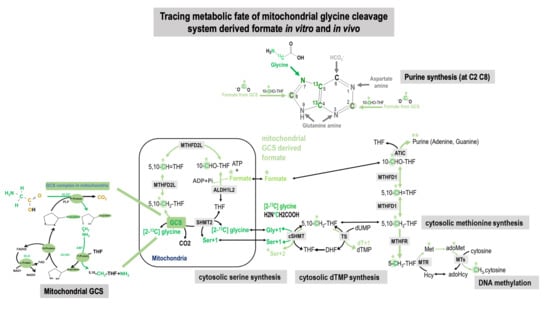
Tracing Metabolic Fate of Mitochondrial Glycine Cleavage System Derived Formate In Vitro and In Vivo
Abstract
:1. Introduction
2. Results
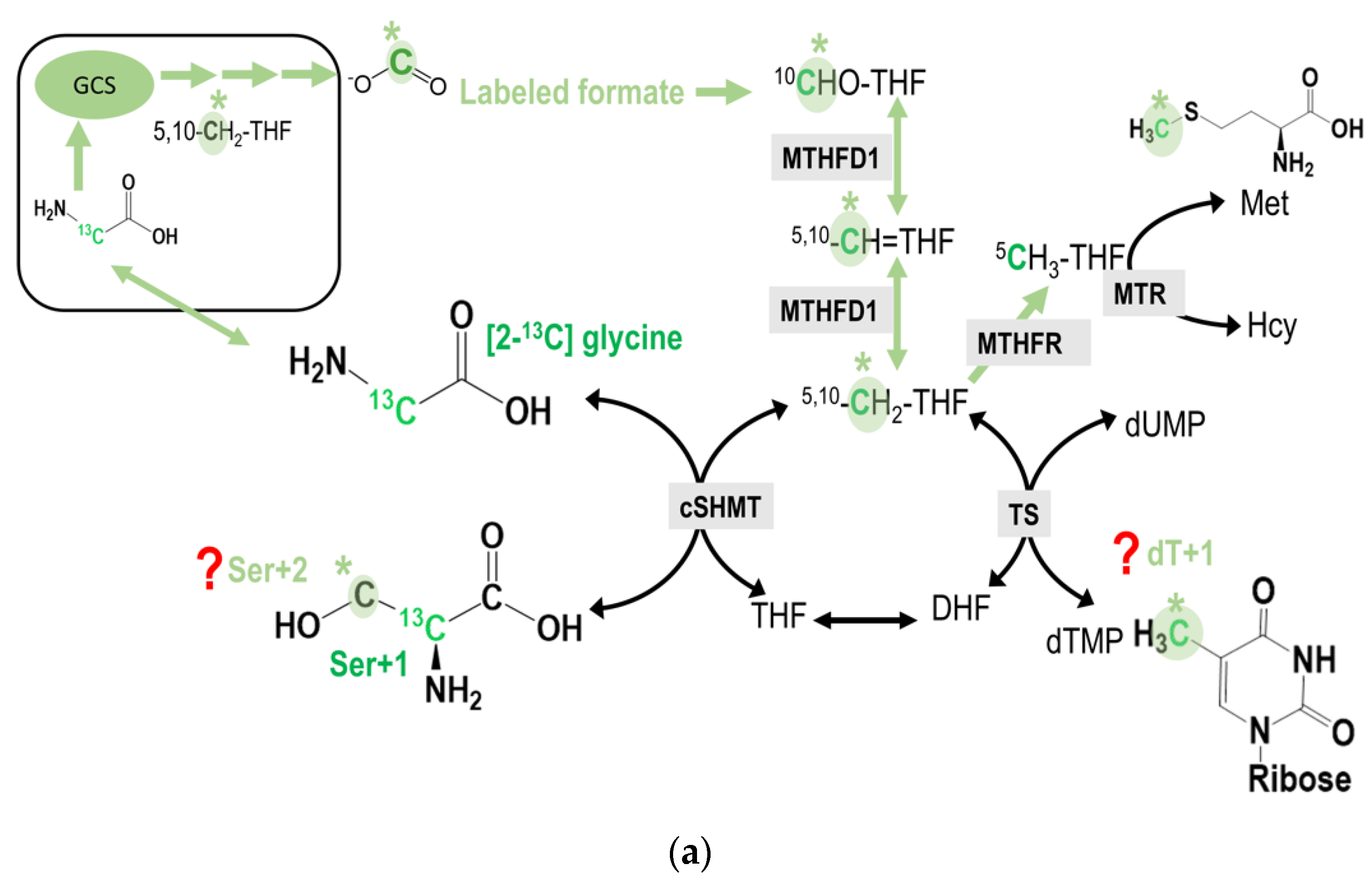
2.1. Tracing Metabolic Fate of C2-Carbon of Glycine via GCS in 1C Metabolism
2.2. Tracing Metabolic Fate of Serine 2-Carbon via GCS in 1C Metabolism
2.3. Metabolic Fate of 2-Carbon from Glycine and Serine via GCS in Cell Models
2.4. Restoring GNMT Expression Decreased Formate Generation from Glycine 2-Carbon via GCS
2.5. Tracing Metabolic Fate of 3-Carbon of Serine in 1C Metabolism
2.6. Utilization of the Glycine 2-Carbon in dTMP Synthesis via Mitochondrial GCS In Vivo
3. Discussion
3.1. Metabolic Fate of C2-Carbon of Glycine and Serine via GCS
3.2. Restoring GNMT Expression Decreased Formate Generation from Glycine 2-Carbon via GCS
3.3. Nutritional Supply Affect the Utilization of GCS-Derived Formate
3.4. The Partitioning of GCS-Derived 1C Unit Utilization is Tissue-Specific
3.5. Limitations of the Study
4. Materials and Methods
4.1. Chemicals and Materials
4.2. Cell-Lines and Culture Conditions
4.3. Stable Isotopic Experiments In Vitro
4.4. Tracing Metabolic Fate of Glycine 2-Carbon, Serine 2-Carbon, and 3-Carbon In Vivo
4.5. Gas Chromatography/Mass Spectrometry Analysis
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Lee, T.Y.; Chiang, E.P.; Shih, Y.T.; Lane, H.Y.; Lin, J.T.; Wu, C.Y. Lower serum folate is associated with development and invasiveness of gastric cancer. World. J. Gastroenterol. 2014, 20, 11313–11320. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; Wu, M.T.; Tang, F.Y.; Chen, D.Y.; Ko, H.A.; Shane, B.; Huang, W.N.; Chiang, E.P. MTHFR C677T polymorphism increases MTX sensitivity via the inhibition of S-adenosylmethionine and de novo purine synthesis. Clin. Sci. 2019, 133, 253–267. [Google Scholar] [CrossRef] [PubMed]
- Adeva-Andany, M.; Souto-Adeva, G.; Ameneiros-Rodriguez, E.; Fernandez-Fernandez, C.; Donapetry-Garcia, C.; Dominguez-Montero, A. Insulin resistance and glycine metabolism in humans. Amino Acids 2018, 50, 11–27. [Google Scholar] [CrossRef] [PubMed]
- Brosnan, J.T.; Brosnan, M.E. Formate and its role in amino acid metabolism. Curr. Opin. Clin. Nutr. Metab. Care 2020, 23, 23–28. [Google Scholar] [CrossRef]
- Garcia-Martinez, L.F.; Appling, D.R. Characterization of the folate-dependent mitochondrial oxidation of carbon 3 of serine. Biochemistry 1993, 32, 4671–4676. [Google Scholar] [CrossRef]
- Morrow, G.P.; MacMillan, L.; Lamarre, S.G.; Young, S.K.; MacFarlane, A.J.; Brosnan, M.E.; Brosnan, J.T. In vivo kinetics of formate metabolism in folate-deficient and folate-replete rats. J. Biol. Chem. 2015, 290, 2244–2250. [Google Scholar] [CrossRef] [Green Version]
- Pietzke, M.; Meiser, J.; Vazquez, A. Formate metabolism in health and disease. Mol. Metab. 2019, 33, 23–37. [Google Scholar] [CrossRef]
- Lamers, Y.; Williamson, J.; Gilbert, L.R.; Stacpoole, P.W.; Gregory, J.F., 3rd. Glycine turnover and decarboxylation rate quantified in healthy men and women using primed, constant infusions of [1,2-(13)C2]glycine and [(2)H3]leucine. J. Nutr. 2007, 137, 2647–2652. [Google Scholar] [CrossRef] [Green Version]
- Kikuchi, G. The glycine cleavage system: Composition, reaction mechanism, and physiological significance. Mol. Cell. Biochem. 1973, 1, 169–187. [Google Scholar] [CrossRef]
- Kikuchi, G.; Hiraga, K.; Yoshida, T. Role of the glycine-cleavage system in glycine and serine metabolism in various organs. Biochem. Soc. Trans. 1980, 8, 504–506. [Google Scholar] [CrossRef] [Green Version]
- Tibbetts, A.S.; Appling, D.R. Compartmentalization of Mammalian folate-mediated one-carbon metabolism. Annu. Rev. Nutr. 2010, 30, 57–81. [Google Scholar] [CrossRef] [PubMed]
- Pai, Y.J.; Leung, K.Y.; Savery, D.; Hutchin, T.; Prunty, H.; Heales, S.; Brosnan, M.E.; Brosnan, J.T.; Copp, A.J.; Greene, N.D. Glycine decarboxylase deficiency causes neural tube defects and features of non-ketotic hyperglycinemia in mice. Nat. Commun. 2015, 6, 6388. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Feng, J.; Cao, Y.; Shen, S.; Cai, Y.; Yang, D.; Yan, R.; Wang, L.; Zhang, H.; Zhong, X.; et al. Glycine cleavage system determines the fate of pluripotent stem cells via the regulation of senescence and epigenetic modifications. Life Sci. Alliance 2019, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, W.C.; Shyh-Chang, N.; Yang, H.; Rai, A.; Umashankar, S.; Ma, S.; Soh, B.S.; Sun, L.L.; Tai, B.C.; Nga, M.E.; et al. Glycine decarboxylase activity drives non-small cell lung cancer tumor-initiating cells and tumorigenesis. Cell 2012, 148, 259–272. [Google Scholar] [CrossRef] [Green Version]
- Go, M.K.; Zhang, W.C.; Lim, B.; Yew, W.S. Glycine decarboxylase is an unusual amino acid decarboxylase involved in tumorigenesis. Biochemistry 2014, 53, 947–956. [Google Scholar] [CrossRef]
- Labuschagne, C.F.; van den Broek, N.J.; Mackay, G.M.; Vousden, K.H.; Maddocks, O.D. Serine, but not glycine, supports one-carbon metabolism and proliferation of cancer cells. Cell Rep. 2014, 7, 1248–1258. [Google Scholar] [CrossRef] [Green Version]
- Stempak, J.M.; Sohn, K.J.; Chiang, E.P.; Shane, B.; Kim, Y.I. Cell and stage of transformation-specific effects of folate deficiency on methionine cycle intermediates and DNA methylation in an in vitro model. Carcinogenesis 2005, 26, 981–990. [Google Scholar] [CrossRef]
- Sohn, K.J.; Jang, H.; Campan, M.; Weisenberger, D.J.; Dickhout, J.; Wang, Y.C.; Cho, R.C.; Yates, Z.; Lucock, M.; Chiang, E.P.; et al. The methylenetetrahydrofolate reductase C677T mutation induces cell-specific changes in genomic DNA methylation and uracil misincorporation: A possible molecular basis for the site-specific cancer risk modification. Int. J. Cancer 2009, 124, 1999–2005. [Google Scholar] [CrossRef] [Green Version]
- Felig, P. Amino acid metabolism in man. Annu. Rev. Biochem. 1975, 44, 933–955. [Google Scholar] [CrossRef]
- Baggott, J.E.; Gorman, G.S.; Tamura, T. 13C enrichment of carbons 2 and 8 of purine by folate-dependent reactions after [13C]formate and [2-13C]glycine dosing in adult humans. Metabolism 2007, 56, 708–715. [Google Scholar] [CrossRef] [Green Version]
- Pimstone, N.R.; Dowdle, E.B.; Eales, L. The incorporation of glycine-2-14C into urinary uric acid in normal and porphyric human subjects. S. Afr. Med. J. 1969, 43, 961–964. [Google Scholar] [PubMed]
- Narkewicz, M.R.; Thureen, P.J.; Sauls, S.D.; Tjoa, S.; Nikolayevsky, N.; Fennessey, P.V. Serine and glycine metabolism in hepatocytes from mid gestation fetal lambs. Pediatr. Res. 1996, 39, 1085–1090. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Narkewicz, M.R.; Sauls, S.D.; Tjoa, S.S.; Teng, C.; Fennessey, P.V. Evidence for intracellular partitioning of serine and glycine metabolism in Chinese hamster ovary cells. Biochem. J. 1996, 313 Pt 3, 991–996. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiang, E.P.; Wang, Y.C.; Tang, F.Y. Folate restriction and methylenetetrahydrofolate reductase 677T polymorphism decreases adoMet synthesis via folate-dependent remethylation in human-transformed lymphoblasts. Leukemia 2007, 21, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Chiang, E.P.; Wang, Y.C.; Chen, W.W.; Tang, F.Y. Effects of insulin and glucose on cellular metabolic fluxes in homocysteine transsulfuration, remethylation, S-adenosylmethionine synthesis, and global deoxyribonucleic acid methylation. J. Clin. Endocrinol. Metab. 2009, 94, 1017–1025. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.C.; Tang, F.Y.; Chen, S.Y.; Chen, Y.M.; Chiang, E.P. Glycine-N methyltransferase expression in HepG2 cells is involved in methyl group homeostasis by regulating transmethylation kinetics and DNA methylation. J. Nutr. 2011, 141, 777–782. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.C.; Chen, Y.M.; Lin, Y.J.; Liu, S.P.; Chiang, E.P. GNMT expression increases hepatic folate contents and folate-dependent methionine synthase-mediated homocysteine remethylation. Mol. Med. 2011, 17, 486–494. [Google Scholar] [CrossRef]
- Wang, Y.C.; Lin, W.L.; Lin, Y.J.; Tang, F.Y.; Chen, Y.M.; Chiang, E.P. A novel role of the tumor suppressor GNMT in cellular defense against DNA damage. Int. J. Cancer 2014, 134, 799–810. [Google Scholar] [CrossRef]
- Anguera, M.C.; Field, M.S.; Perry, C.; Ghandour, H.; Chiang, E.P.; Selhub, J.; Shane, B.; Stover, P.J. Regulation of folate-mediated one-carbon metabolism by 10-formyltetrahydrofolate dehydrogenase. J. Biol. Chem. 2006, 281, 18335–18342. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.C.; Wu, M.T.; Lin, Y.J.; Tang, F.Y.; Ko, H.A.; Chiang, E.P. Regulation of Folate-Mediated One-Carbon Metabolism by Glycine N-Methyltransferase (GNMT) and Methylenetetrahydrofolate Reductase (MTHFR). J. Nutr. Sci. Vitaminol. 2015, 61, S148–S150. [Google Scholar] [CrossRef] [Green Version]
- Herbig, K.; Chiang, E.P.; Lee, L.R.; Hills, J.; Shane, B.; Stover, P.J. Cytoplasmic serine hydroxymethyltransferase mediates competition between folate-dependent deoxyribonucleotide and S-adenosylmethionine biosyntheses. J. Biol. Chem. 2002, 277, 38381–38389. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nijhout, H.F.; Reed, M.C.; Lam, S.L.; Shane, B.; Gregory, J.F., 3rd; Ulrich, C.M. In silico experimentation with a model of hepatic mitochondrial folate metabolism. Theor. Biol. Med. Model. 2006, 3, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ducker, G.S.; Chen, L.; Morscher, R.J.; Ghergurovich, J.M.; Esposito, M.; Teng, X.; Kang, Y.; Rabinowitz, J.D. Reversal of Cytosolic One-Carbon Flux Compensates for Loss of the Mitochondrial Folate Pathway. Cell Metab. 2016, 23, 1140–1153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thureen, P.J.; Narkewicz, M.R.; Battaglia, F.C.; Tjoa, S.; Fennessey, P.V. Pathways of serine and glycine metabolism in primary culture of ovine fetal hepatocytes. Pediatr. Res. 1995, 38, 775–782. [Google Scholar] [CrossRef] [Green Version]
- Lin, B.F.; Kim, J.S.; Hsu, J.C.; Osborne, C.; Lowe, K.; Garrow, T.; Shane, B. Molecular biology in nutrition research: Modeling of folate metabolism. Adv. Food Nutr. Res. 1996, 40, 95–106. [Google Scholar]
- Kastanos, E.K.; Woldman, Y.Y.; Appling, D.R. Role of mitochondrial and cytoplasmic serine hydroxymethyltransferase isozymes in de novo purine synthesis in Saccharomyces cerevisiae. Biochemistry 1997, 36, 14956–14964. [Google Scholar] [CrossRef]
- Lamers, Y.; Williamson, J.; Ralat, M.; Quinlivan, E.P.; Gilbert, L.R.; Keeling, C.; Stevens, R.D.; Newgard, C.B.; Ueland, P.M.; Meyer, K.; et al. Moderate dietary vitamin B-6 restriction raises plasma glycine and cystathionine concentrations while minimally affecting the rates of glycine turnover and glycine cleavage in healthy men and women. J. Nutr. 2009, 139, 452–460. [Google Scholar] [CrossRef] [Green Version]
- Ryan, W.L.; Carver, M.J. Free amino acids of human foetal and adult liver. Nature 1966, 212, 292–293. [Google Scholar] [CrossRef]
- Fujiwara, K.; Motokawa, Y. Mechanism of the glycine cleavage reaction. Steady state kinetic studies of the P-protein-catalyzed reaction. J. Biol. Chem. 1983, 258, 8156–8162. [Google Scholar]
- MacMillan, L.; Tingley, G.; Young, S.K.; Clow, K.A.; Randell, E.W.; Brosnan, M.E.; Brosnan, J.T. Cobalamin Deficiency Results in Increased Production of Formate Secondary to Decreased Mitochondrial Oxidation of One-Carbon Units in Rats. J. Nutr. 2018, 148, 358–363. [Google Scholar] [CrossRef]
- Palmer, A.M.; Kamynina, E.; Field, M.S.; Stover, P.J. Folate rescues vitamin B12 depletion-induced inhibition of nuclear thymidylate biosynthesis and genome instability. Proc. Natl. Acad. Sci. USA 2017, 114, E4095–E4102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bolusani, S.; Young, B.A.; Cole, N.A.; Tibbetts, A.S.; Momb, J.; Bryant, J.D.; Solmonson, A.; Appling, D.R. Mammalian MTHFD2L encodes a mitochondrial methylenetetrahydrofolate dehydrogenase isozyme expressed in adult tissues. J. Biol. Chem. 2011, 286, 5166–5174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shin, M.; Bryant, J.D.; Momb, J.; Appling, D.R. Mitochondrial MTHFD2L is a dual redox cofactor-specific methylenetetrahydrofolate dehydrogenase/methenyltetrahydrofolate cyclohydrolase expressed in both adult and embryonic tissues. J. Biol. Chem. 2014, 289, 15507–15517. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allen, R.W.; Moskowitz, M. Arrest of cell growth in the G1 phase of the cell cycle by serine deprivation. Exp. Cell Res. 1978, 116, 127–137. [Google Scholar] [CrossRef]
- Rowe, P.B.; Sauer, D.; Fahey, D.; Craig, G.; McCairns, E. One-carbon metabolism in lectin-activated human lymphocytes. Arch. Biochem. Biophys. 1985, 236, 277–288. [Google Scholar] [CrossRef]
- Meiser, J.; Vazquez, A. Give it or take it: The flux of one-carbon in cancer cells. FEBS J. 2016, 283, 3695–3704. [Google Scholar] [CrossRef]
- Ducker, G.S.; Rabinowitz, J.D. One-Carbon Metabolism in Health and Disease. Cell Metab. 2017, 25, 27–42. [Google Scholar] [CrossRef] [Green Version]
- Gregory, J.F., 3rd; Cuskelly, G.J.; Shane, B.; Toth, J.P.; Baumgartner, T.G.; Stacpoole, P.W. Primed, constant infusion with [2H3]serine allows in vivo kinetic measurement of serine turnover, homocysteine remethylation, and transsulfuration processes in human one-carbon metabolism. Am. J. Clin. Nutr. 2000, 72, 1535–1541. [Google Scholar] [CrossRef]
- Fell, D.; Selhub, J. Disruption of thymidylate synthesis and glycine-serine interconversion by L-methionine and L-homocystine in Raji cells. Biochim. Biophys. Acta 1990, 1033, 80–84. [Google Scholar] [CrossRef]
- Liu, Q.; Chen, J.; Liu, L.; Zhang, J.; Wang, D.; Ma, L.; He, Y.; Liu, Y.; Liu, Z.; Wu, J. The X protein of hepatitis B virus inhibits apoptosis in hepatoma cells through enhancing the methionine adenosyltransferase 2A gene expression and reducing S-adenosylmethionine production. J. Biol. Chem. 2011, 286, 17168–17180. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.Y.; Guan, D.Y.; Yang, M.; Wang, H.; Shen, Z.H. All-trans-retinoic acid intensifies endoplasmic reticulum stress in N-acetylglucosaminyltransferase V repressed human hepatocarcinoma cells by perturbing homocysteine metabolism. J. Cell. Biochem. 2010, 109, 468–477. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; Chiang, E.P. Low-dose methotrexate inhibits methionine S-adenosyltransferase in vitro and in vivo. Mol. Med. 2012, 18, 423–432. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.Y.; Lin, J.R.; Darbha, R.; Lin, P.; Liu, T.Y.; Chen, Y.M. Glycine N-methyltransferase tumor susceptibility gene in the benzo(a)pyrene-detoxification pathway. Cancer Res. 2004, 64, 3617–3623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Welty, F.K.; Lichtenstein, A.H.; Barrett, P.H.; Dolnikowski, G.G.; Ordovas, J.M.; Schaefer, E.J. Production of apolipoprotein B-67 in apolipoprotein B-67/B-100 heterozygotes: Technical problems associated with leucine contamination in stable isotope studies. J. Lipid Res. 1997, 38, 1535–1543. [Google Scholar] [PubMed]






| From GCS Derived Formate 3 | ||||||
|---|---|---|---|---|---|---|
| [2-13C]-glycine | Leu + 1 + 2 + 3 | Gly + 1 | Ser + 1 4 | Ser + 24 | dT + 14 | Met + 14 |
| L02 | 0.262 ± 0.001 | 0.573 ± 0.018 | 0.250 ± 0.002 | 0.000 ± 0.000 | 0.003 ± 0.001 | 0.000 ± 0.000 |
| Skep1 | 0.211 ± 0.004 | 0.641 ± 0.014 | 0.116 ± 0.001 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.002 ± 0.002 |
| HepG2 | 0.216 ± 0.001 | 0.573 ± 0.005 | 0.306 ± 0.003 | 0.000 ± 0.000 | 0.001 ± 0.001 | 0.000 ± 0.000 |
| An042 | 0.213 ± 0.002 | 0.436 ± 0.010 | 0.296 ± 0.006 | 0.008 ± 0.001 | 0.016 ± 0.003 | 0.000 ± 0.000 |
| GNMT+ | 0.239 ± 0.006 | 0.489 ± 0.010 | 0.377 ± 0.004 | 0.035 ± 0.001 | 0.049 ± 0.002 | 0.012 ± 0.001 |
| GNMT− | 0.233 ± 0.001 | 0.510 ± 0.002 | 0.420 ± 0.010 | 0.124 ± 0.002 | 0.157 ± 0.004 | 0.000 ± 0.000 |
| L-[2-13C]-serine | Leu + 1 + 2 + 3 | Gly + 1 | Ser + 1 4 | Ser + 2 4 | dT + 1 4 | Met + 1 4 |
| L02 | 0.237 ± 0.001 | 0.463 ± 0.011 | 0.591 ± 0.016 | 0.000 ± 0.000 | 0.004 ± 0.0003 | 0.000 ± 0.000 |
| Skep1 | 0.228 ± 0.004 | 0.539 ± 0.012 | 0.687 ± 0.002 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 |
| HepG2 | 0.215 ± 0.001 | 0.398 ± 0.004 | 0.484 ± 0.006 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 |
| An042 | 0.212 ± 0.002 | 0.351 ± 0.009 | 0.365 ± 0.008 | 0.000 ± 0.000 | 0.004 ± 0.002 | 0.001 ± 0.002 |
| GNMT+ | 0.255 ± 0.000 | 0.429 ± 0.001 | 0.514 ± 0.002 | 0.000 ± 0.000 | 0.012 ± 0.001 | 0.000 ± 0.000 |
| GNMT− | 0.229 ± 0.000 | 0.407 ± 0.028 | 0.505 ± 0.013 | 0.018 ± 0.002 | 0.041 ± 0.001 | 0.000 ± 0.000 |
| From GCS Derived Formate 3 | |||||||
|---|---|---|---|---|---|---|---|
| Gly + 1 | Ser + 1 | deAla + 1 4 | Ser + 23 | deAla + 23,4 | dT + 13 | Met + 13 | |
| GNMT+ | |||||||
| MEM 2 | 0.567 ± 0.012 | 0.404 ± 0.011 | 0.379 ± 0.015 | 0.044 ± 0.003 | 0.039 ± 0.001 | 0.044 ± 0.001 | 0.001 ± 0.002 |
| MEM + B12 2 | 0.489 ± 0.010 | 0.377 ± 0.004 | 0.353 ± 0.006 | 0.035 ± 0.001 | 0.033 ± 0.0002 | 0.049 ± 0.002 | 0.012 ± 0.001 |
| MEM + B12 + NEAA 2 | 0.452 ± 0.009 | 0.345 ± 0.005 | 0.322 ± 0.005 | 0.014 ± 0.001 | 0.014 ± 0.001 | 0.012 ± 0.001 | 0.009 ± 0.001 |
| GNMT− | |||||||
| MEM 2 | 0.579 ± 0.019 | 0.458 ± 0.002 | 0.433 ± 0.003 | 0.116 ± 0.0003 | 0.107 ± 0.0001 | 0.134 ± 0.001 | 0.000 ± 0.000 |
| MEM + B12 2 | 0.510 ± 0.002 | 0.420 ± 0.010 | 0.385 ± 0.001 | 0.124 ± 0.002 | 0.109 ± 0.001 | 0.157 ± 0.004 | 0.000 ± 0.000 |
| MEM + B12 + NEAA 2 | 0.469 ± 0.004 | 0.390 ± 0.001 | 0.370 ± 0.004 | 0.062 ± 0.0004 | 0.058 ± 0.001 | 0.096 ± 0.001 | 0.001 ± 0.001 |
| (A) Isotopic enrichments from [2-13C]glycine in serine, dTMP, and methionine 1,2,3 | |||||||
| From GCS Derived Formate3 | |||||||
| Nutritional effects 5,7 | Gly + 1 | Ser + 1 | deAla + 1 | Ser + 23 | deAla + 23,4 | dT + 13 | Met + 13 |
| GNMT+ | |||||||
| MEM + B12 vs. MEM | −14% | −7% | −7% | −20% | −15% | +11% | +1100% |
| MEM + B12 + NEAA vs. MEM | −20% | −15% | −15% | −68% | −64% | −73% | +800% |
| GNMT− | |||||||
| MEM + B12 vs. MEM | −12% | −8% | −11% | 7% | 2% | +17% | ND |
| MEM + B12 + NEAA vs. MEM | −19% | −15% | −15% | −47% | −46% | −28% | ND |
| (B) Vitamin B12 and NEAA supply affect C2-glycine derived formate utilization in serine, dTMP, methionine 5 | |||||||
| From GCS Derived Formate3 | |||||||
| GNMT effects 6,7 (GNMT+ vs. GNNT-) | Gly + 1 | Ser + 1 | deAla + 1 | Ser + 23 | deAla + 2 3,4 | dT + 13 | |
| in MEM | −2% | −12% | −12% | −62% | −64% | −67% | |
| in MEM + B12 | −4% | −10% | −8% | −72% | −70% | −69% | |
| in MEM + B12 + NEAA | −4% | −12% | −13% | −77% | −76% | −88% | |
| (C) GNMT expression affect C2-glycine derived formate utilization in serine and dTMP 6 | |||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, Y.-L.; Sou, N.-L.; Tang, F.-Y.; Ko, H.-A.; Yeh, W.-T.; Peng, J.-H.; Chiang, E.-P.I. Tracing Metabolic Fate of Mitochondrial Glycine Cleavage System Derived Formate In Vitro and In Vivo. Int. J. Mol. Sci. 2020, 21, 8808. https://doi.org/10.3390/ijms21228808
Tan Y-L, Sou N-L, Tang F-Y, Ko H-A, Yeh W-T, Peng J-H, Chiang E-PI. Tracing Metabolic Fate of Mitochondrial Glycine Cleavage System Derived Formate In Vitro and In Vivo. International Journal of Molecular Sciences. 2020; 21(22):8808. https://doi.org/10.3390/ijms21228808
Chicago/Turabian StyleTan, Yee-Ling, Nga-Lai Sou, Feng-Yao Tang, Hsin-An Ko, Wei-Ting Yeh, Jian-Hau Peng, and En-Pei Isabel Chiang. 2020. "Tracing Metabolic Fate of Mitochondrial Glycine Cleavage System Derived Formate In Vitro and In Vivo" International Journal of Molecular Sciences 21, no. 22: 8808. https://doi.org/10.3390/ijms21228808