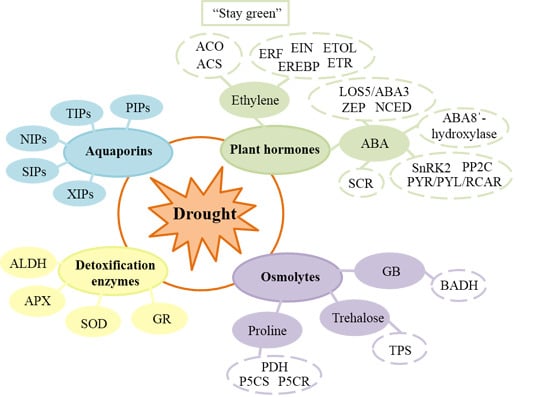
Network Candidate Genes in Breeding for Drought Tolerant Crops
Abstract
:1. Introduction
2. Role of Plant Hormones in Drought Response
2.1. Abscisic Acid (ABA) Biosynthesis, Catabolism and Signaling Pathways Are Major Players under Drought Conditions
| Gene | Gene Function | Abiotic Stress Tolerance | Reference |
|---|---|---|---|
| Zeaxanthin epoxidase (ZEP) | ABA biosynthesis | Improved tolerance to drought stress and salt | [22,25] |
| 9- cis-Epoxycarotenoid dioxygenase (NCED) | ABA biosynthesis | Improved drought tolerance by overexpression in tomato | [23,48] |
| Molybdenum cofactor sulfurase (LOS5/ABA3) | ABA biosynthesis | Increased drought tolerance in maize and soybean combined with higher yields | [30,32] |
| ABA 8′-hydroxylase | ABA catabolism | Upregulated during drought stress and rehydration: reduction in ABA level | [35] |
| Abscisic acid receptor (PYL) | ABA signaling network | Enhanced drought and salinity tolerance | [19,43] |
| Protein phosphatase 2C (PP2C) | ABA signaling network | Induced by drought, repressed by oxidative stress and heat shock | [49,50] |
| Serine/threonine protein kinase (SnRK2) | ABA signaling network | Enhanced tolerance to drought, salt, and freezing stress | [51,52] |
| SCARECROW (SCR) | Transcription factor, structural root differentiation | Binding to regulatory regions of stress-responsive genes; Regulating abscisic acid responses in Arabidopsis | [51] |
2.2. Role of Ethylene Biosynthesis, Catabolism and Signaling Pathway for Drought Tolerance
| Gene | Gene Function | Abiotic Stress Tolerance | Reference |
|---|---|---|---|
| 1-Aminocyclopropane-1-carboxylic acid oxidase (ACO) | Ethylene biosynthesis | Induced expression during wilting in tea plants promotes decreased metabolism and energy consumption by shedding of older leaves | [53] |
| 1-Aminocyclopropane-1-carboxylic acid synthase (ACS) | Ethylene biosynthesis | Delayed senescence in absence of ACS in maize | [54] |
| 1-Aminocyclopropane-1-carboxylic acid synthase 7 (ACS7) | Ethylene biosynthesis | ACS7-knock-out promotes tolerance to salt, osmotic and heat stresses | [55] |
| Ethylene-overproducer 1 (ETO1) | Limitation of ACS in ethylene biosynthesis | ETO1-deficient mutants in Arabidopsis exhibited delayed stomatal closure in response to drought | [56] |
| Ethylene overproducer 1-like (ETOL1) | Ethylene biosynthesis and energy metabolism | ETOL1-overexpressing rice more tolerant to drought and submergence | [57] |
| Ethylene response 1 (ETR1) | Ethylene signaling | Cross-link between ethylene and H2O2 signal transduction in Arabidopsis, stomatal closure; inhibition of seed germination under salt stress | [58,59] |
| Ethylene response 2 (ETR2) | Ethylene signaling | ETR2 promotes seed germination under NaCl stress in Arabidopsis | [59] |
| Ethylene insensitive 2 (EIN2) | Ethylene signaling | EIN2 downregulated in Arabidopsis during salt and osmotic stress, EIN2-deficient mutants sensitive to salt and osmotic stress | [60] |
| Ethylene insensitive 3 (EIN3) | Downstream element of ethylene signaling pathway | EIN3-deficient mutants in Arabidopsis exhibited oxidative stress after exposure to salt stress | [61] |
| Ethylene response factor 1 (ERF1) | Jasmonate and ethylene signaling | ERF1-overexpressing Arabidopsis more tolerant to drought and salt stress | [62] |
| Ethylene responsive element binding protein 1 (EREBP) | Connection between different stress signal transduction pathways | Expression induced in oil palm fruits in response to abiotic stress (drought, cold and salinity) | [63] |
| Ethylene response factor 5 (ERF5) | Vegetative growth and plant development | ERF5-overexpressing tomatoes more tolerant to drought and salt stress | [64] |
| Ethylene response factor 6 (ERF6) | Cell proliferation and leaf growth | ERF5/ERF6-deficient Arabidopsis mutants less affected by stress | [65] |
| Jasmonate and ethylene response factor 1 (JERF1) | Activation of stress responsive genes, proline synthesis and ABA biosynthesis key enzymes | JERF1-overexpressing rice more tolerant to drought stress | [66] |
3. Protection of the Cells against Osmolytic and Oxidative Damages
3.1. Osmolytes: Glycine Betaine, Proline and Trehalose
| Gene | Gene Function | Abiotic Stress Tolerance | Reference |
|---|---|---|---|
| Betaine aldehyde dehydrogenase (BADH) | Osmolyte biosynthesis | BADH from spinach in potato improved drought tolerance | [76] |
| Δ1-pyrroline-5-carboxylate synthetase (P5CS) | Osmolyte biosynthesis | Proline: most frequent osmolyte in water-stressed plants | [77] |
| Proline oxidase/dehydrogenase 1 (PDH1) | Osmolyte biosynthesis | Proline: most frequent osmolyte in water-stressed plants | [77] |
| Δ1-pyrroline-5-carboxylate reductase (P5CR) | Osmolyte biosynthesis | Proline: most frequent osmolyte in water-stressed plants; enhanced drought tolerance in soybean | [78] |
| Trehalose-phosphate synthase 1 (TPS1) | Osmolyte biosynthesis | Transgenic expression in potato improved drought tolerance | [79] |
| DNA-binding protein 4 | Transcription factor | Induced by drought | [51] |
| Dehydration-responsive element binding factor 1 (DREB1B) | Transcription factor | Arabidopsis gene in transgenic potato improved drought tolerance | [80] |
3.2. Aquaporins
| Gene | Gene Function | Abiotic Stress Tolerance | Reference |
|---|---|---|---|
| PIP2;1 | Water transport | PIP2;1 transcription downregulated in Arabidopsis; PIP2;1 degradation benefits drought tolerance | [86,87] |
| GoPIP1 | Water transport | GoPIP1 overexpressing Arabidopsis mutants more sensitive to drought | [88] |
| NtPIP1;1, NtPIP12;1 | Water transport | Downregulation of NtPIP1;1 and NtPIP2;1 in tobacco after drought stress | [89] |
| NtAQP1 | Water transport | NtAQP1 transcription upregulated in tobacco after drought stress | [89] |
| MaPIP1;1 | Water transport | MaPIP1;1 transcription in banana upregulated during drought stress; MaPIP1;1 overexpressing Arabidopsis mutants more tolerant to drought | [90] |
3.3. Protection against Reactive Oxygen Species
| Gene | Gene Function | Abiotic Stress Tolerance | Reference |
|---|---|---|---|
| Aldehyde dehydrogenase family 7 member (ALDH7) | Oxygen radical detoxification | Extenuated oxidative stress | [51,93] |
| Ascorbate peroxidase (APX) | H2O2 metabolism | Preventing oxidative stress | [94] |
| Glutathione reductase (GR) | H2O2 metabolism | Induced by oxidative stress | [95] |
| Superoxide dismutase (SOD) | Oxygen radical detoxification | Improved drought tolerance; Induced by drought | [96–98] |
4. Stay Green Trait
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Eckardt, N.A.; Cominelli, E.; Galbiati, M.; Tonelli, C. The future of science: Food and water for life. Plant Cell 2009, 21, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Chaves, M.M.; Pereira, J.S.; Maroco, J.; Rodrigues, M.L.; Ricardo, C.P.P.; Osorio, M.L.; Carvalho, I.; Faria, T.; Pinheiro, C. How plants cope with water stress in the field? Photosynthesis and growth. Ann. Bot. 2002, 89, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Mba, C.; Guimaraes, E.P.; Gosh, K. Re-orienting crop improvement for the changing climatic conditions of the 21st century. Agric. Food Secur. 2012, 1, 7. [Google Scholar] [CrossRef]
- Dodd, I.C.; Whalley, W.R.; Ober, E.S.; Parry, M.A.J. Genetic and management approaches to boost UK wheat yields by ameliorating water deficits. J. Exp. Bot. 2012, 62, 5241–5248. [Google Scholar] [CrossRef] [PubMed]
- Tuberosa, R. Phenotyping for drought tolerance of crops in the genomic era. Front. Physiol. 2012, 3, 347. [Google Scholar] [CrossRef] [PubMed]
- Grassini, P.; van Bussel, L.G.J.; van Wart, J.; Wolf, J.; Claessens, L.; Yang, H.; Boogaard, H.; de Groot, H.; van Ittersum, M.K.; Gassman, K.G. How good is good enough? Data requirements for reliable crop yield simulations and yield-gap analysis. Field Crops Res. 2015, 177, 49–63. [Google Scholar] [CrossRef] [Green Version]
- Mishra, A.K.; Ines, A.V.M.; Das, N.N.; Khedun, P.C.; Singh, V.P.; Sivakumar, B.; Hansen, J.W. Anatomy of the local-scale drought: Application of assimilated remote sensing products, crop model, and statistical methods to an agricultural drought study. J. Hydrol. 2015, 526, 15–29. [Google Scholar] [CrossRef]
- Daryanto, S.; Wang, L.; Jacinthe, P.-A. Global synthesis of drought effects on food legume production. PLoS ONE 2015, 10, e0127401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.-Y.; Cruz de Carvalho, M.H.; Torres-Jerez, I.; Kang, Y.; Allen, S.N.; Huhman, D.V.; Tang, Y.; Murray, J.; Summer, L.W.; Udvardi, M.K. Global reprogramming of transcription and metabolism in Medicago truncatula during progressive drought and rewatering. Plant Cell Environ. 2014, 37, 2553–2576. [Google Scholar] [CrossRef] [PubMed]
- Reguera, M.; Peleg, Z.; Blumwald, E. Targeting metabolic pathways for genetic engineering abiotic stress-tolerance in crops. Biochim. Biophys. Acta 2012, 1819, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Fleury, D.; Jefferies, S.; Kuchel, H.; Langridge, P. Genetic and genomic tools to improve drought tolerance in wheat. J. Exp. Bot. 2010, 61, 3211–3222. [Google Scholar] [CrossRef] [PubMed]
- Peleg, Z.; Blumwald, E. Hormone balance and abiotic stress tolerance in crop plants. Curr. Opin. Plant Biol. 2011, 14, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Liu, Y.; Li, S.-S.; Han, G.-Z. Insights into the origin and evolution of the plant hormone signaling machinery. Plant Physiol. 2015, 167, 872–886. [Google Scholar] [CrossRef] [PubMed]
- Albacete, A.A.; Martínez-Andújar, C.; Pérez-Alfocea, F. Hormonal and metabolic regulation of source-sink relations under salinity and drought: From plant survival to crop yield stability. Biotechnol. Adv. 2014, 32, 12–30. [Google Scholar] [CrossRef] [PubMed]
- Cramer, G.R.; Urano, K.; Delrot, S.; Pezzotti, M.; Shinozaki, K. Effects of abiotic stress on plants: A systems biology perspective. BMC Plant Biol. 2011, 11, 63. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, T.; Shinozaki, K. Research on plant abiotic stress responses in the post-genome era: Past, present and future. Plant J. 2010, 61, 1041–1052. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, K.; Yamaguchi-Shinozaki, K.; Shinozaki, K. The transcriptional regulatory network in the drought response and its crosstalk in abiotic stress responses including drought, cold, and heat. Front. Plant Sci. 2014, 5, 170. [Google Scholar] [CrossRef] [PubMed]
- Finkelstein, R. Abscisic acid synthesis and response. Arabidopsis Book 2013, 11, e0166. [Google Scholar] [CrossRef] [PubMed]
- Miyakawa, T.; Fujita, Y.; Yamaguchi-Shinozaki, K.; Tanokura, M. Structure and function of abscisic acid receptors. Trends Plant Sci. 2013, 18, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Mehrotra, R.; Bhalothia, P.; Bansal, P.; Basantani, M.K.; Bharti, V.; Mehrotra, S. Abscisic acid and abiotic stress tolerance—Different tiers of regulation. J. Plant Physiol. 2014, 171, 486–496. [Google Scholar] [CrossRef] [PubMed]
- Eskling, M.; Arvidsson, P.-O.; Åkerlund, H.E. The xanthophyll cycle, its regulation and components. Physiol. Plant. 1997, 100, 806–816. [Google Scholar] [CrossRef]
- Thompson, A.J.; Jackson, A.C.; Parker, R.A.; Morpeth, D.R.; Burbidge, A.; Taylor, I.B. Abscisic acid biosynthesis in tomato: Regulation of zeaxanthin epoxidase and 9-cis-epoxycarotenoid dioxygenase mRNAs by light/dark cycles, water stress and abscisic acid. Plant Mol. Biol. 2000, 42, 833–845. [Google Scholar] [CrossRef] [PubMed]
- Iuchi, S.; Kobayashi, M.; Taji, T.; Naramoto, M.; Seki, M.; Kato, T.; Tabata, S.; Kakubari, Y.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Regulation of drought tolerance by gene manipulation of 9-cis-epoxycarotenoid dioxygenase, a key enzyme in abscisic acid biosynthesis in Arabidopsis. Plant J. 2001, 27, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Thomas, H.; Howarth, C.J. Five ways to stay green. J. Exp. Bot. 2000, 51, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Park, H.Y.; Seok, H.Y.; Park, B.K.; Kim, S.H.; Goh, C.H.; Lee, B.-H.; Lee, C.-H.; Moon, Y.-H. Overexpression of Arabidopsis ZEP enhances tolerance to osmotic stress. Biochem. Biophys. Res. Commun. 2008, 375, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Guzman, M.; Apostolova, N.; Belles, J.M.; Barrero, J.M.; Piqueras, P.; Ponce, M.R.; Micol, J.L.; Serrano, R.; Rodríguez, P.L. The short-chain alcohol dehydrogenase ABA2 catalyzes the conversion of xanthoxin to abscisic aldehyde. Plant Cell 2002, 14, 1833–1846. [Google Scholar] [CrossRef] [PubMed]
- Bittner, F.; Oreb, M.; Mendel, R.R. ABA3 is a molybdenum cofactor sulfurase required for activation of aldehyde oxidase and xanthine dehydrogenase in Arabidopsis thaliana. J. Biol. Chem. 2001, 276, 40381–40384. [Google Scholar] [CrossRef] [PubMed]
- Seo, M.; Aoki, H.; Koiwai, H.; Kamiya, Y.; Nambara, E.; Koshiba, T. Comparative studies on the Arabidopsis aldehyde oxidase (AAO) gene family revealed a major role of AAO3 in ABA biosynthesis in seeds. Plant Cell Physiol. 2004, 45, 1694–1703. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.M.; Lee, H.; Ishitani, M.; Zhu, J.K. The ArabidopsisLOS5/ABA3 locus encodes a molybdenum cofactor sulfurase and modulates cold stress- and osmotic stress-responsive gene expression. Plant Cell 2001, 13, 2063–2083. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, J.; Zhang, J.; Hao, L.; Hua, J.; Duan, L.; Zhang, M.; Li, Z. Expression of an Arabidopsis molybdenum factor sulphurase gene in soybean enhances drought tolerance and increases yield under field conditions. Plant Biotechnol. J. 2013, 11, 747–758. [Google Scholar] [CrossRef] [PubMed]
- Xiao, B.Z.; Chen, X.; Xiang, C.B.; Tang, N.; Zhang, Q.F.; Xiong, L.Z. Evaluation of seven function-known candidate genes for their effects on improving drought resistance of transgenic rice under field conditions. Mol. Plant 2009, 2, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Li, Y.; Zhang, J.; Xiao, Y.; Yue, Y.; Duan, L.; Zhang, M.; Li, Z. Overexpression of Arabidopsis molybdenum cofactor sulfurase gene confers drought tolerance in maize (Zea mays L.). PLoS ONE 2013, 8, e52126. [Google Scholar] [CrossRef] [PubMed]
- Cutler, A.J.; Krochko, J.E. Formation and breakdown of ABA. Trends Plant Sci. 1999, 4, 472–478. [Google Scholar] [CrossRef]
- Nambara, E.; Marion-Poll, A. Abscisic acid biosynthesis and catabolism. Annu. Rev. Plant Biol. 2005, 56, 165–185. [Google Scholar] [CrossRef] [PubMed]
- Kushiro, T.; Okamoto, M.; Nakabayashi, K.; Yamagishi, K.; Kitamura, S.; Asami, T.; Hirai, N.; Koshiba, T.; Kamiya, Y.; Nambara, E. The Arabidopsis cytochrome P450 CYP707A encodes ABA 8′-hydroxylases: Key enzymes in ABA catabolism. EMBO J. 2004, 23, 1647–1656. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Piao, H.L.; Kim, H.Y.; Choi, S.M.; Jiang, F.; Hartung, W.; Hwang, I.; Kwak, J.M.; Lee, I.J.; Hwang, I. Activation of glucosidase via stress-induced polymerization rapidly increases active pools of abscisic acid. Cell 2006, 126, 1109–1110. [Google Scholar] [CrossRef] [PubMed]
- Cutler, S.R.; Rodriguez, P.L.; Finkelstein, R.R.; Abrams, S.R. Abscisic acid: Emergence of a core signaling network. Annu. Rev. Plant Biol. 2010, 61, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Umezawa, T.; Nakashima, K.; Miyakawa, T.; Kuromori, T.; Tanokura, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Molecular basis of the core regulatory network in ABA responses: Sensing, signaling and transport. Plant Cell Physiol. 2010, 51, 1821–30. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Fung, P.; Nishimura, N.; Jensen, D.R.; Hiroaki, F.; Zhao, Y.; Lumba, S.; Santiago, J.; Rodrigues, A.; Chow, T.-F.F.; et al. Abscisic acid inhibits PP2Cs via the PYR/PYL family of ABA binding START proteins. Science 2009, 324, 1068–1070. [Google Scholar] [PubMed]
- Schweighofer, A.; Hirt, H.; Meskiene, I. Plant PP2C phosphatases: Emerging functions in stress signaling. Trends Plant Sci. 2004, 9, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Kulik, A.; Wawer, I.; Krzywinska, E.; Bucholc, M.; Dobrowolska, G. SnRK2 protein kinases—Key regulators of plant response to abiotic stresses. OMICS 2011, 15, 859–860. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Jha, S.K.; Bagri, J.; Pandey, G.K. ABA inducible rice protein phosphatase 2C confers ABA insensitivity and abiotic stress tolerance in Arabidopsis. PLoS ONE 2015, 10, e0125168. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Lee, K.; Hwang, H.; Bhatnagar, N.; Kim, D.-Y.; Yoon, I.S.; Buyn, M.-O.; Kim, S.T.; Jung, K.-H.; Kim, B.G. Overexpression of PYL5 in rice enhances drought tolerance, inhibits growth, and modulates gene expression. J. Exp. Bot. 2014, 65, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Shinozaki, K.; Yamaguchi-Shinozaki, K. Gene networks involved in drought stress response and tolerance. J. Exp. Bot. 2007, 58, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Skirycz, A.; Claeys, H.; de Bodt, S.; Oikawa, A.; Shinoda, S.; Andriankaja, M.; Maleux, K.; Eloy, N.B.; Coppens, F.; Yoo, S.-D.; et al. Pause-and-Stop: The effect of osmotic stress on cell proliferation during early leaf development in Arabidopsis and the role for ethylene signaling in cell cycle arrest. Plant Cell 2011, 23, 1876–1888. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ko, J.H.; Yang, S.H.; Han, K.H. Upregulation of an Arabidopsis RING-H2 gene, XERICO, confers drought tolerance through increased abscisic acid biosynthesis. Plant J. 2006, 47, 343–355. [Google Scholar] [CrossRef] [PubMed]
- Golldack, D.; Li, C.; Mohan, H.; Probst, N. Tolerance to drought and salt stress in plants: Unraveling the signaling networks. Front. Plant Sci. 2014, 5, 151. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.J.; Andrews, J.; Mulholland, B.J.; McKee, J.M.T.; Hilton, H.W.; Horridge, J.S.; Farquhar, G.D.; Smeeton, R.C.; Smillie, I.R.A.; Black, C.R.; et al. Overproduction of abscisic acid in tomato increases transpiration efficiency and root hydraulic conductivity and influences leaf expansion. Plant Physiol. 2007, 143, 1905–1917. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, P.L. Protein phosphatase 2C (PP2C) function in higher plants. Plant Mol. Biol. 1998, 38, 919–927. [Google Scholar] [CrossRef] [PubMed]
- Vranová, E.; Langebartels, C.; van Montagu, M.; Inzé, D.; van Camp, W. Oxidative stress, heat shock and drought differentially affect expression of a tobacco protein phosphatase 2C. J. Exp. Bot. 2000, 51, 1763–1764. [Google Scholar] [CrossRef] [PubMed]
- Schafleitner, R.; Rosales, R.O.G.; Gaudin, A.; Aliaga, C.A.A.; Martinez, G.N.; Marca, L.R.T.; Bolivar, L.A.; Delgado, F.M.; Simon, R.; Bonierbale, M. Capturing candidate drought tolerance traits in two native Andean potato clones by transcription profiling of field grown plants under water stress. Plant Physiol. Biochem. 2007, 45, 673–690. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.; Zhang, H.; Tian, S.; Chang, X.; Jing, R. TaSnRK2.4, an SNF1-type serine/threonine protein kinase of wheat (Triticum aestivum L.), confers enhanced multistress tolerance in Arabidopsis. J. Exp. Bot. 2010, 61, 683–696. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Bharalee, R.; Bhorali, P.; Bandyopadhyay, T.; Gohain, B.; Agarwal, N.; Ahmed, P.; Saikia, H.; Borchetia, S.; Kalita, M.C.; et al. Identification of drought tolerant progenies in tea by gene expression analysis. Funct. Integr. Genomics 2012, 12, 543–563. [Google Scholar] [CrossRef] [PubMed]
- Young, T.E.; Meeley, R.B.; Gallie, D.R. ACC synthase expression regulates leaf performance and drought tolerance in maize. Plant J. 2004, 40, 813–825. [Google Scholar]
- Dong, H.; Zhen, Z.; Peng, J.; Chang, L.; Gong, Q.; Wang, N.N. Loss of ACS7 confers abiotic stress tolerance by modulating ABA sensitivity and accumulation in Arabidopsis. J. Exp. Bot. 2011, 62, 4875–4887. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.L.-C.; Yoshida, H.; Lurin, C.; Ecker, J.R. Regulation of ethylene gas biosynthesis by the Arabidopsis ETO1 protein. Nature 2004, 428, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Wu, N.; Cui, F.; You, L.; Li, X.; Xiong, L. A homolog of ETHYLENE OVERPRODUCER, OsETOL1, differentially modulates drought and submergence tolerance in rice. Plant J. 2014, 78, 834–849. [Google Scholar] [CrossRef] [PubMed]
- Desikan, R.; Last, K.; Harrett-Williams, R.; Tagliavia, C.; Harter, K.; Hooley, R.; Hancock, J.T.; Neill, S.J. Ethylene-induced stomatal closure in Arabidopsis occurs via AtrbohF-mediated hydrogen peroxide synthesis. Plant J. 2006, 47, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.L.; Kim, H.; Bakshi, A.; Binder, B.M. The ethylene receptors ETHYLENE RESPONSE1 and ETHYLENE RESPONSE2 have contrasting roles in seed germination of Arabidopsis during salt stress. Plant Physiol. 2014, 165, 1353–1366. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, C.; Li, K.; Sun, F.; Hu, H.; Li, X.; Zhao, Y.; Han, C.; Zhang, W.; Duan, Y.; et al. Arabidopsis EIN2 modulates stress response through abscisic acid response pathway. Plant Mol. Biol. 2007, 64, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Asensi-Fabado, M.A.; Cela, J.; Müller, M.; Arrom, L.; Chang, C.; Munné-Bosch, S. Enhanced oxidative stress in the ethylene-insensitive (ein3-1) mutant of Arabidopsis thaliana exposed to salt stress. J. Plant Physiol. 2012, 169, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.-C.; Liao, P.-M.; Kuo, W.-W.; Lin, T.-P. The Arabidopsis ETHYLENE RESPONSE FACTOR1 regulates abiotic stress-responsive gene expression by binding to different cis-acting elements in response to different stress signals. Plant Physiol. 2013, 162, 1566–1582. [Google Scholar] [CrossRef] [PubMed]
- Omidvar, V.; Abdullah, S.N.A.; Ho, C.L.; Mahmood, M. Isolation and characterization of an ethylene-responsive element binding protein (EgEREBP) from oil palm (Elaeis guineensis). Aust. J. Crop Sci. 2013, 7, 219–226. [Google Scholar]
- Pan, Y.; Seymour, G.B.; Lu, C.; Hu, Z.; Chen, X.; Chen, G. An ethylene response factor (ERF5) promoting adaptation to drought and salt tolerance in tomato. Plant Cell Rep. 2012, 31, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Dubois, M.; Skirycz, A.; Claeys, H.; Maleux, K.; Dhondt, S.; de Bodt, S.; Vanden Bossche, R.; de Milde, L.; Yoshizumi, T.; Matsui, M.; et al. Ethylene response factor6 acts as a central regulator of leaf growth under water-limiting conditions in Arabidopsis. Plant Physiol. 2013, 162, 319–332. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Li, F.; Li, D.; Zhang, H.; Huang, R. Expression of ethylene response factor JERF1 in rice improves tolerance to drought. Planta 2010, 232, 765–774. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.F.; Zhong, S.L.; Grierson, D. Recent advances in ethylene research. J. Exp. Bot. 2009, 60, 3311–3336. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.A.; Bianchetti, R.E.; Freschi, L. Shedding light on ethylene metabolism in higher plants. Front. Plant Sci. 2014, 5, 665. [Google Scholar] [CrossRef] [PubMed]
- Yokoi, S.; Quintero, F.J.; Cubero, B.; Ruiz, M.T.; Bressan, R.A.; Hasegawa, P.M.; Pardo, J.M. Differential expression and function of Arabidopsis thaliana NHX Na+/H+ antiporters in the salt stress response. Plant J. 2002, 30, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Sano, T.; Tamaoki, M.; Nakajima, N.; Kondo, N.; Hasezawa, S. Ethylene inhibits abscisic acid-induced stomatal closure in Arabidopsis. Plant Physiol. 2005, 138, 2337–2343. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.-H.; Yoo, S.-D. Novel connections and gaps in ethylene signaling from the ER membrane to the nucleus. Front. Plant Sci. 2015, 5, 733. [Google Scholar] [CrossRef] [PubMed]
- Chao, Q.; Rothenberg, M.; Solano, R.; Roman, G.; Terzaghi, W.; Ecker, J.R. Activation of the ethylene gas response pathway in Arabidopsis by the nuclear protein ETHYLENE-INSENSITIVE3 and related proteins. Cell 1997, 89, 1133–1144. [Google Scholar] [CrossRef]
- Charfeddine, M.; Saidi, N.M.; Charfeddine, S.; Hammami, A.; Bouzid, R.G. Genome-wide analysis and expression profiling of the ERF transcription factor family in potato (Solanum tuberosum L.). Mol. Biotechnol. 2015, 57, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.K.; Kumar, R.; Solanke, A.U.; Sharma, R.; Tyagi, A.K.; Sharma, A.K. Identification, phylogeny, and transcript profiling of ERF family genes during development and abiotic stress treatments in tomato. Mol. Genet. Genomics 2010, 284, 455–475. [Google Scholar] [CrossRef] [PubMed]
- Burg, M.B.; Ferraris, J.D. Intracellular organic osmolytes: Function and regulation. J. Biol. Chem. 2008, 283, 7309–7313. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Si, H.-J.; Wen, G.; Du, H.-H.; Liu, B.-L.; Wang, D. Enhanced drought and salintiy tolerance in transgenic potato plants with BADH gene from spinach. Plant Biotechnol. Rep. 2011, 5, 71–77. [Google Scholar] [CrossRef]
- Sharma, S.; Villamor, J.G.; Verslues, P.E. Essential role of tissue-specific proline synthesis and catabolism in growth and redox balance at low water potential. Plant Physiol. 2011, 157, 292–304. [Google Scholar] [CrossRef] [PubMed]
- De Ronde, J.A.; Spreeth, M.H.; Cress, W.A. Effect of antisense L-Δ1-pyrroline-5-carboxylate reductase transgenic soybean plants subjected to osmotic and drought stress. Plant Growth Regul. 2000, 32, 13–26. [Google Scholar] [CrossRef]
- Stiller, I.; Dulai, S.; Kondrák, M.; Tarnai, R.; Szabó, L.; Toldi, O.; Bánfalvi, Z. Effects of drought on water content and photosynthetic parameters in potato plants expressing the trehalose-6-phosphate synthase gene of Saccharomyces cerevisiae. Planta 2008, 227, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Movahedi, S.; Sayed Tabatabaei, B.E.; Alizade, H.; Ghobadi, C.; Yamchi, A.; Khaksar, G. Constitutive expression of Arabidopsis DREB1B in transgenic potato enhances drought and freezing tolerance. Biol. Plant 2012, 56, 37–42. [Google Scholar] [CrossRef]
- Verbruggen, N.; Hermans, C. Proline accumulation in plants: A review. Amino Acids 2008, 35, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M.; Foolad, M.R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Maurel, C.; Reizer, J.; Schroeder, J.I.; Chrispeels, M.J. The vacuolar membrane protein γ-TIP creates water specific channels in Xenopus oocytes. EMBO J. 1993, 12, 2241–2247. [Google Scholar] [PubMed]
- Danielson, J.A.H.; Johanson, U. Unexpected complexity of the aquaporin gene family in the moss Physcomitrella patens. BMC Plant Biol. 2008, 8, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Gao, C.; Li, Y.; Li, Y.; Zhu, Y.; Xu, G.; Shen, Q.; Kaldenhoff, R.; Kai, L.; Guo, S. The enhanced drought tolerance of rice plants under ammonium is related to aquaporin (AQP). Plant Sci. 2015, 234, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.Y.; Kim, D.G.; Kim, Y.O.; Kim, J.S.; Kang, H. An expression analysis of a gene family encoding plasma membrane aquaporins in response to abiotic stresses in Arabidopsis thaliana. Plant Mol. Biol. 2004, 54, 713–725. [Google Scholar] [CrossRef] [PubMed]
- Alexandersson, E.; Fraysse, L.; Sjövall-Larsen, S.; Gustavsson, S.; Fellert, M.; Karlsson, M.; Johanson, U.; Kjellbom, P. Whole gene family expression and drought stress regulation of aquaporins. Plant Mol. Biol. 2005, 59, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ban, L.; Wen, H.; Wang, Z.; Dzyubenko, N.; Chapurin, V.; Gao, H.; Wang, X. An aquaporin protein is associated with drought stress tolerance. Biochem. Biophys. Res. Commun. 2015, 459, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Mahdieh, M.; Mostajeran, A.; Horie, T.; Katsuhara, M. Drought stress alters water relations and expression of PIP-type aquaporin genes in Nicotiana tabacum plants. Plant Cell Physiol. 2008, 49, 801–813. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Hu, W.; Liu, J.; Zhang, J.; Jia, C.; Miao, H.; Xu, B.; Jin, Z. A banana aquaporin gene, MaPIP1;1, is involved in tolerance to drought and salt stresses. BMC Plant Biol. 2014, 14, 59. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.K.; Cho, S.K.; Son, O.; Xu, Z.; Hwang, I.; Kim, W.T. Drought stress-induced Rma1H1, a RING membrane-anchor E3 ubiquitin ligase homolog, regulates aquaporin levels via ubiquitination in transgenic Arabidopsis plants. Plant Cell 2009, 21, 622–641. [Google Scholar] [CrossRef] [PubMed]
- Moshelion, M.; Halperin, O.; Wallach, R.; Oren, R.; Way, D.A. Role of aquaporins in determining transpiration and photosynthesis in water-stressed plants: Crop water-use efficiency, growth and yield. Plant Cell Environ. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, S.M.; Andrade, M.O.; Gomes, A.P.S.; Damatta, F.M.; Baracat-Pereira, M.C.; Fontes, E.P.B. Arabidopsis and tobacco plants ectopically expressing the soybean antiquitin-like ALDH7 gene display enhanced tolerance to drought, salinity, and oxidative stress. J. Exp. Bot. 2006, 57, 1909–1918. [Google Scholar] [CrossRef] [PubMed]
- Badawi, G.H.; Kawano, N.; Yamauchi, Y.; Shimada, E.; Sasaki, R.; Kubo, A.; Tanaka, K. Over-expression of ascorbate peroxidase in tobacco chloroplasts enhances the tolerance to salt stress and water deficit. Physiol. Plant. 2004, 121, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Gallé, Á.; Csiszár, J.; Benyó, D.; Laskay, G.; Leviczky, T.; Erdei, L.; Tari, I. Isohydric and anisohydric strategies of wheat genotypes under osmotic stress: Biosynthesis and function of ABA in stress responses. J. Plant Physiol. 2013, 170, 1389–1399. [Google Scholar]
- McKersie, B.D.; Bowley, S.R.; Harjanto, E.; Leprince, O. Water-deficit tolerance and field performance of transgenic alfalfa overexpressing superoxide dismutase. Plant Physiol. 1996, 111, 1177–1181. [Google Scholar] [PubMed]
- Fulda, S.; Mikkat, S.; Stegmann, H.; Horn, R. Physiology and proteomics of drought stress acclimation in sunflower (Helianthus annuus L.). Plant Biol. 2011, 13, 632–642. [Google Scholar] [CrossRef] [PubMed]
- Castillejo, M.A.; Maldonado, A.M.; Ogueta, S.; Jorrin, J.V. Proteomic analysis of responses to drought stress in sunflower (Helianthus annuus) leaves by 2DE gel electrophoresis and mass spectrometry. Open Proteomics J. 2008, 1, 59–71. [Google Scholar] [CrossRef]
- Singh, S.; Brocker, C.; Koppaka, V.; Chen, Y.; Jackson, B.C.; Matsumoto, A.; Thompson, D.C.; Vasiliou, V. Aldehyde dehydrogenases in cellular responses to oxidative/electrophilic stress. Free Radic. Biol. Med. 2013, 56, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Halliwell, B. Purification and properties of dehydroascorbate reductase from spinach leaves. Phytochemistry 1977, 16, 1347–1350. [Google Scholar] [CrossRef]
- Shigeoka, S. Regulation and function of ascorbate peroxidase isoenzymes. J. Exp. Bot. 2002, 53, 1305–1319. [Google Scholar] [CrossRef] [PubMed]
- Gopavajhula, V.R.; Chaitanya, K.V.; Ali Khan, P.A.; Shaik, J.P.; Reddy, P.N.; Alanazi, M. Modeling and analysis of soybean (Glycine max. L.) Cu/Zn, Mn and Fe superoxide dismutases. Genet. Mol. Biol. 2013, 36, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Gregersen, P.L.; Culetic, A.; Boschian, L.; Krupinska, K. Plant senescence and crop productivity. Plant Mol. Biol. 2013, 82, 603–622. [Google Scholar] [CrossRef] [PubMed]
- Kusaba, M.; Tanaka, A.; Tanaka, R. Stay-green plants: What do they tell us about the molecular mechanism of leaf senescence. Photosynth. Res. 2013, 117, 221–234. [Google Scholar] [CrossRef] [PubMed]
- Thomas, H.; Ougham, H. The stay-green trait. J. Exp. Bot. 2014, 65, 3889–3900. [Google Scholar] [CrossRef] [PubMed]
- Hörtensteiner, S. Stay-green regulates chlorophyll and chlorophyll-binding protein degradation during senescence. Trends Plant Sci. 2009, 14, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Grbić, V.; Bleecker, A.B. Ethylene regulates the timing of leaf senescence in Arabidopsis. Plant J. 1995, 8, 595–602. [Google Scholar] [CrossRef]
- Oh, S.A.; Park, J.H.; Lee, G.I.; Paek, K.H.; Park, S.K.; Nam, H.G. Identification of three genetic loci controlling leaf senescence in Arabidopsis thaliana. Plant J. 1997, 12, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Gan, S.S. An abscisic acid-AtNAP transcription factor-SAG113 protein phosphatase 2C regulatory chain for controlling dehydration in senescing Arabidopsis leaves. Plant Physiol. 2012, 158, 961–969. [Google Scholar] [CrossRef] [PubMed]
- Sharabi-Schwager, M.; Lers, A.; Samach, A.; Guy, C.L.; Porat, R. Overexpression of the CBF2 transcriptional activator in Arabidopsis delays leaf senescence and extends plant longevity. J. Exp. Bot. 2010, 61, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Rolando, J.L.; Ramírez, D.A.; Yactayo, W.; Monneveux, P.; Quiroz, R. Leaf greenness as a drought tolerance related trait in potato (Solanum tuberosum L.). Environ. Exp. Bot. 2015, 110, 27–35. [Google Scholar] [CrossRef]
- Lenis, J.I.; Calle, F.; Jaramillo, G.; Perez, J.C.; Ceballos, H.; Cock, J.H. Leaf retention and cassava productivity. Field Crops Res. 2006, 95, 126–134. [Google Scholar] [CrossRef]
- Borras, L.; Slafer, G.A.; Otegui, M.E. Seed dry weight response to source-sink manipulations in wheat, maize and soybean: A quantitative reappraisal. Field Crops Res. 2004, 86, 131–146. [Google Scholar] [CrossRef]
- Gentinetta, E.; Ceppl, D.; Lepori, C.; Perico, G.; Motto, M.; Salamini, F. A major gene for delayed senescence in maize. Pattern of photosynthates accumulation and inheritance. Plant Breed. 1986, 97, 193–203. [Google Scholar] [CrossRef]
- Ding, L.; Wang, K.J.; Jiang, G.M.; Liu, M.Z.; Niu, S.L.; Gao, L.M. Post anthesis changes in photosynthetic traits of maize hybrids released in different years. Field Crop Res. 2005, 93, 108–115. [Google Scholar] [CrossRef]
- Ding, L.; Wang, K.J.; Jiang, G.M.; Liu, M.Z.; Gao, L.M. Photosynthetic rate and yield formation in different maize hybrids. Biol. Plant 2007, 51, 165–168. [Google Scholar] [CrossRef]
- Messmer, R.; Fracheboud, Y.; Bänziger, M.; Stamp, P.; Ribaut, J.-M. Drought stress and tropical maize: QTLs for leaf greenness, plant senescence, and root capacitance. Field Crop Res. 2011, 124, 93–103. [Google Scholar] [CrossRef]
- Borrell, A.K.; Hammer, G.L.; Douglas, A.C.L. Does maintaining green leaf area in sorghum improve yield under drought? I. Leaf growth and senescence. Crop Sci. 2000, 40, 1026–1037. [Google Scholar] [CrossRef]
- Borrell, A.K.; Hammer, G.L.; Henzell, R.G. Does maintaining green leaf area in sorghum improve yield under drought? II. Dry matter production and yield. Crop Sci. 2000, 40, 1037–1048. [Google Scholar] [CrossRef]
- Jordan, D.R.; Hunt, C.H.; Cruickshank, A.W.; Borrell, A.K.; Henzell, R.G. The relationship between the stay-green trait and grain yield in elite sorghum hybrids grown in a range of environments. Crop Sci. 2011, 52, 1153–1161. [Google Scholar] [CrossRef]
- Tian, F.X.; Zhang, M.; Wang, X.; Chen, Y.H.; Wang, W. Antioxidative defence under drought stress in a wheat stay-green mutant. Biol. Plant 2015, 59, 123–130. [Google Scholar] [CrossRef]
- Kooyers, N.J. The evolution of drought escape and avoidance in natural herbaceous populations. Plant Sci. 2015, 234, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Monneveux, P.; Ramínez, D.A.; Pino, M.-T. Drought tolerance in potato (S. tuberosum L.): Can we learn from drought tolerance research in cereals? Plant Sci. 2013, 205, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Sprenger, H.; Rudack, K.; Schudoma, C.; Neumann, A.; Seddig, S.; Peters, R.; Zuther, E.; Kopka, J.; Hincha, D.K.; Walther, D.; et al. Assessment of drought tolerance and its potential yield penalty in potato. Funct. Plant Biol. 2015, 42, 655–667. [Google Scholar] [CrossRef]
- Wilkinson, S.; Kudoyarova, G.R.; Veselov, D.S.; Arkhipova, T.N.; Davies, W.J. Plant hormone interactions: Innovative targets for crop breeding and management. J. Exp. Bot. 2012, 63, 3499–3509. [Google Scholar] [CrossRef] [PubMed]
- Cominelli, E.; Conit, L.; Tonelli, C.; Galbiati, M. Challenges and perspectives to improve crop drought and salinity tolerance. New Biotechnol. 2013, 30, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Cabello, J.V.; Loydeyri, A.F.; Zurbriggen, M.D. Novel perspectives for the engineering of abiotic stress tolerance in plants. Curr. Opin. Plant Biol. 2014, 26, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Dolferus, R. To grow or not to grow: A stressful decision for plants. Plant Sci. 2014, 229, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Alter, S.; Bader, K.C.; Spannagl, M.; Wang, Y.; Bauer, E.; Schön, C.-C.; Mayer, K.F.X. DroughtDB: An expert-curated compilation of plant drought stress genes and their homologs in nine species. Database 2015, 2015, bav046. [Google Scholar] [CrossRef] [PubMed]
- Galindo-González, L.; Pinzón-Latorre, D.; Bergen, A.E.; Jensen, C.D.; Deyholos, M.K. Ion torrent sequencing as a tool for mutation discovery in the flax (Linum usitatissimum L.) genome. Plant Methods 2015, 11, 19. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Liao, F.; Wang, F.; Wen, W.; Li, J.; Mei, H.; Luo, L. Identification of rice transcription factors associated with drought tolerance using ecotilling method. PLoS ONE 2012, 7, e30765. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Sukiran, N.L.; Ma, H.; Su, Z. Moderate drought causes dramatic floral transcriptomic reprogramming to ensure successful reproductive development in Arabidopsis. BMC Plant Biol. 2014, 14, 164. [Google Scholar] [CrossRef] [PubMed]
- Nakaya, A.; Isobe, S.N. Will genomic selection be a practical method for plant breeding? Ann. Bot. 2012, 110, 13003–13016. [Google Scholar] [CrossRef] [PubMed]
- Plauborg, F.; Abrahamsen, P.; Gjettermann, B.; Mollerup, M.; Iversen, B.V.; Liu, F.; Andersen, M.N.; Hansen, S. Modelling of root ABA synthesis, stomatal conductance, transpiration and potato production under water saving irrigation regimes. Agric. Water Manag. 2010, 98, 425–439. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krannich, C.T.; Maletzki, L.; Kurowsky, C.; Horn, R. Network Candidate Genes in Breeding for Drought Tolerant Crops. Int. J. Mol. Sci. 2015, 16, 16378-16400. https://doi.org/10.3390/ijms160716378
Krannich CT, Maletzki L, Kurowsky C, Horn R. Network Candidate Genes in Breeding for Drought Tolerant Crops. International Journal of Molecular Sciences. 2015; 16(7):16378-16400. https://doi.org/10.3390/ijms160716378
Chicago/Turabian StyleKrannich, Christoph Tim, Lisa Maletzki, Christina Kurowsky, and Renate Horn. 2015. "Network Candidate Genes in Breeding for Drought Tolerant Crops" International Journal of Molecular Sciences 16, no. 7: 16378-16400. https://doi.org/10.3390/ijms160716378