Sustained Increase in Serum Glial Fibrillary Acidic Protein after First ST-Elevation Myocardial Infarction
Abstract
:1. Introduction
2. Results
2.1. Characteristics of Included Patients
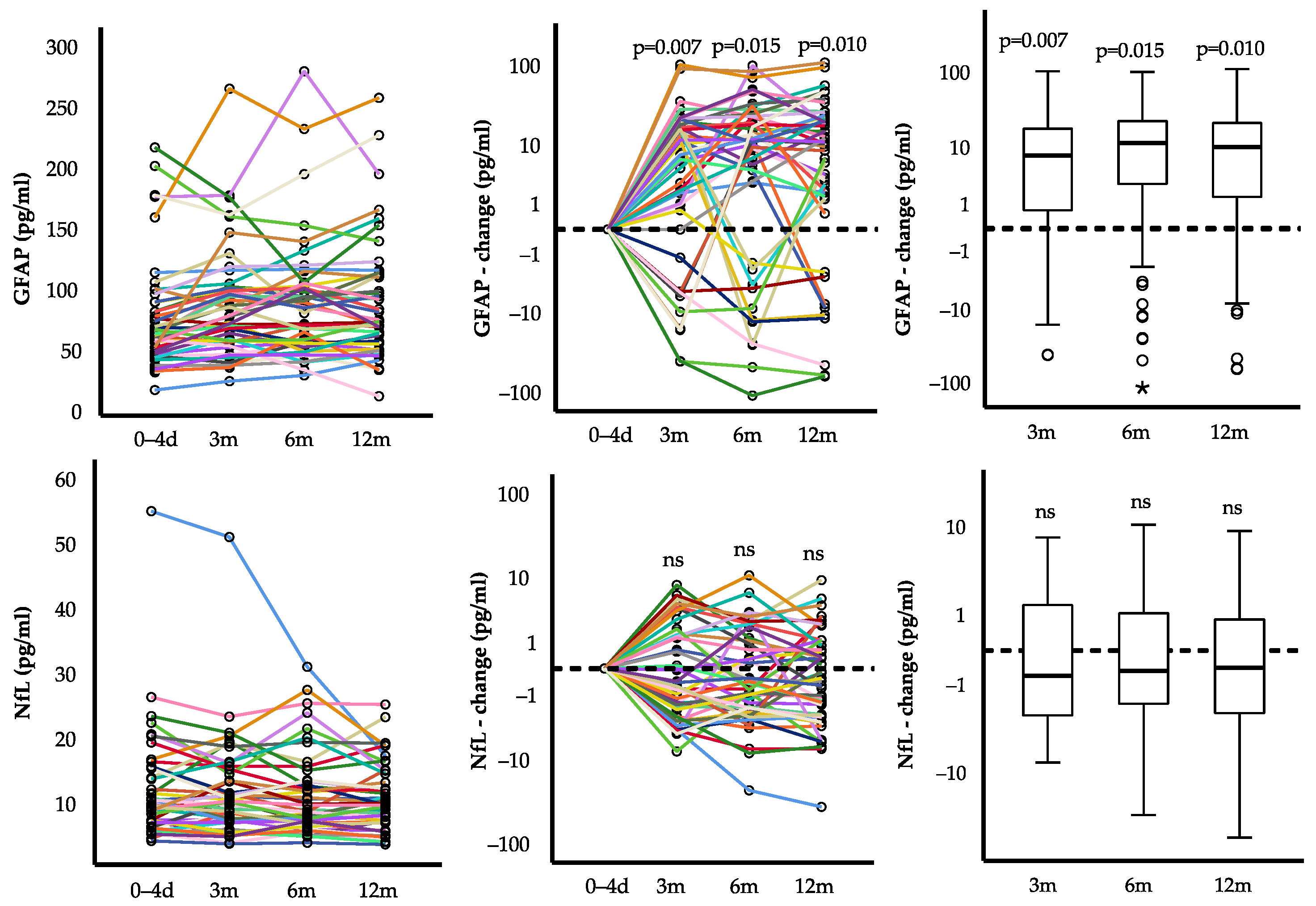
2.2. Course of GFAP and NfL after First STEMI
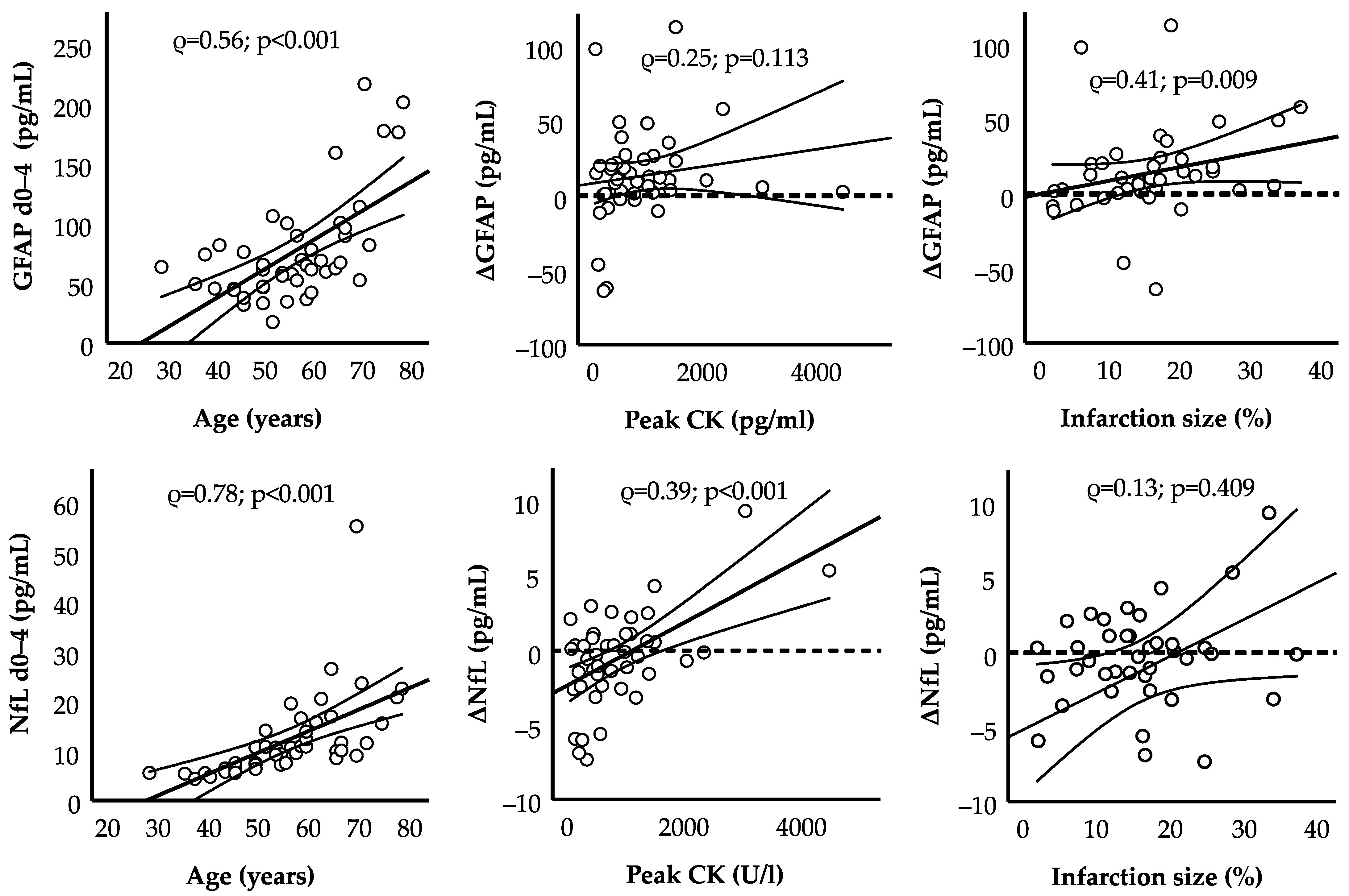
2.3. Clinical Correlates
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Selection of Patients
4.3. Study Flow
4.4. Ultrasensitive Immunoassays for GFAP/NfL
4.5. Cardiac Magnetic Resonance Imaging
4.6. Statistical Evaluation
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Disease, G.B.D.; Injury, I.; Prevalence, C. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: A systematic analysis for the Global Burden of Disease Study. Lancet 2016, 388, 1545–1602. [Google Scholar] [CrossRef]
- Zhang, W.; Luo, P. Myocardial Infarction Predisposes Neurodegenerative Diseases. J. Alzheimers Dis. 2020, 74, 579–587. [Google Scholar] [CrossRef]
- Wolters, F.J.; Segufa, R.A.; Darweesh, S.K.L.; Bos, D.; Ikram, M.A.; Sabayan, B.; Hofman, A.; Sedaghat, S. Coronary heart disease, heart failure, and the risk of dementia: A systematic review and meta-analysis. Alzheimers Dement. 2018, 14, 1493–1504. [Google Scholar] [CrossRef]
- Liu, H.; Luiten, P.G.; Eisel, U.L.; Dejongste, M.J.; Schoemaker, R.G. Depression after myocardial infarction: TNF-alpha-induced alterations of the blood-brain barrier and its putative therapeutic implications. Neurosci. Biobehav. Rev. 2013, 37, 561–572. [Google Scholar] [CrossRef]
- Honig, A.; Lousberg, R.; Wojciechowski, F.L.; Cheriex, E.C.; Wellens, H.J.; van Praag, H.M. Depression following a first heart infarct; similarities with and differences from ‘ordinary’ depression. Ned. Tijdschr. Geneeskd. 1997, 141, 196–199. [Google Scholar]
- Zuidersma, M.; Thombs, B.D.; de Jonge, P. Onset and recurrence of depression as predictors of cardiovascular prognosis in depressed acute coronary syndrome patients: A systematic review. Psychother. Psychosom. 2011, 80, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Cermakova, P.; Eriksdotter, M.; Lund, L.H.; Winblad, B.; Religa, P.; Religa, D. Heart failure and Alzheimer’s disease. J. Intern. Med. 2015, 277, 406–425. [Google Scholar] [CrossRef]
- Thackeray, J.T.; Hupe, H.C.; Wang, Y.; Bankstahl, J.P.; Berding, G.; Ross, T.L.; Bauersachs, J.; Wollert, K.C.; Bengel, F.M. Myocardial Inflammation Predicts Remodeling and Neuroinflammation After Myocardial Infarction. J. Am. Coll. Cardiol. 2018, 71, 263–275. [Google Scholar] [CrossRef]
- Bascunana, P.; Hess, A.; Borchert, T.; Wang, Y.; Wollert, K.C.; Bengel, F.M.; Thackeray, J.T. (11)C-Methionine PET Identifies Astroglia Involvement in Heart-Brain Inflammation Networking After Acute Myocardial Infarction. J. Nucl. Med. 2020, 61, 977–980. [Google Scholar] [CrossRef] [PubMed]
- Middeldorp, J.; Hol, E.M. GFAP in health and disease. Prog. Neurobiol. 2011, 93, 421–443. [Google Scholar] [CrossRef] [PubMed]
- Rajan, K.B.; Aggarwal, N.T.; McAninch, E.A.; Weuve, J.; Barnes, L.L.; Wilson, R.S.; DeCarli, C.; Evans, D.A. Remote Blood Biomarkers of Longitudinal Cognitive Outcomes in a Population Study. Ann. Neurol. 2020, 88, 1065–1076. [Google Scholar] [CrossRef]
- Oeckl, P.; Halbgebauer, S.; Anderl-Straub, S.; Steinacker, P.; Huss, A.M.; Neugebauer, H.; von Arnim, C.A.F.; Diehl-Schmid, J.; Grimmer, T.; Kornhuber, J.; et al. Glial Fibrillary Acidic Protein in Serum is Increased in Alzheimer’s Disease and Correlates with Cognitive Impairment. J. Alzheimers Dis. 2019, 67, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Traub, J.; Otto, M.; Sell, R.; Homola, G.A.; Steinacker, P.; Oeckl, P.; Morbach, C.; Frantz, S.; Pham, M.; Stork, S.; et al. Serum glial fibrillary acidic protein indicates memory impairment in patients with chronic heart failure. ESC. Heart. Fail. 2022, 9, 2626–2634. [Google Scholar] [CrossRef] [PubMed]
- Steinacker, P.; Anderl-Straub, S.; Diehl-Schmid, J.; Semler, E.; Uttner, I.; von Arnim, C.A.F.; Barthel, H.; Danek, A.; Fassbender, K.; Fliessbach, K.; et al. Serum neurofilament light chain in behavioral variant frontotemporal dementia. Neurology 2018, 91, e1390–e1401. [Google Scholar] [CrossRef] [PubMed]
- Mattsson, N.; Andreasson, U.; Zetterberg, H.; Blennow, K.; Alzheimer’s Disease Neuroimaging, I. Association of Plasma Neurofilament Light With Neurodegeneration in Patients With Alzheimer Disease. JAMA Neurol. 2017, 74, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Van der Ende, E.L.; Meeter, L.H.; Poos, J.M.; Panman, J.L.; Jiskoot, L.C.; Dopper, E.G.P.; Papma, J.M.; de Jong, F.J.; Verberk, I.M.W.; Teunissen, C.; et al. Serum neurofilament light chain in genetic frontotemporal dementia: A longitudinal, multicentre cohort study. Lancet Neurol. 2019, 18, 1103–1111. [Google Scholar] [CrossRef]
- Khalil, M.; Pirpamer, L.; Hofer, E.; Voortman, M.M.; Barro, C.; Leppert, D.; Benkert, P.; Ropele, S.; Enzinger, C.; Fazekas, F.; et al. Serum neurofilament light levels in normal aging and their association with morphologic brain changes. Nat. Commun. 2020, 11, 812. [Google Scholar] [CrossRef]
- Smolina, K.; Wright, F.L.; Rayner, M.; Goldacre, M.J. Long-term survival and recurrence after acute myocardial infarction in England, 2004 to 2010. Circ. Cardiovasc. Qual. Outcomes 2012, 5, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Nadlacki, B.; Horton, D.; Hossain, S.; Hariharaputhiran, S.; Ngo, L.; Ali, A.; Aliprandi-Costa, B.; Ellis, C.J.; Adams, R.J.; Visvanathan, R.; et al. Long term survival after acute myocardial infarction in Australia and New Zealand, 2009–2015: A population cohort study. Med. J. Aust. 2021, 214, 519–525. [Google Scholar] [CrossRef]
- Hogel, H.; Rissanen, E.; Barro, C.; Matilainen, M.; Nylund, M.; Kuhle, J.; Airas, L. Serum glial fibrillary acidic protein correlates with multiple sclerosis disease severity. Mult. Scler. 2020, 26, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Abdelhak, A.; Huss, A.; Kassubek, J.; Tumani, H.; Otto, M. Serum GFAP as a biomarker for disease severity in multiple sclerosis. Sci. Rep. 2018, 8, 14798. [Google Scholar] [CrossRef] [Green Version]
- Khalil, M.; Teunissen, C.E.; Otto, M.; Piehl, F.; Sormani, M.P.; Gattringer, T.; Barro, C.; Kappos, L.; Comabella, M.; Fazekas, F.; et al. Neurofilaments as biomarkers in neurological disorders. Nat. Rev. Neurol. 2018, 14, 577–589. [Google Scholar] [CrossRef]
- Lee, E.J.; Lim, Y.M.; Kim, S.; Choi, L.; Kim, H.; Kim, K.; Kim, H.W.; Lee, J.S.; Kim, K.K. Clinical implication of serum biomarkers and patient age in inflammatory demyelinating diseases. Ann. Clin. Transl. Neurol. 2020, 7, 992–1001. [Google Scholar] [CrossRef] [PubMed]
- Manouchehrinia, A.; Piehl, F.; Hillert, J.; Kuhle, J.; Alfredsson, L.; Olsson, T.; Kockum, I. Confounding effect of blood volume and body mass index on blood neurofilament light chain levels. Ann. Clin. Transl. Neurol. 2020, 7, 139–143. [Google Scholar] [CrossRef]
- Rana, O.R.; Schroder, J.W.; Baukloh, J.K.; Saygili, E.; Mischke, K.; Schiefer, J.; Weis, J.; Marx, N.; Rassaf, T.; Kelm, M.; et al. Neurofilament light chain as an early and sensitive predictor of long-term neurological outcome in patients after cardiac arrest. Int. J. Cardiol. 2013, 168, 1322–1327. [Google Scholar] [CrossRef]
- Kirschen, M.P.; Yehya, N.; Graham, K.; Kilbaugh, T.; Berg, R.A.; Topjian, A.; Diaz-Arrastia, R. Circulating Neurofilament Light Chain Is Associated With Survival After Pediatric Cardiac Arrest. Pediatr. Crit. Care. Med. 2020, 21, 656–661. [Google Scholar] [CrossRef]
- Wurm, R.; Arfsten, H.; Muqaku, B.; Ponleitner, M.; Bileck, A.; Altmann, P.; Rommer, P.; Seidel, S.; Hubner, P.; Sterz, F.; et al. Prediction of Neurological Recovery After Cardiac Arrest Using Neurofilament Light Chain is Improved by a Proteomics-Based Multimarker Panel. Neurocrit. Care 2022, 36, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Moseby-Knappe, M.; Mattsson-Carlgren, N.; Stammet, P.; Backman, S.; Blennow, K.; Dankiewicz, J.; Friberg, H.; Hassager, C.; Horn, J.; Kjaergaard, J.; et al. Serum markers of brain injury can predict good neurological outcome after out-of-hospital cardiac arrest. Intensive Care Med. 2021, 47, 984–994. [Google Scholar] [CrossRef]
- Takala, R.S.; Posti, J.P.; Runtti, H.; Newcombe, V.F.; Outtrim, J.; Katila, A.J.; Frantzen, J.; Ala-Seppala, H.; Kyllonen, A.; Maanpaa, H.R.; et al. Glial Fibrillary Acidic Protein and Ubiquitin C-Terminal Hydrolase-L1 as Outcome Predictors in Traumatic Brain Injury. World Neurosurg. 2016, 87, 8–20. [Google Scholar] [CrossRef] [PubMed]
- Larsson, I.M.; Wallin, E.; Kristofferzon, M.L.; Niessner, M.; Zetterberg, H.; Rubertsson, S. Post-cardiac arrest serum levels of glial fibrillary acidic protein for predicting neurological outcome. Resuscitation 2014, 85, 1654–1661. [Google Scholar] [CrossRef]
- Liu, L.R.; Liu, J.C.; Bao, J.S.; Bai, Q.Q.; Wang, G.Q. Interaction of Microglia and Astrocytes in the Neurovascular Unit. Front. Immunol. 2020, 11, 1024. [Google Scholar] [CrossRef]
- Bae, E.; Hwang, I.K.; Yoo, K.Y.; Han, T.H.; Lee, C.H.; Choi, J.H.; Yi, S.S.; Lee, S.Y.; Ryu, P.D.; Yoon, Y.S.; et al. Gliosis in the amygdala following myocardial infarction in the rat. J. Vet. Med. Sci. 2010, 72, 1041–1045. [Google Scholar] [CrossRef]
- Korn, A.; Baylan, U.; Simsek, S.; Schalkwijk, C.G.; Niessen, H.W.M.; Krijnen, P.A.J. Myocardial infarction coincides with increased NOX2 and N(epsilon)-(carboxymethyl) lysine expression in the cerebral microvasculature. Open Heart 2021, 8, e001842. [Google Scholar] [CrossRef]
- Cheng, M.; Su, X.; Liu, D.; Tian, X.; Yan, C.; Zhang, X.; Han, Y. Role of Neutrophil-Derived S100B in Acute Myocardial Infarction Patients From the Han Chinese Population. Front. Cardiovasc. Med. 2020, 7, 595446. [Google Scholar] [CrossRef] [PubMed]
- Yun, G.S.; In, Y.N.; Kang, C.; Park, J.S.; You, Y.; Min, J.H.; Ahn, H.J.; Yoo, I.; Kim, S.W.; Oh, S.K.; et al. Development of a strategy for assessing blood-brain barrier disruption using serum S100 calcium-binding protein B and neuron-specific enolase in early stage of neuroemergencies: A preliminary study. Medicine 2022, 101, e29644. [Google Scholar] [CrossRef] [PubMed]
- Dworak, M.; Stebbing, M.; Kompa, A.R.; Rana, I.; Krum, H.; Badoer, E. Attenuation of microglial and neuronal activation in the brain by ICV minocycline following myocardial infarction. Auton. Neurosci. 2014, 185, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Francis, J.; Chu, Y.; Johnson, A.K.; Weiss, R.M.; Felder, R.B. Acute myocardial infarction induces hypothalamic cytokine synthesis. Am. J. Physiol. Heart Circ. Physiol. 2004, 286, H2264–H2271. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Yin, D.; He, X.; Gao, M.; Choi, Y.; Luo, G.; Wang, H.; Qu, X. Modulation of activated astrocytes in the hypothalamus paraventricular nucleus to prevent ventricular arrhythmia complicating acute myocardial infarction. Int. J. Cardiol. 2020, 308, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.; Wang, J.; Chen, Q.; Zhao, G.; Pang, Y.; Xu, Y.; Ge, J.; Zhu, W. Inflammasome-mediated neurodegeneration following heart disease. Ann. Transl. Med. 2021, 9, 1560. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Bustos, V.; Sebastian, R.; Izquierdo, M.; Rios-Navarro, C.; Bodi, V.; Chorro, F.J.; Ruiz-Sauri, A. Changes in the spatial distribution of the Purkinje network after acute myocardial infarction in the pig. PLoS ONE 2019, 14, e0212096. [Google Scholar] [CrossRef]
- Lattanzi, S.; Silvestrini, M.; Provinciali, L. Cortical laminar necrosis following myocardial infarction. Neurol. Neurochir. Pol. 2016, 50, 271–274. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, A.; Yabluchanskiy, A.; Ghali, R.; Altara, R.; Booz, G.W.; Zouein, F.A. Cerebral blood flow alteration following acute myocardial infarction in mice. Biosci. Rep. 2018, 38, BSR20180382. [Google Scholar] [CrossRef]
- Hu, K.; Gaudron, P.; Bahner, U.; Palkovits, M.; Ertl, G. Changes of atrial natriuretic peptide in brain areas of rats with chronic myocardial infarction. Am. J. Physiol. 1996, 270, H312–H316. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.K.; Augustine, R.A.; Brown, C.H.; Schwenke, D.O. Acute myocardial infarction activates magnocellular vasopressin and oxytocin neurones. J. Neuroendocr. 2019, 31, e12808. [Google Scholar] [CrossRef]
- Thelin, E.P.; Zeiler, F.A.; Ercole, A.; Mondello, S.; Buki, A.; Bellander, B.M.; Helmy, A.; Menon, D.K.; Nelson, D.W. Serial Sampling of Serum Protein Biomarkers for Monitoring Human Traumatic Brain Injury Dynamics: A Systematic Review. Front. Neurol. 2017, 8, 300. [Google Scholar] [CrossRef]
- Antman, E.M.; Anbe, D.T.; Armstrong, P.W.; Bates, E.R.; Green, L.A.; Hand, M.; Hochman, J.S.; Krumholz, H.M.; Kushner, F.G.; Lamas, G.A.; et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction--executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1999 Guidelines for the Management of Patients With Acute Myocardial Infarction). Circulation 2004, 110, 588–636. [Google Scholar] [CrossRef] [PubMed]
- Frey, A.; Gassenmaier, T.; Hofmann, U.; Schmitt, D.; Fette, G.; Marx, A.; Herterich, S.; Boivin-Jahns, V.; Ertl, G.; Bley, T.; et al. Coagulation factor XIII activity predicts left ventricular remodelling after acute myocardial infarction. ESC. Heart Fail. 2020, 7, 2354–2364. [Google Scholar] [CrossRef]
| All Patients (n = 45) | ΔGFAP ≤ 0 (n = 9) | ΔGFAP > 0 (n = 36) | p | |
|---|---|---|---|---|
| Age (years) | 56 (49, 65) | 56 (51, 66) | 55 (46, 65) | 0.399 |
| Female sex (%) | 5 (11.1%) | 1 (11.1%) | 4 (11.1%) | >0.999 |
| Body mass index (kg/m2) | 27 (24, 29) | 26 (24, 30) | 27 (24, 29) | 0.744 |
| Arterial hypertension * | 17 (37.8%) | 4 (44.4%) | 13 (36.1%) | 0.645 |
| Diabetes mellitus † | 7 (15.6%) | 2 (22.2%) | 5 (13.9%) | 0.537 |
| Dyslipidemia ‡ | 8 (17.8%) | 1 (11.1%) | 7 (19.4%) | 0.559 |
| Current smoker | 17 (37.8%) | 1 (11.1%) | 16 (44.4%) | 0.181 |
| Previous coronary artery disease | 10 (22.2%) | 2 (22.2%) | 8 (22.2%) | >0.999 |
| Previous myocardial infarction | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | >0.999 |
| Atrial fibrillation | 2 (4.4%) | 1 (11.1%) | 1 (2.8%) | 0.497 |
| Peripheral vascular disease | 4 (8.9%) | 0 (0.0%) | 4 (11.1%) | 0.295 |
| Pain-to-balloon time (min) | 280 (123, 592) | 210 (114, 784) | 285 (131, 593) | 0.532 |
| Culprit vessel: | ||||
| - Left anterior descending | 19 (42.2%) | 4 (44.4%) | 15 (41.7%) | 0.880 |
| - Circumflex artery | 10 (22.2%) | 2 (22.2%) | 8 (22.2%) | >0.999 |
| - Right coronary artery | 16 (35.6%) | 3 (33.3%) | 13 (36.1%) | 0.876 |
| Stent implantation | 43 (95.6%) | 9 (100.0%) | 34 (94.4%) | 0.469 |
| Classification of coronary artery disease | ||||
| - 1-vessel | 27 (60.0%) | 4 (44.4%) | 23 (63.9%) | 0.287 |
| - 2-vessel | 14 (31.1%) | 3 (33.3%) | 11 (30.6%) | 0.872 |
| - 3-vessel | 4 (8.9%) | 2 (22.2%) | 2 (5.6%) | 0.116 |
| Peak creatine kinase (U/L) | 560 (302, 1136) | 239 (122, 659) | 717 (402, 1326) | 0.096 |
| Peak troponin T (pg/nL) | 2045 (950, 2952) | 1334 (452, 2447) | 2395 (1000, 3372) | 0.145 |
| Peak lactate dehydrogenase (U/L) | 530 (366, 657) | 372 (236, 607) | 533 (375, 734) | 0.117 |
| End-diastolic volume (mL/m2) d 7–9 | 87 (80, 97) | 90 (83, 94) | 86 (79, 100) | 0.924 |
| Left ventricular ejection fraction (%) d 7–9 | 54 (46, 58) | 55 (43, 59) | 54 (47, 58) | 0.836 |
| Relative infarction size (%) d 7–9 | 15.5 (9.4, 20.0) | 10.4 (2.7, 16.1) | 16.2 (11.1, 21.6) | 0.047 |
| End-diastolic volume (mL/m2) 12 m | 84 (74, 96) | 85 (75, 104) | 84 (73, 96) | 0.593 |
| Left ventricular ejection fraction (%) 12 m | 53 (47, 59) | 57 (44, 62) | 53 (47, 58) | 0.716 |
| Relative infarction size (%) 12 m | 8.5 (6.2, 14.8) | 7.2 (2.4, 15.0) | 8.5 (6.6, 14.8) | 0.387 |
| Spearman’s Rho (ρ) | GFAP 0–4 d (pg/mL) | GFAP 12 m (pg/mL) | ΔGFAP (pg/mL) | NfL 0–4 d (pg/mL) | NfL 12 m (pg/mL) | ΔNfL (pg/mL) |
|---|---|---|---|---|---|---|
| Age (years) | 0.56 * | 0.61 * | 0.15 | 0.78 * | 0.74 * | −0.15 |
| Systolic blood pressure at admission (mmHg) | −0.19 | 0.04 | 0.23 | −0.14 | 0.02 | 0.18 |
| Diastolic blood pressure at admission (mmHg) | −0.14 | 0.01 | 0.11 | −0.20 | −0.08 | 0.18 |
| Heart rate at admission (1/min) | −0.14 | −0.13 | 0.10 | −0.15 | −0.08 | 0.11 |
| Pain-to-balloon time (min) | 0.13 | 0.10 | −0.06 | −0.16 | −0.18 | −0.09 |
| Peak creatine kinase (U/L) | −0.14 | 0.03 | 0.25 | −0.19 | 0.04 | 0.39 * |
| Peak troponin T (pg/mL) | 0.08 | 0.10 | 0.03 | 0.00 | 0.08 | 0.00 |
| Peak lactate dehydrogenase (U/L) | 0.05 | 0.14 | 0.17 | −0.03 | 0.07 | 0.08 |
| Peak C-reactive protein (mg/dL) | 0.19 | 0.13 | −0.11 | 0.23 | 0.09 | −0.23 |
| Peak leucocytes (103/µL) | −0.17 | −0.10 | 0.11 | −0.04 | 0.02 | 0.00 |
| Normalized end-diastolic volume (mL/m2) d 7–9 | 0.08 | 0.02 | −0.07 | −0.09 | −0.15 | −0.14 |
| Left-ventricular ejection fraction (%) d 7–9 | −0.24 | −0.21 | 0.02 | −0.18 | −0.26 | −0.06 |
| Relative infarction size (%) d 7–9 | −0.02 | 0.19 | 0.41 * | 0.05 | 0.16 | 0.13 |
| Day 0–4 | 12 Months | Δ12 m—d 0–4 | ~ΔGFAP | p Value | |
|---|---|---|---|---|---|
| Glial fibrillary acidic protein (pg/mL) | 64 (47, 90) | 73 (53, 113) | 10 (1, 23) * | - | |
| Neurofilament light chain (pg/mL) | 11 (7, 15) | 10 (7, 14) | 0 (−2, 1) | 0.32 * | 0.030 |
| Natrium (mmol/L) | 139 (137, 140) | 140 (138, 141) | 1 (−1, 2) * | −0.14 | ns |
| Potassium (mmol/L) | 4.2 (4.0, 4.4) | 4.4 (4.2, 4.6) | 0.2 (0.0, 0.4) * | 0.03 | ns |
| Estimated GFR (mL/min/1.73 m2) | 91 (80, 106) | 82 (72, 99) | −7 (−14, 3) * | −0.14 | ns |
| Urea (mg/dL) | 31 (25, 38) | 31 (26, 37) | 0 (−7, 7) | 0.13 | ns |
| Uric acid (mg/dL) | 5.2 (4.5, 6.0) | 6.0 (5.0, 6.6) | 0.7 (0.0, 1.4) * | 0.17 | ns |
| Cholesterol (mg/dL) | 190 (165, 211) | 161 (145, 190) | −25 (−60, 11) * | −0.08 | ns |
| Low-density lipoprotein (mg/dL) | 95 (78, 114) | 82 (66, 101) | −11 (−42, 5) * | 0.03 | ns |
| High-density lipoprotein (mg/dL) | 39 (33, 46) | 45 (38, 59) | 5 (2, 9) * | −0.02 | ns |
| Triglycerides (mg/dL) | 128 (105, 162) | 162 (98, 195) | 45 (−12, 108) * | −0.14 | ns |
| Aspartate aminotransferase (U/L) | 48 (33, 93) | 28 (21, 32) | −21 (−155, −4) | −0.07 | ns |
| Alanine aminotransferase (U/L) | 45 (31, 65) | 30 (20, 34) | −19 (−27, 3) | −0.23 | ns |
| γ-glutamyltransferase (U/L) | 44 (28, 70) | 32 (19, 38) | −3 (−53, 4) | −0.22 | ns |
| Lactate dehydrogenase (U/L) | 530 (366, 657) | 198 (175, 240) | −293 (−491, −108) * | −0.15 | ns |
| Creatine kinase (U/L) | 560 (302, 1136) | 113 (82, 165) | −426 (−975, −95) * | −0.19 | ns |
| NT-proBNP (pg/mL) | 844 (627, 1368) | 133 (48, 291) | −755 (−1329, −403) * | −0.02 | ns |
| Troponin T (pg/mL) | 2044 (950, 2952) | 6 (5, 9) | −2037 (−2946, −945) * | −0.03 | ns |
| Complement factor C3c (mg/dL) | 121 (107, 132) | 118 (105, 131) | −3 (−14, 8) | −0.02 | ns |
| Albumin (g/dL) | 4.2 (3.9, 4.3) | 4.6 (4.4, 4.9) | 0.5 (0.3, 0.8) * | 0.00 | ns |
| Hemoglobin (g/dL) | 13.9 (13.0, 15.1) | 14.6 (13.8, 15.5) | 0.8 (0.2, 1.7) * | −0.09 | ns |
| Hematocrit (%) | 41 (38, 43) | 43 (41, 45) | 3 (0, 5) * | −0.16 | ns |
| Thrombocytes (103/µL) | 242 (189, 288) | 235 (189, 285) | 3 (−23, 19) | 0.12 | ns |
| Leucocytes (103/µL) | 9.2 (8.1, 10.9) | 6.3 (5.2, 7.2) | −2.8 (−4.2, −1.9) * | −0.14 | ns |
| HbA1c (%) | 5.6 (5.4, 6.7) | 5.7 (5.4, 6.0) | 0.0 (−0.2, 0.3) | 0.15 | ns |
| C-reactive protein (mg/dL) | 1.7 (0.7, 6.2) | 0.1 (0.0, 0.2) | −1.6 (−6.1, −0.6) * | 0.12 | ns |
| Inclusion Criteria |
|---|
| Age ≥ 18 years and written informed consent |
| No previous history of myocardial infarction |
| New onset of chest pain in the past 7 days |
| Confirmed myocardial infarction in cardiac catheterization |
| Fulfillment of STEMI criteria: |
| - Either ST-segment elevations in at least two adjacent leads |
| - ST-segment elevations ≥ 0.1 mV in the extremity leads |
| - ST-segment elevations ≥ 0.2 mV in the chest wall leads |
| - Or new onset left bundle branch block with matching clinic |
| Exclusion criteria |
| Tumor disease or other critical illness with a life expectancy of <1 year |
| Terminal renal failure or hemodialysis |
| Rheumatologic disease with the need for immunomodulatory drugs |
| Autoimmune disease with the need for immunomodulatory drugs |
| Congenital neuromuscular disease |
| Myasthenia gravis |
| Graves’ disease |
| Incapacity to consent |
| Sociological, psychological, mental, or other limitations |
| Continued alcohol or drug abuse |
| Pregnancy or lactation |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Traub, J.; Grondey, K.; Gassenmaier, T.; Schmitt, D.; Fette, G.; Frantz, S.; Boivin-Jahns, V.; Jahns, R.; Störk, S.; Stoll, G.; et al. Sustained Increase in Serum Glial Fibrillary Acidic Protein after First ST-Elevation Myocardial Infarction. Int. J. Mol. Sci. 2022, 23, 10304. https://doi.org/10.3390/ijms231810304
Traub J, Grondey K, Gassenmaier T, Schmitt D, Fette G, Frantz S, Boivin-Jahns V, Jahns R, Störk S, Stoll G, et al. Sustained Increase in Serum Glial Fibrillary Acidic Protein after First ST-Elevation Myocardial Infarction. International Journal of Molecular Sciences. 2022; 23(18):10304. https://doi.org/10.3390/ijms231810304
Chicago/Turabian StyleTraub, Jan, Katja Grondey, Tobias Gassenmaier, Dominik Schmitt, Georg Fette, Stefan Frantz, Valérie Boivin-Jahns, Roland Jahns, Stefan Störk, Guido Stoll, and et al. 2022. "Sustained Increase in Serum Glial Fibrillary Acidic Protein after First ST-Elevation Myocardial Infarction" International Journal of Molecular Sciences 23, no. 18: 10304. https://doi.org/10.3390/ijms231810304








