Generation of Radicals.
Considering the structural complexity and chemical sensitivity of the potential liquid crystalline molecules, we focused on oxidation of 1,2-thiazines as precursors for radical generation, rather than reduction of the appropriate sulfiminyl chlorides (
Figure 2). The 1,2-thiazines are generally stable, they can be prepared in a regiospecific manner and their purification is straightforward [
6,
8,
10]. In contrast, the sulfiminyl chlorides and salts are known to be reactive, highly moisture sensitive and many are strong oxidizing reagents, which may be incompatible with some functional groups [
11,
12]
Figure 2.
Two general methods to generate the thioaminyl radicals.
Figure 2.
Two general methods to generate the thioaminyl radicals.
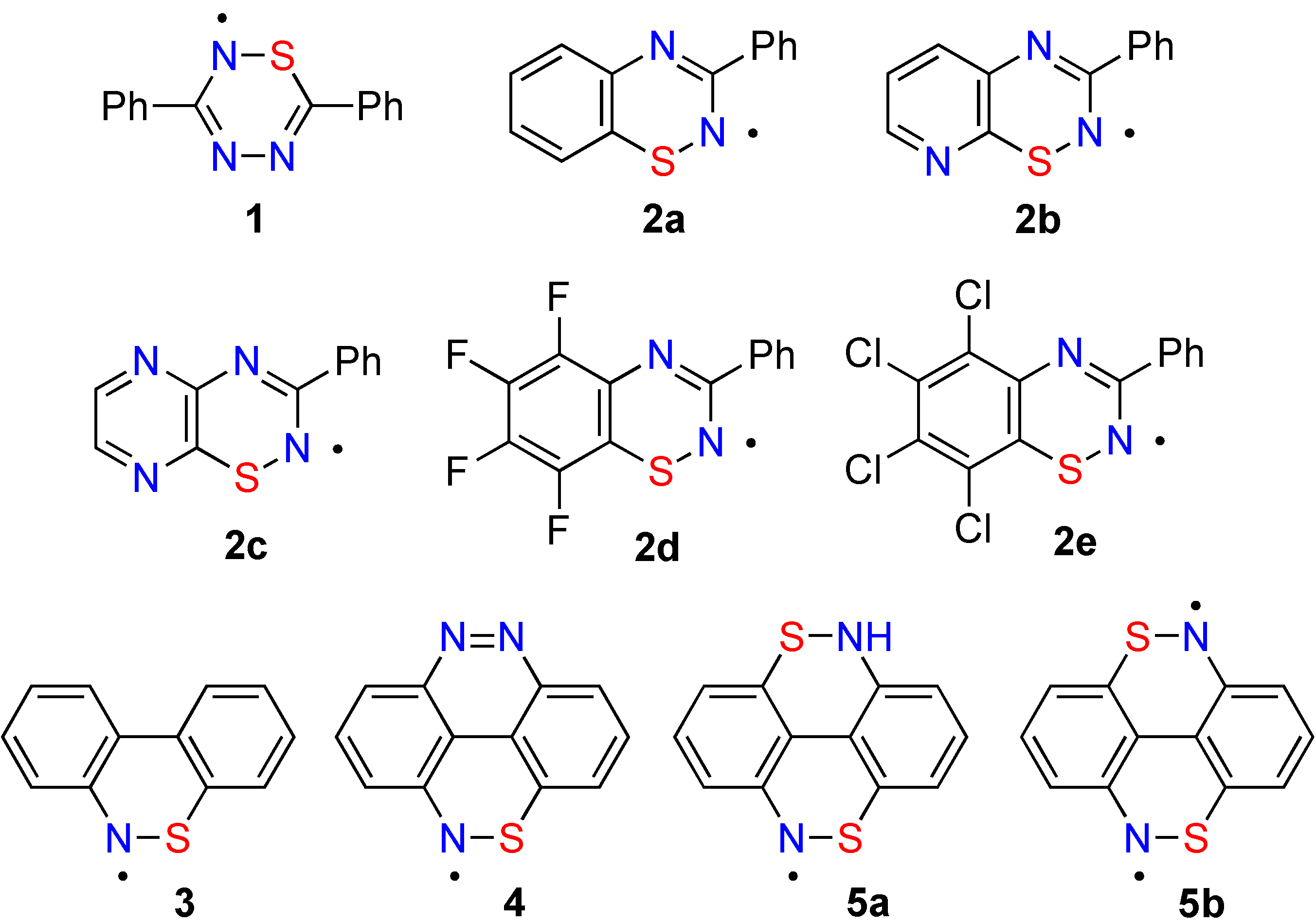
Thus far, we have synthesized nine 1,2-thiazines as precursors to five classes heterocyclic radicals (
A,
B,
E,
F, and
H) shown in
Chart I [
6,
8,
10,
13].
Initially, we used rigorously dried PbO
2/K
2CO
3 in benzene [
14] to generate radicals from the corresponding sulfenamides. Under these conditions, several radicals were formed in respectable yields of about 30-40% (
Table 1). However, the reactions were incomplete, which complicated attempts to isolate and purify the radicals. The pyrazino derivative
2c and halogenated radicals
2d and
2e were formed only in low yields, impractical for preparative work. Attempts to generate
1 or
5b by this method were frustratingly unsuccessful [
6,
13].
Figure 3.
A plot of log(% yield) vs Homolytic Bond Dissociation Energy (HBDE) for selected radicals generated with PbO2/K2CO3 in benzene (circles) and AgO/K2CO3 in MeCN/toluene (red diamonds). The best fit line for 2 (circles): log(%yield) = 15.3-0.18×HBDE (R2 = 0.97).
Figure 3.
A plot of log(% yield) vs Homolytic Bond Dissociation Energy (HBDE) for selected radicals generated with PbO2/K2CO3 in benzene (circles) and AgO/K2CO3 in MeCN/toluene (red diamonds). The best fit line for 2 (circles): log(%yield) = 15.3-0.18×HBDE (R2 = 0.97).
The efficiency of radical generation under these conditions would appear to depend upon the N-H homolytic bond dissociation energy (HBDE) of the precursor (
Table 1 and
Figure 3). The highest yields of about 30% were obtained for
2b-H,
3-H,
4-H and
5a-H, which have calculated HBDE’s <78 kcal/mol. For compounds with higher HBDE values, the yield of radicals diminishes to 0 at about 85 kcal/mol (see
Table 1).
Table 1.
Calculated N-H Homolytic Bond Dissociation Energies (HBDE) and yields of radicals for selected compounds relative to methanol.
An analysis of the data for a series of systematically investigated radicals
2 [
15], shows a strong correlation between HBDE and log (%yield) (
Figure 3). The formation of biradical
5b by oxidation of
5a-H could not be detected spectroscopically. The biradical
5b is predicted to have a singlet ground state, and therefore is ESR silent [
1]. Since the concentration of the monoradical
5a remained approximately constant regardless of the amount of PbO
2, the formation of
5b could not be inferred. The results for
3-5 suggest that the yields of radicals generated using the PbO
2 method may have an upper limit of about 40%. This is consistent with results for acyclic sulfenamides, which typically give radicals in about 30-40% yield [
16]. Thus, it appears that the HBDE can serve as an indicator of the efficiency for generation of radicals under these conditions.
Our recent systematic studies on the thiadiazine series
2 demonstrated that the strength of the oxidizing reagent and the polarity of the solvent play important roles in the efficiency of radical generation [
15]. The results show that AgO/K
2CO
3 in either MeCN or a MeCN/toluene mixture is the most effective method for radical generation [
17]. Under these conditions, radicals
2a,
2d and
2e were generated in yields > 90% in less than 2 min (
Figure 3). Also, the thiatriazinyl radical
1 was generated in about 10% spin yield [
18]. This method holds much promise for the preparation of other radicals such as
3-5 in high yields and perhaps isolation as pure solids.
Stability of Radicals.
Thioaminyl radicals
1-4 are persistent and exhibit moderate to low sensitivity to oxygen. Solutions of
3 and
4 show significant stability to manipulation in air and quick filtration through Florisil. This is remarkable, considering that Ph-Ṅ-S-Ph, the acyclic analog of
3, decomposes completely within 1 hr, [
19] and requires additional substituents to enhance stability [
20].
Detailed studies of thiadiazinyls
2 showed that their stability increases in the order
2c <
2a <
2b <
2e <
2d [
15]. Remarkable is the fact that the tetrafluoro derivative
2d is more stable than the tetrachloro analog
2e, which demonstrates the strong stabilizing effect of fluorine atoms on π-delocalized radicals. The kinetic data shows that
2d has a half-life in solution of about 4 months in the absence of air, while in the presence of oxygen
2e quickly decomposes with a half-life of about 40 min. The main decomposition product for
2 has been identified as the
S-oxide
6.
Among the three radicals that were generated in high yields, only
2d and
2e were sufficiently stable for chromatographic isolation and vacuum sublimation. This allowed a detailed investigation of their structural, spectroscopic, and electrochemical properties [
15].
Solid State Structures.
So far only two radicals have isolated in the solid form suitable for X-ray crystallographic studies. Single crystal analysis revealed that
2d and
2e are nearly planar molecules, which is in agreement with DFT results for all radicals
2 [
15]. The molecules of the tetrafluoro derivative
2d are arranged in ideally parallel interpenetrating mirror-imaged stacks rotated by 166
o (
Figure 4). The stacking appears to be driven by SOMO-SOMO interactions largely localized on the thiadiazinyl rings. Surprisingly, the crystal structure shows little Peierls-type distortion or strong dimeric interactions typically observed for dithiadiazolyls [
12]. Within each column, the S-N bonds form an infinite ladder-type structure with the ladder “rungs” slipped by 0.37 Å. The intra-column S···N distance alternates between 3.193 Å and 3.212 Å, and the inter-column non-bonding S···N distance is about 3.27 Å. These separations correspond to 93% - 95% of the van der Waals separation for the S and N atoms.
Figure 4.
Partial packing diagrams for 2d and 2e viewed along the c axis (2d) and along the a axis (2e).
Figure 4.
Partial packing diagrams for 2d and 2e viewed along the c axis (2d) and along the a axis (2e).
The packing of the tetrachloro analog
2e within the crystal structure appears to be governed by quadrupolar interactions between the chlorinated and non-chlorinated benzene rings at the expense of the overlap between the thiadiazinyls (
Figure 4). This is presumably due to the relatively large size of chlorine atoms, which do not allow for close face-to-face contact. The intermolecular separation within a column is about 3.45 Å, which is about 0.25 Å greater than that in
2d. The S
…S nonbonding distance for molecules in neighboring columns is about 3.4 Å, which corresponds to 94% of the sum of the van der Waals radii.
ESR analysis of the sublimed solid sample of
2d revealed less than 1% of spin concentration at ambient temperature. This presumably results from the dominant antiferromagnetic spin alignment in the solid structure, which is consistent with theoretical analysis. Single point calculations at the B3LYP/6-31+G(d) level for a pair of radicals (overlapping radicals
2d in
Figure 4) at their experimental atomic coordinates show a 5.2 kcal/mol preference for the singlet state over the triplet. Detailed temperature dependence studies will be necessary to better understand the solid state magnetic properties of this radical.
Towards Liquid Crystalline Radicals.
Results obtained for the parent radicals were encouraging and indicated that
2d,
3 and
4 are sufficiently stable to be used as structural elements for liquid crystals. So far we have attempted the preparation of two such derivatives: the potentially discotic radical
7 and potentially calamitic radical
8. The cyclic sulfenamide
7-H exhibits columnar properties with a clearing temperature of 105
oC. This suggests that the radical
7 will also exhibit the desired mesogenic properties. Oxidation of
7-H with the PbO
2/K
2CO
3 method in benzene gave moderate yields of the corresponding persistent radical
7 [
21]. The use of the SO
2Cl
2/pyridine method [
15] allowed the generation of
7 in high yield and isolation in the pure form. The dark-green material appeared to exhibit liquid crystalline behavior at ambient temperature. Unfortunately, detailed studies were hampered by its relatively fast decomposition in the presence of air, presumably accelerated by the presence of pyridinium salt impurities.
More recently, we prepared thiadiazines
8a-H and
8b-H and found that the latter exhibits smectic behavior [
22]. Thiadiazine
8a-H was oxidized to the corresponding radical
8a using the SO
2Cl
2/pyridine method [
15]. The crude radical was isolated as an 80% pure dark-green solid by passing the reaction mixture through Florisil and removing the solvent. The solid melted in air with rapid decomposition at about 78
oC. Again, the presumed presence of pyridinium salt impurities in the crude solid could lower the stability of the radical.
We are currently reinvestigating the preparation of 7 and 8 using the more effective AgO/K2CO3/MeCN method.



 2a-Hc
2a-Hc 1-Hd
1-Hd 2b-Hc
2b-Hc 3-He
3-He 2c-Hc
2c-Hc 4-He
4-He 2d-Hc
2d-Hc 5a-Hf
5a-Hf 2e-Hc
2e-Hc 5af
5af







