In Vivo Biological Behavior of Polymer Scaffolds of Natural Origin in the Bone Repair Process
Abstract
:1. Introduction
2. Results
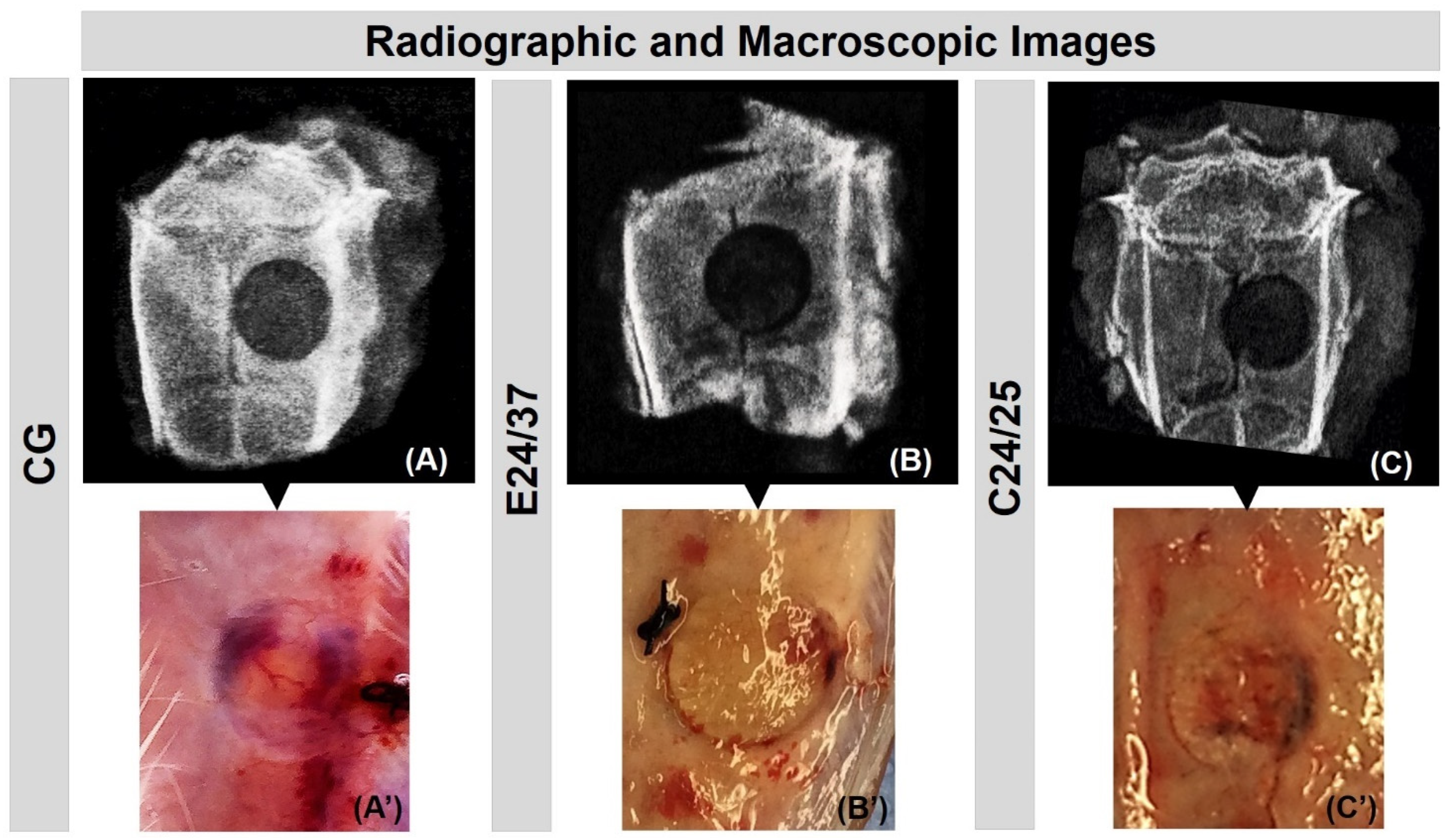
2.1. Radiological and Macroscopic Analysis
2.2. Micro CT Analysis: Three-Dimensional, Two-Dimensional Sections and Quantitative Evaluation
2.3. Confocal Laser Scanning Microscopy Analysis
2.4. Mineralization Analysis of New Bone Formed by the Von Kossa’s Method
2.5. Birefringence Analysis of Collagen Fibers
2.6. Histological Evaluation of Neoformed Bone and Histomorphometric Evaluation
3. Discussion
4. Materials and Methods
4.1. Elastin Matrix Derived from Bovine Auricular Cartilage
4.2. Collagen Matrices Derived from Porcine Intestinal Serosa
4.3. Experimental Design
4.4. Bone Defect Surgeries
4.5. Sequential Fluorescent Labeling
4.6. Histologic Preparation for Confocal Laser Scanning Microscopy Analysis
4.7. Histologic Preparation and Von Kossa’s Staining Method
4.8. Macroscopic and Radiological Analysis
4.9. Micro-CT Scan (μ-CT)
4.10. Histological Procedures–Masson’s Trichrome Staining and Picrosirius Red
4.11. Histomorphometric Evaluation
4.12. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Rodeo, S.A.; Boden, S.D.; Murray, M.M.; Einhorn, T.A. 2011 aoa symposium: Tissue engineering and tissue regeneration: Aoa critical issues. J. Bone Jt. Surg. Ser. A 2013, 95, e109. [Google Scholar] [CrossRef] [PubMed]
- Quinnan, S.M. Segmental Bone Loss Reconstruction Using Ring Fixation. J. Orthop. Trauma 2017, 31, S42–S46. [Google Scholar] [CrossRef] [PubMed]
- Egol, K.A.; Nauth, A.; Lee, M.; Pape, H.C.; Watson, J.T.; Borrelli, J. Bone grafting: Sourcing, timing, strategies, and alternatives. J. Orthop. Trauma 2015, 29, S10–S14. [Google Scholar] [CrossRef]
- Sprio, S.; Fricia, M.; Maddalena, G.F.; Nataloni, A.; Tampieri, A. Osteointegration in cranial bone reconstruction: A goal to achieve. J. Appl. Biomater. Funct. Mater. 2016, 14, e470–e476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Orciani, M.; Fini, M.; Di Primio, R.; Mattioli-Belmonte, M. Biofabrication and bone tissue regeneration: Cell source, approaches, and challenges. Front. Bioeng. Biotechnol. 2017, 5, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saska, S.; Pigossi, S.C.; Oliveira, G.J.P.L.; Teixeira, L.N.; Capela, M.V.; Gonçalves, A.; De Oliveira, P.T.; Messaddeq, Y.; Ribeiro, S.J.L.; Gaspar, A.M.M.; et al. Biopolymer-based membranes associated with osteogenic growth peptide for guided bone regeneration. Biomed. Mater. 2018, 13, 035009. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Moraes, R.; de Guzzi Plepis, A.M.; da Conceição Amaro Martins, V.; Duarte, M.A.H.; Alcalde, M.P.; Buchaim, R.L.; Pomini, K.T.; Machado, E.G.; de Azevedo e Sousa Munhoz, M.; Cunha, F.B.; et al. Suitability of the use of an elastin matrix combined with bone morphogenetic protein for the repair of cranial defects. Am. J. Transl. Res. 2019, 11, 5261–5271. [Google Scholar] [PubMed]
- Munhoz, M.A.S.; Hirata, H.H.; Plepis, A.M.G.; Martins, V.C.A.; Cunha, M.R. Use of collagen/chitosan sponges mineralized with hydroxyapatite for the repair of cranial defects in rats. Injury 2018, 49, 2154–2160. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Lv, Y. Application of collagen scaffold in tissue engineering: Recent advances and new perspectives. Polymers 2016, 8, 42. [Google Scholar] [CrossRef] [Green Version]
- Ferreira, A.M.; Gentile, P.; Chiono, V.; Ciardelli, G. Collagen for bone tissue regeneration. Acta Biomater. 2012, 8, 3191–3200. [Google Scholar] [CrossRef]
- Castilla Bolaños, M.A.; Buttigieg, J.; Briceño Triana, J.C. Development and characterization of a novel porous small intestine submucosa-hydroxyapatite scaffold for bone regeneration. Mater. Sci. Eng. C 2017, 72, 519–525. [Google Scholar] [CrossRef]
- Li, M.; Mondrinos, M.J.; Chen, X.; Gandhi, M.R.; Ko, F.K.; Lelkes, P.I. Co-electrospun poly(lactide-co-glycolide), gelatin, and elastin blends for tissue engineering scaffolds. J. Biomed. Mater. Res. Part A 2006, 79, 963–973. [Google Scholar] [CrossRef]
- Daamen, W.F.; Veerkamp, J.H.; van Hest, J.C.M.; van Kuppevelt, T.H. Elastin as a biomaterial for tissue engineering. Biomaterials 2007, 28, 4378–4398. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, K.D.; Cardoso, L.D.; Oliveira, L.P.; Franzo, V.S.; Pancotti, A.; Miguel, M.P.; Silva, L.A.F.; Vulcani, V.A.S. Histological analysis of elastic cartilages treated with alkaline solution. Arq. Bras. Med. Vet. Zootec. 2020, 72, 647–654. [Google Scholar] [CrossRef]
- Rey, F.; Barzaghini, B.; Nardini, A.; Bordoni, M.; Zuccotti, G.V.; Cereda, C.; Raimondi, M.T.; Carelli, S. Advances in Tissue Engineering and Innovative Fabrication Techniques for 3-D-Structures: Translational Applications in Neurodegenerative Diseases. Cells 2020, 9, 1636. [Google Scholar] [CrossRef] [PubMed]
- Ratner, B.D. A pore way to heal and regenerate: 21st century thinking on biocompatibility. Regen. Biomater. 2016, 3, 107–110. [Google Scholar] [CrossRef] [Green Version]
- Saber-Samandari, S.; Saber-Samandari, S. Biocompatible nanocomposite scaffolds based on copolymer-grafted chitosan for bone tissue engineering with drug delivery capability. Mater. Sci. Eng. C 2017, 75, 721–732. [Google Scholar] [CrossRef] [PubMed]
- Buchaim, R.L.; Goissis, G.; Andreo, J.C.; Roque, D.D.; Roque, J.S.; Buchaim, D.V.; de Rodrigues, A.C. Biocompatibility of anionic collagen matrices and its influence on the orientation of cellular growth. Brazilian Dent. Sci. 2007, 10, 12–20. [Google Scholar] [CrossRef]
- de Azevedo e Sousa Munhoz, M.; Pomini, K.T.; de Guzzi Plepis, A.M.; da Conceição Amaro Martins, V.; Machado, E.G.; de Moraes, R.; Cunha, F.B.; Santos, A.R.; Cardoso, G.B.C.; Duarte, M.A.H.; et al. Elastin-derived scaffolding associated or not with bone morphogenetic protein (BMP) or hydroxyapatite (HA) in the repair process of metaphyseal bone defects. PLoS ONE 2020, 15, e0231112. [Google Scholar] [CrossRef] [Green Version]
- Cunha, M.R.; Santos, A.R.; Goissis, G.; Genari, S.C. Implants of polyanionic collagen matrix in bone defects of ovariectomized rats. J. Mater. Sci. Mater. Med. 2008, 19, 1341–1348. [Google Scholar] [CrossRef] [PubMed]
- Miguel, F.B.; De Almeida Barbosa Júnior, A.; De Paula, F.L.; Barreto, I.C.; Goissis, G.; Rosa, F.P. Regeneration of critical bone defects with anionic collagen matrix as scaffolds. J. Mater. Sci. Mater. Med. 2013, 24, 2567–2575. [Google Scholar] [CrossRef] [PubMed]
- Braile-Sternieri, M.C.V.B.; Goissis, G.; de Giglioti, A.F.; Ramirez, V.D.A.; Pereira, N.P.; de Vasconcellos, A.; Basso-Frazzato, G.G.; Braile, D.M. In vivo evaluation of Vivere bovine pericardium valvular bioprosthesis with a new anti-calcifying treatment. Artif. Organs 2020, 44, E482–E493. [Google Scholar] [CrossRef]
- Maisani, M.; Pezzoli, D.; Chassande, O.; Mantovani, D. Cellularizing hydrogel-based scaffolds to repair bone tissue: How to create a physiologically relevant micro-environment? J. Tissue Eng. 2017, 8, 2041731417712073. [Google Scholar] [CrossRef] [Green Version]
- Pomini, K.T.; Buchaim, D.V.; Andreo, J.C.; Rosso, M.P.d.O.; Della Coletta, B.B.; German, Í.J.S.; Biguetti, A.C.C.; Shinohara, A.L.; Rosa Júnior, G.M.; Shindo, J.V.T.C.; et al. Fibrin sealant derived from human plasma as a scaffold for bone grafts associated with photobiomodulation therapy. Int. J. Mol. Sci. 2019, 20, 1761. [Google Scholar] [CrossRef] [Green Version]
- Rosso, M.P.D.O.; Oyadomari, A.T.; Pomini, K.T.; Botteon, B.; Coletta, D.; Cosin, T.; Seabra, R.; Ferreira, J.; Barraviera, B.; Cassaro, C.V.; et al. Photobiomodulation Therapy Associated with Heterologous Fibrin Biopolymer and Bovine Bone Matrix Helps to Reconstruct Long Bones. Biomolecules 2020, 10, 383. [Google Scholar] [CrossRef] [Green Version]
- Ebrahimi, M.; Botelho, M.G.; Dorozhkin, S.V. Biphasic calcium phosphates bioceramics (HA/TCP): Concept, physicochemical properties and the impact of standardization of study protocols in biomaterials research. Mater. Sci. Eng. C 2017, 71, 1293–1312. [Google Scholar] [CrossRef]
- Pettian, M.S.; De Guzzi Plepis, A.M.; Da Conceição Amaro Martins, V.; Dos Santos, G.R.; Lopes Pinto, C.A.; Galdeano, E.A.; Alves Calegari, A.R.; De Moraes, C.A.; Da Cunha, M.R. Use of an anionic collagen matrix made from bovine intestinal serosa for in vivo repair of cranial defects. PLoS ONE 2018, 13, e0197806. [Google Scholar] [CrossRef] [PubMed]
- Hodde, J.P.; Badylak, S.F.; Brightman, A.O.; Voytik-Harbin, S.L. Glycosaminoglycan content of small intestinal submucosa: A bioscaffold for tissue replacement. Tissue Eng. 1996, 2, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Oryan, A.; Moshiri, A.; Sharifi, P. Advances in injured tendon engineering with emphasis on the role of collagen implants. Hard Tissue 2012, 1, 12. [Google Scholar] [CrossRef]
- Müller, S.A.; Dürselen, L.; Heisterbach, P.; Evans, C.; Majewski, M. Effect of a Simple Collagen Type i Sponge for Achilles Tendon Repair in a Rat Model. Am. J. Sports Med. 2016, 44, 1998–2004. [Google Scholar] [CrossRef]
- Buchaim, R.; Rosso, M.; Andreo, J.; Buchaim, D.; Okamoto, R.; Rodrigues, A.; Shinohara, A.; Roque, J.; Roque, D.; Junior, G.; et al. A New Anionic Bovine Tendon as Scaffold for the Repair of Bone Defects: A Morphological, Histomorphometric and Immunohistochemical Study. Br. J. Med. Med. Res. 2015, 10, 1–11. [Google Scholar] [CrossRef]
- Rocha, L.B.; Goissis, G.; Rossi, M.A. Biocompatibility of anionic collagen matrix as scaffold for bone healing. Biomaterials 2002, 23, 449–456. [Google Scholar] [CrossRef]
- Janicki, P.; Schmidmaier, G. What should be the characteristics of the ideal bone graft substitute? Combining scaffolds with growth factors and/or stem cells. Injury 2011, 42, S77–S81. [Google Scholar] [CrossRef]
- Cardoso, G.B.C.; Maniglio, D.; Volpato, F.Z.; Tondon, A.; Migliaresi, C.; Kaunas, R.R.; Zavaglia, C.A.C. Oleic acid surfactant in polycaprolactone/hydroxyapatite-composites for bone tissue engineering. J. Biomed. Mater. Res. Part B Appl. Biomater. 2016, 104, 1076–1082. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Gu, Q.; Chen, M.; Zhang, C.; Chen, S.; Zhao, J. Controlled delivery of icariin on small intestine submucosa for bone tissue engineering. Mater. Sci. Eng. C. Mater. Biol. Appl. 2017, 71, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Liu, M.; Yao, S.; Ji, Y.; Xiong, Z.; Tang, K.; Chen, K.; Yang, H.; Guo, X. Biomimetic composite scaffold containing small intestinal submucosa and mesoporous bioactive glass exhibits high osteogenic and angiogenic capacity. Tissue Eng. Part A 2018, 24, 1044–1056. [Google Scholar] [CrossRef]
- Sun, J.L.; Jiao, K.; Niu, L.N.; Jiao, Y.; Song, Q.; Shen, L.J.; Tay, F.R.; Chen, J. Intrafibrillar silicified collagen scaffold modulates monocyte to promote cell homing, angiogenesis and bone regeneration. Biomaterials 2017, 113, 203–216. [Google Scholar] [CrossRef] [PubMed]
- Neffe, A.T.; Pierce, B.F.; Tronci, G.; Ma, N.; Pittermann, E.; Gebauer, T.; Frank, O.; Schossig, M.; Xu, X.; Willie, B.M.; et al. One step creation of multifunctional 3D architectured hydrogels inducing bone regeneration. Adv. Mater. 2015, 27, 1738–1744. [Google Scholar] [CrossRef] [PubMed]
- Lohmann, P.; Willuweit, A.; Neffe, A.T.; Geisler, S.; Gebauer, T.P.; Beer, S.; Coenen, H.H.; Fischer, H.; Hermanns-Sachweh, B.; Lendlein, A.; et al. Bone regeneration induced by a 3D architectured hydrogel in a rat critical-size calvarial defect. Biomaterials 2017, 113, 158–169. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, D.A.; de Guzzi Plepis, A.M.; da Conceição Amaro Martins, V.; Cardoso, G.B.C.; Santos, A.R.; Iatecola, A.; Andrade, T.N.; Monteiro, F.M.; Calegari, A.R.A.; Chacon, E.L.; et al. Effects of the combination of low-level laser therapy and anionic polymer membranes on bone repair. Lasers Med. Sci. 2020, 35, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Massimino, L.C.; da Conceição Amaro Martins, V.; Vulcani, V.A.S.; de Oliveira, É.L.; Andreeta, M.B.; Bonagamba, T.J.; Klingbeil, M.F.G.; Mathor, M.B.; de Guzzi Plepis, A.M. Use of collagen and auricular cartilage in bioengineering: Scaffolds for tissue regeneration. Cell Tissue Bank. 2020. [Google Scholar] [CrossRef] [PubMed]
- Horn, M.M.; Martins, V.C.A.; de Guzzi Plepis, A.M. Interaction of anionic collagen with chitosan: Effect on thermal and morphological characteristics. Carbohydr. Polym. 2009, 77, 239–243. [Google Scholar] [CrossRef]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cunha, F.B.; Pomini, K.T.; Plepis, A.M.d.G.; Martins, V.d.C.A.; Machado, E.G.; de Moraes, R.; Munhoz, M.d.A.e.S.; Machado, M.V.R.; Duarte, M.A.H.; Alcalde, M.P.; et al. In Vivo Biological Behavior of Polymer Scaffolds of Natural Origin in the Bone Repair Process. Molecules 2021, 26, 1598. https://doi.org/10.3390/molecules26061598
Cunha FB, Pomini KT, Plepis AMdG, Martins VdCA, Machado EG, de Moraes R, Munhoz MdAeS, Machado MVR, Duarte MAH, Alcalde MP, et al. In Vivo Biological Behavior of Polymer Scaffolds of Natural Origin in the Bone Repair Process. Molecules. 2021; 26(6):1598. https://doi.org/10.3390/molecules26061598
Chicago/Turabian StyleCunha, Fernando Bento, Karina Torres Pomini, Ana Maria de Guzzi Plepis, Virgínia da Conceição Amaro Martins, Eduardo Gomes Machado, Renato de Moraes, Marcelo de Azevedo e Souza Munhoz, Michela Vanessa Ribeiro Machado, Marco Antonio Hungaro Duarte, Murilo Priori Alcalde, and et al. 2021. "In Vivo Biological Behavior of Polymer Scaffolds of Natural Origin in the Bone Repair Process" Molecules 26, no. 6: 1598. https://doi.org/10.3390/molecules26061598






