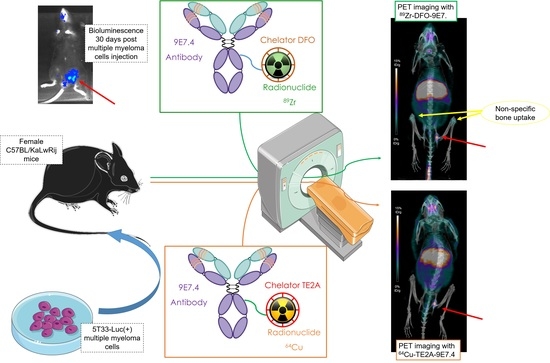
What is the Best Radionuclide for Immuno-PET of Multiple Myeloma? A Comparison Study Between 89Zr- and 64Cu-Labeled Anti-CD138 in a Preclinical Syngeneic Model
Abstract
:1. Introduction
2. Results
2.1. Ex Vivo Biodistribution Experiments
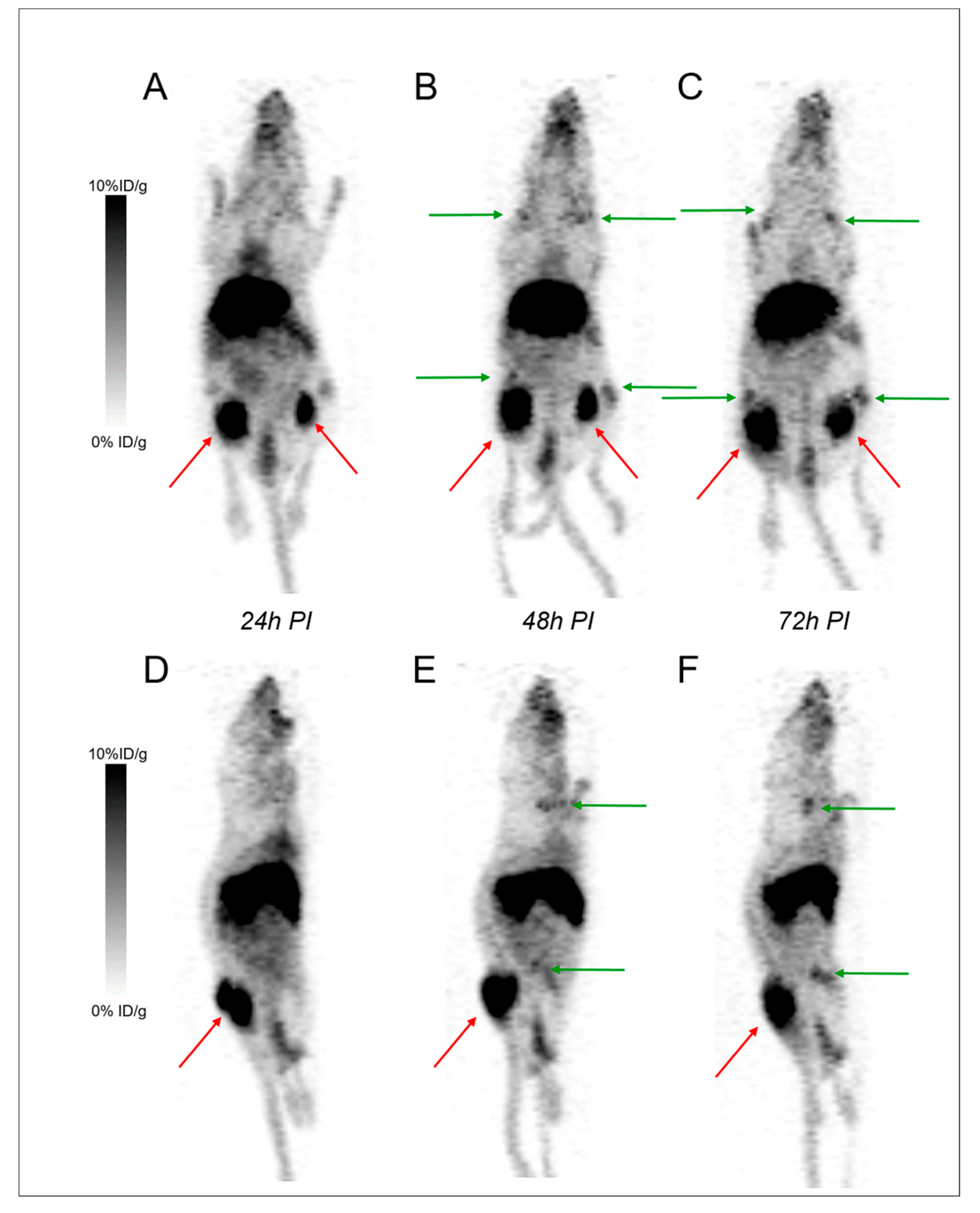
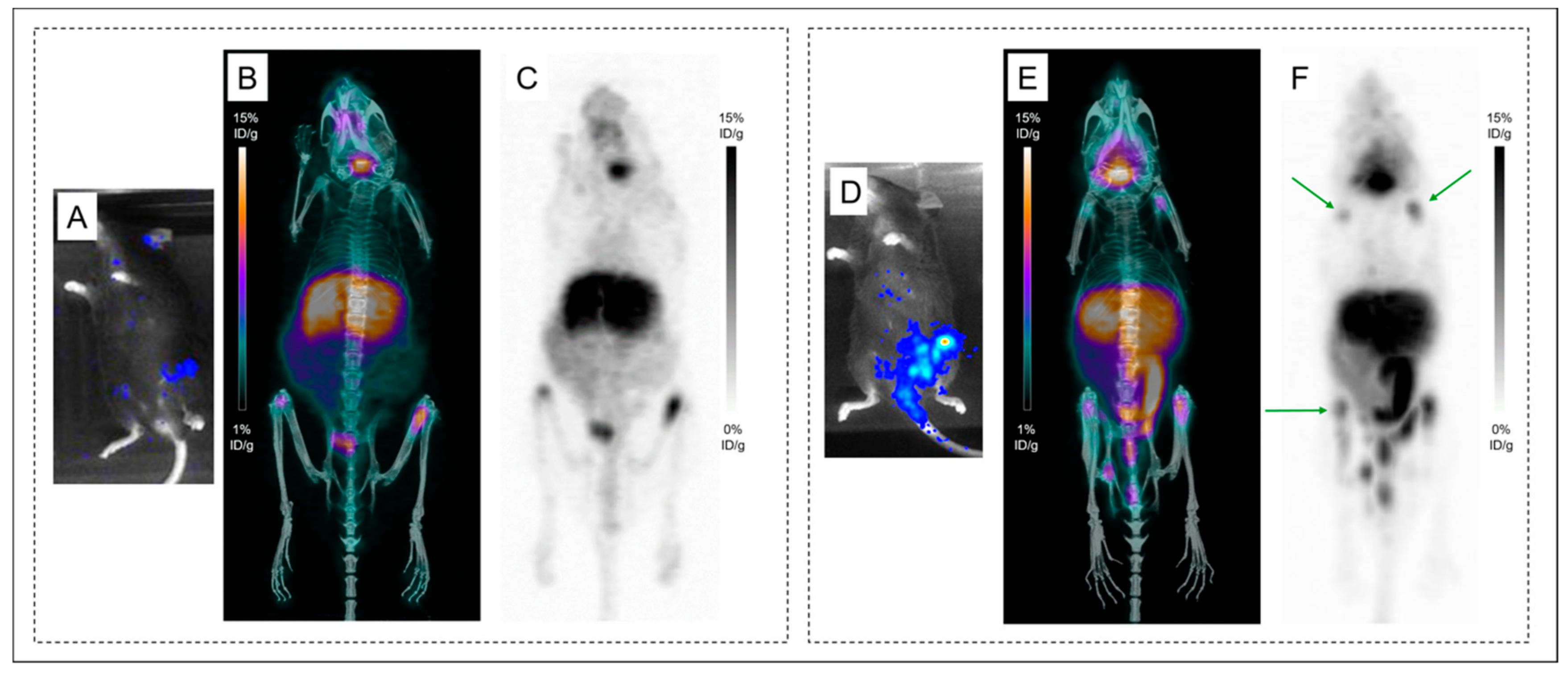
2.2. Small Animal PET and CT Imaging
2.2.1. PET Imaging of Subcutaneous Tumor
2.2.2. PET Imaging of Disseminated Disease
2.2.3. Focus on Bones and Bone Marrows
2.3. Comparison of Both Radioimmunoconjugates
2.3.1. In the Same Mouse
2.3.2. Between Both Experiments
3. Discussion
4. Materials and Methods
4.1. Cell Lines and Cultures
4.2. Preparation of Immunoconjugates and Immuno-PET Tracers
4.2.1. 9E7.4 Antibody
4.2.2. Labeling and Controls with 89Zr
4.2.3. Labeling and Controls with 64Cu
4.2.4. Preparation of 89Zr Oxalate
4.3. Animal Studies
4.3.1. Animal Model: Subcutaneous Tumor Model and IV Disseminated Tumor Model
4.3.2. Bioluminescence Imaging
4.3.3. Small Animal ImmunoPET-CT Imaging
4.3.4. Biodistribution Study
4.3.5. Digital Autoradiography
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 64Cu | copper-64 |
| DFO | desferrioxamine B |
| DFO-SCN | P-isothiocyanatobenzyl-desferrioxamine B; |
| 18F-FDG | [18]-Fluorodeoxyglucose |
| mAb | monoclonal antibody |
| MM | multiple myeloma |
| PET | positron emission tomography |
| PI | post-injection |
| RIT | radioimmunotherapy |
| SC | subcutaneously |
| TE2A | TE2A-benzyl isothiocyanate |
| 89Zr | Zirconium-89 |
References
- Bergsagel, P.L. Where we were, where we are, where we are going: Progress in multiple myeloma. Am. Soc. Clin. Oncol Educ Book 2014, 2014, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Moreau, P.; San Miguel, J.; Sonneveld, P.; Mateos, M.V.; Zamagni, E.; Avet-Loiseau, H.; Hajek, R.; Dimopoulos, M.A.; Ludwig, H.; Einsele, H.; et al. Multiple myeloma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2017, 24, vi133–vi137. [Google Scholar] [CrossRef]
- Harry, P. Recent advances in understanding multiple myeloma. Hematol. Oncol. Stem Cell Ther. 2017, 10, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Brioli, A.; Melchor, L.; Cavo, M.; Morgan, G.J. The impact of intra-clonal heterogeneity on the treatment of multiple myeloma. Br. J. Haematol. 2014, 165, 441–454. [Google Scholar] [CrossRef] [PubMed]
- Lonial, S.; Durie, B.; Palumbo, A.; San-Miguel, J. Monoclonal antibodies in the treatment of multiple myeloma: Current status and future perspectives. Leukemia 2016, 30, 526. [Google Scholar] [CrossRef] [PubMed]
- Bailly, C.; Leforestier, R.; Jamet, B.; Carlier, T.; Bourgeois, M.; Guérard, F.; Touzeau, C.; Moreau, P.; Chérel, M.; Kraeber-Bodéré, F.; et al. PET Imaging for Initial Staging and Therapy Assessment in Multiple Myeloma Patients. Int. J. Mol. Sci. 2017, 18, 445. [Google Scholar] [CrossRef]
- Bailly, C.; Cléry, P.-F.; Faivre-Chauvet, A.; Bourgeois, M.; Guérard, F.; Haddad, F.; Barbet, J.; Chérel, M.; Kraeber-Bodéré, F.; Carlier, T.; et al. Immuno-PET for Clinical Theranostic Approaches. Int. J. Mol. Sci. 2016, 18, 57. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, R.D.; Yang, Y. Syndecan-1: A dynamic regulator of the myeloma microenvironment. Clin. Exp. Metastasis 2008, 25, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; van Andel, H.; de Lau, W.; Hartholt, R.B.; Maurice, M.M.; Clevers, H.; Kersten, M.J.; Spaargaren, M.; Pals, S.T. Syndecan-1 promotes Wnt/β-catenin signaling in multiple myeloma by presenting Wnts and R-spondins. Blood 2017, 131, 982–994. [Google Scholar] [CrossRef] [PubMed]
- Cavo, M.; Terpos, E.; Nanni, C.; Moreau, P.; Lentzsch, S.; Zweegman, S.; Hillengass, J.; Engelhardt, M.; Usmani, S.Z.; Vesole, D.H.; et al. Role of (18)F-FDG PET/CT in the diagnosis and management of multiple myeloma and other plasma cell disorders: A consensus statement by the International Myeloma Working Group. Lancet Oncol. 2017, 18, e206–e217. [Google Scholar] [CrossRef]
- Bailly, C.; Gouard, S.; Lacombe, M.; Saëc, P.R.-L.; Chalopin, B.; Bourgeois, M.; Chouin, N.; Tripier, R.; Halime, Z.; Haddad, F.; et al. Comparison of Immuno-PET of CD138 and PET imaging with 64 CuCl 2 and 18 F-FDG in a preclinical syngeneic model of multiple myeloma. Oncotarget 2018, 9, 9061. [Google Scholar] [CrossRef]
- Kelly, K.R.; Siegel, D.S.; Chanan-Khan, A.A.; Somlo, G.; Heffner, L.T.; Jagannath, S.; Zimmerman, T.; Munshi, N.C.; Madan, S.; Mohrbacher, A.; et al. Indatuximab Ravtansine (BT062) in Combination with Low-Dose Dexamethasone and Lenalidomide or Pomalidomide: Clinical Activity in Patients with Relapsed/Refractory Multiple Myeloma. Blood 2016, 128, 4486. [Google Scholar]
- Heffner, L.T.; Jagannath, S.; Zimmerman, T.M.; Lee, K.P.; Rosenblatt, J.; Lonial, S.; Lutz, R.J.; Czeloth, N.; Osterroth, F.; Ruehle, M.; et al. BT062, an Antibody-Drug Conjugate Directed Against CD138, Given Weekly for 3 Weeks in Each 4 Week Cycle: Safety and Further Evidence of Clinical Activity. Blood 2012, 120, 4042. [Google Scholar]
- Chérel, M.; Gouard, S.; Gaschet, J.; Saï-Maurel, C.; Bruchertseifer, F.; Morgenstern, A.; Bourgeois, M.; Gestin, J.-F.; Bodéré, F.K.; Barbet, J.; et al. 213Bi radioimmunotherapy with an anti-mCD138 monoclonal antibody in a murine model of multiple myeloma. J. Nucl. Med. 2013, 54, 1597–1604. [Google Scholar] [CrossRef]
- Rousseau, C.; Ferrer, L.; Supiot, S.; Bardiès, M.; Davodeau, F.; Faivre-Chauvet, A.; Baumgartner, P.; Wijdenes, J.; Lacombe, M.; Barbet, J.; et al. Dosimetry results suggest feasibility of radioimmunotherapy using anti-CD138 (B-B4) antibody in multiple myeloma patients. Tumour Biol. 2012, 33, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Follacchio, G.A.; De Feo, M.S.; Monteleone, F.; De Vincentis, G.; Liberatore, M. Radiopharmaceuticals labelled with copper radionuclides: Clinical results in human beings. Curr. Radiopharm. 2018, 11, 22–23. [Google Scholar] [CrossRef] [PubMed]
- Jauw, Y.W.S.; Menke-van der Houven van Oordt, C.W.; Hoekstra, O.S.; Hendrikse, N.H.; Vugts, D.J.; Zijlstra, J.M.; Huisman, M.C.; van Dongen, G.A.M.S. Immuno-Positron Emission Tomography with Zirconium-89-Labeled Monoclonal Antibodies in Oncology: What Can We Learn from Initial Clinical Trials? Front. Pharm. 2016, 7, 131. [Google Scholar] [CrossRef] [PubMed]
- Abou, D.S.; Ku, T.; Smith-Jones, P.M. In vivo biodistribution and accumulation of 89Zr in mice. Nucl. Med. Biol. 2011, 38, 675–681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fichou, N.; Gouard, S.; Maurel, C.; Barbet, J.; Ferrer, L.; Morgenstern, A.; Bruchertseifer, F.; Faivre-Chauvet, A.; Bigot-Corbel, E.; Davodeau, F.; et al. Single-Dose Anti-CD138 Radioimmunotherapy: Bismuth-213 is More Efficient than Lutetium-177 for Treatment of Multiple Myeloma in a Preclinical Model. Front. Med. 2015, 2, 76. [Google Scholar] [CrossRef] [Green Version]
- Zeglis, B.M.; Lewis, J.S. The Bioconjugation and Radiosynthesis of 89Zr-DFO-labeled Antibodies. J. Vis. Exp. 2015, 96, e52521. [Google Scholar]
- Holland, J.P.; Divilov, V.; Bander, N.H.; Smith-Jones, P.M.; Larson, S.M.; Lewis, J.S. 89Zr-DFO-J591 for immunoPET of prostate-specific membrane antigen expression in vivo. J. Nucl. Med. 2010, 51, 1293–1300. [Google Scholar] [CrossRef]
- Williams, S.-P. Tissue distribution studies of protein therapeutics using molecular probes: Molecular imaging. AAPS J. 2012, 14, 389–399. [Google Scholar] [CrossRef]
- Chen, F.; Goel, S.; Valdovinos, H.F.; Luo, H.; Hernandez, R.; Barnhart, T.E.; Cai, W. In Vivo Integrity and Biological Fate of Chelator-Free Zirconium-89-Labeled Mesoporous Silica Nanoparticles. ACS Nano 2015, 9, 7950–7959. [Google Scholar] [CrossRef]
- England, C.G.; Ehlerding, E.B.; Hernandez, R.; Rekoske, B.T.; Graves, S.A.; Sun, H.; Liu, G.; McNeel, D.G.; Barnhart, T.E.; Cai, W. Preclinical Pharmacokinetics and Biodistribution Studies of 89Zr-Labeled Pembrolizumab. J. Nucl. Med. 2017, 58, 162–168. [Google Scholar] [CrossRef]
- Wuthier, R.E. A zonal analysis of inorganic and organic constituents of the epiphysis during endochondral calcification. Calcif Tissue Res. 1969, 4, 20–38. [Google Scholar] [CrossRef]
- Rosol, T.J.; Tannehill-Gregg, S.H.; Corn, S.; Schneider, A.; McCauley, L.K. Animal models of bone metastasis. Cancer Treat. Res. 2004, 118, 47–81. [Google Scholar]
- Yoneda, T. Arterial microvascularization and breast cancer colonization in bone. Histol. Histopathol. 1997, 12, 1145–1149. [Google Scholar]
- Hernandez, R.; England, C.G.; Yang, Y.; Valdovinos, H.F.; Liu, B.; Wong, H.C.; Barnhart, T.E.; Cai, W. ImmunoPET imaging of tissue factor expression in pancreatic cancer with 89Zr-Df-ALT-836. J. Control. Release 2017, 264, 160–168. [Google Scholar] [CrossRef]
- Hernandez, R.; Sun, H.; England, C.G.; Valdovinos, H.F.; Ehlerding, E.B.; Barnhart, T.E.; Yang, Y.; Cai, W. CD146-targeted immunoPET and NIRF Imaging of Hepatocellular Carcinoma with a Dual-Labeled Monoclonal Antibody. Theranostics 2016, 6, 1918–1933. [Google Scholar] [CrossRef]
- Aerts, H.J.W.L.; Dubois, L.; Perk, L.; Vermaelen, P.; van Dongen, G.A.M.S.; Wouters, B.G.; Lambin, P. Disparity between in vivo EGFR expression and 89Zr-labeled cetuximab uptake assessed with PET. J. Nucl. Med. 2009, 50, 123–131. [Google Scholar] [CrossRef]
- Perk, L.R.; Visser, G.W.M.; Vosjan, M.J.W.D.; Stigter-van Walsum, M.; Tijink, B.M.; Leemans, C.R.; van Dongen, G.A.M.S. (89)Zr as a PET surrogate radioisotope for scouting biodistribution of the therapeutic radiometals (90)Y and (177)Lu in tumor-bearing nude mice after coupling to the internalizing antibody cetuximab. J. Nucl. Med. 2005, 46, 1898–1906. [Google Scholar]
- Perk, L.R.; Visser, O.J.; Stigter-van Walsum, M.; Vosjan, M.J.W.D.; Visser, G.W.M.; Zijlstra, J.M.; Huijgens, P.C.; van Dongen, G.A.M.S. Preparation and evaluation of (89)Zr-Zevalin for monitoring of (90)Y-Zevalin biodistribution with positron emission tomography. Eur. J. Nucl. Med. Mol. Imaging 2006, 33, 1337–1345. [Google Scholar] [CrossRef]
- Menke-van der Houven van Oordt, C.W.; Gootjes, E.C.; Huisman, M.C.; Vugts, D.J.; Roth, C.; Luik, A.M.; Mulder, E.R.; Schuit, R.C.; Boellaard, R.; Hoekstra, O.S.; et al. 89Zr-cetuximab PET imaging in patients with advanced colorectal cancer. Oncotarget 2015, 6, 30384–30393. [Google Scholar]
- Jansen, M.H.; Veldhuijzen van Zanten, S.E.M.; van Vuurden, D.G.; Huisman, M.C.; Vugts, D.J.; Hoekstra, O.S.; van Dongen, G.A.; Kaspers, G.-J.L. Molecular Drug Imaging: 89Zr-Bevacizumab PET in Children with Diffuse Intrinsic Pontine Glioma. J. Nucl. Med. 2017, 58, 711–716. [Google Scholar] [CrossRef]
- Bahce, I.; Huisman, M.C.; Verwer, E.E.; Ooijevaar, R.; Boutkourt, F.; Vugts, D.J.; van Dongen, G.A.; Boellaard, R.; Smit, E.F. Pilot study of (89)Zr-bevacizumab positron emission tomography in patients with advanced non-small cell lung cancer. EJNMMI Res. 2014, 4, 35. [Google Scholar] [CrossRef]
- Van Loon, J.; Even, A.J.G.; Aerts, H.J.W.L.; Öllers, M.; Hoebers, F.; van Elmpt, W.; Dubois, L.; Dingemans, A.-M.C.; Lalisang, R.I.; Kempers, P.; et al. PET imaging of zirconium-89 labelled cetuximab: A phase I trial in patients with head and neck and lung cancer. Radiother. Oncol. 2017, 122, 267–273. [Google Scholar] [CrossRef]
- Ulaner, G.A.; Hyman, D.M.; Ross, D.S.; Corben, A.; Chandarlapaty, S.; Goldfarb, S.; McArthur, H.; Erinjeri, J.P.; Solomon, S.B.; Kolb, H.; et al. Detection of HER2-Positive Metastases in Patients with HER2-Negative Primary Breast Cancer Using 89Zr-Trastuzumab PET/CT. J. Nucl. Med. 2016, 57, 1523–1528. [Google Scholar] [CrossRef]
- Ulaner, G.A.; Hyman, D.M.; Lyashchenko, S.K.; Lewis, J.S.; Carrasquillo, J.A. 89Zr-Trastuzumab PET/CT for Detection of Human Epidermal Growth Factor Receptor 2-Positive Metastases in Patients With Human Epidermal Growth Factor Receptor 2-Negative Primary Breast Cancer. Clin. Nucl. Med. 2017, 42, 912–917. [Google Scholar] [CrossRef]
- Natarajan, A.; Mayer, A.T.; Reeves, R.E.; Nagamine, C.M.; Gambhir, S.S. Development of Novel ImmunoPET Tracers to Image Human PD-1 Checkpoint Expression on Tumor-Infiltrating Lymphocytes in a Humanized Mouse Model. Mol. Imaging Biol. 2017, 19, 903–914. [Google Scholar] [CrossRef] [Green Version]
- Ghai, A.; Maji, D.; Cho, N.; Chanswangphuwana, C.; Rettig, M.; DiPersio, J.; Akers, W.; Dehdashti, F.; Achilefu, S.; Vij, R.; et al. Preclinical development of CD38-targeted (89Zr)Zr-DFO-daratumumab for imaging multiple myeloma. J. Nucl. Med. 2018, 59, 216–222. [Google Scholar] [CrossRef]
- Caserta, E.; Chea, J.; Minnix, M.; Viola, D.; Vonderfecht, S.; Yazaki, P.; Crow, D.; Khalife, J.; Sanchez, J.F.; Palmer, J.M.; et al. Copper-64-labeled daratumumab as a PET/CT imaging tracer for multiple myeloma. Blood 2018, 131, 741–745. [Google Scholar] [CrossRef]
- Heskamp, S.; Raavé, R.; Boerman, O.; Rijpkema, M.; Goncalves, V.; Denat, F. 89Zr-Immuno-Positron Emission Tomography in Oncology: State-of-the-Art 89Zr Radiochemistry. Bioconjug. Chem. 2017, 28, 2211–2223. [Google Scholar] [CrossRef]
- Ahmedova, A.; Todorov, B.; Burdzhiev, N.; Goze, C. Copper radiopharmaceuticals for theranostic applications. Eur J. Med. Chem 2018, 157, 1406–1425. [Google Scholar] [CrossRef]
- Radl, J.; Croese, J.W.; Zurcher, C.; Van den Enden-Vieveen, M.H.; de Leeuw, A.M. Animal model of human disease. Multiple myeloma. Am. J. Pathol. 1988, 132, 593–597. [Google Scholar]
- Gouard, S.; Pallardy, A.; Gaschet, J.; Faivre-Chauvet, A.; Bruchertseifer, F.; Morgenstern, A.; Maurel, C.; Matous, E.; Kraeber-Bodéré, F.; Davodeau, F.; et al. Comparative analysis of multiple myeloma treatment by CD138 antigen targeting with bismuth-213 and Melphalan chemotherapy. Nucl. Med. Biol. 2014, 41, e30–e35. [Google Scholar] [CrossRef]







| Organs | 89Zr-DFO-9E7.4 (24 h PI) | 89Zr-DFO-9E7.4 (24 h PI) | 89Zr-DFO-9E7.4 (72 h PI) | 89Zr-DFO-9E7.4 (72 h PI) | 89Zr-Oxalate (24 h PI) | 89Zr-Oxalate (24 h PI) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ratio Organs/Blood | Ratio Organs/Blood | Ratio Organs/Blood | ||||||||||
| Median | SD | Median | SD | Median | SD | Median | SD | Median | SD | Median | SD | |
| Tumor | 12.48 | 4.77 | 1.42 | 0.47 | 4.53 | 1.76 | 3.42 | 0.22 | 9.67 | 2.52 | 0.83 | 0.15 |
| Blood | 5.33 | 0.95 | 1.00 | 0.00 | 1.77 | 0.85 | 1.00 | 0.00 | 11.57 | 1.38 | 1.00 | 0.00 |
| Liver | 13.93 | 1.36 | 2.30 | 1.00 | 9.90 | 1.75 | 6.44 | 2.55 | 8.60 | 0.53 | 0.75 | 0.13 |
| Kidney | 5.78 | 2.83 | 0.62 | 0.13 | 3.60 | 1.37 | 2.15 | 0.61 | 12.33 | 1.53 | 1.09 | 0.27 |
| Gut | 7.03 | 0.95 | 1.34 | 0.41 | 6.13 | 2.47 | 3.65 | 0.98 | 5.47 | 1.29 | 0.47 | 0.06 |
| Lungs | 5.70 | 2.79 | 0.86 | 0.22 | 2.77 | 1.14 | 1.60 | 0.16 | 7.90 | 0.79 | 0.69 | 0.01 |
| Muscle | 1.05 | 0.49 | 0.11 | 0.03 | 0.27 | 0.15 | 0.16 | 0.04 | 2.63 | 0.21 | 0.23 | 0.01 |
| Spleen | 10.78 | 1.86 | 2.00 | 0.56 | 7.23 | 1.01 | 4.85 | 2.36 | 10.90 | 1.01 | 0.96 | 0.21 |
| Skin | 3.28 | 0.90 | 0.41 | 0.25 | 1.40 | 0.69 | 0.80 | 0.18 | 3.77 | 0.15 | 0.33 | 0.05 |
| Brain | 0.68 | 0.71 | 0.05 | 0.03 | 0.20 | 0.17 | 0.20 | 0.25 | 0.50 | 0.10 | 0.04 | 0.01 |
| Heart | 4.60 | 2.81 | 0.45 | 0.02 | 0.93 | 0.64 | 0.46 | 0.29 | 8.70 | 1.13 | 0.77 | 0.20 |
| Flat Bone | 2.98 | 1.07 | 0.34 | 0.12 | 3.43 | 1.12 | 2.11 | 0.60 | 39.20 | 2.03 | 3.44 | 0.58 |
| Femur | 3.23 | 1.38 | 0.45 | 0.11 | 3.30 | 0.70 | 2.16 | 0.63 | 38.43 | 1.40 | 3.37 | 0.55 |
| Stomach | 5.30 | 1.35 | 0.62 | 0.46 | 2.77 | 0.45 | 1.82 | 0.76 | 14.67 | 1.53 | 1.29 | 0.28 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bailly, C.; Gouard, S.; Guérard, F.; Chalopin, B.; Carlier, T.; Faivre-Chauvet, A.; Remaud-Le Saëc, P.; Bourgeois, M.; Chouin, N.; Rbah-Vidal, L.; et al. What is the Best Radionuclide for Immuno-PET of Multiple Myeloma? A Comparison Study Between 89Zr- and 64Cu-Labeled Anti-CD138 in a Preclinical Syngeneic Model. Int. J. Mol. Sci. 2019, 20, 2564. https://doi.org/10.3390/ijms20102564
Bailly C, Gouard S, Guérard F, Chalopin B, Carlier T, Faivre-Chauvet A, Remaud-Le Saëc P, Bourgeois M, Chouin N, Rbah-Vidal L, et al. What is the Best Radionuclide for Immuno-PET of Multiple Myeloma? A Comparison Study Between 89Zr- and 64Cu-Labeled Anti-CD138 in a Preclinical Syngeneic Model. International Journal of Molecular Sciences. 2019; 20(10):2564. https://doi.org/10.3390/ijms20102564
Chicago/Turabian StyleBailly, Clément, Sébastien Gouard, François Guérard, Benjamin Chalopin, Thomas Carlier, Alain Faivre-Chauvet, Patricia Remaud-Le Saëc, Mickaël Bourgeois, Nicolas Chouin, Latifa Rbah-Vidal, and et al. 2019. "What is the Best Radionuclide for Immuno-PET of Multiple Myeloma? A Comparison Study Between 89Zr- and 64Cu-Labeled Anti-CD138 in a Preclinical Syngeneic Model" International Journal of Molecular Sciences 20, no. 10: 2564. https://doi.org/10.3390/ijms20102564