Structural Variation (SV) Markers in the Basidiomycete Volvariella volvacea and Their Application in the Construction of a Genetic Map
Abstract
:1. Introduction
2. Results
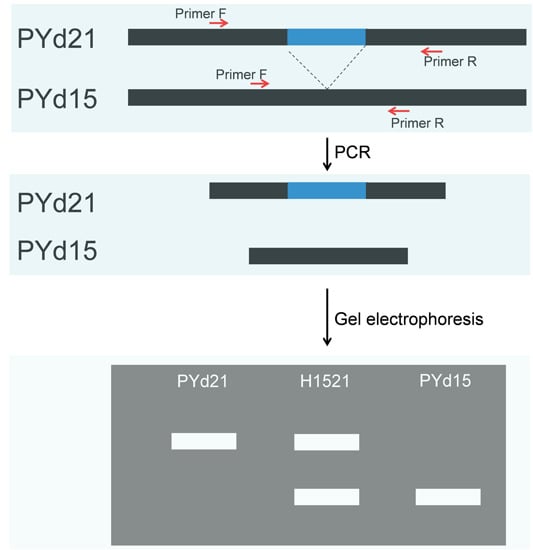
2.1. Developed Structural Variation (SV) Markers
| SV Kind | Number | Rate | Length |
|---|---|---|---|
| insertion | 559 | 59.28 | 160,549 |
| deletion | 371 | 39.34 | 448,893 |
| duplication | 11 | 1.17 | 88,643 |
| inversion | 1 | 0.11 | 47,707 |
| complex | 1 | 0.11 | 1682 |
| Total | 943 | – | 747,474 |

2.2. Genetic Map Construction
| Linkage Group | GD (cM) | No. of Markers | Average Marker Spacing (cM) | No. of Assembled Scaffolds | The Sizes of Assembled Scaffolds (kb) |
|---|---|---|---|---|---|
| Group 1 | 118.211 | 28 | 4.22 | 10 | 4543 |
| Group 2 | 70.185 | 13 | 5.40 | 4 | 2742 |
| Group 3 | 38.397 | 8 | 4.80 | 6 | 661 |
| Group 4 | 108.488 | 18 | 6.03 | 7 | 3136 |
| Group 5 | 34.148 | 10 | 3.41 | 2 | 2395 |
| Group 6 | 17.118 | 6 | 2.85 | 5 | 391 |
| Group 7 | 2.681 | 5 | 0.54 | 1 | 391 |
| Group 8 | 19.473 | 5 | 3.89 | 3 | 3460 |
| Group 9 | 1.605 | 5 | 0.32 | 1 | 880 |
| Group 10 | 1.295 | 4 | 0.32 | 4 | 1048 |
| SUM | 411.601 | 102 | 4.04 | 43 | 19,647 |
| AVERAGE | 4.035 | 10 | 0.40 | – | – |

2.3. Scaffold Anchoring
3. Discussion
4. Experimental Section
4.1. Strains and Growth Conditions

4.2. Genomic DNA Extraction and Genomic Sequencing
4.3. Search SV Loci and Markers Development

4.4. PCR Amplification and Marker Scoring
4.5. Linkage Analysis and Mapping
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Robarts, D.W.H.; Wolfe, A.D. Sequence-related amplified polymorphism (SRAP) markers: A potential resource for studies in plant molecular biology. Appl. Plant Sci. 2014, 2, 1400017. [Google Scholar] [CrossRef] [PubMed]
- Botstein, D.; White, R.L.; Skolnick, M.; Davis, R.W. Construction of a genetic linkage map in man using restriction fragment length polymorphisms. Am. J. Hum. Genet. 1980, 32, 314. [Google Scholar] [PubMed]
- Williams, J.G.K.; Kubelik, A.R.; Livak, K.J.; Rafalski, A.; Tingey, S.V. DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Res. 1990, 18, 6531–6535. [Google Scholar] [CrossRef] [PubMed]
- Ellis, J. Proteins as molecular chaperones. Nature 1987, 328, 378. [Google Scholar] [CrossRef] [PubMed]
- Black, W.C. PCR with arbitrary primers: Approach with care. Insect Mol. Biol. 1993, 2, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Murayama, S.; Yamagishi, H.; Terachi, T. Identification of RAPD and SCAR markers linked to a restorer gene for Ogura cytoplasmic male sterility in padish (Raphanus sativus L.) by bulked seregant analysis. Breed. Sci. 1999, 49, 115–121. [Google Scholar] [CrossRef]
- Velappan, N.; Snodgrass, J.L.; Hakovirta, J.R.; Marronea, B.L.; Burde, S. Rapid identification of pathogenic bacteria by single-enzyme amplified fragment length polymorphism analysis. Diagn. Microbiol. Dis. 2001, 39, 77–83. [Google Scholar] [CrossRef]
- Li, G.; Quiros, C.F. Sequence-related amplified polymorphism (SRAP), a new marker system based on a simple PCR reaction: Its application to mapping and gene tagging in Brassica. Theor. Appl. Genet. 2001, 103, 455–461. [Google Scholar] [CrossRef]
- Zietkiewicz, E.; Rafalski, A.; Labuda, D. Genome fingerprinting by simple sequence repeat (SSR)-anchored polymerase chain reaction amplification. Genomics 1994, 20, 176–183. [Google Scholar] [CrossRef] [PubMed]
- McCouch, S.R.; Chen, X.; Panaud, O.; Temnykh, S.; Xu, Y.; Cho, Y.G.; Huang, N.; Lshii, T.; Blair, M. Microsatellite marker development, mapping and applications in rice genetics and breeding. Plant Mol. Biol. 1997, 35, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Larraya, L.M.; Pérez, G.; Ritter, E.; Pisabarro, A.G.; Ramı́rez, L. Genetic linkage map of the edible basidiomycete Pleurotus ostreatus. Appl. Environ. Microb. 2000, 66, 5290–5300. [Google Scholar] [CrossRef]
- Reddy, M.P.; Sarla, N.; Siddiq, E.A. Inter simple sequence repeat (ISSR) polymorphism and its application in plant breeding. Euphytica 2002, 128, 9–17. [Google Scholar] [CrossRef]
- Meksem, K.; Kahl, G. The Handbook of Plant Genome Mapping: Genetic and Physical Mapping; Wiley-VCH Verlag GMCH & CO. KgaA: Weinheim, Germany, 2005. [Google Scholar]
- Cho, R.J.; Mindrinos, M.; Richards, D.R.; Sapolsky, R.J.; Anderson, M.; Drenkard, E.; Dewdney, J.; Reuber, T.L.; Stammers, M.; Federspiel, N.; et al. Genome-wide mapping with biallelic markers in Arabidopsis thaliana. Nat. Genet. 1999, 23, 203–207. [Google Scholar] [PubMed]
- Wang, C.M.; Liu, P.; Yi, C.; Gu, K.; Sun, F.; Li, L.; Lo, L.C.; Liu, X.; Feng, F.; Lin, G.; et al. A first generation microsatellite-and SNP-based linkage map of Jatropha. PLoS ONE 2011, 6, e23632. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.Y.S.; Barratt, B.J.; Clayton, D.G.; Todd, J.A. Genome-wide association studies: Theoretical and practical concerns. Nat. Rev. Genet. 2005, 6, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Feuk, L.; Carson, A.R.; Scherer, S.W. Structural variation in the human genome. Nat. Rev. Genet. 2006, 7, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zheng, H.; Luo, R.; Wu, H.; Zhu, H.; Li, R.; Cao, H.; Wu, B.; Huang, S.; Shao, H.; et al. Structural variation in two human genomes mapped at single-nucleotide resolution by whole genome de novo assembly. Nat. Biotechnol. 2011, 29, 723–730. [Google Scholar] [CrossRef] [PubMed]
- Kidd, J.M.; Cooper, G.M.; Donahue, W.F.; Hayden, H.S.; Sampas, N.; Graves, T.; Hansen, N.; Teague, B.; Alkan, C.; Antonacci, F.; et al. Mapping and sequencing of structural variation from eight human genomes. Nature 2008, 453, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Dooner, H.K.; He, L. Maize genome structure variation: Interplay between retrotransposon polymorphisms and genic recombination. Plant Cell Online 2008, 20, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Zhao, H.; Kou, Q.; Jiang, J.; Guo, S.; Zhang, H.; Hou, W.; Zou, X.; Sun, H.; Gong, G.; et al. A high resolution genetic map anchoring scaffolds of the sequenced watermelon genome. PLoS ONE 2012, 7, e29453. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.; Aerts, A.; Ahrén, D.; Brun, A.; Danchin, E.G.J.; Duchaussoy, D.F.; Gibon, J.; Kohler, A.; Lindquist, E.; Pereda, V.; et al. The genome of Laccaria bicolor provides insights into mycorrhizal symbiosis. Nature 2008, 452, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Ohm, R.A.; de Jong, J.F.; Lugones, L.G.; Aerts, A.; Kothe, E.; Stajich, J.E.; de Vries, R.P.; Record, E.; Levasseur, A.; Baker, S.E.; et al. Genome sequence of the model mushroom Schizophyllum commune. Nat. Biotechnol. 2010, 28, 957–963. [Google Scholar] [CrossRef] [PubMed]
- Stajich, J.E.; Wilke, S.K.; Ahrén, D.; Au, C.H.; Birren, B.W.; Borodovsky, M.; Burns, C.; Canbäck, B.; Casselton, L.A.; Cheng, C.K.; et al. Insights into evolution of multicellular fungi from the assembled chromosomes of the mushroom Coprinopsis cinerea (Coprinus cinereus). Proc. Natl. Acad. Sci. USA 2010, 107, 11889–11894. [Google Scholar] [CrossRef]
- Chen, S.; Xu, J.; Liu, C.; Zhu, Y.; Nelson, D.R.; Zhou, S.; Li, C.; Wang, L.; Guo, X.; Sun, Y.; et al. Genome sequence of the model medicinal mushroom Ganoderma lucidum. Nat. Commun. 2012, 3, 913. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Gong, J.; Dai, W.; Kang, X.; Huang, Z.; Zhang, H.-M.; Liu, W.; Liu, L.; Ma, J.; Xia, Z.; et al. The genome of Ganderma lucidum provide insights into triterpense biosynthesis and wood degradation. PLoS ONE 2012, 7, e36146. [Google Scholar] [CrossRef] [PubMed]
- Bao, D.; Gong, M.; Zheng, H.; Chen, M.; Zhang, L.; Wang, H.; Jiang, J.; Wu, L.; Zhu, Y.; Zhu, G.; et al. Sequencing and comparative analysis of the straw mushroom (Volvariella volvacea) genome. PLoS ONE 2013, 8, e58294. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Gui, F.; Xie, B.; Deng, Y.; Sun, X.; Lin, M.; Tao, Y.; Li, S. Composition and expression of genes encoding carbohydrate-active enzymes in the straw-degrading mushroom Volvariella volvacea. PLoS ONE 2013, 8, e58780. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.J.; Baek, J.H.; Lee, S.; Kim, C.; Rhee, H.; Kim, H.; Seo, J.-S.; Park, H.-R.; Yoon, D.-E.; Nam, J.-Y.; et al. Whole genome and global gene expression analyses of the model mushroom Flammulina velutipes reveal a high capacity for lignocellulose degradation. PLoS ONE 2014, 9, e93560. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Liu, F.; Jiang, Y.; Wu, G.; Guo, L.; Chen, R.; Chen, B.; Lu, Y.; Dai, Y.; Xie, B. The multigene family of fungal laccases and their expression in the white rot basidiomycete Flammulina velutipes. Gene 2015, 563, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, E.P. Marker-Assisted Selection: Current Status and Future Perspectives in Crops, Livestock, Forestry and Fish; Food & Agriculture Organization of the United Nations: Rome, Italy, 2007. [Google Scholar]
- Zheng, Y.; Zhang, G.; Lin, F.; Wang, Z.; Jin, G.; Yang, L.; Wang, Y.; Chen, X.; Xu, Z.; Zhao, X.; et al. Development of microsatellite markers and construction of genetic map in rice blast pathogen Magnaporthe grisea. Fungal Genet. Biol. 2008, 45, 1340–1347. [Google Scholar] [CrossRef] [PubMed]
- Harushima, Y.; Yano, M.; Shomura, A.; Sato, M.; Shimano, T.; Kuboki, Y.; Yamamoto, T.; Lin, S.Y.; Antonio, B.A.; Parco, A.; et al. A highdensity rice genetic linkage map with 2275 markers using a single F2 population. Genetics 1998, 148, 479–494. [Google Scholar] [PubMed]
- Chang, S.T. Volvariella volvacea. In The Biology and Cultivation of Edible Mushroom; Chang, S.T., Hayes, W.A., Eds.; Academic Press: New York, NY, USA, 1978; pp. 573–605. [Google Scholar]
- Paran, I.; Michelmore, R.W. Development of reliable PCR-based markers linked to downy mildew resistance genes in lettuce. Theor. Appl. Genet. 1993, 85, 985–993. [Google Scholar] [CrossRef] [PubMed]
- Kong, Q.F. SCAR Markers Used in Constructing Genetic Linkage Map of Volvariella volvacea. Master’s Thesis, Fujian Agriculture and Forestry University, Fuzhou, China, 10 April 2010. [Google Scholar]
- Muraguchi, H.; Ito, Y.; Kamada, T.; Yanagi, S.O. A linkage map of the basidiomycete Coprinus cinereus based on random amplified polymorphic DNAs and restriction fragment length polymorphisms. Fungal Genet. Biol. 2003, 40, 93–102. [Google Scholar] [CrossRef]
- Labbé, J.; Zhang, X.; Yin, T.; Schmutz, J.; Grimwood, J.; Martin, F.; Tuskan, G.A.; Tacon, F.L. A genetic linkage map for the ectomycorrhizal fungus Laccaria bicolor and its alignment to the whole-genome sequence assemblies. New Phytol. 2008, 180, 316–328. [Google Scholar] [CrossRef] [PubMed]
- Okuda, Y.; Murakami, S.; Matsumoto, T. A genetic linkage map of Pleurotus pulmonarius based on AFLP markers, and localization of the gene region for the sporeless mutation. Genome 2009, 52, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Kerrigan, R.W.; Royer, J.C.; Baller, L.M.; Kohli, Y.; Horgen, P.A.; Anderson, J.B. Meiotic behavior and linkage relationships in the secondarily homothallic fungus Agaricus bisporus. Genetics 1993, 133, 225–236. [Google Scholar] [PubMed]
- Moquet, F.; Desmerger, C.; Mamoun, M.; Ramos-Guedes-Lafargue, M.; Olivier, J.M. A quantitative trait locus of Agaricus bisporus resistance to Pseudomonas tolaasii is closely linked to natural cap color. Fungal Genet. Biol. 1999, 28, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Foulongne-Oriol, M.; Spataro, C.; Cathalot, V.; Monllor, S.; Savoie, J.M. An expanded genetic linkage map of an intervarietal Agaricus bisporus var. bisporus × A. bisporus var. burnettii hybrid based on AFLP, SSR and CAPS markers sheds light on the recombination behavior of the species. Fungal Genet. Biol. 2010, 47, 226–236. [Google Scholar] [CrossRef] [PubMed]
- Okuda, Y.; Ueda, J.; Obatake, Y.; Murakami, S.; Fukumasa, Y.; Matsumoto, T. Construction of a genetic linkage map based on amplified fragment length polymorphism markers and development of sequence-tagged site markers for marker-assisted selection of the sporeless trait in the oyster mushroom (Pleurotus eryngii). Appl. Environ. Microb. 2012, 78, 1496–1504. [Google Scholar] [CrossRef] [PubMed]
- Tanesaka, E.; Honda, R.; Sasaki, S.; Yoshida, M. Assignment of RAPD marker probes designed from 12 linkage groups of Flammulina velutipes to CHEF-separated chromosomal DNAs. Mycoscience 2012, 53, 238–243. [Google Scholar] [CrossRef]
- Kwan, H.S.; Xu, H.L. Construction of a genetic linkage map of shiitake mushroom Lentinula edodes strain L-54. J. Biochem. Mol. Biol. 2002, 35, 465–471. [Google Scholar] [PubMed]
- Terashima, K.; Matsumoto, T.; Hayashi, E.; Fukumasa-Nakai, Y. A genetic linkage map of Lentinula edodes (shiitake) based on AFLP markers. Mycol. Res. 2002, 106, 911–917. [Google Scholar] [CrossRef]
- Terashima, K.; Matsumoto, T.; Hayashi, E.; Kawasaki, S.; Fukumasa-Nakai, Y. Construction of a linkage map of Lentinula edodes (shiitake) with the HEGS (high-efficiency genome scanning) system: Use of versatile AFLP and PCR-based gene markers. Mycoscience 2006, 47, 336–346. [Google Scholar] [CrossRef]
- Miyazaki, K.; Huang, F.; Zhang, B.; Shiraishi, S.; Sakai, M.; Shimaya, C.; Shishido, K. Genetic map of a basidiomycete fungus, Lentinula edodes (shiitake mushroom), constructed by tetrad analysis. Breed. Sci. 2008, 58, 23–30. [Google Scholar] [CrossRef]
- Gong, W.-B.; Liu, W.; Lu, Y.Y.; Bian, Y.-B.; Zhou, Y.; Kwan, H.S.; Cheung, M.K.; Xiao, Y. Constructing a new integrated genetic linkage map and mapping quantitative trait loci for vegetative mycelium growth rate in Lentinula edodes. Fungal Biol-UK 2014, 118, 295–308. [Google Scholar]
- Li, S.F.; Chang, S.T. Variation in the homothallic basidiomycetes Volvariella volvacea. Mushroom Sci. 1979, 10, 171–184. [Google Scholar]
- Chang, S.T.; Miles, P.G.; Wai, C.C. A study of monosporous isolates of Volvariella volvacea. Mushroom Sci. 1981, 11, 603–621. [Google Scholar]
- Li, S.X.; Chang, S.T. Study on the spore pattern in basidium of Volvareilla volvacea. In Science and Cultivation of Edible Fungi; Van Griensven, L.J.L.D., Ed.; CRC Press/Balkema: Boca Raton, FL, USA, 1991; pp. 115–118. [Google Scholar]
- Chang, S.T.; Li, S.X. Genetical studies on the sexuality pattern of Volvariella volvacea. In Science and Cultivation of Edible Fungi; Van Griensven, L.J.L.D., Ed.; CRC Press/Balkema: Boca Raton, FL, USA, 1991; pp. 119–122. [Google Scholar]
- Lichten, M.; Goldman, A.S.H. Meiotic recombination hotspots. Annu. Rev. Genet. 1995, 29, 423–444. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.T.; Yau, C.K. Volvariella volvacea and its life history. Am. J. Bot. 1971, 1971, 552–561. [Google Scholar] [CrossRef]
- Kües, U. Life history and developmental processes in the basidiomycete Coprinus cinereus. Microbiol. Mol. Biol. Rev. 2000, 64, 316–353. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Zhu, H.; Ruan, J.; Qian, W.; Fang, X.; Shi, Z.; Li, Y.; Li, S.; Shan, G.; Kristiansen, K.; et al. De novo assembly of human genomes with massively parallel short read sequencing. Genome Res. 2010, 20, 265–272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lalitha, S. Primer premier 5. Biotech Softw. Internet Rep. 2000, 1, 270–272. [Google Scholar] [CrossRef]
- Lander, E.S.; Green, P.; Abrahamson, J.; Barlow, A.; Daly, M.J.; Lincoln, S.E.; Newburg, L. MAPMAKER: An interactive computer package for constructing primary genetic linkage maps of experimental and natural populations. Genomics 1987, 1, 174–181. [Google Scholar] [CrossRef]
- Lincoln, S.E.; Daly, M.J.; Lander, E.S. Constructing Genetic Linkage Maps with MAPMAKER/EXP Version 3.0: A Tutorial and Reference Manual; Technical Report for a Whitehead Institute for Biomedical Research: Cambridge, MA, USA, 1993; Volume 1993, pp. 78–79. [Google Scholar]
- Liu, R.H.; Meng, J.L. MapDraw: A microsoft excel macro for drawing genetic linkage maps based on given genetic linkage data. Yi Chuan 2003, 25, 317–321. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, W.; Chen, B.; Zhang, L.; Yan, J.; Lu, Y.; Zhang, X.; Jiang, Y.; Wu, T.; Van Peer, A.F.; Li, S.; et al. Structural Variation (SV) Markers in the Basidiomycete Volvariella volvacea and Their Application in the Construction of a Genetic Map. Int. J. Mol. Sci. 2015, 16, 16669-16682. https://doi.org/10.3390/ijms160716669
Wang W, Chen B, Zhang L, Yan J, Lu Y, Zhang X, Jiang Y, Wu T, Van Peer AF, Li S, et al. Structural Variation (SV) Markers in the Basidiomycete Volvariella volvacea and Their Application in the Construction of a Genetic Map. International Journal of Molecular Sciences. 2015; 16(7):16669-16682. https://doi.org/10.3390/ijms160716669
Chicago/Turabian StyleWang, Wei, Bingzhi Chen, Lei Zhang, Junjie Yan, Yuanping Lu, Xiaoyin Zhang, Yuji Jiang, Taju Wu, Arend Frans Van Peer, Shaojie Li, and et al. 2015. "Structural Variation (SV) Markers in the Basidiomycete Volvariella volvacea and Their Application in the Construction of a Genetic Map" International Journal of Molecular Sciences 16, no. 7: 16669-16682. https://doi.org/10.3390/ijms160716669