The SARS-CoV-2 Virus and the Cholinergic System: Spike Protein Interaction with Human Nicotinic Acetylcholine Receptors and the Nicotinic Agonist Varenicline
Abstract
:1. Introduction
2. Results
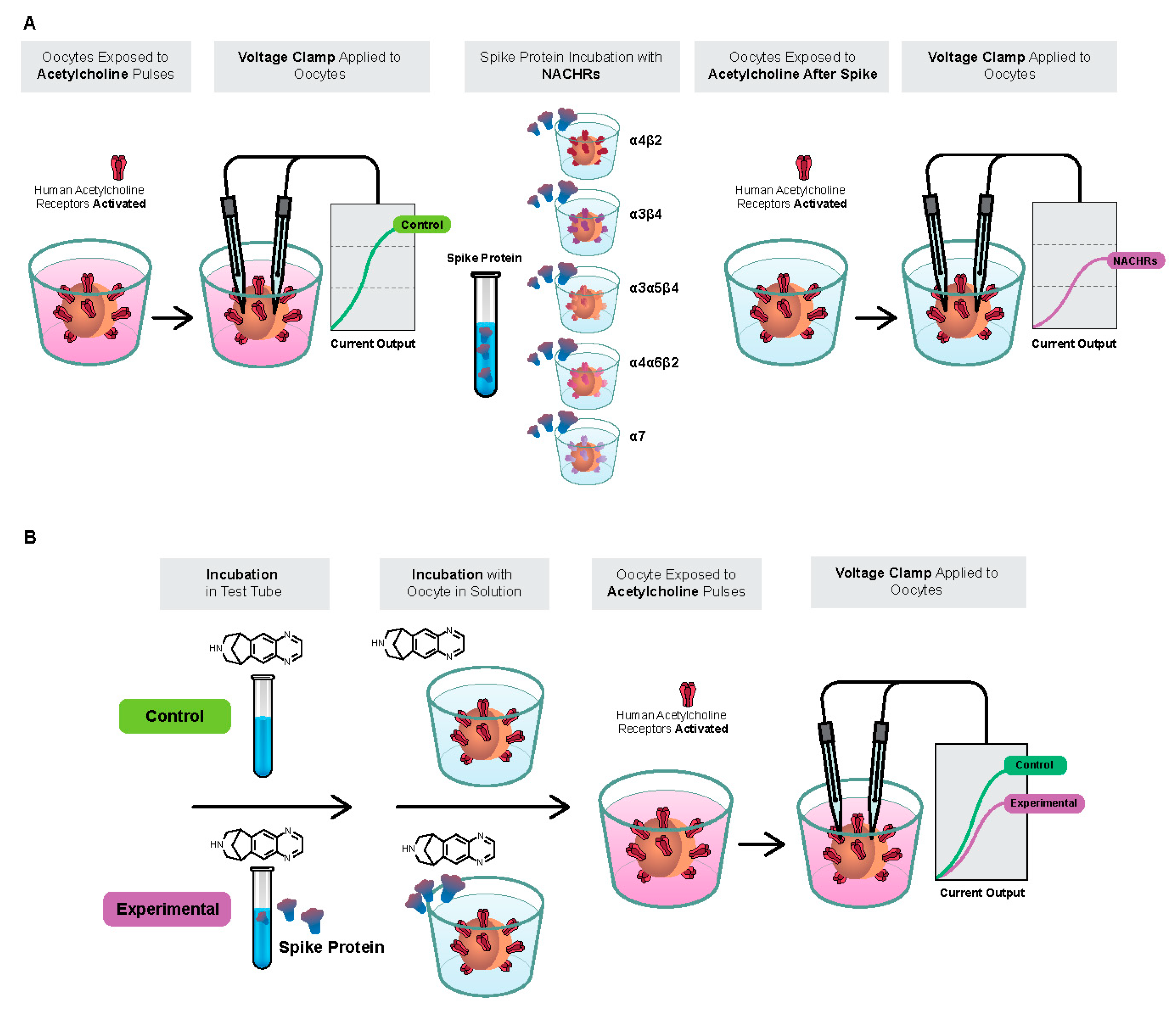
2.1. Spike-RBD Interacts with the Human α4β2 and α4α6β2 nAChRs and Possibly the α3α5β4 nAChR, but Not the α3β4 and α7 nAChRs
2.2. The S2 Subunit of the S Protein Interacts with the Human α4β2 nAChR, but Not the α4α6β2, α3β4, and α3α5β4 nAChRs
2.3. The S1 Subunit of the S Protein Interacts with the Human α4β2 nAChRs at an Allosteric Binding Site That Inhibits Receptor Activity
2.4. The SARS-CoV-2 B.1.1.529 Spike-RBD Protein Interacts with the Human α4β2 nAChR at an Allosteric Binding Site That Potentiates Receptor Activity
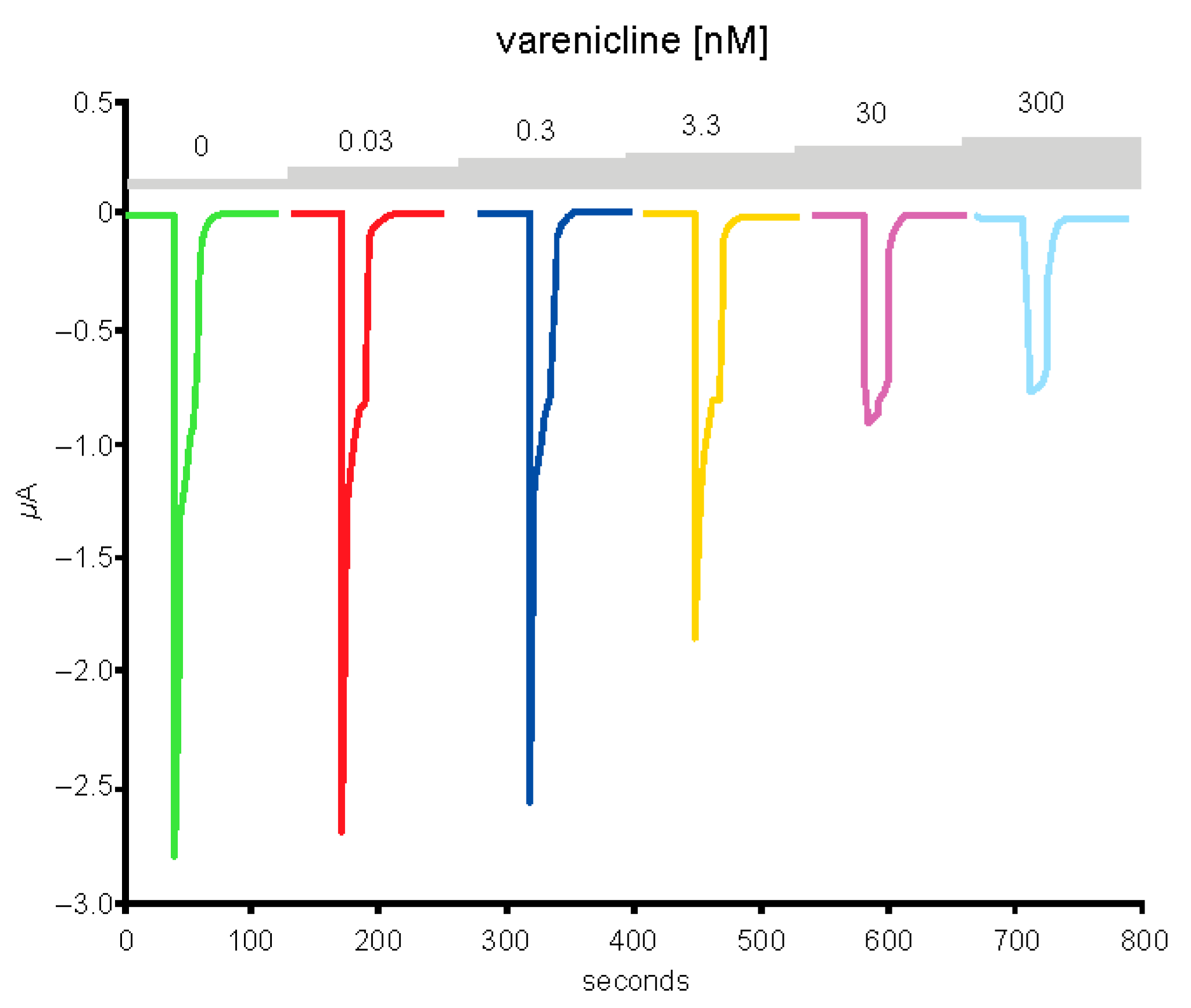
2.5. Preincubation of Varenicline with Spike-RBD Indicates That Varenicline Inhibition of the Human α4β2 nAChR Is Modified by Spike-RBD
2.6. The SARS-CoV-2 B.A.1 (Omicron) Spike-RBD Protein Interacts with the Human α4β2 nAChRs at an Allosteric Binding Site That Potentiates Receptor Activity
3. Discussion
4. Materials and Methods
4.1. Human nAChRs
4.2. SARS-CoV-2 S Protein
4.3. Varenicline Tartrate
4.4. Electrophysiological Recordings
4.5. SARS-CoV-2 B.1.1.7 S Protein Interaction with Human nAChR Subtypes
4.6. SARS-CoV-2 B.1.1.529 (Omicron Variant) S Protein Interaction with Human α4β2 nAChRs
4.7. Varenicline Interaction with Spike-RBD and SARS-CoV-2 B.1.1.529 Spike-RBD
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rajpal, V.R.; Sharma, S.; Sehgal, D.; Singh, A.; Kumar, A.; Vaishnavi, S.; Tiwari, M.; Bhalla, H.; Goel, S.; Raina, S.N. A comprehensive account of SARS-CoV-2 genome structure, incurred mutations, lineages and COVID-19 vaccination program. Future Virol. 2022; ahead of print. [Google Scholar] [CrossRef]
- Thapa Magar, S.; Lokhandwala, H.I.; Batool, S.; Zahoor, F.; Zaidi, S.K.F.; Sahtiya, S.; Khemani, D.; Kumar, S.; Voloshyna, D.; Saleem, F.; et al. A systematic review of neurological manifestations of COVID-19. Cureus 2022, 14, e28309. [Google Scholar] [CrossRef] [PubMed]
- Bertram, S.; Dijkman, R.; Habjan, M.; Heurich, A.; Gierer, S.; Glowacka, I.; Welsch, K.; Winkler, M.; Schneider, H.; Hofmann-Winkler, H.; et al. TMPRSS2 activates the human coronavirus 229E for cathepsin-independent host cell entry and is expressed in viral target cells in the respiratory epithelium. J. Virol. 2013, 87, 6150–6160. [Google Scholar] [CrossRef]
- Xia, S.; Zhu, Y.; Liu, M.; Lan, Q.; Xu, W.; Wu, Y.; Ying, T.; Liu, S.; Shi, Z.; Jiang, S.; et al. Fusion mechanism of 2019-nCoV and fusion inhibitors targeting HR1 domain in spike protein. Cell Mol. Immunol. 2020, 17, 765–767. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Cao, J.; Zhang, X.; Gao, H.; Wang, Y.; Wang, J.; He, J.; Jiang, X.; Zhang, J.; Shen, G.; et al. Receptome profiling identifies KREMEN1 and ASGR1 as alternative functional receptors of SARS-CoV-2. Cell Res. 2022, 32, 24–37. [Google Scholar] [CrossRef] [PubMed]
- Lagoumintzis, G.; Chasapis, C.T.; Alexandris, N.; Kouretas, D.; Tzartos, S.; Eliopoulos, E.; Farsalinos, K.; Poulas, K. Nicotinic cholinergic system and COVID-19: In silico identification of interactions between α7 nicotinic acetylcholine receptor and the cryptic epitopes of SARS-Co-V and SARS-CoV-2 spike glycoproteins. Food Chem. Toxicol. 2021, 149, 112009. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.S.F.; Ibarra, A.A.; Bermudez, I.; Casalino, L.; Gaieb, Z.; Shoemark, D.K.; Gallagher, T.; Sessions, R.B.; Amaro, R.E.; Mullholland, A.J. Simulations support the interaction of the SARS-CoV-2 spike protein with nicotinic acetylcholine receptors. bioRxiv, 2020; ahead of print. [Google Scholar] [CrossRef]
- Farsalinos, K.; Niaura, R.; Le Houezec, J.; Barbouni, A.; Tsatsakis, A.; Kouretas, D.; Vantarakis, A.; Poulas, K. Editorial: Nicotine and SARS-CoV-2: COVID-19 may be a disease of the nicotinic cholinergic system. Toxicol. Rep. 2020, 7, 658–663. [Google Scholar] [CrossRef]
- Farsalinos, K.; Barbouni, A.; Niaura, R. Systematic review of the prevalence of current smoking among hospitalized COVID-19 patients in China: Could nicotine be a therapeutic option? Reply. Intern. Emerg. Med. 2021, 16, 235–236. [Google Scholar] [CrossRef]
- Farsalinos, K.; Barbouni, A.; Poulas, K.; Polosa, R.; Caponnetto, P.; Niaura, R. Current smoking, former smoking, and adverse outcome among hospitalized COVID-19 patients: A systematic review and meta-analysis. Ther. Adv. Chronic. Dis. 2020, 11, 2040622320935765. [Google Scholar] [CrossRef]
- Farsalinos, K.; Bagos, P.G.; Giannouchos, T.; Niaura, R.; Barbouni, A.; Poulas, K. Smoking prevalence among hospitalized COVID-19 patients and its association with disease severity and mortality: An expanded re-analysis of a recent publication. Harm. Reduct. J. 2021, 18, 9. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.S.F.; Ibarra, A.A.; Bermudez, I.; Casalino, L.; Gaieb, Z.; Shoemark, D.K.; Gallagher, T.; Sessions, R.B.; Amaro, R.E.; Mulholland, A.J. A potential interaction between the SARS-CoV-2 spike protein and nicotinic acetylcholine receptors. Biophys. J. 2021, 120, 983–993. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, G.K.; Williams, M. Neuronal nicotinic acetylcholine receptors as novel drug targets. J. Pharm. Exp. 2000, 292, 461–467. [Google Scholar]
- Nau, J.; Luthra, P.; Lanzer, K.; Szaba, F.; Cookenham, T.; Carlson, E. Varenicline prevents SARS-CoV-2 infection in vitro and in rhesus macaques. bioRxiv, 2021; ahead of print. [Google Scholar] [CrossRef]
- Lagoumintzis, G.; Chasapis, C.T.; Alexandris, N.; Tzartos, S.; Eliopoulis, E.; Farsalinos, K.; Poulas, K. COVID-19 and cholinergic anti-inflammatory pathway: In silico identification of an interaction between α7 nicotinic acetylcholine receptor and the cryptic epitopes of SARS-CoV and SARS-CoV-2 spike glycoproteins. bioRxiv, 2020; ahead of print. [Google Scholar] [CrossRef]
- Grant, S.N.; Lester, H.A. Regulation of epithelial sodium channel activity by SARS-CoV-1 and SARS-CoV-2 proteins. Biophys. J. 2021, 120, 2805–2813. [Google Scholar] [CrossRef]
- Godellas, N.E.; Cymes, G.D.; Grosman, C. An experimental test of the nicotinic hypothesis of COVID-19. Proc. Natl. Acad. Sci. USA 2022, 119, e2204242119. [Google Scholar] [CrossRef]
- Sudre, C.H.; Murray, B.; Varsavsky, T.; Graham, M.S.; Penfold, R.S.; Bowyer, R.C.; Pujol, J.C.; Klaser, K.; Antonelli, M.; Canas, L.S.; et al. Attributes and predictors of long COVID. Nat. Med. 2021, 27, 626–631. [Google Scholar] [CrossRef]
- D’Souza, R.D.; Parsa, P.V.; Vijayaraghavan, S. Nicotinic receptors modulate olfactory bulb external tufted cells via an excitation-dependent inhibitory mechanism. J. Neurophysiol. 2013, 110, 1544–1553. [Google Scholar] [CrossRef]
- Qian, J.; Mummalaneni, S.; Larsen, J.; Grider, J.R.; Spielman, A.I.; Ozdener, M.H.; Lyall, V. Nicotinic acetylcholine receptor (CHRN) expression and function in cultured human adult fungiform (HBO) taste cells. PLoS One 2018, 13, e0194089. [Google Scholar] [CrossRef]
- Fotedar, S.; Fotedar, V. Green tobacco sickness: A brief review. Indian J. Occup. Env. Med. 2017, 21, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Finsterer, J.; Scorza, F.A. Perspectives of neuro-COVID: Myasthenia. Front. Neurol. 2021, 12, 635747. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Salinas, G.L.; Martínez-Archundia, M.; Correa-Basurto, J.; García-Machorro, J. Repositioning of ligands that target the spike glycoprotein as potential drugs for SARS-CoV-2 in an in silico study. Molecules 2020, 25, 5615. [Google Scholar] [CrossRef] [PubMed]
- Hogg, R.C.; Bandelier, F.; Benoit, A.; Dosch, R.; Bertrand, D. An automated system for intracellular and intranuclear injection. J. Neurosci. Methods 2008, 169, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Rollema, H.; Coe, J.W.; Chambers, L.K.; Hurst, R.S.; Stahl, S.M.; Williams, K.E. Rationale, pharmacology and clinical efficacy of partial agonists of α4β2 nACh receptors for smoking cessation. Trends Pharm. Sci. 2007, 28, 316–325. [Google Scholar] [CrossRef]









Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carlson, E.C.; Macsai, M.; Bertrand, S.; Bertrand, D.; Nau, J. The SARS-CoV-2 Virus and the Cholinergic System: Spike Protein Interaction with Human Nicotinic Acetylcholine Receptors and the Nicotinic Agonist Varenicline. Int. J. Mol. Sci. 2023, 24, 5597. https://doi.org/10.3390/ijms24065597
Carlson EC, Macsai M, Bertrand S, Bertrand D, Nau J. The SARS-CoV-2 Virus and the Cholinergic System: Spike Protein Interaction with Human Nicotinic Acetylcholine Receptors and the Nicotinic Agonist Varenicline. International Journal of Molecular Sciences. 2023; 24(6):5597. https://doi.org/10.3390/ijms24065597
Chicago/Turabian StyleCarlson, Eric C., Marian Macsai, Sonia Bertrand, Daniel Bertrand, and Jeffrey Nau. 2023. "The SARS-CoV-2 Virus and the Cholinergic System: Spike Protein Interaction with Human Nicotinic Acetylcholine Receptors and the Nicotinic Agonist Varenicline" International Journal of Molecular Sciences 24, no. 6: 5597. https://doi.org/10.3390/ijms24065597






