2.1. Generation of Ionic Liquids
This type of ionic liquids, a form of quaternary ammonium salts R
4N
+Cl
−, can be generated by reaction of a given amine and the corresponding mineral acid [
28,
29].
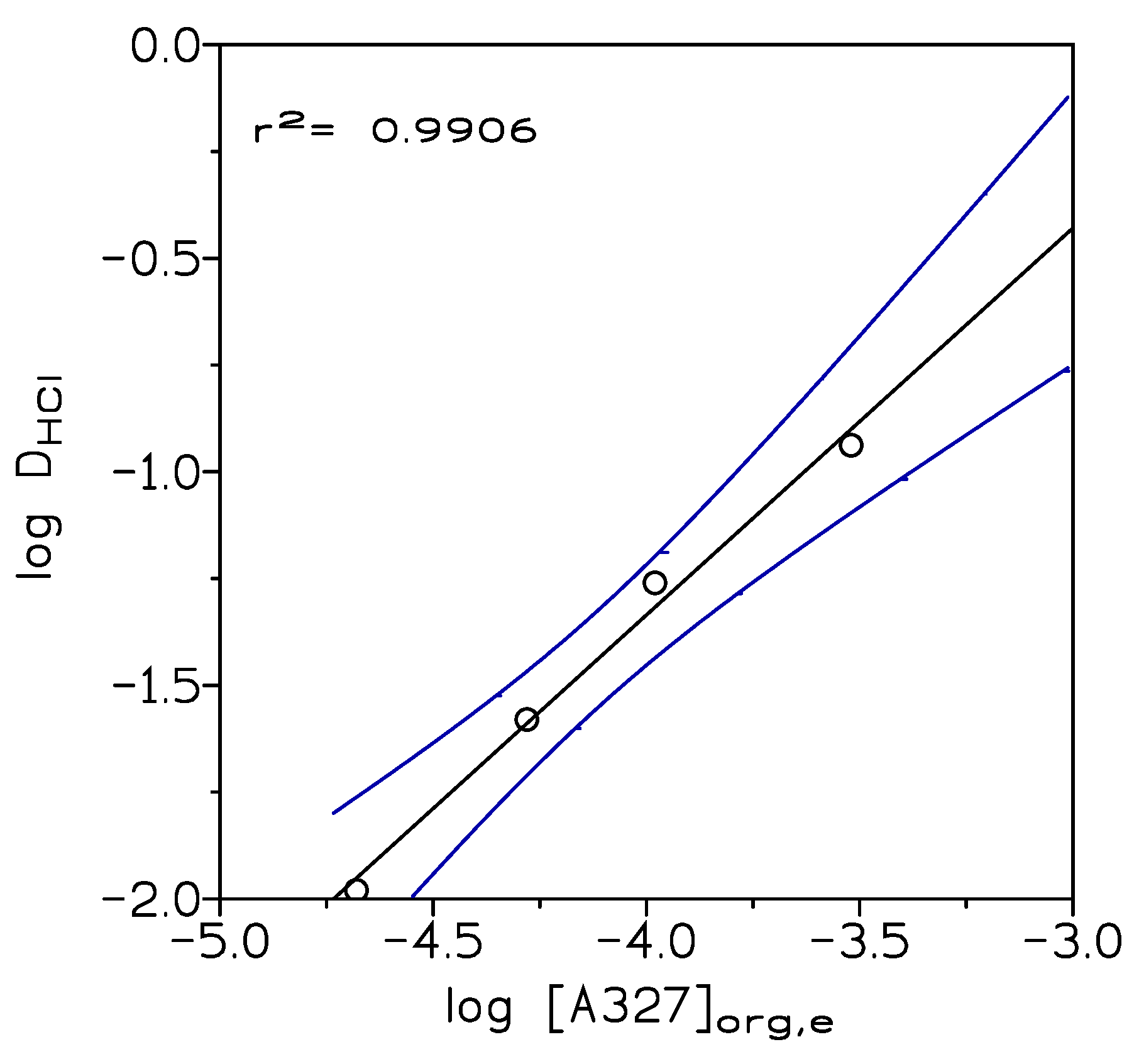
Figure 1 shows the results of extraction of 1 M HCl solution by different amine concentrations in Solvesso 100; it can be seen that a straight line with a slope near 1 was obtained; thus, the ionic liquid was formed according to:
with log K = 2.29 (graphically obtained). The experimental data were numerically treated by a computer program which minimizes function U:
where D
cal and D
exp were the calculated and the experimental distribution coefficients values, respectively.
The numerical treatment proposed formation in the organic phase of a liquid, that is, an ionic liquid, with the stoichiometry indicated in Equation (1) and log K = 2.55 (U = 3.8 × 10−6, σ(log K= 0.558)). In the present study, the ionic liquid was generated in Solvesso 100; this procedure has the advantage of (i) decreasing the viscosity of the ionic liquid and thus of improving phase disengagement, (ii) adjusting the extractant concentration to a given system, avoiding the use of unnecessary extractant concentration excess, and (iii) the yield of an organic phase containing the ionic liquid ready for its further use in metal extraction processing.
2.2. Indium(III) Liquid-Liquid Extraction Results
To investigate the effect of the equilibration time on indium(III) extraction by a A327H+Cl− ionic liquid, various extractions were carried out using O/A relationships of 1 and an organic phase of 1.25% v/v ionic liquid in Solvesso 100 and an aqueous solution containing 0.075 g/L In(III) in 7 M HCl. The temperature was 20 °C and the equilibration time varied from 1 to 60 min. The results show that, under the present experimental conditions, the extraction is not very dependent upon the contact time and equilibrium is achieved after 5 min of contact (94% indium extraction, equivalent to an indium distribution coefficient value of 15.7).
The variation of the temperature (20–60 °C) in the extraction of indium(III) by an ionic liquid was investigated using the same organic and aqueous solutions as described above. The results given in
Table 1 indicated that there is a decrease in indium extraction as the temperature is increased, the extraction reaction is exothermic with ΔH° = −15 kJ/mol.
The metal distribution ratio at 7 M HCl was determined for different indium(III) concentrations. The results are showed in
Table 2. It can be seen that D
In does not depend on the indium concentration. This behavior indicates that metal-polynuclear complexes are not apparently formed in the organic phase.
The variation in the concentration of the ionic liquid in Solvesso 100 in the extraction of indium(III) was also investigated. The temperature was 20 °C, and the aqueous solution contained 0.075 g/L In(III) in 7 M HCl, with the results derived from this investigation showed in
Table 3.
It can be clearly seen that a ten-fold increase of the extractant concentration in the organic phase increased the indium(III) distribution coefficient value nearly 150 times.
Accordingly with the literature [
30], at this HCl concentration in the aqueous solution, indium(III) is present in this solution as InCl
4− species, thus, the extraction of indium(III) can be described by using the following formula:
which is in accordance with the previous assumption that non-polynuclear metal complexes are formed in the organic phase.
Considering this equilibrium, experimental data were treated numerically by a program which minimizes the U function, as described in Equation (1). The results indicated the existence in the organic phase of the ionic liquid In(III) species showed in Equation (3), with log K = 3.74 (U = 0.7889, σ(log K = 0.482)). Thus, the extraction of indium(III) from this 7 M HCl medium matched an anionic exchange reaction.
Indium(III) stripping from metal-loaded organic phases was also investigated. Considering that at low chloride concentrations, indium(III) exists in aqueous solutions as In
3+ (
Figure 2), it is logical to use low acidic solutions as a strippant for this system.
In this study, a 5% v/v A327H
+Cl
− in Solvesso 100 loaded with 0.074 g/L indium(III) and 0.1 M HCl as a strip solution were used. The results of this study were summarized in
Table 4; it can be seen that the percentage of indium(III) stripped, calculated as:
decreased as the Organic/Strip phases relationship increased. In the above equation, [In]
st and [In]
org were the indium concentrations in the equilibrated strip solution and in the organic phase feeding the stripping stage, respectively. In these series of experiments, equilibrium was reached within 5 min of contact between both phases.
Accordingly with these results, the stripping reaction can be written as:
and:
2.3. Indium(III) Transport Results
The system In(III)/A327H+Cl− was also investigated in a supported liquid membrane; in this operation, the transport of indium(III) across the liquid membrane also depends on kinetic factors.
The influence of the stirring speed applied to the feed phase on In(III) transport was first investigated, with the results derived from these tests summarized in
Table 5.
The permeability coefficient increased from 1.0–1.4 × 10−1 cm/min in the range of stirring speeds from 600 to 850 min−1, and beyond that, no appreciable increase in indium(III) transport was observed. These results indicated that the thickness of the feed phase boundary layer decreased continuously with the increase of the stirring speed, and that the boundary layer reached the minimum in the 850–1000 min−1 range. A slight decrease in permeability at 1200 min−1 could be attributed to an increment in the turbulence caused by stirring resulting in displacement of the carrier phase from the support pores. A stirring speed of 865 min−1 in the feed phase was maintained throughout all the experimentation.
Using the same experimental conditions as in
Table 5, the variation of the stirring speed (500–750 min
−1) in the receiving solution showed no changes in the permeability coefficient. Thus, a stirring speed of 500 min
−1 in the receiving solution was used in all the experiments. Furthermore, the variation in the receiving solution composition in the indium(III) transport was investigated. In this case, all the experimental parameters were maintained constant as in
Table 5, but the receiving phase was of 0.01 g/L In(III) and 0.1 M HCl. The results derived from this experiment showed that the presence of indium(III) in the initial receiving phase practically did not change indium permeability: 1.5 × 10
−1 cm/min for the present receiving solution against 1.4 × 10
−1 cm/min in the case of the receiving solution containing only 0.1 M HCl. However, the importance of this experiment was that it demonstrated that indium(III) is transported against its concentration gradient for the first 15 min, driven by the difference in acidity (or chloride concentration) between the feed and the receiving phases (
Table 6).
Moreover, if the chloride concentration in the feed phase, from the HCl medium, was substituted by LiCl, the transport experiment would allowed concluding that this substitution resulted in a decrease in indium transport (P = 5.9 × 10−2 cm/min). Thus, the difference in acidity between the feed and the receiving phases was the main driving force for this system.
The variation (0.01–0.2 g/L) in indium(III) concentration in the feed solution in the metal transport was also investigated, with the results obtained from these tests shown in
Table 7.
In this
Table 7, the initial metal flux J was calculated as:
where [In]
0 represented the initial indium concentration in the feed phase. The percentage of indium recovered in the receiving solution (%R) was calculated by the next relationship:
where [In]
f,t was the metal concentration in the feed solution at a specific timepoint and [In]
r,t was the indium concentration in the receiving phase at the same timepoint.
At these initial indium(III) concentrations in the feed phase, the initial flux was a function of the initial metal concentration in the phase. Therefore, the transport process was controlled by diffusion of indium species. With respect to the recovery of the metal in the receiving phase, acceptable values were obtained in all the cases, though these values could be improved by the use of the smart liquid membrane technology such as pseudo-emulsion membrane strip dispersion [
32].
The effect of the carrier concentration on indium(III) transport was also investigated.
Figure 3 showed the permeability values for the transport of indium(III) across a supported liquid membrane containing different solutions of the ionic liquid in Solvesso 100. The results showed that the permeability increased with the A327H
+Cl
− concentration until the maximum value of P was obtained at the 10% v/v carrier concentration. This limiting permeability value, P
lim, can be explained by assuming that diffusion across the membrane was negligible compared with that of aqueous diffusion, with the transport process controlled by the diffusion of metal species in the stagnant layer of the feed phase. In that case,
where D
aq was the average aqueous diffusion coefficient (6 × 10
−4 cm
2/min) and d
aq was the thickness of the feed phase diffusion layer, thus, P
lim = 1.4 × 10
−1 cm/min, the value of d
aq was 4 × 10
−3 cm; this value represented the minimum thickness of the diffusion layer. The decrease in the permeability value at higher carrier concentrations was probably due to an increase in viscosity of the organic solution, which caused an increase in membrane resistance to indium(III) transport.
At the first approach, the mechanism of mass transfer in flat sheet supported liquid membranes is understodd as the diffusion of the indium species across the membrane pores from the feed phase to the receiving phase with no dispersion at all. Evaluation of mass transfer coefficients is important, because they influence the scaling-up of the technology to the most efficient membrane operation using a hollow fibers module, which gives an important increment in the contact area between the feed-organic phases and the organic–receiving phases. According to Equation (3), the equilibrium constant for the present system can be written as:
Following the same approach as described elsewhere [
33], the next expression can be derived:
where Δ
f and Δ
m are the transport resistances related to diffusion in the feed and membrane phases, respectively. In the above equation, P
In is the indium(III) permeation coefficient, moreover, Equation (11) combines the diffusional and equilibrium parameters involved in the transport of indium(III) from an HCl solution through a membrane containing the carrier. From the above,
Accordingly, a plot of 1/PIn versus 1/B for various carrier concentrations in the membrane phase and 7 M chloride concentration in the feed phase resulted in a straight line (r2 = 0.9115) with an intercept and a slope that can be used to estimate the transport resistance in the feed phase (Δf = 7 min/cm) and in the membrane phase (Δm = 170 min/cm), respectively.
Thus, the mass transfer coefficient in the feed phase is Δ
f−1 = 1.4 × 10
−1 cm/min, whereas the diffusion coefficient in the organic solution,
is calculated as 7.4 × 10
−5 cm
2/min, considering Δ
m is 170 min/cm and the thickness of the membrane support, d
m, is 12.5 × 10
−3 cm.
The diffusion coefficient of the indium(III) ionic liquid species in the bulk organic phase is estimated using the following relationship [
34]:
where ε is the support porosity (75%) and τ is the membrane tortuosity (1.67), thus, D
b,org is estimated at 2.8 × 10
−4 cm
2/min. The diffusion coefficient in the bulk organic phase presented a greater value than D
org; this result can be due to the diffusional resistance caused by support thickness located between the feed and receiving solutions.
If the carrier concentration in the membrane support is constant, the apparent diffusion coefficient for indium(III) can be calculated as:
Using a 10% v/v ionic liquid concentration (2.1 × 10−1 M) and with dm = 12.5 × 10−3 cm, this apparent diffusion coefficient has the value of 7 × 10−7 cm2/min.