Sigma-1 Receptor in Retina: Neuroprotective Effects and Potential Mechanisms
Abstract
:1. Introduction
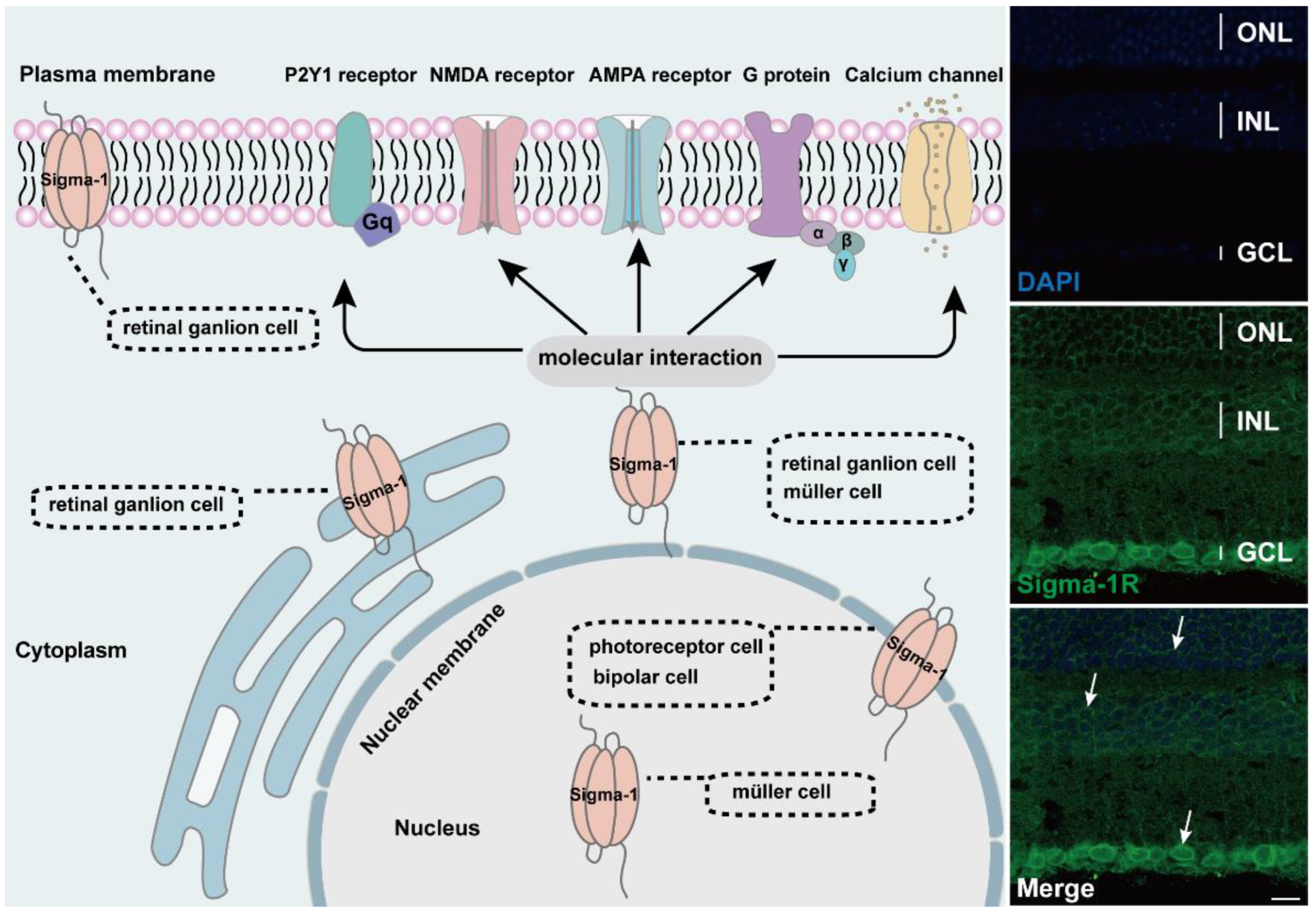
2. Expression and Molecular Interactions of Sigma-1 Receptor in Retina
2.1. Expression of Sigma-1 Receptor in Retinal Neurons
2.2. Molecular Interaction of Sigma-1 Receptor in Retina
3. Relevant Signaling Pathways of Sigma-1 Receptor in Retina
3.1. Nrf2 Signaling Pathway
3.2. MAPK Signaling Pathway
3.3. Other Related Signaling Pathways
4. Neuroprotective Effects of Sigma-1 Receptor
4.1. Suppression of Oxidative Stress
4.2. Alleviation of Pathological Autophagy
4.3. Reduction of Cell Apoptosis
4.4. Anti-Inflammation
5. The Sigma-1 Receptor-Associated Diseases
5.1. Diseases in Retina
5.2. Other Diseases
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Martin, W.R.; Eades, C.G.; Thompson, J.A.; Huppler, R.E.; Gilbert, P.E. The effects of morphine- and nalorphine- like drugs in the nondependent and morphine-dependent chronic spinal dog. J. Pharmacol. Exp. Ther. 1976, 197, 517–532. [Google Scholar] [PubMed]
- Vaupel, D.B. Naltrexone fails to antagonize the sigma effects of PCP and SKF 10,047 in the dog. Eur. J. Pharmacol. 1983, 92, 269–274. [Google Scholar] [CrossRef]
- Katz, J.L.; Spealman, R.D.; Clark, R.D. Stereoselective behavioral effects of N-allylnormetazocine in pigeons and squirrel monkeys. J. Pharmacol. Exp. Ther. 1985, 232, 452–461. [Google Scholar] [PubMed]
- Slifer, B.L.; Balster, R.L. Reinforcing properties of stereoisomers of the putative sigma agonists N-allylnormetazocine and cyclazocine in rhesus monkeys. J. Pharmacol. Exp. Ther. 1983, 225, 522–528. [Google Scholar]
- Su, T.P. Evidence for sigma opioid receptor: Binding of [3H]SKF-10047 to etorphine-inaccessible sites in guinea-pig brain. J. Pharmacol. Exp. Ther. 1982, 223, 284–290. [Google Scholar]
- Hellewell, S.B.; Bowen, W.D. A sigma -like binding site in rat pheochromocytoma (PC12) cells: Decreased affinity for (+)-benzomorphans and lower molecular weight suggest a different sigma receptor form from that of guinea pig brain. Brain Res. 1990, 527, 244–253. [Google Scholar] [CrossRef]
- Walker, J.M.; Bowen, W.D.; Walker, F.O.; Matsumoto, R.R.; De Costa, B.; Rice, K.C. Sigma receptors: Biology and function. Pharmacol. Rev. 1990, 42, 355–402. [Google Scholar]
- Prasad, P.D.; Li, H.W.; Fei, Y.J.; Ganapathy, M.E.; Fujita, T.; Plumley, L.H.; Yang-Feng, T.L.; Leibach, F.H.; Ganapathy, V. Exon-intron structure, analysis of promoter region, and chromosomal localization of the human type 1 sigma receptor gene. J. Neurochem. 1998, 70, 443–451. [Google Scholar] [CrossRef]
- Hanner, M.; Moebius, F.F.; Flandorfer, A.; Knaus, H.G.; Striessnig, J.; Kempner, E.; Glossmann, H. Purification, molecular cloning, and expression of the mammalian sigma1-binding site. Proc. Natl. Acad. Sci. USA 1996, 93, 8072–8077. [Google Scholar] [CrossRef] [Green Version]
- Kekuda, R.; Prasad, P.D.; Fei, Y.J.; Leibach, F.H.; Ganapathy, V. Cloning and functional expression of the human type 1 sigma receptor (hSigmaR1). Biochem. Biophys. Res. Commun. 1996, 229, 553–558. [Google Scholar] [CrossRef]
- Seth, P.; Fei, Y.J.; Li, H.W.; Huang, W.; Leibach, F.H.; Ganapathy, V. Cloning and functional characterization of a sigma receptor from rat brain. J. Neurochem. 1998, 70, 922–931. [Google Scholar] [CrossRef] [PubMed]
- Silve, S.; Dupuy, P.H.; Labit-Lebouteiller, C.; Kaghad, M.; Chalon, P.; Rahier, A.; Taton, M.; Lupker, J.; Shire, D.; Loison, G. Emopamil-binding protein, a mammalian protein that binds a series of structurally diverse neuroprotective agents, exhibits delta8-delta7 sterol isomerase activity in yeast. J. Biol. Chem. 1996, 271, 22434–22440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moebius, F.F.; Reiter, R.J.; Hanner, M.; Glossmann, H. High affinity of sigma 1-binding sites for sterol isomerization inhibitors: Evidence for a pharmacological relationship with the yeast sterol C8-C7 isomerase. Br. J. Pharmacol. 1997, 121, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dussossoy, D.P.C.; Belugou, S.; Feraut, D.; Bord, A.; Goubet, C.; Roque, C.; Vidal, H.; Combes, T.; Loison, G.; Casellas, P. Clocalization of sterol isomerase and sigma1receptor at endoplasmicreticulum and nuclear envelope level. Eur. J. Biochem. 1999, 263, 377–386. [Google Scholar] [CrossRef]
- Senda, T.; Matsuno, K.; Mita, S. The presence of sigma receptor subtypes in bovine retinal membranes. Exp. Eye Res. 1997, 64, 857–860. [Google Scholar] [CrossRef]
- Aydar, E.; Palmer, C.P.; Klyachko, V.A.; Jackson, M.B. The sigma receptor as a ligand-regulated auxiliary potassium channel subunit. Neuron 2002, 34, 399–410. [Google Scholar] [CrossRef] [Green Version]
- Schmidt, H.R.; Zheng, S.; Gurpinar, E.; Koehl, A.; Manglik, A.; Kruse, A.C. Crystal structure of the human sigma1 receptor. Nature 2016, 532, 527–530. [Google Scholar] [CrossRef]
- Schmidt, H.R.; Betz, R.M.; Dror, R.O.; Kruse, A.C. Structural basis for sigma1 receptor ligand recognition. Nat. Struct. Mol. Biol. 2018, 25, 981–987. [Google Scholar] [CrossRef]
- Alonso, G.; Phan, V.; Guillemain, I.; Saunier, M.; Legrand, A.; Anoal, M.; Maurice, T. Immunocytochemical localization of the sigma(1) receptor in the adult rat central nervous system. Neuroscience 2000, 97, s0306–s4522. [Google Scholar] [CrossRef]
- Mavlyutov, T.A.; Duellman, T.; Kim, H.T.; Epstein, M.L.; Leese, C.; Davletov, B.A.; Yang, J. Sigma-1 receptor expression in the dorsal root ganglion: Reexamination using a highly specific antibody. Neuroscience 2016, 331, 148–157. [Google Scholar] [CrossRef] [Green Version]
- Palacios, G.; Muro, A.; Verdu, E.; Pumarola, M.; Vela, J.M. Immunohistochemical localization of the sigma1 receptor in Schwann cells of rat sciatic nerve. Brain Res. 2004, 1007, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Palacios, G.; Muro, A.; Vela, J.M.; Molina-Holgado, E.; Guitart, X.; Ovalle, S.; Zamanillo, D. Immunohistochemical localization of the sigma1-receptor in oligodendrocytes in the rat central nervous system. Brain Res. 2003, 961, s0006–s8993. [Google Scholar] [CrossRef]
- Bangaru, M.L.; Weihrauch, D.; Tang, Q.B.; Zoga, V.; Hogan, Q.; Wu, H.E. Sigma-1 receptor expression in sensory neurons and the effect of painful peripheral nerve injury. Mol. Pain 2013, 9, 47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mavlyutov, T.A.; Epstein, M.L.; Andersen, K.A.; Ziskind-Conhaim, L.; Ruoho, A.E. The sigma-1 receptor is enriched in postsynaptic sites of C-terminals in mouse motoneurons. An anatomical and behavioral study. Neuroscience 2010, 167, 247–255. [Google Scholar] [CrossRef] [Green Version]
- Bouchard, P.; Quirion, R. [3H]1,3-di(2-tolyl)guanidine and [3H](+)pentazocine binding sites in the rat brain: Autoradiographic visualization of the putative sigma1 and sigma2 receptor subtypes. Neuroscience 1997, 76, 467–477. [Google Scholar] [CrossRef]
- Stracina, T.; Novakova, M. Cardiac sigma receptors—An update. Physiol. Res. 2018, 67 (Suppl. 4), S561–S576. [Google Scholar] [CrossRef]
- Bowen, W.D. Sigma receptors: Recent advances and new clinical potentials. Pharm. Acta Helv. 2000, 74, 211–218. [Google Scholar] [CrossRef]
- Su, T.P.; Hayashi, T.; Maurice, T.; Buch, S.; Ruoho, A.E. The sigma-1 receptor chaperone as an inter-organelle signaling modulator. Trends Pharmacol. Sci. 2010, 31, 557–566. [Google Scholar] [CrossRef] [Green Version]
- Hayashi, T.; Su, T.P. Sigma-1 receptor chaperones at the ER-mitochondrion interface regulate Ca(2+) signaling and cell survival. Cell 2007, 131, 596–610. [Google Scholar] [CrossRef] [Green Version]
- Kourrich, S.; Hayashi, T.; Chuang, J.Y.; Tsai, S.Y.; Su, T.P.; Bonci, A. Dynamic Interaction between Sigma-1 Receptor and Kv1.2 Shapes Neuronal and Behavioral Responses to Cocaine. Cell 2013, 152, 236–247. [Google Scholar] [CrossRef] [Green Version]
- Tsai, S.Y.A.; Pokrass, M.J.; Klauer, N.R.; Nohara, H.; Su, T.P. Sigma-1 receptor regulates Tau phosphorylation and axon extension by shaping p35 turnover via myristic acid. Proc. Natl. Acad. Sci. USA 2015, 112, 6742–6747. [Google Scholar] [CrossRef] [Green Version]
- Seung, H.S.; Sumbul, U. Neuronal cell types and connectivity: Lessons from the retina. Neuron 2014, 83, 1262–1272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goldman, D. Muller glial cell reprogramming and retina regeneration. Nat. Rev. Neurosci. 2014, 15, 431–442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mavlyutov, T.A.; Epstein, M.; Guo, L.W. Subcellular localization of the sigma-1 receptor in retinal neurons—An electron microscopy study. Sci. Rep. 2015, 5, 10689. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Zhao, J.; Cui, X.; Mysona, B.A.; Navneet, S.; Saul, A.; Ahuja, M.; Lambert, N.; Gazaryan, I.G.; Thomas, B.; et al. The molecular chaperone sigma 1 receptor mediates rescue of retinal cone photoreceptor cells via modulation of NRF2. Free Radic. Biol. Med. 2019, 134, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Shimazawa, M.; Sugitani, S.; Inoue, Y.; Tsuruma, K.; Hara, H. Effect of a sigma-1 receptor agonist, cutamesine dihydrochloride (SA4503), on photoreceptor cell death against light-induced damage. Exp. Eye Res. 2015, 132, 64–72. [Google Scholar] [CrossRef]
- Barwick, S.R.; Siddiq, M.S.; Wang, J.; Xiao, H.; Marshall, B.; Perry, E.; Smith, S.B. Sigma 1 Receptor Co-Localizes with NRF2 in Retinal Photoreceptor Cells. Antioxidants 2021, 10, 981. [Google Scholar] [CrossRef]
- Ha, Y.; Saul, A.; Tawfik, A.; Williams, C.; Bollinger, K.; Smith, R.; Tachikawa, M.; Zorrilla, E.; Ganapathy, V.; Smith, S.B. Late-onset inner retinal dysfunction in mice lacking sigma receptor 1 (sigmaR1). Investig. Ophthalmol. Vis. Sci. 2011, 52, 7749–7760. [Google Scholar] [CrossRef]
- Mavlyutov, T.A.; Nickells, R.W.; Guo, L.W. Accelerated retinal ganglion cell death in mice deficient in the Sigma-1 receptor. Mol. Vis. 2011, 17, 1034–1043. [Google Scholar]
- Ola, M.S.; Moore, P.; El-Sherbeny, A.; Roon, P.; Agarwal, N.; Sarthy, V.P.; Casellas, P.; Ganapathy, V.; Smith, S.B. Expression pattern of sigma receptor 1 mRNA and protein in mammalian retina. Brain Res. Mol. Brain Res. 2001, 95, 86–95. [Google Scholar] [CrossRef] [Green Version]
- Yang, H.; Fu, Y.; Liu, X.; Shahi, P.K.; Mavlyutov, T.A.; Li, J.; Yao, A.; Guo, S.Z.; Pattnaik, B.R.; Guo, L.W. Role of the sigma-1 receptor chaperone in rod and cone photoreceptor degenerations in a mouse model of retinitis pigmentosa. Mol. Neurodegener. 2017, 12, 68. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.Y.; Cringle, S.J.; Balaratnasingam, C.; Morgan, W.H.; Yu, P.K.; Su, E.N. Retinal ganglion cells: Energetics, compartmentation, axonal transport, cytoskeletons and vulnerability. Prog. Retin. Eye Res. 2013, 36, 217–246. [Google Scholar] [CrossRef] [PubMed]
- Martin, P.M.; Ola, M.S.; Agarwal, N.; Ganapathy, V.; Smith, S.B. The sigma receptor ligand (+)-pentazocine prevents apoptotic retinal ganglion cell death induced in vitro by homocysteine and glutamate. Brain Res. Mol. Brain Res. 2004, 123, 66–75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mueller, B.H.; Park, Y.; Ma, H.Y.; Dibas, A.; Ellis, D.Z.; Clark, A.F.; Yorio, T. Sigma-1 receptor stimulation protects retinal ganglion cells from ischemia-like insult through the activation of extracellular-signal-regulated kinases 1/2. Exp. Eye Res. 2014, 128, 156–169. [Google Scholar] [CrossRef]
- Dun, Y.; Thangaraju, M.; Prasad, P.; Ganapathy, V.; Smith, S.B. Prevention of excitotoxicity in primary retinal ganglion cells by (+)-pentazocine, a sigma receptor-1 specific ligand. Investig. Ophthalmol. Vis. Sci. 2007, 48, 4785–4794. [Google Scholar] [CrossRef]
- Larsen, M.H.; Hay-Schmidt, A.; Ronn, L.C.; Mikkelsen, J.D. Temporal expression of brain-derived neurotrophic factor (BDNF) mRNA in the rat hippocampus after treatment with selective and mixed monoaminergic antidepressants. Eur. J. Pharmacol. 2008, 578, 114–122. [Google Scholar] [CrossRef]
- Ola, M.S.; Moore, P.; Maddox, D.; El-Sherbeny, A.; Huang, W.; Roon, P.; Agarwal, N.; Ganapathy, V.; Smith, S.B. Analysis of sigma receptor (sigmaR1) expression in retinal ganglion cells cultured under hyperglycemic conditions and in diabetic mice. Brain Res. Mol. Brain Res. 2002, 107, 97–107. [Google Scholar] [CrossRef]
- Smith, S.B.; Duplantier, J.; Dun, Y.; Mysona, B.; Roon, P.; Martin, P.M.; Ganapathy, V. In vivo protection against retinal neurodegeneration by sigma receptor 1 ligand (+)-pentazocine. Investig. Ophthalmol. Vis. Sci. 2008, 49, 4154–4161. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Cui, X.; Roon, P.; Smith, S.B. Role of Sigma 1 Receptor in Retinal Degeneration of the Ins2Akita/+ Murine Model of Diabetic Retinopathy. Investig. Ophthalmol. Vis. Sci. 2016, 57, 2770–2781. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.L.; Wang, L.; Zhong, Y.M.; Yang, X.L. Expression of sigma receptor 1 mRNA and protein in rat retina. Neuroscience 2010, 167, 1151–1159. [Google Scholar] [CrossRef]
- Jiang, G.; Mysona, B.; Dun, Y.; Gnana-Prakasam, J.P.; Pabla, N.; Li, W.; Dong, Z.; Ganapathy, V.; Smith, S.B. Expression, Subcellular Localization, and Regulation of Sigma Receptor in Retinal Müller Cells. Investig. Ophthalmol. Vis. Sci. 2006, 47, 5576. [Google Scholar] [CrossRef] [PubMed]
- Ha, Y.; Shanmugam, A.K.; Markand, S.; Zorrilla, E.; Ganapathy, V.; Smith, S.B. Sigma receptor 1 modulates ER stress and Bcl2 in murine retina. Cell Tissue Res. 2014, 356, 15–27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jadzinsky, P.D.; Baccus, S.A. Transformation of visual signals by inhibitory interneurons in retinal circuits. Annu. Rev. Neurosci. 2013, 36, 403–428. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Mysona, B.A.; Wang, J.; Gonsalvez, G.B.; Smith, S.B.; Bollinger, K.E. Sigma 1 receptor regulates ERK activation and promotes survival of optic nerve head astrocytes. PLoS ONE 2017, 12, e0184421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schoenwald, R.D.; Barfknecht, C.F.; Xia, E.; Newton, R.E. The presence of sigma-receptors in the lacrimal gland. J. Ocul. Pharmacol. 1993, 9, 125–139. [Google Scholar] [CrossRef]
- Bucolo, C.; Campana, G.; Di Toro, R.; Cacciaguerra, S.; Spampinato, S. Sigma1 recognition sites in rabbit iris-ciliary body: Topical sigma1-site agonists lower intraocular pressure. J. Pharmacol. Exp. Ther. 1999, 289, 1362–1369. [Google Scholar]
- Wang, W.F.; Ishiwata, K.; Kiyosawa, M.; Kawamura, K.; Oda, K.; Kobayashi, T.; Matsuno, K.; Mochizuki, M. Visualization of sigma1 receptors in eyes by ex vivo autoradiography and in vivo positron emission tomography. Exp. Eye Res. 2002, 75, 723–730. [Google Scholar] [CrossRef]
- Wang, L.; Duncan, G. Silencing of sigma-1 receptor induces cell death in human lens cells. Exp. Cell Res. 2006, 312, 1439–1446. [Google Scholar] [CrossRef]
- Senda, T.; Mita, S.; Kaneda, K.; Kikuchi, M.; Akaike, A. Effect of SA4503, a novel sigma1 receptor agonist, against glutamate neurotoxicity in cultured rat retinal neurons. Eur. J. Pharmacol. 1998, 342, 105–111. [Google Scholar] [CrossRef]
- Tchedre, K.T.; Yorio, T. σ-1 receptors protect RGC-5 cells from apoptosis by regulating intracellular calcium, Bax levels, and caspase-3 activation. Investig. Ophthalmol. Vis. Sci. 2008, 49, 2577–2588. [Google Scholar] [CrossRef] [Green Version]
- Mueller, B.; Park, Y.; Daudt, D.; Ma, H.; Akopova, I.; Stankowska, D.; Clark, A.; Yorio, T. Sigma-1 receptor stimulation attenuates calcium influx through activated L-type Voltage Gated Calcium Channels in purified retinal ganglion cells. Exp. Eye Res. 2013, 107, 21–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.J.; Liu, L.L.; Wu, Y.; Jiang, S.X.; Zhong, Y.M.; Yang, X.L. Sigma receptor 1 is preferentially involved in modulation of N-methyl-D-aspartate receptor-mediated light-evoked excitatory postsynaptic currents in rat retinal ganglion cells. Neurosignals 2011, 19, 110–116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.J.; Liu, L.L.; Jiang, S.X.; Zhong, Y.M.; Yang, X.L. Activation of the sigma receptor 1 suppresses NMDA responses in rat retinal ganglion cells. Neuroscience 2011, 177, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.L.; Deng, Q.Q.; Weng, S.J.; Yang, X.L.; Zhong, Y.M. Activation of the sigma receptor 1 modulates AMPA receptor-mediated light-evoked excitatory postsynaptic currents in rat retinal ganglion cells. Neuroscience 2016, 332, 53–60. [Google Scholar] [CrossRef]
- Vogler, S.; Winters, H.; Pannicke, T.; Wiedemann, P.; Reichenbach, A.; Bringmann, A. Sigma-1 receptor activation inhibits osmotic swelling of rat retinal glial (Muller) cells by transactivation of glutamatergic and purinergic receptors. Neurosci. Lett. 2016, 610, 13–18. [Google Scholar] [CrossRef]
- Zhao, J.; Ha, Y.; Liou, G.I.; Gonsalvez, G.B.; Smith, S.B.; Bollinger, K.E. Sigma Receptor Ligand, (+)-Pentazocine, Suppresses Inflammatory Responses of Retinal Microglia. Investig. Ophthalmol. Vis. Sci. 2014, 55, 3375–3384. [Google Scholar] [CrossRef]
- Zhao, J.; Mysona, B.A.; Qureshi, A.; Kim, L.; Fields, T.; Gonsalvez, G.B.; Smith, S.B.; Bollinger, K.E. (+)-Pentazocine Reduces NMDA-Induced Murine Retinal Ganglion Cell Death Through a sigma R1-Dependent Mechanism. Investig. Ophthalmol. Vis. Sci. 2016, 57, 453–461. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Smith, S.B. A Novel mechanism of sigma 1 receptor neuroprotection: Modulation of miR-214-3p. In Retinal Degenerative Diseases: Mechanisms and Experimental Therapy; Rickman, C.B., Grimm, C., Anderson, R.E., Ash, J.D., LaVail, M.M., Hollyfield, J.G., Eds.; Springer International Publishing Ag: Cham, Switzerland, 2019; Volume 1185, pp. 463–467. 3p. [Google Scholar] [CrossRef]
- Wang, J.; Shanmugam, A.; Markand, S.; Zorrilla, E.; Ganapathy, V.; Smith, S.B. Sigma 1 receptor regulates the oxidative stress response in primary retinal Muller glial cells via NRF2 signaling and system xc(-), the Na(+)-independent glutamate-cystine exchanger. Free Radic. Biol. Med. 2015, 86, 25–36. [Google Scholar] [CrossRef] [Green Version]
- Cuadrado, A.; Rojo, A.I.; Wells, G.; Hayes, J.D.; Cousin, S.P.; Rumsey, W.L.; Attucks, O.C.; Franklin, S.; Levonen, A.L.; Kensler, T.W.; et al. Therapeutic targeting of the NRF2 and KEAP1 partnership in chronic diseases. Nat. Rev. Drug Discov. 2019, 18, 295–317. [Google Scholar] [CrossRef] [Green Version]
- Buendia, I.; Michalska, P.; Navarro, E.; Gameiro, I.; Egea, J.; Leon, R. Nrf2-ARE pathway: An emerging target against oxidative stress and neuroinflammation in neurodegenerative diseases. Pharmacol. Ther. 2016, 157, 84–104. [Google Scholar] [CrossRef]
- Nakagami, Y. Nrf2 Is an Attractive Therapeutic Target for Retinal Diseases. Oxidative Med. Cell. Longev. 2016, 2016, 7469326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Navneet, S.; Cui, X.; Zhao, J.; Wang, J.; Kaidery, N.A.; Thomas, B.; Bollinger, K.E.; Yoon, Y.; Smith, S.B. Excess homocysteine upregulates the NRF2-antioxidant pathway in retinal Muller glial cells. Exp. Eye Res. 2019, 178, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Navneet, S.; Zhao, J.; Wang, J.; Mysona, B.; Barwick, S.; Ammal Kaidery, N.; Saul, A.; Kaddour-Djebbar, I.; Bollag, W.B.; Thomas, B.; et al. Hyperhomocysteinemia-induced death of retinal ganglion cells: The role of Muller glial cells and NRF2. Redox Biol. 2019, 24, 101199. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xiao, H.; Barwick, S.R.; Smith, S.B. Comparison of Sigma 1 Receptor Ligands SA4503 and PRE084 to (+)-Pentazocine in the rd10 Mouse Model of RP. Investig. Ophthalmol. Vis. Sci. 2020, 61, 3. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.Y.; Rothman, R.K.; Su, T.P. Insights into the Sigma-1 receptor chaperone’s cellular functions: A microarray report. Synapse 2012, 66, 42–51. [Google Scholar] [CrossRef] [Green Version]
- Pal, A.; Fontanilla, D.; Gopalakrishnan, A.; Chae, Y.K.; Markley, J.L.; Ruoho, A.E. The sigma-1 receptor protects against cellular oxidative stress and activates antioxidant response elements. Eur. J. Pharmacol. 2012, 682, 12–20. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Zhao, C.S. Sigma-1 receptor activation ameliorates LPS-induced NO production and ROS formation through the Nrf2/HO-1 signaling pathway in cultured astrocytes. Neurosci. Lett. 2019, 711, 134387. [Google Scholar] [CrossRef]
- Wang, J.; Saul, A.; Cui, X.; Roon, P.; Smith, S.B. Absence of Sigma 1 Receptor Accelerates Photoreceptor Cell Death in a Murine Model of Retinitis Pigmentosa. Investig. Ophthalmol. Vis. Sci. 2017, 58, 4545–4558. [Google Scholar] [CrossRef] [Green Version]
- Weng, T.Y.; Hung, D.T.; Su, T.P.; Tsai, S.A. Loss of Sigma-1 Receptor Chaperone Promotes Astrocytosis and Enhances the Nrf2 Antioxidant Defense. Oxidative Med. Cell. Longev. 2017, 2017, 4582135. [Google Scholar] [CrossRef] [Green Version]
- Xiao, H.; Wang, J.; Saul, A.; Smith, S.B. Comparison of Neuroprotective Effects of Monomethylfumarate to the Sigma 1 Receptor Ligand (+)-Pentazocine in a Murine Model of Retinitis Pigmentosa. Investig. Ophthalmol. Vis. Sci. 2020, 61, 5. [Google Scholar] [CrossRef] [Green Version]
- Johnson, G.L.; Lapadat, R. Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 2002, 298, 1911–1912. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Widmann, C.; Gibson, S.; Jarpe, M.; Johnson, G. Mitogen-activated protein kinase: Conservation of a three-kinase module from yeast to human. Physiol. Rev. 1999, 79, 143–180. [Google Scholar] [CrossRef] [PubMed]
- Wortzel, I.; Seger, R. The ERK Cascade: Distinct Functions within Various Subcellular Organelles. Genes Cancer 2011, 2, 195–209. [Google Scholar] [CrossRef] [PubMed]
- Meng, B.; Li, H.; Sun, X.; Qu, W.; Yang, B.; Cheng, F.; Shi, L.; Yuan, H. Sigma-1 receptor stimulation protects against pressure-induced damage through InsR-MAPK signaling in human trabecular meshwork cells. Mol. Med. Rep. 2017, 16, 617–624. [Google Scholar] [CrossRef] [Green Version]
- Moriguchi, S.; Yamamoto, Y.; Ikuno, T.; Fukunaga, K. Sigma-1 receptor stimulation by dehydroepiandrosterone ameliorates cognitive impairment through activation of CaM kinase II, protein kinase C and extracellular signal-regulated kinase in olfactory bulbectomized mice. J. Neurochem. 2011, 117, 879–891. [Google Scholar] [CrossRef]
- Yabuki, Y.; Shinoda, Y.; Izumi, H.; Ikuno, T.; Shioda, N.; Fukunaga, K. Dehydroepiandrosterone administration improves memory deficits following transient brain ischemia through sigma-1 receptor stimulation. Brain Res. 2015, 1622, 102–113. [Google Scholar] [CrossRef]
- Yang, R.; Chen, L.; Wang, H.F.; Xu, B.Z.; Tomimoto, H.; Chen, L. Anti-amnesic effect of neurosteroid PREGS in A beta(25-35)-injected mice through sigma(1) receptor- and alpha 7nAChR-mediated neuroprotection. Neuropharmacology 2012, 63, 1042–1050. [Google Scholar] [CrossRef]
- Cantarella, G.; Bucolo, C.; Di Benedetto, G.; Pezzino, S.; Lempereur, L.; Calvagna, R.; Clementi, S.; Pavone, P.; Fiore, L.; Bernardini, R. Protective effects of the sigma agonist Pre-084 in the rat retina. Br. J. Ophthalmol. 2007, 91, 1382–1384. [Google Scholar] [CrossRef] [Green Version]
- Yagasaki, Y.; Numakawa, T.; Kumamaru, E.; Hayashi, T.; Su, T.P.; Kunugi, H. Chronic antidepressants potentiate via sigma-1 receptors the brain-derived neurotrophic factor-induced signaling for glutamate release. J. Biol. Chem. 2006, 281, 12941–12949. [Google Scholar] [CrossRef] [Green Version]
- Mysona, B.A.; Zhao, J.; Smith, S.; Bollinger, K.E. Relationship between Sigma-1 receptor and BDNF in the visual system. Exp. Eye Res. 2018, 167, 25–30. [Google Scholar] [CrossRef]
- Zhao, L.; Chen, G.; Li, J.; Fu, Y.; Mavlyutov, T.A.; Yao, A.; Nickells, R.W.; Gong, S.; Guo, L.W. An intraocular drug delivery system using targeted nanocarriers attenuates retinal ganglion cell degeneration. J. Control. Release 2017, 247, 153–166. [Google Scholar] [CrossRef] [Green Version]
- Bucolo, C.; Drago, F. Neuroactive steroids protect retinal tissue through sigma1 receptors. Basic Clin. Pharmacol. Toxicol. 2007, 100, 214–216. [Google Scholar] [CrossRef]
- Bucolo, C.D.; Drago, F. Effects of neurosteroids on ischemia-reperfusion injury in the rat retina: Role of sigma1 recognition sites. Eur. J. Pharmacol. 2004, 498, 111–114. [Google Scholar] [CrossRef]
- Wang, J.; Saul, A.; Roon, P.; Smith, S.B. Activation of the molecular chaperone, sigma 1 receptor, preserves cone function in a murine model of inherited retinal degeneration. Proc. Natl. Acad. Sci. USA 2016, 113, E3764–E3772. [Google Scholar] [CrossRef] [Green Version]
- Sun, X.; Cheng, F.; Meng, B.; Yang, B.; Song, W.; Yuan, H. Pregnenolone sulfate decreases intraocular pressure and changes expression of sigma receptor in a model of chronic ocular hypertension. Mol. Biol. Rep. 2012, 39, 6607–6614. [Google Scholar] [CrossRef]
- Kumar, B.; Gupta, S.K.; Srinivasan, B.P.; Nag, T.C.; Srivastava, S.; Saxena, R.; Jha, K.A. Hesperetin rescues retinal oxidative stress, neuroinflammation and apoptosis in diabetic rats. Microvasc. Res. 2013, 87, 65–74. [Google Scholar] [CrossRef]
- Bucolo, C.; Marrazzo, A.; Ronsisvalle, S.; Ronsisvalle, G.; Cuzzocrea, S.; Mazzon, E.; Caputi, A.; Drago, F. A novel adamantane derivative attenuates retinal ischemia-reperfusion damage in the rat retina through sigma1 receptors. Eur. J. Pharmacol. 2006, 536, 200–203. [Google Scholar] [CrossRef]
- Mysona, B.; Kansara, N.; Zhao, J.; Bollinger, K. The Role of Sigma 1 Receptor as a Neuroprotective Target in Glaucoma. Adv. Exp. Med. Biol. 2017, 964, 299–307. [Google Scholar] [CrossRef]
- Ellis, D.; Li, L.; Park, Y.; He, S.; Mueller, B.; Yorio, T. Sigma-1 Receptor Regulates Mitochondrial Function in Glucose- and Oxygen-Deprived Retinal Ganglion Cells. Investig. Ophthalmol. Vis. Sci. 2017, 58, 2755–2764. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Xiao, H.; Barwick, S.; Liu, Y.; Smith, S.B. Optimal timing for activation of sigma 1 receptor in the Pde6b(rd10)/J (rd10) mouse model of retinitis pigmentosa. Exp. Eye Res. 2021, 202, 108397. [Google Scholar] [CrossRef]
- Domènech, E.B.; Marfany, G. The Relevance of Oxidative Stress in the Pathogenesis and Therapy of Retinal Dystrophies. Antioxidants 2020, 9, 347. [Google Scholar] [CrossRef] [Green Version]
- Ozawa, Y. Oxidative stress in the light-exposed retina and its implication in age-related macular degeneration. Redox Biol. 2020, 37, 101779. [Google Scholar] [CrossRef] [PubMed]
- Frey, T.; Antonetti, D.A. Alterations to the blood-retinal barrier in diabetes: Cytokines and reactive oxygen species. Antioxid. Redox Signal. 2011, 15, 1271–1284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cuenca, N.; Fernandez-Sanchez, L.; Campello, L.; Maneu, V.; De la Villa, P.; Lax, P.; Pinilla, I. Cellular responses following retinal injuries and therapeutic approaches for neurodegenerative diseases. Prog. Retin. Eye Res. 2014, 43, 17–75. [Google Scholar] [CrossRef]
- Kourrich, S.; Su, T.P.; Fujimoto, M.; Bonci, A. The sigma-1 receptor: Roles in neuronal plasticity and disease. Trends Neurosci. 2012, 35, 762–771. [Google Scholar] [CrossRef] [Green Version]
- Hayashi, T. The Sigma-1 Receptor in Cellular Stress Signaling. Front. Neurosci. 2019, 13, 733. [Google Scholar] [CrossRef] [Green Version]
- Ha, Y.; Dun, Y.; Thangaraju, M.; Duplantier, J.; Dong, Z.; Liu, K.; Ganapathy, V.; Smith, S.B. Sigma Receptor 1 Modulates Endoplasmic Reticulum Stress in Retinal Neurons. Investig. Ophthalmol. Vis. Sci. 2011, 52, 527–540. [Google Scholar] [CrossRef] [Green Version]
- Christ, M.G.; Huesmann, H.; Nagel, H.; Kern, A.; Behl, C. Sigma-1 Receptor Activation Induces Autophagy and Increases Proteostasis Capacity In Vitro and In Vivo. Cells 2019, 8, 211. [Google Scholar] [CrossRef] [Green Version]
- Boya, P.; Esteban-Martínez, L.; Serrano-Puebla, A.; Gómez-Sintes, R.; Villarejo-Zori, B. Autophagy in the eye: Development, degeneration, and aging. Prog. Retin. Eye Res. 2016, 55, 206–245. [Google Scholar] [CrossRef]
- Christ, M.G.; Clement, A.M.; Behl, C. The Sigma-1 Receptor at the Crossroad of Proteostasis, Neurodegeneration, and Autophagy. Trends Neurosci. 2020, 43, 79–81. [Google Scholar] [CrossRef]
- MacVicar, T.; Mannack, L.; Lees, R.; Lane, J. Targeted siRNA Screens Identify ER-to-Mitochondrial Calcium Exchange in Autophagy and Mitophagy Responses in RPE1 Cells. Int. J. Mol. Sci. 2015, 16, 13356–13380. [Google Scholar] [CrossRef] [Green Version]
- Maher, C.M.; Thomas, J.D.; Haas, D.A.; Longen, C.G.; Oyer, H.M.; Tong, J.Y.; Kim, F.J. Small-Molecule Sigma1 Modulator Induces Autophagic Degradation of PD-L1. Mol. Cancer Res. 2018, 16, 243–255. [Google Scholar] [CrossRef] [Green Version]
- Schrock, J.M.; Spino, C.M.; Longen, C.G.; Stabler, S.M.; Marino, J.C.; Pasternak, G.W.; Kim, F.J. Sequential cytoprotective responses to Sigma1 ligand-induced endoplasmic reticulum stress. Mol. Pharmacol. 2013, 84, 751–762. [Google Scholar] [CrossRef] [Green Version]
- Vollrath, J.T.; Sechi, A.; Dreser, A.; Katona, I.; Wiemuth, D.; Vervoorts, J.; Dohmen, M.; Chandrasekar, A.; Prause, J.; Brauers, E.; et al. Loss of function of the ALS protein SigR1 leads to ER pathology associated with defective autophagy and lipid raft disturbances. Cell Death Dis. 2014, 5, e1290. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Zhang, X.; Wei, Q.; Leng, S.; Li, C.; Han, B.; Bai, Y.; Zhang, H.; Yao, H. Activation of Sigma-1 Receptor Enhanced Pericyte Survival via the Interplay Between Apoptosis and Autophagy: Implications for Blood-Brain Barrier Integrity in Stroke. Transl. Stroke Res. 2020, 11, 267–287. [Google Scholar] [CrossRef]
- Lee, J.; Giordano, S.; Zhang, J. Autophagy, mitochondria and oxidative stress: Cross-talk and redox signalling. Biochem. J. 2012, 441, 523–540. [Google Scholar] [CrossRef] [Green Version]
- Youle, R.J.; Narendra, D.P. Mechanisms of mitophagy. Nat. Rev. Mol. Cell Biol. 2011, 12, 9–14. [Google Scholar] [CrossRef]
- Fukunaga, K.; Shinoda, Y.; Tagashira, H. The role of SIGMAR1 gene mutation and mitochondrial dysfunction in amyotrophic lateral sclerosis. J. Pharmacol. Sci. 2015, 127, 36–41. [Google Scholar] [CrossRef] [Green Version]
- Brimson, J.M.; Prasanth, M.I.; Malar, D.S.; Brimson, S.; Thitilertdecha, P.; Tencomnao, T. Drugs that offer the potential to reduce hospitalization and mortality from SARS-CoV-2 infection: The possible role of the sigma-1 receptor and autophagy. Expert Opin. Ther. Targets 2021, 25, 435–449. [Google Scholar] [CrossRef]
- Yang, H.; Shen, H.; Li, J.; Guo, L.W. SIGMAR1/Sigma-1 receptor ablation impairs autophagosome clearance. Autophagy 2019, 15, 1539–1557. [Google Scholar] [CrossRef] [Green Version]
- Danial, N.N.; Korsmeyer, S.J. Cell death: Critical control points. Cell 2004, 116, 205–219. [Google Scholar] [CrossRef] [Green Version]
- Linden, R.; Rehen, S.K.; Chiarini, L.B. Apoptosis in Developing Retinal Tissue. Prog. Retin. Eye Res. 1999, 18, 133–165. [Google Scholar] [CrossRef]
- Maes, M.E.; Schlamp, C.L.; Nickells, R.W. BAX to basics: How the BCL2 gene family controls the death of retinal ganglion cells. Prog. Retin. Eye Res. 2017, 57, 1–25. [Google Scholar] [CrossRef] [Green Version]
- Yang, S.; Bhardwaj, A.; Cheng, J.; Alkayed, N.J.; Hurn, P.D.; Kirsch, J.R. Sigma receptor agonists provide neuroprotection in vitro by preserving bcl-2. Anesth. Analg. 2007, 104, 1179–1184, tables of contents. [Google Scholar] [CrossRef] [Green Version]
- Zhan, Y.; van de Water, B.; Wang, Y.; Stevens, J.L. The roles of caspase-3 and bcl-2 in chemically-induced apoptosis but not necrosis of renal epithelial cells. Oncogene 1999, 18, 6505–6512. [Google Scholar] [CrossRef] [Green Version]
- Rathnasamy, G.; Foulds, W.S.; Ling, E.A.; Kaur, C. Retinal microglia—A key player in healthy and diseased retina. Prog. Neurobiol. 2019, 173, 18–40. [Google Scholar] [CrossRef]
- Shanmugam, A.; Wang, J.; Markand, S.; Perry, R.L.; Tawfik, A.; Zorrilla, E.; Ganapathy, V.; Smith, S.B. Sigma receptor 1 activation attenuates release of inflammatory cytokines MIP1gamma, MIP2, MIP3alpha, and IL12 (p40/p70) by retinal Muller glial cells. J. Neurochem. 2015, 132, 546–558. [Google Scholar] [CrossRef] [Green Version]
- Rothova, A.; Suttorp-van Schulten, M.S.; Treffers, W.F.; Kijlstra, A. Causes and frequency of blindness in patients with intraocular inflammatory disease. Br. J. Ophthalmol. 1996, 80, 332–336. [Google Scholar] [CrossRef] [Green Version]
- Guadagni, V.; Novelli, E.; Piano, I.; Gargini, C.; Strettoi, E. Pharmacological approaches to retinitis pigmentosa: A laboratory perspective. Prog. Retin. Eye Res. 2015, 48, 62–81. [Google Scholar] [CrossRef]
- Wang, J.; Cui, X.; Roon, P.; Saul, A.; Smith, S.B. The Role of Sigma1R in Mammalian Retina. Adv. Exp. Med. Biol. 2017, 964, 267–284. [Google Scholar] [CrossRef]
- Ha, Y.; Saul, A.; Tawfik, A.; Zorrilla, E.P.; Ganapathy, V.; Smith, S.B. Diabetes accelerates retinal ganglion cell dysfunction in mice lacking sigma receptor 1. Mol. Vis. 2012, 18, 2860–2870. [Google Scholar]
- Eddings, C.R.; Arbez, N.; Akimov, S.; Geva, M.; Hayden, M.R.; Ross, C.A. Pridopidine protects neurons from mutant-huntingtin toxicity via the sigma-1 receptor. Neurobiol. Dis. 2019, 129, 118–129. [Google Scholar] [CrossRef]
- Maurice, T.; Volle, J.N.; Strehaiano, M.; Crouzier, L.; Pereira, C.; Kaloyanov, N.; Virieux, D.; Pirat, J.L. Neuroprotection in non-transgenic and transgenic mouse models of Alzheimer’s disease by positive modulation of sigma1 receptors. Pharmacol. Res. 2019, 144, 315–330. [Google Scholar] [CrossRef]
- Mishina, M.; Ohyama, M.; Ishii, K.; Kitamura, S.; Kimura, Y.; Oda, K.; Kawamura, K.; Sasaki, T.; Kobayashi, S.; Katayama, Y.; et al. Low density of sigma1 receptors in early Alzheimer’s disease. Ann. Nucl. Med. 2008, 22, 151–156. [Google Scholar] [CrossRef]
- Uchida, N.; Ujike, H.; Tanaka, Y.; Sakai, A.; Yamamoto, M.; Fujisawa, Y.; Kanzaki, A.; Kuroda, S. A variant of the sigma receptor type-1 gene is a protective factor for Alzheimer disease. Am. J. Geriatr. Psychiatry 2005, 13, 1062–1066. [Google Scholar] [CrossRef]
- Watanabe, S.; Ilieva, H.; Tamada, H.; Nomura, H.; Komine, O.; Endo, F.; Jin, S.; Mancias, P.; Kiyama, H.; Yamanaka, K. Mitochondria-associated membrane collapse is a common pathomechanism in SIGMAR1- and SOD1-linked ALS. EMBO Mol. Med. 2016, 8, 1421–1437. [Google Scholar] [CrossRef]
- Al-Saif, A.; Al-Mohanna, F.; Bohlega, S. A mutation in sigma-1 receptor causes juvenile amyotrophic lateral sclerosis. Ann. Neurol. 2011, 70, 913–919. [Google Scholar] [CrossRef]
- Benarroch, E.E. Sigma-1 receptor and amyotrophic lateral sclerosis. Neurology 2018, 91, 743–747. [Google Scholar] [CrossRef]
- Mishina, M.; Ishiwata, K.; Ishii, K.; Kitamura, S.; Kimura, Y.; Kawamura, K.; Oda, K.; Sasaki, T.; Sakayori, O.; Hamamoto, M.; et al. Function of sigma1 receptors in Parkinson’s disease. Acta Neurol. Scand. 2005, 112, 103–107. [Google Scholar] [CrossRef]
- Hong, J.; Sha, S.; Zhou, L.; Wang, C.; Yin, J.; Chen, L. Sigma-1 receptor deficiency reduces MPTP-induced parkinsonism and death of dopaminergic neurons. Cell Death Dis. 2015, 6, e1832. [Google Scholar] [CrossRef]
- Fox, S.H.; Metman, L.V.; Nutt, J.G.; Brodsky, M.; Factor, S.A.; Lang, A.E.; Pope, L.E.; Knowles, N.; Siffert, J. Trial of dextromethorphan/quinidine to treat levodopa-induced dyskinesia in Parkinson’s disease. Mov. Disord. 2017, 32, 893–903. [Google Scholar] [CrossRef] [PubMed]
- Quadir, S.G.; Cottone, P.; Sabino, V. Role of Sigma Receptors in Alcohol Addiction. Front. Pharmacol. 2019, 10, 687. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sambo, D.O.; Lin, M.; Owens, A.; Lebowitz, J.J.; Richardson, B.; Jagnarine, D.A.; Shetty, M.; Rodriquez, M.; Alonge, T.; Ali, M.; et al. The sigma-1 receptor modulates methamphetamine dysregulation of dopamine neurotransmission. Nat. Commun. 2017, 8, 2228. [Google Scholar] [CrossRef] [PubMed]
- Sambo, D.O.; Lebowitz, J.J.; Khoshbouei, H. The sigma-1 receptor as a regulator of dopamine neurotransmission: A potential therapeutic target for methamphetamine addiction. Pharmacol. Ther. 2018, 186, 152–167. [Google Scholar] [CrossRef]
- Hedges, D.M.; Obray, J.D.; Yorgason, J.T.; Jang, E.Y.; Weerasekara, V.K.; Uys, J.D.; Bellinger, F.P.; Steffensen, S.C. Methamphetamine Induces Dopamine Release in the Nucleus Accumbens Through a Sigma Receptor-Mediated Pathway. Neuropsychopharmacology 2018, 43, 1405–1414. [Google Scholar] [CrossRef] [Green Version]
- Delint-Ramirez, I.; Garcia-Oscos, F.; Segev, A.; Kourrich, S. Cocaine engages a non-canonical, dopamine-independent, mechanism that controls neuronal excitability in the nucleus accumbens. Mol. Psychiatry 2020, 25, 680–691. [Google Scholar] [CrossRef] [Green Version]
- Narayanan, S.; Mesangeau, C.; Poupaert, J.H.; McCurdy, C.R. Sigma receptors and cocaine abuse. Curr. Top. Med. Chem. 2011, 11, 1128–1150. [Google Scholar] [CrossRef]
- Thomas, J.D.; Longen, C.G.; Oyer, H.M.; Chen, N.; Maher, C.M.; Salvino, J.M.; Kania, B.; Anderson, K.N.; Ostrander, W.F.; Knudsen, K.E.; et al. Sigma1 Targeting to Suppress Aberrant Androgen Receptor Signaling in Prostate Cancer. Cancer Res. 2017, 77, 2439–2452. [Google Scholar] [CrossRef] [Green Version]
- Xu, Q.X.; Li, E.M.; Zhang, Y.F.; Liao, L.D.; Xu, X.E.; Wu, Z.Y.; Shen, J.H.; Xu, L.Y. Overexpression of sigma1 receptor and its positive associations with pathologic TNM classification in esophageal squamous cell carcinoma. J. Histochem. Cytochem. 2012, 60, 457–466. [Google Scholar] [CrossRef] [Green Version]
- Xu, D.; Yi, W.; Chen, Y.; Ma, L.; Wang, J.; Yu, G. Overexpression of Sig1R is closely associated with tumor progression and poor outcome in patients with hilar cholangiocarcinoma. Med. Oncol. 2014, 31, 261. [Google Scholar] [CrossRef]
- Maurice, T.; Su, T.P. The pharmacology of sigma-1 receptors. Pharmacol. Ther. 2009, 124, 195–206. [Google Scholar] [CrossRef] [Green Version]
- Carcole, M.; Kummer, S.; Goncalves, L.; Zamanillo, D.; Merlos, M.; Dickenson, A.H.; Fernandez-Pastor, B.; Cabanero, D.; Maldonado, R. Sigma-1 receptor modulates neuroinflammation associated with mechanical hypersensitivity and opioid tolerance in a mouse model of osteoarthritis pain. Br. J. Pharmacol. 2019, 176, 3939–3955. [Google Scholar] [CrossRef] [PubMed]
- Montilla-Garcia, A.; Tejada, M.A.; Ruiz-Cantero, M.C.; Bravo-Caparros, I.; Yeste, S.; Zamanillo, D.; Cobos, E.J. Modulation by Sigma-1 Receptor of Morphine Analgesia and Tolerance: Nociceptive Pain, Tactile Allodynia and Grip Strength Deficits During Joint Inflammation. Front. Pharmacol. 2019, 10, 136. [Google Scholar] [CrossRef] [PubMed]
- Cirino, T.J.; Eans, S.O.; Medina, J.M.; Wilson, L.L.; Mottinelli, M.; Intagliata, S.; McCurdy, C.R.; McLaughlin, J.P. Characterization of Sigma 1 Receptor Antagonist CM-304 and Its Analog, AZ-66: Novel Therapeutics Against Allodynia and Induced Pain. Front. Pharmacol. 2019, 10, 678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dorsey, S.G. Selective Blockade of the Sigma 1 Receptor Has Beneficial Effects on Both Acute and Chronic Oxaliplatin-Induced Peripheral Neuropathy. Neurotherapeutics 2018, 15, 176–177. [Google Scholar] [CrossRef] [Green Version]
- Bhuiyan, M.S.; Tagashira, H.; Fukunaga, K. Sigma-1 receptor stimulation with fluvoxamine activates Akt-eNOS signaling in the thoracic aorta of ovariectomized rats with abdominal aortic banding. Eur. J. Pharmacol. 2011, 650, 621–628. [Google Scholar] [CrossRef]
- Tagashira, H.; Bhuiyan, S.; Shioda, N.; Fukunaga, K. Distinct cardioprotective effects of 17beta-estradiol and dehydroepiandrosterone on pressure overload-induced hypertrophy in ovariectomized female rats. Menopause 2011, 18, 1317–1326. [Google Scholar] [CrossRef]
- Bhuiyan, M.S.; Tagashira, H.; Shioda, N.; Fukunaga, K. Targeting sigma-1 receptor with fluvoxamine ameliorates pressure-overload-induced hypertrophy and dysfunctions. Expert Opin. Ther. Targets 2010, 14, 1009–1022. [Google Scholar] [CrossRef]
- Syc-Mazurek, S.B.; Libby, R.T. Axon injury signaling and compartmentalized injury response in glaucoma. Prog. Retin. Eye Res. 2019, 73, 100769. [Google Scholar] [CrossRef]
- Li, L.; He, S.; Liu, Y.; Yorio, T.; Ellis, D.Z. Sigma-1R Protects Retinal Ganglion Cells in Optic Nerve Crush Model for Glaucoma. Investig. Ophthalmol. Vis. Sci. 2021, 62, 17. [Google Scholar] [CrossRef]
- Antonetti, D.A.; Silva, P.S.; Stitt, A.W. Current understanding of the molecular and cellular pathology of diabetic retinopathy. Nat. Rev. Endocrinol. 2021, 17, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Ryskamp, D.A.; Korban, S.; Zhemkov, V.; Kraskovskaya, N.; Bezprozvanny, I. Neuronal Sigma-1 Receptors: Signaling Functions and Protective Roles in Neurodegenerative Diseases. Front. Neurosci. 2019, 13, 862. [Google Scholar] [CrossRef] [PubMed]
- Paniagua, N.; Goicoechea, C.; Abalo, R.; Lopez-Miranda, V.; Vela, J.M.; Merlos, M.; Martin Fontelles, M.I.; Giron, R. May a sigma-1 antagonist improve neuropathic signs induced by cisplatin and vincristine in rats? Eur. J. Pain 2019, 23, 603–620. [Google Scholar] [CrossRef]
- Bruna, J.; Videla, S.; Argyriou, A.A.; Velasco, R.; Villoria, J.; Santos, C.; Nadal, C.; Cavaletti, G.; Alberti, P.; Briani, C.; et al. Efficacy of a Novel Sigma-1 Receptor Antagonist for Oxaliplatin-Induced Neuropathy: A Randomized, Double-Blind, Placebo-Controlled Phase IIa Clinical Trial. Neurotherapeutics 2018, 15, 178–189. [Google Scholar] [CrossRef]
- Aydar, E.; Onganer, P.; Perrett, R.; Djamgoz, M.B.; Palmer, C.P. The expression and functional characterization of sigma (sigma) 1 receptors in breast cancer cell lines. Cancer Lett. 2006, 242, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Kim, F.J.; Maher, C.M. Sigma1 Pharmacology in the Context of Cancer. Handb. Exp. Pharmacol. 2017, 244, 237–308. [Google Scholar] [CrossRef] [PubMed]




| Ligand | Activity | In Virto/In Vivo | Function | Reference |
|---|---|---|---|---|
| (+)-PTZ | Agonist | rMC-1, RGC |
| [38,48,49,54,81,95,100,101] |
| SA4503 | Agonist | 661W, Retina |
| [36,57,59,75] |
| Neuroactive steroids | Agonist | Retina |
| [54,93,94,95] |
| PRE-084 | Agonist | 661W, Retina |
| [75,89,94] |
| (+) SKF10047 | Agonist | RGC-5, Retina |
| [60,62,63,64] |
| (−)-MR22 | Agonist | Retina |
| [98] |
| BD-1047 | Antagonist | RGC |
| [36,89] |
| NE-100 | Antagonist | APRE-19 |
| [98] |
| BD-1063 | Antagonist | Microglia |
| [67] |
| Disorders | Study Model | Reference |
|---|---|---|
| Retinal degenerative diseases | ||
| Glaucoma | Cultured primary retinal ganglion cell SigR−/− mice Ischemia-reperfusion injury animal NMDA-induced animal model | [38,39,43,44,45,52,54,61,67,91,92,94,96,99,100] |
| Retinitis pigmentosa | 661W cell line, Wild type mice, Rd10 mice, Rd10/SigR−/− mice | [35,36,37,41,68,75,81,96,101,130,131] |
| Diabetic retinopathy | Cultured primary retinal ganglion cell Ins2Akita/+, Ins2Akita/+ /Sig1R−/− and streptozotocin induced Diabetic retinopathy mice model | [38,39,47,48,108,132] |
| Neurodegenerative diseases | ||
| Huntington disease | Mutant Huntingtin transfected mice, SigR−/− mice | [133] |
| Alzheimer’s disease | Mouse model of Alzheimer’s disease AD patients | [134,135,136] |
| Amyotrophic lateral sclerosis | Mouse model of ALS disease Motor neuron-like cell line model (NSC34) | [137,138,139] |
| Parkinson’s disease | Parkinson’s disease patients MPTP-SigR−/− mice and MPTP-WT mice | [140,141,142] |
| Addiction | ||
| Alcohol addiction | WT and Sig-1R knockout mice | [143] |
| Methamphetamine addiction | WT and Sig-1R knockout mice | [144,145,146] |
| Cocaine addiction | HEK293T cell and WT mice | [147,148] |
| Cancer | ||
| Prostate cancer | LNCaP, VCap,22RV1, PC3, C4-2 and LAPC4 | [149] |
| Esophageal squamous cell carcinoma | Human ESCC cell lines KYSE180, KYSE150, and EC109 | [150] |
| Hilar cholangiocarcinoma | Tissues from hilar cholangiocarcinoma patients | [151] |
| Others | ||
| Peripheral neuropathic pain | Mouse model of osteoarthritis pain CFA-induced inflammatory model Chronic nerve constriction injury (CCI)model Acute and chronic oxaliplatin-induced peripheral pain | [152,153,154,155,156] |
| Cardiovascular disease | Ovariectomized female rats | [157,158,159] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, Z.; Lei, Y.; Qin, H.; Zhang, S.; Li, P.; Yao, K. Sigma-1 Receptor in Retina: Neuroprotective Effects and Potential Mechanisms. Int. J. Mol. Sci. 2022, 23, 7572. https://doi.org/10.3390/ijms23147572
Xu Z, Lei Y, Qin H, Zhang S, Li P, Yao K. Sigma-1 Receptor in Retina: Neuroprotective Effects and Potential Mechanisms. International Journal of Molecular Sciences. 2022; 23(14):7572. https://doi.org/10.3390/ijms23147572
Chicago/Turabian StyleXu, Zifen, Yushuang Lei, Huan Qin, Shiyao Zhang, Ping Li, and Kai Yao. 2022. "Sigma-1 Receptor in Retina: Neuroprotective Effects and Potential Mechanisms" International Journal of Molecular Sciences 23, no. 14: 7572. https://doi.org/10.3390/ijms23147572






