In Vitro Anti-Tubulin Activity on MCF10A Cell Line and In Silico Rigid/Semiflexible-Residues Docking, of Two Lignans from Bursera Fagaroides var. Fagaroides
Abstract
:1. Introduction
2. Results
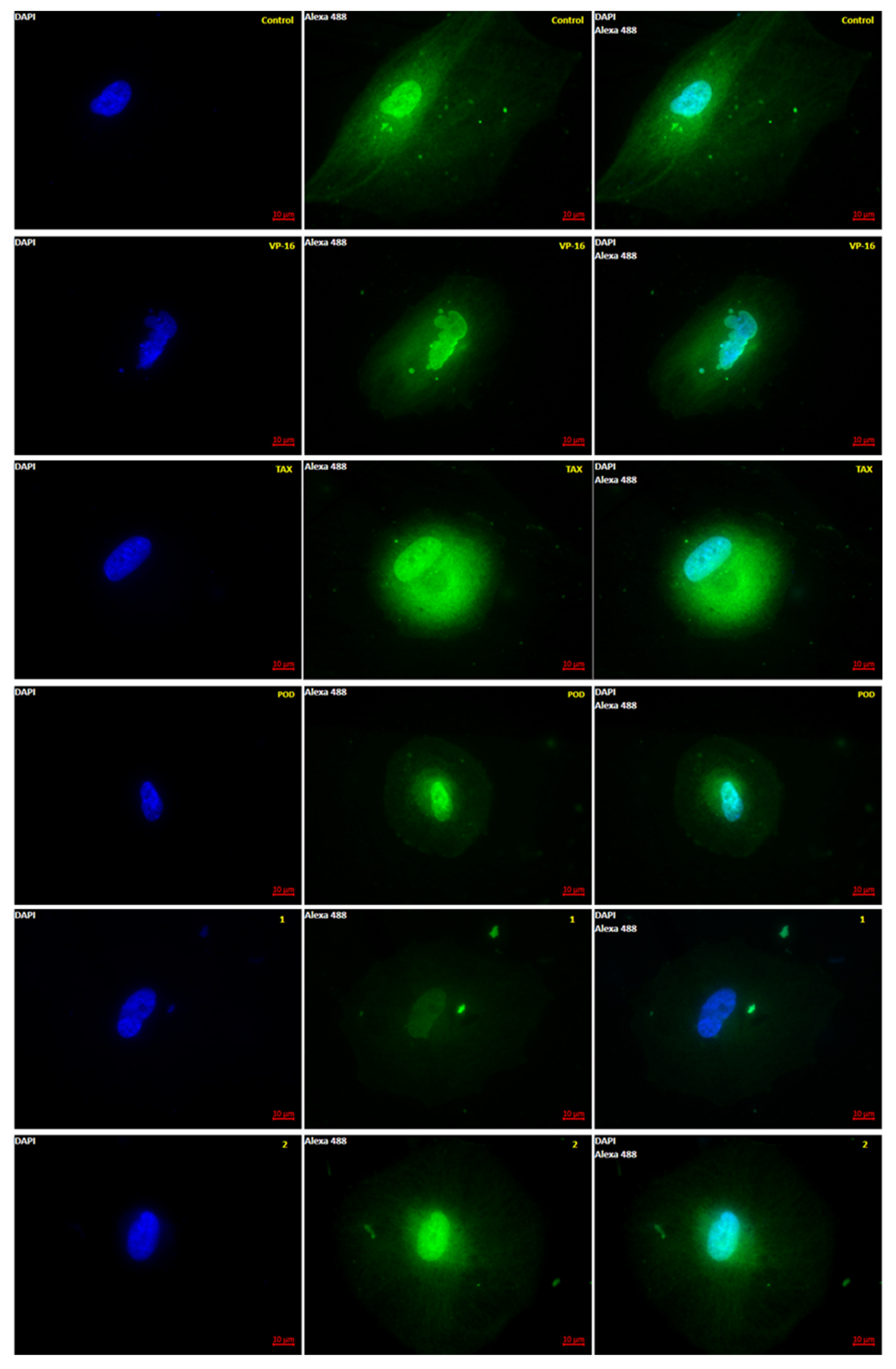
2.1. Microscopy Anti-Tubulin Test
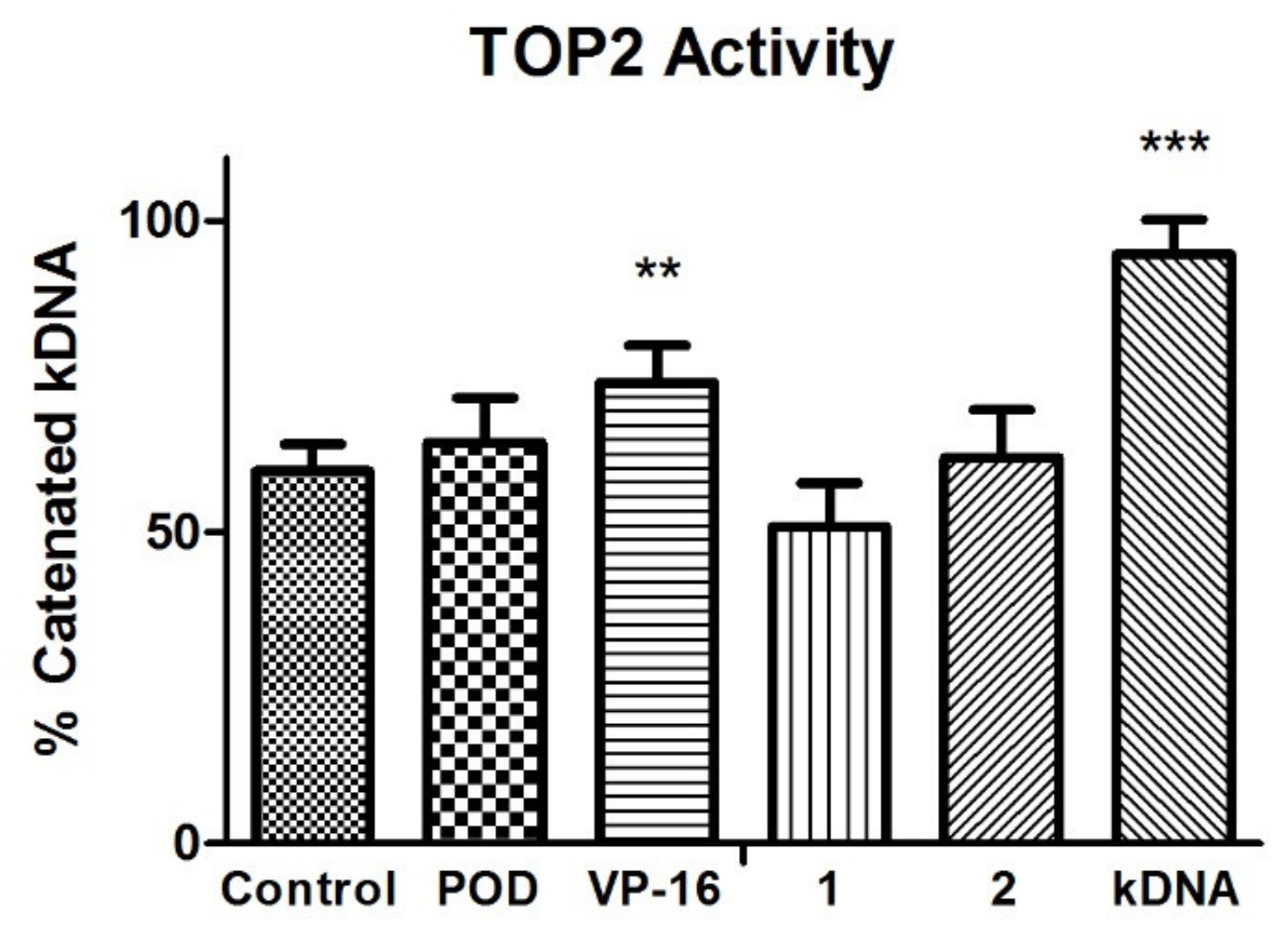
2.2. Topoisomerase II Inhibition Assay
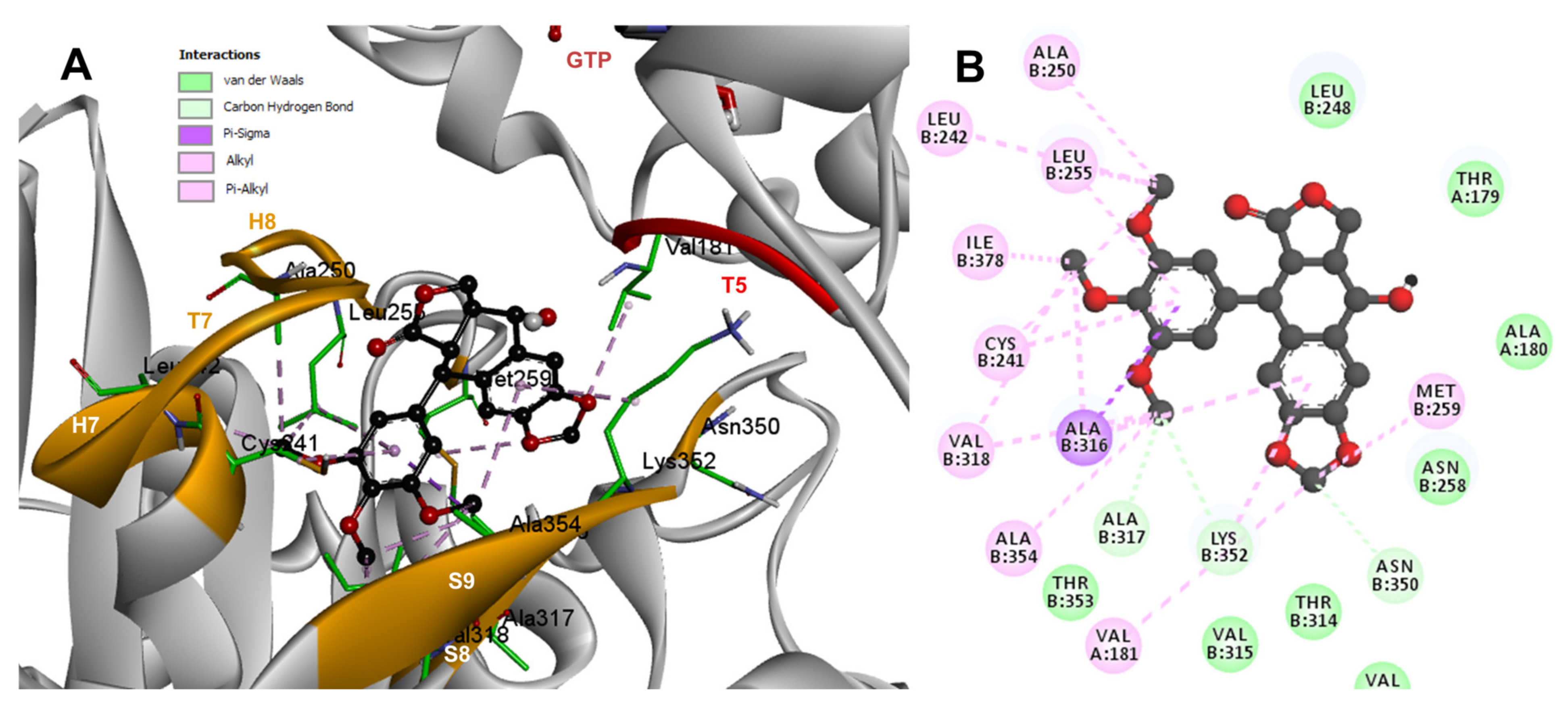
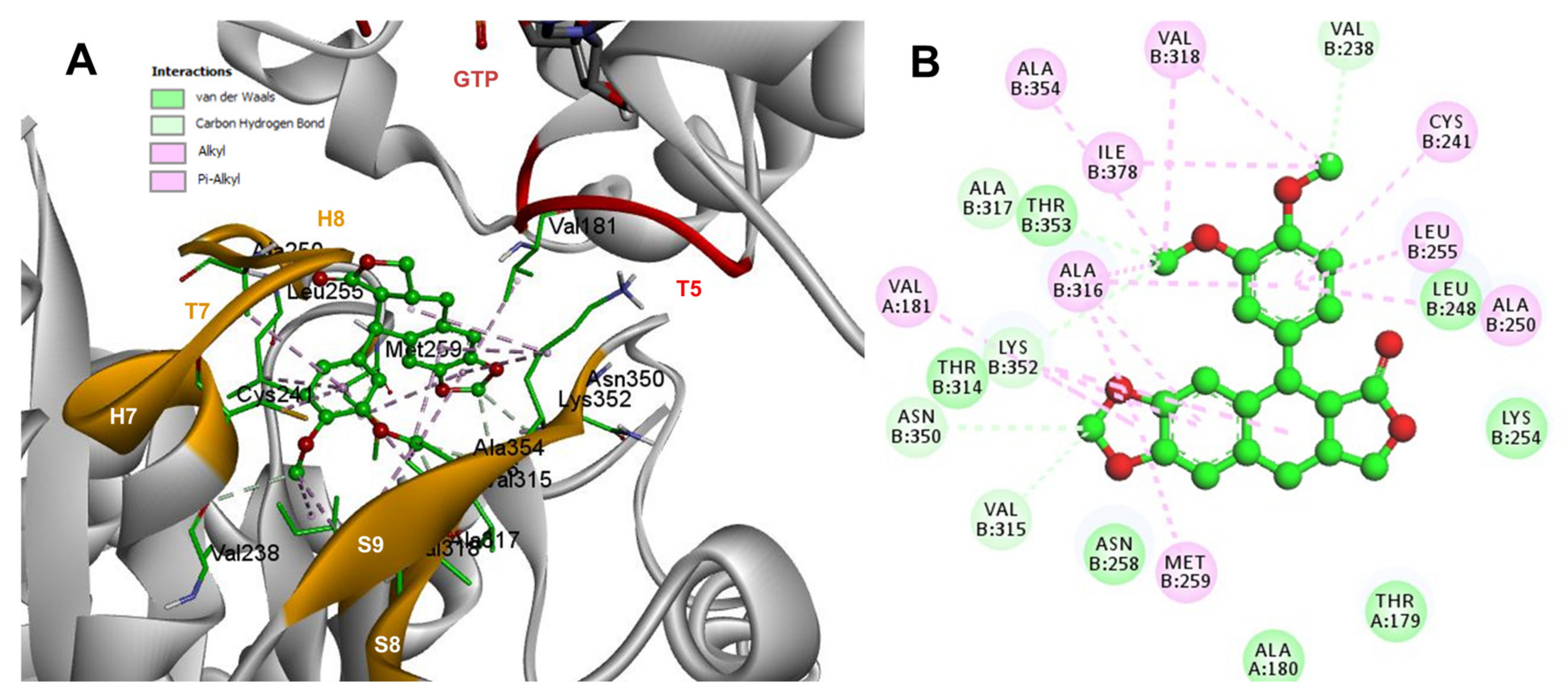
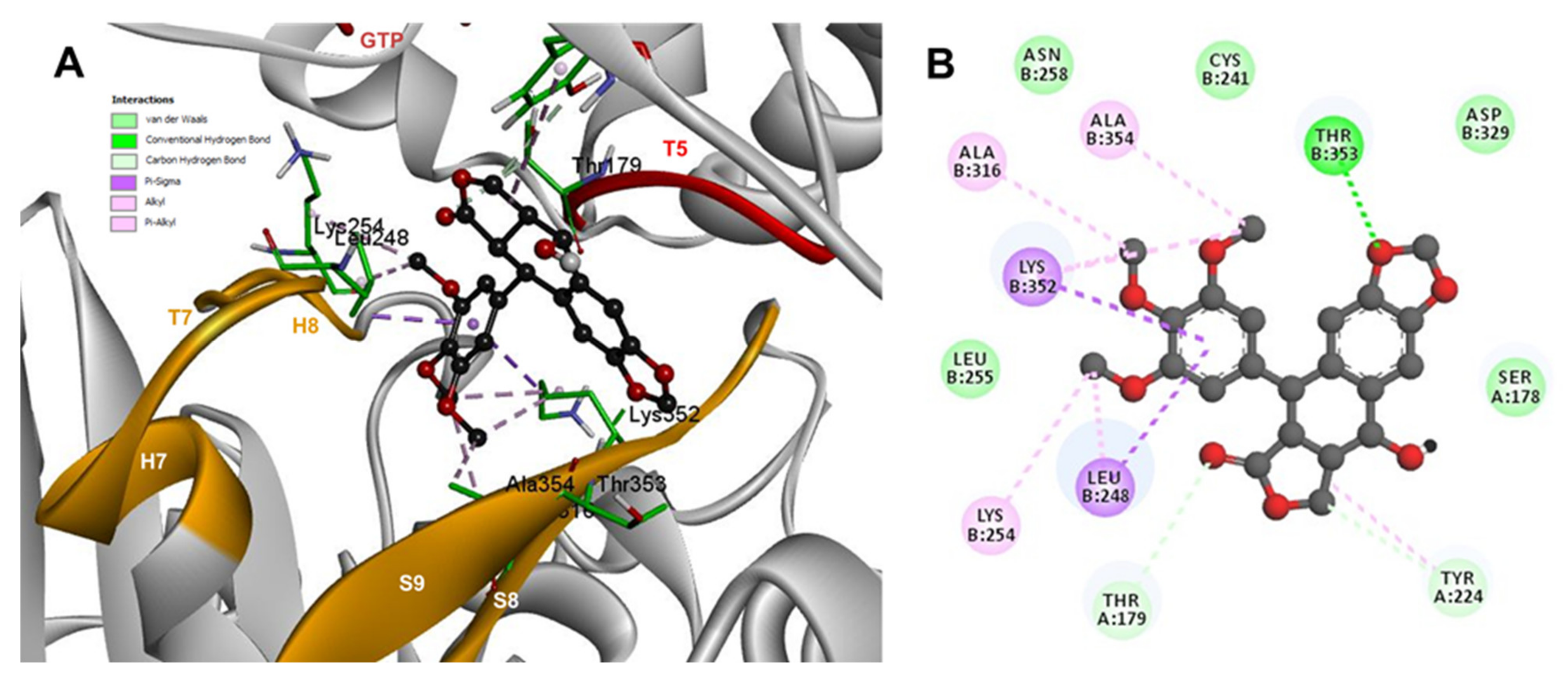
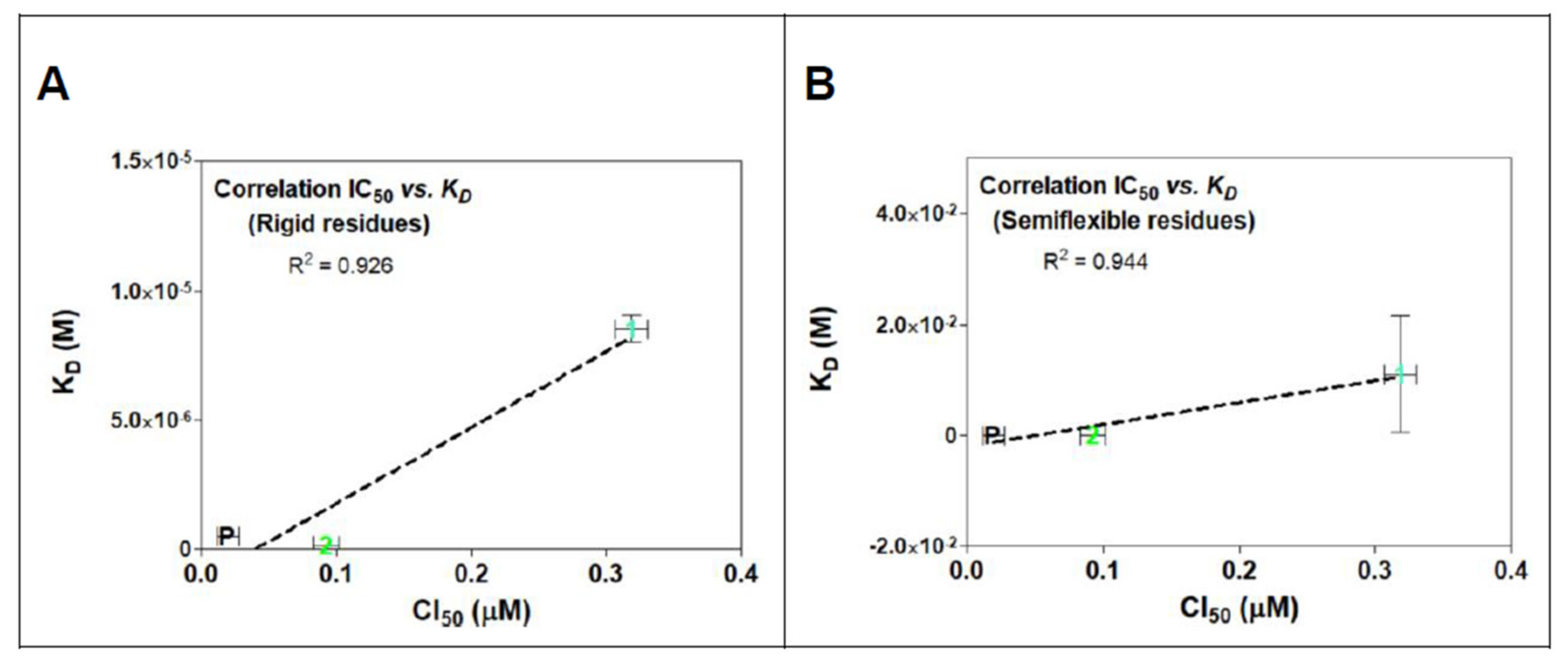
2.3. Rigid- and Semiflexible-Residues Molecular Docking
3. Discussion
4. Materials and Methods
4.1. Microscopy Anti-Tubulin Test
4.2. Microtubules Semi-Quantitation
4.3. Topoisomerase II Inhibition Assay
4.4. Flexible- and Rigid-Residues Molecular Docking
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Teponno, R.B.; Kusari, S.; Spiteller, M. Recent advances in research on lignans and neolignans. Nat. Prod. Rep. 2016, 33, 1044–1092. [Google Scholar] [CrossRef] [Green Version]
- Barker, D. Lignans. Molecules 2019, 24, 1424. [Google Scholar] [CrossRef] [Green Version]
- Durazzo, A.; Lucarini, M.; Camilli, E.; Marconi, S.; Gabrielli, P.; Lisciani, S.; Gambelli, L.; Aguzzi, A.; Novellino, E.; Santini, A.; et al. Dietary Lignans: Definition, Description and Research Trends in Databases Development. Molecules 2018, 23, 3251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamal, A.; Hussaini, S.M.; Malik, M.S. Recent developments towards podophyllotoxin congeners as potential apoptosis inducers. Anticancer Agents Med. Chem. 2015, 15, 565–574. [Google Scholar] [CrossRef]
- Tseng, C.J.; Wang, Y.J.; Liang, Y.C.; Jeng, J.H.; Lee, W.S.; Lin, J.K.; Chen, C.H.; Liu, I.C.; Ho, Y.S. Microtubule damaging agents induce apoptosis in H.L. 60 cells and G2/M cell cycle arrest in H.T. 29 cells. Toxicology 2002, 175, 123–142. [Google Scholar] [CrossRef]
- Canel, C.; Moraes, R.; Dayan, F.; Ferreira, D. Molecules of interest: Podophyllotoxin. Phytochemistry 2000, 54, 115–120. [Google Scholar] [CrossRef]
- Xu, H.; Lv, M.; Tian, X. A review on hemisynthesis, biosynthesis, biological activities, mode of action, and structure-activity relationship of podophyllotoxins. Curr. Med. Chem. 2009, 16, 327–349. [Google Scholar] [CrossRef]
- Longstaff, E.; VonKrogh, G. Condyloma eradication: Self-therapy with 0.15–0.5% podophyllotoxin versus 20–25% podophyllin preparations-An integrated safety assessment. Regul. Toxicol. Pharmacol. 2001, 33, 117–137. [Google Scholar] [CrossRef]
- Peng, X.; Li, M. Medical Treatment of Condyloma Acuminatum, a Review on the Chemistry of Therapeutic Drugs. J. Appl. Virol. 2019, 8, 1–8. [Google Scholar] [CrossRef]
- Dewick, P.M. Medicinal Natural Products: A Biosynthetic Approach, 3rd ed.; John Wiley & Sons: Chichester, UK, 2009; pp. 151–156. [Google Scholar]
- Guerram, M.; Jiang, Z.-Z.; Zhang, L.-Y. Podophyllotoxin, a medicinal agent of plant origin: Past, present and future. Chin. J. Nat. Med. 2012, 10, 161–169. [Google Scholar] [CrossRef]
- Ravelli, R.; Gigant, B.; Curmi, A.; Jourdain, I.; Lachkar, S.; Sobel, A.; Knossow, M. Insight into tubulin regulation from a complex with colchicines and a stathmin-like domain. Nature 2004, 428, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Montecucco, A.; Biamonti, G. Cellular response to etoposide treatment. Cancer Lett. 2007, 252, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.C.; Li, T.K.; Farh, L.; Lin, L.Y.; Lin, T.S.; Yu, Y.J.; Yen, T.J.; Chiang, C.W.; Chan, N.L. Structural basis of type II topoisomerase inhibition by the anticancer drug etoposide. Science 2011, 333, 459–462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thurston, L.S.; Irie, H.; Tani, S.; Han, F.S.; Liu, Z.C.; Cheng, Y.C.; Lee, K.H. Antitumor agents. 78. Inhibition of human DNA topoisomerase II by podophyllotoxin and alpha-peltatin analogs. J. Med. Chem. 1986, 29, 1547–1550. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, J.T.; Lipp, H.P. Camptothecin and podophyllotoxin derivatives: Inhibitors of topoisomerase I and II - mechanisms of action, pharmacokinetics and toxicity profile. Drug Saf. 2006, 29, 209–230. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.Y.; Hwang, B.D.; Haruna, M.; Imakura, Y.; Lee, K.H.; Cheng, Y.C. Podophyllotoxin analogs: Effects on DNA topoisomerase II, tubulin polymerization, human tumor KB cells, and their VP-16-resistant variants. Mol. Pharmacol. 1989, 36, 78–82. [Google Scholar]
- Zhao, W.; Bai, J.K.; Li, H.M.; Chen, T.; Tang, Y.J. Tubulin structure-based drug design for the development of novel 4β-sulfur-substituted podophyllum tubulin inhibitors with anti-tumor activity. Sci. Rep. 2015, 5, 10172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antúnez, M.M.; León, A.; Rojas-Sepúlveda, M.; Marquina, S.; Mendieta-Serrano, M.A.; Salas-Vidal, E.; Villarreal, M.L.; Alvarez, L. Aryldihydronaphthalene-type lignans from Bursera fagaroides var. fagaroides and their antimitotic mechanism of action. RSC Adv. 2016, 6, 4950–4959. [Google Scholar] [CrossRef]
- Desbène, S.; Giorgi-Renault, S. Drugs that inhibit tubulin polymerization: The particular case of podophyllotoxin and analogues. Curr. Med. Chem. Anticancer Agents 2002, 2, 71–90. [Google Scholar] [CrossRef]
- Rojas-Sepúlveda, A.M.; Mendieta-Serrano, M.; Antúnez-Mojica, M.Y.; Salas-Vidal, E.; Marquina, S.; Villarreal, M.L.; Puebla, A.M.; Delgado, J.; Alvarez, L. Cytotoxic podophyllotoxin type-lignans from the steam bark of Bursera fagaroides var. fagaroides. Molecules 2012, 17, 9506–9519. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.H.; Horwitz, S.B. Taxol®: The first microtubule stabilizing agent. Int. J. Mol. Sci. 2017, 18, 1733. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berman, H.M.; Westbrook, J.; Feng, Z.; Gilliland, G.; Bhat, T.N.; Weissig, H.; Shindyalov, I.N.; Bourne, P.E. The Protein Data Bank. Nucleic Acids Res. 2000, 28, 235–242. [Google Scholar] [CrossRef] [Green Version]
- Peña-Morán, O.A.; Villarreal, M.L.; Álvarez-Berber, L.; Meneses-Acosta, A.; Rodríguez-López, V. Cytotoxicity, Post-Treatment Recovery, and Selectivity Analysis of Naturally Occurring Podophyllotoxins from Bursera fagaroides var. fagaroides on Breast Cancer Cell Lines. Molecules 2016, 21, 1013. [Google Scholar] [CrossRef] [PubMed]
- Liebmann, J.E.; Cook, J.A.; Lipschultz, C.; Teague, D.; Fisher, J.; Mitchell, J.B. Cytotoxic studies of paclitaxel (Taxol) in human tumour cell lines. Br. J. Cancer 1993, 68, 1104–1109. [Google Scholar] [CrossRef] [Green Version]
- Steed, H.; Sawyer, M.B. Pharmacology, pharmacokinetics and pharmacogenomics of paclitaxel. Pharmacogenomics 2007, 8, 803–815. [Google Scholar] [CrossRef] [PubMed]
- Mollinedo, F.; Gajate, C. Microtubules, microtubule-interfering agents and apoptosis. Apoptosis 2003, 8, 413–450. [Google Scholar] [CrossRef] [Green Version]
- Kamal, A.; Ali-Hussaini, S.M.; Rahim, A.; Riyaz, S. Podophyllotoxin derivatives: A patent review (2012–2014). Exp. Opin. Ther. Pat. 2015, 25, 1025–1034. [Google Scholar] [CrossRef]
- Lv, M.; Xu, H. Recent advances in semisynthesis, biosynthesis, biological activities, mode of action, and structure-activity relationship of podophyllotoxins: An update (2008–2010). Mini Rev. Med. Chem. 2011, 11, 901–909. [Google Scholar] [CrossRef]
- Zhang, X.; Rakesh, K.P.; Shantharam, C.S.; Manukumar, H.M.; Asiri, A.M.; Marwani, H.M.; Qin, H.L. Podophyllotoxin derivatives as an excellent anticancer aspirant for future chemotherapy: A key current imminent needs. Bioorg Med. Chem. 2018, 26, 340–355. [Google Scholar] [CrossRef]
- Hevener, K.; Verstak, T.A.; Lutat, K.E.; Riggsbee, D.L.; Mooney, J.W. Recent developments in topoisomerase-targeted cancer chemotherapy. Acta Pharm. Sin. B 2018, 8, 844–861. [Google Scholar] [CrossRef]
- Zi, C.T.; Yang, L.; Xu, F.Q.; Dong, F.W.; Yang, D.; Li, Y.; Ding, Z.T.; Zhou, J.; Jiang, Z.H.; Hu, J.M. Synthesis and anticancer activity of dimeric podophyllotoxin derivatives. Drug Des. Devel. Ther. 2018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Negia, A.S.; Gautam, Y.; Alam, S.; Chanda, D.; Luqman, D.; Sarkar, J.; Khan, F.; Konwar, R. Natural antitubulin agents: Importance of 3,4,5-trimethoxyphenyl fragment. Bioorganic Med. Chem. 2015, 23, 373–389. [Google Scholar] [CrossRef] [PubMed]
- Carlson, H.A. Protein flexibility and drug design: How to hit a moving target. Curr. Opin. Chem. Biol. 2002, 6, 447–452. [Google Scholar] [CrossRef]







| Rigid Residues | Flexible Residues | |||
|---|---|---|---|---|
| Compound | ΔGb (Kcal/Mol) | KD (M) | ΔGb (Kcal/Mol) | KD (M) |
| POD | −8.6 ± 0.00 | 4.96 × 10−7 ± 0.00 | −8.6 ± 1.15 | 1.44 × 10−5 ± 5.05 × 10−5 |
| 1 | −6.9 ± 0.03 | 8.53 × 10−6 ± 5.08 × 10−7 | −3.6 ± 1.05 | 0.011 ± 0.03 |
| 2 | −9.4 ± 0.00 | 1.28 × 10−7 ± 0.00 | −10.8 ± 0.81 | 3.10 × 10−8 ± 5.97 × 10−8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-López, V.; Millán-Pacheco, C.; González-Christen, J.; Anaya-Ruíz, M.; Peña-Morán, O.A. In Vitro Anti-Tubulin Activity on MCF10A Cell Line and In Silico Rigid/Semiflexible-Residues Docking, of Two Lignans from Bursera Fagaroides var. Fagaroides. Molecules 2021, 26, 6155. https://doi.org/10.3390/molecules26206155
Rodríguez-López V, Millán-Pacheco C, González-Christen J, Anaya-Ruíz M, Peña-Morán OA. In Vitro Anti-Tubulin Activity on MCF10A Cell Line and In Silico Rigid/Semiflexible-Residues Docking, of Two Lignans from Bursera Fagaroides var. Fagaroides. Molecules. 2021; 26(20):6155. https://doi.org/10.3390/molecules26206155
Chicago/Turabian StyleRodríguez-López, Verónica, César Millán-Pacheco, Judith González-Christen, Maricruz Anaya-Ruíz, and Omar Aristeo Peña-Morán. 2021. "In Vitro Anti-Tubulin Activity on MCF10A Cell Line and In Silico Rigid/Semiflexible-Residues Docking, of Two Lignans from Bursera Fagaroides var. Fagaroides" Molecules 26, no. 20: 6155. https://doi.org/10.3390/molecules26206155







