Patient-Derived Organoids of Cholangiocarcinoma
Abstract
:1. Introduction
2. Results
2.1. Establishment of Human Cholangiocarcinoma Organoid Lines from Surgical Specimens
Organoid-Derived 2D Cell Lines
2.2. Patient-Derived Organoid-Based Xenografts of Cholangiocarcinoma
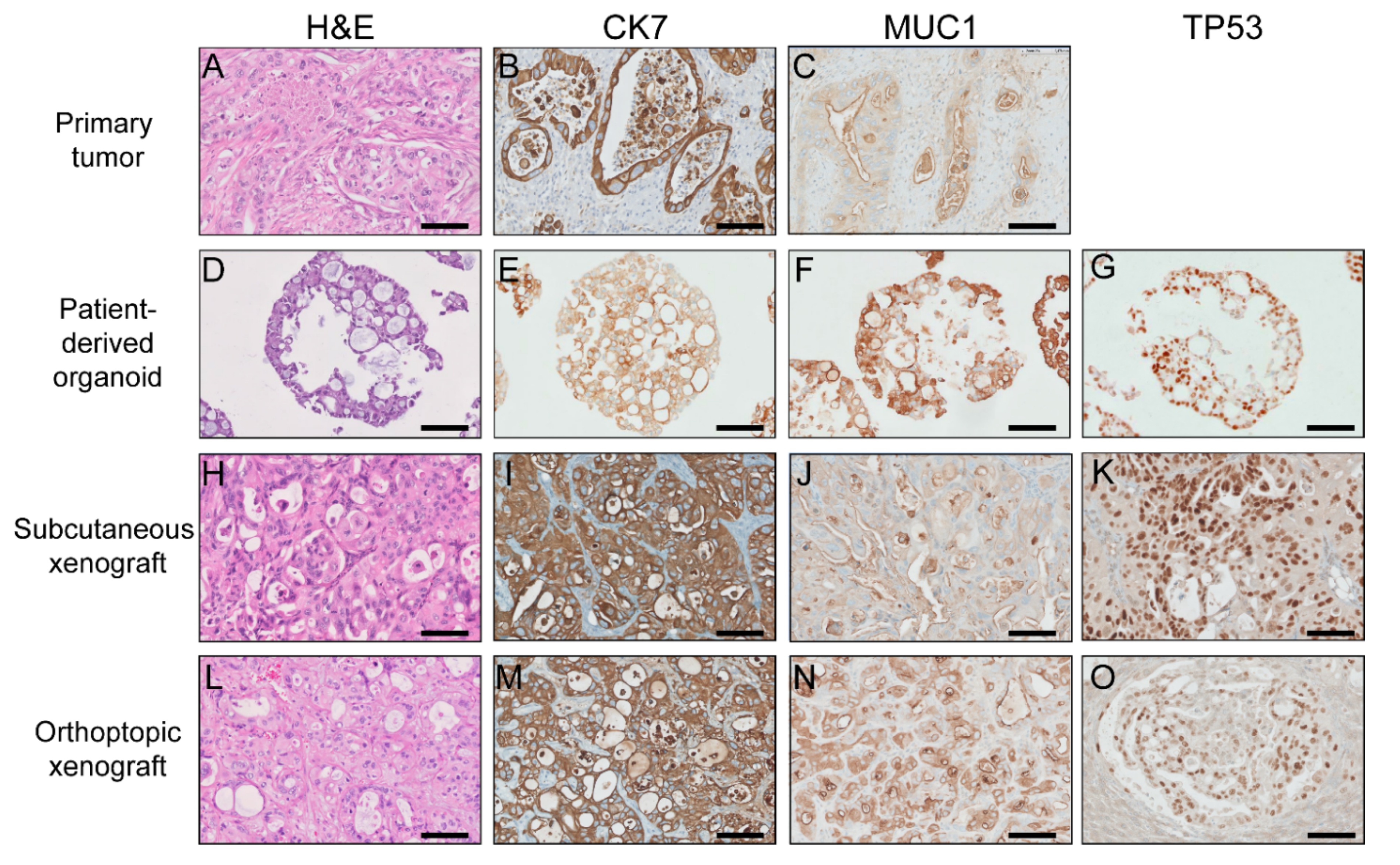
2.3. Patient-Derived Organoids Retained the Histological Characteristics of the Parental Tumor
2.4. Patient-Derived Organoids Recapitulate Parental Tumor Molecular Features
2.5. Therapeutic Response of the Human Preclinical Cholangiocarcinoma Models
3. Discussion
4. Materials and Methods
4.1. Human Samples
4.2. Mice
4.3. Organoid Culture
4.4. Cell Culture
4.5. Treatment
4.6. Patient-Derived Organoids Xenografts
4.7. Histology and Immunohistochemistry
4.8. Next-Generation Sequencing
4.9. Sequencing Data Analysis
4.10. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lendvai, G.; Szekerczés, T.; Illyés, I.; Dóra, R.; Kontsek, E.; Gógl, A.; Kiss, A.; Werling, K.; Kovalszky, I.; Schaff, Z.; et al. Cholangiocarcinoma: Classification, Histopathology and Molecular Carcinogenesis. Pathol. Oncol. Res. 2020, 26, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Bergquist, A.; von Seth, E. Epidemiology of Cholangiocarcinoma. Best Pract. Res. Clin. Gastroenterol. 2015, 29, 221–232. [Google Scholar] [CrossRef] [PubMed]
- DeOliveira, M.; Cunningham, S.; Cameron, J.; Kamangar, F.; Winter, J.; Lillemoe, K.; Choti, M.; Yeo, C.; Schulick, R. Cholangiocarcinoma: Thirty-One-Year Experience with 564 Patients at a Single Institution. Ann. Surg. 2007, 245, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Nakeeb, A.; Pitt, H.; Sohn, T.; Coleman, J.; Abrams, R.; Piantadosi, S.; Hruban, R.; Lillemoe, K.; Yeo, C.; Cameron, J. Cholangiocarcinoma. A Spectrum of Intrahepatic, Perihilar, and Distal Tumors. Ann. Surg. 1996, 224, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Tyson, G.; El-Serag, H. Risk Factors of Cholangiocarcinoma. Hepatology 2011, 54, 173–184. [Google Scholar] [CrossRef]
- Khan, S.; Toledano, M.; Taylor-Robinson, S. Epidemiology, Risk Factors, and Pathogenesis of Cholangiocarcinoma. HPB 2008, 10, 77–82. [Google Scholar] [CrossRef] [Green Version]
- Lazaridis, K.; Gores, G. Cholangiocarcinoma. Gastroenterology 2005, 128, 1655–1667. [Google Scholar] [CrossRef] [PubMed]
- Razumilava, N.; Gores, G.J. Cholangiocarcinoma. Lancet 2014, 383, 2168–2179. [Google Scholar] [CrossRef] [Green Version]
- The American Cancer Society Home Page. Available online: https://www.cancer.org/cancer/bile-duct-cancer/detection-diagnosis-staging/survival-by-stage.html (accessed on 21 July 2021).
- Chan-On, W.; Nairismägi, M.L.; Ong, C.; Lim, W.; Dima, S.; Pairojkul, C.; Lim, K.; McPherson, J.; Cutcutache, I.; Heng, H.; et al. Exome Sequencing Identifies Distinct Mutational Patterns in Liver Fluke-Related and Non-Infection-Related Bile Duct Cancers. Nat. Genet. 2013, 45, 1474–1478. [Google Scholar] [CrossRef]
- Jiao, Y.; Pawlik, T.; Anders, R.; Selaru, F.; Streppel, M.; Lucas, D.; Niknafs, N.; Guthrie, V.; Maitra, A.; Argani, P.; et al. Exome Sequencing Identifies Frequent Inactivating Mutations in BAP1, ARID1A and PBRM1 in Intrahepatic Cholangiocarcinomas. Nat. Genet. 2013, 45, 1470–1473. [Google Scholar] [CrossRef]
- Ong, C.; Subimerb, C.; Pairojkul, C.; Wongkham, S.; Cutcutache, I.; Yu, W.; McPherson, J.; Allen, G.; Ng, C.; Wong, B.; et al. Exome Sequencing of Liver Fluke-Associated Cholangiocarcinoma. Nat. Genet. 2012, 44, 690–693. [Google Scholar] [CrossRef]
- Borger, D.; Tanabe, K.; Fan, K.; Lopez, H.; Fantin, V.; Straley, K.; Schenkein, D.; Hezel, A.; Ancukiewicz, M.; Liebman, H.; et al. Frequent Mutation of Isocitrate Dehydrogenase (IDH)1 and IDH2 in Cholangiocarcinoma Identified through Broad-Based Tumor Genotyping. Oncologist 2012, 17, 72–79. [Google Scholar] [CrossRef] [Green Version]
- Banales, J.; Cardinale, V.; Carpino, G.; Marzioni, M.; Andersen, J.; Invernizzi, P.; Lind, G.; Folseraas, T.; Forbes, S.; Fouassier, L.; et al. Expert Consensus Document: Cholangiocarcinoma: Current Knowledge and Future Perspectives Consensus Statement from the European Network for the Study of Cholangiocarcinoma (ENS-CCA). Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 261–280. [Google Scholar] [CrossRef]
- Brito, A.; Abrantes, A.; Encarnação, J.; Tralhão, J.; Botelho, M. Cholangiocarcinoma: From Molecular Biology to Treatment. Med. Oncol. 2015, 32, 245. [Google Scholar] [CrossRef] [Green Version]
- Nakamura, H.; Arai, Y.; Totoki, Y.; Shirota, T.; Elzawahry, A.; Kato, M.; Hama, N.; Hosoda, F.; Urushidate, T.; Ohashi, S.; et al. Genomic Spectra of Biliary Tract Cancer. Nat. Genet. 2015, 47, 1003–1010. [Google Scholar] [CrossRef]
- Andersen, J.; Spee, B.; Blechacz, B.; Avital, I.; Komuta, M.; Barbour, A.; Conner, E.; Gillen, M.; Roskams, T.; Roberts, L.; et al. Genomic and Genetic Characterization of Cholangiocarcinoma Identifies Therapeutic Targets for Tyrosine Kinase Inhibitors. Gastroenterology 2012, 142, 1021–1031. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rizzo, A.; Ricci, A.D.; Brandi, G. Futibatinib, an Investigational Agent for the Treatment of Intrahepatic Cholangiocarcinoma: Evidence to Date and Future Perspectives. Expert Opin. Investig. Drugs 2021, 30, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Lau, D.K.; Mouradov, D.; Wasenang, W.; Luk, I.Y.; Scott, C.M.; Williams, D.S.; Yeung, Y.H.; Limpaiboon, T.; Iatropoulos, G.F.; Jenkins, L.J.; et al. Genomic Profiling of Biliary Tract Cancer Cell Lines Reveals Molecular Subtypes and Actionable Drug Targets. iScience 2019, 21, 624–637. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Ding, X.; Wang, S.; Moser, C.D.; Shaleh, H.M.; Mohamed, E.A.; Chaiteerakij, R.; Allotey, L.K.; Chen, G.; Miyabe, K.; et al. Antitumor Effect of FGFR Inhibitors on a Novel Cholangiocarcinoma Patient Derived Xenograft Mouse Model Endogenously Expressing an FGFR2-CCDC6 Fusion Protein. Cancer Lett. 2016, 380, 163–173. [Google Scholar] [CrossRef] [Green Version]
- Mancarella, S.; Serino, G.; Dituri, F.; Cigliano, A.; Ribback, S.; Wang, J.; Chen, X.; Calvisi, D.F.; Giannelli, G. Crenigacestat, a Selective NOTCH1 Inhibitor, Reduces Intrahepatic Cholangiocarcinoma Progression by Blocking VEGFA/DLL4/MMP13 Axis. Cell Death Differ. 2020, 27, 2330–2343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ku, J.; Yoon, K.; Kim, I.; Kim, W.; Jang, J.; Suh, K.; Kim, S.; Park, Y.; Hwang, J.; Yoon, Y.; et al. Establishment and Characterisation of Six Human Biliary Tract Cancer Cell Lines. Br. J. Cancer 2002, 87, 187–193. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cavalloni, G.; Peraldo-Neia, C.; Varamo, C.; Casorzo, L.; Dell’Aglio, C.; Bernabei, P.; Chiorino, G.; Aglietta, M.; Leone, F. Establishment and Characterization of a Human Intrahepatic Cholangiocarcinoma Cell Line Derived from an Italian Patient. Tumour Biol. 2016, 37, 4041–4052. [Google Scholar] [CrossRef] [Green Version]
- Hidalgo, M.; Amant, F.; Biankin, A.; Budinská, E.; Byrne, A.; Caldas, C.; Clarke, R.; de Jong, S.; Jonkers, J.; Mælandsmo, G.; et al. Patient-Derived Xenograft Models: An Emerging Platform for Translational Cancer Research. Cancer Discov. 2014, 4, 998–1013. [Google Scholar] [CrossRef] [Green Version]
- Zach, S.; Birgin, E.; Rückert, F. Primary Cholangiocellular Carcinoma Cell Lines. J. Stem Cell Res. Transplant. 2015, 2, 1–7. [Google Scholar]
- Augustyniak, J.; Bertero, A.; Coccini, T.; Baderna, D.; Buzanska, L.; Caloni, F. Organoids Are Promising Tools for Species—Specific in Vitro Toxicological Studies. J. Appl. Toxicol. 2019, 39, 1610–1622. [Google Scholar] [CrossRef]
- Broutier, L.; Mastrogiovanni, G.; Verstegen, M.; Francies, H.; Gavarró, L.; Bradshaw, C.; Allen, G.; Arnes-Benito, R.; Sidorova, O.; Gaspersz, M.; et al. Human Primary Liver Cancer-Derived Organoid Cultures for Disease Modeling and Drug Screening. Nat. Med. 2017, 23, 1424–1435. [Google Scholar] [CrossRef]
- Baiocchi, L.; Sato, K.; Ekser, B.; Kennedy, L.; Francis, H.; Ceci, L.; Lenci, I.; Alvaro, D.; Franchitto, A.; Onori, P.; et al. Cholangiocarcinoma: Bridging the Translational Gap from Preclinical to Clinical Development and Implications for Future Therapy. Expert Opin. Investig. Drugs 2021, 30, 365–375. [Google Scholar] [CrossRef]
- Nuciforo, S.; Fofana, I.; Matter, M.S.; Blumer, T.; Calabrese, D.; Boldanova, T.; Piscuoglio, S.; Wieland, S.; Ringnalda, F.; Schwank, G.; et al. Organoid Models of Human Liver Cancers Derived from Tumor Needle Biopsies. Cell Rep. 2018, 24, 1363–1376. [Google Scholar] [CrossRef] [Green Version]
- Waddell, S.H.; Boulter, L. Developing Models of Cholangiocarcinoma to Close the Translational Gap in Cancer Research. Expert Opin. Investig. Drugs 2021, 30, 439–450. [Google Scholar] [CrossRef]
- Amato, F.; Rae, C.; Prete, M.G.; Braconi, C. Cholangiocarcinoma Disease Modelling Through Patients Derived Organoids. Cells 2020, 9, 832. [Google Scholar] [CrossRef] [Green Version]
- Lampis, A.; Carotenuto, P.; Vlachogiannis, G.; Cascione, L.; Hedayat, S.; Burke, R.; Clarke, P.; Bosma, E.; Simbolo, M.; Scarpa, A.; et al. MIR21 Drives Resistance to Heat Shock Protein 90 Inhibition in Cholangiocarcinoma. Gastroenterology 2018, 154, 1066–1079.e5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Massa, A.; Varamo, C.; Vita, F.; Tavolari, S.; Peraldo-Neia, C.; Brandi, G.; Rizzo, A.; Cavalloni, G.; Aglietta, M. Evolution of the Experimental Models of Cholangiocarcinoma. Cancers 2020, 12, 2308. [Google Scholar] [CrossRef]
- Bartfeld, S.; Bayram, T.; van de Wetering, M.; Huch, M.; Begthel, H.; Kujala, P.; Vries, R.; Peters, P.; Clevers, H. In Vitro Expansion of Human Gastric Epithelial Stem Cells and Their Responses to Bacterial Infection. Gastroenterology 2015, 148, 126–136. [Google Scholar] [CrossRef] [Green Version]
- Saijyo, S.; Kudo, T.; Suzuki, M.; Katayose, Y.; Shinoda, M.; Muto, T.; Fukuhara, K.; Suzuki, T.; Matsuno, S. Establishment of a New Extrahepatic Bile Duct Carcinoma Cell Line, TFK-1. Tohoku J. Exp. Med. 1995, 177, 61–71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rückert, F.; Aust, D.; Böhme, I.; Werner, K.; Brandt, A.; Diamandis, E.P.; Krautz, C.; Hering, S.; Saeger, H.-D.; Grützmann, R.; et al. Five Primary Human Pancreatic Adenocarcinoma Cell Lines Established by the Outgrowth Method. J. Surg. Res. 2012, 172, 29–39. [Google Scholar] [CrossRef]
- Coughlan, A.M.; Harmon, C.; Whelan, S.; O’Brien, E.C.; O’Reilly, V.P.; Crotty, P.; Kelly, P.; Ryan, M.; Hickey, F.B.; O’Farrelly, C.; et al. Myeloid Engraftment in Humanized Mice: Impact of Granulocyte-Colony Stimulating Factor Treatment and Transgenic Mouse Strain. Stem Cells Dev. 2016, 25, 530–541. [Google Scholar] [CrossRef]
- Rizvi, S.; Khan, S.A.; Hallemeier, C.L.; Kelley, R.K.; Gores, G.J. Cholangiocarcinoma—Evolving Concepts and Therapeutic Strategies. Nat. Rev. Clin. Oncol. 2018, 15, 95–111. [Google Scholar] [CrossRef] [Green Version]
- Cardinale, V.; Renzi, A.; Carpino, G.; Torrice, A.; Bragazzi, M.; Giuliante, F.; DeRose, A.; Fraveto, A.; Onori, P.; Napoletano, C.; et al. Profiles of Cancer Stem Cell Subpopulations in Cholangiocarcinomas. Am. J. Pathol. 2015, 185, 1724–1739. [Google Scholar] [CrossRef]
- Kim, D.; Jeong, Y.; Chung, C.; Kim, C.; Kwak, T.; Lee, H.; Kang, D. Preclinical Evaluation of Sorafenib-Eluting Stent for Suppression of Human Cholangiocarcinoma Cells. Int. J. Nanomed. 2013, 8, 1697–1711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, C.; Maass, T.; Krupp, M.; Thieringer, F.; Strand, S.; Wörns, M.; Barreiros, A.; Galle, P.; Teufel, A. A Systems Biology Perspective on Cholangiocellular Carcinoma Development: Focus on MAPK-Signaling and the Extracellular Environment. J. Hepatol. 2009, 50, 1122–1131. [Google Scholar] [CrossRef]
- Sugiyama, H.; Onuki, K.; Ishige, K.; Baba, N.; Ueda, T.; Matsuda, S.; Takeuchi, K.; Onodera, M.; Nakanuma, Y.; Yamato, M.; et al. Potent in Vitro and in Vivo Antitumor Activity of Sorafenib against Human Intrahepatic Cholangiocarcinoma Cells. J. Gastroenterol. 2011, 46, 779–789. [Google Scholar] [CrossRef]
- Huether, A.; Höpfner, M.; Baradari, V.; Schuppan, D.; Scherübl, H. Sorafenib Alone or as Combination Therapy for Growth Control of Cholangiocarcinoma. Biochem. Pharmacol. 2007, 73, 1308–1317. [Google Scholar] [CrossRef]
- Bengala, C.; Bertolini, F.; Malavasi, N.; Boni, C.; Aitini, E.; Dealis, C.; Zironi, S.; Depenni, R.; Fontana, A.; Del Giovane, C.; et al. Sorafenib in Patients with Advanced Biliary Tract Carcinoma: A Phase II Trial. Br. J. Cancer 2010, 102, 68–72. [Google Scholar] [CrossRef] [Green Version]
- El-Khoueiry, A.; Rankin, C.; Ben-Josef, E.; Lenz, H.; Gold, P.; Hamilton, R.; Govindarajan, R.; Eng, C.; Blanke, C. SWOG 0514: A Phase II Study of Sorafenib in Patients with Unresectable or Metastatic Gallbladder Carcinoma and Cholangiocarcinoma. Investig. New Drugs 2012, 30, 1646–1651. [Google Scholar] [CrossRef] [Green Version]
- Pan, T.; Wang, W.; Jia, W.; Xu, G. A Single-Center Experience of Sorafenib Monotherapy in Patients with Advanced Intrahepatic Cholangiocarcinoma. Oncol. Lett. 2017, 13, 2957–2964. [Google Scholar] [CrossRef] [Green Version]
- Pinter, M.; Sieghart, W.; Reisegger, M.; Wrba, F.; Peck-Radosavljevic, M. Sorafenib in Unresectable Intrahepatic Cholangiocellular Carcinoma: A Case Report. Wien. Klin. Wochenschr. 2011, 123, 61–64. [Google Scholar] [CrossRef]
- LaRocca, R.; Hicks, M.; Mull, L.; Foreman, B. Effective Palliation of Advanced Cholangiocarcinoma with Sorafenib: A Two-Patient Case Report. J. Gastrointest. Cancer 2007, 38, 154–156. [Google Scholar] [CrossRef]
- Poddubskaya, E.; Baranova, M.; Allina, D.; Smirnov, P.; Albert, E.; Kirilchev, A.; Aleshin, A.; Sekacheva, M.; Suntsova, M. Personalized Prescription of Tyrosine Kinase Inhibitors in Unresectable Metastatic Cholangiocarcinoma. Exp. Hematol. Oncol. 2018, 7, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Di Sotto, A.; Di Giacomo, S.; Rubini, E.; Macone, A.; Gulli, M.; Mammola, C.; Eufemi, M.; Mancinelli, R.; Mazzanti, G. Modulation of STAT3 Signaling, Cell Redox Defenses and Cell Cycle Checkpoints by β-Caryophyllene in Cholangiocarcinoma Cells: Possible Mechanisms Accounting for Doxorubicin Chemosensitization and Chemoprevention. Cells 2020, 9, 858. [Google Scholar] [CrossRef] [Green Version]
- Di Giorgio, A.; Sgarbura, O.; Rotolo, S.; Schena, C.; Bagalà, C.; Inzani, F.; Russo, A.; Chiantera, V.; Pacelli, F. Pressurized Intraperitoneal Aerosol Chemotherapy with Cisplatin and Doxorubicin or Oxaliplatin for Peritoneal Metastasis from Pancreatic Adenocarcinoma and Cholangiocarcinoma. Ther. Adv. Med. Oncol. 2020, 12, 1758835920940887. [Google Scholar] [CrossRef]
- Pellino, A.; Loupakis, F.; Cadamuro, M.; Dadduzio, V.; Fassan, M.; Guido, M.; Cillo, U.; Indraccolo, S.; Fabris, L. Precision Medicine in Cholangiocarcinoma. Transl. Gastroenterol. Hepatol. 2018, 3, 40. [Google Scholar] [CrossRef]
- Jusakul, A.; Cutcutache, I.; Yong, C.H.; Lim, J.Q.; Huang, M.N.; Padmanabhan, N.; Nellore, V.; Kongpetch, S.; Ng, A.W.T.; Ng, L.M.; et al. Whole-Genome and Epigenomic Landscapes of Etiologically Distinct Subtypes of Cholangiocarcinoma. Cancer Discov. 2017, 7, 1116–1135. [Google Scholar] [CrossRef] [Green Version]
- Vogel, A.; Saborowski, A. Current and Future Systemic Therapies in Biliary Tract Cancer. Visc. Med. 2021, 37, 32–38. [Google Scholar] [CrossRef]
- Seidlitz, T.; Merker, S.R.; Rothe, A.; Zakrzewski, F.; von Neubeck, C.; Grützmann, K.; Sommer, U.; Schweitzer, C.; Schölch, S.; Uhlemann, H.; et al. Human Gastric Cancer Modelling Using Organoids. Gut 2019, 68, 207–217. [Google Scholar] [CrossRef] [Green Version]
- Ponsioen, B.; Post, J.B.; Buissant des Amorie, J.R.; Laskaris, D.; van Ineveld, R.L.; Kersten, S.; Bertotti, A.; Sassi, F.; Sipieter, F.; Cappe, B.; et al. Quantifying Single-Cell ERK Dynamics in Colorectal Cancer Organoids Reveals EGFR as an Amplifier of Oncogenic MAPK Pathway Signalling. Nat. Cell Biol. 2021, 23, 377–390. [Google Scholar] [CrossRef]
- Karkampouna, S.; La Manna, F.; Benjak, A.; Kiener, M.; De Menna, M.; Zoni, E.; Grosjean, J.; Klima, I.; Garofoli, A.; Bolis, M.; et al. Patient-Derived Xenografts and Organoids Model Therapy Response in Prostate Cancer. Nat. Commun. 2021, 12, 1117. [Google Scholar] [CrossRef] [PubMed]
- Drost, J.; Clevers, H. Organoids in Cancer Research. Nat. Rev. Cancer 2018, 18, 407–418. [Google Scholar] [CrossRef]
- Saito, Y.; Muramatsu, T.; Kanai, Y.; Ojima, H.; Sukeda, A.; Hiraoka, N.; Arai, E.; Sugiyama, Y.; Matsuzaki, J.; Uchida, R.; et al. Establishment of Patient-Derived Organoids and Drug Screening for Biliary Tract Carcinoma. Cell Rep. 2019, 27, 1265–1276.e4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sachs, N.; Clevers, H. Organoid Cultures for the Analysis of Cancer Phenotypes. Curr. Opin. Genet. Dev. 2014, 24, 68–73. [Google Scholar] [CrossRef]
- Gu, Q.; Zhang, B.; Sun, H.; Xu, Q.; Tan, Y.; Wang, G.; Luo, Q.; Xu, W.; Yang, S.; Li, J.; et al. Genomic Characterization of a Large Panel of Patient-Derived Hepatocellular Carcinoma Xenograft Tumor Models for Preclinical Development. Oncotarget 2015, 6, 20160–20176. [Google Scholar] [CrossRef] [Green Version]
- Vlachogiannis, G.; Hedayat, S.; Vatsiou, A.; Jamin, Y.; Fernández-Mateos, J.; Khan, K.; Lampis, A.; Eason, K.; Huntingford, I.; Burke, R.; et al. Patient-Derived Organoids Model Treatment Response of Metastatic Gastrointestinal Cancers. Science 2018, 359, 920–926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oliveira, I.S.; Kilcoyne, A.; Everett, J.M.; Mino-Kenudson, M.; Harisinghani, M.G.; Ganesan, K. Cholangiocarcinoma: Classification, Diagnosis, Staging, Imaging Features, and Management. Abdom. Radiol. 2017, 42, 1637–1649. [Google Scholar] [CrossRef]
- Aishima, S.; Asayama, Y.; Taguchi, K.; Sugimachi, K.; Shirabe, K.; Shimada, M.; Sugimachi, K.; Tsuneyoshi, M. The Utility of Keratin 903 as a New Prognostic Marker in Mass-Forming-Type Intrahepatic Cholangiocarcinoma. Mod. Pathol. 2002, 15, 1181–1190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lau, S.; Prakash, S.; Geller, S.; Alsabeh, R. Comparative Immunohistochemical Profile of Hepatocellular Carcinoma, Cholangiocarcinoma, and Metastatic Adenocarcinoma. Hum. Pathol. 2002, 33, 1175–1181. [Google Scholar] [CrossRef] [PubMed]
- Vijgen, S.; Terris, B.; Rubbia-Brandt, L. Pathology of Intrahepatic Cholangiocarcinoma. Hepatobiliary Surg. Nutr. 2017, 6, 22–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kasprzak, A.; Adamek, A. Mucins: The Old, the New and the Promising Factors in Hepatobiliary Carcinogenesis. Int. J. Mol. Sci. 2019, 20, 1288. [Google Scholar] [CrossRef] [Green Version]
- Park, S.; Roh, S.; Kim, Y.; SZ, K.; Park, H.; Jang, K.; Chung, M.; Kang, M.; Lee, D.; Moon, W. Expression of MUC1, MUC2, MUC5AC and MUC6 in Cholangiocarcinoma: Prognostic Impact. Oncol. Rep. 2009, 22, 649–657. [Google Scholar] [PubMed] [Green Version]
- Blechacz, B.; Komuta, M.; Roskams, T.; Gores, G.J. Clinical Diagnosis and Staging of Cholangiocarcinoma. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 512–522. [Google Scholar] [CrossRef] [PubMed] [Green Version]





| Patient ID | Age (Years) | Gender | Tumor Type | Isolation Method | Culture Medium * | Culture Period (Weeks) |
|---|---|---|---|---|---|---|
| 2 | 76 | M | CC | Manually picked | 1 | 1.1 |
| 5 | 54 | M | iCC | Manually picked | 1 | 5.0 |
| 5 | 54 | M | iCC | SC | 1 | 5.0 |
| 5 | 54 | M | iCC | CCL | 1 | 7.0 |
| 7 | 65 | M | iCC | CCL | 1 | 11.3 |
| 14 | 71 | F | iCC | CCL | 1 | 7.1 |
| 15 | 56 | F | CC | CCL | 1 | 7.1 |
| 19 | 74 | M | iCC | CCL | 1 | 7.0 |
| 20 | 76 | M | CC | CCL | 1 | 8.7 |
| 23 | 68 | M | pCC | CCL | 2 | 8.0 |
| 26 | 26 | F | dCC | CCL | 2 | 2.1 |
| 42 | 46 | M | CC | CCL + SC | 2 | 6.4 |
| 44 | 56 | M | pCC | CCL + SC | 2 | 2.0 |
| 50 | 73 | F | CC | CCL + SC | 2 | 6.4 |
| 51 | 72 | F | CC | CCL + SC | 2 | 5.9 |
| 61 | 69 | F | iCC | CCL | 3 | 35.3 |
| 68 | 57 | F | iCC | CCL + SC | 3 | 103.3 |
| 70 | 80 | F | iCC | CCL + SC | 3 | 2.0 |
| 71 | 60 | M | iCC | CCL + SC | 3 | 3.0 |
| 81 | 82 | M | dCC | CCL + SC | 3 | 1.0 |
| 83 | 68 | F | pCC | CCL | 3 | 86.4 |
| 86 | 67 | F | dCC | SC | 3 | 1.0 |
| 95 | 72 | M | iCC | SC | 3 | 2.1 |
| 99 | 63 | F | iCC | CCL + SC | 3 | 2.0 |
| 108 | 53 | M | iCC | CCL + SC | 3 | 20.1 |
| 109 | 78 | F | pCC | CCL + SC | 3 | 15.9 |
| 115 | 65 | M | pCC | CCL + SC | 3 | 4.3 |
| 118 | 53 | F | iCC | CCL + SC | 3 | 6.6 |
| 125 | 69 | M | pCC | CCL + SC | 3 | 7.1 |
| Patient-ID | Sex | Age at Diagnosis | Tumor Location | TNM-Stage | Grading |
|---|---|---|---|---|---|
| P68 | Female | 57 | Intrahepatic | T2a N1 M1 | G2 |
| P83 | Female | 86 | Perihilar | T3 N0 M0 | G3 |
| TFK-1 | Male | 63 | Extrahepatic |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maier, C.F.; Zhu, L.; Nanduri, L.K.; Kühn, D.; Kochall, S.; Thepkaysone, M.-L.; William, D.; Grützmann, K.; Klink, B.; Betge, J.; et al. Patient-Derived Organoids of Cholangiocarcinoma. Int. J. Mol. Sci. 2021, 22, 8675. https://doi.org/10.3390/ijms22168675
Maier CF, Zhu L, Nanduri LK, Kühn D, Kochall S, Thepkaysone M-L, William D, Grützmann K, Klink B, Betge J, et al. Patient-Derived Organoids of Cholangiocarcinoma. International Journal of Molecular Sciences. 2021; 22(16):8675. https://doi.org/10.3390/ijms22168675
Chicago/Turabian StyleMaier, Christopher Fabian, Lei Zhu, Lahiri Kanth Nanduri, Daniel Kühn, Susan Kochall, May-Linn Thepkaysone, Doreen William, Konrad Grützmann, Barbara Klink, Johannes Betge, and et al. 2021. "Patient-Derived Organoids of Cholangiocarcinoma" International Journal of Molecular Sciences 22, no. 16: 8675. https://doi.org/10.3390/ijms22168675









