Chronic Administration of Non-Constitutive Proteasome Inhibitor Modulates Long-Term Potentiation and Glutamate Signaling-Related Gene Expression in Murine Hippocampus
Abstract
:1. Introduction
2. Results
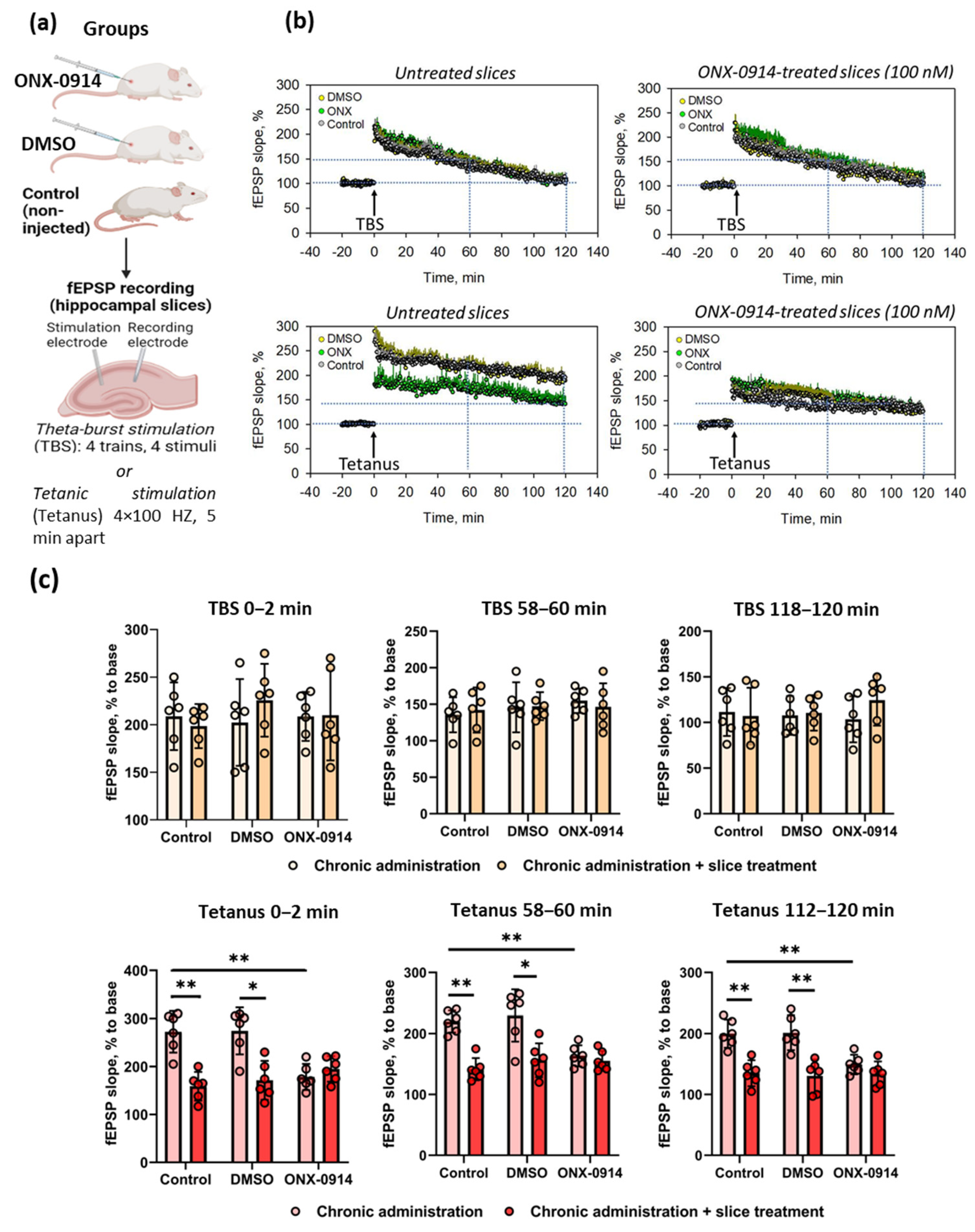
2.1. Chronic Administration of ONX-0914 Modulates LTP in the Hippocampus Following Tetanic Stimulation, but Not Short Theta-Burst Stimulation
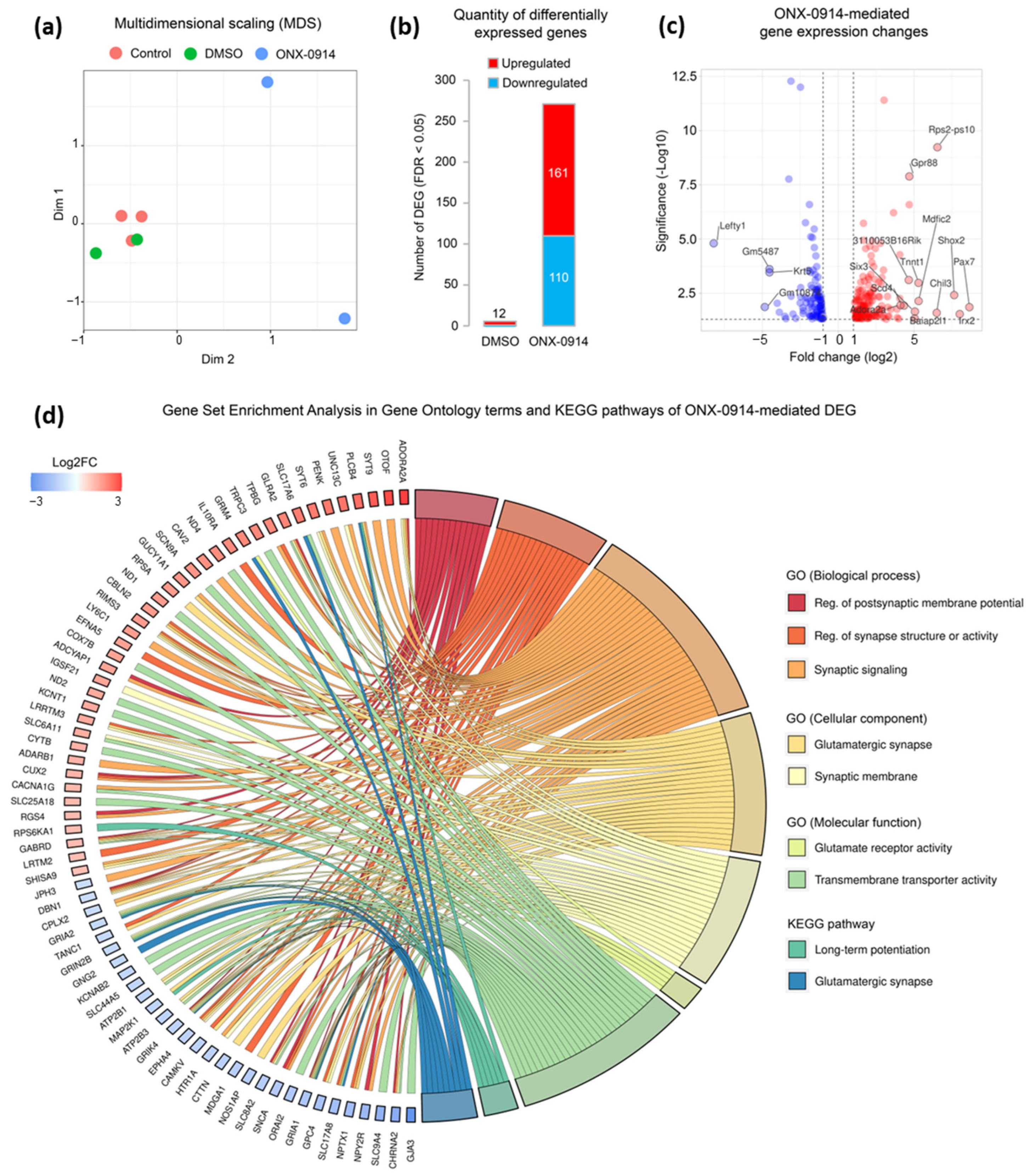
2.2. Chronic Administration of ONX-0914 Results in Altered Expression of Glutamate Signaling-Related Genes
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Administration of ONX-0914
4.3. Preparation and Treatment of Hippocampal Slices
4.4. Electrophysiology
4.5. RNA Extraction and RNA-Seq
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Morozov, A.V.; Karpov, V.L. Biological consequences of structural and functional proteasome diversity. Heliyon 2018, 4, e00894. [Google Scholar] [CrossRef] [PubMed]
- Guillaume, B.; Chapiro, J.; Stroobant, V.; Colau, D.; Van Holle, B.; Parvizi, G.; Bousquet-Dubouch, M.P.; Théate, I.; Parmentier, N.; Van den Eynde, B.J. Two abundant proteasome subtypes that uniquely process some antigens presented by HLA class I molecules. Proc. Natl. Acad. Sci. USA 2010, 107, 18599–18604. [Google Scholar] [CrossRef] [PubMed]
- Ferrington, D.A.; Gregerson, D.S. Immunoproteasomes: Structure, function, and antigen presentation. Prog. Mol. Biol. Transl. Sci. 2012, 109, 75–112. [Google Scholar] [PubMed]
- Díaz-Hernández, M.; Hernández, F.; Martín-Aparicio, E.; Gómez-Ramos, P.; Morán, M.A.; Castaño, J.G.; Ferrer, I.; Avila, J.; Lucas, J.J. Neuronal induction of the immunoproteasome in Huntington’s disease. J. Neurosci. 2003, 23, 11653–11661. [Google Scholar] [CrossRef] [PubMed]
- Orre, M.; Kamphuis, W.; Dooves, S.; Kooijman, L.; Chan, E.T.; Kirk, C.J.; Dimayuga Smith, V.; Koot, S.; Mamber, C.; Jansen, A.H.; et al. Reactive glia show increased immunoproteasome activity in Alzheimer’s disease. Brain 2013, 136, 1415–1431. [Google Scholar] [CrossRef]
- van Scheppingen, J.; Broekaart, D.W.; Scholl, T.; Zuidberg, M.R.; Anink, J.J.; Spliet, W.G.; van Rijen, P.C.; Czech, T.; Hainfellner, J.A.; Feucht, M.; et al. Dysregulation of the (immuno)proteasome pathway in malformations of cortical development. J. Neuroinflammation 2016, 13, 202. [Google Scholar] [CrossRef]
- Fonseca, R.; Vabulas, R.M.; Hartl, F.U.; Bonhoeffer, T.; Nägerl, U.V. A balance of protein synthesis and proteasome-dependent degradation determines the maintenance of LTP. Neuron 2006, 52, 239–245. [Google Scholar] [CrossRef]
- Dong, C.; Upadhya, S.C.; Ding, L.; Smith, T.K.; Hegde, A.N. Proteasome inhibition enhances the induction and impairs the maintenance of late-phase long-term potentiation. Learn. Mem. 2008, 15, 335–347. [Google Scholar] [CrossRef]
- Türker, F.; Cook, E.K.; Margolis, S.S. The proteasome and its role in the nervous system. Cell Chem. Biol. 2021, 28, 903–917. [Google Scholar] [CrossRef]
- Maltsev, A.; Funikov, S.; Burov, A.; Spasskaya, D.; Ignatyuk, V.; Astakhova, T.; Lyupina, Y.; Deikin, A.; Tutyaeva, V.; Bal, N.; et al. Immunoproteasome Inhibitor ONX-0914 Affects Long-Term Potentiation in Murine Hippocampus. J. Neuroimmune Pharmacol. 2021, 16, 7–11. [Google Scholar] [CrossRef]
- Bashir, Z.I.; Bortolotto, Z.A.; Davies, C.H.; Berretta, N.; Irving, A.J.; Seal, A.J.; Henley, J.M.; Jane, D.E.; Watkins, J.C.; Collingridge, G.L. Induction of LTP in the hippocampus needs synaptic activation of glutamate metabotropic receptors. Nature 1993, 363, 347–350. [Google Scholar] [CrossRef] [PubMed]
- Malleret, G.; Alarcon, J.M.; Martel, G.; Takizawa, S.; Vronskaya, S.; Yin, D.; Chen, I.Z.; Kandel, E.R.; Shumyatsky, G.P. Bidirectional regulation of hippocampal long-term synaptic plasticity and its influence on opposing forms of memory. J. Neurosci. 2010, 30, 3813–3825. [Google Scholar] [CrossRef] [PubMed]
- Maltsev, A.; Roshchin, M.; Bezprozvanny, I.; Smirnov, I.; Vlasova, O.; Balaban, P.; Borodinova, A. Bidirectional regulation by “star forces”: Ionotropic astrocyte’s optical stimulation suppresses synaptic plasticity, metabotropic one strikes back. Hippocampus 2023, 33, 18–36. [Google Scholar] [CrossRef]
- Basler, M.; Mundt, S.; Muchamuel, T.; Moll, C.; Jiang, J.; Groettrup, M.; Kirk, C.J. Inhibition of the immunoproteasome ameliorates experimental autoimmune encephalomyelitis. EMBO Mol. Med. 2014, 6, 226–238. [Google Scholar] [CrossRef] [PubMed]
- Phillips, K.G.; Hardingham, N.R.; Fox, K. Postsynaptic action potentials are required for nitric-oxide-dependent long-term potentiation in CA1 neurons of adult GluR1 knock-out and wild-type mice. J. Neurosci. 2008, 28, 14031–14041. [Google Scholar] [CrossRef] [PubMed]
- De Verteuil, D.A.; Rouette, A.; Hardy, M.-P.; Lavallée, S.; Trofimov, A.; Gaucher, E.; Perreault, C. Immunoproteasomes Shape the Transcriptome and Regulate the Function of Dendritic Cells. J. Immunol. 2014, 193, 1121–1132. [Google Scholar] [CrossRef]
- Wagner, L.K.; Gilling, K.E.; Schormann, E.; Kloetzel, P.M.; Heppner, F.L.; Krüger, E.; Prokop, S. Immunoproteasome deficiency alters microglial cytokine response and improves cognitive deficits in Alzheimer’s disease-like APPPS1 mice. Acta Neuropathol. Commun. 2017, 5, 52. [Google Scholar] [CrossRef]
- Min, S.S.; Quan, H.Y.; Ma, J.; Han, J.S.; Jeon, B.H.; Seol, G.H. Chronic brain inflammation impairs two forms of long-term potentiation in the rat hippocampal CA1 area. Neurosci. Lett. 2009, 456, 20–24. [Google Scholar] [CrossRef]
- Bliss, T.V.P.; Collingridge, G.L. Expression of NMDA receptor-dependent LTP in the hippocampus: Bridging the divide. Mol. Brain 2013, 6, 5. [Google Scholar] [CrossRef]
- Parmar, S.; Tadavarty, R.; Sastry, B.R. G-protein coupled receptors and synaptic plasticity in sleep deprivation. World J. Psychiatry 2021, 11, 954–980. [Google Scholar] [CrossRef]
- Kelleher, R.J., 3rd; Govindarajan, A.; Jung, H.Y.; Kang, H.; Tonegawa, S. Translational control by MAPK signaling in long-term synaptic plasticity and memory. Cell 2004, 116, 467–479. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, K.V.; Margolis, S.S. A mammalian nervous-system-specific plasma membrane proteasome complex that modulates neuronal function. Nat. Struct. Mol. Biol. 2017, 24, 419–430. [Google Scholar] [CrossRef] [PubMed]
- He, H.Y.; Ahsan, A.; Bera, R.; McLain, N.; Faulkner, R.; Ramachandran, K.V.; Margolis, S.S.; Cline, H.T. Neuronal membrane proteasomes regulate neuronal circuit activity in vivo and are required for learning-induced behavioral plasticity. Proc. Natl. Acad. Sci. USA 2023, 120, e2216537120. [Google Scholar] [CrossRef]
- Muchamuel, T.; Basler, M.; Aujay, M.A.; Suzuki, E.; Kalim, K.W.; Lauer, C.; Sylvain, C.; Ring, E.R.; Shields, J.; Jiang, J.; et al. A selective inhibitor of the immunoproteasome subunit LMP7 blocks cytokine production and attenuates progression of experimental arthritis. Nat. Med. 2009, 15, 781–787. [Google Scholar] [CrossRef] [PubMed]
- Funikov, S.Y.; Rezvykh, A.P.; Mazin, P.V.; Morozov, A.V.; Maltsev, A.V.; Chicheva, M.M.; Vikhareva, E.A.; Evgen’ev, M.B.; Ustyugov, A.A. FUS(1-359) transgenic mice as a model of ALS: Pathophysiological and molecular aspects of the proteinopathy. Neurogenetics 2018, 19, 189–204. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.D.; McCarthy, D.J.; Smyth, G.K. edgeR: A Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 2010, 26, 139–140. [Google Scholar] [CrossRef]
- Ge, S.X.; Jung, D.; Yao, R. ShinyGO: A graphical gene-set enrichment tool for animals and plants. Bioinformatics 2020, 36, 2628–2629. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maltsev, A.; Funikov, S.; Rezvykh, A.; Teterina, E.; Nebogatikov, V.; Burov, A.; Bal, N.; Ustyugov, A.; Karpov, V.; Morozov, A. Chronic Administration of Non-Constitutive Proteasome Inhibitor Modulates Long-Term Potentiation and Glutamate Signaling-Related Gene Expression in Murine Hippocampus. Int. J. Mol. Sci. 2023, 24, 8172. https://doi.org/10.3390/ijms24098172
Maltsev A, Funikov S, Rezvykh A, Teterina E, Nebogatikov V, Burov A, Bal N, Ustyugov A, Karpov V, Morozov A. Chronic Administration of Non-Constitutive Proteasome Inhibitor Modulates Long-Term Potentiation and Glutamate Signaling-Related Gene Expression in Murine Hippocampus. International Journal of Molecular Sciences. 2023; 24(9):8172. https://doi.org/10.3390/ijms24098172
Chicago/Turabian StyleMaltsev, Alexander, Sergei Funikov, Alexander Rezvykh, Ekaterina Teterina, Vladimir Nebogatikov, Alexander Burov, Natalia Bal, Aleksey Ustyugov, Vadim Karpov, and Alexey Morozov. 2023. "Chronic Administration of Non-Constitutive Proteasome Inhibitor Modulates Long-Term Potentiation and Glutamate Signaling-Related Gene Expression in Murine Hippocampus" International Journal of Molecular Sciences 24, no. 9: 8172. https://doi.org/10.3390/ijms24098172





