Chitin Synthesis in Yeast: A Matter of Trafficking
Abstract
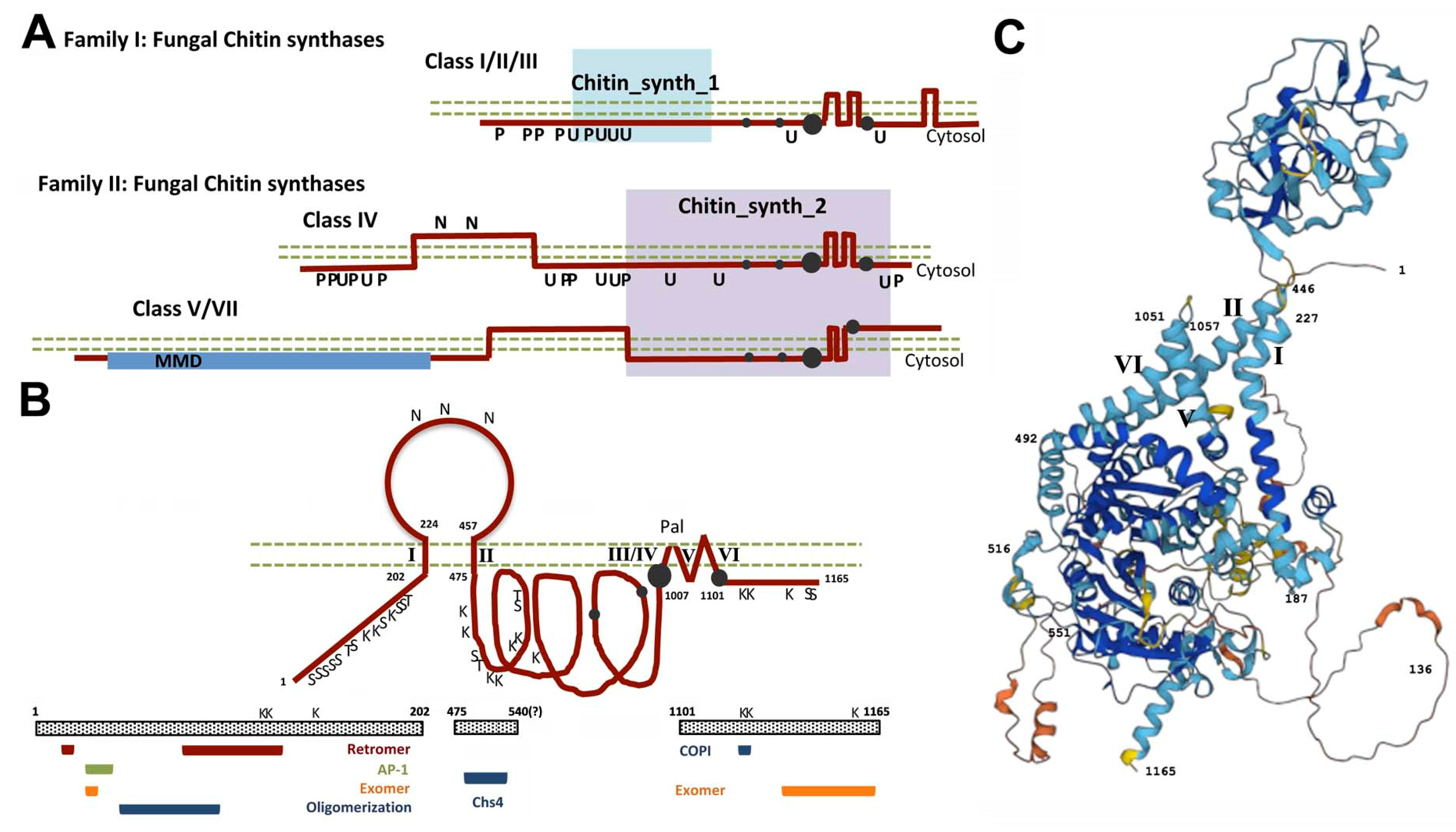
:1. Chitin Synthesis: A Historical Perspective
2. The Road of Chs3 to the PM and Beyond
3. The First Step of the Road: Chs3 Folding and Export from the ER
4. The trans-Golgi Network (TGN): The Central Station of Chs3 Trafficking
4.1. Going Ahead: Sorting Chs3 in the Golgi
4.2. Endosomal Recycling: A Noteworthy Step in the Intracellular Trafficking of Chs3
5. Controlling Residence Time Is the Chief Regulator of Chitin Synthesis
6. Avoiding the Vacuole: A Signature of Chs3 Trafficking
7. Chs3 Outside of Yeast Cells
8. Other Chitin Synthases and Other Modes of Intracellular Transport
9. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cabib, E.; Farkas, V. The control of morphogenesis: An enzymatic mechanism for the initiation of septum formation in yeast. Proc. Natl. Acad. Sci. USA 1971, 68, 2052–2056. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cabib, E.; Keller, F.A. Chitin and yeast budding. Allosteric inhibition of chitin synthetase by a heat-stable protein from yeast. J. Biol. Chem. 1971, 246, 167–173. [Google Scholar] [CrossRef]
- McMurrough, I.; Flores-Carreon, A.; Bartnicki-Garcia, S. Pathway of chitin synthesis and cellular localization of chitin synthetase in Mocor rouxii. J. Biol. Chem. 1971, 246, 3999–4007. [Google Scholar] [CrossRef]
- Cabib, E.; Ulane, R.E. Chitin synthetase activating factor from yeast, a protease. Biochem. Biophys. Res. Commun. 1973, 50, 186–191. [Google Scholar] [CrossRef]
- Ulane, R.E.; Cabib, E. The activating system of chitin synthetase from Saccharomyces cerevisiae. Purification and properties of an inhibitor of the activating factor. J. Biol. Chem. 1974, 249, 3418–3422. [Google Scholar] [CrossRef]
- Zubenko, G.S.; Mitchell, A.P.; Jones, E.W. Septum formation, cell division, and sporulation in mutants of yeast deficient in proteinase B. Proc. Natl. Acad. Sci. USA 1979, 76, 2395–2399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duran, A.; Bowers, B.; Cabib, E. Chitin synthase zymogen is attached to the plasma membrane. Proc. Natl. Acad. Sci. USA 1975, 72, 3952–3955. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duran, A.; Cabib, E.; Bowers, B. Chitin synthetase distribution on the yeast plasma membrane. Science 1979, 203, 363–365. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Herrera, J.; Bartnicki-Garcia, S. Synthesis of cell wall microfibrils in vitro by a “soluble” chitin synthetase from Mucor rouxii. Science 1974, 186, 357–359. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Herrera, J.; Sing, V.O.; van der Woude, W.J.; Bartnicki-Garcia, S. Microfibril assembly by granules of chitin synthetase. Proc. Natl. Acad. Sci. USA 1975, 72, 2706–2710. [Google Scholar] [CrossRef] [PubMed]
- Schekman, R.; Brawley, V. Localized deposition of chitin on the yeast cell surface in response to mating pheromone. Proc. Natl. Acad. Sci. USA 1979, 76, 645–649. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chuang, J.S.; Schekman, R.W. Differential trafficking and timed localization of two chitin synthase proteins, Chs2p and Chs3p. J. Cell Biol. 1996, 135, 597–610. [Google Scholar]
- Roncero, C. The genetic complexity of chitin synthesis in fungi. Curr. Genet. 2002, 41, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Bulawa, C.E.; Slater, M.; Cabib, E.; Au-Young, J.; Sburlati, A.; Adair, W.L.J.; Robbins, P.W. The S.cerevisiae structural gene for chitin synthase is not required for chitin synthesis in vivo. Cell 1986, 46, 213–225. [Google Scholar] [CrossRef]
- Cabib, E.; Sburlati, A.; Bowers, B.; Silverman, S.J. Chitin synthase 1, an auxiliary enzyme for chitin synthesis in Saccharomyces cerevisiae. J. Cell Biol. 1989, 108, 1665–1672. [Google Scholar] [CrossRef] [PubMed]
- Silverman, S.J.; Sburlati, A.; Slater, M.L.; Cabib, E. Chitin synthase 2 is essential for septum formation and cell division in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 1988, 85, 4735–4739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shaw, J.A.; Mol, P.C.; Bowers, B.; Silverman, S.J.; Valdivieso, M.H.; Duran, A.; Cabib, E. The function of chitin synthases 2 and 3 in the Saccharomyces cerevisiae cell cycle. J. Cell Biol. 1991, 114, 111–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roncero, C.; Valdivieso, M.H.; Ribas, J.C.; Duran, A. Isolation and characterization of Saccharomyces cerevisiae mutants resistant to Calcofluor white. J. Bacteriol. 1988, 170, 1950–1954. [Google Scholar] [CrossRef] [Green Version]
- Valdivieso, M.H.; Mol, P.C.; Shaw, J.A.; Cabib, E.; Duran, A. CAL1, a gene required for activity of chitin synthase 3 in Saccharomyces cerevisiae. J. Cell Biol. 1991, 114, 101–109. [Google Scholar] [CrossRef] [Green Version]
- Roncero, C.; Sanchez-Diaz, A.; Valdivieso, M.H. Chitin synthesis and fungal morphogenesis. In The Mycota III. Biochemistry and Molecular Biology; Hoffmeister, D., Ed.; Springer: Cham, Switzerland, 2016; pp. 167–190. [Google Scholar]
- Bulawa, C.E. Genetics and Molecular biology of chitin synthesis in fungi. Annu. Rev. Microbiol. 1993, 47, 505–534. [Google Scholar] [CrossRef]
- Choi, W.; Sburlati, A.; Cabib, E. Chitin synthase 3 from yeast has zymogenic properties that depend on both the CAL1 and CAL3 genes. Proc. Natl. Acad. Sci. USA 1994, 91, 4727–4730. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.; Santos, B.; Duran, A.; Cabib, E. Are Yeast chitin synthases regulated at the transciptional or the posttranslational level? Mol. Cell. Biol. 1994, 14, 7685–7694. [Google Scholar] [CrossRef] [PubMed]
- Varadi, M.; Anyango, S.; Deshpande, M.; Nair, S.; Natassia, C.; Yordanova, G.; Yuan, D.; Stroe, O.; Wood, G.; Laydon, A.; et al. AlphaFold Protein Structure Database: Massively expanding the structural coverage of protein-sequence space with high-accuracy models. Nucleic Acids Res. 2022, 50, D439–D444. [Google Scholar] [CrossRef] [PubMed]
- Daras, G.; Templalexis, D.; Avgeri, F.; Tsitsekian, D.; Karamanou, K.; Rigas, S. Updating Insights into the Catalytic Domain Properties of Plant Cellulose synthase (CesA) and Cellulose synthase-like (Csl) Proteins. Molecules 2021, 26, 4335. [Google Scholar] [CrossRef]
- Farinha, C.M.; Canato, S. From the endoplasmic reticulum to the plasma membrane: Mechanisms of CFTR folding and trafficking. Cell. Mol. Life Sci. 2017, 74, 39–55. [Google Scholar] [CrossRef]
- Ren, Z.; Chhetri, A.; Guan, Z.; Suo, Y.; Yokoyama, K.; Lee, S. Structural basis for inhibition and regulation of a chitin synthase from Candida albicans. Nat. Struct. Mol. Biol. 2022, 29, 653–664. [Google Scholar] [CrossRef]
- Cos, T.; Ford, R.A.; Trilla, J.A.; Duran, A.; Cabib, E.; Roncero, C. Molecular analysis of Chs3p participation in chitin synthase III activity. Eur. J. Biochem. 1998, 256, 419–426. [Google Scholar] [CrossRef]
- Zanolari, B.; Rockenbauch, U.; Trautwein, M.; Clay, L.; Barral, Y.; Spang, A. Transport to the plasma membrane is regulated differently early and late in the cell cycle in Saccharomyces cerevisiae. J. Cell Sci. 2011, 124, 1055–1066. [Google Scholar] [CrossRef] [Green Version]
- Okada, H.; MacTaggart, B.; Ohya, Y.; Bi, E. The kinetic landscape and interplay of protein networks in cytokinesis. iScience 2021, 24, 101917. [Google Scholar] [CrossRef]
- Trilla, J.A.; Duran, A.; Roncero, C. Chs7p, a new protein involved in the control of protein export from the endoplasmic reticulum that is specifically engaged in the regulation of chitin synthesis in Saccharomyces cerevisiae. J. Cell Biol. 1999, 145, 1153–1163. [Google Scholar] [CrossRef] [Green Version]
- Kota, J.; Ljungdahl, P.O. Specialized membrane-localized chaperones prevent aggregation of polytopic proteins in the ER. J. Cell Biol. 2005, 168, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Herzig, Y.; Sharpe, H.J.; Elbaz, Y.; Munro, S.; Schuldiner, M. A systematic approach to pair secretory cargo receptors with their cargo suggests a mechanism for cargo selection by Erv14. PLoS Biol. 2012, 10, e1001329. [Google Scholar] [CrossRef] [PubMed]
- Sacristan, C.; Manzano-Lopez, J.; Reyes, A.; Spang, A.; Muniz, M.; Roncero, C. Dimerization of the chitin synthase Chs3 is monitored at the Golgi and affects its endocytic recycling. Mol. Microbiol. 2013, 90, 252–266. [Google Scholar]
- Kota, J.; Gilstring, C.F.; Ljungdahl, P.O. Membrane chaperone Shr3 assists in folding amino acid permeases preventing precocious ERAD. J. Cell Biol. 2007, 176, 617–628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gohlke, S.; Muthukrishnan, S.; Merzendorfer, H. In Vitro and In Vivo Studies on the Structural Organization of Chs3 from Saccharomyces cerevisiae. Int. J. Mol. Sci. 2017, 18, 702. [Google Scholar] [CrossRef] [Green Version]
- Zhu, Y.; McFarlane, H.E. Regulation of cellulose synthesis via exocytosis and endocytosis. Curr. Opin. Plant Biol. 2022, 69, 102273. [Google Scholar] [CrossRef]
- Dharwada, S.T.; Dalton, L.E.; Bean, B.D.M.; Padmanabhan, N.; Choi, C.; Schluter, C.; Davey, M.; Conibear, E. The chaperone Chs7 forms a stable complex with Chs3 and promotes its activity at the cell surface. Traffic 2018, 19, 285–295. [Google Scholar] [CrossRef] [Green Version]
- Sanz, M.; Carrano, L.; Jimenez, C.; Candiani, G.; Trilla, J.A.; Duran, A.; Roncero, C. Candida albicans strains deficient in CHS7, a key regulator of chitin synthase III, exhibit morphogenetic alterations and attenuated virulence. Microbiology 2005, 151, 2623–2636. [Google Scholar] [CrossRef] [Green Version]
- Rico-Ramírez, A.M.; Roberson, R.W.; Riquelme, M. Imaging the secretory compartments involved in the intracellular traffic of CHS-4, a class IV chitin synthase, in Neurospora crassa. Fungal Genet. Biol. 2018, 117, 30–42. [Google Scholar] [CrossRef]
- Lam, K.K.; Davey, M.; Sun, B.; Roth, A.F.; Davis, N.G.; Conibear, E. Palmitoylation by the DHHC protein Pfa4 regulates the ER exit of Chs3. J. Cell Biol. 2006, 174, 19–25. [Google Scholar] [CrossRef]
- Lam, K.K. The Role of Plamitoylation in Endoplasmic Reticulum Trasnport and Quality Control of the Yeast Polytopic Protein CHS3. Ph.D. Thesis, University of British Columbia, Vancouver, BC, Canada, 2009. [Google Scholar]
- Sanchatjate, S.; Schekman, R. Chs5/6 complex: A multiprotein complex that interacts with and conveys chitin synthase III from the trans-Golgi network to the cell surface. Mol. Biol. Cell 2006, 17, 4157–4166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trautwein, M.; Schindler, C.; Gauss, R.; Dengjel, J.; Hartmann, E.; Spang, A. Arf1p, Chs5p and the ChAPs are required for export of specialized cargo from the Golgi. EMBO J. 2006, 25, 943–954. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.W.; Hamamoto, S.; Orci, L.; Schekman, R. Exomer: A coat complex for transport of select membrane proteins from the trans-Golgi network to the plasma membrane in yeast. J. Cell Biol. 2006, 174, 973–983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santos, B.; Snyder, M. Targeting of chitin synthase 3 to polarized growth sites in yeast requires Chs5p and Myo2p. J. Cell Biol. 1997, 136, 95–110. [Google Scholar] [CrossRef] [Green Version]
- Ziman, M.; Chuang, J.S.; Tsung, M.; Hamamoto, S.; Schekman, R. Chs6p-dependent anterograde transport of Chs3p from the chitosome to the plasma membrane in Saccharomycces cerevisiae. Mol. Biol. Cell 1998, 9, 1565–1576. [Google Scholar] [CrossRef] [Green Version]
- Anton, C.; Taubas, J.V.; Roncero, C. The Functional Specialization of Exomer as a Cargo Adaptor during the Evolution of Fungi. Genetics 2018, 208, 1483–1498. [Google Scholar] [CrossRef] [Green Version]
- Rockenbauch, U.; Ritz, A.M.; Sacristan, C.; Roncero, C.; Spang, A. The complex interactions of Chs5p, the ChAPs, and the cargo Chs3p. Mol. Biol. Cell 2012, 23, 4402–4415. [Google Scholar] [CrossRef] [Green Version]
- Barfield, R.M.; Fromme, J.C.; Schekman, R. The exomer coat complex transports Fus1p to the plasma membrane via a novel plasma membrane sorting signal in yeast. Mol. Biol. Cell 2009, 20, 4985–4996. [Google Scholar] [CrossRef] [Green Version]
- Ritz, A.M.; Trautwein, M.; Grassinger, F.; Spang, A. The prion-like domain in the exomer-dependent cargo Pin2 serves as a trans-Golgi retention motif. Cell Rep. 2014, 7, 249–260. [Google Scholar] [CrossRef] [Green Version]
- Paczkowski, J.E.; Richardson, B.C.; Strassner, A.M.; Fromme, J.C. The exomer cargo adaptor structure reveals a novel GTPase-binding domain. EMBO J. 2012, 31, 4191–4203. [Google Scholar] [CrossRef] [Green Version]
- Richardson, B.C.; Fromme, J.C. The exomer cargo adaptor features a flexible hinge domain. Structure 2013, 21, 486–492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paczkowski, J.E.; Fromme, J.C. Structural basis for membrane binding and remodeling by the exomer secretory vesicle cargo adaptor. Dev. Cell 2014, 30, 610–624. [Google Scholar] [CrossRef]
- Huranova, M.; Muruganandam, G.; Weiss, M.; Spang, A. Dynamic assembly of the exomer secretory vesicle cargo adaptor subunits. EMBO Rep. 2016, 17, 202–219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lesage, G.; Shapiro, J.; Specht, C.A.; Sdicu, A.M.; Menard, P.; Hussein, S.; Tong, A.H.; Boone, C.; Bussey, H. An interactional network of genes involved in chitin synthesis in Saccharomyces cerevisiae. BMC Genet. 2005, 6, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anton, C.; Zanolari, B.; Arcones, I.; Wang, C.; Mulet, J.M.; Spang, A.; Roncero, C. Involvement of the exomer complex in the polarized transport of Ena1 required for Saccharomyces cerevisiae survival against toxic cations. Mol. Biol. Cell 2017, 28, 3672–3685. [Google Scholar] [CrossRef] [Green Version]
- Zhu, L.; Sardana, R.; Jin, D.K.; Emr, S.D. Calcineurin-dependent regulation of endocytosis by a plasma membrane ubiquitin ligase adaptor, Rcr1. J. Cell Biol. 2020, 219, e201909158. [Google Scholar] [CrossRef]
- Anton-Plagaro, C.; Sanchez, N.; Valle, R.; Mulet, J.M.; Duncan, M.C.; Roncero, C. Exomer complex regulates protein traffic at the TGN through differential interactions with cargos and clathrin adaptor complexes. FASEB J. 2021, 35, e21615. [Google Scholar] [CrossRef]
- Hoya, M.; Yanguas, F.; Moro, S.; Prescianotto-Baschong, C.; Doncel, C.; de León, N.; Curto, M.Á.; Spang, A.; Valdivieso, M.H. Traffic Through the Trans-Golgi Network and the Endosomal System Requires Collaboration Between Exomer and Clathrin Adaptors in Fission Yeast. Genetics 2017, 205, 673–690. [Google Scholar] [CrossRef] [Green Version]
- Moro, S.; Moscoso-Romero, E.; Poddar, A.; Mulet, J.M.; Perez, P.; Chen, Q.; Valdivieso, M.H. Exomer Is Part of a Hub Where Polarized Secretion and Ionic Stress Connect. Front. Microbiol. 2021, 12, 708354. [Google Scholar] [CrossRef]
- Ramirez-Macias, I.; Barlow, L.D.; Anton, C.; Spang, A.; Roncero, C.; Dacks, J.B. Evolutionary cell biologyt traces de rise of exomer complex in Fungi from an ancient eukaryotic component. Sci. Rep. 2018, 8, 11154. [Google Scholar] [CrossRef] [Green Version]
- Martzoukou, O.; Diallinas, G.; Amillis, S. Secretory Vesicle Polar Sorting, Endosome Recycling and Cytoskeleton Organization Require the AP-1 Complex in Aspergillus nidulans. Genetics 2018, 209, 1121–1138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bonifacino, J.S. Adaptor proteins involved in polarized sorting. J. Cell Biol. 2014, 204, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Valdivia, R.H.; Baggot, D.; Chuang, J.S.; Schekman, R. The yeast Clathrin adaptor protein complex 1 is required for the efficeint retention of a subset of late Golgi membrane proteins. Dev. Cell 2002, 2, 283–294. [Google Scholar] [CrossRef] [Green Version]
- Copic, A.; Starr, T.L.; Schekman, R. Ent3p and Ent5p exhibit cargo-specific functions in trafficking proteins between the trans-Golgi network and the endosomes in yeast. Mol. Biol. Cell 2007, 18, 1803–1815. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ziman, M.; Chuang, J.S.; Schekman, R.W. Chs1p and Chs3p, two proteins involved in chitin synthesis, populate a compartment of the Saccharomyces cerevisiae endocytic pathway. Mol. Biol. Cell 1996, 7, 1909–1919. [Google Scholar] [CrossRef] [Green Version]
- Day, K.J.; Casler, J.C.; Glick, B.S. Budding Yeast Has a Minimal Endomembrane System. Dev. Cell 2018, 44, 56–72. [Google Scholar] [CrossRef]
- Starr, T.L.; Pagant, S.; Wang, C.W.; Schekman, R. Sorting Signals That Mediate Traffic of Chitin Synthase III between the TGN/Endosomes and to the Plasma Membrane in Yeast. PLoS ONE 2012, 7, e46386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daboussi, L.; Costaguta, G.; Payne, G.S. Phosphoinositide-mediated clathrin adaptor progression at the trans-Golgi network. Nat. Cell Biol. 2012, 14, 239–248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, W.; Du, G.; Zhou, J.; Chen, J. Regulation of Sensing, Transportation, and Catabolism of Nitrogen Sources in Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev. 2018, 82, e00017–e00040. [Google Scholar] [CrossRef] [Green Version]
- Valdivia, R.H.; Schekman, R. The yeasts Rho1p and Pkc1p regulate the transport of chitin synthase III (Chs3p) from internal stores to the plasma membrane. Proc. Natl. Acad. Sci. USA 2003, 100, 10287–10292. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Rodriguez, L.J.; Trilla, J.A.; Castro, C.; Valdivieso, M.H.; Duran, A.; Roncero, C. Characterization of the chitin biosynthesis process as a compensatory mechanism in the fks1 mutant of Saccharomyces cerevisiae. FEBS Lett. 2000, 478, 84–88. [Google Scholar] [CrossRef] [Green Version]
- Schorr, M.; Then, A.; Tahirovic, S.; Hug, N.; Mayinger, P. The phosphoinositide phosphatase Sac1p controls trafficking of the yeast Chs3p chitin synthase. Curr. Biol. 2001, 11, 1421–1426. [Google Scholar] [CrossRef]
- Sakane, H.; Yamamoto, T.; Tanaka, K. The functional relationship between the Cdc50p-Drs2p putative aminophospholipid translocase and the Arf GAP Gcs1p in vesicle formation in the retrieval pathway from yeast early endosomes to the TGN. Cell Struct. Funct. 2006, 31, 87–108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, K.; Surendhran, K.; Nothwehr, S.F.; Graham, T.R. P4-ATPase requirement for AP-1/clathrin function in protein transport from the trans-Golgi network and early endosomes. Mol. Biol. Cell 2008, 19, 3526–3535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sciorra, V.A.; Audhya, A.; Parsons, A.B.; Segev, N.; Boone, C.; Emr, S.D. Synthetic genetic array analysis of the PtdIns 4-kinase Pik1p identifies components in a Golgi-specific Ypt31/rab-GTPase signaling pathway. Mol. Biol. Cell 2005, 16, 776–793. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ortiz, D.; Novick, P.J. Ypt32p regulates the translocation of Chs3p from an internal pool to the plasma membrane. Eur. J. Cell Biol. 2006, 85, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Holthuis, J.C.; Nichols, B.J.; Pelham, H.R. The syntaxin Tlg1p mediates trafficking of chitin synthase III to polarized growth sites in yeast. Mol. Biol. Cell 1998, 9, 3383–3397. [Google Scholar] [CrossRef] [Green Version]
- Morishita, M.; Mendonsa, R.; Wright, J.; Engebrecht, J. Snc1p v-SNARE transport to the prospore membrane during yeast sporulation is dependent on endosomal retrieval pathways. Traffic 2007, 8, 1231–1245. [Google Scholar] [CrossRef] [PubMed]
- Whitfield, S.T.; Burston, H.E.; Bean, B.D.; Raghuram, N.; Maldonado-Báez, L.; Davey, M.; Wendland, B.; Conibear, E. The alternate AP-1 adaptor subunit Apm2 interacts with the Mil1 regulatory protein and confers differential cargo sorting. Mol. Biol. Cell 2016, 27, 588–598. [Google Scholar] [CrossRef]
- Cabib, E.; Bowers, B.; Roberts, R.L. Vectorial synthesis of a polysaccharide by isolated plasma membranes. Proc. Natl. Acad. Sci. USA 1983, 80, 3318–3321. [Google Scholar] [CrossRef] [Green Version]
- DeMarini, D.J.; Adams, A.E.M.; Fares, H.; de Virgilio, C.; Valle, G.; Chuang, J.S.; Pringle, J.R. A septin-based hierarchy of proteins required for localized deposition of chitin in the Saccharomyces cerevisiae cell wall. J. Cell Biol. 1997, 139, 75–93. [Google Scholar] [CrossRef]
- Kozubowski, L.; Panek, H.; Rosenthal, A.; Bloecher, A.; DeMarini, D.J.; Tatchell, K. A Bni4-Glc7 phospatase complex that recruit chitin synthase to the site of bud emergence. Mol. Biol. Cell 2003, 14, 26–39. [Google Scholar] [CrossRef] [PubMed]
- Sanz, M.; Castrejon, F.; Duran, A.; Roncero, C. Saccharomyces cerevisiae Bni4p directs the formation of the chitin ring and also participates in the correct assembly of the septum structure. Microbiology 2004, 150, 3229–3241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reyes, A.; Sanz, M.; Duran, A.; Roncero, C. Chitin synthase III requires Chs4p-dependent translocation of Chs3p into the plasma membrane. J. Cell Sci. 2007, 120, 1998–2009. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sacristan, C.; Reyes, A.; Roncero, C. Neck compartmentalization as the molecular basis for the different endocytic behaviour of Chs3 during budding or hyperpolarized growth in yeast cells. Mol. Microbiol. 2012, 83, 1124–1135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grabinska, K.A.; Magnelli, P.; Robbins, P.W. Prenylation of Saccharomyces cerevisiae Chs4p Affects Chitin Synthase III activity and chitin chain length. Eukaryot. Cell 2007, 6, 328–336. [Google Scholar] [CrossRef] [Green Version]
- Spira, F.; Mueller, N.S.; Beck, G.; von Olshausen, P.; Beig, J.; Wedlich-Söldner, R. Patchwork organization of the yeast plasma membrane into numerous coexisting domains. Nat. Cell Biol. 2012, 14, 640–648. [Google Scholar] [CrossRef] [PubMed]
- Trilla, J.A.; Cos, T.; Duran, A.; Roncero, C. Characterisation of CHS4 (CAL2), a gene of Saccharomyces cerevisiae involved in chitin biosynthesis and allelic to SKT5 and CSD4. Yeast 1997, 13, 795–807. [Google Scholar] [CrossRef]
- Ono, N.; Yabe, T.; Sudoh, M.; Nakajima, T.; Yamada-Okabe, T.; Arisawa, M.; Yamada-Okabe, H. The yeast Chs4p protein stimulates the trypsin-sensitive activity of chitin synthase 3 through an apparent protein-protein interaction. Microbiology 2000, 146, 385–391. [Google Scholar] [CrossRef] [Green Version]
- Sanz, M.; Trilla, J.A.; Duran, A.; Roncero, C. Control of chitin synthesis through Shc1p, a functional homologue of Chs4p specifically induced during sporulation. Mol. Microbiol. 2002, 43, 1183–1195. [Google Scholar]
- Gohlke, S.; Heine, D.; Schmitz, H.P.; Merzendorfer, H. Septin-associated protein kinase Gin4 affects localization and phosphorylation of Chs4, the regulatory subunit of the Baker’s yeast chitin synthase III complex. Fungal. Genet. Biol. 2018, 117, 11–20. [Google Scholar] [CrossRef]
- Oh, Y.; Schreiter, J.H.; Okada, H.; Wloka, C.; Okada, S.; Yan, D.; Duan, X.; Bi, E. Hof1 and Chs4 Interact via F-BAR Domain and Sel1-like Repeats to Control Extracellular Matrix Deposition during Cytokinesis. Curr. Biol. 2017, 27, 2878–2886. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knafler, H.C.; Smaczynska-de Rooij, I.I.; Walker, L.A.; Lee, K.K.; Gow, N.A.R.; Ayscough, K.R. AP-2-Dependent Endocytic Recycling of the Chitin Synthase Chs3 Regulates Polarized Growth in Candida albicans. mBio 2019, 10, e02421–e02418. [Google Scholar] [CrossRef] [PubMed]
- Sudoh, M.; Tatsuno, K.; Ono, N.; Chibana, H.; Yamada-Okabe, H.; Arisawa, M. The Candida albicans CHS4 gene complements a Saccharomyces cerevisiae skt5/chs4 mutation and is involved in chitin biosynthesis. Microbiology 1999, 145, 1613–1622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banks, I.R.; Specht, C.A.; Donlin, M.J.; Gerik, K.J.; Levitz, S.M.; Lodge, J.K. A chitin synthase and its regulator protein are critical for chitosan production and growth of the fungal pathogen Cryptococcus neoformans. Eukaryot. Cell 2005, 4, 1902–1912. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arcones, I.; Sacristán, C.; Roncero, C. Maintaining protein homeostasis: Early and late endosomal dual recycling for the maintenance of intracellular pools of the plasma membrane protein Chs3. Mol. Biol. Cell 2016, 27, 4021–4032. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Macgurn, J.A.; Liu, M.; Emr, S.D. The ART-Rsp5 ubiquitin ligase network comprises a plasma membrane quality control system that protects yeast cells from proteotoxic stress. eLife 2013, 2, e00459. [Google Scholar] [CrossRef]
- Cui, T.; Peterson, T.A.; Burd, C.G. A CDC25 family protein phosphatase gates cargo recognition by the Vps26 retromer subunit. eLife 2017, 6, e24126. [Google Scholar] [CrossRef]
- Zhao, K.; Bleackley, M.; Chisanga, D.; Gangoda, L.; Fonseka, P.; Liem, M.; Kalra, H.; Saffar, H.A.; Keerthikumar, S.; Ang, C.S.; et al. Extracellular vesicles secreted by Saccharomyces cerevisiae are involved in cell wall remodelling. Commun. Biol. 2019, 2, 305. [Google Scholar] [CrossRef] [Green Version]
- Liebana-Jordan, M.; Brotons, B.; Falcon-Perez, J.M.; Gonzalez, E. Extracellular Vesicles in the Fungi Kingdom. Int. J. Mol. Sci. 2021, 22, 7221. [Google Scholar] [CrossRef] [PubMed]
- Moyano-Rodríguez, Y.; Vaquero, D.; Vilalta-Castany, O.; Foltman, M.; Sanchez-Diaz, A.; Queralt, E. PP2A-Cdc55 phosphatase regulates actomyosin ring contraction and septum formation during cytokinesis. Cell. Mol. Life Sci. 2022, 79, 165. [Google Scholar] [CrossRef] [PubMed]
- Teh, E.M.; Chai, C.C.; Yeong, F.M. Retention of Chs2p in the ER requires N-terminal CDK1-phosphorylation sites. Cell Cycle 2009, 8, 2964–2974. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chin, C.F.; Bennett, A.M.; Ma, W.K.; Hall, M.C.; Yeong, F.M. Dependence of Chs2 ER export on dephosphorylation by cytoplasmic Cdc14 ensures that septum formation follows mitosis. Mol. Biol. Cell 2012, 23, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Jakobsen, M.K.; Cheng, Z.; Lam, S.K.; Roth-Johnson, E.; Barfield, R.M.; Schekman, R. Phosphorylation of Chs2p regulates interaction with COPII. J. Cell Sci. 2013, 126, 2151–2156. [Google Scholar] [CrossRef] [Green Version]
- Meitinger, F.; Palani, S.; Pereira, G. The power of MEN in cytokinesis. Cell Cycle 2012, 11, 219–228. [Google Scholar] [CrossRef] [Green Version]
- Foltman, M.; Filali-Mouncef, Y.; Crespo, D.; Sanchez-Diaz, A. Cell polarity protein Spa2 coordinates Chs2 incorporation at the division site in budding yeast. PLoS Genet. 2018, 30, e1007299. [Google Scholar] [CrossRef] [Green Version]
- Devrekanli, A.; Foltman, M.; Roncero, C.; Sanchez-Diaz, A.; Labib, K. Inn1 and Cyk3 regulate chitin synthase during cytokinesis in budding yeasts. J. Cell Sci. 2012, 125, 5453–5466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foltman, M.; Molist, I.; Arcones, I.; Sacristan, C.; Filali-Mouncef, Y.; Roncero, C.; Sanchez-Diaz, A. Ingression Progression Complexes Control Extracellular Matrix Remodelling during Cytokinesis in Budding Yeast. PLoS Genet. 2016, 12, e1005864. [Google Scholar] [CrossRef] [Green Version]
- Hernández-González, M.; Bravo-Plaza, I.; Pinar, M.; de Ríos, V.L.; Arst, H.N., Jr.; Peñalva, M.A. Endocytic recycling via the TGN underlies the polarized hyphal mode of life. PLoS Genet. 2018, 14, e1007291. [Google Scholar] [CrossRef] [Green Version]
- Verdín, J.; Sánchez-León, E.; Rico-Ramírez, A.M.; Martínez-Núñez, L.; Fajardo-Somera, R.A.; Riquelme, M. Off the wall: The rhyme and reason of Neurospora crassa hyphal morphogenesis. Cell Surf. 2019, 5, 100020. [Google Scholar] [CrossRef]
- Martín-Urdíroz, M.; Madrid, M.P.; Roncero, M.I. Role of chitin synthase genes in Fusarium oxysporum. Microbiology 2004, 150, 3175–3187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schuster, M.; Treitschke, S.; Kilaru, S.; Molloy, J.; Harmer, N.J.; Steinberg, G. Myosin-5, kinesin-1 and myosin-17 cooperate in secretion of fungal chitin synthase. EMBO J. 2012, 31, 214–227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schuster, M.; Martin-Urdiroz, M.; Higuchi, Y.; Hacker, C.; Kilaru, S.; Gurr, S.J.; Steinberg, G. Co-delivery of cell-wall-forming enzymes in the same vesicle for coordinated fungal cell wall formation. Nat. Microbiol. 2016, 1, 16149. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez, N.; Roncero, C. Chitin Synthesis in Yeast: A Matter of Trafficking. Int. J. Mol. Sci. 2022, 23, 12251. https://doi.org/10.3390/ijms232012251
Sánchez N, Roncero C. Chitin Synthesis in Yeast: A Matter of Trafficking. International Journal of Molecular Sciences. 2022; 23(20):12251. https://doi.org/10.3390/ijms232012251
Chicago/Turabian StyleSánchez, Noelia, and César Roncero. 2022. "Chitin Synthesis in Yeast: A Matter of Trafficking" International Journal of Molecular Sciences 23, no. 20: 12251. https://doi.org/10.3390/ijms232012251






