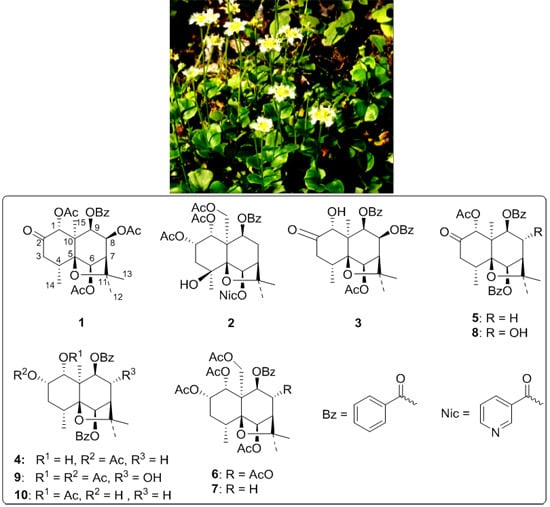
2. Results and Discussion
The ethyl acetate extract from the whole plants of
P. wightiana was subjected to column chromatography on silica gel, preparative HPLC (High Performance Liquid Chromatography) and preparative silica gel TLC (Thin Layer Chromatography), affording four new dihydro-β-agarofuran sesquiterpenes (
1–
4), along with six known compounds (
5–
10) previously isolated from the same plant (
Figure 1) [
1].
Figure 1.
Structures of compounds 1–10 isolated from P. wightiana.
Figure 1.
Structures of compounds 1–10 isolated from P. wightiana.
Compound
1 was obtained as a pale yellow solid with [α]
D19 = −8.661° (
c = 0.26, CH
3OH). The pseudo-molecular ions at
m/
z: 553.2035 [M + Na]
+ in HR-ESI-MS (
Figure S6) in conjunction with an analysis of
1H- and
13C-NMR spectra (
Figures S1 and S2) afforded a molecular formula of C
28H
34O
10. The IR showed the presence of carbonyl (1754, 1734 cm
−1) and aromatic groups (3063, 1602 cm
−1). The UV spectrum showed the presence of benzoate moieties with maximum absorption peaks at 204, 232, 274 and 282 nm.
The
1H- and
13C-NMR spectroscopic data of compound
1 (
Table 1 and
Table 2) showed the structural characteristics of sesquiterpene polyol esters with a dihydro-β-agarofuran skeleton found in the same plant [
1]. The
1H-NMR displayed the presence of three acetyl groups (δ
H 1.60 (s, 3H), 1.78 (s, 3H), 2.05 (s, 3H)), three tertiary methyl groups (δ
H 1.22 (s, 3H), 1.45 (s, 3H), 1.58 (s, 3H)), one secondary methyl group (δ
H 0.90 (d, 7.6 Hz, 3H)) and one benzoate group (δ
H 7.39–7.92, 5H). Additionally, the
13C-NMR and DEPT (Distortionless Enhancement by Polarization Transfer) spectra revealed that
1 also contained one methylene group (δ
C 43.1 (CH
2)), six methine groups with four linked to an oxygen atom (δ
C 38.1 (CH), 53.3 (CH), 69.0 (CH–O–), 72.6 (CH–O–), 76.0 (CH–O–), 77.2 (CH–O–)), three quaternary sp
3 carbons with two bearing an oxygen atom (δ
C 54.0 (C), 84.0 (C–O–), 88.7(C–O–)), one ketone carbonyl group (δ
C 204.8) and four ester carbonyl groups (δ
C 165.4, 169.3, 169.5, 169.8). The complete assignments of all carbon and hydrogen signals were carried out by an analysis of
1H-
1H COSY (
1H-
1H Correlation Spectroscopy), HSQC (Heteronuclear Single Quantum Coherence) (
Figure S3), HMBC (Heteronuclear Multiple Bond Correlation) (
Figure S4) and DEPT spectra (
Table 1). The signals observed at δ
H 5.16 (d,
J = 6.4 Hz), 5.35 (s), 5.53 (dd,
J = 3.2, 6.4 Hz) and 5.82 (s) were assigned to H-9, H-6, H-8 and H-1, respectively.
Table 1.
1H-NMR data of compounds 1–4 (500 MHz; 1 and 2: CD3OD; 3 and 4: CD3Cl).
Table 1.
1H-NMR data of compounds 1–4 (500 MHz; 1 and 2: CD3OD; 3 and 4: CD3Cl).
| No. | 1, δH,
J in Hz | 2, δH,
J in Hz | 3, δH,
J in Hz | 4, δH,
J in Hz |
|---|
| 1 | 5.82, s | 5.54, d, 3.1 | 5.19, d, 4.5 | 4.62, dd, 4.0, 5.4 |
| 1-OH | – | – | 3.19, d, 4.5 | 1.82, d, 5.5 |
| 2 | – | 5.41, m | – | 5.34, q-like, 3.4 |
| 3eq | 2.10, dd, 1.6, 12.9 | 1.90, dd, 2.5, 15.0, | 2.37, br d, 13.1 | 1.89, br d, 13.0 |
| 3ax | 3.15, dd, 7.3, 12.9 | 2.22, m | 3.24, dd, 7.6, 13.1 | 2.41, 4 × d, 3.4, 6.2, 13.0 |
| 4 | 2.73, m | – | 2.86, m | 2.51, m |
| 6 | 5.35, s | 6.30, s | 5.49, s | 5.67, s |
| 7 | 2.56, d, 3.2 | 2.33, br s | 2.78, d, 2.9 | 2.42, d, 3.2 |
| 8eq | – | 2.25, m | – | 2.29, dd, 3.2, 16.4 |
| 8ax | 5.53, dd, 3.2, 6.4 | 2.59, 4 × d, 3.5, 7.0, 16.0 | 5.84, dd, 3.2, 6.6 | 2.58, 4 × d, 3.2, 7.0, 16.4 |
| 9 | 5.16, d, 6.4 | 5.42, d, 7.0 | 5.44, d, 6.6 | 5.08, d, 7.0 |
| 12 | 1.45, s | 1.49, s | 1.52, s | 1.46, s |
| 13 | 1.58, s | 1.53, s | 1.82, s | 1.50, s |
| 14 | 0.90, d, 7.6 | 1.41, s | 1.01, d, 7.6 | 1.21, d, 7.7 |
| 15 | 1.22, s | 4.99, d, 13.0; 4.35, d, 13.0 | 1.22, s | 1.42, s |
| AcO-1 | 1.60, s | 1.47, s | – | – |
| AcO-2 | – | 1.99, s | – | 2.02, s |
| AcO-6 | 2.05, s | – | 2.15, s | – |
| AcO-8 | 1.78, s | – | – | – |
| AcO-15 | – | 2.22, s | – | – |
| 2'/6' | 7.92, d, 8.4 | 7.94, d, 7.8 | 8.04, d, 7.5 | 8.09, d, 8.4 |
| 3'/5' | 7.39, t, 7.9 | 7.39, t, 7.7 | 7.44, t, 7.5 | 7.46, t, 7.6 |
| 4' | 7.54, t, 7.4 | 7.54, t, 7.4 | 7.58, t, 7.5 | 7.57, t, 7.4 |
| 2''/6'' | – | 9.23, s (2''-H); 8.47, d, 8.0 (6''-H) | 7.78, d, 7.5 | 8.07, d, 8.4 |
| 3''/5'' | – | 7.50, dd, 2.6, 8.0 (5''-H) | 7.26, t, 7.8 | 7.50, d, 7.8 |
| 4'' | – | 8.68, br s (4''-H) | 7.47, t, 7.5 | 7.61, t, 7.4 |
Table 2.
13C-NMR and DEPT (Distortionless Enhancement by Polarization Transfer) data of compounds 1–4 (125 MHz; 1 and 2: CD3OD; 3 and 4: CD3Cl).
Table 2.
13C-NMR and DEPT (Distortionless Enhancement by Polarization Transfer) data of compounds 1–4 (125 MHz; 1 and 2: CD3OD; 3 and 4: CD3Cl).
| No. | 1 | 2 | 3 | 4 |
|---|
| 1 | 77.2, CH | 70.9, CH | 75.1, CH | 68.9, CH |
| 2 | 204.8, C | 69.1, CH | 211.5, C | 73.9, CH |
| 3 | 43.1, CH2 | 41.2, CH2 | 42.8, CH2 | 31.2, CH2 |
| 4 | 38.1, CH | 69.7, C | 38.4, CH | 34.1, CH |
| 5 | 88.7, C | 90.8, C | 88.7, C | 90.0, C |
| 6 | 76.0, CH | 79.2, CH | 77.3, CH | 80.2, CH |
| 7 | 53.3, CH | 49.1, CH | 53.7, CH | 49.1, CH |
| 8 | 69.0, CH | 34.3, CH2 | 69.9, CH | 31.6, CH2 |
| 9 | 72.6, CH | 68.0, CH | 72.8, CH | 73.4, CH2 |
| 10 | 54.0, C | 55.3, C | 56.3, C | 51.6, C |
| 11 | 84.0, C | 84.5, C | 83.8, C | 82.8, C |
| 12 | 30.2, CH3 | 24.7, CH3 | 31.2, CH3 | 30.9, CH3 |
| 13 | 25.7, CH3 | 28.2, CH3 | 26.8, CH3 | 26.1, CH3 |
| 14 | 16.9, CH3 | 24.4, CH3 | 18.4, CH3 | 18.8, CH3 |
| 15 | 18.58 *, CH3 | 65.2, CH2 | 18.9, CH3 | 19.2, CH3 |
| AcO-1 | 18.62 *, CH3; 169.5, C | 19.1, CH3; 169.6, C | – | – |
| AcO-2 | – | 19.6, CH3; 170.0, C | – | 21.5, CH3; 171.5, C |
| AcO-6 | 19.6, CH3; 169.8, CH3 | – | 21.2, CH3; 169.4, C | – |
| AcO-8 | 19.2, CH3; 169.3, C | – | – | – |
| AcO-15 | – | 19.8, CH3; 170.7, C | – | – |
| 1' | 128.9, C | 129.1, C | 130.9, C | – |
| 2'/6' | 129.6, CH | 129.8, CH | 129.8, CH | – |
| 3'/5' | 128.3, CH | 128.1, CH | 128.5, CH | – |
| 4' | 133.5, CH | 133.3, CH | 133.14, CH | – |
| 1'' | – | 126.4, C | 130.1, C | – |
| 2''/6'' | – | 150.5, CH (2''); 138.0, CH (6'') | 129.7, CH | – |
| 3''/5'' | – | 124.0, CH (5'') | 128.3, CH | – |
| 4'' | – | 153.0, CH | 133.11, CH | – |
| CO2-6 | – | 164.4, C | – | 165.6, C |
| CO2-8 | – | – | 164.8, C | – |
| CO2-9 | 165.4, C | 165.1, C | 165.2, C | 165.7, C |
The location sites of the ester groups and ketone carbonyl group were determined by an HMBC experiment (
Figure 2). The correlations between δ
H 5.82 (s, H-1)/δ
C 169.5 (CH
3CO
2–), δ
H 5.35 (s, H-6)/δ
C 169.8 (CH
3CO
2–), δ
H 5.53 (dd,
J = 3.2, 6.4 Hz, H-8)/δ
C 169.3 (CH
3CO
2–), and δ
H 5.16 (d,
J = 6.4 Hz, H-9)/δ
C 165.4 (PhCO
2–) showed that three acetoxy groups and one benzoyloxy group were at C-1, C-6, C-8 and C-9, respectively. The correlation between δ
H 5.82 (s, H-1) and δ
C 204.6 (C=O) as well as the downfield chemical shift and singlet of H-1 showed that position 2 was a ketone carbonyl group.
The stereochemistry of compound
1 was determined by a NOESY (Nuclear Overhauser Enhancement Spectroscopy) spectrum (
Figure S5). In the NOESY spectrum (
Figure 3), the correlations of δ
H 5.82 (s, H-1) to 3.15 (dd,
J = 7.3, 12.9 Hz, H-3
ax), 1.58 (s, H-13) to 7.92 (d,
J = 8.4 Hz, H-2'), δ
H 5.35 (s, H-6) to 0.90 (d,
J = 7.6 Hz, H-14), 5.53 (dd,
J = 3.2, 6.4 Hz, H-8) to 1.22 (s, H-15), and δ
H 5.16 (d,
J = 6.4 Hz, H-9) to 5.53 (dd,
J = 3.2, 6.4 Hz, H-8) and 1.22 (s, H-15) showed that three acetoxy groups at C-1, C-6 and C-8 were equatorial and the 9-benzoyloxy group was axial. It is worth noting that 8-OH or 8-AcO in the previously isolated compounds from the same plant are axial [
1], whereas the 8-AcO of compound
1 is located at an equatorial bond.
Figure 2.
Main 1H-13C long-range correlation (⇀) and 1H-1H correlation (—) signals in the HMBC (Heteronuclear Multiple Bond Correlation) and COSY (1H-1H Correlation Spectroscopy) spectra of 1–4.
Figure 2.
Main 1H-13C long-range correlation (⇀) and 1H-1H correlation (—) signals in the HMBC (Heteronuclear Multiple Bond Correlation) and COSY (1H-1H Correlation Spectroscopy) spectra of 1–4.
Figure 3.
Main NOE (Nuclear Overhauser Effect) correlation signals (↔) in the NOESY (Nuclear Overhauser Enhancement Spectroscopy) spectra of 1–4.
Figure 3.
Main NOE (Nuclear Overhauser Effect) correlation signals (↔) in the NOESY (Nuclear Overhauser Enhancement Spectroscopy) spectra of 1–4.
In dihydro-β-agarofuran sesquiterpene polyesters isolated from the same plant, the two six-membered rings are in
trans configuration while both H-1 and H-6 are axial [
1], which are also the general characteristics of natural dihydro-β-agarofuran sesquiterpenes [
4,
5]. Based on the facts above in conjunction with the above-mentioned stereochemistry analysis and biosynthesis point of view, compound
1 is identified as (1
R,4
R,5
S,6
R,7
R,8
S,9
R,10
R)-1,6,8-triacetoxy-9-benzoyloxydihydro-β-agarofuran-2-one.
Compound
2, obtained as a pale yellow solid with [α]
D19 = +18.448° (
c = 0.23, CH
3OH), possessed a molecular formula of C
34H
39NO
12, as deduced from HR-ESI-MS data (
m/
z: 676.2359 [M + Na]
+) (
Figure S12) and
13C-NMR data. The IR showed the presence of ester carbonyl (1751, 1719 cm
−1), hydroxyl (3551 cm
−1) and aromatic groups (3063, 1593 cm
−1). The UV spectrum showed the presence of benzoate moieties with maximum absorption peaks at 202, 228 and 264 nm. Spraying with potassium heptaiodobismuthate solution gave an orange-red spot on thin layer chromatographic plate, indicating
2 belonged to alkaloids.
The
1H- and
13C-NMR (
Figures S7 and S8), DEPT and HSQC spectroscopic data (
Figure S9) of
2 (
Table 1 and
Table 2) showed signals of five carbonyl groups (δ
C = 164.4, 165.1, 169.6, 170.0, 170.7), six methyl groups (δ
H/δ
C = 1.41/24.4, 1.47/19.1, 1.49/24.7, 1.53/28.2, 1.99/19.6, 2.22/19.8, each hydrogen signal being a singlet and 3H), three methylene groups with one linked to an oxygen atom (δ
H/δ
C = 1.90 (dd,
J = 2.5, 15.0 Hz, 1H) and 2.22 (m, 1H)/41.2, 2.25 (m, 1H) and 2.59 (4 × d,
J = 3.5, 7.0, 16.0 Hz, 1H)/34.3, 4.35 (d,
J = 13.0 Hz, 1H) and 4.99 (d,
J = 13.0 Hz, 1H)/65.2), five methine groups with four linked to an oxygen atom (δ
H/δ
C = 6.30 (s, 1H)/79.2, 5.54 (d,
J = 3.1 Hz, 1H)/70.9, 5.42 (d,
J = 7.0 Hz, 1H)/68.0, 5.41 (m, 1H)/69.1, 2.33 (br s, 1H)/49.1), four quaternary sp
3 carbon atoms with three linked to an oxygen atom (δ
C = 55.3, 69.7, 84.5, 90.8), two quaternary sp
2 carbon atoms (δ
C 126.4, 129.1) and nine tertiary sp
2 carbon atoms (δ
H = 7.39–9.23 (9H)/δ
C 124.0–153.0 (nine peaks)). The
1H-NMR, HSQC and
1H-
1H COSY spectra revealed the presence of one phenyl group (δ
H/δ
C = 7.39 (t,
J = 7.7 Hz, 2H)/128.1, 7.54 (t,
J = 7.4 Hz, 1H)/133.3, 7.94 (d,
J = 7.8 Hz, 2H)/129.8) and one 3-pyridyl group (δ
H 9.23 (s, 1H)/150.5, 8.68 (br s, 1H)/153.0, 8.47 (d,
J = 8.0 Hz, 1H)/138.0, 7.50 (dd,
J = 2.6, 8.0 Hz, 1H)/124.0). The complete assignments of signals of all hydrogen atoms on the dihydro-β-agarofuran rings were carried out by an analysis of the
1H-
1H COSY and coupling constants. The signals of H-12, H-13, H-14 and H-15 were assigned on the base of the HMBC analysis (
Figure S10).
In the HMBC spectrum, the correlations between δH 5.54 (d, J = 3.1 Hz, 1H, H-1), 1.47 (s, 3H, 1-CH3C=O) and δC = 169.6, between δH = 5.41 (m, 1H, H-2), 1.99 (s, 3H, 2-CH3C=O) and δC = 170.0, and between δH = 4.35 (d, J = 13.0 Hz, 1H, H-15a), 4.99 (d, J = 13.0 Hz, 1H, H-15b), 2.22 (s, 3H, 15-CH3C=O) and δC = 170.7 revealed the presence of 1-AcO, 2-AcO and 15-AcO groups while the correlations of both δH = 6.30 (s, 1H, H-6) and 8.47 (d, J = 8.0 Hz, 1H, H-6'') to δC = 164.4, and both δH = 5.42 (d, J = 7.0 Hz, 1H, H-9) and 7.94 (d, J = 7.8 Hz, 2H, H-2') to δC = 165.1 indicated the presence of 6-nicotinoyloxy and 9-benzoyloxy groups. The correlations between δH = 5.41 (m, 1H, H-2), 1.90 (dd, J = 2.5, 15.0 Hz, 1H, H-3eq), 2.22 (m, 1H, H-3ax), 1.41 (s, 3H, H-14) and δC = 69.7 (C-4) showed that the unique hydroxyl group was located at C-4.
In the NOESY spectrum (
Figure S11), the cross signals of δ
H = 5.54 (d,
J = 3.1 Hz, 1H, H-1) toδ
H = 5.41 (m, 1H, H-2), 2.22 (m, 1H, H-3
ax) and 7.94 (d,
J = 7.8 Hz, 2H, H-2') showed that 1-AcO and 2-AcO groups were equatorial and axial, respectively. The correlations between δ
H = 1.41 (s, 3H, H-14) and 1.90 (dd,
J = 2.5, 15.0 Hz, 1H, H-3
eq), 4.35 (d,
J = 13.0 Hz, 1H, H-15
a), 4.99 (d,
J = 13.0 Hz, 1H, H-15
b) and 6.30 (s, 1H, H-6), and between δ
H = 6.30 (s, 1H, H-6) and 4.35 (d,
J = 13.0 Hz, 1H, H-15
a), 4.99 (d,
J = 13.0 Hz, 1H, H-15
b) indicated that both 4-OH and 6-nicotinoyloxy groups were located at an equatorial bond. The correlations of δ
H = 5.42 (d,
J = 7.0 Hz, 1H, H-9) to 4.35 (d,
J = 13.0 Hz, 1H, H-15
a) and 2.59 (4 × d,
J = 3.5, 7.0, 16.0 Hz, 1H, H-8
ax) showed 9-benzoyloxy group was axial. Based on the NOESY analysis above as well as the fact that 1-OH or 1-ester group in all known natural dihydro-β-agarofuran sesquiterpenes were found to be equatorial, the absolute configuration of
2 was determined. Thus,
2 was elucidated as (1
R,2
S,4
S,5
S,6
R,7
R,9
S,10
R)-1,2,15-triacetoxy-9-benzoyloxy-6-nicotinoyloxy dihydro-β-agarofuran. This finding on
2 is of a certain significance because this compound is the first nitrogen-containing dihydro-β-agarofuran sesquiterpene polyester isolated from this plant.
Compound
3, obtained as a pale yellow solid with [α]
D19 = +15.364° (
c 0.26, CH
3OH), possessed a molecular formula of C
31H
34O
9, as deduced from HR-ESI-MS data (
m/
z: 573.2073 [M + Na]
+) (
Figure S18) and
13C-NMR data (
Figure S14). The IR showed the presence of carbonyl (1730 cm
−1), hydroxyl (3466 cm
−1), and aromatic groups (3068, 1602, 1558 cm
−1). The UV spectrum showed the presence of benzoate moieties with maximum absorption peaks at 202, 228, 274 and 282 nm.
The
1H-NMR spectrum of compound
3 (
Figure S13) showed the signals of one acetyl group (δ
H = 2.15, s, 3H), two phenyl groups with two sets of aromatic hydrogen signals (δ
H = 7.26–8.04, 10H) and one hydroxy group (δ
H = 3.19, d,
J = 4.5 Hz) which disappeared after the addition of D
2O. The
13C-NMR spectrum showed the presence of one acetic ester (δ
C = 21.2, 169.4) and two benzoic ester moieties (δ
C = 128.3–133.1, eight peaks; 164.8, 165.2). Besides these ester moieties, the
13C-NMR, DEPT and HSQC spectra (
Figure S15) showed the presence of four methyl groups (δ
H/δ
C = 1.52 (s, 3H)/31.2, 1.82 (s, 3H)/26.8, 1.01(d,
J = 7.6 Hz, 3H)/18.4, 1.22(s, 3H)/18.9), one methylene group (δ
H/δ
C = 3.24 (dd,
J = 7.8, 13.1 Hz, 1H), 2.37 (br d,
J = 13.1 Hz, 1H)/42.8), six methine groups with four bearing an oxygen atom (δ
H/δ
C = 2.78 (d,
J = 2.9 Hz, 1H)/53.7, 2.86 (m, 1H)/38.4, 5.19 (d,
J = 4.5 Hz, 1H)/75.1, 5.44 (d,
J = 6.6 Hz, 1H)/72.8, 5.49 (s, 1H)/77.3, 5.84 (dd,
J = 3.2, 6.6 Hz, 1H)/69.9), three quaternary sp
3 carbon atoms (δ
C 56.3, 83.8, 88.7) with two linked to an oxygen atom and one ketone group (δ
C = 211.5), which were very similar to those of the skeleton of (1
R,4
R,5
S,6
R,7
R,8
R,9
R,10
R)-1-acetoxy-6,9-dibenzoyloxy-8-hydroxydihydro-β-agarofuran-2-one isolated from the same plant [
1].
Based on the cross peak (δ
H/δ
H = 3.19/5.19) in the
1H-
1H COSY spectrum, the same coupling constant (
J = 4.5 Hz) in combination with the correlations of δ
H = 5.19 to δ
C = 18.9 (C-15), 56.3 (C-10), 72.8 (C-9) and 211.5 (C-2) in the HMBC spectrum, the signals at δ
H = 5.19 and 3.19 were assigned as H-1 and 1-OH, respectively. In the HMBC spectrum (
Figure S16), the correlations of δ
H = 5.49 (s, 1H) to δ
C = 56.3 (C-10), 69.9 (C-8), 83.8 (C-11) and 88.7 (C-5), δ
H = 5.84 (dd,
J = 3.2, 6.6 Hz, 1H) to δ
C = 53.7 (C-7) and 72.8 (C-9), and δ
H= 5.44 (d,
J = 6.6 Hz, 1H) to δ
C = 18.9 (C-15), 53.7 (C-7), 56.3 (C-10), 69.9 (C-8) and 88.7 (C-5) showed that the peaks at δ
H = 5.49, 5.84, 5.44 belonged to H-6, H-8 and H-9, respectively. The above assignments were further supported by the
1H-
1H COSY spectrum and coupling constants. The complete assignments of the other protons and all the protonated carbons were finished on the base of the analysis of HSQC, COSY spectra and coupling constants.
The substitution sites of three ester groups were established by the analysis of the HMBC spectrum (
Figure 2). Correlations between δ
H = 5.49 (H-6), 2.15 (C
H3C=O-6) and δ
C = 169.4 (C=O), between δ
H = 5.84 (H-8), 7.78 (d,
J = 7.5, 2H, H-2'') and δ
C = 164.8 (C=O), and between δ
H = 5.44 (H-9), 8.04 (d,
J = 7.5 Hz, 2H, H-2') and δ
C = 165.2 (C=O) indicated the presence of 6-acetoxy and 8,9-dibenzoyloxy groups. The long-distance correlations between δ
H = 5.19 (H-1), 3.24 (H-3
ax) and δ
C = 211.5 (C-2) in conjunction with downfield shift of H-3 compared with those of
2 showed that the ketone carbonyl group was located at C-2. Thus, compound
3 was identified as 6-acetoxy-8,9-dibenzoyloxy-1-hydroxydihydro-β-agarofuran-2-one.
In the NOESY spectrum (
Figure S17), the correlations between δ
H = 5.19 (H-1) and 3.24 (H-3
ax), 8.04 (H-2'), between δ
H = 3.19 (1-OH) and 1.27 (H-15), between δ
H = 5.49 (H-6) and 1.01 (H-14), 1.27 (H-15), 5.84 (H-8), and between δ
H = 5.44 (H-9) and 1.22 (H-15), 5.84 (H-8) showed that 1-OH, 6-OAc and 8-benzoyloxyl were equatorial while 9-benzoyloxyl was axial. Accordingly, compound
3 was elucidated as (1
R,4
R,5
S,6
R,7
R,8
S,9
R,10
S)-6-acetoxy-8,9-dibenzoyloxy-1-hydroxydihydro-β-agarofuran-2-one. Interestingly, compound
3 is one diastereomer of the known compound triptogelin A-4 ((1
R,4
R,5
S,6
R,7
R,8
R,9
S,10
S)-6-acetoxy-8,9-dibenzoyloxy-1-hydroxydihydro-β-agarofuran-2-one) (([α]
D23 = –31.0° (
c 1.0, CH
3OH)), isolated from the plant
Tripterygium wilfordii Hook fil. var.
regelii Makino [
6]. The only difference between two compounds lies in the configurations of C-8 and C-9, which is supported by their specific rotations.
Compound
4, obtained as a white solid with [
α]
D21 = +3.152° (
c 0.24, CHCl
3), possessed a molecular formula of C
31H
36O
8, as deduced from ESI-MS data (
m/z: 559.23 [M+Na]
+) (
Figure S24) and
13C-NMR data. The IR showed the presence of ester carbonyl (1718 cm
−1), hydroxyl (3519 cm
−1) and aromatic groups (3062, 3010, 1602, 1584 cm
−1). The UV spectrum showed the presence of benzoate moieties with maximum absorption peaks at 242, 274 and 282 nm.
The
1H- and
13C-NMR spectrum data (
Figures S19 and S20) of compound
4 are identical to those of compound wightianine A ((1
S,2
R,4
R,5
S,6
R,7
R,9
S,10
S)-2-acetoxy-6,9-dibenzoyloxy-1-hydroxydihydro-β-agarofuran) isolated from the same plant [
3], which suggests that compound
4 should be 2-acetoxy-6,9-dibenzoyloxy-1-hydroxydihydro-β-agarofuran. This inference was further confirmed by the HSQC and HMBC spectra (
Figures S21 and S22). However, the NOESY spectrum (
Figure S23) showed that compound
4 should have different stereochemistry from wightianine A. In the NOESY spectrum, the strong correlations between δ
H = 4.62 (dd,
J = 4.0, 5.4 Hz, 1H, H-1) and 5.34 (q-like,
J = 3.4 Hz, 1H, H-2), 2.41 (4 × d,
J = 3.4, 6.2, 13.0 Hz, 1H, H-3
ax), 8.09 (d,
J = 8.4 Hz, 2H, H-2' and H-6') and between δ
H = 1.82 (d,
J = 5.5 Hz, 1H, 1-OH) and 1.42 (s, 3H, H-15) showed that 1-OH and 2-AcO groups were equatorial and axial, respectively. The correlations between δ
H = 5.67 (s, 1H, H-6) and 1.21 (d,
J = 7.7 Hz, 3H, H-14), 1.42 (s, 3H, H-15) and between δ
H = 5.08 (d,
J = 7.0 Hz, 1H, H-9) and 1.42 (s, 3H, H-15), 2.58 (4 × d,
J = 3.2, 7.0, 16.4 Hz, 1H, H-8
ax) showed that the 6-benzoyloxy group was equatorial while 9-benzoyloxy group was axial. Accordingly, compound
4 was identified as (1
R,2
S,4
R,5
S,6
R,7
R,9
S,10
S)-2-acetoxy-6,9-dibenzoyloxy-1-hydroxydihydro-β-agarofuran. The only difference of
4 with wightianine A [
3] is the epimerization at C-1 and C-2. The C-1 configuration in compound
4 is in agreement with the stereochemical characteristics of this class of compounds in which H-1 generally has the axial configuration [
7].
In addition to four new compounds (
1–
4), six known dihydro-β-agarofuran derivatives (
5–
10) were also isolated and characterized by comparison of their physical and spectral data with those in the literature [
1] as (1
R,4
R,5
S,6
R,7
R,9
S,10
R)-1-acetoxy-6,9-dibenzoyloxydihydro-β-agarofuran-2-one (
5), (1
R,2
S,4
R,5
S,6
R,7
R,8
R,9
R,10
S)-1,2,6,8,15-pentaacetoxy-9-benzoyloxydihydro-β-agarofuran (
6), (1
R,2
S,4
R,5
S,6
R,7
R,9
S,10
R)-1,2,6,15-tetraacetoxy-9-benzoyloxydihydro-β-agarofuran (
7), (1
R,4
R,5
S,6
R,7
R,8
R,9
R,10
R)-1-acetoxy-6,9-dibenzoyloxy-8-hydroxydihydro-β-agarofuran-2-one (
8), (1
R,2
S,4
R,5
S,6
R,7
R,8
R,9
R,10
R)-1,2-diacetoxy-6,9-dibenzoyloxy-8-hydroxydihydro-β-agarofuran (
9) and (1
R,2
S, 4
R,5
S,6
R,7
R,9
S,10
R)-1-acetoxy-6,9-dibenzoyloxy-2-hydroxydihydro-β-agarofuran (
10).
New compounds
1–
4 were screened to explore their influence on the proliferation of MKN-45 cells, NB4 cells, and MCF-7 cells by MTT assay.
Cis-platinum (
cis-diaminodichloroplatinum, DDP) as a standard anticancer drug purchased from Sigma Aldrich Co. (St. Louis, MO, USA) was used as a positive control drug. Based on the cytotoxic activities of compounds
5,
7–
10 [
1], the tested concentrations of the compounds were set as 20 μM. The cytotoxicity data were shown in
Table 3.
Table 3.
Inhibition percentages of compounds 1–4 against three cancer cell lines at 20 μM (48 h).
Table 3.
Inhibition percentages of compounds 1–4 against three cancer cell lines at 20 μM (48 h).
| Compound | NB4 | MKN-45 | MCF-7 |
|---|
| 1 | 12.7 ± 3.2 | 8.9 ± 2.3 | 6.0 ± 0.9 |
| 2 | 13.7 ± 5.4 | 9.3 ± 1.6 | 2.9 ± 1.4 |
| 3 | 27.1 ± 3.3 | 9.9 ± 1.9 | 8.3 ± 1.4 |
| 4 | 85.6 ± 3.4 | 30.5 ± 4.5 | 4.5 ± 1.4 |
| cis-diaminodichloroplatinum | 100.0 ± 0.0 | 57.1 ± 2.2 | 44.2 ± 3.5 |
Almost all the test compounds showed some activities against three tested cell lines at the concentration of 20 μM, but were obviously weaker than DDP. Among all the test compounds,
4 showed the higher activity on NB4 with the inhibition rate of 85.6% and MKN-45 cells with the inhibition rate of 30.5% followed by compound
3. Compounds
1 and
2 showed the similar activity against each of the cell lines at 20 μM. For susceptibility of various cell lines, all the compounds showed the order of NB4 cells > MKN-45 cells > MCF-7 cells. In our previous study on the cytotoxic activity of
5,
7–
10, it was found that the presence of hydroxyl groups could improve the activity [
1]. Based on the fact above, we speculated that the higher activity of
4 and
3 compared to
1 or
2 may be related to the presence of hydroxyl groups in the structures of
4 and
3.
The sesquiterpene polyesters with a dihydroagarofuran skeleton have attracted considerable attention from synthetic organic chemists and pharmacologists due to their complex and diverse chemical structures and biological activities, such as insect antifeedant and/or insecticidal activity [
8,
9], cytotoxic activity [
1], antitumor promoting activity [
10], antitubercular [
11], immunosuppressive [
12], anti-HIV [
13], anti-inflammatory activity [
14] and reversal of the multidrug resistance (MDR) phenotype [
15]. To date, hundreds of dihydro-β
-agarofuran compounds have been isolated from dozens of species of plants. It is noteworthy that most of the natural dihydro-β
-agarofurans mainly originated from plants of the Celastraceae family [
5]. Only a minority were found in other family plants such as Lamiaceae [
1]. Nevertheless, the present study and previous works on phytochemistry of
P. wightiana [
1,
2,
3] leading to the discovery of 12 new and 4 known dihydro-β
-agarofuran sesquiterpene polyesters suggest that this plant may be an important resource of dihydro-β
-agarofuran sesquiterpenes.