Acoustic Trauma Modulates Cochlear Blood Flow and Vasoactive Factors in a Rodent Model of Noise-Induced Hearing Loss
Abstract
:1. Introduction
2. Results
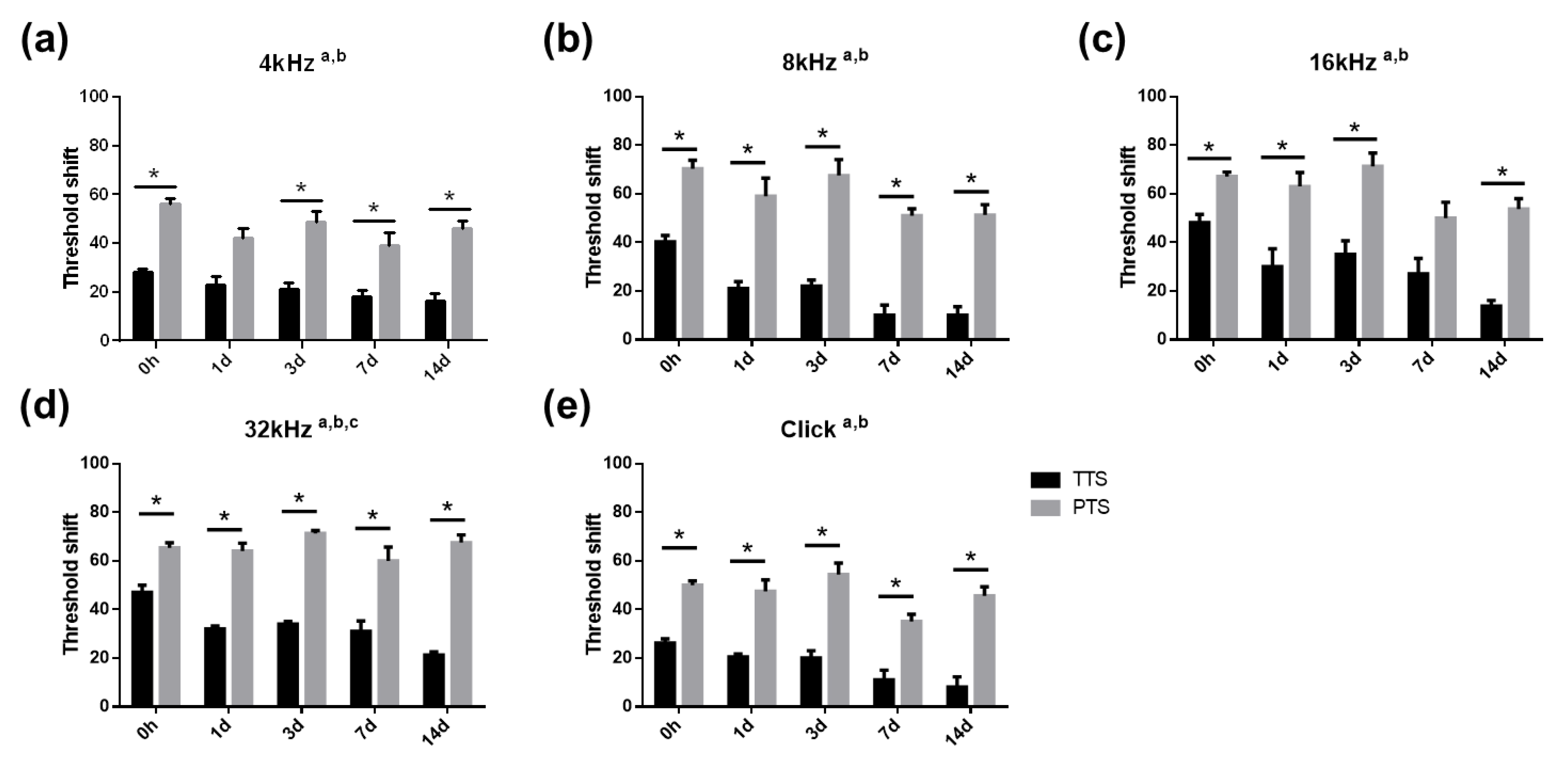
2.1. Auditory Brainstem Response Threshold by Transient Hearing Threshold Shift and Permanent Hearing Threshold Shift
2.2. Survival of SGNs is Modulated by Severity of Noise Trauma
2.3. Changes in Spiral Ligament Fibrocytes
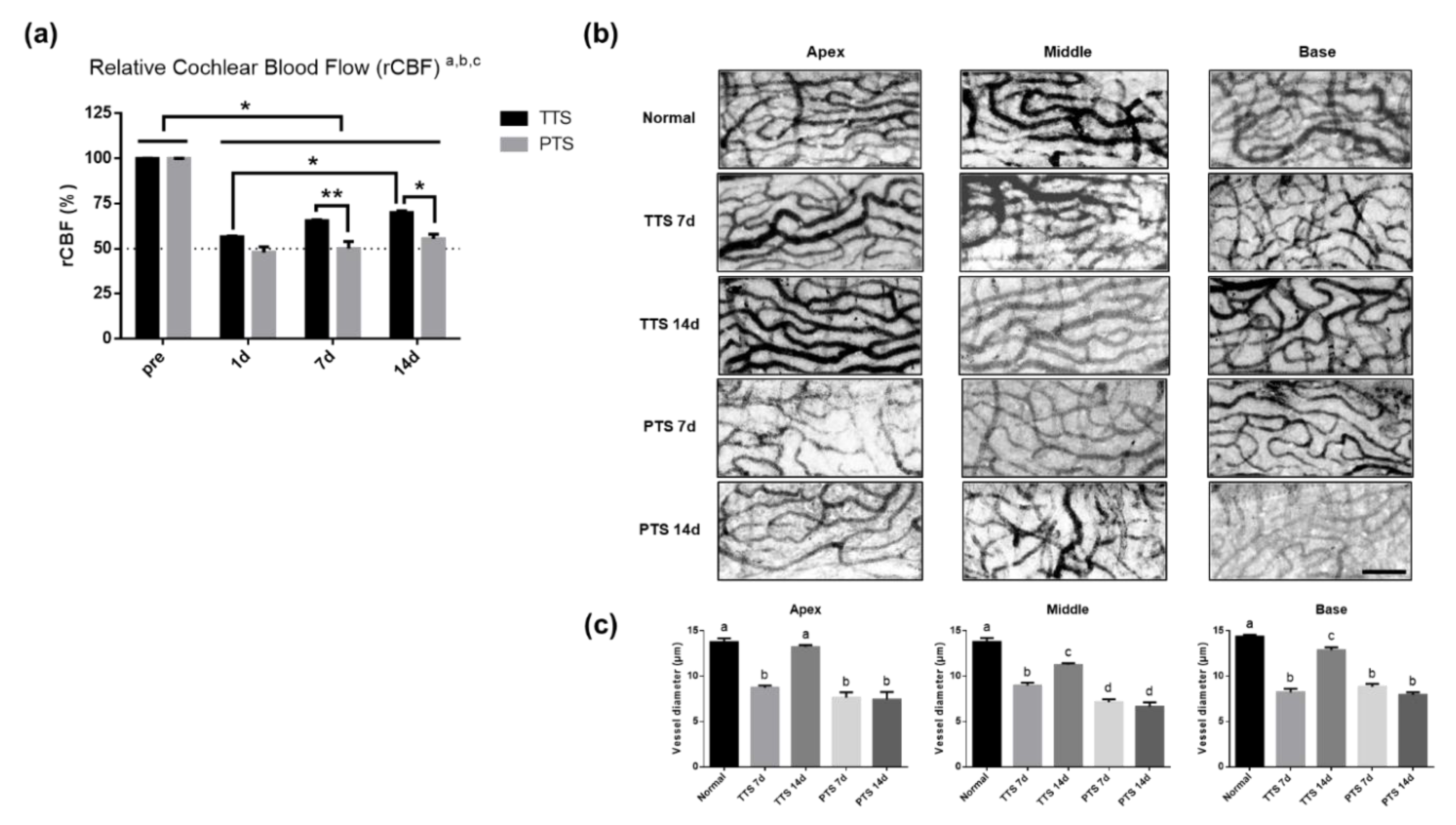
2.4. Acoustic Trauma Modulates Cochlear Microcirculation
2.5. Microvasculature of Stria Vascularis after Acoustic injury
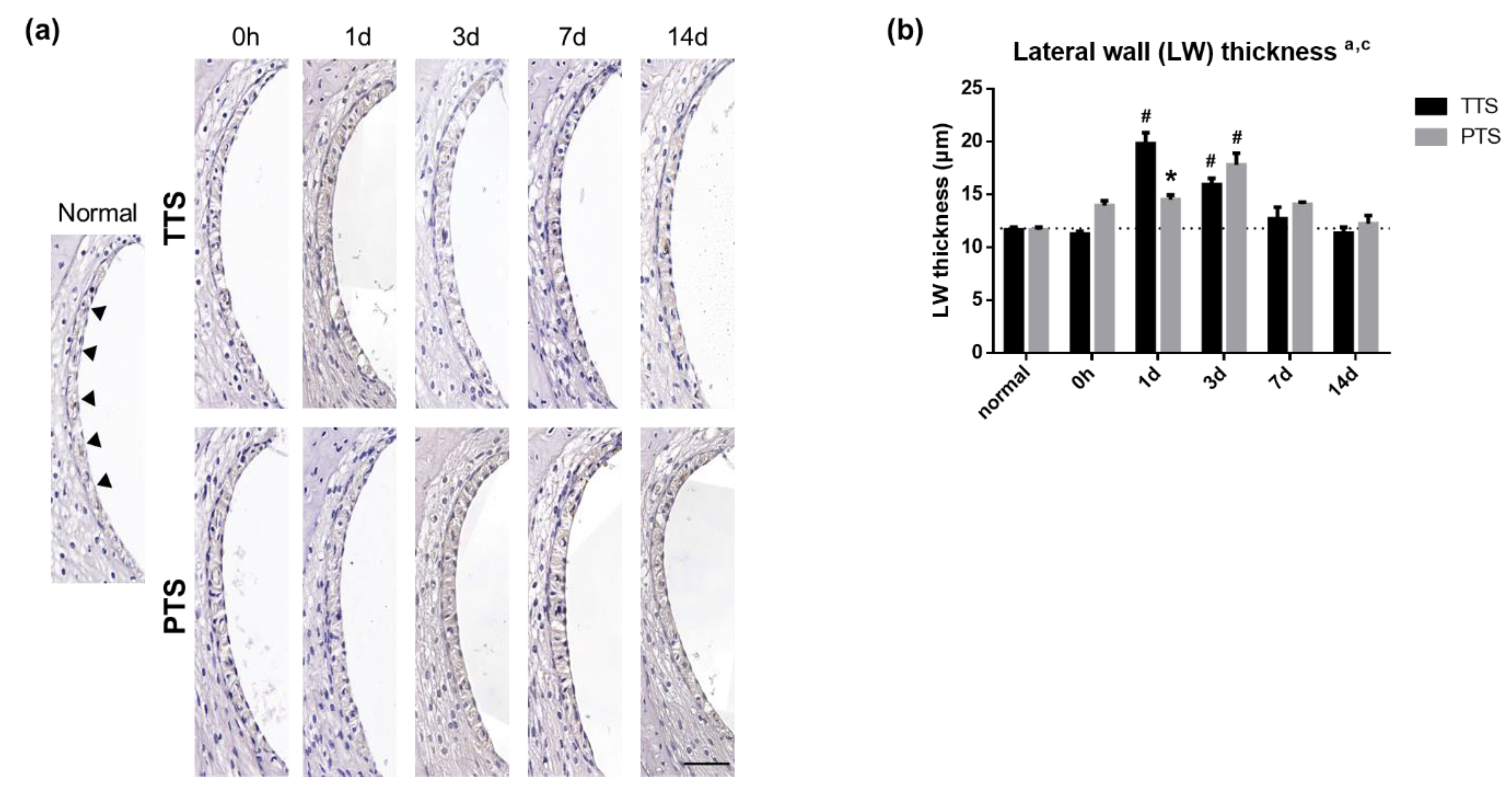
2.6. Stria Vascularis Thickness is Changed by Noise Exposure
2.7. Vasoactive Genes are Differentially Expressed in Both TTS and PTS
2.8. Genes Involved in Oxidative Stress are Modulated by Noise Trauma
2.9. Genes Involved in Proinflammatory Responses are Modulated by Noise Trauma
3. Discussion
4. Materials and Methods
4.1. Experimental Animals and Design
4.2. Noise Exposure
4.3. Auditory Brainstem Response
4.4. Hematoxylin Staining
4.5. Transmission Electron Microscope (TEM)
4.6. Measurement of Cochlear Blood Flow
4.7. Measurement of the Stria Capillary Thickness
4.8. Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
4.9. Image Processing and Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| TTS | transient hearing threshold shift |
| PTS | permanent hearing threshold shift |
| IL | interleukin |
| TNF | tumor necrosis factor |
| NIHL | noise-induced hearing loss |
| SLFs | spiral ligament fibrocytes |
| SGNs | spiral ganglion neurons |
| ADRA1A | alpha-1A adrenergic receptor |
| ADRA1D | alpha-1D adrenergic receptor |
| ET A | endothelin receptor type A |
| ET B | endothelin receptor type B |
| HO-1 | heme oxygenase 1 |
| eNOS | endothelial nitric oxide synthase |
| AT2 | angiotensin 2 receptor type 2 |
| ADORA2A | adenosine A2A receptor |
| PGE2 | prostaglandin E receptor 2 |
| PGI2 | prostaglandin I2 receptor |
| VEGF-A | vascular endothelial growth factor A |
References
- WHO. Global Estimates on Prevalence of Hearing Loss; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- Le, T.N.; Straatman, L.V.; Lea, J.; Westerberg, B. Current insights in noise-induced hearing loss: A literature review of the underlying mechanism, pathophysiology, asymmetry, and management options. J. Otolaryngol. Head Neck Surg. 2017, 46, 41. [Google Scholar] [CrossRef] [PubMed]
- WHO. Deafness and Hearing Loss. Available online: https://www.who.int/news-room/fact-sheets/detail/deafness-and-hearing-loss (accessed on 20 March 2019).
- World Health Organization. Global Costs of Unaddressed Hearing Loss and Cost-Effectiveness of Interventions: A WHO Report; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Kujawa, S.G.; Liberman, M.C. Adding insult to injury: Cochlear nerve degeneration after “temporary” noise-induced hearing loss. J. Neurosci. 2009, 29, 14077–14085. [Google Scholar] [CrossRef] [PubMed]
- Rabinowitz, P.M. Noise-induced hearing loss. Am. Fam. Physician 2000, 61, 2759–2760. [Google Scholar]
- Holborow, C. Deafness as a world problem. Adv. Otorhinolaryngol. 1983, 29, 174. [Google Scholar] [PubMed]
- Lamm, K.; Arnold, W. Noise-induced cochlear hypoxia is intensity dependent, correlates with hearing loss and precedes reduction of cochlear blood flow. Audiol. Neurootol. 1996, 1, 148–160. [Google Scholar] [CrossRef]
- Tabuchi, K.; Nishimura, B.; Tanaka, S.; Hayashi, K.; Hirose, Y.; Hara, A. Ischemia-reperfusion injury of the cochlea: Pharmacological strategies for cochlear protection and implications of glutamate and reactive oxygen species. Curr. Neuropharmacol. 2010, 8, 128–134. [Google Scholar] [CrossRef]
- Arpornchayanon, W.; Canis, M.; Suckfuell, M.; Ihler, F.; Olzowy, B.; Strieth, S. Modeling the measurements of cochlear microcirculation and hearing function after loud noise. Otolaryngol. Head Neck Surg. 2011, 145, 463–469. [Google Scholar] [CrossRef]
- Shi, X. Physiopathology of the cochlear microcirculation. Hear. Res. 2011, 282, 10–24. [Google Scholar] [CrossRef] [Green Version]
- Reif, R.; Zhi, Z.; Dziennis, S.; Nuttall, A.L.; Wang, R.K. Changes in cochlear blood flow in mice due to loud sound exposure measured with Doppler optical microangiography and laser Doppler flowmetry. Quant. Imaging Med. Surg. 2013, 3, 235–242. [Google Scholar]
- Hirose, K.; Liberman, M.C. Lateral wall histopathology and endocochlear potential in the noise-damaged mouse cochlea. J. Assoc. Res. Otolaryngol. 2003, 4, 339–352. [Google Scholar] [CrossRef]
- Hawkins, J.E., Jr. Microcirculation in the labyrinth. Arch. Otorhinolaryngol. 1976, 212, 241–251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wangemann, P. Supporting sensory transduction: Cochlear fluid homeostasis and the endocochlear potential. J. Physiol. 2006, 576, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Asakuma, S.; Snow, J.B., Jr. Effects of furosemide and ethacrynic acid on the endocochlear direct current potential in normal and kanamycin sulfate-treated guinea pigs. Otolaryngol. Head Neck Surg. (1979) 1980, 88, 188–193. [Google Scholar] [CrossRef]
- Anniko, M.; Wroblewski, R. Ionic environment of cochlear hair cells. Hear. Res. 1986, 22, 279–293. [Google Scholar] [CrossRef]
- Hellier, W.P.; Wagstaff, S.A.; O’Leary, S.J.; Shepherd, R.K. Functional and morphological response of the stria vascularis following a sensorineural hearing loss. Hear. Res. 2002, 172, 127–136. [Google Scholar] [CrossRef]
- Hibino, H.; Nin, F.; Tsuzuki, C.; Kurachi, Y. How is the highly positive endocochlear potential formed? The specific architecture of the stria vascularis and the roles of the ion-transport apparatus. Pflugers Arch. 2010, 459, 521–533. [Google Scholar] [CrossRef]
- Patuzzi, R. Ion flow in stria vascularis and the production and regulation of cochlear endolymph and the endolymphatic potential. Hear. Res. 2011, 277, 4–19. [Google Scholar] [CrossRef]
- Shi, X.; Nuttall, A.L. Upregulated iNOS and oxidative damage to the cochlear stria vascularis due to noise stress. Brain Res. 2003, 967, 1–10. [Google Scholar] [CrossRef]
- Shi, X. Pathophysiology of the cochlear intrastrial fluid-blood barrier (review). Hear. Res. 2016, 338, 52–63. [Google Scholar] [CrossRef]
- Park, Y.H.; Chung, J.; Lee, M.Y.; Lee, D.Y.; Kim, Y.H. Cochlear damage caused by the striking noise of Titanium head golf driver. Clin. Exp. Otorhinolaryngol. 2019, 12, 18–26. [Google Scholar] [CrossRef]
- Mizutari, K.; Nakagawa, S.; Mutai, H.; Fujii, M.; Ogawa, K.; Matsunaga, T. Late-phase recovery in the cochlear lateral wall following severe degeneration by acute energy failure. Brain Res. 2011, 1419, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Watanabe, K.; Fujioka, M.; Ogawa, K. Characterization of slow-cycling cells in the mouse cochlear lateral wall. PLoS ONE 2017, 12, e0179293. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.C. Immunocytochemical traits of type IV fibrocytes and their possible relations to cochlear function and pathology. J. Assoc. Res. Otolaryngol. 2009, 10, 369–382. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.L.; Schuknecht, H.F. Atrophy of the spiral ligament. Arch. Otolaryngol. 1972, 96, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Kusunoki, T.; Cureoglu, S.; Schachern, P.A.; Baba, K.; Kariya, S.; Paparella, M.M. Age-related histopathologic changes in the human cochlea: A temporal bone study. Otolaryngol. Head Neck Surg. 2004, 131, 897–903. [Google Scholar] [CrossRef]
- Howarth, A.; Shone, G.R. Ageing and the auditory system. Postgrad. Med. J. 2006, 82, 166–171. [Google Scholar] [CrossRef]
- Hequembourg, S.; Liberman, M.C. Spiral ligament pathology: A major aspect of age-related cochlear degeneration in C57BL/6 mice. J. Assoc. Res. Otolaryngol. 2001, 2, 118–129. [Google Scholar] [CrossRef]
- Locher, H.; de Groot, J.C.; van Iperen, L.; Huisman, M.A.; Frijns, J.H.; Chuva de Sousa Lopes, S.M. Development of the stria vascularis and potassium regulation in the human fetal cochlea: Insights into hereditary sensorineural hearing loss. Dev. Neurobiol. 2015, 75, 1219–1240. [Google Scholar] [CrossRef] [Green Version]
- Minowa, O.; Ikeda, K.; Sugitani, Y.; Oshima, T.; Nakai, S.; Katori, Y.; Suzuki, M.; Furukawa, M.; Kawase, T.; Zheng, Y.; et al. Altered cochlear fibrocytes in a mouse model of DFN3 nonsyndromic deafness. Science 1999, 285, 1408–1411. [Google Scholar] [CrossRef]
- Anniko, M.; Nordemar, H.; Sobin, A. Principles in embryonic development and differentiation of vestibular hair cells. Otolaryngol. Head Neck Surg. 1983, 91, 540–549. [Google Scholar] [CrossRef]
- Anniko, M.; Nordemar, H.; Van De Water, T.R. Embryogenesis of the inner ear. I. Development and differentiation of the mammalian crista ampullaris in vivo and in vitro. Arch. Otorhinolaryngol. 1979, 224, 285–299. [Google Scholar] [CrossRef] [PubMed]
- Billaud, M.; Lohman, A.W.; Straub, A.C.; Looft-Wilson, R.; Johnstone, S.R.; Araj, C.A.; Best, A.K.; Chekeni, F.B.; Ravichandran, K.S.; Penuela, S.; et al. Pannexin1 regulates alpha1-adrenergic receptor- Mediated vasoconstriction. Circ. Res. 2011, 109, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Methven, L.; Simpson, P.C.; McGrath, J.C. Alpha1A/B-knockout mice explain the native alpha1D-adrenoceptor’s role in vasoconstriction and show that its location is independent of the other alpha1-subtypes. Br. J. Pharmacol. 2009, 158, 1663–1675. [Google Scholar] [CrossRef] [PubMed]
- Tanoue, A.; Nasa, Y.; Koshimizu, T.; Shinoura, H.; Oshikawa, S.; Kawai, T.; Sunada, S.; Takeo, S.; Tsujimoto, G. The α1D-adrenergic receptor directly regulates arterial blood pressure via vasoconstriction. J. Clin. Investig. 2002, 109, 765–775. [Google Scholar] [CrossRef] [PubMed]
- Boyd, R.; Ratsep, M.T.; Ding, L.L.; Wang, H.D. ETA and ETB receptors are expressed in vascular adventitial fibroblasts. Am. J. Physiol. Heart Circ. Physiol. 2011, 301, H2271–H2278. [Google Scholar] [CrossRef] [PubMed]
- Niu, J.; Wu, J.; Li, X.; Zhang, F. Association between endothelin-1/endothelin receptor A and inflammation in mouse kidneys following acute ischemia/reperfusion. Mol. Med. Rep. 2015, 11, 3981–3987. [Google Scholar] [CrossRef]
- Rodriguez, J.E.; Romero-Nava, R.; Resendiz-Albor, A.A.; Rosales-Cruz, E.; Hong, E.; Huang, F.; Villafana, S. Expression and localization of the AT1 and AT2 angiotensin II receptors and alpha1A and alpha1D adrenergic receptors in aorta of hypertensive and diabetic rats. Clin. Exp. Hypertens. 2017, 39, 85–92. [Google Scholar] [CrossRef]
- Ibarra-Lara, L.; Del Valle-Mondragon, L.; Soria-Castro, E.; Torres-Narvaez, J.C.; Perez-Severiano, F.; Sanchez-Aguilar, M.; Ramirez-Ortega, M.; Cervantes-Perez, L.G.; Pastelin-Hernandez, G.S.; Oidor-Chan, V.H.; et al. Peroxisome proliferator-activated receptor-alpha stimulation by clofibrate favors an antioxidant and vasodilator environment in a stressed left ventricle. Pharmacol. Rep. 2016, 68, 692–702. [Google Scholar] [CrossRef]
- Justin, A.; Divakar, S.; Ramanathan, M. Cerebral ischemia induced inflammatory response and altered glutaminergic function mediated through brain AT1 and not AT2 receptor. Biomed. Pharmacother. 2018, 102, 947–958. [Google Scholar] [CrossRef]
- Khodadadi, S.; Zabihi, N.A.; Niazmand, S.; Abbasnezhad, A.; Mahmoudabady, M.; Rezaee, S.A. Teucrium polium improves endothelial dysfunction by regulating eNOS and VCAM-1 genes expression and vasoreactivity in diabetic rat aorta. Biomed. Pharmacother. 2018, 103, 1526–1530. [Google Scholar] [CrossRef]
- Shukla, M.; Roy, K.; Kaur, C.; Nayak, D.; Mani, K.V.; Shukla, S.; Kapoor, N. Attenuation of adverse effects of noise induced hearing loss on adult neurogenesis and memory in rats by intervention with Adenosine A2A receptor agonist. Brain Res. Bull. 2019, 147, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yan, S.; Wang, J.; Xu, Y.; Wang, Y.; Zhang, S.; Xu, X.; Yang, Q.; Zeng, X.; Zhou, Y.; et al. Endothelial adenosine A2a receptor-mediated glycolysis is essential for pathological retinal angiogenesis. Nat. Commun. 2017, 8, 584. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, K.; Hohjoh, H.; Inazumi, T.; Tsuchiya, S.; Sugimoto, Y. Prostaglandin E2-induced inflammation: Relevance of prostaglandin E receptors. Biochim. Biophys. Acta 2015, 1851, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Misawa, H.; Ohashi, W.; Tomita, K.; Hattori, K.; Shimada, Y.; Hattori, Y. Prostacyclin mimetics afford protection against lipopolysaccharide/d-galactosamine-induced acute liver injury in mice. Toxicol. Appl. Pharmacol. 2017, 334, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Han, W.K.; Kim, E.H.; Shin, S.A.; Shin, D.S.; Kim, B.J.; Lyu, A.R.; Park, Y.H. Susceptibility of diabetic mice to noise trauma. Biomed. Res. Int. 2018, 2018, 7601232. [Google Scholar] [CrossRef]
- Fujita, T.; Yamashita, D.; Katsunuma, S.; Hasegawa, S.; Tanimoto, H.; Nibu, K. Increased inner ear susceptibility to noise injury in mice with streptozotocin-induced diabetes. Diabetes 2012, 61, 2980–2986. [Google Scholar] [CrossRef]
- Hildesheimer, M.; Henkin, Y.; Pye, A.; Heled, S.; Sahartov, E.; Shabtai, E.L.; Muchnik, C. Bilateral superior cervical sympathectomy and noise-induced, permanent threshold shift in guinea pigs. Hear. Res. 2002, 163, 46–52. [Google Scholar] [CrossRef]
- Seidman, M.D.; Quirk, W.S.; Shirwany, N.A. Mechanisms of alterations in the microcirculation of the cochlea. Ann. N. Y. Acad. Sci. 1999, 884, 226–232. [Google Scholar] [CrossRef]
- Nuttall, A.L. Sound-induced cochlear ischemia/hypoxia as a mechanism of hearing loss. Noise Health 1999, 2, 17–32. [Google Scholar]
- Olivetto, E.; Simoni, E.; Guaran, V.; Astolfi, L.; Martini, A. Sensorineural hearing loss and ischemic injury: Development of animal models to assess vascular and oxidative effects. Hear. Res. 2015, 327, 58–68. [Google Scholar] [CrossRef]
- Axelsson, A.; Dengerink, H. The effects of noise on histological measures of the cochlear vasculature and red blood cells: A review. Hear. Res. 1987, 31, 183–191. [Google Scholar] [CrossRef]
- Scheibe, F.; Haupt, H.; Ludwig, C. Intensity-related changes in cochlear blood flow in the guinea pig during and following acoustic exposure. Eur. Arch. Otorhinolaryngol. 1993, 250, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Attanasio, G.; Buongiorno, G.; Piccoli, F.; Mafera, B.; Cordier, A.; Barbara, M.; Filipo, R. Laser Doppler measurement of cochlear blood flow changes during conditioning noise exposure. Acta Otolaryngol. 2001, 121, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, A.; Hasegawa, M.; Tamura, T.; Homma, T.; Komatsuzaki, A. Effects of frequency and intensity of sound on cochlear blood flow. Acta Otolaryngol. 1992, 112, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Hultcrantz, E.; Angelborg, C.; Beausang-Linder, M. Noise and cochlear blood flow. Arch. Otorhinolaryngol. 1979, 224, 103–106. [Google Scholar] [CrossRef] [PubMed]
- Prazma, J.; Smalley, W.E.; Covington, S.; Pillsbury, H.C. Cochlear blood flow. The effect of six hours of noise exposure. Arch. Otolaryngol. Head Neck Surg. 1988, 114, 657–660. [Google Scholar] [CrossRef] [PubMed]
- Quirk, W.S.; Seidman, M.D. Cochlear vascular changes in response to loud noise. Am. J. Otol. 1995, 16, 322–325. [Google Scholar]
- Luo, Y.; Tang, Y.; Xia, Q.; Liu, J. The expression of endothelin type A and B receptors in the lateral wall of the mouse cochlea. Cell. Mol. Biol. Lett. 2007, 12, 595–603. [Google Scholar] [CrossRef]
- Coessens, B.C. Endothelin: An endothelium-derived vasoactive peptide. J. Reconstr. Microsurg. 1994, 10, 405–410. [Google Scholar] [CrossRef]
- Sadanaga, M.; Liu, J.; Wangemann, P. Endothelin-A receptors mediate vasoconstriction of capillaries in the spiral ligament. Hear. Res. 1997, 112, 106–114. [Google Scholar] [CrossRef]
- Scherer, E.Q.; Arnold, W.; Wangemann, P. Pharmacological reversal of endothelin-1 mediated constriction of the spiral modiolar artery: A potential new treatment for sudden sensorineural hearing loss. BMC Ear Nose Throat Disord. 2005, 5, 10. [Google Scholar] [CrossRef] [PubMed]
- Picciotti, P.M.; Fetoni, A.R.; Paludetti, G.; Wolf, F.I.; Torsello, A.; Troiani, D.; Ferraresi, A.; Pola, R.; Sergi, B. Vascular endothelial growth factor (VEGF) expression in noise-induced hearing loss. Hear. Res. 2006, 214, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Byrne, A.M.; Bouchier-Hayes, D.J.; Harmey, J.H. Angiogenic and cell survival functions of vascular endothelial growth factor (VEGF). J. Cell. Mol. Med. 2005, 9, 777–794. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Zhou, H.; Zhang, J.; Liu, L. Increased endothelial progenitor cell circulation and VEGF production in a rat model of noise-induced hearing loss. Acta Otolaryngol. 2015, 135, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Vlajkovic, S.M.; Housley, G.D.; Thorne, P.R. Adenosine and the auditory system. Curr. Neuropharmacol. 2009, 7, 246–256. [Google Scholar] [CrossRef]
- Shi, X.; Han, W.; Yamamoto, H.; Tang, W.; Lin, X.; Xiu, R.; Trune, D.R.; Nuttall, A.L. The cochlear pericytes. Microcirculation 2008, 15, 515–529. [Google Scholar] [CrossRef] [PubMed]
- Canis, M.; Bertlich, M. Cochlear capillary pericytes. Adv. Exp. Med. Biol. 2019, 1122, 115–123. [Google Scholar]
- Gerhardt, H.; Betsholtz, C. Endothelial-pericyte interactions in angiogenesis. Cell Tissue Res. 2003, 314, 15–23. [Google Scholar] [CrossRef]
- Frank, R.N.; Dutta, S.; Mancini, M.A. Pericyte coverage is greater in the retinal than in the cerebral capillaries of the rat. Investig. Ophthalmol. Vis. Sci. 1987, 28, 1086–1091. [Google Scholar]
- Shepro, D.; Morel, N.M. Pericyte physiology. FASEB J. 1993, 7, 1031–1038. [Google Scholar] [CrossRef]
- Dai, M.; Nuttall, A.; Yang, Y.; Shi, X. Visualization and contractile activity of cochlear pericytes in the capillaries of the spiral ligament. Hear. Res. 2009, 254, 100–107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagenfeld, L.; Weiss, S.; Klemm, M.; Richard, G.; Zeitz, O. Vascular dysfunction in ocular blood flow regulation: Impact of reactive oxygen species in an experimental setup. Investig. Ophthalmol. Vis. Sci. 2014, 55, 5531–5536. [Google Scholar] [CrossRef] [PubMed]
- Minutoli, L.; Puzzolo, D.; Rinaldi, M.; Irrera, N.; Marini, H.; Arcoraci, V.; Bitto, A.; Crea, G.; Pisani, A.; Squadrito, F.; et al. ROS-mediated NLRP3 inflammasome activation in brain, heart, kidney, and testis ischemia/reperfusion injury. Oxid. Med. Cell. Longev. 2016, 2016, 2183026. [Google Scholar] [CrossRef] [PubMed]
- Pak, O.; Sydykov, A.; Kosanovic, D.; Schermuly, R.T.; Dietrich, A.; Schroder, K.; Brandes, R.P.; Gudermann, T.; Sommer, N.; Weissmann, N. Lung ischaemia-reperfusion injury: The role of reactive oxygen species. Adv. Exp. Med. Biol. 2017, 967, 195–225. [Google Scholar] [PubMed]
- Francis, A.; Baynosa, R. Ischaemia-reperfusion injury and hyperbaric oxygen pathways: A review of cellular mechanisms. Diving Hyperb. Med. 2017, 47, 110–117. [Google Scholar] [PubMed]
- Ohlemiller, K.K.; Wright, J.S.; Dugan, L.L. Early elevation of cochlear reactive oxygen species following noise exposure. Audiol. Neurootol. 1999, 4, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Yamane, H.; Nakai, Y.; Takayama, M.; Iguchi, H.; Nakagawa, T.; Kojima, A. Appearance of free radicals in the guinea pig inner ear after noise-induced acoustic trauma. Eur. Arch. Otorhinolaryngol. 1995, 252, 504–508. [Google Scholar] [CrossRef]
- Yamashita, D.; Jiang, H.Y.; Schacht, J.; Miller, J.M. Delayed production of free radicals following noise exposure. Brain Res. 2004, 1019, 201–209. [Google Scholar] [CrossRef] [Green Version]
- Yuan, H.; Wang, X.; Hill, K.; Chen, J.; Lemasters, J.; Yang, S.M.; Sha, S.H. Autophagy attenuates noise-induced hearing loss by reducing oxidative stress. Antioxid. Redox Signal. 2015, 22, 1308–1324. [Google Scholar] [CrossRef]
- Matsunobu, T.; Satoh, Y.; Ogawa, K.; Shiotani, A. Heme oxygenase-1 expression in the guinea pig cochlea induced by intense noise stimulation. Acta Otolaryngol. Suppl. 2009, 129, 18–23. [Google Scholar] [CrossRef]
- Jacono, A.A.; Hu, B.; Kopke, R.D.; Henderson, D.; Van De Water, T.R.; Steinman, H.M. Changes in cochlear antioxidant enzyme activity after sound conditioning and noise exposure in the chinchilla. Hear. Res. 1998, 117, 31–38. [Google Scholar] [CrossRef]
- Cheng, P.-W.; Liu, S.-H.; Young, Y.-H.; Hsu, C.-J.; Lin-Shiau, S.-Y. Protection from noise-induced temporary threshold shift by D-methionine is associated with preservation of ATPase activities. Ear Hear. 2008, 29, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Henderson, D.; Bielefeld, E.C.; Harris, K.C.; Hu, B.H. The role of oxidative stress in noise-induced hearing loss. Ear Hear. 2006, 27, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.M.; Brown, J.N.; Schacht, J. 8-iso-prostaglandin F(2alpha), a product of noise exposure, reduces inner ear blood flow. Audiol. Neurootol. 2003, 8, 207–221. [Google Scholar] [CrossRef] [PubMed]
- Thorne, P.R.; Nuttall, A.L.; Scheibe, F.; Miller, J.M. Sound-induced artifact in cochlear blood flow measurements using the laser Doppler flowmeter. Hear. Res. 1987, 31, 229–234. [Google Scholar] [CrossRef] [Green Version]
- Kurabi, A.; Keithley, E.M.; Housley, G.D.; Ryan, A.F.; Wong, A.C. Cellular mechanisms of noise-induced hearing loss. Hear. Res. 2017, 349, 129–137. [Google Scholar] [CrossRef]
- Kobel, M.; Le Prell, C.G.; Liu, J.; Hawks, J.W.; Bao, J. Noise-induced cochlear synaptopathy: Past findings and future studies. Hear. Res. 2017, 349, 148–154. [Google Scholar] [CrossRef]
- Vetter, D.E. Cellular signaling protective against noise-induced hearing loss—A role for novel intrinsic cochlear signaling involving corticotropin-releasing factor? Biochem. Pharmacol. 2015, 97, 1–15. [Google Scholar] [CrossRef]
- Hirose, K.; Discolo, C.M.; Keasler, J.R.; Ransohoff, R. Mononuclear phagocytes migrate into the murine cochlea after acoustic trauma. J. Comp. Neurol. 2005, 489, 180–194. [Google Scholar] [CrossRef]
- Fujioka, M.; Okano, H.; Ogawa, K. Inflammatory and immune responses in the cochlea: Potential therapeutic targets for sensorineural hearing loss. Front. Pharmacol. 2014, 5, 287. [Google Scholar] [CrossRef]
- Tahera, Y.; Meltser, I.; Johansson, P.; Bian, Z.; Stierna, P.; Hansson, A.C.; Canlon, B. NF-kappaB mediated glucocorticoid response in the inner ear after acoustic trauma. J. Neurosci. Res. 2006, 83, 1066–1076. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.; Omelchenko, I.; Shi, X.; Nuttall, A.L. The influence of NF-κB signal-transduction pathways on the murine inner ear by acoustic overstimulation. J. Neurosci. Res. 2009, 87, 1832–1840. [Google Scholar] [CrossRef] [PubMed]
- Infante, E.B.; Channer, G.A.; Telischi, F.F.; Gupta, C.; Dinh, J.T.; Vu, L.; Eshraghi, A.A.; Van De Water, T.R. Neurotology, Mannitol Protects Hair Cells Against Tumor Necrosis Factor α–Induced Loss. Otol. Neurotol. 2012, 33, 1656–1663. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.J.; Thorne, P.R.; Vlajkovic, S.M. Characterisation of cochlear inflammation in mice following acute and chronic noise exposure. Histochem. Cell Biol. 2016, 146, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhang, L.S.; Zinsmaier, A.K.; Patterson, G.; Leptich, E.J.; Shoemaker, S.L.; Yatskievych, T.A.; Gibboni, R.; Pace, E.; Luo, H.; et al. Neuroinflammation mediates noise-induced synaptic imbalance and tinnitus in rodent models. PLoS Biol. 2019, 17, e3000307. [Google Scholar] [CrossRef] [PubMed]
- Lyu, A.R.; Kim, D.H.; Lee, S.H.; Shin, D.S.; Shin, S.A.; Park, Y.H. Effects of dexamethasone on intracochlear inflammation and residual hearing after cochleostomy: A comparison of administration routes. PLoS ONE 2018, 13, e0195230. [Google Scholar] [CrossRef]
- Lee, S.H.; Lyu, A.R.; Shin, S.A.; Jeong, S.H.; Lee, S.A.; Park, M.J.; Park, Y.H. Cochlear glucocorticoid receptor and serum corticosterone expression in a rodent model of noise-induced hearing loss: Comparison of timing of Dexamethasone administration. Sci. Rep. 2019, 9, 12646. [Google Scholar] [CrossRef]
- Fischer, A.H.; Jacobson, K.A.; Rose, J.; Zeller, R. Hematoxylin and eosin staining of tissue and cell sections. CSH Protoc. 2008, 2008, pdb.prot4986. [Google Scholar] [CrossRef]
- Spurr, A.R. A low-viscosity epoxy resin embedding medium for electron microscopy. J. Ultrastruct. Res. 1969, 26, 31–43. [Google Scholar] [CrossRef]
- Reynolds, E.S. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J. Cell Biol. 1963, 17, 208. [Google Scholar] [CrossRef]
- Wangemann, P.; Itza, E.M.; Albrecht, B.; Wu, T.; Jabba, S.V.; Maganti, R.J.; Lee, J.H.; Everett, L.A.; Wall, S.M.; Royaux, I.E.; et al. Loss of KCNJ10 protein expression abolishes endocochlear potential and causes deafness in Pendred syndrome mouse model. BMC Med. 2004, 2, 30. [Google Scholar] [CrossRef] [PubMed]





© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shin, S.-A.; Lyu, A.-R.; Jeong, S.-H.; Kim, T.H.; Park, M.J.; Park, Y.-H. Acoustic Trauma Modulates Cochlear Blood Flow and Vasoactive Factors in a Rodent Model of Noise-Induced Hearing Loss. Int. J. Mol. Sci. 2019, 20, 5316. https://doi.org/10.3390/ijms20215316
Shin S-A, Lyu A-R, Jeong S-H, Kim TH, Park MJ, Park Y-H. Acoustic Trauma Modulates Cochlear Blood Flow and Vasoactive Factors in a Rodent Model of Noise-Induced Hearing Loss. International Journal of Molecular Sciences. 2019; 20(21):5316. https://doi.org/10.3390/ijms20215316
Chicago/Turabian StyleShin, Sun-Ae, Ah-Ra Lyu, Seong-Hun Jeong, Tae Hwan Kim, Min Jung Park, and Yong-Ho Park. 2019. "Acoustic Trauma Modulates Cochlear Blood Flow and Vasoactive Factors in a Rodent Model of Noise-Induced Hearing Loss" International Journal of Molecular Sciences 20, no. 21: 5316. https://doi.org/10.3390/ijms20215316




