Scheme 1.
Indolizines (A) in [8+2] cycloaddition reaction forming cycl[3.2.2]azines (B). The reaction may proceed with alkynes or alkenes via dihydro- (C1) or tetrahydro- (C2) cyclazines.
Scheme 1.
Indolizines (A) in [8+2] cycloaddition reaction forming cycl[3.2.2]azines (B). The reaction may proceed with alkynes or alkenes via dihydro- (C1) or tetrahydro- (C2) cyclazines.
Scheme 2.
Different images of the structure of cyclazine.
Scheme 2.
Different images of the structure of cyclazine.
Scheme 3.
Possible benzo- aza- and benzoazacyclazines discussed in this review.
Scheme 3.
Possible benzo- aza- and benzoazacyclazines discussed in this review.
Scheme 4.
Symthesis of cyclazines from indolizines (
Table 1).
Scheme 4.
Symthesis of cyclazines from indolizines (
Table 1).
Scheme 5.
Synthesis of bis-cyclazinylethanes from bis-indolizinylethanes.
Scheme 5.
Synthesis of bis-cyclazinylethanes from bis-indolizinylethanes.
Scheme 6.
i N-bromsuccinimide. 6 h; ii K2CO3.
Scheme 6.
i N-bromsuccinimide. 6 h; ii K2CO3.
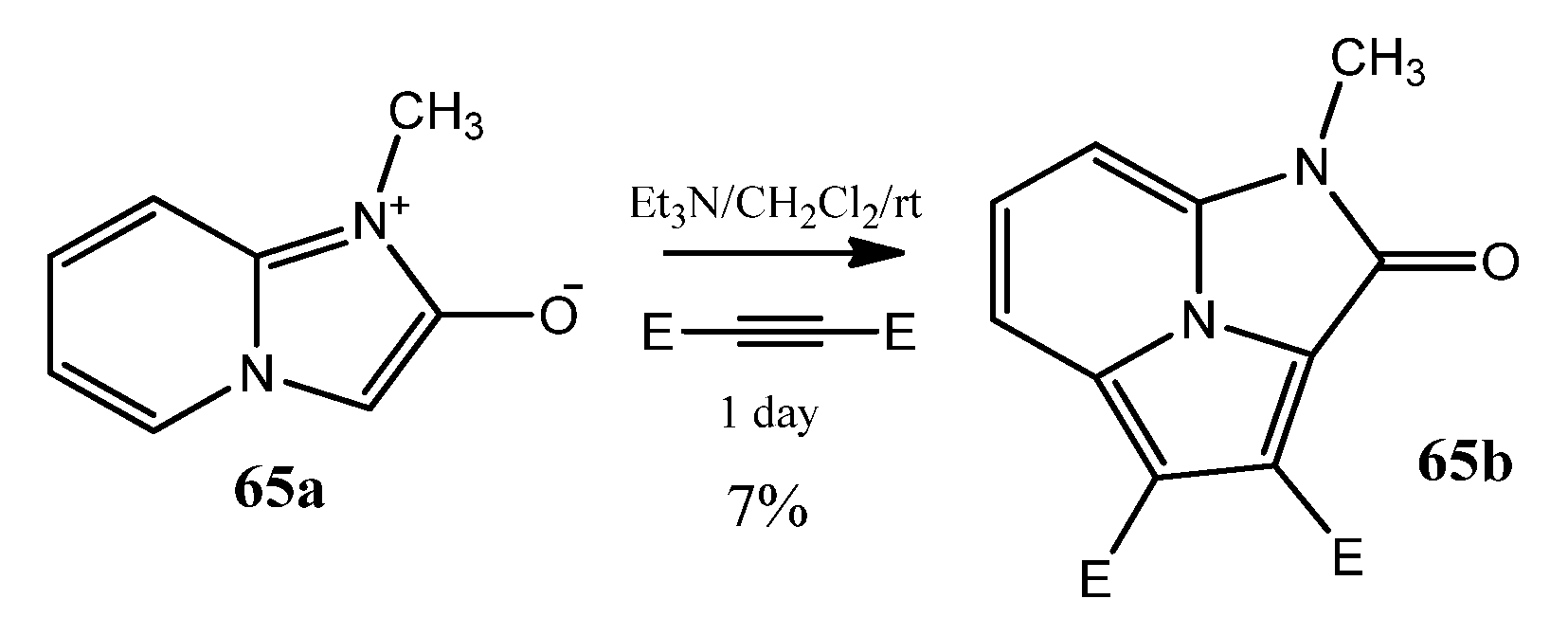
Scheme 7.
Synthesis of cyclazino-cyclazinone.
Scheme 7.
Synthesis of cyclazino-cyclazinone.
Scheme 8.
Synthesis of cyclazines from 1,8-cycloannelated indolizines (see
Table 2).
Scheme 8.
Synthesis of cyclazines from 1,8-cycloannelated indolizines (see
Table 2).
Scheme 9.
Abnormal cycloaddition to 1,8-cycloannelated indolizine combined with dehydrogenation.
Scheme 9.
Abnormal cycloaddition to 1,8-cycloannelated indolizine combined with dehydrogenation.
Scheme 10.
10 mol % Pd(OAc)2, DMSO, O2 (1 atm), no base.
Scheme 10.
10 mol % Pd(OAc)2, DMSO, O2 (1 atm), no base.
Scheme 11.
Bengal rose KI, air, DMSO, 8 h, 20 W, blue LED.
Scheme 11.
Bengal rose KI, air, DMSO, 8 h, 20 W, blue LED.
Scheme 12.
Bengal Rose, TFA, CH2Cl2, O2, 10 h, 20 W blue LED.
Scheme 12.
Bengal Rose, TFA, CH2Cl2, O2, 10 h, 20 W blue LED.
Scheme 13.
10 mol % Pd(OAc)2, 1 eq benzoquinone, O2, 2 eq KOAc, DMF. 120°, 12 h.
Scheme 13.
10 mol % Pd(OAc)2, 1 eq benzoquinone, O2, 2 eq KOAc, DMF. 120°, 12 h.
Scheme 14.
Cycloaddition of indolizines with 3(5)-leaving groups.
Scheme 14.
Cycloaddition of indolizines with 3(5)-leaving groups.
Scheme 15.
Examples of 3(5)-substituted indolizines involved in cycloaddition (see
Table 7).
Scheme 15.
Examples of 3(5)-substituted indolizines involved in cycloaddition (see
Table 7).
Scheme 16.
Cycloaddition to 3-CN-substituted indolizines (see
Table 8).
Scheme 16.
Cycloaddition to 3-CN-substituted indolizines (see
Table 8).
Scheme 17.
Structure of 1:2 adduct of 3-cyanoindolizine and DMAD.
Scheme 17.
Structure of 1:2 adduct of 3-cyanoindolizine and DMAD.
Scheme 18.
Cycloaddition to 3-CN-substituted benzoindolizines (see
Table 9).
Scheme 18.
Cycloaddition to 3-CN-substituted benzoindolizines (see
Table 9).
Scheme 19.
CsF, MeCN, 90°. A—ortho-substituted benzene with SiMe3 and OSO2CF3; B—1-Aminobenzotriazole/Pb(OAc)4.
Scheme 19.
CsF, MeCN, 90°. A—ortho-substituted benzene with SiMe3 and OSO2CF3; B—1-Aminobenzotriazole/Pb(OAc)4.
Scheme 20.
CsF, MeCN, 90°. 43r (R7R8 = H, 93%), 43s (R7,R8 = benzo, 62%), Method A.
Scheme 20.
CsF, MeCN, 90°. 43r (R7R8 = H, 93%), 43s (R7,R8 = benzo, 62%), Method A.
Scheme 21.
Cycloaddition to benzoindolizines see
Table 11).
Scheme 21.
Cycloaddition to benzoindolizines see
Table 11).
Scheme 22.
Pd-C/PhMe/Δ/30h. 47a R2 = H (33%), 47b R2 = MeS (27%).
Scheme 22.
Pd-C/PhMe/Δ/30h. 47a R2 = H (33%), 47b R2 = MeS (27%).
Scheme 23.
MePh + HOAc/Δ/20 h.
Scheme 23.
MePh + HOAc/Δ/20 h.
Scheme 24.
Example of cycloaddition to fused indolizines.
Scheme 24.
Example of cycloaddition to fused indolizines.
Scheme 25.
Pd-C/PhMe/Δ/14 h.
Scheme 25.
Pd-C/PhMe/Δ/14 h.
Scheme 26.
Microwave three-component synthesis of cyclazines (see
Table 12).
Scheme 26.
Microwave three-component synthesis of cyclazines (see
Table 12).
Scheme 28.
TEA (1.2 eq), ethyl propiolate (1.5 eq), CH3CN + DMF, rt, 24 h.
Scheme 28.
TEA (1.2 eq), ethyl propiolate (1.5 eq), CH3CN + DMF, rt, 24 h.
Scheme 29.
Synthesis of benzocyclazines from pyridinium ylides (
Table 13).
Scheme 29.
Synthesis of benzocyclazines from pyridinium ylides (
Table 13).
Scheme 30.
Multistep reaction ending with cyclazines.
Scheme 30.
Multistep reaction ending with cyclazines.
Scheme 31.
Synthesis of azacyclazines from azaindolizines (
Table 14).
Scheme 31.
Synthesis of azacyclazines from azaindolizines (
Table 14).
Scheme 32.
Synthesis of azacyclazinone from mesoionic structure.
Scheme 32.
Synthesis of azacyclazinone from mesoionic structure.
Scheme 33.
Pd(OAc)
2, Cu(OAc)
2, DMSO, 110 °C, 12 h [
90]. Pd-Se complex Cu(OAc)
2, KOtBu, DMAC, 120 °C, 16 h [
91].
Scheme 33.
Pd(OAc)
2, Cu(OAc)
2, DMSO, 110 °C, 12 h [
90]. Pd-Se complex Cu(OAc)
2, KOtBu, DMAC, 120 °C, 16 h [
91].
Scheme 34.
Pd-NHC complex, Cu(OAc)2, TBAB, DMA, 90 °C, 12 h.
Scheme 34.
Pd-NHC complex, Cu(OAc)2, TBAB, DMA, 90 °C, 12 h.
Scheme 35.
DMF, PEG-1500, sealed tube Pd(OAc)2, Cu(OAc)2, TBAB 90 °C, 12 h.
Scheme 35.
DMF, PEG-1500, sealed tube Pd(OAc)2, Cu(OAc)2, TBAB 90 °C, 12 h.
Scheme 36.
Reaction of azaindolizines with benzynes (
Table 18).
Scheme 36.
Reaction of azaindolizines with benzynes (
Table 18).
Scheme 37.
Reaction of imidazopyridines with benzynes (
Table 19).
Scheme 37.
Reaction of imidazopyridines with benzynes (
Table 19).
Scheme 38.
Pd/xphos, K2CO3, DMF, 160 °C, 24 h.
Scheme 38.
Pd/xphos, K2CO3, DMF, 160 °C, 24 h.
Scheme 39.
Cycloaddition of tetraazapentalene derivatives.
Scheme 39.
Cycloaddition of tetraazapentalene derivatives.
Scheme 40.
Synthesis of diazacyclazine.
Scheme 40.
Synthesis of diazacyclazine.
Scheme 41.
Cyclization of 6-ethynylpyridinium salts.
Scheme 41.
Cyclization of 6-ethynylpyridinium salts.
Scheme 42.
Pd(PPh3)2Cl2/CuI/MeCN/Et3N/rt.
Scheme 42.
Pd(PPh3)2Cl2/CuI/MeCN/Et3N/rt.
Scheme 43.
79a–c Me, n-Bu, Ph (70–80%).
Scheme 43.
79a–c Me, n-Bu, Ph (70–80%).
Scheme 44.
Pd(OAc)2 10 mol %. PPh3, K2CO3. PhMe/115 °C/60 h.
Scheme 44.
Pd(OAc)2 10 mol %. PPh3, K2CO3. PhMe/115 °C/60 h.
Scheme 45.
Cyclization of Br-substituted 3-arylimidazopyridines (
Table 21).
Scheme 45.
Cyclization of Br-substituted 3-arylimidazopyridines (
Table 21).
Scheme 46.
Unusual ring closure of 5-chlorindolizines.
Scheme 46.
Unusual ring closure of 5-chlorindolizines.
Scheme 47.
Example of concurrence between [8+2] and [8+2] cycloaddition.
Scheme 47.
Example of concurrence between [8+2] and [8+2] cycloaddition.
Scheme 48.
Concurrence in cycloaddition for 2-arylimidazopyridines (
Table 22).
Scheme 48.
Concurrence in cycloaddition for 2-arylimidazopyridines (
Table 22).
Scheme 49.
Theoretically possible mechanisms of [8+2] cycloaddition to indolizines.
Scheme 49.
Theoretically possible mechanisms of [8+2] cycloaddition to indolizines.
Scheme 50.
Abnormal cycloadditon to 6-nitroindolizine.
Scheme 50.
Abnormal cycloadditon to 6-nitroindolizine.
Scheme 51.
Possible channels of transformation of initially formed zwitter-ion.
Scheme 51.
Possible channels of transformation of initially formed zwitter-ion.
Scheme 52.
3-Vinyl derivatives of aza/benzo/indolizines isolated as intermediates (see
Table 24).
Scheme 52.
3-Vinyl derivatives of aza/benzo/indolizines isolated as intermediates (see
Table 24).
Scheme 53.
Dihydrocyclazines obtained in the synthesis.
Scheme 53.
Dihydrocyclazines obtained in the synthesis.
Scheme 54.
Unusual cycloaddion to 6-nitroindolizine with the loss of NO2 group.
Scheme 54.
Unusual cycloaddion to 6-nitroindolizine with the loss of NO2 group.
Scheme 55.
1:1 and 1:2 cycloadducts of 5-methylindolizine and DMAD.
Scheme 55.
1:1 and 1:2 cycloadducts of 5-methylindolizine and DMAD.
Scheme 56.
Different cycloadducts obtained from benzoindolizine.
Scheme 56.
Different cycloadducts obtained from benzoindolizine.
Scheme 57.
Structure of some adducts and cycloadducts of indoliznes and alkenes.
Scheme 57.
Structure of some adducts and cycloadducts of indoliznes and alkenes.
Table 1.
Substituents, reaction conditions and yields of reactions of indolizines with substituted acetylenes giving cyclazines (
Scheme 4).
Table 1.
Substituents, reaction conditions and yields of reactions of indolizines with substituted acetylenes giving cyclazines (
Scheme 4).
| N | R/R’ | R1 | R2 | R6 | R7 | R8 | Cat/Solv/T°/Time | Yield % | Ref. |
|---|
| 1 | E/E a | H | H | H | H | H | Pd-C/MePh/Δ | 50–66 | [22] |
| 2 | E/E | H | H | H | H | H | Pd-C/MePh/Δ/24 h | 68 | [23] |
| 3 | E/E | H | H | H | H | H | MnO2/MePh/Δ/16 h | 55 | [4] |
| 4 | E/H | H | H | H | H | H | Pd-C/MePh/Δ/24 h | 11 | [24] |
| 5 | E/E | H | Ph | H | H | H | Pd-C/MePh/Δ/20 h | 28 | [25] |
| 6 | E/E | H | Me | H | H | H | Pd-C/MePh/Δ | 60 | [27] |
| 7a | E/E | H | H | H | H | Me | Pd-C/MePh/Δ/1.5 h | 53 | [28] |
| 7b | E/E | H | H | H | H | Pr | Pd-C/MePh/Δ/1.5 h | 53 | [28] |
| 7c | E/E | H | H | H | H | Ph | Pd-C/MePh/Δ/1.5 h | 53 | [28] |
| 8a | E/E | H | Ph | H | H | H | Pd-C/MePh/Δ | 39 | [26] |
| 8b | E/E | H | p-tBuPh | H | H | H | Pd-C/MePh/Δ | 75 | [26] |
| 9a | E/E | H | H | H | H | H | Pd-C/MePh/Δ/100 h | 33 | [29] |
| 9b | E/E | H | H | H | H | Me | Pd-C/MePh/D/100 h | 34 | [29] |
| 9c | E/E | H | H | Me | H | Me | Pd-C/MePh/Δ/100 h | 25 | [29] |
| 10 | E/E | CONH2 | MeS | H | H | H | Pd-C/MePh/Δ/24 h | 70 | [30] |
| 11a | E/H | CONH2 | MeS | H | H | H | Pd-C/MePh/Δ/100 h | 45 | [31] |
| 11b | E/H | CONH2 | MeS | H | Me | H | Pd-C/MePh/Δ/100 h | 48 | [31] |
| 12a | E/E | H | MeS | H | H | H | Pd-C/MePh/Δ/100 h | 38 | [29] |
| 12b | E/E | H | MeS | H | H | Me | Pd-C/MePh/Δ/100 h | 40 | [29] |
| 12c | E/E | H | MeS | Me | H | Me | Pd-C/MePh/Δ/100 h | 49 | [29] |
| 13a | E/E | H | H | H | NMe2 | H | Pd-C/MePh/Δ | 32 | [2] |
| 13b | E/E | H | MeS | H | NMe2 | H | Pd-C/MePh/Δ | 17 | [2] |
| 14a | E’/E’ b | F | p-Brh | H | H | H | Cu(OAc)2/PhMe/Δ/5 h | 63 | [34] |
| 14b | E’/E’ | F | p-MeOPh | H | H | H | Cu(OAc)2/PhMe/Δ/8 h | 70 | [34] |
| 15 | E/H | E | Me | H | H | H | Pd-C/PhH/Δ/24 h | NG c | [39] |
| 16a | E/H | 2-Py | Me | H | H | H | No/NO2Ph/Δ/20 h | 43 | [40] |
| 16b | E/E | 2-Py | Me | H | H | H | No/NO2Ph/Δ/20 h | 47 | [40] |
| 17 | E/E | H | Me | o-OHPhCO | H | COMe | Pd-C/MePh/Δ/16 h | 54 | [35] |
| 18 | E/H | H | t-Bu | H | H | H | O2/MePh/Δ/4 h | 79 | [36] |
| 19 | Me2B/H | H | H | H | H | H | (1) MePh/Δ/3 d (2) DDQ | 55 | [37] |
| 20a | E/E | H | Styryl | H | H | H | No/PhMe/50o/31 h | 44 | [38] |
| 20b | E/H | H | Styryl | H | H | H | No/PhMe/20o/120 h | 64 | [38] |
Table 2.
Substituents and yields of cycloaddition of acetylenes to indolizines
23–
25 annelated by a ring across the positions C1 and C-8 (
Scheme 8).
Table 2.
Substituents and yields of cycloaddition of acetylenes to indolizines
23–
25 annelated by a ring across the positions C1 and C-8 (
Scheme 8).
| N | R/R’ | R8-R1 | R2 | Cat/Solv/T°/Time | Yield % | Ref. |
|---|
| 23a | E/H | (CH2)4 | Me | Pd-C/PhMe/Δ/50 h | 75 | [42] |
| 23b | E/H | (CH2)3 | Me | Pd-C/PhMe/Δ/50 h | 75 | [42] |
| 23c | E/E | (CH2)4 | Me | Pd-C/PhMe/Δ/50 h | 85 | [42] |
| 23d | E/E | (CH2)3 | Ph | Pd-C/PhMe/Δ/50 h | 58 | [42] |
| 23d | E/E | (CH2)4 | Ph | Pd-C/PhMe/Δ/50 h | 61 | [42] |
| 23e | E/E | (CH2)3 | Me | Pd-C/PhMe/Δ/50 h | 77 | [42] |
| 24f | COPh/COPh | (CH2)4 | Me | DDQ/THF/0°/1 h | 86 | [42] |
| 23g | COPh/COPh | (CH2)4 | Ph | DDQ/THF/0°/1 h | 92 | [42] |
| 23h | COPh/COPh | (CH2)3 | Me | DDQ/THF/0°/1 h | 68 | [42] |
| 23i | COPh/COPh | (CH2)3 | Ph | DDQ/THF/0°/1 h | 52 | [42] |
| 24a | E/E | O=C(CH2)2 | Ph | DDQ/MePh/Δ/2 h | 92 | [45] |
| 24b | E/E | O=C(CH2)2 | E’ | DDQ/MePh/Δ/2 h | 98 | [45] |
| 25a | COPh/COPh | CH2-NE’-CH2 | H | DDQ/THF/0°/10 m | 66 | [46] |
| 25b | COPh/COPh | CH2-NE’-(CH2)2 | H | DDQ/THF/0°/10 m | 84 | [46] |
Table 3.
Substituents and yields of cycloaddition of diarylalkynes to indolizines (
Scheme 10).
Table 3.
Substituents and yields of cycloaddition of diarylalkynes to indolizines (
Scheme 10).
| N | R | R’ | R1 | R2 | Yield % | Ref. |
|---|
| 26a | Ph | Ph | E’ | E’ | 92 | [47] |
| 26b | 4-MePh | 4-MePh | E | H | 88 | [47] |
| 26c | Ph | Ph | E | E | 98 | [47] |
| 26d | 4-FPh | 4-FPh | E | E | 85 | [47] |
| 26e | 3-BrPh | 3-BrPh | E | E | 56 | [47] |
| 26f | 4-NO2Ph | 4-MeOPh | E | E | 80 (6:1) | [47] |
| 26g | 4-NO2Ph | Ph | E | E | 68 (20:1) | [47] |
| 26h | 4-FPh | 4-MeOPh | E | E | 91 (2:1) | [47] |
| 26i | C≡CPh | Ph | E | E | 41;39 | [47] |
| 26j | Ph | Ph | CO2nBu | CO2nBu | 79 | [47] |
| 26k | Ph | Ph | E | Ph | 20 * | [47] |
| 26l | Ph | Ph | CO2tBu | H | 55 | [47] |
| 26l | Ph | Ph | H | E | 70 | [47] |
| 26m | Ph | Ph | CONMe2 | H | 76 | [47] |
| 26n | Ph | Ph | E’ | H | 87 | [47] |
| 26o | Ph | Ph | E | H | 90 | [47] |
| 26p | Ph | Ph | CN | H | 59 | [47] |
Table 4.
Substituents and yields of photochemical cycloaddition to indolizines (
Scheme 11).
Table 4.
Substituents and yields of photochemical cycloaddition to indolizines (
Scheme 11).
| N | R’ | R” | R1 | R2 | R6 | R7 | R8 | Yield, % | Ref. |
|---|
| 27a | CHO | Ph | H | Ph | H | H | H | 87 | [48] |
| 27b | CHO | n-C5H11 | H | Ph | H | H | H | 61 | [48] |
| 27c | CHO | 2-Thienyl | H | Ph | H | H | H | 66 | [48] |
| 27d | CHO | 3-Cl-Ph | H | Ph | H | H | H | 79 | [48] |
| 27e | CHO | 3-Ac-Ph | H | Ph | H | H | H | 58 | [48] |
| 27f | CHO | 4-Me-Ph | H | Ph | H | H | H | 77 | [48] |
| 27g | CHO | 3,4-Me2Ph | H | Ph | H | H | H | 57 | [48] |
| 27h | CHO | 2-Naphtyl | H | Ph | H | H | H | 59 | [48] |
| 27i | COPh | Ph | H | Ph | H | H | H | 58 | [48] |
| 27j | Ph | Ph | H | Ph | H | H | H | 0 | [48] |
| 27k | CHO | Ph | H | 4-OMe-Ph | H | H | H | 70 | [48] |
| 27l | CHO | Ph | H | 4-F-Ph | H | H | H | 81 | [48] |
| 27m | CHO | Ph | H | 4-NO2-Ph | H | H | H | 58 | [48] |
| 27n | CHO | Ph | H | 4-Br-Ph | H | H | H | 64 | [48] |
| 27o | CHO | Ph | H | 4-CF3-Ph | H | H | H | 71 | [48] |
| 27 | CHO | Ph | H | 2-F-Ph | H | H | H | 73 | [48] |
| 27p | CHO | Ph | H | 3-Me-Ph | H | H | H | 63 | [48] |
| 27q | CHO | Ph | H | 3,4-Cl2-Ph | H | H | H | 58 | [48] |
| 27r | CHO | Ph | H | 2,4-Cl2-Ph | H | H | H | 78 | [48] |
| 27s | CHO | Ph | H | 3,4-(OMe)2-Ph | H | H | H | 84 | [48] |
| 27t | CHO | Ph | H | 1,3-Benzo-dioxolyl-5 | H | H | H | 67 | [48] |
| 27u | CHO | Ph | H | Furyl | H | H | H | 62 | [48] |
| 27v | CHO | Ph | H | 2-Naphtyl | H | H | H | 60 | [48] |
| 27w | CHO | Ph | E’ | H | H | H | H | 59 | [48] |
| 27x | CHO | Ph | H | Ph | Et | H | H | 61 | [48] |
| 27y | CHO | Ph | H | Ph | H | Me | H | 73 | [48] |
| 27z | CHO | Ph | H | Ph | H | OMe | H | 70 | [48] |
| 27a1 | CHO | Ph | H | Ph | Me | H | Me | 61 | [48] |
| 27b1 | E’ | H | H | Ph | H | H | H | 88 | [48] |
| 27c1 | E’ | H | H | Ph | H | H | H | 70 | [48] |
| 27d1 | E’ | E’ | H | Ph | H | H | H | 77 | [48] |
| 27e1 | E’ | E’ | H | Ph | H | H | H | 70 | [48] |
Table 5.
Substituents and yields of oxidative cycloaddition of alkenes to indolizines (
Scheme 12).
Table 5.
Substituents and yields of oxidative cycloaddition of alkenes to indolizines (
Scheme 12).
| N | R | R2 | R5 | R6 | R7 | R8 | Yield, % | Ref. |
|---|
| 28a | E | Ph | H | H | H | H | 80 | [49] |
| 28b | CO2nBu | Ph | H | H | H | H | 69 | [49] |
| 28c | CO2CH2CHOH | Ph | H | H | H | H | 68 | [49] |
| 28d | CONHt-Bu | Ph | H | H | H | H | 67 | [49] |
| 28e | 2-Py | Ph | H | H | H | H | 65 | [49] |
| 28f | E | Ph | Me | H | H | H | 78 | [49] |
| 28g | 2-Py | Ph | H | H | H | Me | 69 | [49] |
| 28h | E | Ph | H | H | Me | H | 77 | [49] |
| 28i | E | Ph | H | Et | H | H | 70 | [49] |
| 28j | E | 4-FPh | H | H | H | H | 75 | [49] |
| 28k | E | 4-BrPh | H | H | H | H | 78 | [49] |
| 28l | E | 4-MePh | H | H | H | H | 74 | [49] |
| 28m | E | 4-MeOPh | H | H | H | H | 71 | [49] |
| 28n | E | 3-FPh | H | H | H | H | 75 | [49] |
| 28o | E | 3-ClPh | H | H | H | H | 66 | [49] |
| 28p | E | 3-BrPh | H | H | H | H | 70 | [49] |
| 28q | E | 3-MePh | H | H | H | H | 71 | [49] |
| 28r | 2-Py | 3-MePh | H | H | H | H | 66 | [49] |
| 28s | E | 3-MeOPh | H | H | H | H | 68 | [49] |
| 28t | E | 3-FPh | H | H | H | H | 65 | [49] |
| 28u | E | 3,4-Cl2Ph | H | H | H | H | 70 | [49] |
| 28v | E | 3,4-(MeO)2Ph | H | H | H | H | 66 | [49] |
| 28x | E | 2-Thienyl | H | H | H | H | 69 | [49] |
Table 6.
Yields of catalytic cycloaddition of acrylates (
Scheme 13) to 1-cyanoindolizine.
Table 6.
Yields of catalytic cycloaddition of acrylates (
Scheme 13) to 1-cyanoindolizine.
| N | R | Yield, % | Ref. |
|---|
| 29a | Me | 39 | [50] |
| 29b | Ph | 52 | [50] |
| 29c | 4-Cl-Ph | 46 | [50] |
| 29e | 4-OMePh | 59 | [50] |
| 29f | 1-Naphtyl | 45 | [50] |
Table 7.
Substituents, conditions and yields in cycloaddition of acetylenes to 3(5)-substituted indolizines giving cyclazines (
Scheme 15).
Table 7.
Substituents, conditions and yields in cycloaddition of acetylenes to 3(5)-substituted indolizines giving cyclazines (
Scheme 15).
| N | R/R’ | R3/R5 | R1 | R2 | R6 | R7 | R8 | Cat/Solv/T°/Time | Yield % | Ref. |
|---|
| 30a | E/H | OCOMe/H | H | H | H | H | H | No/Ac2O/Δ/0.5 h | 86 | [51] |
| 30b | E/H | OCOEt/H | H | H | H | H | H | No/Ac2O/Δ/0.5 h | 100 | [51] |
| 30c | E/H | OCOMe/H | Ph | H | H | H | H | No/Ac2O/Δ/0.5 h | 90 | [51] |
| 30d | E/H | OCOMe/H | CH2E’ | E’ | H | H | H | No/Ac2O/Δ/0.5 h | 100 | [51] |
| 31 | E/E | i-PrS/H | i-PrS | i-PrS | H | H | H | No/PhMe/Δ/40 h | 96 | [52] |
| 32a | E/E | NE’NHE’/H | Ph | H | H | H | H | No/PhH/Δ/5 h | 5.6 | [53] |
| 32b | E/E | NE’NHE’/H | H | H | H | H | H | No/PhH/Δ/5 h | 32 | [53] |
| 32c | E/E | NE’NHE’/H | NE’NHE’ | H | H | H | H | No/PhH/Δ/5 h | 66 | [53] |
| 33a | E/E | H/OTms | H | H | OMe | OMe | OH | No/PhMe/Δ/23 h | 56 | [54] |
| 33b | E/E | H/OTms | H | H | OMe | Ph | OH | No/PhMe/Δ/23 h | 79 | [54] |
| 33c | E/E | H/OTms | H | H | OMe | n-Bu | OH | No/PhMe/Δ/23 h | 52 | [54] |
| 33d | E/E | H/OTms | H | H | OMe | n-BuC≡C | OH | No/PhMe/Δ/23 h | 67 | [54] |
| 33e | E/E | H/OTms | H | H | OMe | PhC≡C | OH | No/PhMe/Δ/23 h | 66 | [54] |
| 33f | E/E | H/OTms | H | H | n-Bu | n-Bu | OH | No/PhMe/Δ/23 h | 53 | [54] |
| 34 | E/E | H/Morph | H | p-NO2Ph | H | H | H | No/PhH/Δ/16 h | 82 | [55] |
| 35 | E/E | H/Br | Me | t-Bu | H | H | H | No/PhMe/80°/2 h | 53 | [56] |
Table 8.
Substituents, conditions and yields in cycloaddition of acetylene acetylenes to 3-CN-substituted indolizines giving cyclazines (
Scheme 16).
Table 8.
Substituents, conditions and yields in cycloaddition of acetylene acetylenes to 3-CN-substituted indolizines giving cyclazines (
Scheme 16).
| N | R/R’ | R1 | R2 | R6 | R7 | R8 | Cat/Solv/T°/Time | Yield % | Ref. |
|---|
| 36a | E/E | H | H | H | H | H | Pd-C/PhH/Δ/24 h | 40 | [58] |
| 36b | E/E | H | H | Me | H | Me | Pd-C/PhH/Δ/24 h | 25 | [58] |
| 37a | E/E | H | MeS | H | H | H | Pd-C/PhH/Δ/30 h | 22 | [59] |
| 37b | E/E | H | MeS | Me | H | Me | Pd-C/PhH/Δ/30 h | 12 | [59] |
| 38a | E/E | H | H | H | H | H | Pd’C/MePh/Δ/24 h | 40 | [60] |
| 38b | E/E | H | H | H | Me | H | Pd’C/MePh/Δ/25–74 h | 10-7 | [60] |
| 38c | E/E | H | H | H | PhCH2 | H | Pd’C/MePh/Δ/30 h | 5 | [60] |
| 38d | E/E | H | H | H | Ph | H | Pd’C/MePh/Δ/215 h | 13 | [60] |
| 38e | E/E | H | H | Me | H | Me | Pd’C/MePh/Δ/77 h | 25 | [60] |
| 38f | E/E | H | H | H | CN | H | Pd’C/MePh/Δ/260 h | 7 | [60] |
| 38g | E/E | H | H | H | E | H | Pd’C/MePh/Δ/336 h | 2 | [60] |
Table 9.
Substituents, conditions and yields in cycloaddition of acetylene to 3-CN-substituted benzoindolizines giving benzocyclazines (
Scheme 18).
Table 9.
Substituents, conditions and yields in cycloaddition of acetylene to 3-CN-substituted benzoindolizines giving benzocyclazines (
Scheme 18).
| N | R/R’ | R7 | Cat/Solv/T°/Time | Yield % | Ref. |
|---|
| 40a | COPh/COPh | H | Pd-C/PhH/rt/46 h | 82 | [62,63] |
| 40b | COPh/COPh | Me | Pd-C/PhH/rt/24 h | 82 | [62] |
| 40c | COPh/COPh | Ph | Pd-C/PhH/rt/24 h | 69 | [62] |
| 40d | COPh/COPh | COPh | Pd-C/PhH/Δ/5.5 h | 59 | [62] |
| 41 | E/E | H | Pd-C/PhMe/Δ/20 h | 54 | [64] |
| 42a | E/E | H | Pd-C/PhMe/Δ/2 h | 13 | [65] |
| 42b | E”/E” a | H | Pd-C/PhMe/Δ/20 h | 14 (R’ = H) | [65] |
| 42c | Ac/Ac | H | Pd-C/PhMe/Δ/20 h | 8 | [65] |
| 42d | E/H | H | Pd-C/PhMe/Δ/34 h | NG | [65] |
| 42e | E/SiMe3 | H | Pd-C/PhMe/Δ/72 h | 11:14 b | [65] |
| 42f | E/Ph | H | Pd-C/PhMe/Δ/72 h | 52:6 b | [65] |
| 42g | Ac/Ph | H | Pd-C/PhMe/Δ/72 h | 48:7 b | [65] |
Table 10.
Substituents and yields of cycloaddition of benzynes to indolizines (
Scheme 19).
Table 10.
Substituents and yields of cycloaddition of benzynes to indolizines (
Scheme 19).
| N | Benzyne | R1 | R2 | R3 | R5 | R6 | R7 | R8 | Yield, % | Ref. |
|---|
| 43a | A | H | Ph | H | H | H | H | H | 23 | [3] |
| 43b | A | Me | Ph | H | H | H | H | H | 49 | [3] |
| 43c | A | H | Ph | H | H | H | H | Me | 44 | [3] |
| 43d | A | H | Ph | H | H | H | Me | H | 55 | [3] |
| 43e | A | CN | H | H | H | H | Me | H | 51 | [3] |
| 43f | A | E | H | H | H | H | H | H | 50 | [3] |
| 43g | A | E | E | H | H | H | H | H | 50 | [3] |
| 43h | A | E’ | E’ | H | H | H | H | H | 37 | [3] |
| 43i | A | E’ | E’ | H | H | H | Me | H | 30 | [3] |
| 43j | A | E | E | CN | H | Me | H | H | 75 | [3] |
| 43k | A | COPh | Ph | H | H | H | Me | H | 51 | [3] |
| 43l | B | Me | Ph | H | H | H | H | H | 40 | [3] |
| 43m | B | H | Ph | H | H | H | H | Me | 18 | [3] |
| 43m | B | H | Ph | H | H | H | Me | H | 42 | [3] |
| 43o | A | E | H | H | H | H | 7,8-Benzo | 60 | [3] |
| 43p | A | CN | H | H | H | H | 7,8-Benzo | 52 | [3] |
| 43q | A | E’ | E’ | H | H | H | 7,8-Benzo | 58 | [3] |
Table 11.
Substituents and yields (ratio of isomers) of cycloaddition to benzoindolizines (
Scheme 21).
Table 11.
Substituents and yields (ratio of isomers) of cycloaddition to benzoindolizines (
Scheme 21).
| N | R1 | R2 | Solv/T°/Time/Oxidant | Yield, % | Ref. |
|---|
| 44a | Mes2B | H | PhMe/rt/30m/DDQ | 89 | [37] |
| 44b | 2-(Mes2B)Ph | H | PhMe/Δ/3d/DDQ | 75 | [37] |
| 44c | 4-(Mes2B)Ph | H | PhMe/Δ/3d/DDQ | 83 | [37] |
| 45a | 4-(Mes2B)Ph | 2-Py | (1) PhMe/120 °C/5–6d; (2) DDQ/rt/0.5 h | 79:2 | [5] |
| 45b | 2-(Mes2B)Ph | 2-Py | (1) PhMe/120 °C/5–6d; (2) DDQ/rt/0.5 h | 82:4 | [5] |
| 45c | 2-(Mes2B)Ph | 2-Isoquinolyl | (1) PhMe/120 °C/5–6d; (2) DDQ/rt/0.5 h | 72:18 | [5] |
| 45d | 2-(Mes2B)Ph | 2-Benzotiazolyl | (1) PhMe/120 °C/5–6d; (2) DDQ/rt/0.5 h | 62:19 | [5] |
| 45e | 2-(Mes2B)Ph | 2-Tiazolyl | (1) PhMe/120 °C/5–6d; (2) DDQ/rt/0.5 h | 68:14 | [5] |
| 46 | Benzo | 2,2,6,6-tetramethylpiperidine, n-BuLi, PhBr/THF/−78 °C/1 h | 6.6 | [66] |
Table 12.
Substituents and yields of microwave three-component synthesis of cyclazines (
Scheme 26).
Table 12.
Substituents and yields of microwave three-component synthesis of cyclazines (
Scheme 26).
| N | R | R’ | R2 | R7 | R8 | Yield, % | Ref. |
|---|
| 51a | E | E | Ph | H | H | 90 | [71] a |
| 51b | E | E | 4-Me-Ph | H | H | 92 | [71] |
| 51c | E | E | 4-Cl-Ph | H | H | 60 | [71] |
| 51d | E | E | 4-NO2-Ph | H | H | 20 | [71] |
| 51e | E | E | Polycyclic R | H | H | 78 | [71] |
| 51f | E | E | 1-Cyclohexenyl | H | H | 74 | [71] |
| 51g * | H | E’ | Ph | H | H | 78 | [71] |
| 51h * | H | E’ | Ph | Me | H | 80 | [71] |
| 51i * | H | E’ | 4-Me-Ph | Me | H | 74 | [71] |
| 51j * | H | E’ | 4-Cl-Ph | H | Me | 65 | [71] |
| 51k * | H | E’ | 4-NO2-Ph | H | Me | 22 | [71] |
| 52a | E | E | Ph | H | H | 37 | [72] b |
| 52b | E | E | 4-NO2Ph | H | H | 78 | [72] |
| 52c | E | E | 4-ClPh | H | H | 46 | [72] |
| 52d | E | E | 4-MeOPh | H | H | 23 | [72] |
| 52e | E | E | 4-MePh | H | H | 39 | [72] |
| 52f | E | E | 4-OHPh | H | H | 36 | [72] |
| 52g | E | E | 4-BrPh | H | H | 49 | [72] |
| 52h | E | E | 4-FPh | H | H | 42 | [72] |
Table 13.
Substituents and yields of benzocyclazines from pyridinium ylide (
Scheme 29).
Table 13.
Substituents and yields of benzocyclazines from pyridinium ylide (
Scheme 29).
| N | R | A, Yield, % | B, Yield, % | C, Yield, % | Ref. |
|---|
| 55a | H | 11 (20) | 4 (35) | 39 | [76] |
| 55b | Me | 5 (22) | 2 (12) | 33 | [76] |
| 55c | Ph | 3 (44) | 2 (10) | 38 | [76] |
| 55d | PhCO | 0.5 (21) | 17 (24) | 25 | [76] |
| 55e | MeOCO | 3 (33) | 32 | - | [76] |
| 55f | MeCO | Trace (27) | 18 | - | [76] |
| 56a | 1,2-Me | 12 | - | - | [77] |
| 56b | 1,3-Me | 5 | - | - | [77] |
Table 14.
Substituents, conditions and yields of azacyclazines from azaindolizines (
Scheme 31).
Table 14.
Substituents, conditions and yields of azacyclazines from azaindolizines (
Scheme 31).
| N | R/R’ | X | R2 | Z | R7 | Y | Cat/Solv/T°/Time | Yield, % | Ref. |
|---|
| 58 | E/E | N | Ph | H | H | H | Pd-C/MePh/Δ/25 h | 29 | [83] |
| 59 | E/E | H | Ph | N | Me | H | Pd-C/MePh/Δ/21 h | 28 | [84] |
| 60a | E/E | H | Me | H | Me | N | Pd-C/MePh/Δ/ | 66 | [85] |
| 60b | E/E | H | Me | H | O= | NMe | Pd-C/MePh/Δ/ | 59 | [85] |
| 61 | E/E | N | SMe | H | (CH=CH)2 | Pd-C/MePh/Δ/30 h | 6 | [88] |
| 62a | E/E | N | SMe | H | H | H | Pd-C/MePh/Δ/30 h | 36 | [87] |
| 62b | E/E | N | SMe | MeC | H | MeC | Pd-C/MePh/Δ/30 h | 40 | [87] |
| 63 | E/H | N | t-Bu | H | H | H | O2/MePh/Δ/4 h | 75 | [36] |
| 64 | Ph/Ph | N | H | H | H | H | Pd(OAc)2/Cu(OAc)2 LiOAc/DMAc/120°/8 h | 49 | [89] |
Table 15.
Substituents and yields of reaction of 2-arylimidazopiridines and diarylacetylenes (
Scheme 33).
Table 15.
Substituents and yields of reaction of 2-arylimidazopiridines and diarylacetylenes (
Scheme 33).
| N | Ar | R2 | R6 | R7 | R8 | Yield, % | Ref. |
|---|
| 66a | Ph | Ph | H | H | H | 78 | [90] |
| 66b | Ph | Ph | H | H | Me | 76 | [90] |
| 66c | Ph | Ph | H | Me | H | 75 | [90] |
| 66d | Ph | Ph | Cl | H | H | 68 | [90] |
| 66e | Ph | 4-MePh | H | H | H | 79 | [90] |
| 66f | Ph | 4-FPh | H | H | H | 75 | [90] |
| 66g | Ph | 4-ClPh | H | H | Me | 73 | [90] |
| 66h | Ph | 4-CNPh | H | H | H | 71 | [90] |
| 66i | Ph | 4-NO2Ph | H | H | H | 67 | [90] |
| 66j | Ph | CF3 | H | H | H | 64 | [90] |
| 66k | Ph | i-Bu | H | H | H | 63 | [90] |
| 66l | 4-MePh | Ph | H | H | H | 77 | [90] |
| 66m | 4-MePh | Ph | H | Me | H | 74 | [90] |
| 66n | 4-MeOPh | Ph | H | H | H | 70 | [90] |
| 66o | 4-MeOPh | Ph | H | Me | H | 69 | [90] |
| 66p | 4-MeOPh | i-Bu | H | H | H | 61 | [90] |
| 67a | Ph | Ph | H | H | H | 68 | [91] |
| 67b | Ph | 1-Naphtyl | H | H | H | 69 | [91] |
| 67c | Ph | Ph | H | Cl | Cl | 57 | [91] |
| 67d | Ph | Ph | H | Br | Br | 51 | [91] |
| 67e | Ph | Ph | Me | H | H | 76 | [91] |
| 67f | Ph | 4-CN-Ph | H | H | H | 64 | [91] |
| 67g | Ph | 4-F-Ph | H | H | H | 68 | [91] |
| 67h | Ph | 4-Br-Ph | H | H | H | 66 | [91] |
| 67i | Ph | 4-MeO-Ph | H | H | H | 73 | [91] |
| 67j | Ph | 4-F-Ph | Me | H | H | 61 | [91] |
| 67k | Ph | 2-Tienyl | H | H | H | 67 | [91] |
| 67l | Ph | Ph | H | H | H | 63 | [91] |
| 67m | 4-Br-Ph | Ph | H | H | H | 78 | [91] |
| 67n | E’ | Ph | H | H | H | 31 | [91] |
Table 16.
Substituents and yields of reaction of 2-arylimidazopiridines and diarylacetylenes (
Scheme 34).
Table 16.
Substituents and yields of reaction of 2-arylimidazopiridines and diarylacetylenes (
Scheme 34).
| N | Ar/Ar | R2 | R8 | Yield, % | Ref. |
|---|
| 69a | Ph | Me | H | 67 | [92] |
| 69b | 4-Me-Ph | Me | H | 71 | [92] |
| 69c | 2-Me-Ph | Me | H | 60 | [92] |
| 69e | 4-MeO-Ph | Me | H | 63 | [92] |
| 69f | 4-F-Ph | Me | H | 68 | [92] |
| 69g | 4-Cl-Ph | Me | H | 68 | [92] |
| 69h | 4-Br-Ph | Me | H | 63 | [92] |
| 69i | Ph | t-Bu | H | 78 | [92] |
| 69j | 4-Me-Ph | t-Bu | H | 75 | [92] |
| 69k | 4-F-Ph | t-Bu | H | 74 | [92] |
| 69l | 4-Cl-Ph | t-Bu | H | 80 | [92] |
| 69m | 4-Br-Ph | t-Bu | H | 66 | [92] |
| 69n | Ph | t-Bu | N | 63 | [92] |
| 69o | 4-Cl-Ph | t-Bu | N | 68 | [92] |
| 69p | Ph | Me | Me | 61 | [92] |
| 69q | 4-F-Ph | Me | Me | 59 | [92] |
| 69r | Ph | Mes | H | 66 | [92] |
| 69s | 4-Cl-Ph | Me | H | 72 | [92] |
Table 17.
Substituents and yields of reaction of imidazopyrimidines and diarylacetylenes (
Scheme 35).
Table 17.
Substituents and yields of reaction of imidazopyrimidines and diarylacetylenes (
Scheme 35).
| N | Ar | R2 | Yield, % | Ref. |
|---|
| 70a | Ph | Ph | 76 | [11] |
| 70b | Ph | 2-MeOPh | 72 | [11] |
| 70c | 4-FPh | 2-MeOPh | 78 | [11] |
| 70d | E | 2-MeOPh | 74 | [11] |
| 70e | E | 4-MeOPh | 70 | [11] |
| 70f | 4-FPh | 3-CNPh | 61 | [11] |
| 70g | Ph | 3-CNPh | 82 | [11] |
| 70h | 2-Pyridyl | 3-CNPh | 65 | [11] |
| 70i | 4-MePh | 4-CNPh | 68 | [11] |
| 70j | Ph | 2-NO2Ph | 64 | [11] |
| 70k | Ph | 4-MeOPh | 74 | [11] |
| 70l | 4-MePh | 4-MeOPh | 65 | [11] |
Table 18.
Substituents and yields of reaction of azaindolizines and benzynes (
Scheme 36).
Table 18.
Substituents and yields of reaction of azaindolizines and benzynes (
Scheme 36).
| N | R2 | X | Y * | Yield, % | Ref. |
|---|
| 71a | 2-HOPh | H | A | 23 | [6] |
| 71b | 2-HO-4-MeOPh | H | A | 21 | [6] |
| 71c | 2-HO-5-FPh | H | A | 19 | [6] |
| 71d | 2-HO-5-MePh | H | A | 21 | [6] |
| 72a | Ph | H | B | 74 | [93] |
| 72b | 4-MeOPh | H | B | 39 | [93] |
| 72c | 4-FPh | H | B | 49 | [93] |
| 72d | 4-NO2Ph | H | B | 51 | [93] |
| 72e | 3,5-(BnO)2Ph | H | B | 49 | [93] |
| 72f | Ph | N | B | 58 | [93] |
| 72g | 4-MeOPh | N | B | 54 | [93] |
| 72h | 4-FPh | N | B | 38 | [93] |
| 72i | 4-MeOPh | H | C | 43 (3’) | [93] |
| 72j | 4-MeOPh | N | C | 34 (3’) | [93] |
| 72k | 4-MeOPh | H | D | 39 (4’) | [93] |
| 72l | 4-MeOPh | H | D | 18 (5’) | [93] |
| 72m | E’ | H | B | 51 | [93] |
Table 19.
Substituents and yields of reaction of imidazopiridines and benzynes (
Scheme 37).
Table 19.
Substituents and yields of reaction of imidazopiridines and benzynes (
Scheme 37).
| N | R2 | R6 | R7 | R8 | Method * | Reagent | Yield, % | Ref. |
|---|
| 73a | Ph | H | H | H | A | [Benzyne] | 75 | [94] |
| 73b | 4-MeO-Ph | H | H | H | A | [Benzyne] | 72 | [94] |
| 73c | 4-Et-Ph | H | H | H | A | [Benzyne] | 81 | [94] |
| 73d | 4-F-Ph | H | H | H | A | [Benzyne] | 69 | [94] |
| 73e | 4-CN-Ph | H | H | H | A | [Benzyne] | 82 | [94] |
| 73f | 2-F-Ph | H | H | H | A | [Benzyne] | 70 | [94] |
| 73g | 2-Cl-Ph | H | H | H | A | [Benzyne] | 66 | [94] |
| 73h | 2-Me-Ph | H | H | H | A | [Benzyne] | 71 | [94] |
| 73i | 2-Br-Ph * | H | H | H | A | [Benzyne] | 55 (de-brom) | [94] |
| 73j | 4-Cl-Ph | Me | H | H | A | [Benzyne] | 83 | [94] |
| 73k | 2-Cl-Ph | H | H | Me | A | [Benzyne] | 63 | [94] |
| 73l | Ph | Me | H | H | A | [Benzyne] | 71 | [94] |
| 73m | 4-Et-Ph | H | H | Me | A | [Benzyne] | 65 | [94] |
| 73n | 4-Et-Ph | Me | H | H | A | [Benzyne] | 60 | [94] |
| 73o | 4-MeO-Ph | Me | H | H | A | [Benzyne] | 72 | [94] |
| 73p | 2-F-Ph | H | H | Me | A | [Benzyne] | 60 | [94] |
| 73q | Ph | H | H | Me | A | [Benzyne] | 75 | [94] |
| 73r | 4-Cl-Ph | H | Me | H | A | [Benzyne] | 32 | [94] |
| 73s | 4-Et-Ph | Br * | H | H | A | [Benzyne] | 54 (de-brom) | [94] |
| 73t | H | H | CO2Me | H | A | [Benzyne] | 50 | [94] |
| 73u | H | Cl | H | H | A | [Benzyne] | 78 | [94] |
| 73v | H | H | H | H | A | [Benzyne] | 45 | [94] |
| 73x | H | H | H | H | B | [1-MeO-benzyne-2] | 70 | [94] |
| 73y | H | H | H | H | C | [1-Me-benzyne-3] | 65 (2 isom.) | [94] |
| 73 | 4-Et-Ph | H | H | H | C | [1-Me-benzyne-3] | 72 (2 isom.) | [94] |
| 73z | 4-Cl-Ph | H | H | H | C | [1-Me-benzyne-3] | 69 (2 isom.) | [94] |
| 73a1 | 4-CN-Ph | H | H | H | D | [1-MeO-benzyne-2] | 80 (3’) | [94] |
| 73b1 | 4-CN-Ph | H | H | H | E | [1-Me-benzyne-3] | 75 (2 isom.) | [94] |
| 73c1 | 4-Et-Ph | H | H | H | F | [1-MeO-benzyne-2] | 73 | [94] |
| 73d1 | t-Bu | H | H | H | G | [Benzyne] | 62 | [94] |
Table 20.
Substituents and yields of reaction of imidazopyridines and 1,2-dihalobenzenes (
Scheme 38).
Table 20.
Substituents and yields of reaction of imidazopyridines and 1,2-dihalobenzenes (
Scheme 38).
| N | R2 | R6 | R7 | R8 | Yield, % | Benzene * | Ref |
|---|
| 74a | Ph | H | H | H | 77 | A | [95] |
| 74b | 4-Me-Ph | H | H | H | 79 | A | [95] |
| 74c | 4-MeOPh | H | H | H | 54 | A | [95] |
| 74d | 4-ClPh | H | H | H | 45 | A | [95] |
| 74e | 4-CF3Ph | H | H | H | 74 | A | [95] |
| 74f | 3-MeOPh | H | H | H | 83 | A | [95] |
| 74g | 2-MePh | H | H | H | 60 | A | [95] |
| 74h | 2-Naphtyl | H | H | H | 52 | A | [95] |
| 74i | Ph | H | H | Me | 83 | A | [95] |
| 74j | Ph | H | Me | H | 87 | A | [95] |
| 74k | Ph | H | Cl | H | 27 | A | [95] |
| 74l | Ph | Me | H | H | 73 | A | [95] |
| 74m | Ph | H | H | N | 95 | A | [95] |
| 74n | Ph | H | H | H | 80 | B | [95] |
| 74o | Ph | H | H | H | 69 | C | [95] |
| 74p | Ph | H | H | H | 82 | D | [95] |
| 74q | Ph | H | H | H | 86 | E | [95] |
Table 21.
Substituents and yields of the ring closure reaction on
Scheme 45.
Table 21.
Substituents and yields of the ring closure reaction on
Scheme 45.
| N | Ar | R8 | Yield, % | Ref. |
|---|
| 81a | Ph | H | 100 | [8] **, [101] * |
| 81b | 4-(Ph)-Ph | H | 100 | [8] |
| 81c | 4-t-Bu-Ph | H | 89 | [8] |
| 81d | 4-BnO-Ph | H | 90 | [8] |
| 81e | 4-(O(CH2)2OBz)-Ph | H | 89 | [8] |
| 81f | 4-NMe2-Ph | H | 98 | [8] |
| 81g | 4-F-Ph | H | 98 | [8] |
| 81i | 4-Cl-Ph | H | 94 | [8] |
| 81j | 4-CO2Me-Ph | H | 95 | [8] |
| 81k | 4-CN-Ph | H | 99 | [8] |
| 81l | 3,4,5-(Me)3-Ph | H | 93 | [8] |
| 81m | 2-EtO-4-NO2-Ph | H | 92 | [8] |
| 81n | 2,3,4,5-(F)4-Ph | H | 66 | [8] |
| 81o | 2-MeO-Ph | H | 98 | [8] |
| 81p | 1-Naphtyl | H | 97 | [8] |
| 81q | 2-Fluorenonyl | H | 61 | [8] |
| 81r | 2-Furyl | H | 40 | [8] |
| 81s | 2-Tienyl | H | 65 | [8] |
| 81t | 2-Propenyl | H | 87 | [8] |
| 81u | 2-Styryl | H | 28 | [8] |
Table 22.
Substituents, yields and ratio of isomers in concurrent 2+4 and 2+8 cycloadditions of 2-arylazaindolizines and acetylenes (
Scheme 48).
Table 22.
Substituents, yields and ratio of isomers in concurrent 2+4 and 2+8 cycloadditions of 2-arylazaindolizines and acetylenes (
Scheme 48).
| N | Ar | Ar | R2 | R5 | R6 | R7 | R8 | Yield, % (4+2/8+2) | Ref. |
|---|
| 84a | Ph | Ph | H | H | H | H | H | 85 (82/18) | [104] a |
| 84b | 4-FPh | 4-FPh | H | H | H | H | H | 79 (68/32) | [104] |
| 84c | 4-Me-Ph | 4-Me-Ph | H | H | H | H | H | 89 (60/40) | [104] |
| 84d | Ph | Ph | H | H | H | H | Me | 88 (46/54) | [104] |
| 84e | Ph | Ph | H | Me | H | H | H | 74 (100/0) | [104] |
| 85a | Ph | Ph | H | H | H | H | H | 65/18 | [105] b |
| 85b | Ph | Ph | 4-Me | H | H | H | H | 72/15 | [105] |
| 85c | Ph | Ph | 4-MeO | H | H | H | H | 75/13 | [105] |
| 85d | Ph | Ph | 4-F | H | H | H | H | 50/33 | [105] |
| 85e | Ph | Ph | 4-CF3 | H | H | H | H | 39/42 | [105] |
| 85f | Ph | Ph | 2-Me | H | H | H | H | 40/45 | [105] |
| 85g | Ph | Ph | 2-Tienyl | H | H | H | H | 80/9 | [105] |
| 85h | Ph | Ph | 1-Naphtyl | H | H | H | H | 43/35 | [105] |
| 85i | Ph | Ph | H | H | H | Me | H | 55/29 | [105] |
| 85j | Ph | Ph | H | H | H | H | Me | 60/26 | [105] |
| 85k | Ph | Ph | H | H | Cl | H | H | 69/7 | [105] |
| 85l | Ph | Ph | H | H | CF3 | H | H | 77/0 | [105] |
| 85m | 4-MeO-Ph | 4-MeO-Ph | H | H | H | H | H | 61/16 | [105] |
| 85n | 4-Cl-Ph | 4-Cl-Ph | H | H | H | H | H | 60/20 | [105] |
| 85o | 4-CF3-Ph | 4-CF3-Ph | H | H | H | H | H | 63/15 | [105] |
| 85p | 2-Me-Ph | 2-Me-Ph | Ph | H | H | H | H | 66/20 | [105] |
| 85q | E’ | E’ | H | H | H | H | H | 51/14 | [105] |
| 85r | Ph | Me | Ph | H | H | H | H | 56/14 | [105] |
| 85s | Ph | n-Pr | Ph | H | H | H | H | 47/16 | [105] |
| 85t | Ph | Ph | Ph | Me | H | H | H | 60/0 | [105] |
| 85u | Ph | Ph | 4-Me | Me | H | H | H | 65/0 | [105] |
| 85v | Ph | Ph | 4-CF3 | Me | H | H | H | 51/0 | [105] |
| 85w | Ph | Ph | 4-F | Me | H | H | H | 56/0 | [105] |
| 85x | Ph | Ph | 2-Me | Me | H | H | H | 52/0 | [105] |
| 85y | Ph | Ph | 3-Me | Me | H | H | H | 68/0 2 isomers | [105] |
| 85z | Ph | Ph | Mes | H | H | H | H | 0/77 | [105] |
| 85a1 | Ph | Ph | Mes | H | H | H | Me | 0/81 | [105] |
| 85b1 | Ph | Ph | Mes | H | H | Me | H | 0/79 | [105] |
| 85c1 | Ph | Ph | Mes | H | Me | H | H | 0/77 | [105] |
| 85d1 | Ph | Ph | Mes | H | H | MeO | H | 0/86 | [105] |
| 85e1 | Ph | Ph | Mes | H | Cl | H | H | 0/65 | [105] |
| 86a | 4-BrPh | 4-BrPh | Ph | H | H | H | H | 22/27 | [7] c |
| 86b | 4-BrPh | 4-BrPh | 4-CNPh | H | H | H | H | 22/35 | [7] c |
Table 23.
Possible mechanisms of [8+2] cycloaddition depending on the nature of groups in alkene and indolizne as shown on
Scheme 49.
Table 23.
Possible mechanisms of [8+2] cycloaddition depending on the nature of groups in alkene and indolizne as shown on
Scheme 49.
| Substituent in Alkene | Indolizine | 6-Nitroindolizine |
|---|
| Nitroethylene | (i), (ii) | (i), (ii) |
| Methyl acrylate | (iii) | (iii) |
| Acrylonitrile | (iii) | (iii) |
| Ethylene | (iii) | (iii) |
| N,N-Dimethylaminoethylene | (iii) | (iv), (v) |
Table 24.
Stereochemistry, conditions and yields of synthesis of vinyl indolisines on
Scheme 52.
Table 24.
Stereochemistry, conditions and yields of synthesis of vinyl indolisines on
Scheme 52.
| N | R | Yield (trans/cis), % | Conditions | Ref. |
|---|
| 89a | R = H | 13:4 | No/Me2CO/rt/15 h | [108] |
| 89b | R6 = Me | 11:5 | | [108] |
| 89c | R7 = Me | 16:0 | | [108] |
| 89d | R8 = Me | 20:0 | | [108] |
| 90 | | 18:13 | Pd/PhMe/Δ/21 h | [85] |
| 91a | n = 1 | 62 * | No/0°/THF | [46] |
| 91b | n = 2 | 62 * | No/0°/THF | [46] |
| 92a | R2 = SMe | 26:17 | Pd-C/PhMe/Δ/30 h | [29] |
| 92b | R2 = H | 0:11 | Pd-C/PhMe/Δ/30 h | [29] |
| 93 | | 4 * | Pd-C/PhMe/Δ/30 h | [88] |
| 94 | | 14 ** | No/PhMe/rt/120 h | [109] |