scholar] [CrossRef]
- Urdampilleta, J.D. Estudo Citotaxonômico em Espécies de Paullinieae (Sapindaceae). Ph.D. Thesis, Universidade Estadual de Campinas, Campinas, Brazil, 2009. [Google Scholar]
- De Sena, L.H.M.; Matos, V.P.; de Medeiros, J.É.; Santos, H.H.D.; Rocha, A.P.; Ferreira, R.L.C. Storage of pitombeira seeds [Talisia esculenta (A. St. Hil) Radlk - Sapindaceae] in different enviroments and packagings. Rev. Árvore 2016, 40, 435–445. [Google Scholar] [CrossRef]
- Dos Santos, W.L.; Freire, M.D.G.M.; Bogorni, P.C.; Vendramim, J.D.; Macedo, M.L.R. Effect of the aqueous extracts of the seeds of Talisia esculenta and Sapindus saponaria on fall armyworm. Brazilian Arch. Biol. Technol. 2008, 51, 373–383. [Google Scholar] [CrossRef] [Green Version]
- Dos Sena, L.H.M. Conservação de Sementes e Produção de Mudas de Pitombeira (Talisia esculenta (A. St. Hil.) Radlk.). Master’s Thesis, Universidade Federal Rural de Pernambuco, Recife, Pernambuco, 2014. [Google Scholar]
- Brasil. Alimentos Regionais Brasileiros, 2nd ed.; Ministério da Saúde: Brasília, Brazil, 2015; ISBN 8533404921.
- Lorenzi, H. Árvores Brasileiras: Manual de Identificação e Cultivo de plAntas Arbóreas nativas do Brasil. Plantarum 2009, 2, 384. [Google Scholar]
- Bieski, I.G.C.; Rios Santos, F.; de Oliveira, R.M.; Espinosa, M.M.; Macedo, M.; Albuquerque, U.P.; de Oliveira Martins, D.T. Ethnopharmacology of Medicinal Plants of the Pantanal Region (Mato Grosso, Brazil). Evidence-Based Complement. Altern. Med. 2012. [Google Scholar] [CrossRef] [Green Version]
- Vásquez, S.P.F.; de Mendonça, M.S.; do Noda, S.N. Etnobotânica de plantas medicinais em comunidades ribeirinhas do Município de Manacapuru, Amazonas, Brasil. Acta Amaz. 2014, 44, 457–472. [Google Scholar] [CrossRef]
- Badke, M.R.; de Budó, M.L.D.; da Silva, F.M.; Ressel, L.B. Plantas medicinais na prática do cotidiano popular. Esc. Anna Nery 2011, 15, 132–139. [Google Scholar] [CrossRef] [Green Version]
- De Nascimento, J.M.; da Conceição, G.M. Plantas medicinais e indicações terapêuticas da comunidade quilombola Olho d’água do Raposo, Caxias, Maranhão, Brasil. Rev. Biol. e Farmácia 2011, 6. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, V.B.; Zuchetto, M.; Oliveira, C.F.; Paula, C.S.; Duarte, A.F.S.; Miguel, M.D.; Miguel, O.G. Efeito de diferentes técnicas extrativas no rendimento, atividade antioxidante, doseamentos totais e no perfil por clae-dad de Dicksonia sellowiana (Presl.). Hook, dicksoniaceae. Rev. Bras. Plantas Med. 2016, 18, 230–239. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, S.F. Influência de malhas fotoconversoras nos aspectos anatômicos e fisiológicos de mudas de Talisia esculenta (A. St.-Hil.) Radlk. Master’s Thesis, Universidade Federal de Lavras, Lavras, Brazil, 2014. [Google Scholar]
- Silva, M.L.M. Biologia reprodutiva e maturação de sementes de Talisia esculenta (Cambess.) Radlk. Ph.D. Thesis, Universidade Federal da Paraíba, Areia, Brazil, 2019. [Google Scholar]
- Alves, E.U.; Silva, K.B.; Gonçalves, E.P.; Cardoso, E.D.A.; Alves, A.U. Germination and vigour of Talisia esculenta (St. Hil) Radlk seeds as a function of different fermentation periods. Semin. Ciências Agrárias 2009, 30, 761. [Google Scholar] [CrossRef] [Green Version]
- Teixeira, N.; Melo, J.C.S.; Batista, L.F.; Paula-Souza, J.; Fronza, P.; Brandão, M.G.L. Edible fruits from Brazilian biodiversity: A review on their sensorial characteristics versus bioactivity as tool to select research. Food Res. Int. 2019, 119, 325–348. [Google Scholar] [CrossRef]
- Neri-Numa, I.A.; de Carvalho-Silva, L.B.; Macedo Ferreira, J.E.; Tomazela Machado, A.R.; Malta, L.G.; Tasca Gois Ruiz, A.L.; de Carvalho, J.E.; Pastore, G.M. Preliminary evaluation of antioxidant, antiproliferative and antimutagenic activities of pitomba (Talisia esculenta). LWT-Food Sci. Technol. 2014, 59, 1233–1238. [Google Scholar] [CrossRef] [Green Version]
- Souza, M.P. Caracterização química e avaliação do potencial antioxidante dos frutos mari-mari (Cassia leiandra), pajurá (Couepia bracteosa) e pitomba (Talisia esculenta). Master’sThesis, Universidade Federal do Amazonas, Manaus, Brazil, 2016. [Google Scholar]
- De Souza, M.P.; Bataglion, G.A.; da Silva, F.M.A.; de Almeida, R.A.; Paz, W.H.P.; Nobre, T.A.; Marinho, J.V.N.; Salvador, M.J.; Fidelis, C.H.V.; Acho, L.D.R.; et al. Phenolic and aroma compositions of pitomba fruit (Talisia esculenta Radlk.) assessed by LC–MS/MS and HS-SPME/GC–MS. Food Res. Int. 2016, 83, 87–94. [Google Scholar] [CrossRef]
- Tirloni, C.A.S.; Silva, A.O.; Palozi, R.A.C.; Vasconcelos, P.C.D.P.; Souza, R.I.C.; Dos Santos, A.C.; De Almeida, V.P.; Budel, J.M.; De Souza, L.M.; Gasparotto, A. Biological Characterization of an Edible Species from Brazilian Biodiversity: From Pharmacognostic Data to Ethnopharmacological Investigation. J. Med. Food 2018, 21, 1276–1287. [Google Scholar] [CrossRef]
- Júnior, J.H.S. Avaliação da atividade antimicrobiana do extrato hidroetanólico de Talisia esculenta Radlk. Master’s Thesis, Universidade Federal do Maranhão, São Luís, Brazil, 2019. [Google Scholar]
- Sturm, S.; Seger, C. Liquid chromatography–nuclear magnetic resonance coupling as alternative to liquid chromatography–mass spectrometry hyphenations: Curious option or powerful and complementary routine tool? J. Chromatogr. A 2012, 1259, 50–61. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E. Fisiologia Vegetal, 5th ed.; Artmed: Porto Alegre, Brazil, 2013. [Google Scholar]
- Silva, M.R.; Lacerda, D.B.C.L.; Santos, G.G.; Martins, D.M.D.O. Caracterização química de frutos nativos do cerrado. Ciência Rural 2008, 38, 1790–1793. [Google Scholar] [CrossRef] [Green Version]
- Marin, A.M.F.; Siqueira, E.M.A.; Arruda, S.F. Minerals, phytic acid and tannin contents of 18 fruits from the Brazilian savanna. Int. J. Food Sci. Nutr. 2009, 60, 177–187. [Google Scholar] [CrossRef]
- Yi, Y.-S. Regulatory Roles of Flavonoids on Inflammasome Activation during Inflammatory Responses. Mol. Nutr. Food Res. 2018, 62, e1800147. [Google Scholar] [CrossRef] [PubMed]
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. Int.J. Mol. Sci. 2019, 20, 3328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, H.; Kawai, T.; Akira, S. Pathogen Recognition by the Innate Immune. Int. Rev. Immunol. 2011, 30, 16–34. [Google Scholar] [CrossRef] [PubMed]
- Franchi, L.; Warner, N.; Viani, K.; Nuñez, G. Function of Nod-like Receptors in Microbial Recognition and Host Defense. Immunol. Rev. 2009, 227, 106–128. [Google Scholar] [CrossRef] [Green Version]
- Davis, B.K.; Wen, H.; Ting, J.P. The Inflammasome NLRs in Immunity, Inflammation, and Associated Diseases. Annu. Rev. Immunol. 2011, 29, 707–735. [Google Scholar] [CrossRef] [Green Version]
- Martinon, F.; Mayor, A.; Tschopp, J. The Inflammasomes: Guardians of the Body. Annu. Rev. Immunol. 2009, 27, 229–265. [Google Scholar] [CrossRef] [Green Version]
- Dinarello, C.A. Immunological and Inflammatory Functions of the Interleukin-1 Family. Annu. Rev. Immunol. 2009, 27, 519–550. [Google Scholar] [CrossRef]
- Turner, M.D.; Nedjai, B.; Hurst, T.; Pennington, D.J. Cytokines and chemokines: At the crossroads of cell signalling and inflammatory disease. Biochim. Biophys. Acta J. 2014, 1843, 2563–2582. [Google Scholar] [CrossRef] [Green Version]
- Shuang, L.; Wen-Qing, Y.; TAO Ye-Zheng, M.L.; Xing, L. Serotonergic neurons in the median raphe nucleus mediate anxiety- and depression- like behavior. Acta Physiol. Sin. 2018, 70, 228–236. [Google Scholar] [CrossRef]
- Jhang, J.-J.; Lu, C.-C.; Ho, C.-Y.; Cheng, Y.-T.; Yen, G.-C. Protective Effects of Catechin against Monosodium Urate-Induced Inflammation through the Modulation of NLRP3 Inflammasome Activation. J. Agric. Food Chem. 2015, 63, 7343–7352. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, L.; Sun, D.; He, Y.; Jiang, Y.; Cheng, K.W.; Chen, F. DHA protects against monosodium urate-induced inflammation through modulation of oxidative stress. Food Funct. 2019, 10, 4010–4021. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Tardivel, A.; Thorens, B.; Choi, I.; Tschopp, J. Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat. Immunol. 2009, 11, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Yazdi, A.S.; Menu, P. A role for mitochondria in NLRP3 inflammasome activation. Nature 2011, 469, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Owona, B.A.; Abia, W.A.; Moundipa, P.F. Natural compounds flavonoids as modulators of inflammasomes in chronic diseases. Int. Immunopharmacol. 2020, 84, 106498. [Google Scholar] [CrossRef] [PubMed]
- Leopoldini, M.; Marino, T.; Russo, N.; Toscano, M. Antioxidant Properties of Phenolic Compounds: H-Atom versus Electron Transfer Mechanism. J. Phys. Chem. 2004, 108, 4916–4922. [Google Scholar] [CrossRef]
- Badhani, B.; Sharma, N.; Kakkar, R. Gallic acid: A versatile antioxidant with promising therapeutic and industrial applications. RSC Adv. 2015, 5, 27540–27557. [Google Scholar] [CrossRef]
- Katz, D.L.; Doughty, K.; Ali, A. Cocoa and Chocolate in Human Health and Disease. Antioxid. ad Redos Sinaling 2011, 15, 2779–2811. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Pan, Y.; Zhang, Q.-Y.; Wang, F.-M.; Kong, L.-D. Quercetin and Allopurinol Ameliorate Kidney Injury in STZ-Treated Rats with Regulation of Renal NLRP3 Inflammasome Activation and Lipid Accumulation. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [Green Version]
- Xu, D.; Hu, M.; Wang, Y.; Cui, Y. Antioxidant Activities of Quercetin and Its Complexes for Medicinal Application. Molecules 2019, 24, 1123. [Google Scholar] [CrossRef] [Green Version]
- Kawamura, K.; Qi, F.; Kobayashi, J. Potential relationship between the biological effects of low-dose irradiation and mitochondrial ROS production. J. Radiat. Res. 2018, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Hu, X.; Liu, Y.; Dong, S.; Wen, Z.; He, W.; Zhang, S.; Huang, Q. ROS signaling under metabolic stress: Cross-talk between AMPK and AKT pathway. Mol. Cancer 2017, 16, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jalmi, S.K.; Sinha, A.K. ROS mediated MAPK signaling in abiotic and biotic stress- striking similarities and differences. Front. Plant Sci. 2015, 6, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stoiber, W.; Obermayer, A.; Steinbacher, P.; Krautgartner, W. The Role of Reactive Oxygen Species (ROS) in the Formation of Extracellular Traps (ETs) in Humans. Biomolecules 2015, 5, 702–723. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vurusaner, B.; Poli, G.; Basaga, H. Tumor suppressor genes and ROS: Complex networks of interactions. Free Radic. Biol. Med. 2012, 52, 7–18. [Google Scholar] [CrossRef]
- Pei, K.; Ou, J.; Huang, J.; Ou, S. p-Coumaric acid and its conjugates: Dietary sources, pharmacokinetic properties and biological activities. J. Sci. Food Agric. 2016, 96, 2952–2962. [Google Scholar] [CrossRef]
- Pinheiro, A.Q.; Melo, D.F.; Macedo, L.M.; Freire, M.G.M.; Rocha, M.F.G.; Sidrim, J.J.C.; Brilhante, R.S.N.; Teixeira, E.H.; Campello, C.C.; Pinheiro, D.C.S.N.; et al. Antifungal and marker effects of Talisia esculenta lectin on Microsporum canis in vitro. J. Appl. Microbiol. 2009, 107, 2063–2069. [Google Scholar] [CrossRef]
- Oliveira, M.R.T.R.; Napimoga, M.H.; Cogo, K.; Gonçalves, R.B.; Macedo, M.L.R.; Freire, M.G.M.; Groppo, F.C. Inhibition of bacterial adherence to saliva-coated through plant lectins. J. Oral Sci. 2007, 49, 141–145. [Google Scholar] [CrossRef] [Green Version]
- Riet-Correa, F.; Medeiros, R.M.T.; Pfister, J.A.; Mendonça, F.S. Toxic plants affecting the nervous system of ruminants and horses in Brazil. Pesqui. Veterinária Bras. 2017, 37, 1357–1368. [Google Scholar] [CrossRef] [Green Version]
- Riet-Correa, F.; Bezerra, C.W.; Medeiros, M.A.; da Silva, T.R.; Neto, E.G.M.; Medeiros, R.M.T. Poisoning by Talisia esculenta (A. St.-Hil.) Radlk in sheep and cattle. J. Vet. Diagnostic Investig. 2014, 26, 412–417. [Google Scholar] [CrossRef] [Green Version]
- Melo, J.K.A.; Soares, G.S.L.; Ramos, T.R.R.; Almeida, V.M.; Nascimento, A.L.O.; Silva Filho, G.B.; Chaves, H.A.S.; Mendonça, F.S. Spontaneous poisoning by Talisia esculenta in cattle. Pesqui. Veterinária Bras. 2019, 39, 949–953. [Google Scholar] [CrossRef]
- Freire, M.G.; Desouza, I.; Silva, A.C.; Macedo, M.L.; Lima, M.; Tamashiro, W.M.S.; Antunes, E.; Marangoni, S. Inflammatory responses induced in mice by lectin from Talisia esculenta seeds. Toxicon 2003, 42, 275–280. [Google Scholar] [CrossRef]
- Da Mota, T.M.; Silva, A.E.V.N.; de Melo Filho, E.V.; de Siqueira, J.O.; Ferreira, D.R.C.; Groschke, H.M.; da Braz, R.S.; Teixeira, M.W. Intoxicação por sementes de pitombeira (Talisia esculenta) em um cão—Relato de caso. Clín. Vet. 2016, 21, 78–84. [Google Scholar]
- De Castro, R.M. Flora da Bahia–Moraceae. Master’s Thesis, Universidade Estadual de Feira de Santana, Florianópolis, Brazil, 2006. [Google Scholar]
- Leite, V.G.; Mansano, V.F.; Pádua Teixeira, S. Floral development of Moraceae species with emphasis on the perianth and androecium. Flora 2018, 240, 116–132. [Google Scholar] [CrossRef]
- De Filho, A.B.L.; da Silva, J.M.; dos de Santana, M.A.; de Melo, E.M. Espécies da família moraceae com potencial fitoterápico em dois fragmento de floresta urbana no município de camaragibe. JEPEX-UFRPE 2013. Available online: http://www.eventosufrpe.com.br/2013/cd/resumos/R0533-1.pdf (accessed on 21 July 2020).
- Jacomassi, E.; Moscheta, I.S.; Machado, S.R. Morfoanatomia e histoquímica de órgãos reprodutivos de Brosimum gaudichaudii (Moraceae). Rev. Bras. Botânica 2010, 33, 115–129. [Google Scholar] [CrossRef] [Green Version]
- Mitsuko Aoyama, E.; Mazzoni-Viveiros, C. ADAPTAÇÕES ESTRUTURAIS DAS PLANTAS AO AMBIENTE 2006. Available online: http://www.biodiversidade.pgibt.ibot.sp.gov.br/Web/pdf/Adaptacoes_estruturais_das_Plantas_ao_Ambiente_Elisa_Aoyama.pdf (accessed on 21 July 2020).
- Ribeiro, J.E.L.S.; Pederneiras, L.C. Brosimum in Flora do Brasil 2020 em construção. Jardim Botânico do Rio de Janeiro. Available online: http://floradobrasil.jbrj.gov.br/ (accessed on 26 June 2020).
- Palhares, D.; de Paula, J.E.; dos Santos Silveira, C.E. Morphology of stem and subterranean system of Brosimum gaudichaudii (Moraceae). Acta Botanica Hungarica. 2006, 48, 89–101. [Google Scholar] [CrossRef]
- Da Cunha, L.C.; de Paula, J.R.; de Sá, V.A.; da e Amorim, M.E.P.; Barros, I.C.M.; Brito, L.A.B.; da Silveira, N. Acute toxicity of Brosimum gaudichaudii Trécul. root extract in mice: Determination of both approximate and median lethal doses. Rev. Bras. Farmacogn. 2008, 18, 532–538. [Google Scholar] [CrossRef]
- De Faria, R.A.P.G.; de Coelho, M.F.B.; de Albuquerque, M.C.F.; de Azevedo, R.A.B. Fenologia de Brosimum gaudichaudii TRÉCUL. (Moraceae) no cerrado de Mato Grosso. Ciência Florest. 2015, 25, 67–75. [Google Scholar] [CrossRef] [Green Version]
- Jacomassi, E.; Moscheta, I.S.; Machado, S.R. Morfoanatomia e histoquímica de Brosimum gaudichaudii Trécul (Moraceae). Acta Bot. Brasilica 2007, 21, 575–597. [Google Scholar] [CrossRef] [Green Version]
- Jacomassi, E. Morfoanatomia e histoquímica de orgãos vegetativos reprodutivos de Brosimum gaudichaudii Trécul (Moraceae). Inst. Biociências – Dep. Botânica 2006. [Google Scholar]
- Faria, R.A.P.G.; Silva, A.N.; Albuquerque, M.C.F.; Coelho, M.F.B. Características biométricas e emergência de plântulas de Brosimum gaudichaudii Tréc. oriundas de diferentes procedências do cerrado mato-grossense. Rev. Bras. Plantas Med. 2009, 11, 414–421. [Google Scholar] [CrossRef]
- Ribeiro, R.V.; Bieski, I.G.C.; Balogun, S.O.; de Martins, D.T.O. Ethnobotanical study of medicinal plants used by Ribeirinhos in the North Araguaia microregion, Mato Grosso, Brazil. J. Ethnopharmacol. 2017, 205, 69–102. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, V.E.G.; Carvalho, D.A. Levantamento etnobotânico de plantas medicinais no domínio dos cerrados na região do Alto Rio Grande - Minas Gerais. Rev. Bras. Plantas Med. 2007, 9, 17–35. [Google Scholar]
- de Braga, C.M. Histórico da Utilização de Plantas Medicinais; Monography; Universidade de Brasília/Universidade Federal de Gioás: Brasília, Brazil, 2011. [Google Scholar]
- dos Passos, M.M.B.; da Albino, R.C.; Feitoza-Silva, M.; de Oliveira, D.R. A disseminação cultural das garrafadas no Brasil: Um paralelo entre medicina popular e legislação sanitária. Saúde em Debate 2018, 42, 248–262. [Google Scholar] [CrossRef]
- Da Borges, J.C. Atividade antimicrobiana de extrato de Brosimum gaudichaudii trécul. contra bactérias isoladas de lesões de pés diabéticos. Master’s Thesis, Universidade Federal de Tocantins, Palmas, Brazil, 2016. [Google Scholar]
- Martins, D. Morfologia e Anatomia do caule e do sistema subterrâneo de Brosimum gaudichaudii Tréc. (Moraceae). Master’s Thesis, Universidade de Brasília, Brasília, Brazil, 2004. [Google Scholar]
- Lourenço, M.V. Estudo comparativo dos constituintes químicos de Brosimum gaudichaudii Trécul e do medicamento “V.”. Ph.D. Thesis, Universidade Estadual Paulista, Araraquara, Brazil, 2001. [Google Scholar]
- Martins, F.S.; Pascoa, H.; De Paula, J.R.; Da Conceição, E.C. Technical aspects on production of fluid extract from Brosimum gaudichaudii Trécul roots. Pharmacogn. Mag. 2015, 11, 226–231. [Google Scholar] [CrossRef] [Green Version]
- Vieira, I.J.C.; Mathias, L.; Monteiro, V.D.F.F.; Braz-Filho, R.; Rodrigues-Filho, E. A New Coumarln from Brosimum gaudichaudii Trecul. Nat. Prod. Lett. 1999, 13, 47–52. [Google Scholar] [CrossRef]
- De Monteiro, V.F.F.; Mathias, L.; Vieira, I.J.C.; Schripsema, J.; Braz-Filho, R. Prenylated Coumarins, Chalcone and New Cinnamic Acid and Dihydrocinnamic Acid Derivatives from Brosimum gaudichaudii. J. Braz. Chem. Soc. 2002, 13, 281–287. [Google Scholar] [CrossRef]
- Land, L.R.B.; Borges, F.M.; Borges, D.O.; Pascoal, G.B. Composição centesimal, compostos bioativos e parâmetros físico-químicos da mama-cadela (Brosimum gaudichaudii Tréc) proveniente do Cerrado Mineiro. DEMETRA Aliment. Nutr. Saúde 2017, 12, 509–518. [Google Scholar] [CrossRef] [Green Version]
- Silva, S.M.F.Q.; Pinheiro, S.M.B.; Queiroz, M.V.F.; Pranchevicius, M.C.; Castro, J.G.D.; Perim, M.C.; Carreiro, S.C. Atividade in vitro de extratos brutos de duas espécies vegetais do cerrado sobre leveduras do gênero Candida. Cien. Saude Colet. 2012, 17, 1649–1656. [Google Scholar] [CrossRef] [Green Version]
- Monteiro, J.M.; de Souza, J.S.N.; Neto, E.M.F.L.; Scopel, K.; Trindade, E.F. Does total tannin content explain the use value of spontaneous medicinal plants from the Brazilian semi-arid region? Rev. Bras. Farmacogn. 2014, 24, 116–123. [Google Scholar] [CrossRef] [Green Version]
- De Moreira, C.C.S. Atividade Antimicrobiana da Casca e Folha do Brosimum gaudichaudii Trécul; Monography; Centro Universitário Luterano de Palmas: Palmas, Brazil, 2019. [Google Scholar]
- De Quintão, W.S.C. Desenvolvimento, caracterização e avaliação in vitro de nanoemulsões O/A a partir de extratos de Brosimum gaudichaudii (mama cadela) como alternativa parao tratamento tópico de vitiligo. Master’s Thesis, Universidade de Brasília, Brasília, Brazil, 2018. [Google Scholar]
- De Filho, A.C.P.M.; de Castro, C.F.S. Identificação das classes de metabólitos secundários nos extratos etanólicos foliares de Brosimum gaudichaudii, Qualea grandiflora, Rollinia laurifolia e Solanum cernuum. Rev. Multitexto 2019, 7, 22–32. [Google Scholar]
- Leão, A.R.; da Cunha, L.C.; Parente, L.M.L.; Castro, L.C.M.; Chaul, A.; Carvalho, H.E.; Rodrigues, V.B.; Bastos, M.A. Avaliação clínica toxicólogica preliminar do Viticromin em pacientes com vitiligo. Rev. Eletrônica Farmácia 2007, 2, 15–23. [Google Scholar] [CrossRef]
- Del Río, J.A.; Díaz, L.; García-Bernal, D.; Blanquer, M.; Ortuño, A.; Correal, E.; Moraleda, J.M. Furanocoumarins: Biomolecules of therapeutic interest. Stud. Nat. Prod. Chem. 2014, 43, 145–195. [Google Scholar]
- Lang, K.S.; Muhm, A.; Moris, A.; Stevanovic, S.; Rammensee, H.-G.; Caroli, C.C.; Wernet, D.; Schittek, B.; Knauss-Scherwitz, E.; Garbe, C. HLA-A2 Restricted, Melanocyte-Specific CD8+ T Lymphocytes Detected in Vitiligo Patients are Related to Disease Activity and are Predominantly Directed Against MelanA/MART1. J. Investig. Dermatol. 2001, 116, 891–897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mankarious, M.; Matthews, N.C.; Snowden, J.A.; Alfred, A. Extracorporeal Photopheresis (ECP) and the Potential of Novel Biomarkers in Optimizing Management of Acute and Chronic Graft vs. Host Disease (GvHD). Front. Immunol. 2020, 11, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Da Borges, J.C.; Perim, M.C.; de Castro, R.O.; de Araújo, T.A.S.; da Peixoto Sobrinho, T.J.S.; da Silva, A.C.O.; Mariano, S.M.B.; Carreiro, S.C.; da Pranchevicius, M.C.S. Evaluation of antibacterial activity of the bark and leaf extracts of Brosimum gaudichaudii Trécul against multidrug resistant strains. Nat. Prod. Res. 2017, 31, 2931–2935. [Google Scholar] [CrossRef]
- Ferreira, P.C. Toxicidade e Atividade Tripanocida do Extrato Bruto de Brosimum gaudichaudii Trécul (Moraceae) (Mama-cadela) no Pré-tratamento e Tratamento de Camundongos Infectados por Trypanosoma cruzi. Master’s Thesis, Universidade Federal de Uberlândia, Uberlândia, Brazil, 2008. [Google Scholar]
- Jeon, S.J.; Park, H.J.; Gao, Q.; Lee, H.E.; Park, S.J.; Hong, E.; Jang, D.S.; Shin, C.Y.; Cheong, J.H.; Ryu, J.H. Positive effects of β-amyrin on pentobarbital-induced sleep in mice via GABAergic neurotransmitter system. Behav. Brain Res. 2015, 291, 232–236. [Google Scholar] [CrossRef]
- Wei, C.-C.; Chang, C.-H.; Liao, V.H.-C. Anti-Parkinsonian effects of β-amyrin are regulated via LGG-1 involved autophagy pathway in Caenorhabditis elegans. Phytomedicine 2017, 36, 118–125. [Google Scholar] [CrossRef]
- Sunil, C.; Irudayaraj, S.S.; Duraipandiyan, V.; Al-Dhabi, N.A.; Agastian, P.; Ignacimuthu, S. Antioxidant and free radical scavenging effects of β-amyrin isolated from S. cochinchinensis Moore. leaves. Ind. Crops Prod. 2014, 61, 510–516. [Google Scholar] [CrossRef]
- Askari, V.R.; Fereydouni, N.; Baradaran Rahimi, V.; Askari, N.; Sahebkar, A.H.; Rahmanian-Devin, P.; Samzadeh-Kermani, A. β-Amyrin, the cannabinoid receptors agonist, abrogates mice brain microglial cells inflammation induced by lipopolysaccharide/interferon-γ and regulates Mφ1/Mφ2 balances. Biomed. Pharmacother. 2018, 101, 438–446. [Google Scholar] [CrossRef]
- Dong, L.; Xu, W.-W.; Li, H.; Bi, K.-H. In vitro and in vivo anticancer effects of marmesin in U937 human leukemia cells are mediated via mitochondrial-mediated apoptosis, cell cycle arrest, and inhibition of cancer cell migration. Oncol. Rep. 2018, 39, 597–602. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-K.; Jeon, S.W. Neuroinflammation and the Immune-Kynurenine Pathway in Anxiety Disorders. Curr. Neuropharmacol. 2018, 16, 574–582. [Google Scholar] [CrossRef] [PubMed]
- Marques, G.; Gutiérrez, A.; Del Río, J.C. Chemical characterization of lignin and lipophilic fractions from leaf fibers of curaua (Ananas erectifolius). J. Agric. Food Chem. 2007, 55, 1327–1336. [Google Scholar] [CrossRef] [Green Version]
- Varanda, E.A.; Pozetti, G.L.; Lourenço, M.V.; Vilegas, W.; Raddi, M.S.G. Genotoxicity of Brosimum gaudichaudii measured by the Salmonella/microsome assay and chromosomal aberrations in CHO cells. J. Ethnopharmacol. 2002, 81, 257–264. [Google Scholar] [CrossRef]
- e Amorim, M.E.P.; da Cunha, L.C.; da Silveira, N.A. Estudo Da Toxicidade Aguda E Subaguda De Brosimum gaudichaudii Trécul (Mamacadela) Em Ratos (Rattus Norvergicus) P.O. Rev. Eletrônica Farmácia 2004, 1, 52. [Google Scholar] [CrossRef]
- Orme, M.L.; Breckenridge, A.M. Coumarin anticoagulants. In Meyler’s Side Effects of Drugs; Elsevier: Dordrecht, The Netherlands, 2016; Volume 58, pp. 702–737. [Google Scholar]
- Delprete, P.G.; Jardim, J.G. Systematics, taxonomy and floristics of Brazilian Rubiaceae: An overview about the current status and future challenges. Rodriguésia 2012, 63, 101–128. [Google Scholar] [CrossRef] [Green Version]
- Baldin, T. Anatomia do Lenho do Genero Calycophyllum A. DC. (Rubiaceae). Master’s Thesis, Universidade Federal de Santa Maria, Santa Maria, Brazil, 2015. [Google Scholar]
- Bellé, A.S. Extração de Genipina a Partir do Jenipapo (Genipa americana Linnaeus) para Imobilização de Enzimas. Master’s Thesis, Universidade Federal do Rio Grande do Sul, Proto Alegre, Brazil, 2017. [Google Scholar]
- Erbano, M.; Duarte, M.R. Morfoanatomia de folha e caule de Genipa americana L., Rubiaceae. Rev. Bras. Farmacogn. 2010, 20, 825–832. [Google Scholar] [CrossRef]
- Mielke, M.S.; de Almeida, A.-A.F.; Gomes, F.P.; Aguilar, M.A.G.; Mangabeira, P.A.O. Leaf gas exchange, chlorophyll fluorescence and growth responses of Genipa americana seedlings to soil flooding. Environ. Exp. Bot. 2003, 50, 221–231. [Google Scholar] [CrossRef]
- Luzia, D.M.M. Propriedades funcionais de óleos extraídos de sementes de frutos do cerrado brasileiro. Master’s Thesis, Universidade Estadual Paulista, São José do Rio Preto, Brazil, 2012. [Google Scholar]
- Ferreira, W.R.; Ranal, M.; Dorneles, M.C.; Santana, D.G. Crescimento de mudas de Genipa americana L. submetidas a condições de pré-semeadura. Braz. J. Bioc. 2007, 5, 1026–1028. [Google Scholar]
- Da Santos, W.C. Germinação e Vigor de Sementes de Genipa americana L. em Função do Estresse hídrico em Diferentes Temperaturas; Monography; Universidade Federal da Paraíba: Areia, Brazil, 2018. [Google Scholar]
- De Barbosa, D.A. Avaliação Fitoquímica e Farmacológica de Genipa americana L. (Rubiaceae). Master’s Thesis, Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil, 2008. [Google Scholar]
- Santos, L.; Salles, M.; Pinto, C.; Pinto, O.; Rodrigues, I. O saber etnobotânico sobre plantas medicinais na comunidade da Brenha, Redenção, CE. Agrar. Acad. 2018, 5, 127–145. [Google Scholar] [CrossRef]
- Souza, R.K.D.; Mendonça, A.C.A.M.; da Silva, M.A.P. Ethnobotanical, phytochemical and pharmacological aspects Rubiaceae species in Brazil. Rev. Cuba. Plantas Med. 2013, 18, 140–156. [Google Scholar]
- Bieski, I.G.C.; Leonti, M.; Arnason, J.T.; Ferrier, J.; Rapinski, M.; Violante, I.M.P.; Balogun, S.O.; Pereira, J.F.C.A.; Figueiredo, R.D.C.F.; Lopes, C.R.A.S.; et al. Ethnobotanical study of medicinal plants by population of Valley of Juruena Region, Legal Amazon, Mato Grosso, Brazil. J. Ethnopharmacol. 2015, 173, 383–423. [Google Scholar] [CrossRef] [PubMed]
- Cartaxo, S.L.; de Almeida Souza, M.M.; de Albuquerque, U.P. Medicinal plants with bioprospecting potential used in semi-arid northeastern Brazil. J. Ethnopharmacol. 2010, 131, 326–342. [Google Scholar] [CrossRef] [PubMed]
- Yazbek, P.B.; Tezoto, J.; Cassas, F.; Rodrigues, E. Plants used during maternity, menstrual cycle and other women’s health conditions among Brazilian cultures. J. Ethnopharmacol. 2016, 179, 310–331. [Google Scholar] [CrossRef]
- Odonne, G.; Valadeau, C.; Alban-Castillo, J.; Stien, D.; Sauvain, M.; Bourdy, G. Medical ethnobotany of the Chayahuita of the Paranapura basin (Peruvian Amazon). J. Ethnopharmacol. 2013, 146, 127–153. [Google Scholar] [CrossRef]
- De Carvalho, T.L.G.S. Etnofarmacologia e fisiologia de plantas medicinais do quilombo Tiningú, Santarém, Pará. Master’s Thesis, Universidade Federal do Oeste do Pará, Santarém, Brazil.
- Bueno, M.J.A.; Martínez, B.B.; Bueno, J.C. Manual de Plantas Medicinais e Fitoterápicos: Utilizados na Cicatrização de Feridas; UNIVÁS: Pouso Alegre, Brazil, 2016; ISBN 9788533415973. [Google Scholar]
- Rodrigues, V.G.S. Cultivo, Uso e Manipulação de Plantas Medicinais; Embrapa-Empres: Rondônia, Brazil, 2004; Volume 1, pp. 1–25. [Google Scholar]
- Santiago, E.F.; Paoli, A.A.S. Respostas morfológicas em Guibourtia hymenifolia (Moric.) J. Leonard (Fabaceae) e Genipa americana L. (Rubiaceae), submetidas ao estresse por deficiência nutricional e alagamento do substrato. Rev. Bras. Botânica 2007, 30, 131–140. [Google Scholar] [CrossRef] [Green Version]
- De Crestana, C.S.M. Ecologia e polinização de Genipa americana L. (Rubiaceae) na estação ecológica de Moji-guaçu, estado de São Paulo. Rev. Inst. Flor 1995, 7, 169–195. [Google Scholar]
- Carvalho, P.E.R. Jenipapeiro; Embrapa-Bol: Colombo, Brazil, 2003; Volume 14, pp. 2–14. ISSN 1517-5278. [Google Scholar]
- De Carvalho, J.E.U.; do Nascimento, W.M.O. Sensibilidade de sementes de jenipapo (Genipa americana L.) ao dessecamento e ao congelamento. Rev. Bras. Frutic. 2000, 22, 53–56. [Google Scholar]
- Silva, L.; Alves, J.; da Silva Siqueira, E.; de Souza Neto, M.; Abreu, L.; Tavares, J.; Porto, D.; de Santis Ferreira, L.; Demarque, D.; Lopes, N.; et al. Isolation and Identification of the Five Novel Flavonoids from Genipa americana Leaves. Molecules 2018, 23, 2521. [Google Scholar] [CrossRef] [Green Version]
- Ono, M.; Ueno, M.; Masuoka, C.; Ikeda, T.; Nohara, T. Iridoid Glucosides from the Fruit of Genipa americana. Chem. Pharm. Bull. (Tokyo) 2005, 53, 1342–1344. [Google Scholar] [CrossRef] [Green Version]
- Bellé, A.S.; Hackenhaar, C.R.; Spolidoro, L.S.; Rodrigues, E.; Klein, M.P.; Hertz, P.F. Efficient enzyme-assisted extraction of genipin from genipap (Genipa americana L.) and its application as a crosslinker for chitosan gels. Food Chem. 2018, 246, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Alves, J.S.F.; de Medeiros, L.A.; de Fernandes-Pedrosa, M.F.; Araújo, R.M.; Zucolotto, S.M. Iridoids from leaf extract of Genipa americana. Rev. Bras. Farmacogn. 2017, 27, 641–644. [Google Scholar] [CrossRef] [Green Version]
- Neri-Numa, I.A.; Pessôa, M.G.; Arruda, H.S.; Pereira, G.A.; Paulino, B.N.; Angolini, C.F.F.; Ruiz, A.L.T.G.; Pastore, G.M. Genipap (Genipa americana L.) fruit extract as a source of antioxidant and antiproliferative iridoids. Food Res. Int. 2020, 134, 109252. [Google Scholar] [CrossRef] [PubMed]
- Náthia-Neves, G.; Tarone, A.G.; Tosi, M.M.; Maróstica Júnior, M.R.; Meireles, M.A.A. Extraction of bioactive compounds from genipap (Genipa americana L.) by pressurized ethanol: Iridoids, phenolic content and antioxidant activity. Food Res. Int. 2017, 102, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Ramos-de-la-Peña, A.M.; Renard, C.M.G.C.; Wicker, L.; Montañez, J.C.; García-Cerda, L.A.; Contreras-Esquivel, J.C. Environmental friendly cold-mechanical/sonic enzymatic assisted extraction of genipin from genipap (Genipa americana). Ultrason. Sonochem. 2014, 21, 43–49. [Google Scholar] [CrossRef]
- Pacheco, P.; Da Paz, J.G.; Da Silva, C.O.; Pascoal, G.B. Composição centesimal, compostos bioativos e parâmetros físico-químicos do jenipapo (Genipa americana L.) in natura. DEMETRA Aliment. Nutr. Saúde 2014, 9, 1041–1054. [Google Scholar] [CrossRef]
- Costa, R.G.; Da Silva, D.A.; Alves, S.F. Obtenção e caracterização do extrato fluido de Genipa americana Linnaeus. Rev. Eletrônica Farmácia 2019, 16, 1–7. [Google Scholar] [CrossRef]
- Mendes, W.B.S.; Andrade, T.O.; Moraes, S.M. Estudo Fitoquímico e Avaliação da Atividade Antiacetilcolinestersásica da Folha e Casca do Jenipapaeiro (Genipa americana L.). 57° Congresso Brasileiro de Química, 2017. Available online: http://www.abq.org.br/cbq/2017/trabalhos/7/11151-24611.html (accessed on 21 July 2020).
- Costa, R.B.; Campana, P.T.; Chambergo, F.S.; Napoleão, T.H.; Paiva, P.M.G.; Pereira, H.J.V.; Oliva, M.L.V.; Gomes, F.S. Purification and characterization of a lectin with refolding ability from Genipa americana bark. Int. J. Biol. Macromol. 2018, 119, 517–523. [Google Scholar] [CrossRef]
- Kwon, S.-H.; Lee, H.-K.; Kim, J.-A.; Hong, S.-I.; Kim, H.-C.; Jo, T.-H.; Park, Y.-I.; Lee, C.-K.; Kim, Y.-B.; Lee, S.-Y.; et al. Neuroprotective effects of chlorogenic acid on scopolamine-induced amnesia via anti-acetylcholinesterase and anti-oxidative activities in mice. Eur. J. Pharmacol. 2010, 649, 210–217. [Google Scholar] [CrossRef]
- Wang, J.; Chen, L.; Liang, Z.; Li, Y.; Yuan, F.; Liu, J.; Tian, Y.; Hao, Z.; Zhou, F.; Liu, X.; et al. Genipin Inhibits LPS-Induced Inflammatory Response in BV2 Microglial Cells. Neurochem. Res. 2017, 42, 2769–2776. [Google Scholar] [CrossRef]
- Yu, S.X.; Du, C.T.; Chen, W.; Lei, Q.Q.; Li, N.; Qi, S.; Zhang, X.J.; Hu, G.Q.; Deng, X.M.; Han, W.Y.; et al. Genipin inhibits NLRP3 and NLRC4 inflammasome activation via autophagy suppression. Sci. Rep. 2016, 5, 17935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, X.; Lin, B.; Gao, Y.; Lei, X.; Wang, X.; Li, Y.; Li, T. International Immunopharmacology Genipin attenuates mitochondrial-dependent apoptosis, endoplasmic reticulum stress, and in fl ammation via the PI3K/AKT pathway in acute lung injury. Int. Immunopharmacol. 2019, 76, 105842. [Google Scholar] [CrossRef] [PubMed]
- Pugazhenthi, S.; Nesterova, A.; Sable, C.; Heidenreich, K.A.; Boxer, L.M.; Heasley, L.E.; Reusch, J.E. Akt/Protein Kinase B Up-regulates Bcl-2 Expression through cAMP-response Element-binding Protein. J. Biol. Chem. 2000, 275, 10761–10766. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Z.; Zhang, T.; Jia, D.; Sun, W.; Wang, C.; Gu, A.; Yang, X. Genipin inhibits the growth of human bladder cancer cells via inactivation of PI3K/Akt signaling. Oncol. Lett. 2017, 15, 2619–2624. [Google Scholar] [CrossRef]
- Wei, M.; Wu, Y.; Liu, H.; Xie, C. Genipin Induces Autophagy and Suppresses Cell Growth of Oral Squamous Cell Carcinoma via PI3K/AKT/MTOR Pathway. Drug Des. Devel. Ther. 2020, 14, 395–405. [Google Scholar] [CrossRef] [Green Version]
- Zhao, B.; Lian-kun, S.; Jiang, X.; Zhang, Y.; Kang, J.; Meng, H.; Li, H.; Jing, S. Genipin protects against cerebral ischemia- reperfusion injury by regulating the UCP2-SIRT3 signaling pathway. Eur. J. Pharmacol. 2018, 845, 56–64. [Google Scholar] [CrossRef]
- Ayyasamy, V.; Owens, K.M.; Desouki, M.M.; Liang, P.; Bakin, A.; Thangaraj, K.; Buchsbaum, D.J.; LoBuglio, A.F.; Singh, K.K. Cellular Model of Warburg Effect Identifies Tumor Promoting Function of UCP2 in Breast Cancer and Its Suppression by Genipin. PLoS ONE 2011, 6, e24792. [Google Scholar] [CrossRef] [Green Version]
- Wang, R.; MoYung, K.; Zhao, Y.; Poon, K. A Mechanism for the Temporal Potentiation of Genipin to the Cytotoxicity of Cisplatin in Colon Cancer Cells. Int. J. Med. Sci. 2016, 13, 507–516. [Google Scholar] [CrossRef] [Green Version]
- Lou, J.; Wang, Y.; Wang, X.; Jiang, Y. Uncoupling Protein 2 Regulates Palmitic Acid-Induced Hepatoma Cell Autophagy. Biomed Res. Int. 2014, 2014, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Yu, J.; Shi, L.; Shen, X.; Zhao, Y. UCP2 regulates cholangiocarcinoma cell plasticity via mitochondria-to-AMPK signals. Biochem. Pharmacol. 2019, 166, 174–184. [Google Scholar] [CrossRef]
- Ahani, N.; Sangtarash, M.H.; Houshmand, M.; Eskandani, M.A. Genipin induces cell death via intrinsic apoptosis pathways in human glioblastoma cells. J. Cell. Biochem. 2019, 120, 2047–2057. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, M.K.; Shen, H.; Tang, F.R.; Arfuso, F.; Rajesh, M.; Wang, L.; Kumar, A.P.; Bian, J.; Goh, B.C.; Bishayee, A.; et al. Potential role of genipin in cancer therapy. Pharmacol. Res. 2018, 133, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Dando, I.; Fiorini, C.; Pozza, E.D.; Padroni, C.; Costanzo, C.; Palmieri, M.; Donadelli, M. UCP2 inhibition triggers ROS-dependent nuclear translocation of GAPDH and autophagic cell death in pancreatic adenocarcinoma cells. Biochim. Biophys. Acta Mol. Cell Res. 2013, 1833, 672–679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Habtemariam, S.; Lentini, G. Plant-derived anticancer agents: Lessons from the pharmacology of geniposide and its aglycone, genipin. Biomedicines 2018, 6, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jo, M.J.; Jeong, S.; Yun, H.K.; Kim, D.Y.; Kim, B.R.; Kim, J.L.; Na, Y.J.; Park, S.H.; Jeong, Y.A.; Kim, B.G.; et al. Genipin induces mitochondrial dysfunction and apoptosis via downregulation of Stat3/mcl-1 pathway in gastric cancer. BMC Cancer 2019, 19, 739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, B.R.; Jeong, Y.A.; Na, Y.J.; Park, S.H.; Jo, M.J.; Kim, J.L.; Jeong, S.; Lee, S.-Y.; Kim, H.J.; Oh, S.C.; et al. Genipin suppresses colorectal cancer cells by inhibiting the Sonic Hedgehog pathway. Oncotarget 2017, 8, 101952–101964. [Google Scholar] [CrossRef] [Green Version]
- Xue, A.; Zheng, L.; Tan, G.; Wu, S.; Wu, Y.; Cheng, L.; Qu, J. Genipin-Crosslinked Donor Sclera for Posterior Scleral Contraction/Reinforcement to Fight Progressive Myopia. Investig. Opthalmology Vis. Sci. 2018, 59, 3564. [Google Scholar] [CrossRef] [Green Version]
- Manickam, B.; Sreedharan, R.; Elumalai, M. ‘Genipin’ – The Natural Water Soluble Cross-linking Agent and Its Importance in the Modified Drug Delivery Systems: An Overview. Curr. Drug Deliv. 2014, 11, 139–145. [Google Scholar] [CrossRef]
- Cho, M.; Jung, S.W.; Lee, S.; Son, K.; Park, G.H.; Jung, J.-W.; Shin, Y.S.; Seo, T.; Hyosun, C.; Kang, H. Genipin Enhances Kaposi’s Sarcoma-Associated Herpesvirus Genome Maintenance. PLoS ONE 2016, 11, e0163693. [Google Scholar] [CrossRef] [Green Version]
- Nonato, D.T.T.; Vasconcelos, S.M.M.; Mota, M.R.L.; de Barros Silva, P.G.; Cunha, A.P.; Ricardo, N.M.P.S.; Pereira, M.G.; Assreuy, A.M.S.; Chaves, E.M.C. The anticonvulsant effect of a polysaccharide-rich extract from Genipa americana leaves is mediated by GABA receptor. Biomed. Pharmacother. 2018, 101, 181–187. [Google Scholar] [CrossRef]
- Madeira, J.C.; da Silva, G.V.L.; Batista, J.J.; Saraiva, G.D.; Santos, G.R.C.; Assreuy, A.M.S.; Mourão, P.A.S.; Pereira, M.G. An arabinogalactan-glycoconjugate from Genipa americana leaves present anticoagulant, antiplatelet and antithrombotic effects. Carbohydr. Polym. 2018, 202, 554–562. [Google Scholar] [CrossRef] [PubMed]
- De Júnior, D.L.S.; da Silva, Í.M.; Benjamim, L.E.; Teotônio, O.; Gonçalves, F.J.; Tôrres, C.M.; Salviano, R.C.; Leandro; Lopes, M.J.P.; de Aquino, P.E.A.; et al. Efeito antimicrobiano e modulador do extrato hidroalcoólico de Genipa americana (Jenipapo). Rev. Saúde (Sta. Maria) 2019, 45. [Google Scholar]
- Da Souza, R.O.S.; Sousa, P.L.; de Menezes, R.R.P.P.B.; Sampaio, T.L.; Tessarolo, L.D.; Silva, F.C.O.; Pereira, M.G.; Martins, A.M.C. Trypanocidal activity of polysaccharide extract from Genipa americana leaves. J. Ethnopharmacol. 2018, 210, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Moura, M.N. Hipóteses Filogenéticas Baseadas em Caracteres Moleculares e Estudos do Tamanho do Genoma em Dyckia Schult. & Schult.f. E Encholirium Mart. ex Schult. & Schult.f. (Bromeliaceae). Master’s Thesis, Universidade Federal de Viçosa, Viçosa, Brazil, 2014. [Google Scholar]
- Pacchioni, F.V.T.L.; Oliveira, R.; Matias, R.; Wendt, C.L.G.R. Perfil toxicológico da planta Genipa americana L. com potencial farmacológico em Danio rerio. 9° Semin. Inic. Cient. 2018. Available online: https://repositorio.pgsskroton.com.br/bitstream/123456789/22514/1/UNIDERP%20-%20Felipe%20Villar%20Telles%20Lunardelli.pdf (accessed on 21 July 2020).
- Manetti, L.M.; Delaporte, R.H.; Laverde, A., Jr. Metabólitos secundários da família bromeliaceae. Quim. Nova 2009, 32, 1885–1897. [Google Scholar] [CrossRef]
- Souza, C.P.F. Caracterização de Variedades de Abacaxi e Sua Potencial Utilização Como Fonte de Fibras. Master’s Thesis, Universidade Federal do Recôncavo da Bahia, Cruz das Almas, Brazil, 2015. [Google Scholar]
- Vallés, D.; Cantera, A.M.B. Antiacanthain A: New proteases isolated from Bromelia antiacantha Bertol. (Bromeliaceae). Int. J. Biol. Macromol. 2018, 113, 916–923. [Google Scholar] [CrossRef]
- Da Cruz, M.P. Estudo fitoquímico e avaliação da atividade antimicrobiana de Bromelia laciniosa Mart. ex Schult. f. (Bromeliaceae). Master’s Thesis, Universidade Federal do Vale do São Francisco, Petrolina, Brazil, 2017. [Google Scholar]
- Filippon, S.; Fernandes, C.D.; Ferreira, D.K.; da Silva, D.L.S.; Altrak, G.; Duarte, A.S.; dos Reis, M.S. Bromelia antiacantha Bertol.(Bromeliaceae): Caracterização demográfica e potencial de manejo em uma população no Planalto Norte Catarinense. Biodiversidade Bras. 2012, 2, 83–91. [Google Scholar]
- Manetti, L.M.; Turra, A.F.; Takemura, O.S.; Svidzinski, T.I.E.; Laverde Junior, A. Avaliação das atividades antimicrobiana, citotóxica, moluscicida e antioxidante de Bromelia antiacantha Bertol. (Bromeliaceae). Rev. Bras. Plantas Med. 2010, 12, 406–413. [Google Scholar] [CrossRef] [Green Version]
- Dettke, G.A.; Milaneze-Gutierre, M.A. Anatomia vegetativa de Bromelia antiacantha Bertol. (Bromeliaceae, Bromelioideae). Balduinia 2014, 13, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Vallés, D.; Furtado, S.; Cantera, A.M.B. Characterization of news proteolytic enzymes from ripe fruits of Bromelia antiacantha Bertol. (Bromeliaceae). Enzyme Microb. Technol. 2007, 40, 409–413. [Google Scholar] [CrossRef]
- Bersi, G.; Vallés, D.; Penna, F.; Cantera, A.M.; Barberis, S. Valorization of fruit by-products of Bromelia antiacantha Bertol.: Protease obtaining and its potential as additive for laundry detergents. Biocatal. Agric. Biotechnol. 2019, 18, 101099. [Google Scholar] [CrossRef]
- Nogueira, A.C.; Côrtes, I.M.R.; Verçoza, F.C. A família Bromeliaceae na Área de Proteção Ambiental de Grumari, Rio de Janeiro, RJ, Brasil. Nat. Line 2011, 9, 91–95. [Google Scholar]
- Monteiro, R.F.; Mantovani, A.; Forzza, R.C. Morphological Phylogenetic Analysis of Two Early-Diverging Genera of Bromelioideae (Bromeliaceae). Rodriguésia 2015, 66, 505–521. [Google Scholar] [CrossRef]
- Grandi, T.S.M. Tratado de Plantas Medicinais Mineiras, Nativas e Cultivadas, 1st ed.; Adequatio Estúdio: Belo Horizonte, Brazil, 2014; ISBN 978-85-68322-00-0. [Google Scholar]
- Yazbek, P.B.; Matta, P.; Passero, L.F.; Santos, G.; Braga, S.; Assunção, L.; Sauini, T.; Cassas, F.; Garcia, R.J.F.; Honda, S.; et al. Plants utilized as medicines by residents of Quilombo da Fazenda, Núcleo Picinguaba, Ubatuba, São Paulo, Brazil: A participatory survey. J. Ethnopharmacol. 2019, 244, 112123. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, F.H.; Martins, G.V.; Oliveira, C.; Contrera, M.G.D.; Ferro, D.; Regrado, S.C.H.; Lopes, R.A.; Sala, M.A. Hepatotoxicidade de plantas medicinais na preparação fitoterápica usada popularmente como vermífugo contendo Mentha villosa L., Bromelia antiacantha Bertol, Chenopodium ambrosioides L., Citrus sinensis L., Punica granatum L. e Curcubita pepo L. em camund. Rev. Científica da Univ. Fr. 2005, 5, 215–222. [Google Scholar]
- Filippon, S. Aspectos da demografia, fenologia e uso tradicionaldo Caraguatá (Bromelia antiacantha Bertol.) no Planalto Norte Catarinense. Master’s Thesis, Universidade Federal de Santa Catarina, Florianópolis, Brazil, 2009. [Google Scholar]
- Nasser, N.P.A.; Scheeren, N.B.; Ramos, R.F.; Bellé, C.; Nora, D.D.; Betemps, D.L. Germinação de sementes de Bromelia antiacantha em diferentes fotoperíodos. Rev. Eletrônica Científica UERGS 2019, 5, 296–301. [Google Scholar] [CrossRef] [Green Version]
- Fabri, R.L.; da Costa, J.A.B.M. Perfil farmacognóstico e avaliação das atividades citotóxica e antibacteriana de Bromelia antiacantha Bertol. Rev. Electron. Farmácia 2012, 9, 37–48. [Google Scholar] [CrossRef]
- Santos, V.N.C.; de Freitas, R.A.; Deschamps, F.C.; Biavatti, M.W. Ripe fruits of Bromelia antiacantha: Investigations on the chemical and bioactivity profile. Rev. Bras. Farmacogn. 2009, 19, 358–365. [Google Scholar] [CrossRef]
- Zanella, C.M. Caracterização genética, morfológica e fitoquímica de populações de Bromelia antiacantha (Bertol.) do Rio Grande do Sul. Master’s Thesis, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil, 2009. [Google Scholar]
- Manetti, L.M.; Turra, A.F.; Takemura, O.S.; Júnior, A.L. Avaliação da atividade hemolítica de Bromelia antiacantha Bertol. (Bromeliaceae). Arq. Ciênc. Saúde UNIPAR 2010, 14, 43–47. [Google Scholar]
- Krumreich, F.D.; Corrêa, A.P.A.; da Silva, S.D.S.; Zambiazi, R.C. Composição físico-química e de compostos bioativos em frutos de Bromelia antiacantha Bertol. Rev. Bras. Frutic. 2015, 37, 450–456. [Google Scholar] [CrossRef]
- Jang, J.; Kim, S.-M.; Yee, S.-M.; Kim, E.-M.; Lee, E.-H.; Choi, H.-R.; Lee, Y.-S.; Yang, W.-K.; Kim, H.-Y.; Kim, K.-H.; et al. Daucosterol suppresses dextran sulfate sodium (DSS)-induced colitis in mice. Int. Immunopharmacol. 2019, 72, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Huang, X.; Liao, T.; Li, G.; Yu, X.; You, Y.; Huang, Y. Daucosterol induces autophagic-dependent apoptosis in prostate cancer via JNK activation. Biosci. Trends 2019, 13, 160–167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, B.; Jiang, P.; Liu, W.; Xu, H.; Li, Y.; Li, Z.; Ma, H.; Yu, Y.; Li, X.; Ye, X. Role of Daucosterol Linoleate on Breast Cancer: Studies on Apoptosis and Metastasis. J. Agric. Food Chem. 2018, 66, 6031–6041. [Google Scholar] [CrossRef] [PubMed]
- Zingue, S.; Gbaweng Yaya, A.J.; Michel, T.; Ndinteh, D.T.; Rutz, J.; Auberon, F.; Maxeiner, S.; Chun, F.K.H.; Tchinda, A.T.; Njamen, D.; et al. Bioguided identification of daucosterol, a compound that contributes to the cytotoxicity effects of Crateva adansonii DC (capparaceae) to prostate cancer cells. J. Ethnopharmacol. 2020, 247, 112251. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, M.A.; Farimani, M.M. Inactivation of PI3K/Akt pathway and upregulation of PTEN gene are involved in daucosterol, isolated from Salvia sahendica, induced apoptosis in human breast adenocarcinoma cells. South African J. Bot. 2014, 93, 37–47. [Google Scholar] [CrossRef] [Green Version]
- Zeng, J.; Liu, X.; Li, X.; Zheng, Y.; Liu, B.; Xiao, Y. Daucosterol Inhibits the Proliferation, Migration, and Invasion of Hepatocellular Carcinoma Cells via Wnt/β-Catenin Signaling. Molecules 2017, 22, 862. [Google Scholar] [CrossRef]
- Jiang, L.; Yuan, X.; Yang, N.; Ren, L.; Zhao, F.; Luo, B.; Bian, Y.; Xu, J.; Lu, D.; Zheng, Y.; et al. Daucosterol protects neurons against oxygen–glucose deprivation/reperfusion-mediated injury by activating IGF1 signaling pathway. J. Steroid Biochem. Mol. Biol. 2015, 152, 45–52. [Google Scholar] [CrossRef]
- Podolak, I.; Galanty, A.; Sobolewska, D. Saponins as cytotoxic agents: A review. Phytochem. Rev. 2010, 9, 425–474. [Google Scholar] [CrossRef] [Green Version]
- Ellington, A.A. Induction of macroautophagy in human colon cancer cells by soybean B-group triterpenoid saponins. Carcinogenesis 2004, 26, 159–167. [Google Scholar] [CrossRef] [Green Version]
- Venkatakrishnan, K.; Chiu, H.-F.; Cheng, J.-C.; Chang, Y.-H.; Lu, Y.-Y.; Han, Y.-C.; Shen, Y.-C.; Tsai, K.-S.; Wang, C.-K. Comparative studies on the hypolipidemic, antioxidant and hepatoprotective activities of catechin-enriched green and oolong tea in a double-blind clinical trial. Food Funct. 2018. [Google Scholar] [CrossRef]
- Nagao, T.; Meguro, S.; Hase, T.; Otsuka, K.; Komikado, M.; Tokimitsu, I.; Yamamoto, T.; Yamamoto, K. A Catechin-rich Beverage Improves Obesity and Blood Glucose Control in Patients With Type 2 Diabetes. Obesity 2009, 17, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Inami, S.; Takano, M.; Yamamoto, M.; Murakami, D.; Tajika, K.; Yodogawa, K.; Yokoyama, S.; Ohno, N.; Ohba, T.; Sano, J.; et al. Tea Catechin Consumption Reduces Circulating Oxidized Low-Density Lipoprotein. Int. Heart J. 2007, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsuyama, T.; Tanaka, Y.; Kamimaki, I.; Nagao, T.; Tokimitsu, I. Catechin Safely Improved Higher Levels of Fatness, Blood Pressure, and Cholesterol in Children. Obesity 2008, 16. [Google Scholar] [CrossRef] [PubMed]
- Alañón, M.E.; Castle, S.M.; Serra, G.; Lévèques, A.; Poquet, L.; Actis-Goretta, L.; Spencer, J.P.E. Acute study of dose-dependent effects of (−)-epicatechin on vascular function in healthy male volunteers: A randomized controlled trial. Clin. Nutr. 2020. [Google Scholar] [CrossRef] [PubMed]
- Hollands, W.J.; Tapp, H.; Defernez, M.; Perez Moral, N.; Winterbone, M.S.; Philo, M.; Lucey, A.J.; Kiely, M.E.; Kroon, P.A. Lack of acute or chronic effects of epicatechin-rich and procyanidin-rich apple extracts on blood pressure and cardiometabolic biomarkers in adults with moderately elevated blood pressure: A randomized, placebo-controlled crossover trial. Am. J. Clin. Nutr. 2018, 108, 1006–1014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dower, J.I.; Geleijnse, J.M.; Gijsbers, L.; Zock, P.L.; Kromhout, D.; Hollman, P.C.H. Effects of the pure flavonoids epicatechin and quercetin on vascular function and cardiometabolic health: A randomized, double-blind, placebo-controlled, crossover trial. Am. J. Clin. Nutr. 2015. [Google Scholar] [CrossRef]
- Edwards, R.L.; Lyon, T.; Litwin, S.E.; Rabovsky, A.; Symons, J.D.; Jalili, T. Quercetin Reduces Blood Pressure in Hypertensive Subjects. J. Nutr. 2007. [Google Scholar] [CrossRef]
- Duranti, G.; Ceci, R.; Patrizio, F.; Sgrò, P.; Di Luigi, L.; Sabatini, S.; Felici, F.; Bazzucchi, I. Chronic consumption of quercetin reduces erythrocytes oxidative damage: Evaluation at resting and after eccentric exercise in humans. Nutr. Res. 2018. [Google Scholar] [CrossRef]
- Shi, Y.; Williamson, G. Quercetin lowers plasma uric acid in pre-hyperuricaemic males: A randomised, double-blinded, placebo-controlled, cross-over trial. Br. J. Nutr. 2016, 115, 800–806. [Google Scholar] [CrossRef] [Green Version]
- Javadi, F.; Ahmadzadeh, A.; Eghtesadi, S.; Aryaeian, N.; Zabihiyeganeh, M.; Rahimi Foroushani, A.; Jazayeri, S. The Effect of Quercetin on Inflammatory Factors and Clinical Symptoms in Women with Rheumatoid Arthritis: A Double-Blind, Randomized Controlled Trial. J. Am. Coll. Nutr. 2017. [Google Scholar] [CrossRef]
- Kondratiuk, V.E.; Synytsia, Y.P. Effect of quercetin on the echocardiographic parameters of left ventricular diastolic function in patients with gout and essential hypertension. Wiad. Lek. 2018, 71, 1554–1559. [Google Scholar] [PubMed]
- Khorshidi, M.; Moini, A.; Alipoor, E.; Rezvan, N.; Gorgani-Firuzjaee, S.; Yaseri, M.; Hosseinzadeh-Attar, M.J. The effects of quercetin supplementation on metabolic and hormonal parameters as well as plasma concentration and gene expression of resistin in overweight or obese women with polycystic ovary syndrome. Phyther. Res. 2018, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sajadi Hezaveh, Z.; Azarkeivan, A.; Janani, L.; Hosseini, S.; Shidfar, F. The effect of quercetin on iron overload and inflammation in β-thalassemia major patients: A double-blind randomized clinical trial. Complement. Ther. Med. 2019, 46. [Google Scholar] [CrossRef] [PubMed]
- Rezvan, N.; Moini, A.; Janani, L.; Mohammad, K.; Saedisomeolia, A.; Nourbakhsh, M.; Gorgani-Firuzjaee, S.; Mazaherioun, M.; Hosseinzadeh-Attar, M. Effects of Quercetin on Adiponectin-Mediated Insulin Sensitivity in Polycystic Ovary Syndrome: A Randomized Placebo-Controlled Double-Blind Clinical Trial. Horm. Metab. Res. 2016. [Google Scholar] [CrossRef]
- Patrizio, F.; Ditroilo, M.; Felici, F.; Duranti, G.; De Vito, G.; Sabatini, S.; Sacchetti, M.; Bazzucchi, I. The acute effect of Quercetin on muscle performance following a single resistance training session. Eur. J. Appl. Physiol. 2018. [Google Scholar] [CrossRef]
- Talirevic, E.; Sehovic, J. Quercetin in the Treatment of Dyslipidemia. Med. Arch. 2012, 66, 87–88. [Google Scholar] [CrossRef] [Green Version]
- Boots, A.W.; Drent, M.; de Boer, V.C.J.; Bast, A.; Haenen, G.R.M.M. Quercetin reduces markers of oxidative stress and inflammation in sarcoidosis. Clin. Nutr. 2011, 30, 506–512. [Google Scholar] [CrossRef] [Green Version]
- Katada, S.; Watanabe, T.; Mizuno, T.; Kobayashi, S.; Takeshita, M.; Osaki, N.; Kobayashi, S.; Katsuragi, Y. Effects of Chlorogenic Acid-Enriched and Hydroxyhydroquinone-Reduced Coffee on Postprandial Fat Oxidation and Antioxidative Capacity in Healthy Men: A Randomized, Double-Blind, Placebo-Controlled, Crossover Trial. Nutrients 2018, 10, 525. [Google Scholar] [CrossRef] [Green Version]
- Saitou, K.; Ochiai, R.; Kozuma, K.; Sato, H.; Koikeda, T.; Osaki, N.; Katsuragi, Y. Effect of Chlorogenic Acids on Cognitive Function: A Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients 2018, 10, 1337. [Google Scholar] [CrossRef] [Green Version]
- Mubarak, A.; Bondonno, C.P.; Liu, A.H.; Considine, M.J.; Mas, E.; Croft, K.D.; Hodgson, J.M. Acute effects of chlorogenic acid on nitric oxide status, endothelial function and blood pressure in healthy volunteers: A randomised trial. J. Agric. Food Chem. 2012, 53, S191–S192. [Google Scholar] [CrossRef]
- Vieyra-Garcia, P.; Fink-Puches, R.; Porkert, S.; Lang, R.; Pöchlauer, S.; Ratzinger, G.; Tanew, A.; Selhofer, S.; Paul-Gunther, S.; Hofer, A.; et al. Evaluation of Low-Dose, Low-Frequency Oral Psoralen–UV-A Treatment With or Without Maintenance on Early-Stage Mycosis Fungoides. JAMA Dermatol. 2019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brass, D.; Fouweather, T.; Stocken, D.D.; Macdonald, C.; Wilkinson, J.; Lloyd, J.; Farr, P.M.; Reynolds, N.J.; Hampton, P.J. An observer-blinded randomized controlled pilot trial comparing localized immersion psoralen-ultraviolet A with localized narrowband ultraviolet B for the treatment of palmar hand eczema. Br. J. Dermatol. 2018, 179, 8–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khanna, N.; Nazli, T.; Siddiqui, K.; Kalaivani, M. Rais-ur-Rahman A non-inferiority randomized controlled clinical trial comparing Unani formulation & psoralen plus ultraviolet A sol in chronic plaque psoriasis. Indian J. Med. Res. 2018, 66–72. [Google Scholar] [CrossRef]
- Brazzelli, V.; Grassi, S.; Merante, S.; Grasso, V.; Ciccocioppo, R.; Bossi, G.; Borroni, G. Narrow-band UVB phototherapy and psoralen-ultraviolet A photochemotherapy in the treatment of cutaneous mastocytosis: A study in 20 patients. Photodermatol. Photoimmunol. Photomed. 2016, 32, 238–246. [Google Scholar] [CrossRef]
- Bansal, S.; Sahoo, B.; Garg, V. Psoralen-narrowband UVB phototherapy for the treatment of vitiligo in comparison to narrowband UVB alone. Photodermatol. Photoimmunol. Photomed. 2013. [Google Scholar] [CrossRef]
- Calzavara-Pinton, P.; Ortel, B.; Carlino, A.; Honigsmann, H.; Panfilis, G. A reappraisal of the use of 5-methoxypsoralen in the therapy of psoriasis. Exp. Dermatol. 1992. [Google Scholar] [CrossRef]
- Stefanaki, C.; Fasoulaki, X.; Kouris, A.; Caroni, C.; Papagianaki, K.; Mavrogianni, P.; Nicolaidou, E.; Gregoriou, S.; Antoniou, C. A randomized trial of efficacy of beta-sitosterol and its glucoside as adjuvant to cryotherapy in the treatment of anogenital warts. J. Dermatolog. Treat. 2015, 1–4. [Google Scholar] [CrossRef]
- Donald, P.R.; Lamprecht, J.H.; Freeston, M.; Al, E. A randomised placebo-controlled trial of the efficacy of beta-sitosterol and its glucoside as adjuvants in the treatment of pulmonary tuberculosis. Int. J. Tuberc. Lung. Dis. 1997, 1, 518–522. [Google Scholar]


| Compounds | Molecular Structures | Chromatographic Methods for the Isolation and Identification | Solvent Used/Essential Oil | Plant Part Used | Collection Site | References |
|---|---|---|---|---|---|---|
| Myricetin (1, F) |  | HPLC | Hydroalcoholic 5:95 (v/v water, ethanol) | Fruit | N.I. | [32] |
| Quercetin (2, F) |  | |||||
| Catechin (3, F) |  | LC-MS | Acetone Methanol | Pulp Fruit peel Seed | Parintins, Amazonas-Brazil | [33] |
| Epicatechin (4, F) |  | Acetone | Fruit peel Seed | |||
| Gallic acid (5, PA) |  | Acetone | Pulp | |||
| Luteolin (6, F) |  | Acetone Methanol | Seed | |||
| Naringenin (7, F) |  | Acetone Methanol | Fruit peel Seed | |||
| p-Coumaric acid (8, PA) |  | Acetone | Pulp | |||
| Quinic acid (9, CL) |  | Acetone | Pulp | |||
| Rutin (10, F) |  | Acetone Methanol | Fruit peel | |||
| Acacetin (11, F) |  | UHPLC–MS/MS | Methanol | Fruit | Manaus, Amazonas -Brazil | [34] |
| Caffeic acid (12, PA) |  | |||||
| Catechin (3, F) |  | |||||
| Chlorogenic acid (13, PA) |  | |||||
| Epicatechin (4, F) |  | |||||
| Eriodictyol (14, F) |  | |||||
| Ferulic acid (15, PA) |  | |||||
| Gallic acid (5, PA) |  | |||||
| p-Coumaric acid (8, PA) |  | |||||
| Quercetin (2, F) |  | |||||
| Quinic acid (9, CL) |  | |||||
| Rutin (10, F) |  | |||||
| Syringic acid (16, PA) |  | |||||
| β-Bisabolene (17, T) |  | HS-SPME-GC-MS | DVB/CAR/PDMS | |||
| Linalool (18, T) |  | |||||
| Dihydroxybenzoic acid hexoside (19, PA) | * | UHPLC | Hydroalcoholic 1:1 (v/v methanol, water) | Leaf Stem | Dourados, Mato Grosso do Sul-Brazil | [35] |
| Kaempferol-diglycoside (20, F) | * | |||||
| Methylquercetin-diglycoside (21, F) | * | |||||
| Quercetin-diglycoside (22, F) | * | |||||
| Quercetin-rhamnoside (23, F) | * | |||||
| Dicaffeoylquinic acid (24, PA) | * | |||||
| Acacetin (11, F) |  | UHPLC–MS/MS | Hydroalcoholic 1:4 (m/v, 70% ethanol) | Leaf | São Luís, Maranhão-Brazil | [36] |
| Caffeic acid (12, PA) |  | |||||
| Catechin (3, F) |  | |||||
| Gallic acid (5, PA) |  | |||||
| Quercetin (2, F) |  | |||||
| Quinic acid (9, CL) |  | |||||
| Rutin (10, F) |  |
| Compounds | Molecular Structures | Chromatographic Methods for the Isolation and Identification | Solvent Used/Essential Oil | Plant Part Used | Collection Site | References |
|---|---|---|---|---|---|---|
| Psoralen (25, C) |  | HPLC | Methanol | Root cortex Leaf Twigs Latex Heartwood of the root Root cortex | São Paulo-Brazil | [91] |
| Bergapten (26, C) |  | |||||
| 5,7,3‘,4’-Tetrahydroxy-6-C-glucopyranosylflavone (27, F) |  | Water | Root cortex | |||
| 28 4 5,7,3‘,4’-tetrahydroxy-3-O-β-d-galactopyranosylflavonol (F) |  | |||||
| Psoralen (25, C) |  | |||||
| Bergapten (26, C) |  | |||||
| Psoralen (25, C) |  | HPLC | Ethanol (v/v 95%) | Root | Jussára, Goiás-Brazil | [92] |
| Bergapten (26, C) |  | |||||
| Gaudichaudine (29, C) |  | HPLC | Dichloromethane | Root bark | Araguari, Minas Gerais-Brazil | [93] |
| Luvangetin (30, C) |  | |||||
| (+)-(2’S,3’R)-3′-hydroxymarmesin (31, C) |  | |||||
| Xanthyletin (32, C) |  | |||||
| Psoralen (25, C) |  | |||||
| Bergapten (26, C) |  | |||||
| Marmesin (33, C) |  | CC | Dichloromethane | Root bark | Araguari, Minas Gerais-Brazil | [94] |
| 1′,2′-Dehydromarmesin (34, C) |  | |||||
| 8-Methoxymarmesin (35, C) |  | |||||
| 1′-Hydroxy-3′-O-β-glucopyranosylmarmesin (36, C) |  | |||||
| 2′,4′,4-Trihydroxy-3′3-diprenylchalcone (37, CH) |  | |||||
| β-Amyrin (38, T) |  |
| Compounds | Molecular Structures | Chromatographic Methods for the Isolation and Identification | Solvent Used/Essential Oil | Plant Part Used | Collection Site | References |
|---|---|---|---|---|---|---|
| Asystasioside D (39, T) |  | UHPLC | Hydroalcoholic 70:30 (v/v ethanol, water) | Leaf | Natal, Rio Grande do Norte-Brazil | [139] |
| Geniposidic acid (40, T) |  | |||||
| Tarenoside (41, T) |  | |||||
| Teneoside A (42, T) |  | |||||
| Kaempferol-3-O-hexoside-deoxyhexoside-7-O-deoxyhexoside (43, F) |  | |||||
| Isorhamnetin-3-O-hexoside-deoxyhexoside-7-O-deoxyhexoside (44, F) |  | |||||
| Quercetin-3-O-hexoside-deoxyhexoside (45, F) |  | |||||
| Kaempferol-3-O-hexoside-deoxyhexoside (46, F) |  | |||||
| Isorhamnetin-3-O-hexoside-deoxyhexoside (47, F) |  | |||||
| Geniposidic acid (40, T) |  | [140] | ||||
| Geniposide (48, T) |  | |||||
| Genipin-gentiobioside (49, T) |  | |||||
| Genameside A (50, T) |  | |||||
| Genameside B (51, T) |  | |||||
| Genameside C (52, T) |  | |||||
| Genameside D (53, T) |  | |||||
| Gardenoside (54, T) |  | |||||
| Genipin (55, T) |  | HPLC | Hydroalcoholic (v/v, 80% methanol, ultra-pure water) | Fruit | São Paulo-Brazil | [141] |
| 1-Hydroxy-7-(hydroxymethyl)-1,4aH,5H,7aH-cyclopenta[c]pyran-4-carbaldehyde (56, T) |  | HPLC | Hydroalcoholic (v/v, 70 % ethanol, water) | Leaf | Natal, Rio Grande do Norte-Brazil | [142] |
| 7-(Hydroxymethyl)-1-methoxy-1H,4aH,5H,7aH-cyclopenta[c]pyran-4-carbaldehyde (57, T) |  | |||||
| Geniposidic acid (40, T) |  | UHPLC | Methanol | Mesocarp Endocarp | Campinas, São Paulo-Brazil | [143] |
| Gardenosid (54, T) |  | |||||
| Genipin-1-β-gentiobioside (49, T) |  | |||||
| Geniposide (48, T) |  | |||||
| 6′’-O-p-Coumaroyl-1-β-gentiobioside geniposidic acid (59, T) |  | |||||
| 6′′-O-p-Coumaroylgenipin-gentiobioside (60, T) |  | |||||
| Genipin (55, T) |  | |||||
| 6′-O-p-Coumaroyl-geniposidic acid (61, T) |  | |||||
| 6′-O-Feruloyl-geniposidic acid (62, T) |  | |||||
| Genipin (55, T) |  | HPLC | Ethanol | Mesocarp Seeds Fruit peel Whole fruit Endocarp | Paraibuna, São Paulo-Brazil | [144] |
| Geniposide (48, T) |  |
| Compounds | Molecular Structures | Chromatographic Methods for the Isolation and Identification | Solvent Used/Essential Oil | Plant Part Used | Collection Site | References |
|---|---|---|---|---|---|---|
| Alkaloid | * | _ | Methanol | Fruit | Rio Pomba, Minas Gerais-Brazil | [193] |
| Flavonoid | * | |||||
| Tannin | * | |||||
| Terpene | * | |||||
| Anthraquinone | * | |||||
| Coumarin | * | |||||
| Alkaloid | * | Leaf | ||||
| Flavonoid | * | |||||
| Tannin | * | |||||
| Terpene | * | |||||
| Coumarin | * | |||||
| Flavonoid | * | _ | Methanol | Fruit Leaf | Umuarama, Paraná-Brazil | [182] |
| Tannin | * | |||||
| Saponin | * | |||||
| Flavonoid | * | _ | Water | Fruit | Vale do Itajaí, Santa Catarina-Brazil | [194] |
| Flavonoid | * | _ | Hydroalcoholic | Leaf | Viamão, Rio Grande do Sul-Brazil | [195] |
| Anthocyanin | * | |||||
| Flavonoid | * | Bract | ||||
| Anthocyanin | * | |||||
| Daucosterol (63, S) |  | CC | Methanol | Leaf | Umuarama, Paraná-Brazil | [196] |
| Species | Compounds | Investigated Pathology | Search Result | References |
|---|---|---|---|---|
| Talisia esculenta | Catechin (3) | Hypercholesterolemia | Hypolipidemic and hepatoprotective effect | [207] |
| Obesity and type 2 diabetes | Reduction of visceral fat, blood pressure and cholesterol | [208] | ||
| Coronary artery disease | Reduction of oxidized LDL in plasma | [209] | ||
| Child obesity | Reduction in waist circumference, systolic blood pressure and LDL levels | [210] | ||
| Epicatechin (4) | Vascular dysfunction | Improved cardiovascular health | [211] | |
| Arterial hypertension | Ineffective on blood pressure, blood lipid profile and glucose control | [212] | ||
| Cardiovascular diseases | Cardioprotective effect and improved insulin resistance | [213] | ||
| Quercetin (2) | Arterial hypertension | Lowering blood pressure | [214] | |
| Increase in free radicals produced after eccentric exercises | Antioxidant and protective effect | [215] | ||
| Hyperuricemia | Significant reduction in elevated plasma uric acid concentrations | [216] | ||
| Rheumatoid arthritis | Significant improvement in clinical symptoms and reduced levels of TNF-α | [217] | ||
| Gout and primary hypertension | Improvement of echocardiographic parameters, left ventricular diastolic function, purine metabolism, renal function, and normalization of blood pressure | [218] | ||
| Overweight or obesity with polycystic ovary syndrome | Significant reduction in gene expression and plasma resistin concentration and considerable decrease in the level of luteinizing hormone and testosterone | [219] | ||
| β-Thalassemia major | Reduced iron overload | [220] | ||
| Chlorogenic acid (13) | Polycystic ovary syndrome | Improvement of insulin resistance and hormonal profile of women with the syndrome. | [221] | |
| Neuromuscular dysfunction | Improvement of neuromuscular performance | [222] | ||
| Dyslipidemia | Decrease in cholesterol, triglycerides and LDL values, and increased HDL levels | [223] | ||
| Sarcoidosis | Reduction of oxidative stress and inflammation | [224] | ||
| Hypertension and fat accumulation | Reduction of blood pressure and body fat | [225] | ||
| Cognitive dysfunction | Improvement of cognitive functions | [226] | ||
| Arterial hypertension | Reduction of systolic and diastolic blood pressure | [227] | ||
| Brosimum gaudichaudii | Psoralen (25) | Fungal ringworm | Oral treatment with a low dose and low frequency of psoralen-UV-A, was safe and effective | [228] |
| Chronic palmar eczema of the hand | Reduced severity of chronic palmar eczema of the hand | [229] | ||
| Chronic moderate to severe plaque psoriasis | Psoralen plus ultraviolet A, are therapeutic options for chronic moderate to severe plaque psoriasis | [230] | ||
| Cutaneous mastocytosis | Efficacy for the treatment of moderate to severe chronic psoriasis | [231] | ||
| Vitiligo | Increased extent of skin repigmentation | [232] | ||
| Bergaptene (26) | Psoriasis | Reduction of signs and symptoms of psoriasis | [233] | |
| Genipa americana | Genipin (55) | Absence of clinical trials in the current literature | _ | _ |
| Asystasioside D (39) | ||||
| Geniposidic acid (40) | ||||
| Tarenoside (41) | ||||
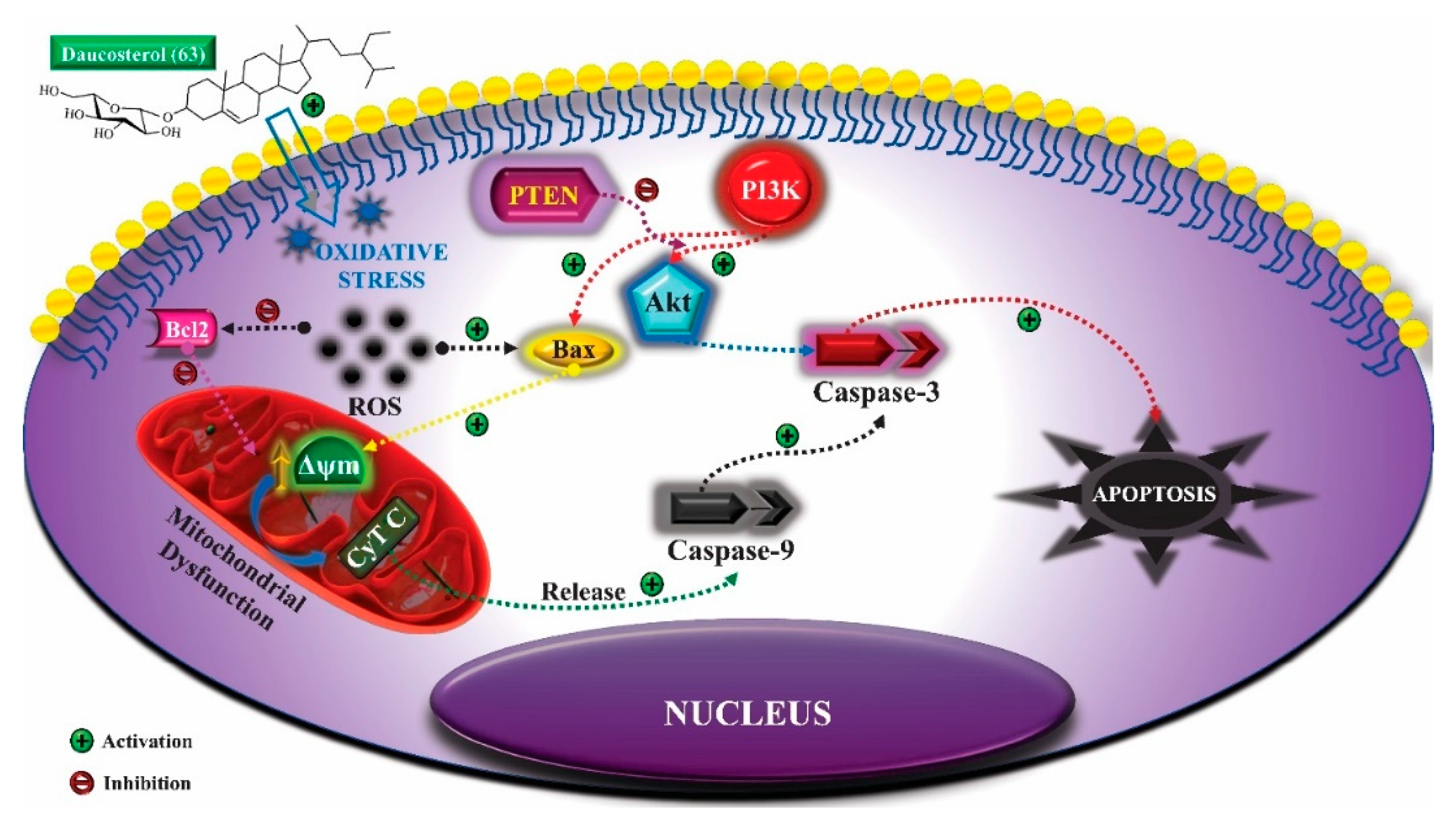
| Bromelia antiacantha | Daucosterol (63) | Anogenital warts | The treatment led to the elimination of injury | [234] |
| Pulmonary Tuberculosis | Improvement in imaging tests and weight gain of the patient | [235] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Carvalho, A.T.; Paes, M.M.; Cunha, M.S.; Brandão, G.C.; Mapeli, A.M.; Rescia, V.C.; Oesterreich, S.A.; Villas-Boas, G.R. Ethnopharmacology of Fruit Plants: A Literature Review on the Toxicological, Phytochemical, Cultural Aspects, and a Mechanistic Approach to the Pharmacological Effects of Four Widely Used Species. Molecules 2020, 25, 3879. https://doi.org/10.3390/molecules25173879
de Carvalho AT, Paes MM, Cunha MS, Brandão GC, Mapeli AM, Rescia VC, Oesterreich SA, Villas-Boas GR. Ethnopharmacology of Fruit Plants: A Literature Review on the Toxicological, Phytochemical, Cultural Aspects, and a Mechanistic Approach to the Pharmacological Effects of Four Widely Used Species. Molecules. 2020; 25(17):3879. https://doi.org/10.3390/molecules25173879
Chicago/Turabian Stylede Carvalho, Aline T., Marina M. Paes, Mila S. Cunha, Gustavo C. Brandão, Ana M. Mapeli, Vanessa C. Rescia, Silvia A. Oesterreich, and Gustavo R. Villas-Boas. 2020. "Ethnopharmacology of Fruit Plants: A Literature Review on the Toxicological, Phytochemical, Cultural Aspects, and a Mechanistic Approach to the Pharmacological Effects of Four Widely Used Species" Molecules 25, no. 17: 3879. https://doi.org/10.3390/molecules25173879








