1. Introduction
Preeclampsia is a pregnancy-specific disorder characterized by sudden onset of hypertension with either proteinuria or end-organ dysfunction, or both, after the 20th week of gestation in a previously normotensive woman, occurring in 3–5% of pregnancies in industrialized countries [
1,
2].
The pathogenesis of preeclampsia remains poorly understood, even though preeclampsia has been recognized for at least 100 years [
3]. In the last 20 years, multiple theories about the ultimate cause of preeclampsia have been developed with little agreement, except for the conclusion that preeclampsia is a multifactorial disease [
4,
5].
Many approaches have been developed for predicting preeclampsia at an early stage and promising insights have been discovered [
5,
6,
7,
8]. Nevertheless, the etiology of preeclampsia remains incompletely understood, although the placenta has been identified as the central organ in the pathogenesis of preeclampsia. Impaired placentation and placental function in early pregnancy remains the leading hypothesis [
9,
10,
11,
12], while emerging hypotheses focus on the maternal cardiovascular susceptibility to preeclampsia and pregnancy adaptions [
13]. Structural and functional cardiovascular changes were found in women 1 year after preeclamptic pregnancies [
14], where the involvement of angiogenic factors such as soluble fms-like tyrosine kinase-1 (sFlt-1) and placental growth factor (PlGF) and placental factors such as placental protein 13 (PP13) and the dysbalance thereof may be used to predict severity and long-term cardiovascular complications of preeclampsia [
15,
16].
While the identification of etiological factors is without doubt an important task, the management of adverse concomitant effects and consequences of preeclampsia may be even more relevant. Women with pregnancies complicated by preeclampsia appear to be at increased risk of metabolic and cardiovascular diseases in later life, and pregnancy complications and coronary heart disease may have common disease mechanisms [
17,
18]. As cardiovascular disease (CVD) is a leading cause of death, earlier recognition of those at risk seems vital. Therefore, the diagnosis of preeclampsia, or adverse pregnancy more generally, could be an opportunity for the implementation of primary prevention strategies [
17,
19,
20,
21].
Pregnancy is associated with huge cardiovascular and metabolic changes and can be considered as a “stress test” of the somatic and cardiovascular system, suggesting that preeclampsia manifesting in pregnancy is akin to a “failed stress test”. “Failing the stress-test”, that is, absence of the typical autonomically regulated cardiovascular and cardiorespiratory adaptations to pregnancy, may be predictive of cardiovascular disorders in later life, when the system is put under similar strain [
21,
22].
The autonomic nervous system plays a central role in cardiovascular and cardiorespiratory adaptation to pregnancy-related hemodynamic changes [
23,
24,
25]. Previous research has shown that the increases in peripheral vascular resistance and blood pressure that characterize preeclampsia are mediated, at least in part, by a substantial increase in sympathetic vasoconstrictor activity [
26,
27]. Autonomic nervous system functioning during pregnancy can be noninvasively assessed by analyzing continuous measures of cardiovascular variables, baroreceptor reflex sensitivity (BRS), and the mutual adjustment of blood pressure, heart rate, and respiration, partitioned for influences of the sympathetic and the parasympathetic branch of the autonomic nervous system [
28,
29,
30,
31]. Now, if we can determine abnormalities in blood pressure regulation in affected women at a time at which their preeclampsia is no longer present by definition (i.e., from 12 weeks after delivery onwards) by use of a simple and time-efficient test in the laboratory, this test may be used to evaluate the effectiveness of pharmacological or behavioral interventions for reducing affected women’s cardiovascular risk. Therefore, the aim of this study was to follow up on the regulation of the physiological response to everyday stressful events in preeclampsia several weeks postpartum.
3. Discussion
The results of the present study showed that the short-term blood pressure regulation in women previously affected by preeclampsia returns to normal several weeks postpartum. However, heart rate and systemic vascular resistance responses in women previously affected by preeclampsia indicated impaired ability to flexibly respond to moderate stress; this indicates that their overarching regulation mechanisms may be altered after all.
In the healthy organism, physiological mechanisms of blood pressure regulation maintain the arterial blood pressure at a largely constant level even in stressful conditions, ensuring adequate tissue perfusion throughout. However, the intense hemodynamic modifications during pregnancy result in a decrease of baroreceptor sensitivity; this is even more pronounced in preeclampsia, and it jeopardizes the proper adjustment of the physiological factors regulating the arterial blood pressure [
29,
32]. Impaired baroreceptor sensitivity can still be observed after pregnancy, depending on the time since delivery. Walther et al. reported reduced baroreflex sensitivity four days after delivery and concluded that the maternal cardiovascular system is still affected by pregnancy at that time [
33]. With greater distance since delivery, BRS returns to levels recorded at the beginning of pregnancy [
28,
34]. The present findings indicate that this similarly applies to women who have had preeclampsia. Fifteen to seventeen weeks after delivery, the BRS of women with pregnancies complicated by preeclampsia did not differ from that of mothers with uncomplicated pregnancies, and neither did it differ from the BRS of women who did not give birth. Thus, persistently impaired BRS does not seem to explain the increased cardiovascular risk in later life of women who have had preeclampsia.
The assessment of BRS provides some indication of the regulatory activity of the autonomic nervous system in stressful conditions, but for more specific information, more profound analysis of the regulating factors is required. Important additional information is provided by more fine-grained analysis of the variations of the state of the complex regulatory system over time. Phase synchronization indexes, which indicate mutual adjustments of blood pressure, R–R intervals, and respiration across time, supply this information [
35,
36]. However, in the present study, mutual adjustment of the mainly sympathetically modulated low-frequency variations of heart rate and systolic blood pressure did not differ between groups, thus basically confirming the findings obtained from the BRS.
Nevertheless, the conclusion that blood pressure regulation has entirely returned to normal several weeks postpartum is still premature. From the mathematical approach for the estimation of BRS alone, it follows that baroreceptor sensitivity decreases with increasing levels of heart rate. While some heart rate acceleration during demanding conditions is adaptive, not all individuals may show this adaptation to the same extent. In other words, it is vital to also consider the contributing factors when interpreting the presence or absence of differences in baroreceptor sensitivity between groups (i.e., heart rate, stroke volume, and systemic vascular resistance).
To maintain a constant arterial blood pressure—the primary regulated variable in stressful situations—increases of heart rate (or stroke volume) must be compensated by decreased vascular resistance in line with the fundamental Darcy’s law (or Ohm’s law) of hemodynamics. In preeclamptic women, factors such as PP13, responsible for vasodilation and decreased vascular resistance, are reduced early in gestation [
37]. Their dysregulation may thus not only be involved in endothelial dysfunction during pregnancy, but markers such as PlGF and sFlt-1 may also function as predictors for long-term cardiovascular health [
38]. Later in pregnancy, PP13 is massively increased and returns to basal levels within two weeks in healthy women, while it takes more than eight weeks until it returns to normal levels in women who experienced preeclampsia [
39]. Since the effects of PP13 last even after its disappearance, PP13 might still influence the maternal vascular system months after pregnancy [
37]. Maybe only then do the true effects of preeclampsia on the maternal system become obvious.
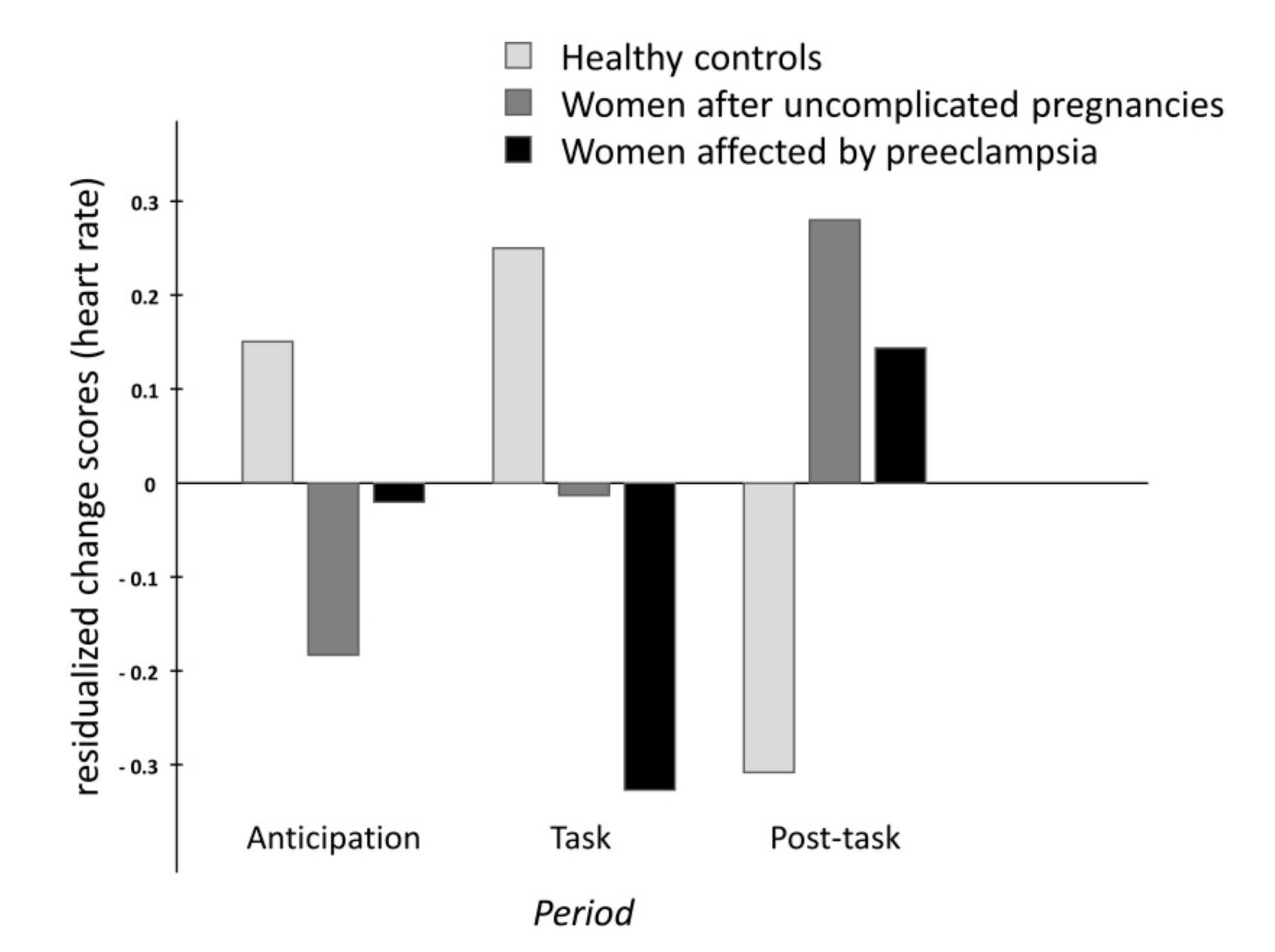
In the present study, heart rate markedly increased and vascular resistance decreased accordingly in response to the stress manipulation only in women without previous pregnancy. By contrast, women who were pregnant, and in particular women with former preeclampsia, hardly showed any heart rate responses at all. Hence, in women recovering from preeclampsia, there was no necessity for short-term blood pressure regulation via activation of baroreceptors and subsequent adaptation of vascular resistance. From this, it follows that the similar outcomes in healthy women and women with preeclampsia in terms of baroreceptor sensitivity and maintenance of largely constant arterial blood pressure levels during brief periods of stress do not justify the conclusion that blood pressure regulation is unimpaired after preeclampsia. Instead, it appears to be that blunted cardiac responses in affected women did not make blood pressure regulation necessary to the same extent as in healthy women.
In women without previous pregnancy, the flexible cardiac response to the active performance task, which provides adaptive energy mobilization and oxygen supply, indicates proper functioning of their autonomic nervous system-mediated cardiovascular regulation [
40]. In contrast, the blunted heart rate response in women previously affected by preeclampsia may be related to their generally elevated levels of systemic vascular resistance, and the more rigid vasoadaptation associated with this. This idea is fueled by the elevated systemic vascular resistance levels in women with a history of preeclampsia observed at baseline, which is in line with previous research [
41].
Various proinflammatory factors, such as TNF-alpha, IL-10, IL-6, leptin, and CX3CL1, are elevated in preeclamptic women [
42,
43,
44] and are also known to mediate cardiovascular remodeling outside of pregnancy, in crosstalk with reactive oxygen species, angiotensin II, and other proinflammatory cytokines [
45,
46,
47,
48,
49]. In pregnancy, vascular remodeling may also be induced by an elevated maternal inflammatory profile. TNF-alpha induces collagen I deposition in the maternal vasculature, and MMP1 and -7 activity induce extracellular matrix degradation [
50], while CYP2J2, elevated in preeclampsia, may also be involved in uteroplacental and vascular remodeling [
51].
Thus, the blunted cardiac responses in women with former preeclampsia may arise from an attempt by the organism to protect itself against undue elevations of arterial blood pressure, which would occur as a result of the failure to lower the systemic vascular resistance synchronously with the rising heart rate (and/or stroke volume). With more severe stress as well as in later life when, due to normal aging alone, vasoadaptation becomes even more rigid, this compensatory mechanism may not suffice to prevent harmful elevations of blood pressure. One factor that may account for basally elevated levels of systemic vascular resistance in preeclampsia is the influence of endothelium-derived vasoconstrictors [
20], which are linked to endothelial dysfunction and may be key components in the etiology of preeclampsia [
52,
53]. Impaired endothelial nitric oxide synthase (eNOS) function and decreased NO action, and on the other hand, overrepresentation of inflammatory and antiangiogenic factors, contribute to a preeclamptic phenotype. Restoring this balance is currently in focus for new therapeutic approaches such as capturing or silencing sFLt-1, and although still inconclusive, evidence also shows that the NO donor and vasodilator pentaerythritol tetranitrate (PETN) reduces the incidence of preeclampsia in a high-risk study population [
54,
55,
56]. The vasoconstrictor angiotensin II mediates stimulation of factors such as human placental lactogen and sFlt-1, as well as transcription of inflammatory cytokines through angiotensin II type 1 receptor activation (AT1R) [
57,
58]. Increased agonistic autoantibodies against AT1R (AT1-AA) in preeclampsia [
59,
60] also contribute to AT1R activation, whereas AT1-AA blockade was shown to reduce preeclamptic symptoms in rats [
61]. Other endothelium-derived factors promote monocyte adhesion and migration by monocyte chemotactic protein-1 (MCP-1) and intercellular adhesion molecule-1 (ICAM-1) expression, for example, contributing to vascular inflammation [
62]. Hypertension-induced shear stress can induce endothelium-derived vasoconstrictors such as endothelin-1 and angiotensin II [
63,
64]. These do not only regulate the vascular tone, but also vascular smooth muscle cell (VSMC) growth and migration into the intima [
65,
66], where they proliferate in response to cytokines and growth factors such as platelet-derived growth factor (PDGF) [
67]. Angiotensin II, endothelin-1, and other cytokines lead to VSMC dedifferentiation to a migratory proliferative type that is fundamental to vascular pathogenesis and remodeling [
65,
68], which eventually leads to collagen and elastin deposition by VSMCs into the intima and increased vascular rigidity [
69].
When looking more closely at the contribution of the autonomic nervous system, it is important to note that scientists primarily focus on sympathetic nervous system control, although parasympathetic regulation appears to play an important role in pregnancy [
70]. Moreover, as heart rate levels remain below the intrinsic heart rate, moderate active performance stressors such as the one used in the present study primarily trigger parasympathetically mediated cardiorespiratory adaptation, i.e., fast vagal withdrawal [
29,
35]. Another plausible explanation for the blunted heart rate responses to stress in women with former preeclampsia is impaired adaptation to changing demands through vagal withdrawal, brought about by subordinate functioning of the parasympathetic branch of the autonomic nervous system. This notion is corroborated by the present finding that the mutual adjustments of respiration and heart rate as well as the mutual adjustments of respiration and blood pressure during the stress manipulation tended to be lower in women with previous preeclampsia compared to healthy women. In line with that, previous research has shown that sympathetically mediated regulation prevails during pregnancy, and it has been suggested that impaired parasympathetic regulation along with sympathetic overactivity may contribute to the pathophysiology of preeclampsia [
23,
26,
71].
Taken together, arterial blood pressure was maintained at a largely constant level throughout the stress manipulation in healthy women as well as in women with history of preeclampsia; however, different reasons seemed to have accounted for this outcome.
No differences were observed between women with severe compared to mild preeclampsia. However, while severity of preeclampsia was defined in the current study by recommended criteria, these criteria used to differentiate severe from mild preeclampsia are currently a subject of debate [
12]. A further limitation may be that on average, women with preeclampsia had greater body mass, which is an important determinant of cardiovascular function. However, it should be noted that stroke volume and systemic vascular resistance were calculated relative to body surface area, thus mitigating the potential confoundment, and results remained unchanged if the variables were adjusted for body mass index (BMI). Nevertheless, possible effects of body mass cannot be completely ruled out. On the other hand, body mass did not differ between women with preeclampsia and women with uncomplicated pregnancies. The groups did not differ in parity either, which may also impact the development of blood pressure in later life [
72]. In addition, the fact that we have no information about the women’s stress responses in the time before they became pregnant and the relatively small sample size should also be considered as important limitations of this study. As the risk factors for preeclampsia are the same as those predisposing to cardiovascular disease and might have the same effect on stress responses, problems may already precede pregnancy and then persist after delivery. The latter to some extent limits the interpretation of the analysis, especially within the women with preeclampsia. Due to the relatively small sample size, some analyses are at the lower limit with regard to the statistical power, in particular those analyzing differences between subtypes of preeclampsia. We hope that reporting these results nevertheless may encourage other researchers to use similar methods in larger cohorts of women with former preeclampsia, while separating women according to the subtypes of preeclampsia. Furthermore, there are a variety of excellent methods for the description of important metabolic and vascular changes during pregnancy and beyond which the present study did not include. Lack of such variables without doubt limits the overall explanatory power of the study. However, the point of the present study was to introduce a different approach that represents a novel addition to these metabolic and vascular variables. The approach of the present study provides new and additional information that is not easily gained from metabolic and vascular variables, particularly with regard to the functionality of the neuro-regulation of cardiovascular adaptation on a short-term basis.
Recent longitudinal assessment of preeclampsia has suggested masked hypertension at 12 weeks postpartum [
73]. In line with this observation, the present findings indicated moderately elevated blood pressure levels in women who were previously affected by preeclampsia. This suggests that the operating point of the arterial baroreflex may remain readjusted to a (slightly) higher pressure several weeks after delivery, although all blood pressure levels in the present study were in the nonpathological range. More importantly, the present study highlights that the identification of altered regulation mechanisms and impaired functioning of specific elements of the cardiovascular regulatory circuit in women with history of preeclampsia may be of greater scientific and finally clinical prognostic value than the monitoring of blood pressure levels and baroreceptor functioning alone. As preeclampsia has become a well-recognized risk factor for life-threatening cardiovascular complications in later life, several attempts have been made to include obstetric history in routine screening of cardiovascular risk. In the future, standardized stress response testing may significantly add to the identification of risk. Given the relatively novel stress parameters evaluated here, it appears that according to such novel lab testing to evaluate increased risk of developing cardiovascular complications (with or without relevant symptoms for metabolic syndrome), the risk is not clearly established early after delivery in women who experience preeclampsia during their pregnancy. Further studies are warranted to verify these findings and also to identify when such stress measures tested here become indicative of future cardiovascular complications for women who experienced preeclampsia. Finally, to date, very little guidance exists on the use of tailored prevention strategies in women with history of preeclampsia.