1. Introduction
Reactive oxygen species (ROS), such as superoxide and hydrogen peroxide, are constantly generated from oxygen in all aerobic metabolism and pathogenic processes [
1]. Oxidative stress occurs when the balance between the generation of ROS and the antioxidant defense capacity of the body is destroyed [
2]. Oxidative stress can lead to a cascade of reactions that damage lipids, proteins and/or DNA, and cause a number of human diseases [
3]. During the neonatal period, ROS play an important role in the onset of many diseases, such as periventricular leukomalacia, chronic lung disease, and bronchopulmonary dysplasia [
4]. However, neonates are especially prone to oxidative stress, because they are often exposed to high oxygen concentrations due to the rapid passage from the intrauterine to the extrauterine environment, and they have limited antioxidant defense [
4]. Therefore, improving antioxidant capacity in newborns is crucial.
Maternal nutrition during pregnancy causes permanent adaptations in the offspring, which probably occur because of epigenetic regulation and changes in metabolic programming [
5]. The composition of diets in gestation has been reported to modulate maternal intestinal adaptations to pregnancy, affect placental function, and impair fetal gut development and immune status [
6,
7]. Therefore, the antioxidant capacity of offspring can be improved by regulating maternal nutrition. Increasing maternal fiber intake is an effective means to improve the antioxidant capacity of offspring [
2]. Wang et al. [
8] also reported that increasing maternal fiber intake during pregnancy enhances the antioxidative capacity of mothers and their offspring through increasing the total superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) activity and decreasing the serum malondialdehyde (MDA) concentration. The gut microbiota plays an important role in regulating human health [
9]. A previous study reported that diet dominates host genotype in influencing the gut microbiota [
10], and changes in dietary components can quickly lead to alterations in the composition of the microbiota [
11]. The maternal gut microbiota drives early postnatal innate immune development [
12]. Moreover, maternal fiber intake during gestation alters the intestinal microbiota in offspring [
13], and the gut microbiota in vaginally delivered infants resembles that of their mothers [
14]. Research has shown that the gut microbiota inhibits the NF-κB pathway, thus leading to the production of inflammatory cytokines and chemokines (tumor necrosis factor (TNF-α), interleukin-6 (IL-6), and MCP1), and suppressing the inflammatory response [
15]. The inflammatory response is often accompanied by changes in oxidative stress, which are strongly linked to alterations in the gut microbiota [
16,
17]. HO-1, an important antioxidative enzyme regulating the ROS levels of cells, can be induced by enteric microbiota [
18,
19]. However, whether dietary fiber in gestation can change the composition of the gut microbiota and subsequently improve the antioxidative capacity and immune status of offspring by altering the maternal gut microbiota has not been reported.
Generally, soluble fibers (SFs) are fermented more than insoluble fibers (ISFs), but both trigger specific alterations in the composition and predicted functions of colonic bacterial communities [
20]. Whether bacteria exist that specifically utilize SF and ISF, and how single or mixed types of dietary fiber in gestation might influence enteric microbial functions, host health, and metabolism, still remain unclear. In the present study, inulin (a typical fermentable SF) and natural cellulose (a typical ISF) [
21], were selected as supplementary dietary fibers in a pregnant sow model, which is usually used as an animal model for humans to estimate diet–microbiota–health interactions [
22].
3. Discussion
Newborns are vulnerable to free radical oxidative damage, thus resulting in oxidative stress [
23], because neonates (i) are often exposed to high oxygen concentrations, (ii) have diminished antioxidant activity, and (iii) have infections or inflammation due to environmental microbiota. Oxidative stress probably contributes to the severity of several newborn conditions to an extent that may cause organ injury or even death [
24]. Oxidative stress decreases the average daily feed intake, average daily gain, and nutrient digestibility in a pig model [
25]. Pregnant women usually show increased oxidative damage during the third trimester of pregnancy [
26]. Our previous study has indicated that maternal oxidative stress status might be transmitted to offspring by affecting placental oxidative stress, and improvements in maternal antioxidant capacity benefit fetal and neonatal development and health [
27]. Thus, improving maternal oxidative stress status, which can be regulated by nutritional means, provides a favorable means of enhancing the antioxidant capacity of offspring.
Dietary fiber is considered a key component in a healthful diet in pregnant women [
28]. Pregnant rats fed a high-fiber diet (oat bran and wheat bran, 1:1,
w/
w, 250 g/kg diet) showed a higher antioxidative capacity than those fed a high-fat diet, and this response was also present in future generations, as represented by higher liver total SOD, and Cu- and Zn-containing SOD activity [
2]. A similar study also shoed that a gestation diet supplemented with inulin enhances the serum activity of total SOD and GSH-Px and decreases the concentration of MDA in mothers and their offspring [
6]. Complex enzymatic and nonenzymatic systems play vital roles in protecting organisms from oxidative damage [
29]. In the present study, sows fed R1 and R2 diets showed increased CAT activity in sow plasma, and the GSH-Px activity increased in piglet plasma. SOD, CAT, and GSH-Px are three crucial endogenous antioxidant enzymes that play important roles in preventing oxidative damage. SOD converts ROS into hydrogen peroxide (H
2O
2), and then CAT and GSH-Px degrade the H
2O
2 to water and oxygen [
30,
31]. In addition, sows fed R1 and R2 diets showed decreased plasma MDA concentrations, and the piglets showed increased T-AOC in our study. T-AOC and MDA are two non-enzymatic indicators of antioxidant status and cell damage, respectively. In detail, T-AOC is an important integrative index reflecting the total antioxidant capacity of the body [
32], whereas MDA is a secondary product of lipid oxidation and is closely associated with cell damage, for which MDA has been widely considered an index to monitor the degree of lipid peroxidation [
33]. The liver, an important metabolic organ, plays a crucial role in nutrient metabolism and transformation, and in the defense against the invasion of bacteria and bacterial products [
34]. We also found that the offspring of sows fed R1 and R2 diets showed increased liver CAT and GSH-Px activity, and elevated mRNA expression of Nrf2 and HO-1 in the liver. Nrf2, a key transcription factor, plays an essential role in regulating the activity of endogenous antioxidant enzymes to resist oxidative stress [
35]. A previous study in piglets also showed that the activity of antioxidative enzymes is enhanced by increasing the mRNA expression level of Nrf2 [
36]. Moreover, Nrf2 regulates the activity of HO-1, an important antioxidative enzyme regulating the ROS levels of cells [
19] and serving as a sensitive and reliable indicator of cellular oxidative stress [
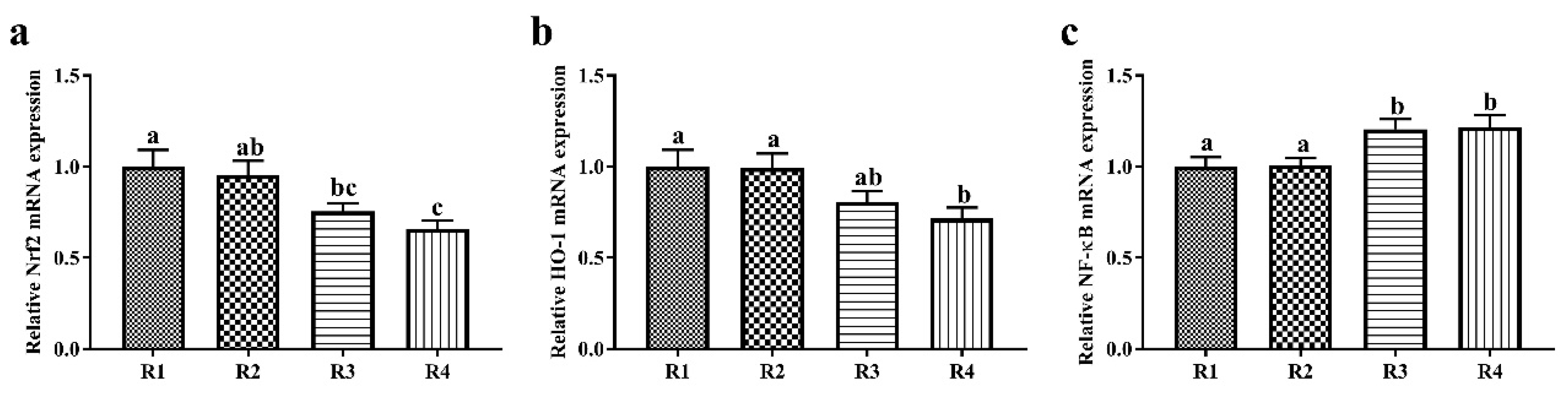
37]. Therefore, R1 and R2 diets enhanced the antioxidative capacity of offspring through upregulating Nrf2 and HO-1 mRNA expression and improving the antioxidant enzyme activity.
Oxidative stress is often associated with the inflammatory response. Cytokines have crucial roles in the immune and inflammatory response [
38,
39]. In the present study, we found that sows fed R1 and R2 had lower plasma IL-6, and piglets showed lower plasma TNF-α. TNF-α and IL-6 are both pro-inflammatory cytokines. IL-6 is a pleiotropic cytokine participating in the physiology of virtually every organ system, and it activates the hypothalamic-pituitary-adrenal axis and regulates hepatic protein synthesis during the acute response [
40]. In addition, IL-6 plays an important role in regulating the balance between the IL-17-producing Th17 cells and regulatory T cells, which have prominent roles in immune functions [
41]. TNF-α is secreted by activated macrophages and has some metabolic effects on lipid metabolism [
42]. TNF-α signaling induces activation of the transcription factor NF-κB and programmed cell death [
43]. The NF-κB signaling pathway is considered a key inducer of inflammation. Accordingly, lower mRNA expression levels of NF-κB were observed in the liver in R1 and R2 piglets. Similar research has shown that a maternal SF diet increases the plasma concentrations of anti-inflammatory factors, such as interleukin 10 (IL-10) and transforming growth factor β, in offspring [
44]. Therefore, the R1 and R2 diets decreased the inflammatory response of offspring through decreasing the plasma TNF-α level and liver NF-κB mRNA expression. Many studies have shown that high dietary fiber intake is associated with decreased inflammation [
45,
46]. Accumulating evidence indicates that maternal inflammation also has long-term consequences for offspring by affecting the intrauterine environment [
47]. In our current study, R1 and R2 diets improved the antioxidative capacity and decreased the plasma pro-inflammatory cytokine concentration in sows and piglets. Continued oxidative stress leads to chronic inflammation [
48] and detrimentally affects growth performance [
49]. Our previous study showed increased average daily gain of piglets during lactation when the ISF/SF ratios in the pregnancy diet were 3.89 and 5.59 [
50]. Therefore, the dietary fiber composition in the maternal pregnancy diet has an important effect on the health of mothers and offspring, and there is a threshold ratio of insoluble to soluble fiber to ensure that the dietary fiber is effective.
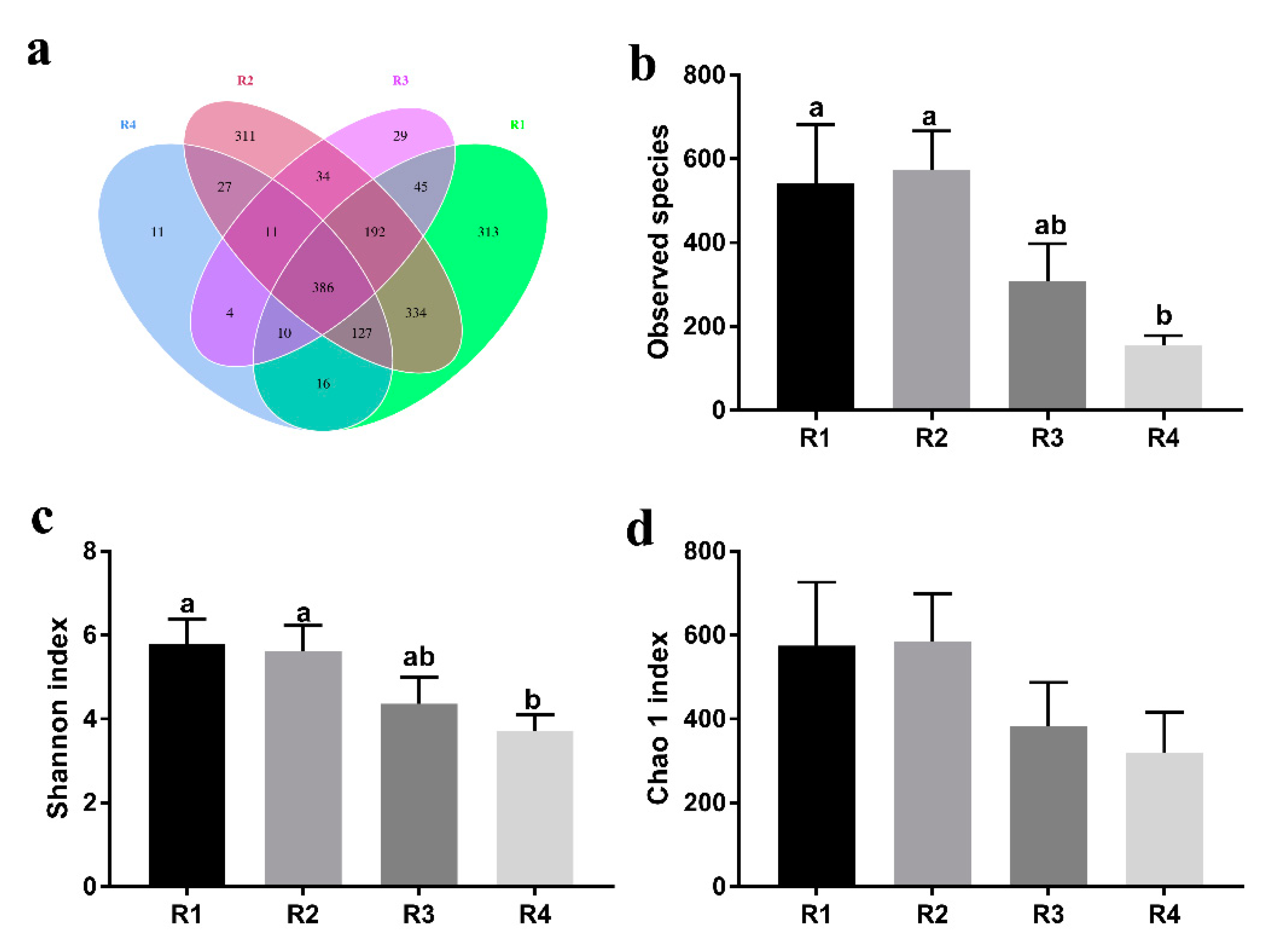
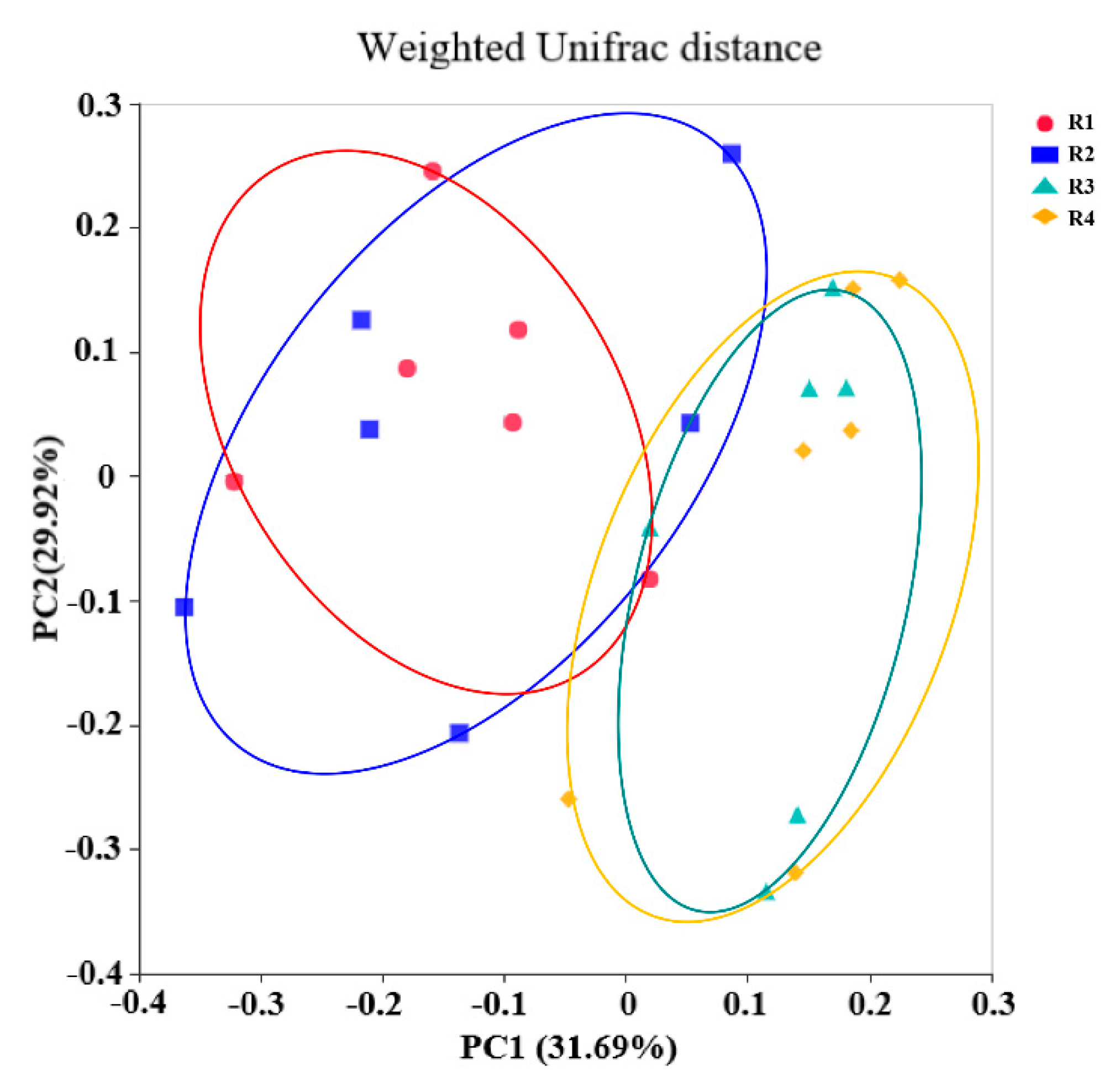
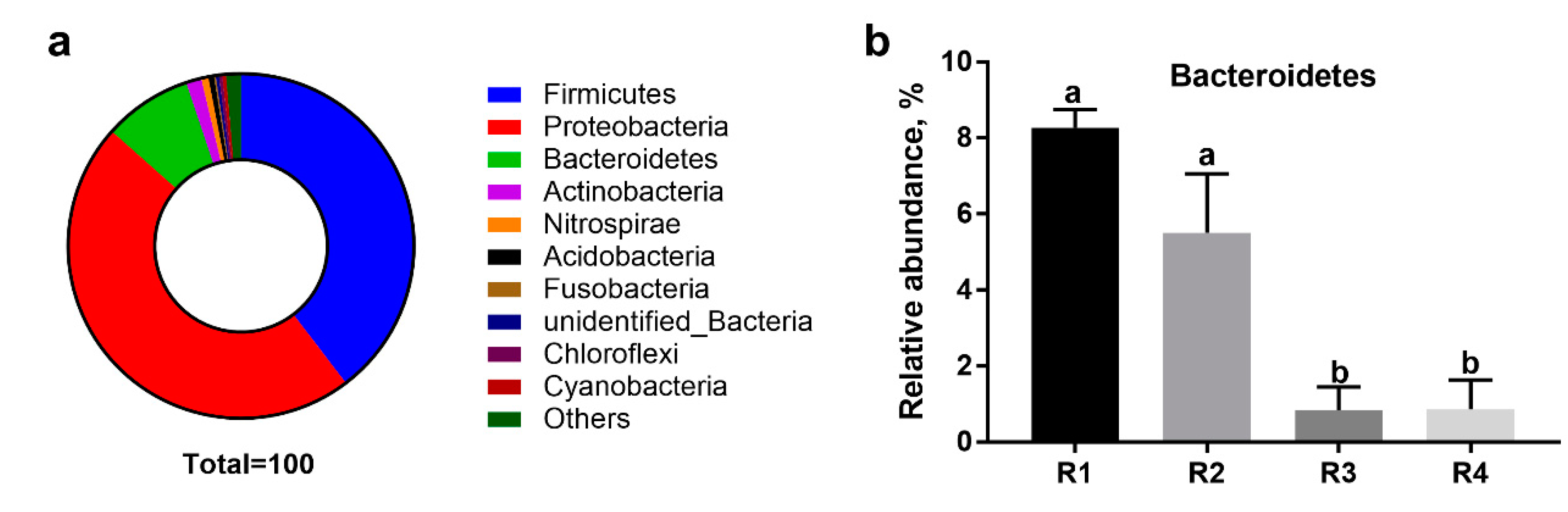
The gut microbiota has an indispensable role in host health by promoting the development of the immune system, decreasing inflammation, and competitively inhibiting pathogens [
51]. Consumption of dietary fiber is an effective strategy for modulating the microbiota [
52]. The composition of the gut microbiota is also affected by the types of dietary fiber [
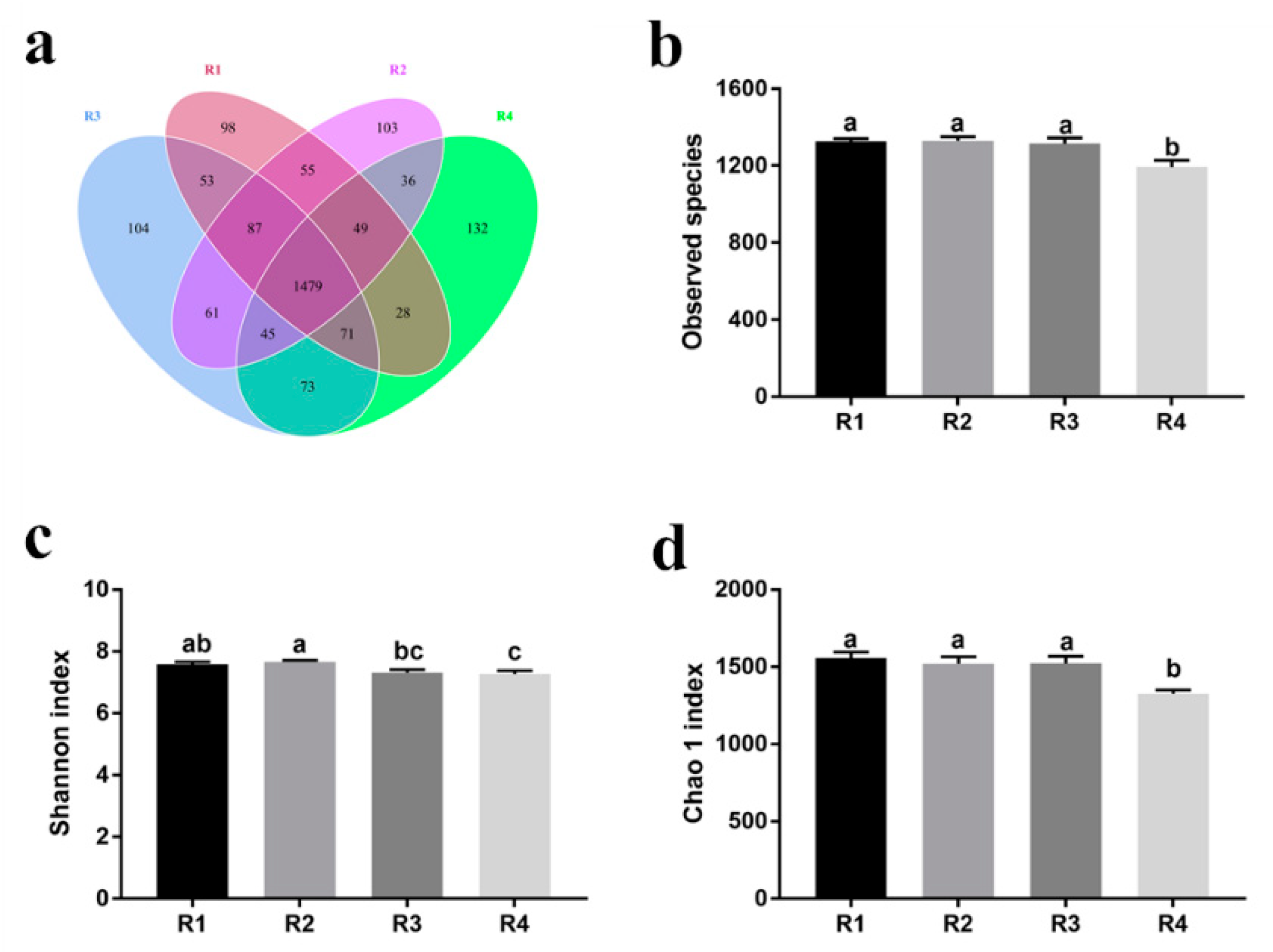
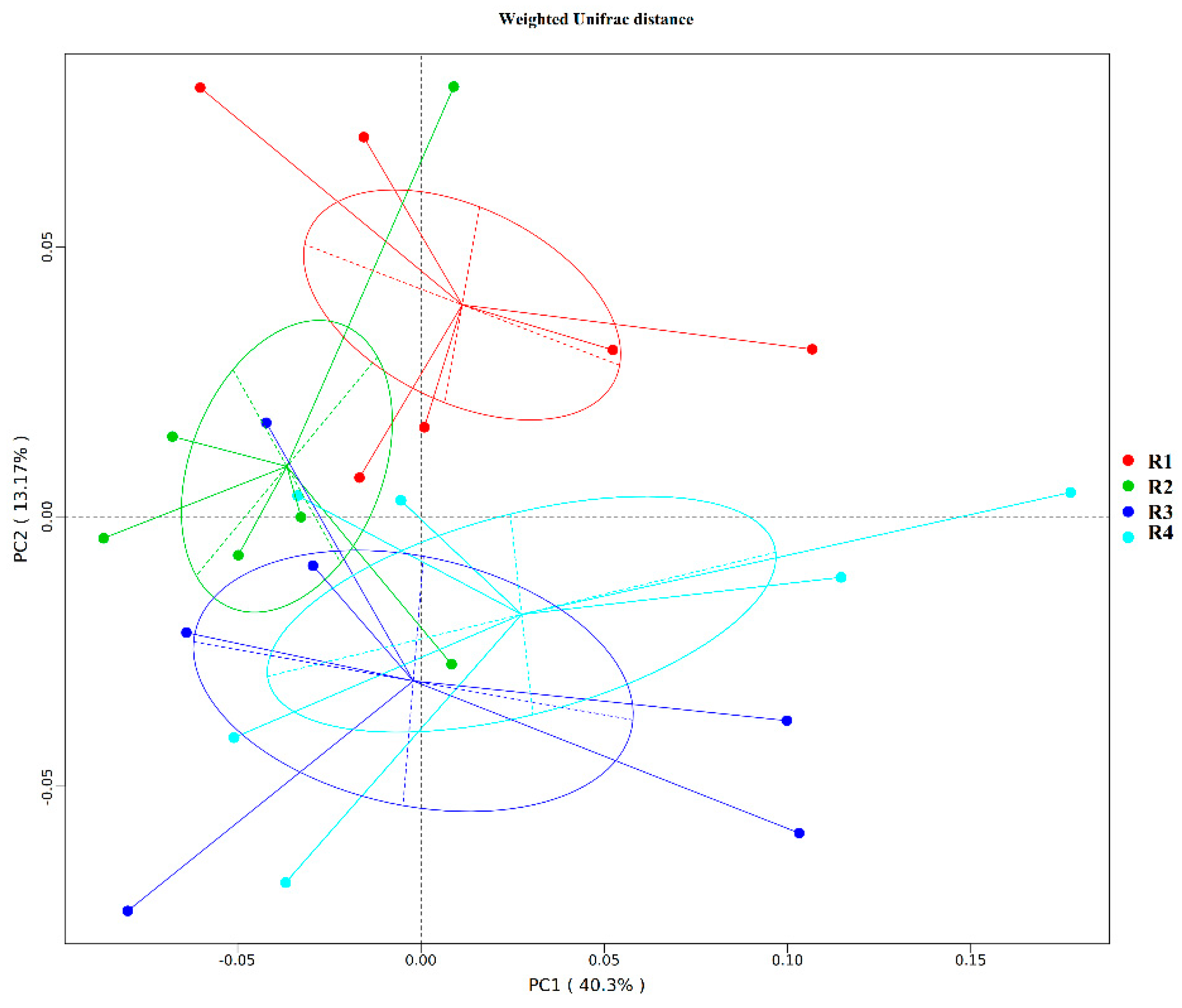
20]. In the current study, we found that sows in R1 and R2 on day 110 of pregnancy had significantly different microbiota community structures than those in R3 and R4, and R1 and R2 diets increased the α-diversity indexes of the sow fecal microbiota, which also indicated a threshold ratio of insoluble to soluble fiber. Low microbial diversity is often associated with metabolic syndrome and inflammation [
53]. Moreover, the R1 and R2 diets decreased the abundance of
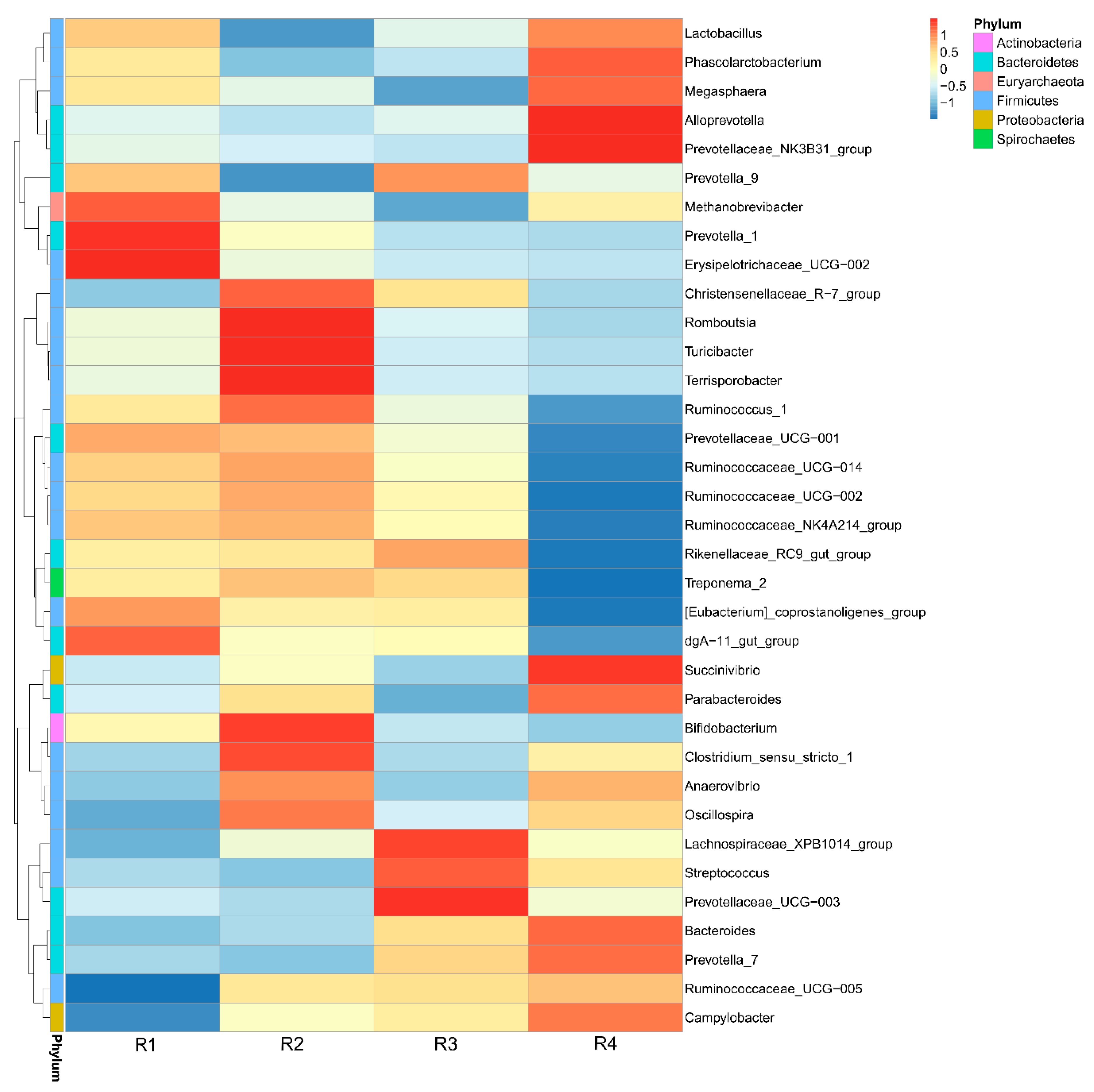
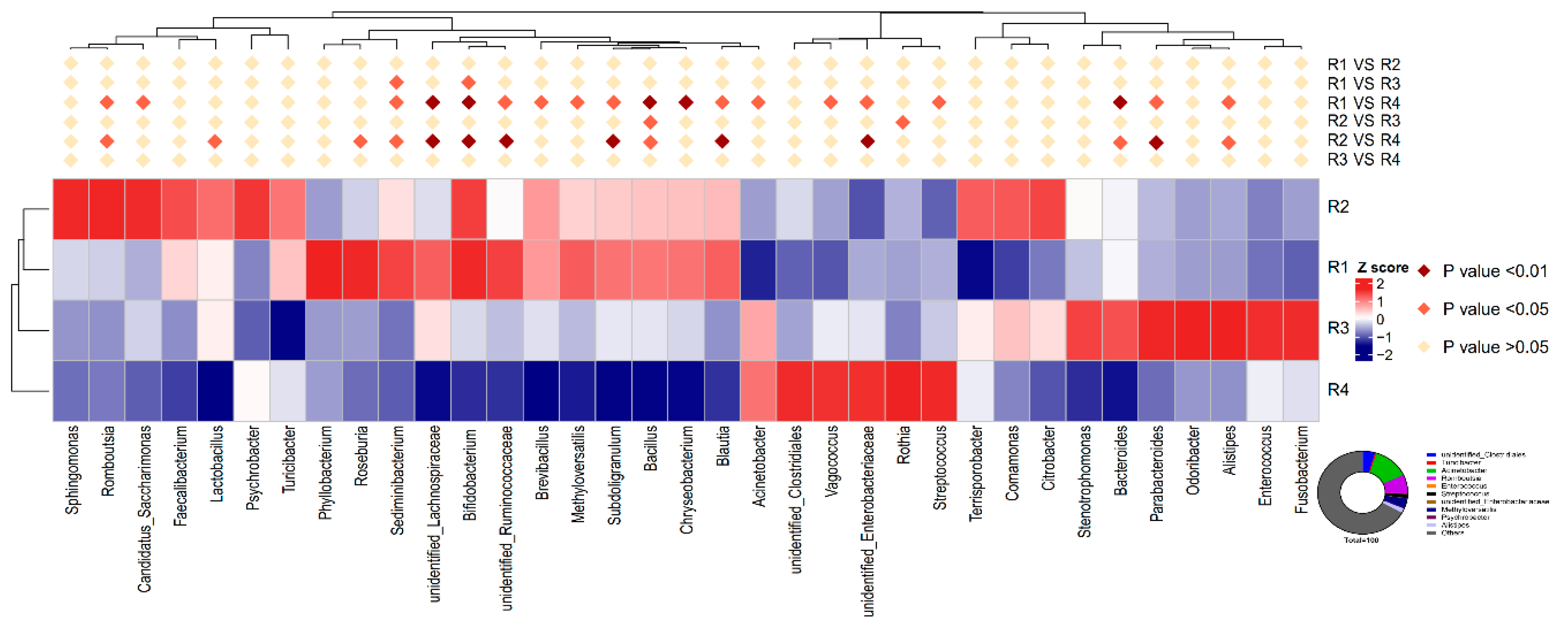
Streptococcus and increased the abundance of
Bifidobacterium in sow feces. A higher abundance of
Streptococcus is related to numerous inflammatory responses [
54], whereas
Bifidobacterium decreases inflammation through inhibiting the growth of pathogens via the production of organic acids and releasing soluble factors that alleviate the secretion of pro-inflammatory cytokines by immune cells [
55,
56]. The gut microbiota of individuals is dominated by different fiber-utilizing bacteria, which ferment dietary fiber into SCFAs, including acetate, propionate, and butyrate, which are important for human health. SCFAs, mainly butyrate, suppress the LPS- and cytokine-stimulated production of pro-inflammatory mediators, including TNF-α and IL-6, via interaction with the orphan G protein-coupled receptors GPR41 and GPR43 [
57,
58]. In the current study, sows in R1 and R2 had higher fecal SCFA concentrations, which might have resulted from soluble fiber being more easily fermented to produce SCFAs than ISF. In addition, SCFAs produced by the intestinal microbiota or their specific GPR43 agonist have been reported to inhibit oxidative stress [
59]. Interestingly, piglets in R1 and R2 also showed increased SCFA concentrations in the colon. SCFAs, especially butyrate, produced by microbial fermentation in the colon, were reported to raise Nrf2 in colonocytes [
60] and modulate the activity of the transcription factor NF-κB [
61], which might be the reason for the decreased inflammatory response and increased antioxidative capacity. Different SCFA concentrations are related to the distinct compositions of the microbiota among the four treatments. The offspring of sows fed R1 and R2 diets showed increased abundance of degrading bacteria, such as
Bacteroidetes,
Romboutsia,
Ruminococcaceae, and
Parabacteroides, in the colonic contents. Recently, sequencing has indicated that placenta and umbilical cord blood is not sterile, and the microbiota within the neonate’s meconium shares significant similarity with that of the placenta, thus suggesting that maternal transfer of microbiota is possible and might occur during gestation [
62,
63,
64]. Furthermore, previous research has also shown that vaginally delivered infants acquire bacterial communities resembling their own mother’s vaginal microbiota [
65]. Therefore, the maternal microbiota plays a vital role in the composition of the intestinal flora of the offspring. Pregnancy-related changes in the maternal microbiota are dependent on the mother’s periconceptional diet [
66]. Therefore, diet composition during pregnancy has important effects on the structure of the gut microbiota of offspring [
67]. Paßlack et al. [
13] showed that the addition of inulin to a gestation diet modulates not only the intestinal microbiota in sows but also their offspring. In the current study, similar structures of microbiota were also observed between mothers and their offspring: R1 resembled R2, and R3 resembled R4, and the observed changes in the relative abundance of
Streptococcus and
Bifidobacterium in the offspring was similar to that in the mother. Moreover, decreased abundance of
Enterobacteriaceae was also observed in the colonic contents of piglets in R1 and R2, in agreement with the results for inflammatory factors. The capacity of
Enterobacteriaceae to induce host inflammation through endotoxin production is well known [
68]. Increased
Bifidobacterium inhibits the overgrowth of
Enterobacteriaceae [
69]. Moreover, the relative abundance of
Acinetobacter in the colonic contents of R1 was lower than that of R4, but not significantly different from that of R2.
Acinetobacter, a highly concerning pathogen belonging to the gram-negative
Coccobacillus, has become an increasingly common nosocomial problem [
70]. Members of the genus
Acinetobacter have been implicated in a wide spectrum of infectious diseases [
71] and have a strong ability to acquire or upregulate antibiotic drug resistance determinants [
72]. These findings might also suggest that the offspring of sows in R1 and R2 had a strong ability to resist
Acinetobacter infection. The increased beneficial bacteria and decreased harmful bacteria might be another reason for the lower inflammation and oxidative stress in R1 and R2 piglets.
Thus, the composition of dietary fiber in the pregnancy diet has an important role in improving antioxidative capacity and decreasing the inflammatory response of mothers and their offspring through modulating the composition of the gut microbiota. Not only the dietary fiber level but also the ratio of insoluble dietary fiber to soluble dietary fiber should be considered in pregnancy diets.