Imaging and Genetic Tools for the Investigation of the Endocannabinoid System in the CNS
Abstract
:1. The Endocannabinoid System (ECS)
1.1. Cannabinoid Receptors
Intracellular Organelle-Associated CB1R
1.2. Endocannabinoids: Endogenous CBR Ligands
1.3. The Enzymes
2. Imaging the ECS
2.1. The Most Used Tools in ECS Imaging
2.2. CB1R Imaging
| Technique | Advantages | Disadvantages | References |
|---|---|---|---|
| Radioligands | |||
| [18F] radioactive ligands coupled with PET 1 and autoradiography |
|
| [54,55,56] |
| [11C] radioactive ligands coupled with PET 1 and autoradiography |
|
| [57,72] |
| Optical microscopy | |||
| Fluorescence light microscopy (immunocytochemistry and immunohistochemistry) |
|
| [61,62] |
| Fluorescent probes |
|
| [47] |
| FRET 2 |
|
| [48,63] |
| Electron microscopy | |||
| TEM 3 |
|
| [12,65,66,67,68] |
| Super-resolution microscopy | |||
| STORM 4 |
|
| [69,70,71] |
2.3. CB2R
| Technique | Advantages | Disadvantages | References |
|---|---|---|---|
| Radioligands | |||
| [18F] and [11C] radioactive ligands coupled with PET 1 and autoradiography |
|
| [74,75] |
| Optical microscopy | |||
| Fluorescence light microscopy (immunocytochemistry and immunohistochemistry) |
|
| [76,77] |
| Fluorescent probes |
|
| [78,79,80] |
| FRET 2 |
|
| [48,81] |
| Electron microscopy | |||
| TEM 3 |
|
| [82,83,84] |
2.4. Imaging the Endocannabinoids
2.5. Imaging eCB Metabolic Enzymes
| Technique | Advantages | Disadvantages | References |
|---|---|---|---|
| Radioligands | |||
| [18F] and [11C] radioactive ligands coupled with PET 1 and autoradiography |
|
| [90,91,92] |
| Optical microscopy | |||
| Fluorescence light microscopy (immunocytochemistry and immunohistochemistry) |
|
| [99,100] |
| Fluorescent probes |
|
| [96,97,98] |
| Electron microscopy | |||
| TEM 2 |
|
| [102,103] |
3. The Best Genetic Models to Investigate the ECS
3.1. C. elegans as a Model
3.1.1. The Endocannabinoid System in C. elegans
3.1.2. Genetic Approaches
3.2. Zebrafish as a Model for ECS Studies
3.2.1. The Endocannabinoid System in Zebrafish
3.2.2. Genetic Approaches for ECS Manipulation in Zebrafish
3.2.3. CB1R Models in Zebrafish
| Target Sequence of cnr1 | Annotations | References |
|---|---|---|
| 5′-CGGACTTTGAGGCCGGGAACAGCAT-3′ | Translation-blocking | [127] |
| 5′-CTAGAGGAAACCTGTCGGAGGAAAT-3′ | Translation-blocking | [127,139] |
| 5′-GAATGACTACGCTTACATGGACATC-3′ | Target the 5′UTR | [132] |
| 5′-AACAGCATGGTCAGAGATGCTCTAG-3′ | Translation-blocking | [132] |
| 5′-GTGCTATCAACAACATACCTTTGTG-3 | Splice-blocking | [133,145] |
| 5′-CTTTGAGGCCGGGAACAGCATGGTC-3 | Splice-blocking | [145] |
| 5′-GAACAGCATGGTCAGAGATGCTCTA-3′ | Translation-blocking | [136] |
| 5′-TCAGAACCATCACCTCCG-3′ 5′-TCAGAACCATCACCTCCG-3′ | Target first exon | [146] |
3.2.4. CB2R Models in Zebrafish
| Target Sequence of cnr2 | Annotations | References |
|---|---|---|
| 5′-CTGCTCTTGTGTGGTCAAAACCATG-3′ | CB2R AUG | [132] |
| 5′-ATGGCGTTTACGGGCTCTGT-3′ | 5′ end of exon 2 | [138] |
| 5′-GCCATGAAACAAACAGTACCTGTGG-3′ | Splice-blocking | [135,145] |
| 5′-GTTCCAGTTTGTTCTCCATTTTCCC-3′ | Translation-blocking | [145] |
3.3. Mouse Models
3.3.1. CB1R Mouse Models
| Genetic Background | General Phenotype | Outcome | References |
|---|---|---|---|
| Spontaneous Behaviour | |||
| CD1 (Ledent group) | Significantly higher anxiety-like and aggressive behaviour, increased affected maternal care | Increased locomotor activity and time spent exploring unknown objects; decreased spontaneous alteration in the Y-maze | [154] |
| Increased anxiety-like behaviour | [170] | ||
| Increased AMT 1 and FAAH 2 activity with age; mild-anxiety-like behaviour of young mice compared to old mice | [152] | ||
| Increased anxiety-like effect under unfamiliar environment | [171] | ||
| Increased anxiety-like behaviour in the elevated plus maze | [155] | ||
| High level of anxiety with different types of anxiogenic stimuli under unfamiliar conditions | [153] | ||
| No significant alterations in anxiety-like behaviours under total darkness conditions | [172] | ||
| Delayed pup retrieval and fewer ultrasonic vocalizations | [173] | ||
| Higher levels of offensive aggression when housed in group | [174] | ||
| C57BL/6J (Zimmer group) | Hypoactive, increased mortality, reduced anxiety | Reduced locomotion and rearing in open-field test | [158] |
| Increased mortality rate and ring catalepsy; reduced locomotor activity and hypoalgesia | [142] | ||
| Less burying behaviour and fewer contacts with the probe in the shock-probe burying test | [175] | ||
| C57BL/6N (Lutz group) | Aversiveness-dependent anxiogenic-like phenotype and acute fear response, enhanced contextual fear memory, increased wakefulness | Reduced spontaneous caloric intake and decreased body weight | [176] (p. 200) |
| Sustained fear response only after application of an intense foot shock (0.7 mA and 1.5 mA) | [166] | ||
| Lack of within-session extinction during permanent tones and repeated tone presentations at variable intervals | [177] | ||
| Decreased distance covered in the active and in the inactive phases of the cycle on wheel running activity | [178] | ||
| Disrupted classical fear conditioning pattern by favouring passive responses | [179] | ||
| Increased fear expression abolished by repeated social stress | [180] | ||
| Enhanced freezing levels in the conditioning context and increased contextual fear after high-intensity conditioning foot shock (1.5 mA) | [181] | ||
| Increased cortical excitability and reduced NREM 3 sleep and NREM 3 bout duration | [182] | ||
| More time awake and less time in NREM 3 and REM 4; slower EEG 5 theta rhythm during REM 4 and habituated more rapidly to the arousing effect of the cage-switch test | [183] | ||
| Hypoactivity, impaired eyeblink, and normal cerebellum-dependent locomotor coordination and learning | [162] | ||
| In vivo response to drugs | |||
| CD1 (Ledent group) | Insensitive to THC and CBD, sensitive to nicotine, ethanol, cocaine, and amphetamine | Insensitive to THC-induced antinociceptive properties, reduced horizontal activity, and decreased rectal temperature | [154] |
| Sensitive to SR141716A anxiety reduction | [155] | ||
| Enhanced nicotine-induced antinociceptive effects and absent rewarding effects | [184] | ||
| Decreased ethanol self-administration with increased sensitivity to its acute intoxicating effects | [185] | ||
| Decreased ethanol-induced conditional place preference and increased striatal dopamine D2 receptors | [186] | ||
| Reduced ethanol self-administration and ethanol-conditioned place preference | [187] | ||
| Decreased locomotor responses to cocaine and D-amphetamine | [188] | ||
| Insensitive to CBD-induced anxiolytic actions | [157] | ||
| C57BL/6J (Zimmer group) | Insensitive to cannabinoid drugs and sensitive to cocaine and ethanol; absence of ethanol withdrawal | Insensitive to THC-induced ring catalepsy, hypomobility, and hypothermia | [142] |
| Insensitive to THC-, WIN 55,212-2-, and methanandamide; disruption in the working memory task | [159] | ||
| No enhancement of growth rates or survival after CP55,940, WIN55,212-2, or 2-AG administration | [189] | ||
| Reduced voluntary alcohol consumption and absent alcohol–dopamine release in the nucleus accumbens | [190] | ||
| Absence of ethanol withdrawal symptoms and of foot-shock stress-induced alcohol preference | [191] | ||
| Reduced ethanol preference and insensitive to SR141716A-induced reduction in ethanol preference in young mice | [192] | ||
| Insensitive to THC- and O-1812-induced decrease in lever press; sensitive to methanandamide-, ethanol-, and morphine-induced decrease in lever press | [193] | ||
| Abolished CP55,940-induced antinociceptive effects and associated motor deficits | [194] | ||
| Absent THC-induced expression of ΔFosB in the striatum | [195] | ||
| Sensitive to the locomotor-stimulant effects of RTI-371 (a cocaine analogue) | [196] | ||
| Absence of ethanol-induced activation of caspase 3 and of reduction in DNMT1 6, DNMT3A 6, and DNA methylation | [197] | ||
| C57BL/6N (Lutz group) | Sensitive to KA 7 and insensitive to THC, reduced sensitivity to rewarding properties. | KA 7 injection induced more severe seizures and decreased survival rate | [164] |
| Decreased sucrose consumption under operant conditions or a two-bottle free choice; decreased sensitivity to rewarding properties of sucrose | [198] | ||
| Insensitive to cannabinoid-induced neurosphere generation | [199] | ||
| Insensitive to THC-induced tetrad effects | [200] | ||
| Insensitive to THC-induced increase in pregnenolone in the nucleus accumbens | [201] | ||
| Learning and aging | |||
| CD1 (Ledent group) | Defective neurogenesis, increased aggressiveness and conditioned responses | Increased aggressive response, higher sensitivity to exhibit depressive-like responses, and increased conditioned responses in the active avoidance model | [202] |
| Enhanced retention of the habituation task | [203] | ||
| Reduction in bromodeoxyuridine-labelled cells in dentate gyrus and subventricular zone | [204] | ||
| C57BL/6J (Zimmer group) | Impaired extinction, age-related memory decline, accelerated decline in cognitive function | Increased perseverance in a reversal task | [159] |
| Similar or better performance on 6–7-week-old mice and worst performance on 3–5-month-old mice in several learning and memory paradigms | [205] | ||
| Impaired extinction process in the Morris water maze using a spaced extinction procedure | [206] | ||
| Enhanced habituation (non-associative learning) displayed with decreased number of ambulations | [207] | ||
| Impaired delay eyeblink conditioning performance | [208] | ||
| Deficits in a sensory-selective reinforcer devaluation task | [209] | ||
| Improved performance in the Morris water maze at 6 weeks old and inferior performance at 12 months old | [161] | ||
| Superior learning ability in the eight-arm radial maze at 2 months old and impaired performance at 12 months old | [160] | ||
| C57BL/6N (Lutz group) | Impaired extinction of aversive memories and of fear, impaired habituation | Strongly impaired short-term and long-term extinction in auditory fear-conditioning tests, with unaffected memory acquisition and consolidation | [163] |
| Normal extinction of the stimulus–response association in an appetitively motivated learning task | [165] | ||
| Severely impaired in extinction and in habituation of the fear response to a tone | [166] | ||
3.3.2. Conditional CB1R Deletions
| Strain Designation | Cell-Type-Specific Deletion | Outcome | References |
|---|---|---|---|
| CB1RCaMKIIa-Cre | Forebrain principal neurons | More severe KA 1-induced seizures and decreased survival | [36,164] (p. 200) |
| Sustained fear response only after intense electric shock | [214] | ||
| CB1RDlx5/6-Cre | GABAergic neurons | No change in KA 1-induced seizures | [213] |
| Impaired target selection of cortical GABAergic interneurons | [217] | ||
| CB1R is localized on presynaptic boutons of about 30% in alBNST 2 | [218] | ||
| Neuronal loss and increased neuroinflammation in the hippocampus | [161] | ||
| Abolished anxiogenic effect under a high-dose treatment of CB1R agonist (CP-55940) | [219] | ||
| Conserved impairment of SWM 3 and in vivo LTD 4 of synaptic strength at CA3-CA1 synapses caused by an acute exposure to exogenous cannabinoids | [36] | ||
| Abolished CB1R agonist (CP-55940)-induced increase in HVS 5 | [220] | ||
| Insensitive to quinolinic-acid-induced neurotoxicity | [136] | ||
| Insensitive to THC-induced memory impairment in novel object recognition | [221,222] | ||
| CB1RNex-Cre | Glutamatergic cortical neurons | Aberrant fasciculation and pathfinding in both corticothalamic and thalamocortical axons | [223] |
| Absence of neuronal loss and increased neuroinflammation in the hippocampus | [161] | ||
| Unbalanced neurogenic fate determination | [224] | ||
| Conserved impairment of SWM 3 and in vivo LTD 4 of synaptic strength at CA3-CA1 synapses caused by an acute exposure to exogenous cannabinoids | [36] | ||
| Reduced decrease in fast ECoG 6 oscillations and stronger cannabinoid-induced increase in HVS 5 | [220] | ||
| Sensitive to excitotoxic damage induced with quinolinic acid administration | [136] | ||
| CB1RD1-Cre | Neurons expressing dopamine D1 receptors | Insensitive to THC-induced catalepsy | [200] |
| Abolished CB1R agonist (CP-55940)-induced increase in HVS 5 | [220] | ||
| CB1Rsns-Cre | Dorsal root ganglia neurons | Reduced LTD 4 at dorsal horn nociceptor synapses | [215] |
| CB1RGabra6-Cre | Cerebellar granule cells | Abolition of short-term plasticity at parallel fibre synapses and lack of LTD | [225] |
| Activated cerebellar microglia and increased cerebellar neuroinflammation | [226] | ||
| Normal eyeblink conditioning and normal cerebellum-dependent locomotor coordination and learning | [162] | ||
| CB1RGfap-CreERT2 | Astrocytes | Abolished impairment of SWM 3 and in vivo LTD 4 of synaptic strength at CA3-CA1 synapses | [36] |
| Impaired object recognition memory and decreased LTP 7 at CA3-CA1 synapses | [37] | ||
| CB1RSim1-Cre | Neurons expressing Sim 1 8 (hypothalamus and mediobasal amygdala) | Increased locomotor activity in open field, unconditioned anxiety, and cued fear expression under basal conditions | [216] |
| CB1RTph2-CreERT2 | Central serotoninergic neurons | Anxiety and decreased cued fear expression | [180] |
3.3.3. MtCB1R Models
| Genetic Background | General Phenotype | Outcome | References |
|---|---|---|---|
| C57BL/6N DN22-CB1R 1-KI | Specific impairment of mtCB1R and of cannabinoid effects on mitochondrial dynamics but no influence on CB1R general functions | mtCB1R decreases synaptic activity and induces catalepsy | [27] |
| mtCB1R mediates corticosterone-induced memory impairment | [26] | ||
| Astroglial mtCB1R reduces mitochondrial respiration with complex I destabilization | [25] | ||
| C57BL/6N GFAP 2-CB1R-KO | Specific impairment of mtCB1R and of cannabinoid effects on mitochondrial dynamics but no influence on CB1R general functions | Astroglial mtCB1R reduces mitochondrial respiration with complex I destabilization | [25] |
3.3.4. CB2R Mouse Models
| Genetic Background | General Phenotype | Outcome | References |
|---|---|---|---|
| Neurodegeneration, neuroinflammation, and synaptic plasticity | |||
| C57BL/6J CB2R −/− Buk | Increased neurodegenerative symptoms, impaired neuroprogenitor proliferation, impairment of neuroprotective proteins | Protective role for CB2R in experimental autoimmune encephalitis | [231] |
| Augmented multiple sclerosis severity (similar to pharmacological inhibition) | [232] | ||
| Link between CB2R and the onset of Huntington’s disease | [233] | ||
| Amelioration of Alzheimer’s disease-like pathology | [234] | ||
| CB2R-mediated modulation of cocaine action | [235] | ||
| Incomplete activation of microglia in neuroinflammation | [236] | ||
| C57BL/6J CB2R −/− Del | Higher corticosterone levels after stress in the prefrontal cortex (PFC), higher hippocampal and PFC neuron excitability | CB2R activation mediates PFC neuron excitability | [237] |
| Chronic CB2R activation in the hippocampus increases excitatory synaptic transmission | [238] | ||
| C57BL/6J CB2R−/− Lop | Increased neurodegenerative symptoms, agonist treatment can reduce inflammatory phenotypes | Development of a new reporter mouse line and involvement in neuroinflammation | [230] |
| Tau protein levels increase CB2R during early stages of neurodegeneration | [239] | ||
| CB2R depletion reduces inflammatory pain behaviours and markers of neuroinflammation | [240] | ||
| Nociception and neuropathic pain | |||
| C57BL/6J CB2R −/− Buk | Normal cannabidiol analgesia, altered opioid receptor expression | Neuropathic pain is mediated by glycinergic neurons | [241] |
| CB2R mediates analgesic effects in neuroinflammation, neuropathies | [242] | ||
| C57BL/6J CB2R −/− Del | Reduced morphine analgesia, no effect of AM1710 on paclitaxel-induced allodynia | Chronic CB2R activation reverses paclitaxel-induced neuropathy | [243] |
| Activation of CB2R alone or with CB1R decreases neuropathic-pain-related behaviour in mice | [244] | ||
| Possible mechanism to suppress chemotherapy-induced neuropathy | [245] | ||
| Behaviour | |||
| C57BL/6J CB2R −/− Buk | Impaired memory consolidation, schizophrenic behavioural phenotypes | Induces schizophrenia-related behaviours such as locomotor activities, anxiety- and depressive-like behaviours, and cognitive deficits | [246] |
| CB2R role in cognitive processes, particularly in short- and long-term memory | [247] | ||
| CB2R is expressed in red nucleus glutamate receptors and modulates motor behaviour | [248] | ||
| C57BL/6J CB2R −/− Del | Impairment of contextual long-term memory, enhancement of spatial working memory, decrease in neuropathic pain-related behaviour in mice | CB2R plays different roles in regulating memory with different outcomes depending on the brain areas | [249] |
3.3.5. CB1R × B2R Genetic Models
3.3.6. Genetic Mouse Models of eCB Metabolic Enzymes
3.3.7. 2-AG Biosynthesis and Catabolism
| Genetic Background | General Phenotype | Outcome | References |
|---|---|---|---|
| C57BL/6N DAGLα−/− Tanimura | Improved learning habituation, increased seizure risk | Reduced 2-AG; abolished DSE 1 | [258] |
| Abolished DSE 1 at MC-GC 2 | [266] | ||
| Improved odour habituation; enhanced LTP 3 | [267] | ||
| Unchanged CB1R-G protein signalling, compensatory 2-AG synthesis | [268] | ||
| Localization: Mostly post-synaptic | [269] | ||
| C57BL/6 DAGLα−/− Gao | Impaired spatial learning and memory | 80% 2-AG reduction; impaired synaptic plasticity; reduced neurogenesis | [259] |
| Main contribution to brain 2-AG and eicosanoid synthesis | [261] | ||
| Decreased 2-AG; decreased LTD 4; impaired learning and memory | [270] | ||
| 2-AG signals preferentially to neurons in short-distance synaptic regulation | [34] | ||
| Unspecified DAGLα-KOLex; gene trap | No overt phenotype reported | Reduced 2-AG; small AEA reduction; abolished DSI 5 | [260] |
| C57BL/6N DAGLα−/− | Sex-specific pro-anxiety and anhedonia | CB1R-dependent anxiety; anhedonia | [262] |
| C57BL/6J DAGLα−/− | Anxiety and fear similar to CB1R-KO | Increased fear, anxiety; loss of maternal care | [263] |
| Unspecified DAGLαfl/fl | Increased susceptibility to post-traumatic stress | Decreased stress resilience (AAV 6 directed in basolateral amygdala) | [264] |
| 129SvEv × C57BL/6 DAGLβ-GTLex; gene trap | Reduced macrophage response, reduced hepatic eCB | 50% 2-AG reduction; impaired synaptic plasticity; reduced neurogenesis | [259] |
| Negligible contribution to brain 2-AG and eicosanoid synthesis | [261] | ||
| C57BL/6N DAGLβ−/− | No overt phenotype reported | Normal 2-AG; DSE 1 normal | [258] |
| Unspecified DAGLβ-GTLex; gene trap | No overt phenotype reported | Normal 2-AG; small AEA reduction | [260] |
| Unspecified DAGLα-GTLex × DAGLβ-GTLex | Greater 2-AG reduction than single KO | Greater 2-AG reduction than single KO | [260] |
| Unspecified RNAi DAGLα/β | No overt phenotype reported | Equal DAGLα/β contribution to autaptic CA1/3 neuron 2-AG levels | [265] |
| 129SvEv × C57BL/6J MAGL-GTLex; gene trap | Enhanced learning, analgesic CB1R agonist tolerance | CB1R desensitization; altered synaptic plasticity | [271] |
| Altered synaptic plasticity; enhanced LTD 4; enhanced learning | [272] | ||
| CB1R desensitization; prolonged climbing fibre DSE 1 | [273] | ||
| C57BL6/Ntac MAGL−/− | CB1R agonist tolerance, analgesic tolerance | CB1R desensitization; lack of characteristic CB1R effects | [274] |
| C57BL/6 MAGL−/− Taschler | CB1R agonist tolerance, anxious and obsessive-compulsive behaviour | Increased 2-AG; CB1R agonist tolerance; impaired lipolysis | [275] |
| CB1R desensitization in all regions; compensatory serine hydrolase activity | [276] | ||
| CB1R desensitization and disturbed E/I ratio in limbic pathways; stress-like cannabimimetic behaviour | [277] | ||
| C57BL/6N MAGL−/− Uchigashima | CB1R agonist tolerance, analgesic tolerance | Low MAGL expression in MC-GC 2 spines, mostly astrocytic | [266] |
| Unspecified GluN2C:MAGL−/− | No overt phenotype reported | Prolonged 2-AG signalling, but less than total KO | [278] |
| C57BL/6 MAGLlox/lox | Floxed animals with no overt phenotype. Specific effect for neuronal, astrocytic cKO. No effect in microglial cKO | Neuron and astrocyte MAGL coordinate CB1R signalling termination; neuron-2-AG/astrocyte-AA 7 shuttle. | [40] |
| Neuron and astrocyte MAGL both contribute to terminate synaptic eCB signalling; differential effects in different fibre types/synaptic events | [38] | ||
| Neuron and astrocyte MAGL both contribute to terminate synaptic eCB signalling; effect restricted to molecular layer | [39] | ||
| Increased 2-AG; decreased AA 7 and PGE/F 8 (global KO) | [85] | ||
| C57BL/6N MAGLflox/flox | Decreased microglial inflammatory response | Cell-type-specific change in gene expression, astrocytic 2-AG promotes immune vigilance in microglia | [279] |
| C57BL6/J CaMKII:MAGLTg | Lean and hypothermic response to hypercaloric diet | MAGL overexpression; decreased 2-AG correlated to weight loss and hyperthermia | [280] |
| C57BL6/J GFAP:MAGL−/− | Decreased microglial inflammatory response | Reduced neuroinflammation independent of CB1R and total 2-AG | [281] |
3.3.8. AEA Synthesis and Catabolism
| Genetic Background | General Phenotype | Outcome | References |
|---|---|---|---|
| 129SvJ × C57BL/6 NAPE-PLD−/− Cravatt | Healthy and viable through lifespan, no significant AEA decrease | Decreased saturated and monounsaturated NAEs, no change in AEA | [284] |
| Subcellular localization: Preferentially in dendrites | [269] | ||
| Lower AEA; higher DHA/DHEA; significant effect of dietary fatty acids | [291] | ||
| AEA signals preferentially to astrocytes; astrocytic Ca2+ mobilization and synaptic plasticity | [34] | ||
| C57BL/6 × 129SV NAPE-PLD−/− Palmiter | Healthy and viable throughout, significant AEA decrease | Decreased AEA/NAE levels, ABDH4 as main compensatory synthesizer | [285] |
| Region- and lipid-specific NAE/MAG alteration; decreased NAE | [255] | ||
| C57BL/6 NAPE-PLD−/− Tsuboi | Healthy and viable throughout, significant AEA decrease | Decreased AEA/NAE levels, compensatory synthesis pathways involved | [252] |
| Decreased AEA/NAE levels, compensatory synthesis pathways involved | [292] | ||
| C57BL/6 GDE1−/− | Healthy and viable through lifespan, no significant AEA decrease | Unchanged NAE/AEA levels | [287] |
| C57BL/6 GDE1−/− × NAPE-PLD−/− | Healthy and viable through lifespan, no significant AEA decrease | Unchanged NAE/AEA levels | [287] |
| 129SvEv × C57BL/6 ABHD4-KO | Healthy and viable through lifespan, no significant AEA decrease | No significant AEA decrease; decreased GP/pNAPE | [288] |
| 129SvJ × C57BL/6 FAAH−/− | Significant AEA increase, pain and inflammation resistance, super sensitivity to AEA treatment | Increased AEA; CB1R-dependent sensitivity to AEA | [293] |
| Increased GABAergic inhibition of synaptic transmission | [294] | ||
| Decreased AEA transport; diffusion- and transporter-mediated systems | [295] | ||
| CB1R-mediated hypoalgesia | [296] | ||
| Increased neurogenesis (AEA-dependent) | [297] | ||
| Decreased post-mortem AEA/EA accumulation | [298] | ||
| Increased astrogliogenesis (CB1R-dependent) | [199] | ||
| Alternative phosphocholine-NAE metabolic route | [299] | ||
| Accumulation of N-acyltaurines, which act as TRPV1/4 agonists | [300] | ||
| Resistance to acetaminophen analgesia, TRPV1-dependant | [301] | ||
| NAE/NAT accumulation in CNS (primarily long-chain saturated forms) | [302] | ||
| Altered iLTD | [303] | ||
| Increased TRPV1 activity; modulated glutamate release | [304] | ||
| Hyperalgesia to certain types of pain; analgesia to others | [305] | ||
| Increased AEA and phosphamides; no changes in PGE/F effect | [85] | ||
| 129SvJ × C57BL/6 FAAH−/− × Eno2:FAAH | Wild-type pain responses, resistance to inflammation | Increased AEA in CNS; no change in periphery | [306] |
4. Concluding Remarks
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dudok, B.; Soltesz, I. Imaging the endocannabinoid signaling system. J. Neurosci. Methods 2021, 367, 109451. [Google Scholar] [CrossRef]
- Cermenati, G.; Mitro, N.; Audano, M.; Melcangi, R.C.; Crestani, M.; De Fabiani, E.; Caruso, D. Lipids in the nervous system: From biochemistry and molecular biology to pathophysiology. Biochim. Et Biophys. Acta (BBA) Mol. Cell Biol. Lipids 1851, 1, 51–60. [Google Scholar] [CrossRef]
- Cristino, L.; Bisogno, T.; Di Marzo, V. Cannabinoids and the expanded endocannabinoid system in neurological disorders. Nat. Rev. Neurol. 2019, 16, 9–29. [Google Scholar] [CrossRef]
- Zou, M.; Li, D.; Li, L.; Wu, L.; Sun, C. Role of the endocannabinoid system in neurological disorders. Int. J. Dev. Neurosci. 2019, 76, 95–102. [Google Scholar] [CrossRef]
- Puighermanal, E.; Busquets-Garcia, A.; Maldonado, R.; Ozaita, A. Cellular and intracellular mechanisms involved in the cognitive impairment of cannabinoids. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 3254–3263. [Google Scholar] [CrossRef]
- Kaczocha, M.; Haj-Dahmane, S. Mechanisms of endocannabinoid transport in the brain. Br. J. Pharmacol. 2021, 179, 4300–4310. [Google Scholar] [CrossRef]
- Lu, H.-C.; Mackie, K. Review of the Endocannabinoid System. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2020, 6, 607–615. [Google Scholar] [CrossRef]
- Lowe, H.; Toyang, N.; Steele, B.; Bryant, J.; Ngwa, W. The Endocannabinoid System: A Potential Target for the Treatment of Various Diseases. Int. J. Mol. Sci. 2021, 22, 9472. [Google Scholar] [CrossRef]
- Russo, E.B. Taming THC: Potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. Br. J. Pharmacol. 2011, 163, 1344–1364. [Google Scholar] [CrossRef]
- Iozzo, M.; Sgrignani, G.; Comito, G.; Chiarugi, P.; Giannoni, E. Endocannabinoid System and Tumour Microenvironment: New Intertwined Connections for Anticancer Approaches. Cells 2021, 10, 3396. [Google Scholar] [CrossRef]
- Katona, I. Endocannabinoid receptors: CNS localization of the CB1 cannabinoid receptor. Curr. Top. Behav. Neurosci. 2009, 1, 65–86. [Google Scholar] [CrossRef] [PubMed]
- Bénard, G.; Massa, F.; Puente, N.; Lourenço, J.; Bellocchio, L.; Soria-Gómez, E.; Matias, I.; Delamarre, A.; Metna-Laurent, M.; Cannich, A.; et al. Mitochondrial CB1 receptors regulate neuronal energy metabolism. Nat. Neurosci. 2012, 15, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.-C.; Mackie, K. An Introduction to the Endogenous Cannabinoid System. Biol. Psychiatry 2015, 79, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Zou, S.; Kumar, U. Cannabinoid Receptors and the Endocannabinoid System: Signaling and Function in the Central Nervous System. Int. J. Mol. Sci. 2018, 19, 833. [Google Scholar] [CrossRef]
- Ramírez-Orozco, R.E.; García-Ruiz, R.; Morales, P.; Villalón, C.M.; Villafán-Bernal, J.R.; Marichal-Cancino, B.A. Potential metabolic and behavioural roles of the putative endocannabinoid receptors GPR18, GPR55 and GPR119 in feeding. Curr. Neuropharmacol. 2019, 17, 947–960. [Google Scholar] [CrossRef]
- Iglesias, L.P.; Fernandes, H.B.; de Miranda, A.S.; Perez, M.M.; Faccioli, L.H.; Sorgi, C.A.; Bertoglio, L.J.; Aguiar, D.C.; Wotjak, C.T.; Moreira, F.A. TRPV1 modulation of contextual fear memory depends on stimulus intensity and endocannabinoid signaling in the dorsal hippocampus. Neuropharmacology 2023, 224, 109314. [Google Scholar] [CrossRef]
- Muller, C.; Morales, P.; Reggio, P.H. Cannabinoid Ligands Targeting TRP Channels. Front. Mol. Neurosci. 2019, 11, 487. [Google Scholar] [CrossRef]
- Iannotti, F.A.; Vitale, R.M. The Endocannabinoid System and PPARs: Focus on Their Signalling Crosstalk, Action and Transcriptional Regulation. Cells 2021, 10, 586. [Google Scholar] [CrossRef]
- Battista, N.; Di Tommaso, M.; Bari, M.; Maccarrone, M. The endocannabinoid system: An overview. Front. Behav. Neurosci. 2012, 6, 9. [Google Scholar] [CrossRef]
- Ryberg, E.; Larsson, N.; Sjögren, S.; Hjorth, S.; Hermansson, N.-O.; Leonova, J.; Elebring, T.; Nilsson, K.; Drmota, T.; Greasley, P.J. The orphan receptor GPR55 is a novel cannabinoid receptor. Br. J. Pharmacol. 2007, 152, 1092–1101. [Google Scholar] [CrossRef]
- Yang, H.; Zhou, J.; Lehmann, C. GPR55—A putative “type 3” cannabinoid receptor in inflammation. J. Basic Clin. Physiol. Pharmacol. 2015, 27, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Morales, P.; Lago-Fernandez, A.; Hurst, D.P.; Sotudeh, N.; Brailoiu, E.; Reggio, P.H.; Abood, M.E.; Jagerovic, N. Therapeutic Exploitation of GPR18: Beyond the Cannabinoids? J. Med. Chem. 2020, 63, 14216–14227. [Google Scholar] [CrossRef] [PubMed]
- Robledo-Menendez, A.; Vella, M.; Grandes, P.; Soria-Gomez, E. Cannabinoid control of hippocampal functions: The where matters. FEBS J. 2022, 289, 2162–2175. [Google Scholar] [CrossRef] [PubMed]
- Hebert-Chatelain, E.; Desprez, T.; Serrat, R.; Bellocchio, L.; Soria-Gomez, E.; Busquets-Garcia, A.; Zottola, A.C.P.; Delamarre, A.; Cannich, A.; Vincent, P.; et al. A cannabinoid link between mitochondria and memory. Nature 2016, 539, 555–559. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Blasco, D.; Busquets-Garcia, A.; Hebert-Chatelain, E.; Serrat, R.; Vicente-Gutierrez, C.; Ioannidou, C.; Gómez-Sotres, P.; Lopez-Fabuel, I.; Resch-Beusher, M.; Resel, E.; et al. Glucose metabolism links astroglial mitochondria to cannabinoid effects. Nature 2020, 583, 603–608. [Google Scholar] [CrossRef]
- Skupio, U.; Welte, J.; Serrat, R.; Eraso-Pichot, A.; Julio-Kalajzić, F.; Gisquet, D.; Cannich, A.; Delcasso, S.; Matias, I.; Fundazuri, U.B.; et al. Mitochondrial cannabinoid receptors gate corticosterone impact on novel object recognition. Neuron 2023, 111, 1887–1897.e6. [Google Scholar] [CrossRef]
- Soria-Gomez, E.; Zottola, A.C.P.; Mariani, Y.; Desprez, T.; Barresi, M.; Río, I.B.-D.; Muguruza, C.; Le Bon-Jego, M.; Julio-Kalajzić, F.; Flynn, R.; et al. Subcellular specificity of cannabinoid effects in striatonigral circuits. Neuron 2021, 109, 1513–1526.e11. [Google Scholar] [CrossRef]
- Pertwee, R.G. The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: Δ9-tetrahydrocannabinol, cannabidiol and Δ9-tetrahydrocannabivarin. Br. J. Pharmacol. 2008, 153, 199–215. [Google Scholar] [CrossRef]
- Simard, M.; Archambault, A.-S.; Lavoie, J.-P.C.; Dumais, É.; Di Marzo, V.; Flamand, N. Biosynthesis and metabolism of endocannabinoids and their congeners from the monoacylglycerol and N-acyl-ethanolamine families. Biochem. Pharmacol. 2022, 205, 115261. [Google Scholar] [CrossRef]
- Baggelaar, M.P.; Maccarrone, M.; van der Stelt, M. 2-Arachidonoylglycerol: A signaling lipid with manifold actions in the brain. Prog. Lipid Res. 2018, 71, 1–17. [Google Scholar] [CrossRef]
- Maccarrone, M. Phytocannabinoids and endocannabinoids: Different in nature. Rend. Lincei. Sci. Fis. Nat. 2020, 31, 931–938. [Google Scholar] [CrossRef]
- Nomura, D.K.; Morrison, B.E.; Blankman, J.L.; Long, J.Z.; Kinsey, S.G.; Marcondes, M.C.G.; Ward, A.M.; Hahn, Y.K.; Lichtman, A.H.; Conti, B.; et al. Endocannabinoid Hydrolysis Generates Brain Prostaglandins That Promote Neuroinflammation. Science 2011, 334, 809–813. [Google Scholar] [CrossRef] [PubMed]
- Fisette, A.; Tobin, S.; Décarie-Spain, L.; Bouyakdan, K.; Peyot, M.-L.; Madiraju, S.M.; Prentki, M.; Fulton, S.; Alquier, T. α/β-Hydrolase Domain 6 in the Ventromedial Hypothalamus Controls Energy Metabolism Flexibility. Cell Rep. 2016, 17, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Noriega-Prieto, J.A.; Falcón-Moya, R.; Eraso-Pichot, A.; Fundazuri, U.B.; Guttipatti, P.; Belisle, L.; Rodríguez-Moreno, A.; van der Stelt, M.; Cheer, J.; Marsicano, G.; et al. Distinct endocannabinoids specifically signal to astrocytes and neurons. bioRxiv 2023. [Google Scholar] [CrossRef]
- Tsuboi, K.; Uyama, T.; Okamoto, Y.; Ueda, N. Endocannabinoids and related N-acylethanolamines: Biological activities and metabolism. Inflamm. Regen. 2018, 38, 28. [Google Scholar] [CrossRef]
- Han, J.; Kesner, P.; Metna-Laurent, M.; Duan, T.; Xu, L.; Georges, F.; Koehl, M.; Abrous, D.N.; Mendizabal-Zubiaga, J.; Grandes, P.; et al. Acute Cannabinoids Impair Working Memory through Astroglial CB1 Receptor Modulation of Hippocampal LTD. Cell 2012, 148, 1039–1050. [Google Scholar] [CrossRef]
- Robin, L.M.; da Cruz, J.F.O.; Langlais, V.C.; Martin-Fernandez, M.; Metna-Laurent, M.; Busquets-Garcia, A.; Bellocchio, L.; Soria-Gomez, E.; Papouin, T.; Varilh, M.; et al. Astroglial CB1 Receptors Determine Synaptic D-Serine Availability to Enable Recognition Memory. Neuron 2018, 98, 935–944.e5. [Google Scholar] [CrossRef]
- Liu, X.; Chen, Y.; Vickstrom, C.R.; Li, Y.; Viader, A.; Cravatt, B.F.; Liu, Q.-S. Coordinated regulation of endocannabinoid-mediated retrograde synaptic suppression in the cerebellum by neuronal and astrocytic monoacylglycerol lipase. Sci. Rep. 2016, 6, 35829. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, X.; Vickstrom, C.R.; Liu, M.J.; Zhao, L.; Viader, A.; Cravatt, B.F.; Liu, Q.-S. Neuronal and Astrocytic Monoacylglycerol Lipase Limit the Spread of Endocannabinoid Signaling in the Cerebellum. Eneuro 2016, 3, ENEURO.0048-16.2016. [Google Scholar] [CrossRef]
- Viader, A.; Blankman, J.L.; Zhong, P.; Liu, X.; Schlosburg, J.E.; Joslyn, C.M.; Liu, Q.-S.; Tomarchio, A.J.; Lichtman, A.H.; Selley, D.E.; et al. Metabolic Interplay between Astrocytes and Neurons Regulates Endocannabinoid Action. Cell Rep. 2015, 12, 798–808. [Google Scholar] [CrossRef]
- Chen, C. Inhibiting degradation of 2-arachidonoylglycerol as a therapeutic strategy for neurodegenerative diseases. Pharmacol. Ther. 2023, 244, 108394. [Google Scholar] [CrossRef]
- Maccarrone, M. Metabolism of the Endocannabinoid Anandamide: Open Questions after 25 Years. Front. Mol. Neurosci. 2017, 10, 166. [Google Scholar] [CrossRef]
- Martin, G.G.; Seeger, D.R.; McIntosh, A.L.; Milligan, S.; Chung, S.; Landrock, D.; Dangott, L.J.; Golovko, M.Y.; Murphy, E.J.; Kier, A.B.; et al. Sterol Carrier Protein-2/Sterol Carrier Protein-x/Fatty Acid Binding Protein-1 Ablation Impacts Response of Brain Endocannabinoid to High-Fat Diet. Lipids 2019, 54, 583–601. [Google Scholar] [CrossRef]
- Kaczocha, M.; Glaser, S.T.; Maher, T.; Clavin, B.; Hamilton, J.; Rourke, J.O.; Rebecchi, M.; Puopolo, M.; Owada, Y.; Thanos, P.K. Fatty Acid Binding Protein Deletion Suppresses Inflammatory Pain through Endocannabinoid/N-Acylethanolamine-Dependent Mechanisms. Mol. Pain 2015, 11, 52. [Google Scholar] [CrossRef]
- Sulzer, D.; Edwards, R.H. The physiological role of alpha-synuclein and its relationship to Parkinson’s Disease. J. Neurochem. 2019, 150, 475–486. [Google Scholar] [CrossRef]
- Albarran, E.; Sun, Y.; Liu, Y.; Raju, K.; Dong, A.; Li, Y.; Wang, S.; Südhof, T.C.; Ding, J.B. Postsynaptic synucleins mediate endocannabinoid signaling. Nat. Neurosci. 2023, 26, 997–1007. [Google Scholar] [CrossRef]
- Martín-Couce, L.; Martín-Fontecha, M.; Palomares, Ó.; Mestre, L.; Cordomí, A.; Hernangomez, M.; Palma, S.; Pardo, L.; Guaza, C.; López-Rodríguez, M.L.; et al. Chemical Probes for the Recognition of Cannabinoid Receptors in Native Systems. Angew. Chem. Int. Ed. 2012, 51, 6896–6899. [Google Scholar] [CrossRef] [PubMed]
- Imperatore, R.; Palomba, L.; Morello, G.; Di Spiezio, A.; Piscitelli, F.; Di Marzo, V.; Cristino, L. Formation of OX-1R/CB1R heteromeric complexes in embryonic mouse hypothalamic cells: Effect on intracellular calcium, 2-arachidonoyl-glycerol biosynthesis and ERK phosphorylation. Pharmacol. Res. 2016, 111, 600–609. [Google Scholar] [CrossRef]
- Zádor, F.; Ötvös, F.; Benyhe, S.; Zimmer, A.; Páldy, E. Inhibition of forebrain μ-opioid receptor signaling by low concentrations of rimonabant does not require cannabinoid receptors and directly involves μ-opioid receptors. Neurochem. Int. 2012, 61, 378–388. [Google Scholar] [CrossRef] [PubMed]
- Lammertsma, A. Radioligand studies: Imaging and quantitative analysis. Eur. Neuropsychopharmacol. 2002, 12, 513–516. [Google Scholar] [CrossRef] [PubMed]
- Gatley, S.J.; Lan, R.; Volkow, N.D.; Pappas, N.; King, P.; Wong, C.T.; Gifford, A.N.; Pyatt, B.; Dewey, S.L.; Makriyannis, A. Imaging the Brain Marijuana Receptor: Development of a Radioligand that Binds to Cannabinoid CB1 Receptors In Vivo. J. Neurochem. 2002, 70, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Cristino, L.; Imperatore, R.; Di Marzo, V. Chapter Four—Techniques for the Cellular and Subcellular Localization of Endocannabinoid Receptors and Enzymes in the Mammalian Brain. In Methods in Enzymology; Reggio, P.H., Ed.; Academic Press: Cambridge, MA, USA, 2017; Volume 593, pp. 61–98. [Google Scholar] [CrossRef]
- Amenta, A.; Caprioglio, D.; Minassi, A.; Panza, L.; Passarella, D.; Fasano, V.; Imperio, D. Recent advances in the development of CB1R selective probes. Front. Nat. Prod. 2023, 2, 1196321. [Google Scholar] [CrossRef]
- Burns, H.D.; Van Laere, K.; Sanabria-Bohórquez, S.; Hamill, T.G.; Bormans, G.; Eng, W.-S.; Gibson, R.; Ryan, C.; Connolly, B.; Patel, S.; et al. [18F]MK-9470, a positron emission tomography (PET) tracer for in vivo human PET brain imaging of the cannabinoid-1 receptor. Proc. Natl. Acad. Sci. USA 2007, 104, 9800–9805. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-P.; Huang, H.-L.; Huang, J.-K.; Hung, M.-S.; Wu, C.-H.; Song, J.-S.; Lee, C.-J.; Yu, C.-S.; Shia, K.-S. Fluorine-18 isotope labeling for positron emission tomography imaging. Direct evidence for DBPR211 as a peripherally restricted CB1 inverse agonist. Bioorganic Med. Chem. 2018, 27, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Takkinen, J.S.; López-Picón, F.R.; Kirjavainen, A.K.; Pihlaja, R.; Snellman, A.; Ishizu, T.; Löyttyniemi, E.; Solin, O.; Rinne, J.O.; Haaparanta-Solin, M. [18F]FMPEP-d2 PET imaging shows age- and genotype-dependent impairments in the availability of cannabinoid receptor 1 in a mouse model of Alzheimer’s disease. Neurobiol. Aging 2018, 69, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, T.; Fujinaga, M.; Shimoda, Y.; Mori, W.; Zhang, Y.; Wakizaka, H.; Ogawa, M.; Zhang, M.-R. Radiosynthesis and evaluation of new PET ligands for peripheral cannabinoid receptor type 1 imaging. Bioorganic Med. Chem. Lett. 2017, 27, 4114–4117. [Google Scholar] [CrossRef]
- Tsou, K.; Brown, S.; Sañudo-Peña, M.; Mackie, K.; Walker, J. Immunohistochemical distribution of cannabinoid CB1 receptors in the rat central nervous system. Neuroscience 1998, 83, 393–411. [Google Scholar] [CrossRef]
- Elphick, M.R.; Egertová, M. Localisation of cannabinoid receptors in the rat brain using antibodies to the intracellular C-terminal tail of CB1. J. Comp. Neurol. 2000, 422, 159–171. [Google Scholar] [CrossRef]
- Katona, I.; Rancz, E.A.; Acsády, L.; Ledent, C.; Mackie, K.; Hájos, N.; Freund, T.F. Distribution of CB1 Cannabinoid Receptors in the Amygdala and their Role in the Control of GABAergic Transmission. J. Neurosci. 2001, 21, 9506–9518. [Google Scholar] [CrossRef]
- Navarrete, M.; Araque, A. Endocannabinoids Mediate Neuron-Astrocyte Communication. Neuron 2008, 57, 883–893. [Google Scholar] [CrossRef]
- Marinelli, S.; Pacioni, S.; Cannich, A.; Marsicano, G.; Bacci, A. Self-modulation of neocortical pyramidal neurons by endocannabinoids. Nat. Neurosci. 2009, 12, 1488–1490. [Google Scholar] [CrossRef] [PubMed]
- Hojo, M.; Sudo, Y.; Ando, Y.; Minami, K.; Takada, M.; Matsubara, T.; Kanaide, M.; Taniyama, K.; Sumikawa, K.; Uezono, Y. μ-Opioid Receptor Forms a Functional Heterodimer with Cannabinoid CB1 Receptor: Electrophysiological and FRET Assay Analysis. J. Pharmacol. Sci. 2008, 108, 308–319. [Google Scholar] [CrossRef] [PubMed]
- Cristino, L.; Luongo, L.; Imperatore, R.; Boccella, S.; Becker, T.; Morello, G.; Piscitelli, F.; Busetto, G.; Maione, S.; Di Marzo, V. Orexin-A and Endocannabinoid Activation of the Descending Antinociceptive Pathway Underlies Altered Pain Perception in Leptin Signaling Deficiency. Neuropsychopharmacology 2015, 41, 508–520. [Google Scholar] [CrossRef] [PubMed]
- Metna-Laurent, M.; Marsicano, G. Rising stars: Modulation of brain functions by astroglial type-1 cannabinoid receptors. Glia 2014, 63, 353–364. [Google Scholar] [CrossRef] [PubMed]
- Steindel, F.; Lerner, R.; Häring, M.; Ruehle, S.; Marsicano, G.; Lutz, B.; Monory, K. Neuron-type specific cannabinoid-mediated G protein signalling in mouse hippocampus. J. Neurochem. 2013, 124, 795–807. [Google Scholar] [CrossRef] [PubMed]
- Nyíri, G.; Cserép, C.; Szabadits, E.; MacKie, K.; Freund, T. CB1 cannabinoid receptors are enriched in the perisynaptic annulus and on preterminal segments of hippocampal GABAergic axons. Neuroscience 2005, 136, 811–822. [Google Scholar] [CrossRef]
- Katona, I.; Freund, T.F. Multiple Functions of Endocannabinoid Signaling in the Brain. Annu. Rev. Neurosci. 2012, 35, 529–558. [Google Scholar] [CrossRef]
- Dudok, B.; Barna, L.; Ledri, M.; I Szabó, S.; Szabadits, E.; Pintér, B.; Woodhams, S.G.; Henstridge, C.M.; Balla, G.Y.; Nyilas, R.; et al. Cell-specific STORM super-resolution imaging reveals nanoscale organization of cannabinoid signaling. Nat. Neurosci. 2014, 18, 75–86. [Google Scholar] [CrossRef]
- Zöldi, M.; Katona, I. STORM Super-Resolution Imaging of CB1 Receptors in Tissue Preparations. In Endocannabinoid Signaling: Methods and Protocols; Maccarrone, M., Ed.; Springer: New York, NY, USA, 2023; pp. 437–451. [Google Scholar] [CrossRef]
- Zhou, R.; Han, B.; Xia, C.; Zhuang, X. Membrane-associated periodic skeleton is a signaling platform for RTK transactivation in neurons. Science 2019, 365, 929–934. [Google Scholar] [CrossRef]
- Horti, A.G.; Fan, H.; Kuwabara, H.; Hilton, J.; Ravert, H.T.; Holt, D.P.; Alexander, M.; Kumar, A.; Rahmim, A.; Scheffel, U.; et al. 11C-JHU75528: A radiotracer for PET imaging of CB1 cannabinoid receptors. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2006, 47, 1689–1696. [Google Scholar]
- Basagni, F.; Rosini, M.; Decker, M. Functionalized Cannabinoid Subtype 2 Receptor Ligands: Fluorescent, PET, Photochromic and Covalent Molecular Probes. ChemMedChem 2020, 15, 1374–1389. [Google Scholar] [CrossRef] [PubMed]
- Haider, A.; Spinelli, F.; Herde, A.M.; Mu, B.; Keller, C.; Margelisch, M.; Weber, M.; Schibli, R.; Mu, L.; Ametamey, S.M. Evaluation of 4-oxo-quinoline-based CB2 PET radioligands in R6/2 chorea huntington mouse model and human ALS spinal cord tissue. Eur. J. Med. Chem. 2018, 145, 746–759. [Google Scholar] [CrossRef] [PubMed]
- Savonenko, A.V.; Melnikova, T.; Wang, Y.; Ravert, H.; Gao, Y.; Koppel, J.; Lee, D.; Pletnikova, O.; Cho, E.; Sayyida, N.; et al. Cannabinoid CB2 Receptors in a Mouse Model of Aβ Amyloidosis: Immunohistochemical Analysis and Suitability as a PET Biomarker of Neuroinflammation. PLoS ONE 2015, 10, e0129618. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Wang, H. The spatiotemporal expression changes of CB2R in the hippocampus of rats following pilocarpine-induced status epilepticus. Epilepsy Res. 2018, 148, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-Y.; Shen, H.; Jordan, C.J.; Liu, Q.-R.; Gardner, E.L.; Bonci, A.; Xi, Z.-X. CB2 receptor antibody signal specificity: Correlations with the use of partial CB2-knockout mice and anti-rat CB2 receptor antibodies. Acta Pharmacol. Sin. 2018, 40, 398–409. [Google Scholar] [CrossRef]
- Bai, M.; Sexton, M.; Stella, N.; Bornhop, D.J. MBC94, a Conjugable Ligand for Cannabinoid CB2 Receptor Imaging. Bioconjugate Chem. 2008, 19, 988–992. [Google Scholar] [CrossRef]
- Wu, Z.; Shao, P.; Zhang, S.; Bai, M. Targeted zwitterionic near infrared fluorescent probe for improved imaging of type 2 cannabinoid receptors. J. Biomed. Opt. 2014, 19, 036006. [Google Scholar] [CrossRef]
- Singh, S.; Oyagawa, C.R.M.; Macdonald, C.; Grimsey, N.L.; Glass, M.; Vernall, A.J. Chromenopyrazole-based High Affinity, Selective Fluorescent Ligands for Cannabinoid Type 2 Receptor. ACS Med. Chem. Lett. 2019, 10, 209–214. [Google Scholar] [CrossRef]
- Mensching, L.; Rading, S.; Nikolaev, V.; Karsak, M. Monitoring Cannabinoid CB2 -Receptor Mediated cAMP Dynamics by FRET-Based Live Cell Imaging. Int. J. Mol. Sci. 2020, 21, 7880. [Google Scholar] [CrossRef]
- Onaivi, E.S.; Ishiguro, H.; Gong, J.-P.; Patel, S.; Meozzi, P.A.; Myers, L.; Perchuk, A.; Mora, Z.; Tagliaferro, P.A.; Gardner, E.; et al. Brain Neuronal CB2 Cannabinoid Receptors in Drug Abuse and Depression: From Mice to Human Subjects. PLoS ONE 2008, 3, e1640. [Google Scholar] [CrossRef]
- Esteban, S.R.d.M.; Benito-Cuesta, I.; Terradillos, I.; Martínez-Relimpio, A.M.; Arnanz, M.A.; Ruiz-Pérez, G.; Korn, C.; Raposo, C.; Sarott, R.C.; Westphal, M.V.; et al. Cannabinoid CB2 Receptors Modulate Microglia Function and Amyloid Dynamics in a Mouse Model of Alzheimer’s Disease. Front. Pharmacol. 2022, 13, 841766. [Google Scholar] [CrossRef] [PubMed]
- Xing, C.; Zhuang, Y.; Xu, T.-H.; Feng, Z.; Zhou, X.E.; Chen, M.; Wang, L.; Meng, X.; Xue, Y.; Wang, J.; et al. Cryo-EM Structure of the Human Cannabinoid Receptor CB2-Gi Signaling Complex. Cell 2020, 180, 645–654.e13. [Google Scholar] [CrossRef] [PubMed]
- Leishman, E.; Cornett, B.; Spork, K.; Straiker, A.; Mackie, K.; Bradshaw, H.B. Broad impact of deleting endogenous cannabinoid hydrolyzing enzymes and the CB1 cannabinoid receptor on the endogenous cannabinoid-related lipidome in eight regions of the mouse brain. Pharmacol. Res. 2016, 110, 159–172. [Google Scholar] [CrossRef] [PubMed]
- Marchioni, C.; de Souza, I.D.; Acquaro, V.R.; Crippa, J.A.d.S.; Tumas, V.; Queiroz, M.E.C. Recent advances in LC-MS/MS methods to determine endocannabinoids in biological samples: Application in neurodegenerative diseases. Anal. Chim. Acta 2018, 1044, 12–28. [Google Scholar] [CrossRef] [PubMed]
- Berman, P.; Sulimani, L.; Gelfand, A.; Amsalem, K.; Lewitus, G.M.; Meiri, D. Cannabinoidomics—An analytical approach to understand the effect of medical Cannabis treatment on the endocannabinoid metabolome. Talanta 2020, 219, 121336. [Google Scholar] [CrossRef]
- Nielsen, M.M.B.; Lambertsen, K.L.; Clausen, B.H.; Meyer, M.; Bhandari, D.R.; Larsen, S.T.; Poulsen, S.S.; Spengler, B.; Janfelt, C.; Hansen, H.S. Mass spectrometry imaging of biomarker lipids for phagocytosis and signalling during focal cerebral ischaemia. Sci. Rep. 2016, 6, 39571. [Google Scholar] [CrossRef]
- Dong, A.; He, K.; Dudok, B.; Farrell, J.S.; Guan, W.; Liput, D.J.; Puhl, H.L.; Cai, R.; Wang, H.; Duan, J.; et al. A fluorescent sensor for spatiotemporally resolved imaging of endocannabinoid dynamics in vivo. Nat. Biotechnol. 2021, 40, 787–798. [Google Scholar] [CrossRef]
- Chen, Z.; Mori, W.; Fu, H.; Schafroth, M.A.; Hatori, A.; Shao, T.; Zhang, G.; Van, R.S.; Zhang, Y.; Hu, K.; et al. Design, Synthesis, and Evaluation of 18F-Labeled Monoacylglycerol Lipase Inhibitors as Novel Positron Emission Tomography Probes. J. Med. Chem. 2019, 62, 8866–8872. [Google Scholar] [CrossRef]
- He, Y.; Gobbi, L.C.; Herde, A.M.; Rombach, D.; Ritter, M.; Kuhn, B.; Wittwer, M.B.; Heer, D.; Hornsperger, B.; Bell, C.; et al. Discovery, synthesis and evaluation of novel reversible monoacylglycerol lipase radioligands bearing a morpholine-3-one scaffold. Nucl. Med. Biol. 2022, 108–109, 24–32. [Google Scholar] [CrossRef]
- Jacobson, M.R.; Watts, J.J.; Da Silva, T.; Tyndale, R.F.; Rusjan, P.M.; Houle, S.; Wilson, A.A.; Ross, R.A.; Boileau, I.; Mizrahi, R. Fatty acid amide hydrolase is lower in young cannabis users. Addict. Biol. 2020, 26, e12872. [Google Scholar] [CrossRef]
- Varlow, C.; Boileau, I.; Wey, H.-Y.; Liang, S.H.; Vasdev, N. Classics in Neuroimaging: Imaging the Endocannabinoid Pathway with PET. ACS Chem. Neurosci. 2020, 11, 1855–1862. [Google Scholar] [CrossRef]
- Hou, L.; Rong, J.; Haider, A.; Ogasawara, D.; Varlow, C.; Schafroth, M.A.; Mu, L.; Gan, J.; Xu, H.; Fowler, C.J.; et al. Positron Emission Tomography Imaging of the Endocannabinoid System: Opportunities and Challenges in Radiotracer Development. J. Med. Chem. 2020, 64, 123–149. [Google Scholar] [CrossRef]
- Wilson, A.A.; Hicks, J.W.; Sadovski, O.; Parkes, J.; Tong, J.; Houle, S.; Fowler, C.J.; Vasdev, N. Radiosynthesis and Evaluation of [11C-Carbonyl]-Labeled Carbamates as Fatty Acid Amide Hydrolase Radiotracers for Positron Emission Tomography. J. Med. Chem. 2012, 56, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Guberman, M.; Kosar, M.; Omran, A.; Carreira, E.M.; Nazaré, M.; Grether, U. Reverse-Design toward Optimized Labeled Chemical Probes—Examples from the Endocannabinoid System. Chimia 2022, 76, 425. [Google Scholar] [CrossRef]
- Mock, E.D.; Mustafa, M.; Gunduz-Cinar, O.; Cinar, R.; Petrie, G.N.; Kantae, V.; Di, X.; Ogasawara, D.; Varga, Z.V.; Paloczi, J.; et al. Discovery of a NAPE-PLD inhibitor that modulates emotional behavior in mice. Nat. Chem. Biol. 2020, 16, 667–675. [Google Scholar] [CrossRef] [PubMed]
- van Rooden, E.J.; Kreekel, R.; Hansen, T.; Janssen, A.P.A.; van Esbroeck, A.C.M.; Dulk, H.D.; Berg, R.J.B.H.N.v.D.; Codée, J.D.C.; van der Stelt, M. Two-step activity-based protein profiling of diacylglycerol lipase. Org. Biomol. Chem. 2018, 16, 5250–5253. [Google Scholar] [CrossRef]
- Bisogno, T.; Howell, F.; Williams, G.; Minassi, A.; Cascio, M.G.; Ligresti, A.; Matias, I.; Schiano-Moriello, A.; Paul, P.; Williams, E.-J.; et al. Cloning of the first sn1-DAG lipases points to the spatial and temporal regulation of endocannabinoid signaling in the brain. J. Cell Biol. 2003, 163, 463–468. [Google Scholar] [CrossRef]
- Yoshida, T.; Fukaya, M.; Uchigashima, M.; Miura, E.; Kamiya, H.; Kano, M.; Watanabe, M. Localization of Diacylglycerol Lipase-α around Postsynaptic Spine Suggests Close Proximity between Production Site of an Endocannabinoid, 2-Arachidonoyl-glycerol, and Presynaptic Cannabinoid CB1 Receptor. J. Neurosci. 2006, 26, 4740–4751. [Google Scholar] [CrossRef]
- Cristino, L.; Starowicz, K.; De Petrocellis, L.; Morishita, J.; Ueda, N.; Guglielmotti, V.; Di Marzo, V. Immunohistochemical localization of anabolic and catabolic enzymes for anandamide and other putative endovanilloids in the hippocampus and cerebellar cortex of the mouse brain. Neuroscience 2008, 151, 955–968. [Google Scholar] [CrossRef]
- Gulyas, A.I.; Cravatt, B.F.; Bracey, M.H.; Dinh, T.P.; Piomelli, D.; Boscia, F.; Freund, T.F. Segregation of two endocannabinoid-hydrolyzing enzymes into pre- and postsynaptic compartments in the rat hippocampus, cerebellum and amygdala. Eur. J. Neurosci. 2004, 20, 441–458. [Google Scholar] [CrossRef]
- Egertová, M.; Cravatt, B.; Elphick, M. Comparative analysis of fatty acid amide hydrolase and cb1 cannabinoid receptor expression in the mouse brain: Evidence of a widespread role for fatty acid amide hydrolase in regulation of endocannabinoid signaling. Neuroscience 2003, 119, 481–496. [Google Scholar] [CrossRef]
- Zimmer, A. Genetic Manipulation of the Endocannabinoid System. In Endocannabinoids; Pertwee, R.G., Ed.; Springer International Publishing: Berlin/Heidelberg, Germany, 2015; pp. 129–183. [Google Scholar] [CrossRef]
- Buckley, N.E. The peripheral cannabinoid receptor knockout mice: An update. Br. J. Pharmacol. 2008, 153, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Oltrabella, F.; Melgoza, A.; Nguyen, B.; Guo, S. Role of the endocannabinoid system in vertebrates: Emphasis on the zebrafish model. Dev. Growth Differ. 2017, 59, 194–210. [Google Scholar] [CrossRef] [PubMed]
- Estrada-Valencia, R.; De Lima, M.E.; Colonnello, A.; Rangel-López, E.; Saraiva, N.R.; De Ávila, D.S.; Aschner, M.; San-tamaría, A. The Endocannabinoid System in Caenorhabditis elegans. In Reviews of Physiology, Bio-chemistry and Pharmacology; Pedersen, S.H.F., Ed.; Springer Nature: Cham, Switzerland, 2021; Volume 184, pp. 1–31. [Google Scholar] [CrossRef]
- Chiu, H.; Alqadah, A.; Chuang, C.-F.; Chang, C. C. elegans as a genetic model to identify novel cellular and molecular mechanisms underlying nervous system regeneration. Cell Adhes. Migr. 2011, 5, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Lehtonen, M.; Reisner, K.; Auriola, S.; Wong, G.; Callaway, J.C. Mass-spectrometric identification of anandamide and 2-arachidonoylglycerol in nematodes. Chem. Biodivers. 2008, 5, 2431–2441. [Google Scholar] [CrossRef] [PubMed]
- McPartland, J.M.; Matias, I.; Di Marzo, V.; Glass, M. Evolutionary origins of the endocannabinoid system. Gene 2006, 370, 64–74. [Google Scholar] [CrossRef]
- Lucanic, M.; Held, J.M.; Vantipalli, M.C.; Klang, I.M.; Graham, J.B.; Gibson, B.W.; Lithgow, G.J.; Gill, M.S. N-acylethanolamine signalling mediates the effect of diet on lifespan in Caenorhabditis elegans. Nature 2011, 473, 226–229. [Google Scholar] [CrossRef]
- Harrington, A.J.; Hamamichi, S.; Caldwell, G.A.; Caldwell, K.A. C. elegans as a model organism to investigate molecular pathways involved with Parkinson’s disease. Dev. Dyn. 2010, 239, 1282–1295. [Google Scholar] [CrossRef]
- Pastuhov, S.I.; Matsumoto, K.; Hisamoto, N. Endocannabinoid signaling regulates regenerative axon navigation in Caenorhabditis elegansvia the GPCRs NPR-19 and NPR-32. Genes Cells 2016, 21, 696–705. [Google Scholar] [CrossRef]
- McPartland, J.M.; Glass, M. Nematicidal effects of hemp (Cannabis sativa) may not be mediated by cannabinoid receptors. New Zealand Journal of Crop and Horticultural Science 2001, 29, 301–307. [Google Scholar] [CrossRef]
- Galles, C.; Prez, G.M.; Penkov, S.; Boland, S.; Porta, E.O.J.; Altabe, S.G.; Labadie, G.R.; Schmidt, U.; Knölker, H.-J.; Kurzchalia, T.V.; et al. Endocannabinoids in Caenorhabditis elegans are essential for the mobilization of cholesterol from internal reserves. Sci. Rep. 2018, 8, 6398. [Google Scholar] [CrossRef] [PubMed]
- Pastuhov, S.I.; Fujiki, K.; Nix, P.; Kanao, S.; Bastiani, M.; Matsumoto, K.; Hisamoto, N. Endocannabinoid-Goα signalling inhibits axon regeneration in Caenorhabditis elegans by antagonizing Gqα-PKC-JNK signalling. Nat. Commun. 2012, 3, 1136. [Google Scholar] [CrossRef]
- Oakes, M.D.; Law, W.J.; Clark, T.; Bamber, B.A.; Komuniecki, R. Cannabinoids Activate Monoaminergic Signaling to Modulate Key C. elegans Behaviors. J. Neurosci. 2017, 37, 2859–2869. [Google Scholar] [CrossRef] [PubMed]
- Oakes, M.; Law, W.J.; Komuniecki, R. Cannabinoids Stimulate the TRP Channel-Dependent Release of Both Serotonin and Dopamine to Modulate Behavior in C. elegans. J. Neurosci. 2019, 39, 4142–4152. [Google Scholar] [CrossRef]
- Vanin, A.P.; Tamagno, W.A.; Alves, C.; Mesacasa, L.; Santin, L.F.; Sutorillo, N.T.; Bilibio, D.; Müller, C.; Galon, L.; Kaizer, R.R. Neuroprotective potential of Cannabis sativa-based oils in Caenorhabditis elegans. Sci. Rep. 2022, 12, 15376. [Google Scholar] [CrossRef] [PubMed]
- Nance, J.; Frøkjær-Jensen, C. The Caenorhabditis elegans Transgenic Toolbox. Genetics 2019, 212, 959–990. [Google Scholar] [CrossRef]
- Stewart, A.M.; Braubach, O.; Spitsbergen, J.; Gerlai, R.; Kalueff, A.V. Zebrafish models for translational neuroscience research: From tank to bedside. Trends Neurosci. 2014, 37, 264–278. [Google Scholar] [CrossRef]
- Bailone, R.L.; Fukushima, H.C.S.; de Aguiar, L.K.; Borra, R.C. The endocannabinoid system in zebrafish and its potential to study the effects of Cannabis in humans. Lab. Anim. Res. 2022, 38, 5. [Google Scholar] [CrossRef]
- Klee, E.W.; Schneider, H.; Clark, K.J.; Cousin, M.A.; Ebbert, J.O.; Hooten, W.M.; Karpyak, V.M.; Warner, D.O.; Ekker, S.C. Zebrafish: A model for the study of addiction genetics. Hum. Genet. 2011, 131, 977–1008. [Google Scholar] [CrossRef]
- Krug, R.G.; Clark, K.J. Elucidating cannabinoid biology in zebrafish (Danio rerio). Gene 2015, 570, 168–179. [Google Scholar] [CrossRef]
- Demin, K.A.; Meshalkina, D.A.; Kysil, E.V.; Antonova, K.A.; Volgin, A.D.; Yakovlev, O.A.; Alekseeva, P.A.; Firuleva, M.M.; Lakstygal, A.M.; de Abreu, M.S.; et al. Zebrafish models relevant to studying central opioid and endocannabinoid systems. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2018, 86, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Martin, I.; de Velasco, E.M.F.; Rodriguez, R.E. Characterization of cannabinoid-binding sites in zebrafish brain. Neurosci. Lett. 2007, 413, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Watson, S.; Chambers, D.; Hobbs, C.; Doherty, P.; Graham, A. The endocannabinoid receptor, CB1, is required for normal axonal growth and fasciculation. Mol. Cell Neurosci. 2008, 38, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Postlethwait, J.; Amores, A.; Force, A.; Yan, Y.-L. Chapter 8 The Zebrafish Genome. In Methods in Cell Biology; Detrich, H.W., Westerfield, M., Zon, L.I., Eds.; Academic Press: Cambridge, MA, USA, 1998; Volume 60, pp. 149–163. [Google Scholar] [CrossRef]
- Blackburn, P.R.; Campbell, J.M.; Clark, K.J.; Ekker, S.C. The CRISPR system—Keeping zebrafish gene targeting fresh. Zebrafish 2013, 10, 116–118. [Google Scholar] [CrossRef] [PubMed]
- Campbell, D.S.; Okamoto, H. Local caspase activation interacts with Slit-Robo signaling to restrict axonal arborization. J. Cell Biol. 2013, 203, 657–672. [Google Scholar] [CrossRef] [PubMed]
- Pai, W.-Y.; Hsu, C.-C.; Lai, C.-Y.; Chang, T.-Z.; Tsai, Y.-L.; Her, G.M. Cannabinoid receptor 1 promotes hepatic lipid accumulation and lipotoxicity through the induction of SREBP-1c expression in zebrafish. Transgenic Res. 2013, 22, 823–838. [Google Scholar] [CrossRef] [PubMed]
- Nishio, S.-I.; Gibert, Y.; Berekelya, L.; Bernard, L.; Brunet, F.; Guillot, E.; Le Bail, J.-C.; Sánchez, J.A.; Galzin, A.M.; Triqueneaux, G.; et al. Fasting Induces CART Down-Regulation in the Zebrafish Nervous System in a Cannabinoid Receptor 1-Dependent Manner. Mol. Endocrinol. 2012, 26, 1316–1326. [Google Scholar] [CrossRef]
- Shimada, Y.; Hirano, M.; Nishimura, Y.; Tanaka, T. A High-Throughput Fluorescence-Based Assay System for Appetite-Regulating Gene and Drug Screening. PLoS ONE 2012, 7, e52549. [Google Scholar] [CrossRef]
- Liu, Y.-J.; Fan, H.-B.; Jin, Y.; Ren, C.-G.; Jia, X.-E.; Wang, L.; Chen, Y.; Dong, M.; Zhu, K.-Y.; Dong, Z.-W.; et al. Cannabinoid Receptor 2 Suppresses Leukocyte Inflammatory Migration by Modulating the JNK/c-Jun/Alox5 Pathway. J. Biol. Chem. 2013, 288, 13551–13562. [Google Scholar] [CrossRef]
- Esain, V.; Kwan, W.; Carroll, K.J.; Cortes, M.; Liu, S.Y.; Frechette, G.M.; Sheward, L.M.V.; Nissim, S.; Goessling, W.; North, T.E. Cannabinoid Receptor-2 Regulates Embryonic Hematopoietic Stem Cell Development via Prostaglandin E2 and P-Selectin Activity. Stem. Cells 2015, 33, 2596–2612. [Google Scholar] [CrossRef]
- Chiarlone, A.; Bellocchio, L.; Blázquez, C.; Resel, E.; Soria-Gómez, E.; Cannich, A.; Ferrero, J.J.; Sagredo, O.; Benito, C.; Romero, J.; et al. A restricted population of CB1 cannabinoid receptors with neuroprotective activity. Proc. Natl. Acad. Sci. USA 2014, 111, 8257–8262. [Google Scholar] [CrossRef] [PubMed]
- Fin, L.; Bergamin, G.; Steiner, R.A.; Hughes, S.M. The Cannabinoid Receptor Interacting Proteins 1 of zebrafish are not required for morphological development, viability or fertility. Sci. Rep. 2017, 7, 4858. [Google Scholar] [CrossRef]
- Acevedo-Canabal, A.; Colón-Cruz, L.; Rodriguez-Morales, R.; Varshney, G.K.; Burgess, S.; González-Sepúlveda, L.; Yudowski, G.; Behra, M. Altered Swimming Behaviors in Zebrafish Larvae Lacking Cannabinoid Receptor 2. Cannabis Cannabinoid Res. 2019, 4, 88–101. [Google Scholar] [CrossRef] [PubMed]
- Zuccarini, G.; D’atri, I.; Cottone, E.; Mackie, K.; Shainer, I.; Gothilf, Y.; Provero, P.; Bovolin, P.; Merlo, G.R. Interference with the cannabinoid receptor CB1R results in miswiring of GnRH3 and AgRP1 axons in zebrafish embryos. Int. J. Mol. Sci. 2019, 21, 168. [Google Scholar] [CrossRef] [PubMed]
- JBrowse 20:904765..913703. (n.d.). Available online: https://zfin.org/jbrowse/?nav=1&overview=1&tracklist=1&loc=20%3A904765..913703&tracks=ZFIN%20Gene%2CKnockdown%20Reagent%2CTranscript&data=data%2FGRCz11&highlight= (accessed on 3 September 2023).
- Di Marzo, V.; Matias, I. Endocannabinoid control of food intake and energy balance. Nat. Neurosci. 2005, 8, 585–589. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, A.; Zimmer, A.M.; Hohmann, A.G.; Herkenham, M.; Bonner, T.I. Increased mortality, hypoactivity, and hypoalgesia in cannabinoid CB1 receptor knockout mice. Proc. Natl. Acad. Sci. USA 1999, 96, 5780–5785. [Google Scholar] [CrossRef]
- Zhang, M.; Han, L.; Xu, Y. Roles of cocaine- and amphetamine-regulated transcript in the central nervous system. Clin. Exp. Pharmacol. Physiol. 2011, 39, 586–592. [Google Scholar] [CrossRef]
- McLaughlin, P.J.; Winston, K.M.; Limebeer, C.L.; Parker, L.A.; Makriyannis, A.; Salamone, J.D. The cannabinoid CB1 antagonist AM 251 produces food avoidance and behaviors associated with nausea but does not impair feeding efficiency in rats. Psychopharmacol. 2005, 180, 286–293. [Google Scholar] [CrossRef]
- Liu, L.Y.; Alexa, K.; Cortes, M.; Schatzman-Bone, S.; Kim, A.J.; Mukhopadhyay, B.; Cinar, R.; Kunos, G.; North, T.E.; Goessling, W. Cannabinoid receptor signaling regulates liver development and metabolism. Development 2016, 143, 609–622. [Google Scholar] [CrossRef]
- Sun, Y.; Chen, X.; Xiao, D. Tetracycline-inducible expression systems: New strategies and practices in the transgenic mouse modeling. Acta Biochim. Biophys. Sin. 2007, 39, 235–246. [Google Scholar] [CrossRef]
- JBrowse 16:33111425..33115368. (n.d.). Available online: https://zfin.org/jbrowse/?nav=1&overview=1&tracklist=1&loc=16%3A33111425..33115368&tracks=ZFIN%20Gene%2CKnockdown%20Reagent%2CTranscript&data=data%2FGRCz11&highlight= (accessed on 3 September 2023).
- Basu, S.; Dittel, B.N. Unraveling the complexities of cannabinoid receptor 2 (CB2) immune regulation in health and disease. Immunol. Res. 2011, 51, 26–38. [Google Scholar] [CrossRef] [PubMed]
- Adhikary, S.; Kocieda, V.P.; Yen, J.-H.; Tuma, R.F.; Ganea, D. Signaling through cannabinoid receptor 2 suppresses murine dendritic cell migration by inhibiting matrix metalloproteinase 9 expression. Blood 2012, 120, 3741–3749. [Google Scholar] [CrossRef] [PubMed]
- Hoggatt, J.; Pelus, L.M. Eicosanoid regulation of hematopoiesis and hematopoietic stem and progenitor trafficking. Leukemia 2010, 24, 1993–2002. [Google Scholar] [CrossRef] [PubMed]
- Busquets-Garcia, A.; Puighermanal, E.; Pastor, A.; de la Torre, R.; Maldonado, R.; Ozaita, A. Differential role of anandamide and 2-Arachidonoylglycerol in memory and anxiety-like responses. Biol. Psychiatry 2011, 70, 479–486. [Google Scholar] [CrossRef]
- Maccarrone, M.; Attinà, M.; Bari, M.; Cartoni, A.; Ledent, C.; Finazzi-Agrò, A. Anandamide degradation and N-acylethanolamines level in wild-type and CB1 cannabinoid receptor knockout mice of different ages. J. Neurochem. 2001, 78, 339–348. [Google Scholar] [CrossRef]
- Urigüen, L.; Pérez-Rial, S.; Ledent, C.; Palomo, T.; Manzanares, J. Impaired action of anxiolytic drugs in mice deficient in cannabinoid CB1 receptors. Neuropharmacology 2004, 46, 966–973. [Google Scholar] [CrossRef]
- Ledent, C.; Valverde, O.; Cossu, G.; Petitet, F.; Aubert, J.-F.; Beslot, F.; Bohme, G.A.; Imperato, A.; Pedrazzini, T.; Roques, B.P.; et al. Unresponsiveness to Cannabinoids and Reduced Addictive Effects of Opiates in CB1 Receptor Knockout Mice. Science 1999, 283, 401–404. [Google Scholar] [CrossRef]
- Haller, J.; Varga, B.; Ledent, C.; Freund, T.F. CB1 cannabinoid receptors mediate anxiolytic effects: Convergent genetic and pharmacological evidence with CB1-specific agents. Behav. Pharmacol. 2004, 15, 299–304. [Google Scholar] [CrossRef]
- Álvaro-Bartolomé, M.; Esteban, S.; Gutiérrez, G.; Manzanares, J.; Valverde, O.; García-Sevilla, J. Regulation of Fas receptor/Fas-asssociated protein with death domain apoptotic complex and associated signalling systems by cannabinoid receptors in the mouse brain. Br. J. Pharmacol. 2010, 160, 643–656. [Google Scholar] [CrossRef]
- Austrich-Olivares, A.; García-Gutiérrez, M.S.; Illescas, L.; Gasparyan, A.; Manzanares, J. Cannabinoid CB1 Receptor Involvement in the Actions of CBD on Anxiety and Coping Behaviors in Mice. Pharmaceuticals 2022, 15, 473. [Google Scholar] [CrossRef]
- Steiner, H.; Bonner, T.I.; Zimmer, A.M.; Kitai, S.T.; Zimmer, A. Altered gene expression in striatal projection neurons in CB1 cannabinoid receptor knockout mice. Proc. Natl. Acad. Sci. USA 1999, 96, 5786–5790. [Google Scholar] [CrossRef] [PubMed]
- Varvel, S.A.; Lichtman, A.H. Evaluation of CB1 receptor knockout mice in the morris water maze. Experiment 2002, 301, 915–924. [Google Scholar] [CrossRef] [PubMed]
- Albayram, O.; Bilkei-Gorzo, A.; Zimmer, A. Loss of CB1 receptors leads to differential age-related changes in reward-driven learning and memory. Front. Aging Neurosci. 2012, 4, 34. [Google Scholar] [CrossRef] [PubMed]
- Albayram, O.; Alferink, J.; Pitsch, J.; Piyanova, A.; Neitzert, K.; Poppensieker, K.; Mauer, D.; Michel, K.; Legler, A.; Becker, A.; et al. Role of CB1 cannabinoid receptors on GABAergic neurons in brain aging. Proc. Natl. Acad. Sci. USA 2011, 108, 11256–11261. [Google Scholar] [CrossRef]
- Albergaria, C.; Silva, N.T.; Darmohray, D.M.; Carey, M.R. Cannabinoids modulate associative cerebellar learning via alterations in behavioral state. eLife 2020, 9, 1–20. [Google Scholar] [CrossRef]
- Marsicano, G.; Wotjak, C.T.; Azad, S.C.; Bisogno, T.; Rammes, G.; Cascio, M.G.; Hermann, H.; Tang, J.; Hofmann, C.; Zieglgänsberger, W.; et al. The endogenous cannabinoid system controls extinction of aversive memories. Nature 2002, 418, 530–534. [Google Scholar] [CrossRef]
- Marsicano, G.; Goodenough, S.; Monory, K.; Hermann, H.; Eder, M.; Cannich, A.; Azad, S.C.; Cascio, M.G.; Gutiérrez, S.O.; van der Stelt, M.; et al. CB1 Cannabinoid Receptors and On-Demand Defense Against Excitotoxicity. Science 2003, 302, 84–88. [Google Scholar] [CrossRef]
- Hölter, S.M.; Kallnik, M.; Wurst, W.; Marsicano, G.; Lutz, B.; Wotjak, C.T. Cannabinoid CB1 receptor is dispensable for memory extinction in an appetitively-motivated learning task. Eur. J. Pharmacol. 2005, 510, 69–74. [Google Scholar] [CrossRef]
- Kamprath, K.; Marsicano, G.; Tang, J.; Monory, K.; Bisogno, T.; Marzo, V.D.; Lutz, B.; Wotjak, C.T. Cannabinoid CB1 receptor mediates fear extinction via habituation-like processes. J. Neurosci. 2006, 26, 6677–6686. [Google Scholar] [CrossRef]
- Robbe, D.; Kopf, M.; Remaury, A.; Bockaert, J.; Manzoni, O.J. Endogenous cannabinoids mediate long-term synaptic depression in the nucleus accumbens. Proc. Natl. Acad. Sci. USA 2002, 99, 8384–8388. [Google Scholar] [CrossRef]
- Ibrahim, M.M.; Deng, H.; Zvonok, A.; Cockayne, D.A.; Kwan, J.; Mata, H.P.; Vanderah, T.W.; Lai, J.; Porreca, F.; Makriyannis, A.; et al. Activation of CB 2 cannabinoid receptors by AM1241 inhibits experimental neuropathic pain: Pain inhibition by receptors not present in the CNS. Proc. Natl. Acad. Sci. USA 2003, 100, 10529–10533. [Google Scholar] [CrossRef] [PubMed]
- Pan, B.; Hillard, C.J.; Liu, Q.-S. D2Dopamine Receptor Activation Facilitates Endocannabinoid-Mediated Long-Term Synaptic Depression of GABAergic Synaptic Transmission in Midbrain Dopamine Neurons via cAMP-Protein Kinase A Signaling. J. Neurosci. 2008, 28, 14018–14030. [Google Scholar] [CrossRef] [PubMed]
- Haller, J.; Bakos, N.; Szirmay, M.; Ledent, C.; Freund, T.F. The effects of genetic and pharmacological blockade of the CB1 cannabinoid receptor on anxiety: Anxiety and cannabinoids. Eur. J. Neurosci. 2002, 16, 1395–1398. [Google Scholar] [CrossRef] [PubMed]
- Haller, J.; Varga, B.; Ledent, C.; Barna, I.; Freund, T.F. Context-dependent effects of CB1 cannabinoid gene disruption on anxiety-like and social behaviour in mice. Eur. J. Neurosci. 2004, 19, 1906–1912. [Google Scholar] [CrossRef]
- Thiemann, G.; Watt, C.A.; Ledent, C.; Molleman, A.; Hasenöhrl, R.U. Modulation of anxiety by acute blockade and genetic deletion of the CB1 cannabinoid receptor in mice together with biogenic amine changes in the forebrain. Behav. Brain Res. 2009, 200, 60–67. [Google Scholar] [CrossRef]
- Schechter, M.; Pinhasov, A.; Weller, A.; Fride, E. Blocking the postpartum mouse dam’s CB1 receptors impairs maternal behavior as well as offspring development and their adult social–emotional behavior. Behav. Brain Res. 2012, 226, 481–492. [Google Scholar] [CrossRef]
- Rodriguez-Arias, M.; Navarrete, F.; Daza-Losada, M.; Navarro, D.; Aguilar, M.A.; Berbel, P.; Miñarro, J.; Manzanares, J. CB1 cannabinoid receptor-mediated aggressive behavior. Neuropharmacology 2013, 75, 172–180. [Google Scholar] [CrossRef]
- Degroot, A.; Nomikos, G.G. Genetic deletion and pharmacological blockade of CB1 receptors modulates anxiety in the shock-probe burying test. Eur. J. Neurosci. 2004, 20, 1059–1064. [Google Scholar] [CrossRef]
- Cota, D.; Marsicano, G.; Tschöp, M.; Grübler, Y.; Flachskamm, C.; Schubert, M.; Auer, D.; Yassouridis, A.; Thöne-Reineke, C.; Ortmann, S.; et al. The endogenous cannabinoid system affects energy balance via central orexigenic drive and peripheral lipogenesis. J. Clin. Investig. 2003, 112, 423–431. [Google Scholar] [CrossRef]
- Plendl, W.; Wotjak, C.T. Dissociation of within- and between-Session Extinction of Conditioned Fear. J. Neurosci. 2010, 30, 4990–4998. [Google Scholar] [CrossRef]
- Dubreucq, S.; Koehl, M.; Abrous, D.N.; Marsicano, G.; Chaouloff, F. CB1 receptor deficiency decreases wheel-running activity: Consequences on emotional behaviours and hippocampal neurogenesis. Exp. Neurol. 2010, 224, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Metna-Laurent, M.; Soria-Gómez, E.; Verrier, D.; Conforzi, M.; Jégo, P.; Lafenêtre, P.; Marsicano, G. Bimodal Control of Fear-Coping Strategies by CB1Cannabinoid Receptors. J. Neurosci. 2012, 32, 7109–7118. [Google Scholar] [CrossRef] [PubMed]
- Dubreucq, S.; Matias, I.; Cardinal, P.; Häring, M.; Lutz, B.; Marsicano, G.; Chaouloff, F. Genetic dissection of the role of cannabinoid type-1 receptors in the emotional consequences of repeated social stress in mice. Neuropsychopharmacology 2012, 37, 1885–1900. [Google Scholar] [CrossRef]
- Jacob, W.; Marsch, R.; Marsicano, G.; Lutz, B.; Wotjak, C.T. Cannabinoid CB1 receptor deficiency increases contextual fear memory under highly aversive conditions and long-term potentiation in vivo. Neurobiol. Learn. Mem. 2012, 98, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Pava, M.J.; Hartog, C.R.D.; Blanco-Centurion, C.; Shiromani, P.J.; Woodward, J.J. Endocannabinoid Modulation of Cortical Up-States and NREM Sleep. PLoS ONE 2014, 9, e88672. [Google Scholar] [CrossRef]
- Silvani, A.; Berteotti, C.; Bastianini, S.; Martire, V.L.; Mazza, R.; Pagotto, U.; Quarta, C.; Zoccoli, G. Multiple Sleep Alterations in Mice Lacking Cannabinoid Type 1 Receptors. PLoS ONE 2014, 9, e89432. [Google Scholar] [CrossRef]
- Castañé, A.; Valjent, E.; Ledent, C.; Parmentier, M.; Maldonado, R.; Valverde, O. Lack of CB1 cannabinoid receptors modifies nicotine behavioural responses, but not nicotine abstinence. Neuropharmacology 2002, 43, 857–867. [Google Scholar] [CrossRef]
- Naassila, M.; Pierrefiche, O.; Ledent, C.; Daoust, M. Decreased alcohol self-administration and increased alcohol sensitivity and withdrawal in CB1 receptor knockout mice. Neuropharmacology 2003, 46, 243–253. [Google Scholar] [CrossRef]
- Houchi, H.; Babovic, D.; Pierrefiche, O.; Ledent, C.; Daoust, M.; Naassila, M. CB1 Receptor Knockout Mice Display Reduced Ethanol-Induced Conditioned Place Preference and Increased Striatal Dopamine D2 Receptors. Neuropsychopharmacology 2004, 30, 339–349. [Google Scholar] [CrossRef]
- Thanos, P.K.; Dimitrakakis, E.S.; Rice, O.; Gifford, A.; Volkow, N.D. Ethanol self-administration and ethanol conditioned place preference are reduced in mice lacking cannabinoid CB1 receptors. Behav. Brain Res. 2005, 164, 206–213. [Google Scholar] [CrossRef]
- Corbille, A.-G.; Valjent, E.; Marsicano, G.; Ledent, C.; Lutz, B.; Herve, D.; Girault, J.-A. Role of Cannabinoid Type 1 Receptors in Locomotor Activity and Striatal Signaling in Response to Psychostimulants. J. Neurosci. 2007, 27, 6937–6947. [Google Scholar] [CrossRef] [PubMed]
- Fride, E.; Foox, A.; Rosenberg, E.; Faigenboim, M.; Cohen, V.; Barda, L.; Blau, H.; Mechoulam, R. Milk intake and survival in newborn cannabinoid CB1 receptor knockout mice: Evidence for a “CB3” receptor. Eur. J. Pharmacol. 2003, 461, 27–34. [Google Scholar] [CrossRef]
- Hungund, B.L.; Szakall, I.; Adam, A.; Basavarajappa, B.S.; Vadasz, C. Cannabinoid CB1 receptor knockout mice exhibit markedly reduced voluntary alcohol consumption and lack alcohol-induced dopamine release in the nucleus accumbens. J. Neurochem. 2003, 84, 698–704. [Google Scholar] [CrossRef] [PubMed]
- Racz, I.; Bilkei-Gorzo, A.; Toth, Z.E.; Michel, K.; Palkovits, M.; Zimmer, A. A Critical Role for the Cannabinoid CB1Receptors in Alcohol Dependence and Stress-Stimulated Ethanol Drinking. J. Neurosci. 2003, 23, 2453–2458. [Google Scholar] [CrossRef]
- Wang, L.; Liu, J.; Harvey-White, J.; Zimmer, A.; Kunos, G. Endocannabinoid signaling via cannabinoid receptor 1 is involved in ethanol preference and its age-dependent decline in mice. Biol. Sci. 2003, 100, 1393–1398. [Google Scholar] [CrossRef] [PubMed]
- Baskfield, C.Y.; Martin, B.R.; Wiley, J.L. Differential Effects of Δ9-Tetrahydrocannabinol and Methanandamide in CB1 Knockout and Wild-Type Mice. Experiment 2004, 309, 86–91. [Google Scholar] [CrossRef]
- Sain, N.M.; Liang, A.; Kane, S.A.; Urban, M.O. Antinociceptive effects of the non-selective cannabinoid receptor agonist CP 55,940 are absent in CB1−/− and not CB2−/− mice in models of acute and persistent pain. Neuropharmacology 2009, 57, 235–241. [Google Scholar] [CrossRef]
- Lazenka, M.F.; Selley, D.E.; Sim-Selley, L.J. ΔFosB induction correlates inversely with CB1 receptor desensitization in a brain region-dependent manner following repeated Δ9-THC administration. Neuropharmacology 2014, 77, 224–233. [Google Scholar] [CrossRef]
- Hiranita, T.; Wilkinson, D.S.; Hong, W.C.; Zou, M.-F.; Kopajtic, T.A.; Soto, P.L.; Lupica, C.R.; Newman, A.H.; Katz, J.L. 2-Isoxazol-3-Phenyltropane Derivatives of Cocaine: Molecular and Atypical System Effects at the Dopamine Transporter. Experiment 2014, 349, 297–309. [Google Scholar] [CrossRef]
- Nagre, N.N.; Subbanna, S.; Shivakumar, M.; Psychoyos, D.; Basavarajappa, B.S. CB1-receptor knockout neonatal mice are protected against ethanol-induced impairments of DNMT1, DNMT3A, and DNA methylation. J. Neurochem. 2014, 132, 429–442. [Google Scholar] [CrossRef]
- Sanchis-Segura, C.; Cline, B.H.; Marsicano, G.; Lutz, B.; Spanagel, R. Reduced sensitivity to reward in CB1 knockout mice. Psychopharmacol. 2004, 176, 223–232. [Google Scholar] [CrossRef]
- Aguado, T.; Palazuelos, J.; Monory, K.; Stella, N.; Cravatt, B.; Lutz, B.; Marsicano, G.; Kokaia, Z.; Guzmán, M.; Galve-Roperh, I. The endocannabinoid system promotes astroglial differentiation by acting on neural progenitor cells. J. Neurosci. 2006, 26, 1551–1561. [Google Scholar] [CrossRef] [PubMed]
- Monory, K.; Blaudzun, H.; Massa, F.; Kaiser, N.; Lemberger, T.; Schütz, G.; Wotjak, C.T.; Lutz, B.; Marsicano, G. Genetic dissection of behavioural and autonomic effects of Δ9-tetrahydrocannabinol in mice. PLoS Biol. 2007, 5, e269. [Google Scholar] [CrossRef]
- Vallee, M.; Vitiello, S.; Bellocchio, L.; Hébert-Chatelain, E.; Monlezun, S.; Martín-García, E.; Kasanetz, F.; Baillie, G.L.; Panin, F.; Cathala, A.; et al. Pregnenolone Can Protect the Brain from Cannabis Intoxication. Science 2014, 343, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.; Ledent, C.; Parmentier, M.; Maldonado, R.; Valverde, O. Involvement of CB1 cannabinoid receptors in emotional behaviour. Psychopharmacol. 2002, 159, 379–387. [Google Scholar] [CrossRef]
- Thiemann, G.; Fletcher, B.; Ledent, C.; Molleman, A.; Hasenöhrl, R.U. The genetic versus pharmacological invalidation of the cannabinoid CB1 receptor results in differential effects on ‘non-associative’ memory and forebrain monoamine concentrations in mice. Neurobiol. Learn. Mem. 2007, 88, 416–423. [Google Scholar] [CrossRef]
- Jin, K.; Xie, L.; Kim, S.H.; Parmentier-Batteur, S.; Sun, Y.; Mao, X.O.; Childs, J.; Greenberg, D.A. Defective Adult Neurogenesis in CB1 Cannabinoid Receptor Knockout Mice. Mol. Pharmacol. 2004, 66, 204–208. [Google Scholar] [CrossRef]
- Bilkei-Gorzo, A.; Racz, I.; Valverde, O.; Otto, M.; Michel, K.; Sarstre, M.; Zimmer, A. Early age-related cognitive impairment in mice lacking cannabinoid CB1 receptors. Proc. Natl. Acad. Sci. USA 2005, 102, 15670–15675. [Google Scholar] [CrossRef]
- Varvel, S.A.; Anum, E.A.; Lichtman, A.H. Disruption of CB1 receptor signaling impairs extinction of spatial memory in mice. Psychopharmacol. 2004, 179, 863–872. [Google Scholar] [CrossRef]
- Degroot, A.; Salhoff, C.; Davis, R.J.; Nomikos, G.G. Genetic deletion of CB1 receptors improves non-associative learning. Behav. Brain Res. 2005, 162, 161–164. [Google Scholar] [CrossRef]
- Kishimoto, Y.; Kano, M. Endogenous Cannabinoid Signaling through the CB1Receptor Is Essential for Cerebellum-Dependent Discrete Motor Learning. J. Neurosci. 2006, 26, 8829–8837. [Google Scholar] [CrossRef] [PubMed]
- Crombag, H.S.; Johnson, A.W.; Zimmer, A.M.; Zimmer, A.; Holland, P.C. Deficits in sensory-specific devaluation task performance following genetic deletions of cannabinoid (CB1) receptor. Learn. Mem. 2009, 17, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Marsicano, G.; Lutz, B. Expression of the cannabinoid receptor CB1 in distinct neuronal subpopulations in the adult mouse forebrain. Eur. J. Neurosci. 1999, 11, 4213–4225. [Google Scholar] [CrossRef] [PubMed]
- Mackie, K. Cannabinoid receptors as therapeutic targets. Annu. Rev. Pharmacol. Toxicol. 2006, 46, 101–122. [Google Scholar] [CrossRef]
- Matsuda, L.A.; Bonner, T.I.; Lolait, S.J. Localization of cannabinoid receptor mRNA in rat brain. J. Comp. Neurol. 1993, 327, 535–550. [Google Scholar] [CrossRef]
- Monory, K.; Massa, F.; Egertová, M.; Eder, M.; Blaudzun, H.; Westenbroek, R.; Kelsch, W.; Jacob, W.; Marsch, R.; Ekker, M.; et al. The Endocannabinoid System Controls Key Epileptogenic Circuits in the Hippocampus. Neuron 2006, 51, 455–466. [Google Scholar] [CrossRef]
- Kamprath, K.; Plendl, W.; Marsicano, G.; Deussing, J.M.; Wurst, W.; Lutz, B.; Wotjak, C.T. Endocannabinoids mediate acute fear adaptation via glutamatergic neurons independently of corticotropin-releasing hormone signaling. Genes Brain Behav. 2009, 8, 203–211. [Google Scholar] [CrossRef]
- Kato, A.; Punnakkal, P.; Pernía-Andrade, A.J.; von Schoultz, C.; Sharopov, S.; Nyilas, R.; Katona, I.; Zeilhofer, H.U. Endocannabinoid-dependent plasticity at spinal nociceptor synapses. J. Physiol. 2012, 590, 4717–4733. [Google Scholar] [CrossRef]
- Dubreucq, S.; Kambire, S.; Conforzi, M.; Metna-Laurent, M.; Cannich, A.; Soria-Gomez, E.; Richard, E.; Marsicano, G.; Chaouloff, F. Cannabinoid type 1 receptors located on single-minded 1–expressing neurons control emotional behaviors. Neuroscience 2012, 204, 230–244. [Google Scholar] [CrossRef]
- Berghuis, P.; Rajnicek, A.M.; Morozov, Y.M.; Ross, R.A.; Mulder, J.; Urbán, G.M.; Monory, K.; Marsicano, G.; Matteoli, M.; Canty, A.; et al. Hardwiring the Brain: Endocannabinoids Shape Neuronal Connectivity. Science 2007, 316, 1212–1216. [Google Scholar] [CrossRef]
- Puente, N.; Elezgarai, I.; Lafourcade, M.; Reguero, L.; Marsicano, G.; Georges, F.; Manzoni, O.J.; Grandes, P. Localization and function of the cannabinoid CB1 receptor in the anterolateral bed nucleus of the stria terminalis. PLoS ONE 2010, 5, e8869. [Google Scholar] [CrossRef] [PubMed]
- Rey, A.A.; Purrio, M.; Viveros, M.-P.; Lutz, B. Biphasic Effects of Cannabinoids in Anxiety Responses: CB1 and GABAB Receptors in the Balance of GABAergic and Glutamatergic Neurotransmission. Neuropsychopharmacology 2012, 37, 2624–2634. [Google Scholar] [CrossRef] [PubMed]
- Sales-Carbonell, C.; Rueda-Orozco, P.E.; Soria-Gómez, E.; Buzsáki, G.; Marsicano, G.; Robbe, D. Striatal GABAergic and cortical glutamatergic neurons mediate contrasting effects of cannabinoids on cortical network synchrony. Proc. Natl. Acad. Sci. USA 2012, 110, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Puighermanal, E.; Busquets-Garcia, A.; Gomis-González, M.; Marsicano, G.; Maldonado, R.; Ozaita, A. Dissociation of the pharmacological effects of THC by mTOR blockade. Neuropsychopharmacology 2013, 38, 1334–1343. [Google Scholar] [CrossRef] [PubMed]
- Puighermanal, E.; Marsicano, G.; Busquets-Garcia, A.; Lutz, B.; Maldonado, R.; Ozaita, A. Cannabinoid modulation of hippocampal long-term memory is mediated by mTOR signaling. Nat. Neurosci. 2009, 12, 1152–1158. [Google Scholar] [CrossRef]
- Wu, C.-S.; Zhu, J.; Wager-Miller, J.; Wang, S.; O’leary, D.; Monory, K.; Lutz, B.; Mackie, K.; Lu, H.-C. Requirement of cannabinoid CB1 receptors in cortical pyramidal neurons for appropriate development of corticothalamic and thalamocortical projections. Eur. J. Neurosci. 2010, 32, 693–706. [Google Scholar] [CrossRef]
- Díaz-Alonso, J.; Aguado, T.; Wu, C.-S.; Palazuelos, J.; Hofmann, C.; Garcez, P.; Guillemot, F.; Lu, H.-C.; Lutz, B.; Guzmán, M.; et al. The CB1 cannabinoid receptor drives corticospinal motor neuron differentiation through the Ctip2/Satb2 transcriptional regulation axis. J. Neurosci. 2012, 32, 16651–16665. [Google Scholar] [CrossRef]
- Carey, M.R.; Myoga, M.H.; McDaniels, K.R.; Marsicano, G.; Lutz, B.; Mackie, K.; Regehr, W.G. Presynaptic CB1 receptors regulate synaptic plasticity at cerebellar parallel fiber synapses. J. Neurophysiol. 2011, 105, 958–963. [Google Scholar] [CrossRef]
- Cutando, L.; Busquets-Garcia, A.; Puighermanal, E.; Gomis-González, M.; Delgado-García, J.M.; Gruart, A.; Maldonado, R.; Ozaita, A. Microglial activation underlies cerebellar deficits produced by repeated cannabis exposure. J. Clin. Investig. 2013, 123, 2816–2831. [Google Scholar] [CrossRef]
- Buckley, N.E.; McCoy, K.L.; Mezey, E.; Bonner, T.; Zimmer, A.; Felder, C.C.; Glass, M.; Zimmer, A. Immunomodulation by cannabinoids is absent in mice deficient for the cannabinoid CB2 receptor. Eur. J. Pharmacol. 2000, 396, 141–149. [Google Scholar] [CrossRef]
- García-Gutiérrez, M.S.; Manzanares, J. Overexpression of CB2 cannabinoid receptors decreased vulnerability to anxiety and impaired anxiolytic action of alprazolam in mice. J. Psychopharmacol. 2010, 25, 111–120. [Google Scholar] [CrossRef] [PubMed]
- La Porta, C.; Bura, S.A.; Aracil-Fernández, A.; Manzanares, J.; Maldonado, R. Role of CB1 and CB2 cannabinoid receptors in the development of joint pain induced by monosodium iodoacetate. Pain 2013, 154, 160–174. [Google Scholar] [CrossRef] [PubMed]
- López, A.; Aparicio, N.; Pazos, M.R.; Grande, M.T.; Barreda-Manso, M.A.; Benito-Cuesta, I.; Vázquez, C.; Amores, M.; Ruiz-Pérez, G.; García-García, E.; et al. Cannabinoid CB2 receptors in the mouse brain: Relevance for Alzheimer’s disease. J. Neuroinflammation 2018, 15, 158. [Google Scholar] [CrossRef] [PubMed]
- Palazuelos, J.; Davoust, N.; Julien, B.; Hatterer, E.; Aguado, T.; Mechoulam, R.; Benito, C.; Romero, J.; Silva, A.; Guzmán, M.; et al. The CB2 Cannabinoid Receptor Controls Myeloid Progenitor Trafficking. J. Biol. Chem. 2008, 283, 13320–13329. [Google Scholar] [CrossRef]
- Sisay, S.; Pryce, G.; Jackson, S.J.; Tanner, C.; Ross, R.A.; Michael, G.J.; Selwood, D.L.; Giovannoni, G.; Baker, D. Genetic Background Can Result in a Marked or Minimal Effect of Gene Knockout (GPR55 and CB2 Receptor) in Experimental Autoimmune Encephalomyelitis Models of Multiple Sclerosis. PLoS ONE 2013, 8, e76907. [Google Scholar] [CrossRef]
- Bouchard, J.; Truong, J.; Bouchard, K.; Dunkelberger, D.; Desrayaud, S.; Moussaoui, S.; Tabrizi, S.J.; Stella, N.; Muchowski, P.J. Cannabinoid Receptor 2 Signaling in Peripheral Immune Cells Modulates Disease Onset and Severity in Mouse Models of Huntington’s Disease. J. Neurosci. 2012, 32, 18259–18268. [Google Scholar] [CrossRef]
- Schmöle, A.-C.; Lundt, R.; Toporowski, G.; Hansen, J.N.; Beins, E.; Halle, A.; Zimmer, A. Cannabinoid Receptor 2-Deficiency Ameliorates Disease Symptoms in a Mouse Model with Alzheimer’s Disease-Like Pathology. J. Alzheimer’s Dis. 2018, 64, 379–392. [Google Scholar] [CrossRef]
- Zhang, H.-Y.; De Biase, L.; Chandra, R.; Shen, H.; Liu, Q.-R.; Gardner, E.; Lobo, M.K.; Xi, Z.-X. Repeated cocaine administration upregulates CB2 receptor expression in striatal medium-spiny neurons that express dopamine D1 receptors in mice. Acta Pharmacol. Sin. 2021, 43, 876–888. [Google Scholar] [CrossRef]
- Olabiyi, B.F.; Schmoele, A.-C.; Beins, E.C.; Zimmer, A. Pharmacological blockade of cannabinoid receptor 2 signaling does not affect LPS/IFN-γ-induced microglial activation. Sci. Rep. 2023, 13, 11105. [Google Scholar] [CrossRef]
- Boon, F.S.D.; Chameau, P.; Schaafsma-Zhao, Q.; van Aken, W.; Bari, M.; Oddi, S.; Kruse, C.G.; Maccarrone, M.; Wadman, W.J.; Werkman, T.R. Excitability of prefrontal cortical pyramidal neurons is modulated by activation of intracellular type-2 cannabinoid receptors. Proc. Natl. Acad. Sci. USA 2012, 109, 3534–3539. [Google Scholar] [CrossRef]
- Kim, J.; Li, Y. Chronic activation of CB2 cannabinoid receptors in the hippocampus increases excitatory synaptic transmission. J. Physiol. 2015, 593, 871–886. [Google Scholar] [CrossRef] [PubMed]
- Galán-Ganga, M.; Rodríguez-Cueto, C.; Merchán-Rubira, J.; Hernández, F.; Ávila, J.; Posada-Ayala, M.; Lanciego, J.L.; Luengo, E.; Lopez, M.G.; Rábano, A.; et al. Cannabinoid receptor CB2 ablation protects against TAU induced neurodegeneration. Acta Neuropathol. Commun. 2021, 9, 90. [Google Scholar] [CrossRef] [PubMed]
- Guenther, K.G.; Xu, Z.; Romero, J.; Hillard, C.J.; Mackie, K.; Hohmann, A.G. Conditional deletion of CB2 cannabinoid receptors from peripheral sensory neurons eliminates CB2-mediated antinociceptive efficacy in a mouse model of carrageenan-induced inflammatory pain. Neuropharmacology 2023, 237, 109601. [Google Scholar] [CrossRef]
- Xiong, W.; Cui, T.; Cheng, K.; Yang, F.; Chen, S.-R.; Willenbring, D.; Guan, Y.; Pan, H.-L.; Ren, K.; Xu, Y.; et al. Cannabinoids suppress inflammatory and neuropathic pain by targeting α3 glycine receptors. J. Exp. Med. 2012, 209, 1121–1134. [Google Scholar] [CrossRef]
- Klauke, A.-L.; Racz, I.; Pradier, B.; Markert, A.; Zimmer, A.; Gertsch, J. The cannabinoid CB2 receptor-selective phytocannabinoid beta-caryophyllene exerts analgesic effects in mouse models of inflammatory and neuropathic pain. Eur. Neuropsychopharmacol. 2014, 24, 608–620. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Guindon, J.; Cornett, B.L.; Makriyannis, A.; Mackie, K.; Hohmann, A.G. Chronic Cannabinoid Receptor 2 Activation Reverses Paclitaxel Neuropathy without Tolerance or Cannabinoid Receptor 1–Dependent Withdrawal. Biol. Psychiatry 2014, 77, 475–487. [Google Scholar] [CrossRef]
- Desroches, J.; Charron, S.; Bouchard, J.-F.; Beaulieu, P. Endocannabinoids decrease neuropathic pain-related behavior in mice through the activation of one or both peripheral CB1 and CB2 receptors. Neuropharmacology 2014, 77, 441–452. [Google Scholar] [CrossRef]
- Lin, X.; Xu, Z.; Carey, L.; Romero, J.; Makriyannis, A.; Hillard, C.J.; Ruggiero, E.; Dockum, M.; Houk, G.; Mackie, K.; et al. A peripheral CB2 cannabinoid receptor mechanism suppresses chemotherapy-induced peripheral neuropathy: Evidence from a CB2 reporter mouse. Pain 2021, 163, 834–851. [Google Scholar] [CrossRef]
- Ortega-Alvaro, A.; Aracil-Fernández, A.; García-Gutiérrez, M.S.; Navarrete, F.; Manzanares, J. Deletion of CB2 Cannabinoid Receptor Induces Schizophrenia-Related Behaviors in Mice. Neuropsychopharmacology 2011, 36, 1489–1504. [Google Scholar] [CrossRef]
- García-Gutiérrez, M.S.; Ortega-Álvaro, A.; Busquets-García, A.; Pérez-Ortiz, J.M.; Caltana, L.; Ricatti, M.J.; Brusco, A.; Maldonado, R.; Manzanares, J. Synaptic plasticity alterations associated with memory impairment induced by deletion of CB2 cannabinoid receptors. Neuropharmacology 2013, 73, 388–396. [Google Scholar] [CrossRef]
- Zhang, H.-Y.; Shen, H.; Gao, M.; Ma, Z.; Hempel, B.J.; Bi, G.-H.; Gardner, E.L.; Wu, J.; Xi, Z.-X. Cannabinoid CB2 receptors are expressed in glutamate neurons in the red nucleus and functionally modulate motor behavior in mice. Neuropharmacology 2021, 189, 108538. [Google Scholar] [CrossRef]
- Li, Y.; Kim, J. CB2 Cannabinoid Receptor Knockout in Mice Impairs Contextual Long-Term Memory and Enhances Spatial Working Memory. Neural Plast. 2016, 2016, 9817089. [Google Scholar] [CrossRef] [PubMed]
- Sigel, E.; Baur, R.; Rácz, I.; Marazzi, J.; Smart, T.G.; Zimmer, A.; Gertsch, J. The major central endocannabinoid directly acts at GABA A receptors. Proc. Natl. Acad. Sci. USA 2011, 108, 18150–18155. [Google Scholar] [CrossRef] [PubMed]
- Ward, S.J.; Castelli, F.; Reichenbach, Z.W.; Tuma, R.F. Surprising outcomes in cannabinoid CB1/CB2 receptor double knockout mice in two models of ischemia. Life Sci. 2017, 195, 1–5. [Google Scholar] [CrossRef]
- Tsuboi, K.; Okamoto, Y.; Ikematsu, N.; Inoue, M.; Shimizu, Y.; Uyama, T.; Wang, J.; Deutsch, D.G.; Burns, M.P.; Ulloa, N.M.; et al. Enzymatic formation of N-acylethanolamines from N-acylethanolamine plasmalogen through N-acylphosphatidylethanolamine-hydrolyzing phospholipase D-dependent and -independent pathways. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2011, 1811, 565–577. [Google Scholar] [CrossRef] [PubMed]
- Ueda, N.; Tsuboi, K.; Uyama, T. Metabolism of endocannabinoids and related N-acylethanolamines: Canonical and alternative pathways. FEBS J. 2013, 280, 1874–1894. [Google Scholar] [CrossRef]
- Howlett, A.C.; Abood, M.E. CB1 & CB2 Receptor Pharmacology. Adv. Pharmacol. 2017, 80, 169–206. [Google Scholar] [CrossRef] [PubMed]
- Leishman, E.; Mackie, K.; Luquet, S.; Bradshaw, H.B. Lipidomics profile of a NAPE-PLD KO mouse provides evidence of a broader role of this enzyme in lipid metabolism in the brain. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2016, 1861, 491–500. [Google Scholar] [CrossRef]
- Maldonado, R.; Robledo, P.; Berrendero, F. Endocannabinoid system and drug addiction: New insights from mutant mice approaches. Curr. Opin. Neurobiol. 2013, 23, 480–486. [Google Scholar] [CrossRef]
- Monory, K.; Lutz, B. Genetic models of the endocannabinoid system. Curr. Top. Behav. Neurosci. 2009, 1, 111–139. [Google Scholar] [CrossRef]
- Tanimura, A.; Yamazaki, M.; Hashimotodani, Y.; Uchigashima, M.; Kawata, S.; Abe, M.; Kita, Y.; Hashimoto, K.; Shimizu, T.; Watanabe, M.; et al. The Endocannabinoid 2-Arachidonoylglycerol Produced by Diacylglycerol Lipase α Mediates Retrograde Suppression of Synaptic Transmission. Neuron 2010, 65, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Vasilyev, D.V.; Goncalves, M.B.; Howell, F.V.; Hobbs, C.; Reisenberg, M.; Shen, R.; Zhang, M.-Y.; Strassle, B.W.; Lu, P.; et al. Loss of retrograde endocannabinoid signaling and reduced adult neurogenesis in diacylglycerol lipase knock-out mice. J. Neurosci. 2010, 30, 2017–2024. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, H.; Miyamae, T.; Hansen, G.; Zambrowicz, B.; Flynn, M.; Pedicord, D.; Blat, Y.; Westphal, R.S.; Zaczek, R.; Lewis, D.A.; et al. Postsynaptic diacylglycerol lipase α mediates retrograde endocannabinoid suppression of inhibition in mouse prefrontal cortex. J. Physiol. 2011, 589, 4857–4884. [Google Scholar] [CrossRef]
- Hsu, K.-L.; Tsuboi, K.; Adibekian, A.; Pugh, H.; Masuda, K.; Cravatt, B.F. DAGLβ inhibition perturbs a lipid network involved in macrophage inflammatory responses. Nat. Chem. Biol. 2012, 8, 999–1007. [Google Scholar] [CrossRef] [PubMed]
- Shonesy, B.C.; Bluett, R.J.; Ramikie, T.S.; Báldi, R.; Hermanson, D.J.; Kingsley, P.J.; Marnett, L.J.; Winder, D.G.; Colbran, R.J.; Patel, S. Genetic Disruption of 2-Arachidonoylglycerol Synthesis Reveals a Key Role for Endocannabinoid Signaling in Anxiety Modulation. Cell Rep. 2014, 9, 1644–1653. [Google Scholar] [CrossRef]
- Jenniches, I.; Ternes, S.; Albayram, O.; Otte, D.M.; Bach, K.; Bindila, L.; Michel, K.; Lutz, B.; Bilkei-Gorzo, A.; Zimmer, A. Anxiety, Stress, and Fear Response in Mice With Reduced Endocannabinoid Levels. Biol. Psychiatry 2016, 79, 858–868. [Google Scholar] [CrossRef]
- Bluett, R.J.; Báldi, R.; Haymer, A.; Gaulden, A.D.; Hartley, N.D.; Parrish, W.P.; Baechle, J.; Marcus, D.J.; Mardam-Bey, R.; Shonesy, B.C.; et al. Endocannabinoid signalling modulates susceptibility to traumatic stress exposure. Nat. Commun. 2017, 8, 14782. [Google Scholar] [CrossRef]
- Jain, T.; Wager-Miller, J.; Mackie, K.; Straiker, A. Diacylglycerol lipaseα (DAGLα) and DAGLβ cooperatively regulate the production of 2-Arachidonoyl glycerol in autaptic hippocampal neurons. Mol. Pharmacol. 2013, 84, 296–302. [Google Scholar] [CrossRef]
- Uchigashima, M.; Yamazaki, M.; Yamasaki, M.; Tanimura, A.; Sakimura, K.; Kano, M.; Watanabe, M. Molecular and morphological configuration for 2-Arachidonoylglycerol-mediated retrograde signaling at mossy cell–granule cell synapses in the dentate gyrus. J. Neurosci. 2011, 31, 7700–7714. [Google Scholar] [CrossRef]
- Sugaya, Y.; Cagniard, B.; Yamazaki, M.; Sakimura, K.; Kano, M. The Endocannabinoid 2-Arachidonoylglycerol Negatively Regulates Habituation by Suppressing Excitatory Recurrent Network Activity and Reducing Long-Term Potentiation in the Dentate Gyrus. J. Neurosci. 2013, 33, 3588–3601. [Google Scholar] [CrossRef]
- Aaltonen, N.; Ribas, C.R.; Lehtonen, M.; Savinainen, J.R.; Laitinen, J.T. Brain regional cannabinoid CB1 receptor signalling and alternative enzymatic pathways for 2-arachidonoylglycerol generation in brain sections of diacylglycerol lipase deficient mice. Eur. J. Pharm. Sci. 2014, 51, 87–95. [Google Scholar] [CrossRef]
- Reguero, L.; Puente, N.; Elezgarai, I.; Ramos-Uriarte, A.; Gerrikagoitia, I.; Bueno-López, J.-L.; Doñate, F.; Grandes, P. Subcellular localization of NAPE-PLD and DAGL-α in the ventromedial nucleus of the hypothalamus by a preembedding immunogold method. Histochem. 2013, 141, 543–550. [Google Scholar] [CrossRef]
- Schurman, L.D.; Carper, M.C.; Moncayo, L.V.; Ogasawara, D.; Richardson, K.; Yu, L.; Liu, X.; Poklis, J.L.; Liu, Q.-S.; Cravatt, B.F.; et al. Diacylglycerol Lipase-Alpha Regulates Hippocampal-Dependent Learning and Memory Processes in Mice. J. Neurosci. 2019, 39, 5949–5965. [Google Scholar] [CrossRef] [PubMed]
- Schlosburg, J.E.; Blankman, J.L.; Long, J.Z.; Nomura, D.K.; Pan, B.; Kinsey, S.G.; Nguyen, P.T.; Ramesh, D.; Booker, L.; Burston, J.J.; et al. Chronic monoacylglycerol lipase blockade causes functional antagonism of the endocannabinoid system. Nat. Neurosci. 2010, 13, 1113–1119. [Google Scholar] [CrossRef] [PubMed]
- Pan, B.; Wang, W.; Zhong, P.; Blankman, J.L.; Cravatt, B.F.; Liu, Q.-S. Alterations of Endocannabinoid Signaling, Synaptic Plasticity, Learning, and Memory in Monoacylglycerol Lipase Knock-out Mice. J. Neurosci. 2011, 31, 13420–13430. [Google Scholar] [CrossRef]
- Zhong, P.; Pan, B.; Gao, X.-P.; Blankman, J.L.; Cravatt, B.F.; Liu, Q.-S. Genetic deletion of monoacylglycerol lipase alters endocannabinoid-mediated retrograde synaptic depression in the cerebellum. J. Physiol. 2011, 589, 4847–4855. [Google Scholar] [CrossRef]
- Chanda, P.K.; Gao, Y.; Mark, L.; Btesh, J.; Strassle, B.W.; Lu, P.; Piesla, M.J.; Zhang, M.-Y.; Bingham, B.; Uveges, A.; et al. Monoacylglycerol Lipase Activity Is a Critical Modulator of the Tone and Integrity of the Endocannabinoid System. Mol. Pharmacol. 2010, 78, 996–1003. [Google Scholar] [CrossRef] [PubMed]
- Taschler, U.; Radner, F.P.; Heier, C.; Schreiber, R.; Schweiger, M.; Schoiswohl, G.; Preiss-Landl, K.; Jaeger, D.; Reiter, B.; Koefeler, H.C.; et al. Monoglyceride Lipase Deficiency in Mice Impairs Lipolysis and Attenuates Diet-induced Insulin Resistance. J. Biol. Chem. 2011, 286, 17467–17477. [Google Scholar] [CrossRef]
- Navia-Paldanius, D.; Aaltonen, N.; Lehtonen, M.; Savinainen, J.R.; Taschler, U.; Radner, F.P.; Zimmermann, R.; Laitinen, J.T. Increased tonic cannabinoid CB1R activity and brain region-specific desensitization of CB1R Gi/o signaling axis in mice with global genetic knockout of monoacylglycerol lipase. Eur. J. Pharm. Sci. 2015, 77, 180–188. [Google Scholar] [CrossRef]
- Imperatore, R.; Morello, G.; Luongo, L.; Taschler, U.; Romano, R.; De Gregorio, D.; Belardo, C.; Maione, S.; Di Marzo, V.; Cristino, L. Genetic deletion of monoacylglycerol lipase leads to impaired cannabinoid receptor CB1R signaling and anxiety-like behavior. J. Neurochem. 2015, 135, 799–813. [Google Scholar] [CrossRef]
- Tanimura, A.; Uchigashima, M.; Yamazaki, M.; Uesaka, N.; Mikuni, T.; Abe, M.; Hashimoto, K.; Watanabe, M.; Sakimura, K.; Kano, M. Synapse type-independent degradation of the endocannabinoid 2-arachidonoylglycerol after retrograde synaptic suppression. Proc. Natl. Acad. Sci. USA 2012, 109, 12195–12200. [Google Scholar] [CrossRef]
- Zhu, D.; Zhang, J.; Hashem, J.; Gao, F.; Chen, C. Inhibition of 2-arachidonoylglycerol degradation enhances glial immunity by single-cell transcriptomic analysis. J. Neuroinflamm. 2023, 20, 17. [Google Scholar] [CrossRef]
- Jung, K.-M.; Clapper, J.R.; Fu, J.; D’Agostino, G.; Guijarro, A.; Thongkham, D.; Avanesian, A.; Astarita, G.; Di Patrizio, N.V.; Frontini, A.; et al. 2-Arachidonoylglycerol Signaling in Forebrain Regulates Systemic Energy Metabolism. Cell Metab. 2012, 15, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Grabner, G.F.; Eichmann, T.O.; Wagner, B.; Gao, Y.; Farzi, A.; Taschler, U.; Radner, F.P.W.; Schweiger, M.; Lass, A.; Holzer, P.; et al. Deletion of Monoglyceride Lipase in Astrocytes Attenuates Lipopolysaccharide-induced Neuroinflammation. J. Biol. Chem. 2016, 291, 913–923. [Google Scholar] [CrossRef] [PubMed]
- Blankman, J.L.; Simon, G.M.; Cravatt, B.F. A Comprehensive Profile of Brain Enzymes that Hydrolyze the Endocannabinoid 2-Arachidonoylglycerol. Chem. Biol. 2007, 14, 1347–1356. [Google Scholar] [CrossRef]
- Zhao, S.; Mugabo, Y.; Iglesias, J.; Xie, L.; Delghingaro-Augusto, V.; Lussier, R.; Peyot, M.-L.; Joly, E.; Taïb, B.; Davis, M.A.; et al. α/β-Hydrolase Domain-6-Accessible Monoacylglycerol Controls Glucose-Stimulated Insulin Secretion. Cell Metab. 2014, 19, 993–1007. [Google Scholar] [CrossRef]
- Leung, D.; Saghatelian, A.; Simon, G.M.; Cravatt, B.F. Inactivation of N-Acyl Phosphatidylethanolamine Phospholipase D Reveals Multiple Mechanisms for the Biosynthesis of Endocannabinoids. Biochemistry 2006, 45, 4720–4726. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, L.; Harvey-White, J.; Huang, B.X.; Kim, H.-Y.; Luquet, S.; Palmiter, R.D.; Krystal, G.; Rai, R.; Mahadevan, A.; et al. Multiple pathways involved in the biosynthesis of anandamide. Neuropharmacology 2008, 54, 1–7. [Google Scholar] [CrossRef]
- Igarashi, M.; Watanabe, K.; Tsuduki, T.; Kimura, I.; Kubota, N. NAPE-PLD controls OEA synthesis and fat absorption by regulating lipoprotein synthesis in an in vitro model of intestinal epithelial cells. FASEB J. 2018, 33, 3167–3179. [Google Scholar] [CrossRef]
- Simon, G.M.; Cravatt, B.F. Characterization of mice lacking candidate N-acyl ethanolamine biosynthetic enzymes provides evidence for multiple pathways that contribute to endocannabinoid production in vivo. Mol. Biosyst. 2010, 6, 1411–1418. [Google Scholar] [CrossRef]
- Lee, H.-C.; Simon, G.M.; Cravatt, B.F. ABHD4 regulates multiple classes of N-Acyl Phospholipids in the mammalian central nervous system. Biochemistry 2015, 54, 2539–2549. [Google Scholar] [CrossRef]
- Murakami, M.; Takamiya, R.; Miki, Y.; Sugimoto, N.; Nagasaki, Y.; Suzuki-Yamamoto, T.; Taketomi, Y. Segregated functions of two cytosolic phospholipase A2 isoforms (cPLA2α and cPLA2ε) in lipid mediator generation. Biochem. Pharmacol. 2022, 203, 115176. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.K.; Hussain, Z.; Morito, K.; Takahashi, N.; Sikder, M.M.; Tanaka, T.; Ohta, K.-I.; Ueno, M.; Takahashi, H.; Yamamoto, T.; et al. Formation of N-acyl-phosphatidylethanolamines by cytosolic phospholipase A2ε in an ex vivo murine model of brain ischemia. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2022, 1867, 159222. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Metherel, A.H.; Kitson, A.P.; Alashmali, S.M.; Hopperton, K.E.; Trépanier, M.-O.; Jones, P.J.; Bazinet, R.P. Dietary fatty acids augment tissue levels of n-acylethanolamines in n-acylphosphatidylethanolamine phospholipase D (NAPE-PLD) knockout mice. J. Nutr. Biochem. 2018, 62, 134–142. [Google Scholar] [CrossRef]
- Inoue, M.; Tsuboi, K.; Okamoto, Y.; Hidaka, M.; Uyama, T.; Tsutsumi, T.; Tanaka, T.; Ueda, N.; Tokumura, A. Peripheral tissue levels and molecular species compositions of N-acyl-phosphatidylethanolamine and its metabolites in mice lacking N-acyl-phosphatidylethanolamine-specific phospholipase D. J. Biochem. 2017, 162, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Cravatt, B.F.; Demarest, K.; Patricelli, M.P.; Bracey, M.H.; Giang, D.K.; Martin, B.R.; Lichtman, A.H. Supersensitivity to anandamide and enhanced endogenous cannabinoid signaling in mice lacking fatty acid amide hydrolase. Proc. Natl. Acad. Sci. USA 2001, 98, 9371–9376. [Google Scholar] [CrossRef]
- Azad, S.C.; Monory, K.; Marsicano, G.; Cravatt, B.F.; Lutz, B.; Zieglgänsberger, W.; Rammes, G. Circuitry for associative plasticity in the amygdala involves endocannabinoid signaling. J. Neurosci. 2004, 24, 9953–9961. [Google Scholar] [CrossRef]
- Ortega-Gutiérrez, S.; Hawkins, E.G.; Viso, A.; López-Rodríguez, M.L.; Cravatt, B.F. Comparison of anandamide transport in FAAH wild-type and knockout neurons: Evidence for contributions by both FAAH and the CB1 receptor to anandamide uptake. Biochemistry 2004, 43, 8184–8190. [Google Scholar] [CrossRef]
- Lichtman, A.H.; Shelton, C.C.; Advani, T.; Cravatt, B.F. Mice lacking fatty acid amide hydrolase exhibit a cannabinoid receptor-mediated phenotypic hypoalgesia. Pain 2004, 109, 319–327. [Google Scholar] [CrossRef]
- Aguado, T.; Monory, K.; Palazuelos, J.; Stella, N.; Cravatt, B.; Lutz, B.; Marsicano, G.; Kokaia, Z.; Guzmán, M.; Galve-Roperh, I. The endocannabinoid system drives neural progenitor proliferation. FASEB J. 2005, 19, 1704–1706. [Google Scholar] [CrossRef]
- Patel, S.; Carrier, E.J.; Ho, W.-S.V.; Rademacher, D.J.; Cunningham, S.; Reddy, D.S.; Falck, J.R.; Cravatt, B.F.; Hillard, C.J. The postmortal accumulation of brain N-arachidonylethanolamine (anandamide) is dependent upon fatty acid amide hydrolase activity. J. Lipid Res. 2005, 46, 342–349. [Google Scholar] [CrossRef]
- Mulder, A.M.; Cravatt, B.F. Endocannabinoid metabolism in the absence of fatty acid amide hydrolase (FAAH): Discovery of phosphorylcholine derivatives of N-Acyl ethanolamines. Biochemistry 2006, 45, 11267–11277. [Google Scholar] [CrossRef] [PubMed]
- Saghatelian, A.; McKinney, M.K.; Bandell, M.; Patapoutian, A.; Cravatt, B.F. A FAAH-regulated class of N-Acyl taurines that activates TRP Ion channels. Biochemistry 2006, 45, 9007–9015. [Google Scholar] [CrossRef] [PubMed]
- Mallet, C.; Barrière, D.A.; Ermund, A.; Jönsson, B.A.G.; Eschalier, A.; Zygmunt, P.M.; Högestätt, E.D. TRPV1 in Brain Is Involved in Acetaminophen-Induced Antinociception. PLoS ONE 2010, 5, e12748. [Google Scholar] [CrossRef] [PubMed]
- Long, J.Z.; LaCava, M.; Jin, X.; Cravatt, B.F. An anatomical and temporal portrait of physiological substrates for fatty acid amide hydrolase. J. Lipid Res. 2011, 52, 337–344. [Google Scholar] [CrossRef]
- Mathur, B.N.; Tanahira, C.; Tamamaki, N.; Lovinger, D.M. Voltage drives diverse endocannabinoid signals to mediate striatal microcircuit-specific plasticity. Nat. Neurosci. 2013, 16, 1275–1283. [Google Scholar] [CrossRef]
- Musella, A.; De Chiara, V.; Rossi, S.; Prosperetti, C.; Bernardi, G.; Maccarrone, M.; Centonze, D. TRPV1 channels facilitate glutamate transmission in the striatum. Mol. Cell Neurosci. 2009, 40, 89–97. [Google Scholar] [CrossRef]
- Carey, L.M.; Slivicki, R.A.; Leishman, E.; Cornett, B.; Mackie, K.; Bradshaw, H.; Hohmann, A.G. A pro-nociceptive phenotype unmasked in mice lacking fatty-acid amide hydrolase. Mol. Pain 2016, 12, 1744806916649192. [Google Scholar] [CrossRef]
- Cravatt, B.F.; Saghatelian, A.; Hawkins, E.G.; Clement, A.B.; Bracey, M.H.; Lichtman, A.H. Functional disassociation of the central and peripheral fatty acid amide signaling systems. Proc. Natl. Acad. Sci. USA 2004, 101, 10821–10826. [Google Scholar] [CrossRef]
- Xie, X.; Li, Y.; Xu, S.; Zhou, P.; Yang, L.; Xu, Y.; Qiu, Y.; Yang, Y.; Li, Y. Genetic Blockade of NAAA Cell-specifically Regulates Fatty Acid Ethanolamides (FAEs) Metabolism and Inflammatory Responses. Front. Pharmacol. 2022, 12, 817603. [Google Scholar] [CrossRef]
- Bilsland, L.G.; Dick, J.R.T.; Pryce, G.; Petrosino, S.; Di Marzo, V.; Baker, D.; Greensmith, L. Increasing cannabinoid levels by pharmacological and genetic manipulation delays disease progression in SOD1 mice. FASEB J. 2006, 20, 1003–1005. [Google Scholar] [CrossRef] [PubMed]
- Webb, M.; Luo, L.; Ma, J.Y.; Tham, C.-S. Genetic deletion of Fatty Acid Amide Hydrolase results in improved long-term outcome in chronic autoimmune encephalitis. Neurosci. Lett. 2008, 439, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Clement, A.B.; Hawkins, E.G.; Lichtman, A.H.; Cravatt, B.F. Increased seizure susceptibility and proconvulsant activity of anandamide in mice lacking fatty acid amide hydrolase. J. Neurosci. 2003, 23, 3916–3923. [Google Scholar] [CrossRef]
- Sugaya, Y.; Yamazaki, M.; Uchigashima, M.; Kobayashi, K.; Watanabe, M.; Sakimura, K.; Kano, M. Crucial Roles of the Endocannabinoid 2-Arachidonoylglycerol in the Suppression of Epileptic Seizures. Cell Rep. 2016, 16, 1405–1415. [Google Scholar] [CrossRef]
- Wilkerson, J.L.; Donvito, G.; Grim, T.W.; Abdullah, R.A.; Ogasawara, D.; Cravatt, B.F.; Lichtman, A.H. Investigation of Diacylglycerol Lipase Alpha Inhibition in the Mouse Lipopolysaccharide Inflammatory Pain Model. Experiment 2017, 363, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yang, N.; Dong, J.; Tian, W.; Chang, L.; Ma, J.; Guo, J.; Tan, J.; Dong, A.; He, K.; et al. Deficiency in endocannabinoid synthase DAGLB contributes to early onset Parkinsonism and murine nigral dopaminergic neuron dysfunction. Nat. Commun. 2022, 13, 3490. [Google Scholar] [CrossRef] [PubMed]
- Kondev, V.; Bluett, R.; Najeed, M.; Rosas-Vidal, L.E.; Grueter, B.A.; Patel, S. Ventral hippocampal diacylglycerol lipase-alpha deletion decreases avoidance behaviors and alters excitation-inhibition balance. Neurobiol. Stress 2023, 22, 100510. [Google Scholar] [CrossRef]
- Moreira, F.A.; Kaiser, N.; Monory, K.; Lutz, B. Reduced anxiety-like behaviour induced by genetic and pharmacological inhibition of the endocannabinoid-degrading enzyme fatty acid amide hydrolase (FAAH) is mediated by CB1 receptors. Neuropharmacology 2008, 54, 141–150. [Google Scholar] [CrossRef]
- Basavarajappa, B.S.; Yalamanchili, R.; Cravatt, B.F.; Cooper, T.B.; Hungund, B.L. Increased ethanol consumption and preference and decreased ethanol sensitivity in female FAAH knockout mice. Neuropharmacology 2006, 50, 834–844. [Google Scholar] [CrossRef]
- Vinod, K.Y.; Sanguino, E.; Yalamanchili, R.; Manzanares, J.; Hungund, B.L. Manipulation of fatty acid amide hydrolase functional activity alters sensitivity and dependence to ethanol. J. Neurochem. 2007, 104, 233–243. [Google Scholar] [CrossRef]
- Ramesh, D.; Ross, G.R.; Schlosburg, J.E.; Owens, R.A.; Abdullah, R.A.; Kinsey, S.G.; Long, J.Z.; Nomura, D.K.; Sim-Selley, L.J.; Cravatt, B.F.; et al. Blockade of Endocannabinoid Hydrolytic Enzymes Attenuates Precipitated Opioid Withdrawal Symptoms in Mice. Experiment 2011, 339, 173–185. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, L.D.; Smith, T.L.; Donvito, G.; Cravatt, B.F.; Newton, J.; Spiegel, S.; Reeves, T.M.; Phillips, L.L.; Lichtman, A.H. Diacylglycerol Lipase-β Knockout Mice Display a Sex-Dependent Attenuation of Traumatic Brain Injury-Induced Mortality with No Impact on Memory or Other Functional Consequences. Cannabis Cannabinoid Res. 2021, 6, 508–521. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Zhu, D.; Zhang, J.; Gao, F.; Hashem, J.; Kingsley, P.; Marnett, L.J.; Mackie, K.; Chen, C. Enhancing endocannabinoid signalling in astrocytes promotes recovery from traumatic brain injury. Brain 2022, 145, 179–193. [Google Scholar] [CrossRef] [PubMed]
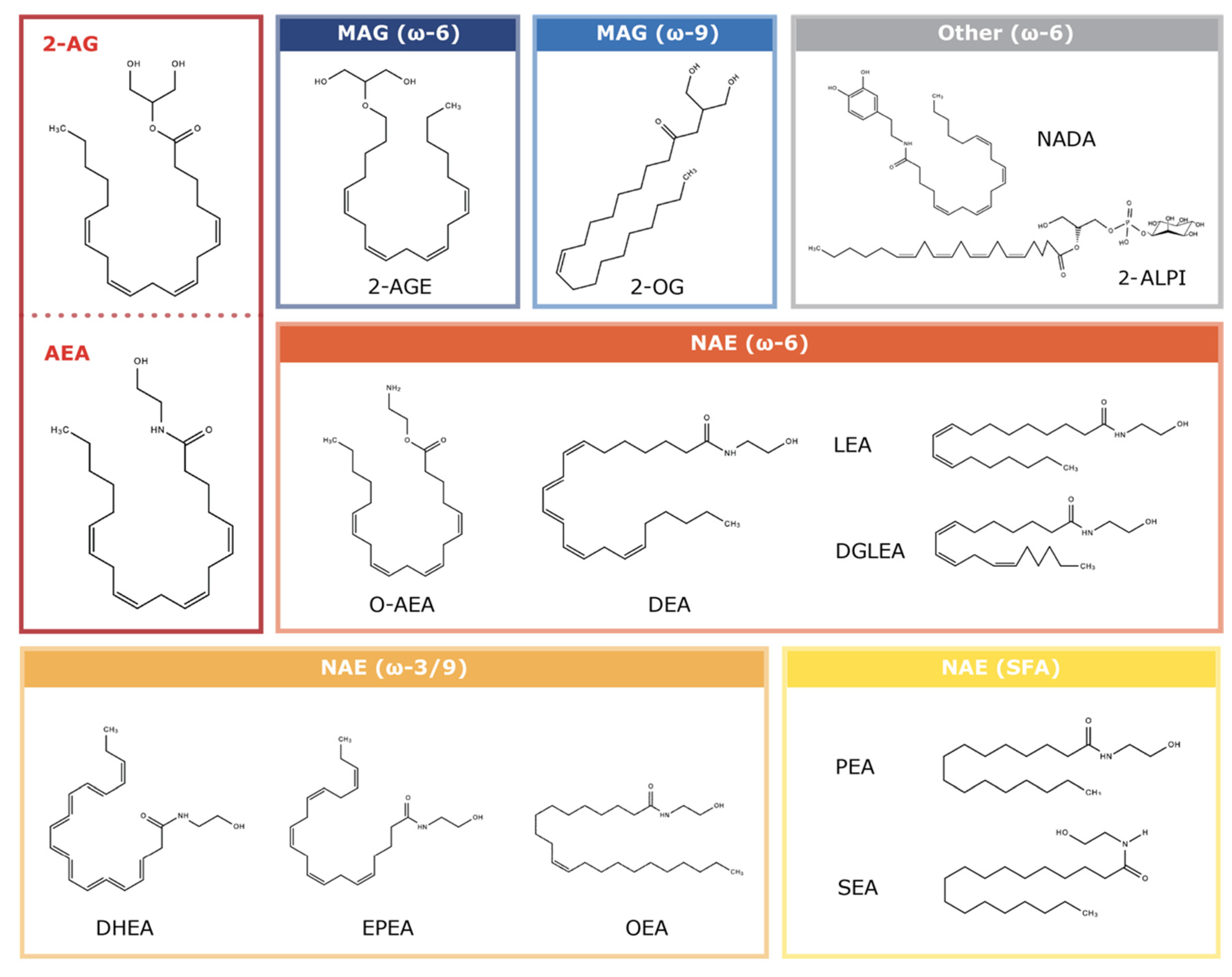
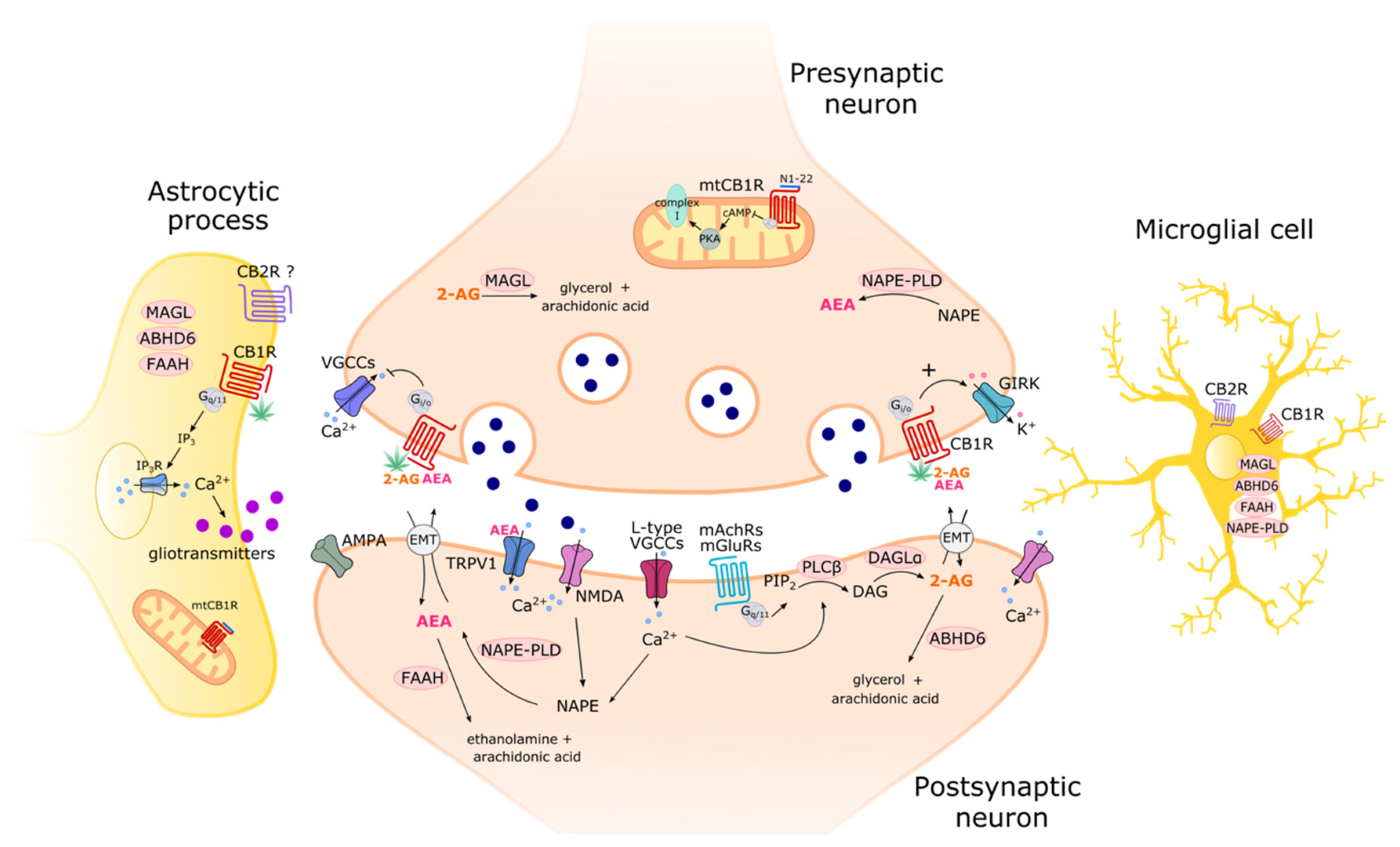

| Cannabinoid Receptors |
| CB1R, CB2R, GPR55, GPR18, GPR119, TRPs, PPARs |
| Endocannabinoids |
| 2-AG, AEA, PEA, DHEA, EPEA, OEA, NADA, 2-AGE, O-AEA |
| Enzymes |
| Biosynthesis: DAGLα/β, NAPE-PLD, ABHD4, GDE1, PLC, PLA, PTN22, SHIP1 Degradation: FAAH, MAGL, NAAA, ABHD6/12, COX-2, CYP2-4, 12/15-LO Transport process: EMT, FABP5/7, SCP2/x |
| Technique | Advantages | Disadvantages | ECS Components |
|---|---|---|---|
| Radioligands | |||
| Radioactive ligands coupled with PET 1, autoradiography, radioligand binding assays |
|
| CB1R, CB2R, enzymes |
| Optical microscopy | |||
| Fluorescence light microscopy (Immunocytochemistry and immunohistochemistry) |
|
| CB1R, CB2R, enzymes |
| FRET 2 |
|
| CB1R, CB2R |
| Electron microscopy | |||
| TEM 3 |
|
| CB1R, CB2R, enzymes |
| Super-resolution microscopy | |||
| STORM 4 |
|
| CB1R |
| Mass spectrometry imaging (MSI) | |||
| MALDI 5 |
|
| eCB |
| Bioengineered sensors | |||
| GRABeCB2.0 6 |
|
| eCB |
| Technique | Advantages | Disadvantages | References |
|---|---|---|---|
| Mass spectrometry imaging | |||
| MALDI 1 |
|
| [88] |
| Bioengineered sensors | |||
| GRABeCB2.0 2 |
|
| [89] |
| Zebrafish Stage | Genetic Approach | Technique | Outcome | References |
|---|---|---|---|---|
| Juvenile | cnr1 KO (total) | Morpholino | Defects in axonal growth and fasciculation | [128] |
| Juvenile | cnr1 and cnr2 KO (total) | Morpholino | CART-3 1 expression and yolk sac size | [132] |
| Juvenile | cnr1 KO (total) | Morpholino | Decreased locomotor activity and suppression of feeding behaviour | [133] |
| Juvenile Adult | cnr1 overexpression (hepatic) | Tetoff transgenic system | Loss of lipid accumulation | [131] |
| Juvenile | cnr2 KO (total) | CoDA ZFN 2 | Regulation of leukocyte migration | [134] |
| Juvenile | cnr2 KO (total) | Morpholino | Reduced runx1 expression and decreased HSCs 3 | [135] |
| Juvenile | cnr1 KO (total) | Morpholino | Reduced microRNA dre-let-7d levels | [136] |
| Juvenile Adult | cnr1 and cnr2 KO | Morpholino TALEN 4 | Smaller livers with fewer hepatocytes, reduced liver-specific gene expression and proliferation | [134] |
| Juvenile Adult | cnrip1a and cnrip1b (total) | CRISPR 5/Cas9 | No phenotype is detected, fish lacking these genes both maternally and zygotically are viable. | [137] |
| Juvenile | cnr2 KO (total) | CRISPR/Cas9 | Mutants show an anxiety-like behaviour: they show an altered PDR 6 and decreased CO 7 | [138] |
| Juvenile | cnr1 KO (total) | Morpholino | Reduced number of GnRH3 neurons, fibre misrouting, and altered fasciculation | [139] |
| Disorder | Model | Outcome | References |
|---|---|---|---|
| ALS 1 | FAAH−/− × SOD1-Tg | Attenuated symptoms; no increase in lifespan (CB1R-independent) | [308] |
| Diet-induced obesity | ABHD6−/− | No changes in total MAG content | [283] |
| ABHD6−/− | Increased 2-AG/CB1R signalling drives weight gain and hypothermia | [33] | |
| CaMKII:MAGLTg | MAGL overexpression; decreased 2-AG correlated to weight loss and hyperthermia | [280] | |
| EAE 2 model of MS 3 | FAAH−/− | Enhanced recovery/remission from EAE 2 | [309] |
| Kainate-induced epilepsy | FAAH−/− | Increased seizure susceptibility | [310] |
| DAGLα−/− Tanimura | Decreased CB1R DSI 4; increased seizure incidence | [311] | |
| Pain and neuroinflammation (LPS 5 induced) | GFAP:MAGL−/− | Reduced neuroinflammation independent of CB1R and total 2-AG | [281] |
| DAGLα−/− Gao | No effect on pain sensitization | [312] | |
| DAGLβ-GTLex; gene trap | Reduced pain sensitization | [312] | |
| Parkinson’s disease | MAGL-GTLex; gene trap | Reduced neuroinflammation; neuroprotection; prostaglandin and not ECS related | [32] |
| DAGLβ-GTLex; gene trap | DAGLβ is main 2-AG synthesizer in SN 6; contributes to disease progression in mice and rare-variant patients | [313] | |
| Stress + anxiety | DAGLαfl/fl | Decreased stress resilience (BLA 7 AAV 8-directed) | [264] |
| DAGLαfl/fl | Reduced stress behaviour | [314] | |
| DAGLα−/− | Increased anxiety; anhedonia | [262] | |
| DAGLα−/− | Increased fear, anxiety; loss of maternal care | [263] | |
| FAAH−/− | Reduced anxiety | [315] | |
| Substance use disorders | FAAH−/− | Increased ethanol consumption and preference | [316] |
| FAAH−/− | Increased alcohol sensitivity and withdrawal | [317] | |
| FAAH−/− | Reduced morphine withdrawal | [318] | |
| TBI 9 | DAGLβ-GTLex; gene trap | Sex-dependant increase in survival; attenuated sphingolipid TBI 9 markers | [319] |
| GFAP:MAGLflox/flox | Reduced neuroinflammation, neuroprotection, CB1R-PPARγ-dependent | [320] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kouchaeknejad, A.; Van Der Walt, G.; De Donato, M.H.; Puighermanal, E. Imaging and Genetic Tools for the Investigation of the Endocannabinoid System in the CNS. Int. J. Mol. Sci. 2023, 24, 15829. https://doi.org/10.3390/ijms242115829
Kouchaeknejad A, Van Der Walt G, De Donato MH, Puighermanal E. Imaging and Genetic Tools for the Investigation of the Endocannabinoid System in the CNS. International Journal of Molecular Sciences. 2023; 24(21):15829. https://doi.org/10.3390/ijms242115829
Chicago/Turabian StyleKouchaeknejad, Armin, Gunter Van Der Walt, Maria Helena De Donato, and Emma Puighermanal. 2023. "Imaging and Genetic Tools for the Investigation of the Endocannabinoid System in the CNS" International Journal of Molecular Sciences 24, no. 21: 15829. https://doi.org/10.3390/ijms242115829







