Trimeric Small Interfering RNAs and Their Cholesterol-Containing Conjugates Exhibit Improved Accumulation in Tumors, but Dramatically Reduced Silencing Activity
Abstract
:1. Introduction
2. Results and Discussion
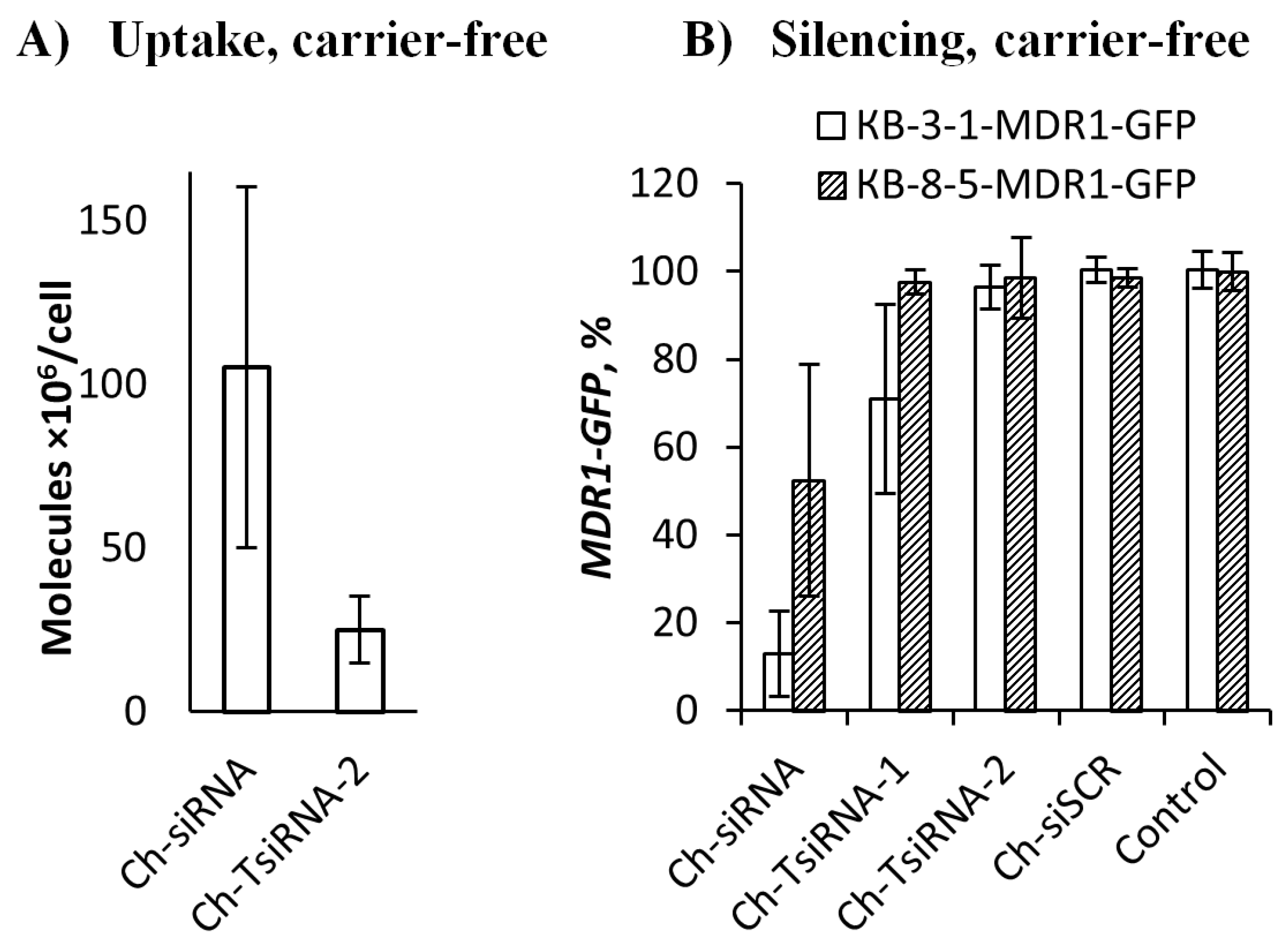
2.1. In Vitro
2.2. In Vivo
3. Materials and Methods
3.1. Synthesis of Monomeric and Trimeric siRNAs, Their Cholesterol-Containing Analogues and Duplex Annealing
3.2. Cell Culture
3.3. Gene Silencing Assay
3.4. Stem-loop RT-PCR Cellular Accumulation Assay
3.5. Mice
3.6. Stem-Loop RT-PCR Analysis of siRNA and TsiRNA Concentration in Mice Blood and Internal Organs After Intravenous Administration
3.7. Analysis of siRNA and TsiRNA Accumulation and Silencing Activity in KB-8-5 Xenograft Tumors in SCID Mice After Intravenous Administration
3.8. Statistical Analyses
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Adams, D.; Gonzalez-Duarte, A.; O’Riordan, W.D.; Yang, C.C.; Ueda, M.; Kristen, A.V.; Tournev, I.; Schmidt, H.H.; Coelho, T.; Berk, J.L.; et al. Patisiran, an RNAi Therapeutic, for Hereditary Transthyretin Amyloidosis. N. Engl. J. Med. 2018, 379, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Tian, G.; Pan, R.; Zhang, B.; Qu, M.; Lian, B.; Jiang, H.; Gao, Z.; Wu, J. Liver-Targeted Combination Therapy Basing on Glycyrrhizic Acid-Modified DSPE-PEG-PEI Nanoparticles for Co-delivery of Doxorubicin and Bcl-2 siRNA. Front. Pharmacol. 2019, 10, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Osborn, M.F.; Coles, A.H.; Biscans, A.; Haraszti, R.A.; Roux, L.; Davis, S.; Ly, S.; Echeverria, D.; Hassler, M.R.; Godinho, B.; et al. Hydrophobicity drives the systemic distribution of lipid-conjugated siRNAs via lipid transport pathways. Nucleic Acids Res. 2018, 47, 1070–1081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nair, J.K.; Willoughby, J.L.; Chan, A.; Charisse, K.; Alam, M.R.; Wang, Q.; Hoekstra, M.; Kandasamy, P.; Kel’in, A.V.; Milstein, S.; et al. Multivalent N-acetylgalactosamine-conjugated siRNA localizes in hepatocytes and elicits robust RNAi-mediated gene silencing. J. Am. Chem. Soc. 2014, 136, 16958–16961. [Google Scholar] [CrossRef] [Green Version]
- Chernikov, I.V.; Vlassov, V.V.; Chernolovskaya, E.L. Current Development of siRNA Bioconjugates: From Research to the Clinic. Front. Pharmacol. 2019, 10, 444. [Google Scholar] [CrossRef] [Green Version]
- Lonn, P.; Kacsinta, A.D.; Cui, X.S.; Hamil, A.S.; Kaulich, M.; Gogoi, K.; Dowdy, S.F. Enhancing Endosomal Escape for Intracellular Delivery of Macromolecular Biologic Therapeutics. Sci. Rep. 2016, 6, 1–9. [Google Scholar] [CrossRef]
- Robbins, M.; Judge, A.; Liang, L.; McClintock, K.; Yaworski, E.; MacLachlan, I. 2′-O-methyl-modified RNAs act as TLR7 antagonists. Mol. Ther. J. Am. Soc. Gene Ther. 2007, 15, 1663–1669. [Google Scholar] [CrossRef]
- Gvozdeva, O.V.; Dovydenko, I.S.; Venyaminova, A.G.; Zenkova, M.A.; Vlassov, V.V.; Chernolovskaya, E.L. 42- and 63-bp anti-MDR1-siRNAs bearing 2′-OMe modifications in nuclease-sensitive sites induce specific and potent gene silencing. FEBS Lett. 2014, 588, 1037–1043. [Google Scholar] [CrossRef] [Green Version]
- Gvozdeva, O.V.; Gladkih, D.V.; Chernikov, I.V.; Meschaninova, M.I.; Venyaminova, A.G.; Zenkova, M.A.; Vlassov, V.V.; Chernolovskaya, E.L. Nuclease-resistant 63-bp trimeric siRNAs simultaneously silence three different genes in tumor cells. FEBS Lett. 2018, 592, 122–129. [Google Scholar] [CrossRef] [Green Version]
- Petrova, N.S.; Chernikov, I.V.; Meschaninova, M.I.; Dovydenko, I.S.; Venyaminova, A.G.; Zenkova, M.A.; Vlassov, V.V.; Chernolovskaya, E.L. Carrier-free cellular uptake and the gene-silencing activity of the lipophilic siRNAs is strongly affected by the length of the linker between siRNA and lipophilic group. Nucleic Acids Res. 2012, 40, 2330–2344. [Google Scholar] [CrossRef] [Green Version]
- Volkov, A.A.; Kruglova, N.S.; Meschaninova, M.I.; Venyaminova, A.G.; Zenkova, M.A.; Vlassov, V.V.; Chernolovskaya, E.L. Selective protection of nuclease-sensitive sites in siRNA prolongs silencing effect. Oligonucleotides 2009, 19, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Chernikov, I.V.; Gladkikh, D.V.; Meschaninova, M.I.; Ven’yaminova, A.G.; Zenkova, M.A.; Vlassov, V.V.; Chernolovskaya, E.L. Cholesterol-Containing Nuclease-Resistant siRNA Accumulates in Tumors in a Carrier-free Mode and Silences MDR1 Gene. Mol. Ther. Nucleic Acids 2017, 6, 209–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chernikov, I.V.; Gladkikh, D.V.; Meschaninova, M.I.; Karelina, U.A.; Ven’yaminova, A.G.; Zenkova, M.A.; Vlassov, V.V.; Chernolovskaya, E.L. Fluorophore Labeling Affects the Cellular Accumulation and Gene Silencing Activity of Cholesterol-Modified Small Interfering RNAs In Vitro. Nucleic Acid Ther. 2019, 29, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Snead, N.M.; Wu, X.; Li, A.; Cui, Q.; Sakurai, K.; Burnett, J.C.; Rossi, J.J. Molecular basis for improved gene silencing by Dicer substrate interfering RNA compared with other siRNA variants. Nucleic Acids Res. 2013, 41, 6209–6221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Golombek, S.K.; May, J.N.; Theek, B.; Appold, L.; Drude, N.; Kiessling, F.; Lammers, T. Tumor targeting via EPR: Strategies to enhance patient responses. Adv. Drug Deliv. Rev. 2018, 130, 17–38. [Google Scholar] [CrossRef] [PubMed]
- Kutova, O.M.; Guryev, E.L.; Sokolova, E.A.; Alzeibak, R.; Balalaeva, I.V. Targeted Delivery to Tumors: Multidirectional Strategies to Improve Treatment Efficiency. Cancers 2019, 11, 68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolfrum, C.; Shi, S.; Jayaprakash, K.N.; Jayaraman, M.; Wang, G.; Pandey, R.K.; Rajeev, K.G.; Nakayama, T.; Charrise, K.; Ndungo, E.M.; et al. Mechanisms and optimization of in vivo delivery of lipophilic siRNAs. Nat. Biotechnol. 2007, 25, 1149–1157. [Google Scholar] [CrossRef]
- Huggins, I.J.; Medina, C.A.; Springer, A.D.; van den Berg, A.; Jadhav, S.; Cui, X.; Dowdy, S.F. Site Selective Antibody-Oligonucleotide Conjugation via Microbial Transglutaminase. Molecules 2019, 24, 3287. [Google Scholar] [CrossRef] [Green Version]
- Ibtehaj, N.; Huda, R. High-dose BAFF receptor specific mAb-siRNA conjugate generates Fas-expressing B cells in lymph nodes and high-affinity serum autoantibody in a myasthenia mouse model. Clin. Immunol. 2017, 176, 122–130. [Google Scholar] [CrossRef]
- Esposito, C.L.; Nuzzo, S.; Catuogno, S.; Romano, S.; de Nigris, F.; de Franciscis, V. STAT3 Gene Silencing by Aptamer-siRNA Chimera as Selective Therapeutic for Glioblastoma. Mol. Ther. Nucleic Acids 2018, 10, 398–411. [Google Scholar] [CrossRef] [Green Version]
- Nakamoto, K.; Akao, Y.; Furuichi, Y.; Ueno, Y. Enhanced Intercellular Delivery of cRGD-siRNA Conjugates by an Additional Oligospermine Modification. ACS Omega 2018, 3, 8226–8232. [Google Scholar] [CrossRef] [Green Version]
- Zheng, M.; Librizzi, D.; Kilic, A.; Liu, Y.; Renz, H.; Merkel, O.M.; Kissel, T. Enhancing in vivo circulation and siRNA delivery with biodegradable polyethylenimine-graft-polycaprolactone-block-poly(ethylene glycol) copolymers. Biomaterials 2012, 33, 6551–6558. [Google Scholar] [CrossRef] [PubMed]
- Biscans, A.; Coles, A.; Echeverria, D.; Khvorova, A. The valency of fatty acid conjugates impacts siRNA pharmacokinetics, distribution, and efficacy in vivo. J. Control. Release 2019, 302, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.M.; Dahlman, J.E.; Neuman, K.K.; Prata, C.A.H.; Krampert, M.C.; Hadwiger, P.M.; Vornlocher, H.P. Ligand Conjugated Multimeric siRNAs Enable Enhanced Uptake and Multiplexed Gene Silencing. Nucleic Acid Ther. 2019, 29, 231–244. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brinkmann, U. Functional polymorphisms of the human multidrug resistance (MDR1) gene: Correlation with P glycoprotein expression and activity in vivo. Novartis Found. Symp. 2002, 243, 207–210. [Google Scholar] [PubMed]
- Landesman, Y.; Svrzikapa, N.; Cognetta, A., 3rd; Zhang, X.; Bettencourt, B.R.; Kuchimanchi, S.; Dufault, K.; Shaikh, S.; Gioia, M.; Akinc, A.; et al. In vivo quantification of formulated and chemically modified small interfering RNA by heating-in-Triton quantitative reverse transcription polymerase chain reaction (HIT qRT-PCR). Silence 2010, 1, 16. [Google Scholar] [CrossRef] [Green Version]
- Czimmerer, Z.; Hulvely, J.; Simandi, Z.; Varallyay, E.; Havelda, Z.; Szabo, E.; Varga, A.; Dezso, B.; Balogh, M.; Horvath, A.; et al. A versatile method to design stem-loop primer-based quantitative PCR assays for detecting small regulatory RNA molecules. PLoS ONE 2013, 8, e55168. [Google Scholar] [CrossRef] [Green Version]
Sample Availability: Samples of the compounds Ch-siRNA, Ch-siSCRm are available from the authors. |

| Designation | Sequence 1 | IC50, nM |
|---|---|---|
| siRNA | 5′-GGCUUGACAAGUUGUAUAUGG-3′ 3′-AACCGAACUGUUCAACAUAUA-5′ | 3.8 ± 3.5 |
| Ch-siRNA | Ch-5′-GGCUUGACAAGUUGUAUAUGG-3′ 3′-AACCGAACUGUUCAACAUAUA-5′ | 29 ± 3 |
| Ch-siSCRm | Ch-5′-CAAGUCUCGUAUGUAGUGGUU-3′ 3′-UUGUUCAGAGCAUACAUCACC-5′ | |
| TsiRNA-1 | 5′-GGCUUGACAAGUUGUAUAUGGGGCUUGACAAGUUGUAUAUGGGGCUUGACAAGUUGUAUAUGG-3′ 3′-AACCGAACUGUUCAACAUAUAAACCGAACUGUUCAACAUAUAAACCGAACUGUUCAACAUAUA-5′ | 0.65 ± 0.14 |
| Ch-TsiRNA-1 | Ch-5′-GGCUUGACAAGUUGUAUAUGGGGCUUGACAAGUUGUAUAUGGGGCUUGACAAGUUGUAUAUGG-3′ 3′-AACCGAACUGUUCAACAUAUAAACCGAACUGUUCAACAUAUAAACCGAACUGUUCAACAUAUA-5′ | 16 ± 10 |
| TsiRNA-2 | 5′-CAGAGGCCGCUGUUCGUUUGAGCGCGAGGUCGGGAUGGAUUUGGCUUGACAAGUUGUAUAUGG-3′ 3′-UCGUCUCCGGCGACAAGCAAACUCGCGCUCCAGCCCUACCUAAACCGAACUGUUCAACAUAUA | 13 ± 7 |
| Ch-TsiRNA-2 | Ch-5′-CAGAGGCCGCUGUUCGUUUGAGCGCGAGGUCGGGAUGGAUUUGGCUUGACAAGUUGUAUAUGG-3′ 3′-UCGUCUCCGGCGACAAGCAAACUCGCGCUCCAGCCCUACCUAAACCGAACUGUUCAACAUAUA–5′ | 87 ± 13 |
| Designation 1 | Strand 1 | Mass Calculated | Mass Found |
|---|---|---|---|
| siRNA | Sense | 6838.2 | 6838.1 |
| Antisense | 6732.2 | 6732.4 | |
| Ch-siRNA | Ch-Sense | 7428.8 | 7428.7 |
| Antisense | 6732.2 | 6732.4 | |
| Ch-siSCRm | Ch-Sense | 7351.6 | 7352.2 |
| Antisense | 6687.1 | 6688 | |
| TsiRNA-1 | Sense | 20,682.5 | 20,684.5 |
| Antisense | 20,320.6 | 20321 | |
| Ch-TsiRNA-1 | Ch-Sense | 21,229.7 | 21,230.7 |
| Antisense | 20,320.6 | 20321 | |
| TsiRNA-2 | Sense | 20,547.1 | 20,545.1 |
| Antisense | 20135 | 20,134.3 | |
| Ch-TsiRNA-2 | Ch-Sense | 21,138.7 | 21137 |
| Antisense | 20,135 | 20,134.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chernikov, I.V.; Gladkikh, D.V.; Karelina, U.A.; Meschaninova, M.I.; Ven’yaminova, A.G.; Vlassov, V.V.; Chernolovskaya, E.L. Trimeric Small Interfering RNAs and Their Cholesterol-Containing Conjugates Exhibit Improved Accumulation in Tumors, but Dramatically Reduced Silencing Activity. Molecules 2020, 25, 1877. https://doi.org/10.3390/molecules25081877
Chernikov IV, Gladkikh DV, Karelina UA, Meschaninova MI, Ven’yaminova AG, Vlassov VV, Chernolovskaya EL. Trimeric Small Interfering RNAs and Their Cholesterol-Containing Conjugates Exhibit Improved Accumulation in Tumors, but Dramatically Reduced Silencing Activity. Molecules. 2020; 25(8):1877. https://doi.org/10.3390/molecules25081877
Chicago/Turabian StyleChernikov, Ivan V., Daniil V. Gladkikh, Ulyana A. Karelina, Mariya I. Meschaninova, Alya G. Ven’yaminova, Valentin V. Vlassov, and Elena L. Chernolovskaya. 2020. "Trimeric Small Interfering RNAs and Their Cholesterol-Containing Conjugates Exhibit Improved Accumulation in Tumors, but Dramatically Reduced Silencing Activity" Molecules 25, no. 8: 1877. https://doi.org/10.3390/molecules25081877






