Mithramycin A Alleviates Osteoarthritic Cartilage Destruction by Inhibiting HIF-2α Expression
Abstract
:1. Introduction
2. Results
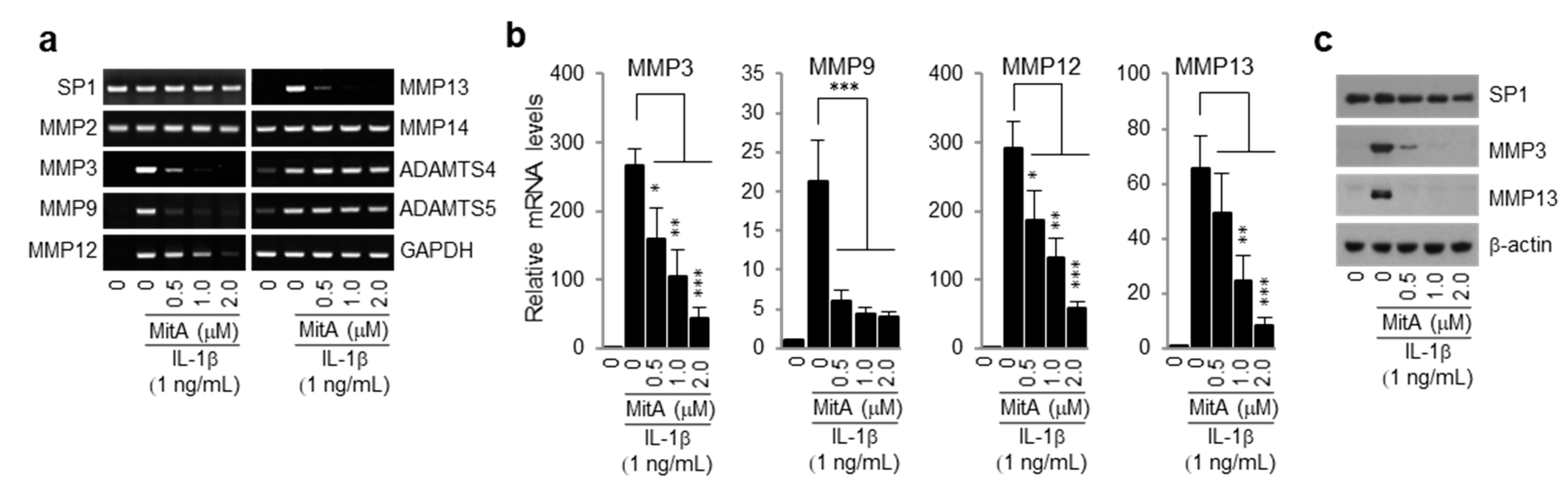
2.1. Mithramycin A (MitA) Inhibits Induction of MMPs by IL-1β in Mouse Primary Chondrocytes
2.2. MitA Administration to Mouse Knee Joint Delays the Progression of Experimental OA
2.3. Effect of MitA on OA is Independent of SP1 Level
2.4. MitA Suppresses HIF-2α Induction by Inhibiting NF-κB Activation
3. Discussion
4. Materials and Methods
4.1. Chondrocyte Culture and MitA Treatment
4.2. Adenoviruses, Infection of Chondrocytes, and Intra-Articular (IA) Injection in Mice
4.3. RNA Analysis
4.4. Western Blot Analysis and Subcellular Fractionation
4.5. siRNA Knockdown (KD) of SP1 and HIF-2α Reporter Assay
4.6. Mice and Experimental OA
4.7. Histology, Immunohistochemistry, and Skeletal Staining
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| DMM | Destabilization of medial meniscus |
| HIF-2α | Hypoxia-inducible factor-2α |
| IL-1β | Interleukin-1β |
| MitA | Mithramycin A |
| MMP | Matrix metalloproteinase |
| NF-κB | Nuclear factor kappa B |
| OA | Osteoarthritis |
| SP1 | Specificity protein 1 |
References
- Goldring, M.B.; Goldring, S.R. Osteoarthritis. J. Cell. Physiol. 2007, 213, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Ouyang, H.; Dass, C.R.; Xu, J. Current research on pharmacologic and regenerative therapies for osteoarthritis. Bone Res. 2016, 4, 15040. [Google Scholar] [CrossRef] [PubMed]
- Bian, Q.; Wang, Y.J.; Liu, S.F.; Li, Y.P. Osteoarthritis: Genetic factors, animal models, mechanisms, and therapies. Front. Biosci. 2012, 4, 74–100. [Google Scholar] [CrossRef]
- Burr, D.B.; Gallant, M.A. Bone remodelling in osteoarthritis. Nat. Rev. Rheumatol. 2012, 8, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Heinegard, D.; Saxne, T. The role of the cartilage matrix in osteoarthritis. Nat. Rev. Rheumatol. 2011, 7, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the extracellular matrix in development and disease. Nat. Rev. Mol. Cell Biol. 2014, 15, 786–801. [Google Scholar] [CrossRef] [PubMed]
- Blom, A.B.; van Lent, P.L.; Libregts, S.; Holthuysen, A.E.; van der Kraan, P.M.; van Rooijen, N.; van den Berg, W.B. Crucial role of macrophages in matrix metalloproteinase-mediated cartilage destruction during experimental osteoarthritis: Involvement of matrix metalloproteinase 3. Arthritis Rheum 2007, 56, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Glasson, S.S.; Askew, R.; Sheppard, B.; Carito, B.; Blanchet, T.; Ma, H.L.; Flannery, C.R.; Peluso, D.; Kanki, K.; Yang, Z.; et al. Deletion of active adamts5 prevents cartilage degradation in a murine model of osteoarthritis. Nature 2005, 434, 644–648. [Google Scholar] [CrossRef] [PubMed]
- Little, C.B.; Barai, A.; Burkhardt, D.; Smith, S.M.; Fosang, A.J.; Werb, Z.; Shah, M.; Thompson, E.W. Matrix metalloproteinase 13-deficient mice are resistant to osteoarthritic cartilage erosion but not chondrocyte hypertrophy or osteophyte development. Arthritis Rheumatol. 2009, 60, 3723–3733. [Google Scholar] [CrossRef] [PubMed]
- Ritchlin, C. Fibroblast biology. Effector signals released by the synovial fibroblast in arthritis. Arthritis Res. 2000, 2, 356–360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, P.; Guan, P.P.; Guo, C.; Zhu, F.; Konstantopoulos, K.; Wang, Z.Y. Fluid shear stress-induced osteoarthritis: Roles of cyclooxygenase-2 and its metabolic products in inducing the expression of proinflammatory cytokines and matrix metalloproteinases. FASEB J. 2013, 27, 4664–4677. [Google Scholar] [CrossRef] [PubMed]
- Towle, C.A.; Hung, H.H.; Bonassar, L.J.; Treadwell, B.V.; Mangham, D.C. Detection of interleukin-1 in the cartilage of patients with osteoarthritis: A possible autocrine/paracrine role in pathogenesis. Osteoarthr. Cartil. 1997, 5, 293–300. [Google Scholar] [CrossRef]
- Kapoor, M.; Martel-Pelletier, J.; Lajeunesse, D.; Pelletier, J.P.; Fahmi, H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat. Rev. Rheumatol. 2011, 7, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, J.C.; Martel-Pelletier, J.; Pelletier, J.P. The role of cytokines in osteoarthritis pathophysiology. Biorheology 2002, 39, 237–246. [Google Scholar] [PubMed]
- Arend, W.P.; Dayer, J.M. Inhibition of the production and effects of interleukin-1 and tumor necrosis factor alpha in rheumatoid arthritis. Arthritis Rheumatol. 1995, 38, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Fukai, A.; Mabuchi, A.; Ikeda, T.; Yano, F.; Ohba, S.; Nishida, N.; Akune, T.; Yoshimura, N.; Nakagawa, T.; et al. Transcriptional regulation of endochondral ossification by hif-2alpha during skeletal growth and osteoarthritis development. Nat. Med. 2010, 16, 678–686. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Jeon, J.; Shin, M.; Won, Y.; Lee, M.; Kwak, J.S.; Lee, G.; Rhee, J.; Ryu, J.H.; Chun, C.H.; et al. Regulation of the catabolic cascade in osteoarthritis by the zinc-ZIP8-MTF1 axis. Cell 2014, 156, 730–743. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Kim, J.; Ryu, J.H.; Oh, H.; Chun, C.H.; Kim, B.J.; Min, B.H.; Chun, J.S. Hypoxia-inducible factor-2alpha is a catabolic regulator of osteoarthritic cartilage destruction. Nat. Med. 2010, 16, 687–693. [Google Scholar] [CrossRef] [PubMed]
- Liacini, A.; Sylvester, J.; Li, W.Q.; Zafarullah, M. Inhibition of interleukin-1-stimulated MAP kinases, activating protein-1 (AP-1) and nuclear factor kappa B (NF-kappa B) transcription factors down-regulates matrix metalloproteinase gene expression in articular chondrocytes. Matrix Biol. 2002, 21, 251–262. [Google Scholar] [CrossRef]
- Kennedy, B.J.; Torkelson, J.L. Long-term follow-up of stage iii testicular carcinoma treated with mithramycin (plicamycin). Med. Pediatr. Oncol. 1995, 24, 327–328. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.H.; Kennedy, B.J. Mithramycin in the treatment of disseminated testicular neoplasms. N. Engl. J. Med. 1965, 272, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Remsing, L.L.; Bahadori, H.R.; Carbone, G.M.; McGuffie, E.M.; Catapano, C.V.; Rohr, J. Inhibition of c-src transcription by mithramycin: Structure-activity relationships of biosynthetically produced mithramycin analogues using the c-src promoter as target. Biochemistry 2003, 42, 8313–8324. [Google Scholar] [CrossRef] [PubMed]
- Hall, T.J.; Schaeublin, M.; Chambers, T.J. The majority of osteoclasts require mrna and protein synthesis for bone resorption in vitro. Biochem. Biophys. Res. Commun. 1993, 195, 1245–1253. [Google Scholar] [CrossRef] [PubMed]
- Ray, R.; Snyder, R.C.; Thomas, S.; Koller, C.A.; Miller, D.M. Mithramycin blocks protein binding and function of the sv40 early promoter. J. Clin. Investig. 1989, 83, 2003–2007. [Google Scholar] [CrossRef] [PubMed]
- Lombo, F.; Menendez, N.; Salas, J.A.; Mendez, C. The aureolic acid family of antitumor compounds: Structure, mode of action, biosynthesis, and novel derivatives. Appl. Microbiol. Biotechnol. 2006, 73, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Z.; Norflus, F.; Singh, B.; Swindell, M.K.; Buzescu, R.; Bejarano, M.; Chopra, R.; Zucker, B.; Benn, C.L.; DiRocco, D.P.; et al. Sp1 is up-regulated in cellular and transgenic models of huntington disease, and its reduction is neuroprotective. J. Biol. Chem. 2006, 281, 16672–16680. [Google Scholar] [CrossRef] [PubMed]
- Beishline, K.; Azizkhan-Clifford, J. Sp1 and the ‘hallmarks of cancer’. FEBS J. 2015, 282, 224–258. [Google Scholar] [CrossRef] [PubMed]
- Sleiman, S.F.; Langley, B.C.; Basso, M.; Berlin, J.; Xia, L.; Payappilly, J.B.; Kharel, M.K.; Guo, H.; Marsh, J.L.; Thompson, L.M.; et al. Mithramycin is a gene-selective sp1 inhibitor that identifies a biological intersection between cancer and neurodegeneration. J. Neurosci. 2011, 31, 6858–6870. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.S.; Nam, J.S.; Jung, J.Y.; Cho, N.P.; Cho, S.D. Modulation of specificity protein 1 by mithramycin a as a novel therapeutic strategy for cervical cancer. Sci. Rep. 2014, 4, 7162. [Google Scholar] [CrossRef] [PubMed]
- Ryu, H.; Lee, J.; Hagerty, S.W.; Soh, B.Y.; McAlpin, S.E.; Cormier, K.A.; Smith, K.M.; Ferrante, R.J. Eset/setdb1 gene expression and histone h3 (k9) trimethylation in huntington’s disease. Proc. Natl. Acad. Sci. USA 2006, 103, 19176–19181. [Google Scholar] [CrossRef] [PubMed]
- Deacon, K.; Onion, D.; Kumari, R.; Watson, S.A.; Knox, A.J. Elevated SP-1 transcription factor expression and activity drives basal and hypoxia-induced vascular endothelial growth factor (VEGF) expression in non-small cell lung cancer. J. Biol. Chem. 2012, 287, 39967–39981. [Google Scholar] [CrossRef] [PubMed]
- Osada, N.; Kosuge, Y.; Ishige, K.; Ito, Y. Mithramycin, an agent for developing new therapeutic drugs for neurodegenerative diseases. J. Pharmacol. Sci. 2013, 122, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Liacini, A.; Sylvester, J.; Li, W.Q.; Zafarullah, M. Mithramycin downregulates proinflammatory cytokine-induced matrix metalloproteinase gene expression in articular chondrocytes. Arthritis Res. Ther. 2005, 7, R777–R783. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goldring, M.B.; Otero, M.; Plumb, D.A.; Dragomir, C.; Favero, M.; El Hachem, K.; Hashimoto, K.; Roach, H.I.; Olivotto, E.; Borzi, R.M.; et al. Roles of inflammatory and anabolic cytokines in cartilage metabolism: Signals and multiple effectors converge upon MMP-13 regulation in osteoarthritis. Eur. Cell Mater. 2011, 21, 202–220. [Google Scholar] [CrossRef] [PubMed]
- Rigoglou, S.; Papavassiliou, A.G. The NF-kappab signalling pathway in osteoarthritis. Int. J. Biochem. Cell Biol. 2013, 45, 2580–2584. [Google Scholar] [CrossRef] [PubMed]
- Roman-Blas, J.A.; Jimenez, S.A. Nf-kappab as a potential therapeutic target in osteoarthritis and rheumatoid arthritis. Osteoarthr. Cartil. 2006, 14, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Lv, H.; Li, X.; Su, H.; Zhang, X.; Yang, J. Danshen attenuates osteoarthritis-related cartilage degeneration through inhibition of nf-kappab signaling pathway in vivo and in vitro. Biochem. Cell Biol. 2017, 95, 644–651. [Google Scholar] [CrossRef] [PubMed]
- Yao, N.; Chen, N.; Xu, X.; Sun, D.; Liu, W.; Li, G.; Bi, X.; Li, S.; Chen, Z.; Chen, G.; et al. Protective effect of shenmai injection on knee articular cartilage of osteoarthritic rabbits and IL-1beta-stimulated human chondrocytes. Exp. Ther. Med. 2017, 13, 3013–3020. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Li, Y.; Han, R.; Cai, G.; He, C.; Wang, G.; Jia, D. T140 blocks the SDF-1/CXCR4 signaling pathway and prevents cartilage degeneration in an osteoarthritis disease model. PLoS ONE 2017, 12, e0176048. [Google Scholar] [CrossRef] [PubMed]
- Sylvester, J.; Ahmad, R.; Zafarullah, M. Role of sp1 transcription factor in interleukin-1-induced adamts-4 (aggrecanase-1) gene expression in human articular chondrocytes. Rheumatol. Int. 2013, 33, 517–522. [Google Scholar] [CrossRef] [PubMed]
- Marcu, K.B.; Otero, M.; Olivotto, E.; Borzi, R.M.; Goldring, M.B. NF-kappab signaling: Multiple angles to target OA. Curr. Drug Targets 2010, 11, 599–613. [Google Scholar] [CrossRef] [PubMed]
- Madhavan, S.; Anghelina, M.; Sjostrom, D.; Dossumbekova, A.; Guttridge, D.C.; Agarwal, S. Biomechanical signals suppress tak1 activation to inhibit nf-kappab transcriptional activation in fibrochondrocytes. J. Immunol. 2007, 179, 6246–6254. [Google Scholar] [CrossRef] [PubMed]
- Pulai, J.I.; Chen, H.; Im, H.J.; Kumar, S.; Hanning, C.; Hegde, P.S.; Loeser, R.F. Nf-kappa b mediates the stimulation of cytokine and chemokine expression by human articular chondrocytes in response to fibronectin fragments. J. Immunol. 2005, 174, 5781–5788. [Google Scholar] [CrossRef] [PubMed]
- Knobloch, T.J.; Madhavan, S.; Nam, J.; Agarwal, S., Jr.; Agarwal, S. Regulation of chondrocytic gene expression by biomechanical signals. Crit. Rev. Eukaryot. Gene Expr. 2008, 18, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.J.; Chueh, A.C.; Togel, L.; Corner, G.A.; Ahmed, N.; Goel, S.; Byun, D.S.; Nasser, S.; Houston, M.A.; Jhawer, M.; et al. Apoptotic sensitivity of colon cancer cells to histone deacetylase inhibitors is mediated by an sp1/sp3-activated transcriptional program involving immediate-early gene induction. Cancer Res. 2010, 70, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Otjacques, E.; Binsfeld, M.; Rocks, N.; Blacher, S.; Vanderkerken, K.; Noel, A.; Beguin, Y.; Cataldo, D.; Caers, J. Mithramycin exerts an anti-myeloma effect and displays anti-angiogenic effects through up-regulation of anti-angiogenic factors. PLoS ONE 2013, 8, e62818. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Sanyal, S.; Kulkarni, K.K.; Jana, K.; Roy, S.; Das, C.; Dasgupta, D. Anticancer drug mithramycin interacts with core histones: An additional mode of action of the DNA groove binder. FEBS Open Bio 2014, 4, 987–995. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.C.; MaruYama, T.; Chun, C.H.; Park, Y. Alleviation of murine osteoarthritis by cartilage-specific deletion of ikappabzeta. Arthritis Rheumatol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Sleiman, S.F.; Berlin, J.; Basso, M.; Karuppagounder, S.S.; Rohr, J.; Ratan, R.R. Histone deacetylase inhibitors and mithramycin a impact a similar neuroprotective pathway at a crossroad between cancer and neurodegeneration. Pharmaceuticals 2011, 4, 1183–1195. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.C.; Jong, H.S.; Kim, T.Y.; Song, S.H.; Lee, D.S.; Lee, J.W.; Kim, T.Y.; Kim, N.K.; Bang, Y.J. AKAP12/Gravin is inactivated by epigenetic mechanism in human gastric carcinoma and shows growth suppressor activity. Oncogene 2004, 23, 7095–7103. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.C.; Ryu, S.; Hao, R.; Wang, B.; Kapur, M.; Fan, C.M.; Yao, T.P. HDAC4 promotes Pax7-dependent satellite cell activation and muscle regeneration. EMBO Rep. 2014, 15, 1175–1183. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.C.; Cohen, T.J.; Barrientos, T.; Wang, B.; Li, M.; Simmons, B.J.; Yang, J.S.; Cox, G.A.; Zhao, Y.; Yao, T.P. A direct HDAC4-map kinase crosstalk activates muscle atrophy program. Mol. Cell 2012, 47, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Ueta, C.; Iwamoto, M.; Kanatani, N.; Yoshida, C.; Liu, Y.; Enomoto-Iwamoto, M.; Ohmori, T.; Enomoto, H.; Nakata, K.; Takada, K.; et al. Skeletal malformations caused by overexpression of CBFA1 or its dominant negative form in chondrocytes. J. Cell Biol. 2001, 153, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Glasson, S.S.; Chambers, M.G.; Van Den Berg, W.B.; Little, C.B. The OARSI histopathology initiative—Recommendations for histological assessments of osteoarthritis in the mouse. Osteoarthr. Cartil. 2010, 18 (Suppl. 3), S17–S23. [Google Scholar] [CrossRef] [PubMed]




© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, M.-C.; Choi, W.H. Mithramycin A Alleviates Osteoarthritic Cartilage Destruction by Inhibiting HIF-2α Expression. Int. J. Mol. Sci. 2018, 19, 1411. https://doi.org/10.3390/ijms19051411
Choi M-C, Choi WH. Mithramycin A Alleviates Osteoarthritic Cartilage Destruction by Inhibiting HIF-2α Expression. International Journal of Molecular Sciences. 2018; 19(5):1411. https://doi.org/10.3390/ijms19051411
Chicago/Turabian StyleChoi, Moon-Chang, and Woo Hee Choi. 2018. "Mithramycin A Alleviates Osteoarthritic Cartilage Destruction by Inhibiting HIF-2α Expression" International Journal of Molecular Sciences 19, no. 5: 1411. https://doi.org/10.3390/ijms19051411



