MicroRNAs in Hepatocellular Carcinoma Pathogenesis: Insights into Mechanisms and Therapeutic Opportunities
Abstract
:1. Introduction
1.1. Hepatocellular Carcinoma
1.2. microRNAs
2. Role of miRNAs in Hepatocellular Carcinoma Cell Survival
2.1. miRNAs in Regulating Hepatocellular Carcinoma Cell Cycle and Proliferation
2.2. Regulatory Role of miRNAs in Hepatocellular Carcinoma Cell Death Pathways
2.2.1. Apoptosis Related miRNAs
2.2.1.1. Pro-Apoptotic miRNAs in Hepatocellular Carcinoma
2.2.1.2. Anti-Apoptotic miRNAs in Hepatocellular Carcinoma
2.2.2. Necroptosis Related miRNAs
2.2.3. Autophagy Related miRNAs
2.2.3.1. Pro-Autophagic miRNAs in Hepatocellular Carcinoma
2.2.3.2. Anti-Autophagic miRNAs in Hepatocellular Carcinoma
3. miRNAs Regulatory Role in Tumor Cell Stemness
3.1. miRNAs Inhibiting Hepatocellular Carcinoma Stemness
3.2. miRNAs Promoting Hepatocellular Carcinoma Stemness
4. miRNAs Regulatory Role in Hepatocellular Carcinoma Metastasis
4.1. Anti-Metastatic miRNAs in Hepatocellular Carcinoma
4.2. Pro-Metastatic miRNAs (metastamiRs) in Hepatocellular Carcinoma
5. Exosomal miRNAs in Hepatocellular Carcinoma Progression
5.1. EMT Related Exosomal miRNAs in Hepatocellular Carcinoma
5.2. Exosomal miRNAs Involved in Non-EMT Related Hepatocellular Carcinoma Progression and Metastsis
6. miRNAs in Tumor Microenvironment Remodelling
6.1. miRNAs Regulating Cancer-Associated Fibroblasts
6.2. miRNAs in Regulating Tumor-Associated Macrophages
6.3. miRNAs in Regulating Natural Killer Cells, T Cells, and Dendritic Cells
6.4. miRNAs Involved in Tumor Angiogenesis Regulation
6.4.1. Anti-Angiogenic miRNAs in Hepatocellular Carcinoma
6.4.2. Pro-Angiogenic miRNAs in Hepatocellular Carcinoma
7. miRNAs in Hepatocellular Carcinoma Drug Resistance
7.1. miRNAs in Chemotherapy Response
7.1.1. miRNAs Promoting Hepatocellular Carcinoma Cells Sensitivity to Chemotherapy
7.1.2. miRNAs Promoting Hepatocellular Carcinoma Cells Resistance to Chemotherapy
7.2. miRNAs in Targeted Therapy Response
7.2.1. miRNAs Improving Sorafenib Response
7.2.2. miRNAs Inducing Sorafenib Resistance
7.2.3. miRNAs Improving Lenvatinib Response
7.2.4. miRNAs Inducing Lenvatinib Resistance
8. miRNA-Based Therapeutics for Hepatocellular Carcinoma Therapy
8.1. Restoring Tumor Suppressive miRNA Function
8.2. Inhibition of Oncogenic miRNA Function
9. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.K.; Tsui, Y.M.; Ho, D.W.; Ng, I.O. Cellular heterogeneity and plasticity in liver cancer. Semin. Cancer Biol. 2021, 82, 134–149. [Google Scholar] [CrossRef] [PubMed]
- Australian Institute of Health and Welfare. Cancer in Australia 2019 (Cancer Series No. 119; Cat. No. CAN 123); AIHW: Canberra, Australia, 2019. [Google Scholar]
- Chen, E.-B.; Zhou, Z.-J.; Xiao, K.; Zhu, G.-Q.; Yang, Y.; Wang, B.; Zhou, S.-L.; Chen, Q.; Yin, D.; Wang, Z. The miR-561-5p/CX3CL1 signaling axis regulates pulmonary metastasis in hepatocellular carcinoma involving CX3CR1+ natural killer cells infiltration. Theranostics 2019, 9, 4779. [Google Scholar] [CrossRef]
- Baecker, A.; Liu, X.; La Vecchia, C.; Zhang, Z.-F. Worldwide incident hepatocellular carcinoma cases attributable to major risk factors. Eur. J. Cancer Prev. Off. J. Eur. Cancer Prev. Organ. ECP 2018, 27, 205. [Google Scholar] [CrossRef]
- Kanwal, F.; Kramer, J.; Asch, S.M.; Chayanupatkul, M.; Cao, Y.; El-Serag, H.B. Risk of hepatocellular cancer in HCV patients treated with direct-acting antiviral agents. Gastroenterology 2017, 153, 996–1005.e1001. [Google Scholar] [CrossRef]
- Tian, Z.; Xu, C.; Yang, P.; Lin, Z.; Wu, W.; Zhang, W.; Ding, J.; Ding, R.; Zhang, X.; Dou, K. Molecular pathogenesis: Connections between viral hepatitis-induced and non-alcoholic steatohepatitis-induced hepatocellular carcinoma. Front. Immunol. 2022, 13, 984728. [Google Scholar] [CrossRef] [PubMed]
- Hoshida, Y.; Nijman, S.M.B.; Kobayashi, M.; Chan, J.A.; Brunet, J.-P.; Chiang, D.Y.; Villanueva, A.; Newell, P.; Ikeda, K.; Hashimoto, M. Integrative transcriptome analysis reveals common molecular subclasses of human hepatocellular carcinoma. Cancer Res. 2009, 69, 7385–7392. [Google Scholar] [CrossRef] [PubMed]
- Dhanasekaran, R.; Bandoh, S.; Roberts, L.R. Molecular pathogenesis of hepatocellular carcinoma and impact of therapeutic advances. F1000Research 2016, 5, 879. [Google Scholar] [CrossRef]
- Dariya, B.; Peela, S.; Nagaraju, G.P.; Nagaraju, G.P.; Peela, S. Chapter 1—Cell origin, biology, and pathophysiology of hepatocellular carcinoma. In Theranostics and Precision Medicine for the Management of Hepatocellular Carcinoma; Academic Press: Cambridge, MA, USA, 2022; pp. 1–9. [Google Scholar] [CrossRef]
- Statello, L.; Guo, C.-J.; Chen, L.-L.; Huarte, M. Gene regulation by long non-coding RNAs and its biological functions. Nat. Rev. Mol. Cell Biol. 2021, 22, 96–118. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Borchert, G.M.; Lanier, W.; Davidson, B.L. RNA polymerase III transcribes human microRNAs. Nat. Struct. Mol. Biol. 2006, 13, 1097–1101. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Kim, M.; Han, J.; Yeom, K.H.; Lee, S.; Baek, S.H.; Kim, V.N. MicroRNA genes are transcribed by RNA polymerase II. EMBO J. 2004, 23, 4051–4060. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, L.A.; Murphy, P.R. MicroRNA: Biogenesis, Function and Role in Cancer. Curr. Genom. 2010, 11, 537–561. [Google Scholar] [CrossRef] [PubMed]
- Lund, E.; Dahlberg, J.E. Substrate selectivity of exportin 5 and Dicer in the biogenesis of microRNAs. Cold Spring Harb. Symp. Quant. Biol. 2006, 71, 59–66. [Google Scholar] [CrossRef]
- Pistritto, G.; Trisciuoglio, D.; Ceci, C.; Garufi, A.; D’Orazi, G. Apoptosis as anticancer mechanism: Function and dysfunction of its modulators and targeted therapeutic strategies. Aging 2016, 8, 603. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar] [CrossRef]
- Guo, H.; Ingolia, N.T.; Weissman, J.S.; Bartel, D.P. Mammalian microRNAs predominantly act to decrease target mRNA levels. Nature 2010, 466, 835–840. [Google Scholar] [CrossRef] [PubMed]
- Di Leva, G.; Garofalo, M.; Croce, C.M. MicroRNAs in cancer. Annu. Rev. Pathol. Mech. Dis. 2014, 9, 287–314. [Google Scholar] [CrossRef]
- Peng, Y.; Croce, C.M. The role of MicroRNAs in human cancer. Signal Transduct. Target. Ther. 2016, 1, 15004. [Google Scholar] [CrossRef]
- Siddhartha, R.; Garg, M. MicroRNAs in Cancer: Diagnostics and Therapeutics. In Handbook of Oncobiology: From Basic to Clinical Sciences; Springer Nature: Berlin/Heidelberg, Germany, 2024; p. 819. [Google Scholar]
- Mizuguchi, Y.; Takizawa, T.; Yoshida, H.; Uchida, E. Dysregulated miRNA in progression of hepatocellular carcinoma: A systematic review. Hepatol. Res. 2016, 46, 391–406. [Google Scholar] [CrossRef]
- Wei, L.; Wang, X.; Lv, L.; Liu, J.; Xing, H.; Song, Y.; Xie, M.; Lei, T.; Zhang, N.; Yang, M. The emerging role of microRNAs and long noncoding RNAs in drug resistance of hepatocellular carcinoma. Mol. Cancer 2019, 18, 147. [Google Scholar] [CrossRef]
- Ramakrishnan, K.; Vishwakarma, R.; Dev, R.R.; Raju, R.; Rehman, N. Etiologically Significant microRNAs in Hepatitis B Virus-Induced Hepatocellular Carcinoma. OMICS J. Integr. Biol. 2024, 28, 280–290. [Google Scholar] [CrossRef] [PubMed]
- Tavakoli Pirzaman, A.; Alishah, A.; Babajani, B.; Ebrahimi, P.; Sheikhi, S.A.; Moosaei, F.; Salarfar, A.; Doostmohamadian, S.; Kazemi, S. The role of microRNAs in hepatocellular cancer: A narrative review focused on tumor microenvironment and drug resistance. Technol. Cancer Res. Treat. 2024, 23, 15330338241239188. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Sinha, S.; Farfel, A.; Luker, K.E.; Parker, B.A.; Yeung, K.T.; Luker, G.D.; Ghosh, P. Growth signaling autonomy in circulating tumor cells aids metastatic seeding. Proc. Natl. Acad. Sci. USA Nexus 2024, 3, pgae014. [Google Scholar] [CrossRef] [PubMed]
- Hwang, H.W.; Mendell, J.T. MicroRNAs in cell proliferation, cell death, and tumorigenesis. Br. J. Cancer 2006, 94, 776–780. [Google Scholar] [CrossRef] [PubMed]
- Duronio, R.J.; Xiong, Y. Signaling pathways that control cell proliferation. Cold Spring Harb. Perspect. Biol. 2013, 5, a008904. [Google Scholar] [CrossRef]
- Bueno, M.J.; Malumbres, M. MicroRNAs and the cell cycle. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2011, 1812, 592–601. [Google Scholar] [CrossRef]
- Xu, X.; Zou, H.; Luo, L.; Wang, X.; Wang, G. MicroRNA-9 exerts antitumor effects on hepatocellular carcinoma progression by targeting HMGA2. FEBS Open Bio 2019, 9, 1784–1797. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhu, C.; Chang, Q.; Peng, P.; Yang, J.; Liu, C.; Liu, Y.; Chen, X.; Liu, Y.; Cheng, R. MiR-424-5p regulates cell cycle and inhibits proliferation of hepatocellular carcinoma cells by targeting E2F7. PLoS ONE 2020, 15, e0242179. [Google Scholar] [CrossRef]
- Zhang, Y.; You, W.; Zhou, H.; Chen, Z.; Han, G.; Zuo, X.; Zhang, L.; Wu, J.; Wang, X. Downregulated miR-621 promotes cell proliferation via targeting CAPRIN1 in hepatocellular carcinoma. Am. J. Cancer Res. 2018, 8, 2116. [Google Scholar]
- Hu, B.; Yang, X.B.; Yang, X.; Sang, X.T. LncRNA CYTOR affects the proliferation, cell cycle and apoptosis of hepatocellular carcinoma cells by regulating the miR-125b-5p/KIAA1522 axis. Aging 2020, 13, 2626–2639. [Google Scholar] [CrossRef]
- Hua, S.; Quan, Y.; Zhan, M.; Liao, H.; Li, Y.; Lu, L. miR-125b-5p inhibits cell proliferation, migration, and invasion in hepatocellular carcinoma via targeting TXNRD1. Cancer Cell Int. 2019, 19, 203. [Google Scholar] [CrossRef]
- Zhang, N.-S.; Dai, G.-L.; Liu, S.-J. MicroRNA-29 family functions as a tumor suppressor by targeting RPS15A and regulating cell cycle in hepatocellular carcinoma. Int. J. Clin. Exp. Pathol. 2017, 10, 8031. [Google Scholar]
- Chen, Z.; Zhuang, W.; Wang, Z.; Xiao, W.; Don, W.; Li, X.; Chen, X. MicroRNA-450b-3p inhibits cell growth by targeting phosphoglycerate kinase 1 in hepatocellular carcinoma. J. Cell. Biochem. 2019, 120, 18805–18815. [Google Scholar] [CrossRef] [PubMed]
- Ribbeck, K.; Raemaekers, T.; Carmeliet, G.; Mattaj, I.W. A role for NuSAP in linking microtubules to mitotic chromosomes. Curr. Biol. 2007, 17, 230–236. [Google Scholar] [CrossRef]
- Hou, S.; Hua, L.; Wang, W.; Li, M.; Xu, L. Nucleolar spindle associated protein 1 (NUSAP1) facilitates proliferation of hepatocellular carcinoma cells. Transl. Cancer Res. 2019, 8, 2113–2120. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ju, L.; Xiao, F.; Liu, H.; Luo, X.; Chen, L.; Lu, Z.; Bian, Z. Downregulation of nucleolar and spindle-associated protein 1 expression suppresses liver cancer cell function. Exp. Ther. Med. 2019, 17, 2969–2978. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Hooiveld, G.J.; Seehawer, M.; Caruso, S.; Heinzmann, F.; Schneider, A.T.; Frank, A.K.; Cardenas, D.V.; Sonntag, R.; Luedde, M.; et al. microRNA 193a-5p Regulates Levels of Nucleolar- and Spindle-Associated Protein 1 to Suppress Hepatocarcinogenesis. Gastroenterology 2018, 155, 1951–1966.e1926. [Google Scholar] [CrossRef]
- Yang, Y.; Zhao, Z.; Hou, N.; Li, Y.; Wang, X.; Wu, F.; Sun, R.; Han, J.; Sun, H.; Song, T. MicroRNA-214 targets Wnt3a to suppress liver cancer cell proliferation. Mol. Med. Rep. 2017, 16, 6920–6927. [Google Scholar] [CrossRef]
- Li, Y.; Li, Y.; Chen, Y.; Xie, Q.; Dong, N.; Gao, Y.; Deng, H.; Lu, C.; Wang, S. MicroRNA-214-3p inhibits proliferation and cell cycle progression by targeting MELK in hepatocellular carcinoma and correlates cancer prognosis. Cancer Cell Int. 2017, 17, 102. [Google Scholar] [CrossRef]
- Dai, X.; Huang, R.; Hu, S.; Zhou, Y.; Sun, X.; Gui, P.; Yu, Z.; Zhou, P. A novel miR-0308-3p revealed by miRNA-seq of HBV-positive hepatocellular carcinoma suppresses cell proliferation and promotes G1/S arrest by targeting double CDK6/Cyclin D1 genes. Cell Biosci. 2020, 10, 24. [Google Scholar] [CrossRef]
- Dai, X.; Chen, C.; Yang, Q.; Xue, J.; Chen, X.; Sun, B.; Luo, F.; Liu, X.; Xiao, T.; Xu, H. Exosomal circRNA_100284 from arsenite-transformed cells, via microRNA-217 regulation of EZH2, is involved in the malignant transformation of human hepatic cells by accelerating the cell cycle and promoting cell proliferation. Cell Death Dis. 2018, 9, 454. [Google Scholar] [CrossRef]
- Zhang, M.; Li, M.; Li, N.; Zhang, Z.; Liu, N.; Han, X.; Liu, Q.; Liao, C. miR-217 suppresses proliferation, migration, and invasion promoting apoptosis via targeting MTDH in hepatocellular carcinoma. Oncol. Rep. 2017, 37, 1772–1778. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Lu, Y.X.; Wang, F.; Sun, J.; Bian, J.X.; Wu, H.Y. miRNA-217 inhibits proliferation of hepatocellular carcinoma cells by regulating KLF5. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 7874–7883. [Google Scholar]
- Li, S.; Peng, F.; Ning, Y.; Jiang, P.; Peng, J.; Ding, X.; Zhang, J.; Jiang, T.; Xiang, S. SNHG16 as the miRNA let-7b-5p sponge facilitates the G2/M and epithelial-mesenchymal transition by regulating CDC25B and HMGA2 expression in hepatocellular carcinoma. J. Cell. Biochem. 2020, 121, 2543–2558. [Google Scholar] [CrossRef]
- Zhao, G.; Han, C.; Zhang, Z.; Wang, L.; Xu, J. Increased expression of microRNA-31-5p inhibits cell proliferation, migration, and invasion via regulating Sp1 transcription factor in HepG2 hepatocellular carcinoma cell line. Biochem. Biophys. Res. Commun. 2017, 490, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Liu, E.; Kang, J.; Yang, X.; Liu, H. MiR-3613-3p affects cell proliferation and cell cycle in hepatocellular carcinoma. Oncotarget 2017, 8, 93014. [Google Scholar] [CrossRef]
- Pollutri, D.; Patrizi, C.; Marinelli, S.; Giovannini, C.; Trombetta, E.; Giannone, F.A.; Baldassarre, M.; Quarta, S.; Vandewynckel, Y.-P.; Vandierendonck, A. The epigenetically regulated miR-494 associates with stem-cell phenotype and induces sorafenib resistance in hepatocellular carcinoma. Cell Death Dis. 2018, 9, 4. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Yu, C.; Wu, M.; Wu, X.; Wan, L.; Zhu, X. MicroRNA-191 promotes hepatocellular carcinoma cell proliferation by has_circ_0000204/miR-191/KLF6 axis. Cell Prolif. 2019, 52, e12635. [Google Scholar] [CrossRef]
- Yao, B.; Niu, Y.; Li, Y.; Chen, T.; Wei, X.; Liu, Q. High-matrix-stiffness induces promotion of hepatocellular carcinoma proliferation and suppression of apoptosis via miR-3682-3p-PHLDA1-FAS pathway. J. Cancer 2020, 11, 6188–6203. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Gong, L.; Wang, J.; Tu, Q.; Yao, L.; Zhang, J.-R.; Han, X.-J.; Zhu, S.-J.; Wang, S.-M.; Li, Y.-H.; et al. miR-10b exerts oncogenic activity in human hepatocellular carcinoma cells by targeting expression of CUB and sushi multiple domains 1 (CSMD1). BMC Cancer 2016, 16, 806. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Xiang, B.; Qi, L.; Zhu, S.; Li, L. miR-221-3p promotes hepatocellular carcinogenesis by downregulating O6-methylguanine-DNA methyltransferase. Cancer Biol. Ther. 2020, 21, 915–926. [Google Scholar] [CrossRef] [PubMed]
- Yan, G.; Elbadawi, M.; Efferth, T. Multiple cell death modalities and their key features. World Acad. Sci. J. 2020, 2, 39–48. [Google Scholar] [CrossRef]
- Walczak, H. Death receptor-ligand systems in cancer, cell death, and inflammation. Cold Spring Harb. Perspect. Biol. 2013, 5, a008698. [Google Scholar] [CrossRef]
- Wang, J.; Li, Y.; Ma, Q.; Huang, J. miR-378 in combination with ultrasonic irradiation and SonoVue microbubbles transfection inhibits hepatoma cell growth. Mol. Med. Rep. 2020, 21, 2493–2501. [Google Scholar] [CrossRef]
- Dong, X.; Wang, F.; Xue, Y.; Lin, Z.; Song, W.; Yang, N.; Li, Q. MicroRNA-9-5p downregulates Klf4 and influences the progression of hepatocellular carcinoma via the AKT signaling pathway. Int. J. Mol. Med. 2019, 43, 1417–1429. [Google Scholar] [CrossRef]
- Liao, Z.-B.; Tan, X.-L.; Dong, K.-S.; Zhang, H.-W.; Chen, X.-P.; Chu, L.; Zhang, B.-X. miRNA-448 inhibits cell growth by targeting BCL-2 in hepatocellular carcinoma. Dig. Liver Dis. 2019, 51, 703–711. [Google Scholar] [CrossRef]
- Wang, X.; Zeng, J.; Wang, L.; Zhang, X.; Liu, Z.; Zhang, H.; Dong, J. Overexpression of microRNA-133b is associated with the increased survival of patients with hepatocellular carcinoma after curative hepatectomy: Involvement of the EGFR/PI3K/Akt/mTOR signaling pathway. Oncol. Rep. 2017, 38, 141–150. [Google Scholar] [CrossRef]
- Li, C.; Hashimi, S.M.; Good, D.A.; Cao, S.; Duan, W.; Plummer, P.N.; Mellick, A.S.; Wei, M.Q. Apoptosis and micro RNA aberrations in cancer. Clin. Exp. Pharmacol. Physiol. 2012, 39, 739–746. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, X.; Zhou, Y.; Cheng, L.; Zhang, Y.; Zhang, Y. Long Noncoding RNA NEAT1 Promotes Cell Proliferation and Invasion And Suppresses Apoptosis In Hepatocellular Carcinoma By Regulating miRNA-22-3p/akt2 In Vitro And In Vivo. OncoTargets Ther. 2019, 12, 8991–9004. [Google Scholar] [CrossRef]
- Wang, Y.; Tai, Q.; Zhang, J.; Kang, J.; Gao, F.; Zhong, F.; Cai, L.; Fang, F.; Gao, Y. MiRNA-206 inhibits hepatocellular carcinoma cell proliferation and migration but promotes apoptosis by modulating cMET expression. Acta Biochim. Biophys. Sin. 2019, 51, 243–253. [Google Scholar] [CrossRef]
- Lee, H.; Jeong, A.J.; Ye, S.-K. Highlighted STAT3 as a potential drug target for cancer therapy. BMB Rep. 2019, 52, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Xue, H.; Sun, Y.; Zhang, L.; Xue, F.; Ge, R. CircularRNA-9119 protects hepatocellular carcinoma cells from apoptosis by intercepting miR-26a/JAK1/STAT3 signaling. Cell Death Dis. 2020, 11, 605. [Google Scholar] [CrossRef] [PubMed]
- Uhlik, M.T.; Abell, A.N.; Cuevas, B.D.; Nakamura, K.; Johnson, G.L. Wiring diagrams of MAPK regulation by MEKK1, 2, and 3. Biochem. Cell Biol. 2004, 82, 658–663. [Google Scholar] [CrossRef]
- Cuevas, B.D.; Abell, A.N.; Johnson, G.L. Role of mitogen-activated protein kinase kinase kinases in signal integration. Oncogene 2007, 26, 3159–3171. [Google Scholar] [CrossRef] [PubMed]
- Su, B.; Cheng, J.; Yang, J.; Guo, Z. MEKK2 is required for T-cell receptor signals in JNK activation and interleukin-2 gene expression. J. Biol. Chem. 2001, 276, 14784–14790. [Google Scholar] [CrossRef]
- Mirza, A.A.; Kahle, M.P.; Ameka, M.; Campbell, E.M.; Cuevas, B.D. MEKK2 regulates focal adhesion stability and motility in invasive breast cancer cells. Biochim. Biophys. Acta BBA Mol. Cell Res. 2014, 1843, 945–954. [Google Scholar] [CrossRef]
- Mazur, P.K.; Reynoird, N.; Khatri, P.; Jansen, P.W.T.C.; Wilkinson, A.W.; Liu, S.; Barbash, O.; Van Aller, G.S.; Huddleston, M.; Dhanak, D. SMYD3 links lysine methylation of MAP3K2 to Ras-driven cancer. Nature 2014, 510, 283–287. [Google Scholar] [CrossRef]
- Li, X.; Azhati, B.; Wang, W.; Rexiati, M.; Xing, C.; Wang, Y. Circular RNA UBAP2 promotes the proliferation of prostate cancer cells via the miR-1244/MAP3K2 axis. Oncol. Lett. 2021, 21, 486. [Google Scholar] [CrossRef]
- Wang, M.; Lv, G.; Jiang, C.; Xie, S.; Wang, G. miR-302a inhibits human HepG2 and SMMC-7721 cells proliferation and promotes apoptosis by targeting MAP3K2 and PBX3. Sci. Rep. 2019, 9, 2032. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.; Zhang, L.; Xian, Y.; Yu, Z. MicroRNA-33a promotes cell proliferation and inhibits apoptosis by targeting PPARα in human hepatocellular carcinoma. Exp. Ther. Med. 2017, 13, 2507–2514. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Liu, Q.; Xu, Y.; Gong, X.; Zhang, R.; Zhou, C.; Su, Z.; Jin, J.; Shi, H.; Shi, J.; et al. PPARα induces cell apoptosis by destructing Bcl2. Oncotarget 2015, 6, 44635–44642. [Google Scholar] [CrossRef] [PubMed]
- Maggiora, M.; Oraldi, M.; Muzio, G.; Canuto, R.A. Involvement of PPARα and PPARγ in apoptosis and proliferation of human hepatocarcinoma HepG2 cells. Cell Biochem. Funct. 2010, 28, 571–577. [Google Scholar] [CrossRef]
- Zheng, X.; Li, S.; Yang, H. Roles of Toll-Like Receptor 3 in Human Tumors. Front. Immunol. 2021, 12, 667454. [Google Scholar] [CrossRef]
- Khvalevsky, E.; Rivkin, L.; Rachmilewitz, J.; Galun, E.; Giladi, H. TLR3 signaling in a hepatoma cell line is skewed towards apoptosis. J. Cell. Biochem. 2007, 100, 1301–1312. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Cai, W.; Liang, Y.; Yao, J.; Wang, X.; Shen, J. In situ self-assembly of Au-antimiR-155 nanocomplexes mediates TLR3-dependent apoptosis in hepatocellular carcinoma cells. Aging 2020, 13, 241–261. [Google Scholar] [CrossRef]
- Xu, C.; Shi, L.; Chen, W.; Fang, P.; Li, J.; Jin, L.; Pan, Z.; Pan, C. MiR-106b inhibitors sensitize TRAIL-induced apoptosis in hepatocellular carcinoma through increase of death receptor 4. Oncotarget 2017, 8, 41921–41931. [Google Scholar] [CrossRef]
- Christofferson, D.E.; Yuan, J. Necroptosis as an alternative form of programmed cell death. Curr. Opin. Cell Biol. 2010, 22, 263–268. [Google Scholar] [CrossRef]
- Belizário, J.; Vieira-Cordeiro, L.; Enns, S. Necroptotic Cell Death Signaling and Execution Pathway: Lessons from Knockout Mice. Mediat. Inflamm. 2015, 2015, 128076. [Google Scholar] [CrossRef]
- Gong, Y.; Fan, Z.; Luo, G.; Yang, C.; Huang, Q.; Fan, K.; Cheng, H.; Jin, K.; Ni, Q.; Yu, X.; et al. The role of necroptosis in cancer biology and therapy. Mol. Cancer 2019, 18, 100. [Google Scholar] [CrossRef] [PubMed]
- Visalli, M.; Bartolotta, M.; Polito, F.; Oteri, R.; Barbera, A.; Arrigo, R.; Di Giorgio, R.M.; Navarra, G.; Aguennouz, M.H. miRNA expression profiling regulates necroptotic cell death in hepatocellular carcinoma. Int. J. Oncol. 2018, 53, 771–780. [Google Scholar] [CrossRef]
- van Zandwijk, N.; Pavlakis, N.; Kao, S.C.; Linton, A.; Boyer, M.J.; Clarke, S.; Huynh, Y.; Chrzanowska, A.; Fulham, M.J.; Bailey, D.L.; et al. Safety and activity of microRNA-loaded minicells in patients with recurrent malignant pleural mesothelioma: A first-in-man, phase 1, open-label, dose-escalation study. Lancet Oncol. 2017, 18, 1386–1396. [Google Scholar] [CrossRef]
- Morselli, E.; Galluzzi, L.; Kepp, O.; Mariño, G.; Michaud, M.; Vitale, I.; Maiuri, M.C.; Kroemer, G. Oncosuppressive functions of autophagy. Antioxid. Redox Signal. 2011, 14, 2251–2269. [Google Scholar] [CrossRef] [PubMed]
- Kenific, C.M.; Thorburn, A.; Debnath, J. Autophagy and metastasis: Another double-edged sword. Curr. Opin. Cell Biol. 2010, 22, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Fung, C.; Lock, R.; Gao, S.; Salas, E.; Debnath, J. Induction of autophagy during extracellular matrix detachment promotes cell survival. Mol. Biol. Cell 2008, 19, 797–806. [Google Scholar] [CrossRef] [PubMed]
- Frankel, L.B.; Lund, A.H. MicroRNA regulation of autophagy. Carcinogenesis 2012, 33, 2018–2025. [Google Scholar] [CrossRef]
- Cao, W.; Li, J.; Yang, K.; Cao, D. An overview of autophagy: Mechanism, regulation and research progress. Bull. Du Cancer 2021, 108, 304–322. [Google Scholar] [CrossRef]
- Wang, S.; Li, H.; Yuan, M.; Fan, H.; Cai, Z. Role of AMPK in autophagy. Front. Physiol. 2022, 13, 1015500. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Pan, Q.; Yu, Y.; Zhong, X.P. microRNA-519d Induces Autophagy and Apoptosis of Human Hepatocellular Carcinoma Cells Through Activation of the AMPK Signaling Pathway via Rab10. Cancer Manag. Res. 2020, 12, 2589–2602. [Google Scholar] [CrossRef]
- Zhou, L.; Liu, S.; Han, M.; Feng, S.; Liang, J.; Li, Z.; Li, Y.; Lu, H.; Liu, T.; Ma, Y. MicroRNA-185 induces potent autophagy via AKT signaling in hepatocellular carcinoma. Tumor Biol. 2017, 39, 1010428317694313. [Google Scholar] [CrossRef]
- Guo, J.; Ma, Y.; Peng, X.; Jin, H.; Liu, J. LncRNA CCAT1 promotes autophagy via regulating ATG7 by sponging miR-181 in hepatocellular carcinoma. J. Cell. Biochem. 2019, 120, 17975–17983. [Google Scholar] [CrossRef] [PubMed]
- Ying, H.; Zheng, H.; Scott, K.; Wiedemeyer, R.; Yan, H.; Lim, C.; Huang, J.; Dhakal, S.; Ivanova, E.; Xiao, Y. Mig-6 controls EGFR trafficking and suppresses gliomagenesis. Proc. Natl. Acad. Sci. USA 2010, 107, 6912–6917. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.; Tian, Y.; Hong, D.; Wang, F.; Li, Z. Mig-6 Inhibits Autophagy in HCC Cell Lines by Modulating miR-193a-3p. Int. J. Med. Sci. 2022, 19, 338. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Li, Y.; Liao, J.; Liu, M.; Zhu, L.; Liao, K. MicroRNA-7 inhibits hepatocellular carcinoma cell invasion and metastasis by regulating Atg5-mediated autophagy. Transl. Cancer Res. 2020, 9, 3965–3972. [Google Scholar] [CrossRef]
- Fu, X.T.; Shi, Y.H.; Zhou, J.; Peng, Y.F.; Liu, W.R.; Shi, G.M.; Gao, Q.; Wang, X.Y.; Song, K.; Fan, J.; et al. MicroRNA-30a suppresses autophagy-mediated anoikis resistance and metastasis in hepatocellular carcinoma. Cancer Lett. 2018, 412, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Jin, F.; Wang, Y.; Li, M.; Zhu, Y.; Liang, H.; Wang, C.; Wang, F.; Zhang, C.-Y.; Zen, K.; Li, L. MiR-26 enhances chemosensitivity and promotes apoptosis of hepatocellular carcinoma cells through inhibiting autophagy. Cell Death Dis. 2018, 8, e2540. [Google Scholar] [CrossRef]
- Su, Y.; Yao, L.; An, H.; Liu, J.; Ye, F.; Shen, J.; Ni, Z.; Huang, B.; Lin, J. MicroRNA-204-5p Inhibits Hepatocellular Carcinoma by Targeting the Regulator of G Protein Signaling 20. ACS Pharmacol. Transl. Sci. 2023, 6, 1817–1828. [Google Scholar] [CrossRef]
- Ye, R.; Lu, X.; Liu, J.; Duan, Q.; Xiao, J.; Duan, X.; Yue, Z.; Liu, F. CircSOD2 Contributes to Tumor Progression, Immune Evasion and Anti-PD-1 Resistance in Hepatocellular Carcinoma by Targeting miR-497-5p/ANXA11 Axis. Biochem Genet. 2023, 61, 597–614. [Google Scholar] [CrossRef]
- Chen, Z.; Du, J.; Yang, C.; Si, G.; Chen, Y. circ-CFH promotes the development of HCC by regulating cell proliferation, apoptosis, migration, invasion, and glycolysis through the miR-377-3p/RNF38 axis. Open Life Sci. 2022, 17, 248–260. [Google Scholar] [CrossRef]
- Zhang, T.; Zhang, Y.; Liu, J.; Ma, Y.; Ye, Q.; Yan, X.; Ding, L. MicroRNA-377-3p inhibits hepatocellular carcinoma growth and metastasis through negative regulation of CPT1C-mediated fatty acid oxidation. Cancer Metab. 2022, 10, 2. [Google Scholar] [CrossRef]
- Wang, C.; Li, C.; Hao, R. miR-559 Inhibits Proliferation, Autophagy, and Angiogenesis of Hepatocellular Carcinoma Cells by Targeting PARD3. Mediat. Inflamm. 2022, 2022, 3121492. [Google Scholar] [CrossRef]
- Zheng, X.S.; Liu, H.J.; Zhang, L.L.; Li, H.; Wang, C.J.; Xin, Y.J.; Hao, R. MiR-559 targets GP73 to suppress proliferation and invasion of hepatocellular carcinoma in vitro. Kaohsiung J. Med. Sci. 2020, 36, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Liang, H.; Wu, S.; Zhang, Y.; Yu, Z. MicroRNA-638 induces apoptosis and autophagy in human liver cancer cells by targeting enhancer of zeste homolog 2 (EZH2). Environ. Toxicol. Pharmacol. 2021, 82, 103559. [Google Scholar] [CrossRef]
- Guan, J.; Liu, Z.; Xiao, M.; Hao, F.; Wang, C.; Chen, Y.; Lu, Y.; Liang, J. MicroRNA-199a-3p inhibits tumorigenesis of hepatocellular carcinoma cells by targeting ZHX1/PUMA signal. Am. J. Transl. Res. 2017, 9, 2457–2465. [Google Scholar] [PubMed]
- Fornari, F.; Milazzo, M.; Chieco, P.; Negrini, M.; Calin, G.A.; Grazi, G.L.; Pollutri, D.; Croce, C.M.; Bolondi, L.; Gramantieri, L. MiR-199a-3p regulates mTOR and c-Met to influence the doxorubicin sensitivity of human hepatocarcinoma cells. Cancer Res. 2010, 70, 5184–5193. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, Z.; Zhang, R.; Jia, C.; Mao, R.; Mahati, S.; Zhang, Y.; Wu, G.; Sun, Y.n.; Jia, X.y. MiR-27a-3p enhances the cisplatin sensitivity in hepatocellular carcinoma cells through inhibiting PI3K/Akt pathway. Biosci. Rep. 2021, 41, BSR20192007. [Google Scholar] [CrossRef]
- Zhang, R.; Guo, C.; Liu, T.; Li, W.; Chen, X. MicroRNA miR-495 regulates the development of Hepatocellular Carcinoma by targeting C1q/tumor necrosis factor-related protein-3 (CTRP3). Bioengineered 2021, 12, 6902–6912. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yang, S.; Song, J.; Yang, W.; Ji, Y.; Zhang, F.; Rao, J. Dysregulation of miR-23b-5p promotes cell proliferation via targeting FOXM1 in hepatocellular carcinoma. Cell Death Discov. 2021, 7, 47. [Google Scholar] [CrossRef]
- Xu, X.; Jiang, W.; Han, P.; Zhang, J.; Tong, L.; Sun, X. MicroRNA-128-3p Mediates Lenvatinib Resistance of Hepatocellular Carcinoma Cells by Downregulating c-Met. J. Hepatocell. Carcinoma 2022, 9, 113–126. [Google Scholar] [CrossRef]
- Hu, C.; Cui, S.; Zheng, J.; Yin, T.; Lv, J.; Long, J.; Zhang, W.; Wang, X.; Sheng, S.; Zhang, H. MiR-875-5p inhibits hepatocellular carcinoma cell proliferation and migration by repressing astrocyte elevated gene-1 (AEG-1) expression. Transl. Cancer Res. 2018, 7, 158. [Google Scholar] [CrossRef]
- Zhang, Y.; Wei, Y.; Li, X.; Liang, X.; Wang, L.; Song, J.; Zhang, X.; Zhang, C.; Niu, J.; Zhang, P.; et al. microRNA-874 suppresses tumor proliferation and metastasis in hepatocellular carcinoma by targeting the DOR/EGFR/ERK pathway. Cell Death Dis. 2018, 9, 130. [Google Scholar] [CrossRef]
- Qin, X.; Chen, J.; Wu, L.; Liu, Z. MiR-30b-5p acts as a tumor suppressor, repressing cell proliferation and cell cycle in human hepatocellular carcinoma. Biomed. Pharmacother. 2017, 89, 742–750. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Ai, R.; Yuan, X.; Dong, S.; Zhao, D.; Sun, X.; Miao, T.; Guan, W.; Guo, P.; Yu, S.; et al. LINC00886 Facilitates Hepatocellular Carcinoma Tumorigenesis by Sequestering microRNA-409-3p and microRNA-214-5p. J. Hepatocell. Carcinoma 2023, 10, 863–881. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Li, M.; Li, Q.; Guo, Z.; Sun, X.; Zhang, X.; Liu, Y.; Yao, W.; Xiao, P. MicroRNA-98-5p Inhibits Cell Proliferation and Induces Cell Apoptosis in Hepatocellular Carcinoma via Targeting IGF2BP1. Oncol. Res. 2017, 25, 1117–1127. [Google Scholar] [CrossRef]
- Ma, Y.-S.; Lv, Z.-W.; Yu, F.; Chang, Z.-Y.; Cong, X.-L.; Zhong, X.-M.; Lu, G.-X.; Zhu, J.; Fu, D. MicroRNA-302a/d inhibits the self-renewal capability and cell cycle entry of liver cancer stem cells by targeting the E2F7/AKT axis. J. Exp. Clin. Cancer Res. 2018, 37, 252. [Google Scholar] [CrossRef]
- Sun, L.; Wang, L.; Chen, T.; Yao, B.; Wang, Y.; Li, Q.; Yang, W.; Liu, Z. microRNA-1914, which is regulated by lncRNA DUXAP10, inhibits cell proliferation by targeting the GPR39-mediated PI3K/AKT/mTOR pathway in HCC. J. Cell. Mol. Med. 2019, 23, 8292–8304. [Google Scholar] [CrossRef]
- Dong, G.; Zhang, S.; Shen, S.; Sun, L.; Wang, X.; Wang, H.; Wu, J.; Liu, T.; Wang, C.; Wang, H.; et al. SPATS2, negatively regulated by miR-145-5p, promotes hepatocellular carcinoma progression through regulating cell cycle. Cell Death Dis. 2020, 11, 837. [Google Scholar] [CrossRef]
- Simile, M.M.; Peitta, G.; Tomasi, M.L.; Brozzetti, S.; Feo, C.F.; Porcu, A.; Cigliano, A.; Calvisi, D.F.; Feo, F.; Pascale, R.M. MicroRNA-203 impacts on the growth, aggressiveness and prognosis of hepatocellular carcinoma by targeting MAT2A and MAT2B genes. Oncotarget 2019, 10, 2835. [Google Scholar] [CrossRef]
- Hui, L.; Zheng, F.; Bo, Y.; Sen-Lin, M.; Ai-Jun, L.; Wei-Ping, Z.; Yong-Jie, Z.; Lei, Y. MicroRNA let-7b inhibits cell proliferation via upregulation of p21 in hepatocellular carcinoma. Cell Biosci. 2020, 10, 83. [Google Scholar] [CrossRef]
- Feng, Y.; Xia, S.; Hui, J.; Xu, Y. Circular RNA circBNC2 facilitates glycolysis and stemness of hepatocellular carcinoma through the miR-217/high mobility group AT-hook 2 (HMGA2) axis. Heliyon 2023, 9, e17120. [Google Scholar] [CrossRef] [PubMed]
- Wan, L.; Yuan, X.; Liu, M.; Xue, B. miRNA-223-3p regulates NLRP3 to promote apoptosis and inhibit proliferation of hep3B cells. Exp. Ther. Med. 2018, 15, 2429–2435. [Google Scholar] [CrossRef]
- Liu, Z.; Li, W.; Pang, Y.; Zhou, Z.; Liu, S.; Cheng, K.; Qin, Q.; Jia, Y.; Liu, S. SF3B4 is regulated by microRNA-133b and promotes cell proliferation and metastasis in hepatocellular carcinoma. EBioMedicine 2018, 38, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Q.; Song, J. Inhibition of autophagy potentiates the proliferation inhibition activity of microRNA-7 in human hepatocellular carcinoma cells. Oncol. Lett. 2017, 14, 3566–3572. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, J.; Liu, Y.; Zeng, X.; Wei, M.; Wu, S.; Xiong, Q.; Song, F.; Yuan, X.; Xiao, Y. Hepatitis B virus induces autophagy to promote its replication by the axis of miR-192-3p-XIAP through NF kappa B signaling. Hepatology 2019, 69, 974–992. [Google Scholar] [CrossRef]
- Ting, G.; Li, X.; Kwon, H.Y.; Ding, T.; Zhang, Z.; Chen, Z.; Li, C.; Liu, Y.; Yang, Y. microRNA-219-5p targets NEK6 to inhibit hepatocellular carcinoma progression. Am. J. Transl. Res. 2020, 12, 7528–7541. [Google Scholar]
- Li, L.; Jia, L.; Ding, Y. Upregulation of miR-375 inhibits human liver cancer cell growth by modulating cell proliferation and apoptosis via targeting ErbB2. Oncol. Lett. 2018, 16, 3319–3326. [Google Scholar] [CrossRef]
- Wang, D.; Sun, X.; Wei, Y.; Liang, H.; Yuan, M.; Jin, F.; Chen, X.; Liu, Y.; Zhang, C.-Y.; Li, L.; et al. Nuclear miR-122 directly regulates the biogenesis of cell survival oncomiR miR-21 at the posttranscriptional level. Nucleic Acids Res. 2018, 46, 2012–2029. [Google Scholar] [CrossRef]
- Li, Y.; Wang, X.; Li, Z.; Liu, B.; Wu, C. MicroRNA-4651 represses hepatocellular carcinoma cell growth and facilitates apoptosis via targeting FOXP4. Biosci. Rep. 2020, 40, BSR20194011. [Google Scholar] [CrossRef]
- Liu, Q.; Shi, H.; Yang, J.; Jiang, N. Long non-coding RNA NEAT1 promoted hepatocellular carcinoma cell proliferation and reduced apoptosis through the regulation of Let-7b-IGF-1R Axis. OncoTargets Ther. 2019, 12, 10401. [Google Scholar] [CrossRef]
- Komoll, R.M.; Hu, Q.; Olarewaju, O.; von Döhlen, L.; Yuan, Q.; Xie, Y.; Tsay, H.C.; Daon, J.; Qin, R.; Manns, M.P.; et al. MicroRNA-342-3p is a potent tumour suppressor in hepatocellular carcinoma. J. Hepatol. 2021, 74, 122–134. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Liu, Q.; Jiang, Y.; Cai, Z.; Liu, H.; Zuo, H. Engineered small extracellular vesicles loaded with miR-654-5p promote ferroptosis by targeting HSPB1 to alleviate sorafenib resistance in hepatocellular carcinoma. Cell Death Discov. 2023, 9, 362. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, J. Circular RNA 0016142 Knockdown Induces Ferroptosis in Hepatocellular Carcinoma Cells via Modulation of the MicroRNA-188-3p/Glutathione Peroxidase 4 Axis. Biochem. Genet. 2023, 62, 333–351. [Google Scholar] [CrossRef]
- Xing, K.; Bian, X.; Shi, D.; Dong, S.; Zhou, H.; Xiao, S.; Bai, J.; Wu, W. miR-612 Enhances RSL3-Induced Ferroptosis of Hepatocellular Carcinoma Cells via Mevalonate Pathway. J. Hepatocell. Carcinoma 2023, 10, 2173–2185. [Google Scholar] [CrossRef]
- Duan, X.; Li, W.; Hu, P.; Jiang, B.; Yang, J.; Zhou, L.; Mao, X.; Tian, B. MicroRNA-183-5p contributes to malignant progression through targeting PDCD4 in human hepatocellular carcinoma. Biosci. Rep 2020, 40, BSR20201761. [Google Scholar] [CrossRef]
- Hu, Z.; Li, L.; Li, M.; Zhang, X.; Zhang, Y.; Ran, J.; Li, L. miR-21-5p Inhibits Ferroptosis in Hepatocellular Carcinoma Cells by Regulating the AKT/mTOR Signaling Pathway through MELK. J. Immunol. Res. 2023, 2023, 8929525. [Google Scholar] [CrossRef]
- Shen, H.; Li, H.; Zhou, J. Circular RNA hsa_circ_0032683 inhibits the progression of hepatocellular carcinoma by sponging microRNA-338-5p. Bioengineered 2022, 13, 2321–2335. [Google Scholar] [CrossRef] [PubMed]
- Cheng, N.; Wu, J.; Yin, M.; Xu, J.; Wang, Y.; Chen, X.; Nie, Z.; Yin, J. LncRNA CASC11 promotes cancer cell proliferation in hepatocellular carcinoma by inhibiting miRNA-188-5p. Biosci. Rep. 2019, 39, BSR20190251. [Google Scholar] [CrossRef]
- Chen, Y.L.; Xu, Q.P.; Guo, F.; Guan, W.H. MicroRNA-302d downregulates TGFBR2 expression and promotes hepatocellular carcinoma growth and invasion. Exp. Ther. Med. 2017, 13, 681–687. [Google Scholar] [CrossRef]
- Wang, Q.; Yang, X.; Zhou, X.; Wu, B.; Zhu, D.; Jia, W.; Chu, J.; Wang, J.; Wu, J.; Kong, L. MiR-3174 promotes proliferation and inhibits apoptosis by targeting FOXO1 in hepatocellular carcinoma. Biochem. Biophys. Res. Commun. 2020, 526, 889–897. [Google Scholar] [CrossRef]
- Li, Y.; Wu, B.; Sun, R.; Zhao, M.; Li, N. miR-93-5p knockdown repressed hepatocellular carcinoma progression via increasing ERBB4 and TETs-dependent DNA demethylation. Autoimmunity 2021, 54, 547–560. [Google Scholar] [CrossRef] [PubMed]
- Walcher, L.; Kistenmacher, A.-K.; Suo, H.; Kitte, R.; Dluczek, S.; Strauß, A.; Blaudszun, A.-R.; Yevsa, T.; Fricke, S.; Kossatz-Boehlert, U. Cancer Stem Cells—Origins and Biomarkers: Perspectives for Targeted Personalized Therapies. Front. Immunol. 2020, 11, 1280. [Google Scholar] [CrossRef]
- Holczbauer, Á.; Factor, V.M.; Andersen, J.B.; Marquardt, J.U.; Kleiner, D.E.; Raggi, C.; Kitade, M.; Seo, D.; Akita, H.; Durkin, M.E. Modeling pathogenesis of primary liver cancer in lineage-specific mouse cell types. Gastroenterology 2013, 145, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Li-Li, L.; Da, F.; Yu-Shui, M.; Xizhong, S. The power and the promise of liver cancer stem cell markers. Stem Cells Dev. 2011, 20, 2023–2030. [Google Scholar] [CrossRef]
- Min, W.; Juan, X.; Jianxin, J.; Renyi, Q. CD133 and ALDH may be the molecular markers of cholangiocarcinoma stem cells. Int. J. Cancer 2011, 128, 1996–1997. [Google Scholar] [CrossRef]
- Shimaa, E.; Raja, C.; Mohammed, Z. Ultrasensitive Label-free Electrochemical Immunosensors for Multiple Cell Surface Biomarkers on Liver Cancer Stem Cells. Electroanalysis 2017, 29, 1994–2000. [Google Scholar] [CrossRef]
- Li, H. Efficacy analysis of combined detection of 5 Serological Tumor markers including MIF and PIVKA-II for early diagnosis of Primary Hepatic Cancer. Pak. J. Med. Sci. 2021, 37, 1456–1460. [Google Scholar] [CrossRef]
- Msy Rulan, A.; Indriyani; Ahmad Azmi, N.; Isabella Kurnia, L. Specific Markers for Hepatic Progenitor Cells. OnLine J. Biol. Sci. 2017, 17, 187–192. [Google Scholar] [CrossRef]
- Lv, D.; Chen, L.; Du, L.; Zhou, L.; Tang, H. Emerging Regulatory Mechanisms Involved in Liver Cancer Stem Cell Properties in Hepatocellular Carcinoma. Front. Cell Dev. Biol. 2021, 9, 691410. [Google Scholar] [CrossRef]
- Xie, D.; Pei, Q.; Li, J.; Wan, X.; Ye, T. Emerging Role of E2F Family in Cancer Stem Cells. Front. Oncol. 2021, 11, 723137. [Google Scholar] [CrossRef]
- Dou, Z.; Lu, F.; Hu, J.; Wang, H.; Li, B.; Li, X. MicroRNA-6838-5p suppresses the self-renewal and metastasis of human liver cancer stem cells through downregulating CBX4 expression and inactivating ERK signaling. Biol. Chem. 2023, 404, 29–39. [Google Scholar] [CrossRef]
- Zhang, X.; Jiang, P.; Shuai, L.; Chen, K.; Li, Z.; Zhang, Y.; Jiang, Y.; Li, X. miR-589-5p inhibits MAP3K8 and suppresses CD90+ cancer stem cells in hepatocellular carcinoma. J. Exp. Clin. Cancer Res. 2016, 35, 176. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Liu, Y.; Guo, Y.; Liu, B.; Zhao, Y.; Li, P.; Song, F.; Zheng, H.; Yu, J.; Song, T.; et al. Regulatory MiR-148a-ACVR1/BMP circuit defines a cancer stem cell-like aggressive subtype of hepatocellular carcinoma. Hepatology 2015, 61, 574–584. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Xu, Y.; Wei, S.; Gao, W.; Chen, L.; Zhou, T.; Wang, Z.; Ying, M.; Zheng, Q. miRNA-148b suppresses hepatic cancer stem cell by targeting neuropilin-1. Biosci. Rep. 2015, 35, e00229. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.M.; Shi, X.L.; Xing, K.L.; Zhou, H.X.; Lu, L.L.; Wu, W.Z. miR-296-5p suppresses stem cell potency of hepatocellular carcinoma cells via regulating Brg1/Sall4 axis. Cell. Signal. 2020, 72, 109650. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.-M.; Li, L.-X.; Bian, X.-Y.; Shi, X.-J.; Lu, L.-L.; Zhou, H.-X.; Pan, T.-J.; Zhou, J.; Fan, J.; Wu, W.-Z. miR-296-5p suppresses EMT of hepatocellular carcinoma via attenuating NRG1/ERBB2/ERBB3 signaling. J. Exp. Clin. Cancer Res. 2018, 37, 294. [Google Scholar] [CrossRef]
- Chai, S.; Ng, K.Y.; Tong, M.; Lau, E.Y.; Lee, T.K.; Chan, K.W.; Yuan, Y.F.; Cheung, T.T.; Cheung, S.T.; Wang, X.Q.; et al. Octamer 4/microRNA-1246 signaling axis drives Wnt/β-catenin activation in liver cancer stem cells. Hepatology 2016, 64, 2062–2076. [Google Scholar] [CrossRef]
- Lin, X.; Zuo, S.; Luo, R.; Li, Y.; Yu, G.; Zou, Y.; Zhou, Y.; Liu, Z.; Liu, Y.; Hu, Y.; et al. HBX-induced miR-5188 impairs FOXO1 to stimulate β-catenin nuclear translocation and promotes tumor stemness in hepatocellular carcinoma. Theranostics 2019, 9, 7583–7598. [Google Scholar] [CrossRef]
- Shi, D.-M.; Bian, X.-Y.; Qin, C.-D.; Wu, W.-Z. miR-106b-5p promotes stem cell-like properties of hepatocellular carcinoma cells by targeting PTEN via PI3K/Akt pathway. OncoTargets Ther. 2018, 11, 571–585. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, B.; Xiao, S.; Li, Y.; Chen, Q. miR-125a/b inhibits tumor-associated macrophages mediated in cancer stem cells of hepatocellular carcinoma by targeting CD90. J. Cell. Biochem. 2018, 120, 3046–3055. [Google Scholar] [CrossRef]
- Li, B.; Liu, D.; Yang, P.; Li, H.-Y.; Wang, D. miR-613 inhibits liver cancer stem cell expansion by regulating SOX9 pathway. Gene 2019, 707, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Seol, H.S.; Akiyama, Y.; Lee, S.-E.; Shimada, S.; Jang, S.J. Loss of miR-100 and miR-125b results in cancer stem cell properties through IGF2 upregulation in hepatocellular carcinoma. Sci. Rep. 2020, 10, 21412. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Xu, T.; Wang, H.; Wu, N.; Liu, F.; Jia, X.; Mi, J.; Lv, J.; Gao, H. Dysregulation of the miR-325–3p/DPAGT1 axis supports HBV-positive HCC chemoresistance. Biochem. Biophys. Res. Commun. 2019, 519, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.C.; Yang, Y.J.; Zhang, J.Q.; Guo, M.; Xiang, L.; Yu, S.F.; Ping, H.; Zhuo, L. microRNA-448 inhibits stemness maintenance and self-renewal of hepatocellular carcinoma stem cells through the MAGEA6-mediated AMPK signaling pathway. J. Cell. Physiol. 2019, 234, 23461–23474. [Google Scholar] [CrossRef]
- Zhong, F.; Wang, Y. YY1-regulated lncRNA SOCS2-AS1 suppresses HCC cell stemness and progression via miR-454-3p/CPEB1. Biochem. Biophys. Res. Commun. 2023, 679, 98–109. [Google Scholar] [CrossRef]
- Chaffer, C.L.; Weinberg, R.A. A perspective on cancer cell metastasis. Science 2011, 331, 1559–1564. [Google Scholar] [CrossRef]
- Geiger, T.R.; Peeper, D.S. Metastasis mechanisms. Biochim. Biophys. Acta 2009, 1796, 293–308. [Google Scholar] [CrossRef]
- Loh, C.-Y.; Chai, J.Y.; Tang, T.F.; Wong, W.F.; Sethi, G.; Shanmugam, M.K.; Chong, P.P.; Looi, C.Y. The E-Cadherin and N-Cadherin Switch in Epithelial-to-Mesenchymal Transition: Signaling, Therapeutic Implications, and Challenges. Cells 2019, 8, 1118. [Google Scholar] [CrossRef]
- Ruiz-Manriquez, L.M.; Carrasco-Morales, O.; Sanchez, Z.E.; Osorio-Perez, S.M.; Estrada-Meza, C.; Pathak, S.; Banerjee, A.; Bandyopadhyay, A.; Duttaroy, A.K.; Paul, S. MicroRNA-mediated regulation of key signaling pathways in hepatocellular carcinoma: A mechanistic insight. Front. Genet. 2022, 13, 910733. [Google Scholar] [CrossRef]
- Mitsunobu, M.; Toyosaka, A.; Oriyama, T.; Okamoto, E.; Nakao, N. Intrahepatic metastases in hepatocellular carcinoma: The role of the portal vein as an efferent vessel. Clin. Exp. Metastasis 1996, 14, 520–529. [Google Scholar] [CrossRef]
- Katyal, S.; Oliver, J.H., 3rd; Peterson, M.S.; Ferris, J.V.; Carr, B.S.; Baron, R.L. Extrahepatic metastases of hepatocellular carcinoma. Radiology 2000, 216, 698–703. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Zhang, J.; Lin, Y.; Sun, X.-M.; Zhang, J.-N.; Cheng, Z.-Q. Identification of MiR-211-5p as a tumor suppressor by targeting ACSL4 in Hepatocellular Carcinoma. J. Transl. Med. 2020, 18, 326. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.; Liu, X.; Li, Z.; Wang, G.; Feng, Z.; Liu, Y.; Yang, H.; Tan, C.; Zhang, Z.; Li, K. LncRNA LINC00667 aggravates the progression of hepatocellular carcinoma by regulating androgen receptor expression as a miRNA-130a-3p sponge. Cell Death Discov. 2021, 7, 387. [Google Scholar] [CrossRef]
- Cai, Q.Q.; Dong, Y.W.; Wang, R.; Qi, B.; Guo, J.X.; Pan, J.; Liu, Y.Y.; Zhang, C.Y.; Wu, X.Z. MiR-124 inhibits the migration and invasion of human hepatocellular carcinoma cells by suppressing integrin αV expression. Sci. Rep. 2017, 7, 40733. [Google Scholar] [CrossRef]
- Zhao, B.; Lu, Y.; Cao, X.; Zhu, W.; Kong, L.; Ji, H.; Zhang, F.; Lin, X.; Guan, Q.; Ou, K. MiRNA-124 inhibits the proliferation, migration and invasion of cancer cell in hepatocellular carcinoma by downregulating lncRNA-UCA1. OncoTargets Ther. 2019, 12, 4509. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; He, Z.; Hu, X.; Ma, P. MiRNA-15a-3p inhibits the metastasis of hepatocellular carcinoma by interacting with HMOX1. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 12694–12700. [Google Scholar]
- Sung, Y.K.; Hwang, S.Y.; Park, M.K.; Bae, H.I.; Kim, W.H.; Kim, J.C.; Kim, M. Fatty acid-CoA ligase 4 is overexpressed in human hepatocellular carcinoma. Cancer Sci. 2003, 94, 421–424. [Google Scholar] [CrossRef]
- Sung, Y.K.; Park, M.K.; Hong, S.H.; Hwang, S.Y.; Kwack, M.H.; Kim, J.C.; Kim, M.K. Regulation of cell growth by fatty acid-CoA ligase 4 in human hepatocellular carcinoma cells. Exp. Mol. Med. 2007, 39, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Ding, C.; Chen, Y.; Hu, W.; Lu, Y.; Wu, W.; Zhang, Y.; Yang, B.; Wu, H.; Peng, C.; et al. ACSL4 promotes hepatocellular carcinoma progression via c-Myc stability mediated by ERK/FBW7/c-Myc axis. Oncogenesis 2020, 9, 42. [Google Scholar] [CrossRef]
- Nagasue, N.; Kohno, H.; Chang, Y.C.; Hayashi, T.; Utsumi, Y.; Nakamura, T.; Yukaya, H. Androgen and estrogen receptors in hepatocellular carcinoma and the surrounding liver in women. Cancer 1989, 63, 112–116. [Google Scholar] [CrossRef]
- Grosso, A.R.; Martins, S.; Carmo-Fonseca, M. The emerging role of splicing factors in cancer. EMBO Rep. 2008, 9, 1087–1093. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Goren, A.; Ast, G. Insights into the connection between cancer and alternative splicing. Trends Genet. 2008, 24, 7–10. [Google Scholar] [CrossRef]
- Shen, Q.; Nam, S.W. SF3B4 as an early-stage diagnostic marker and driver of hepatocellular carcinoma. BMB Rep. 2018, 51, 57–58. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Guan, J.-L. Focal adhesion kinase and its signaling pathways in cell migration and angiogenesis. Adv. Drug Deliv. Rev. 2011, 63, 610–615. [Google Scholar] [CrossRef] [PubMed]
- Qi, J.H.; Claesson-Welsh, L. VEGF-induced activation of phosphoinositide 3-kinase is dependent on focal adhesion kinase. Exp. Cell Res. 2001, 263, 173–182. [Google Scholar] [CrossRef]
- Kim, I.; Kim, H.G.; Moon, S.O.; Chae, S.W.; So, J.N.; Koh, K.N.; Ahn, B.C.; Koh, G.Y. Angiopoietin-1 induces endothelial cell sprouting through the activation of focal adhesion kinase and plasmin secretion. Circ. Res. 2000, 86, 952–959. [Google Scholar] [CrossRef]
- Liu, L.; Yang, X.; Li, N.F.; Lin, L.; Luo, H. Circ_0015756 promotes proliferation, invasion and migration by microRNA-7-dependent inhibition of FAK in hepatocellular carcinoma. Cell Cycle 2019, 18, 2939–2953. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.C.; Dostie, J.; Diem, M.D.; Feng, W.; Mann, M.; Rappsilber, J.; Dreyfuss, G. eIF4A3 is a novel component of the exon junction complex. Rna 2004, 10, 200–209. [Google Scholar] [CrossRef]
- Xia, Q.; Kong, X.-T.; Zhang, G.-A.; Hou, X.-J.; Qiang, H.; Zhong, R.-Q. Proteomics-based identification of DEAD-box protein 48 as a novel autoantigen, a prospective serum marker for pancreatic cancer. Biochem. Biophys. Res. Commun. 2005, 330, 526–532. [Google Scholar] [CrossRef]
- Lin, Y.; Liang, R.; Mao, Y.; Ye, J.; Mai, R.; Gao, X.; Liu, Z.; Wainwright, T.; Li, Q.; Luo, M. Comprehensive analysis of biological networks and the eukaryotic initiation factor 4A-3 gene as pivotal in hepatocellular carcinoma. J. Cell. Biochem. 2020, 121, 4094–4107. [Google Scholar] [CrossRef]
- Zhu, Y.; Ren, C.; Yang, L. Effect of eukaryotic translation initiation factor 4A3 in malignant tumors. Oncol. Lett. 2021, 21, 358. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, Y.; Bao, C.; Zhang, X.; Li, H. Eukaryotic initiation Factor 4AIII facilitates hepatocellular carcinoma cell proliferation, migration, and epithelial-mesenchymal transition process via antagonistically binding to WD repeat domain 66 with miRNA-2113. J. Cell. Physiol. 2020, 235, 8199–8209. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Lu, J.; Cao, J.; Ma, B.; Gao, C.; Qi, F. MicroRNA-18a promotes hepatocellular carcinoma proliferation, migration, and invasion by targeting Bcl2L10. OncoTargets Ther. 2018, 11, 7919–7934. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Cai, X.; Liu, E.; Tian, X.; Tian, C. MicroRNA-18a promotes proliferation and metastasis in hepatocellular carcinoma via targeting KLF4. Oncotarget 2017, 8, 68263–68269. [Google Scholar] [CrossRef]
- El-Mezayen, H.; Yamamura, K.; Yusa, T.; Nakao, Y.; Uemura, N.; Kitamura, F.; Itoyama, R.; Yamao, T.; Higashi, T.; Hayashi, H.; et al. MicroRNA-25 Exerts an Oncogenic Function by Regulating the Ubiquitin Ligase Fbxw7 in Hepatocellular Carcinoma. Ann. Surg. Oncol. 2021, 28, 7973–7982. [Google Scholar] [CrossRef]
- Yu, L.-X.; Zhang, B.-L.; Yang, M.-Y.; Liu, H.; Xiao, C.-H.; Zhang, S.-G.; Liu, R. MicroRNA-106b-5p promotes hepatocellular carcinoma development via modulating FOG2. OncoTargets Ther. 2019, 12, 5639–5647. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chang, W.; Chang, W.; Chang, X.; Zhai, S.; Pan, G.; Dang, S. MicroRNA-376c-3p Facilitates Human Hepatocellular Carcinoma Progression via Repressing AT-Rich Interaction Domain 2. J. Cancer 2018, 9, 4187–4196. [Google Scholar] [CrossRef]
- Shi, X.; Liu, T.T.; Yu, X.N.; Balakrishnan, A.; Zhu, H.R.; Guo, H.Y.; Zhang, G.C.; Bilegsaikhan, E.; Sun, J.L.; Song, G.Q.; et al. microRNA-93-5p promotes hepatocellular carcinoma progression via a microRNA-93-5p/MAP3K2/c-Jun positive feedback circuit. Oncogene 2020, 39, 5768–5781. [Google Scholar] [CrossRef]
- Lv, G.; Wu, M.; Wang, M.; Jiang, X.; Du, J.; Zhang, K.; Li, D.; Ma, N.; Peng, Y.; Wang, L.; et al. miR-320a regulates high mobility group box 1 expression and inhibits invasion and metastasis in hepatocellular carcinoma. Liver Int. 2017, 37, 1354–1364. [Google Scholar] [CrossRef]
- Xie, F.; Yuan, Y.; Xie, L.; Ran, P.; Xiang, X.; Huang, Q.; Qi, G.; Guo, X.; Xiao, C.; Zheng, S. miRNA-320a inhibits tumor proliferation and invasion by targeting c-Myc in human hepatocellular carcinoma. OncoTargets Ther. 2017, 10, 885–894. [Google Scholar] [CrossRef]
- Che, J.; Su, Z.; Yang, W.; Xu, L.; Li, Y.; Wang, H.; Zhou, W. Tumor-suppressor p53 specifically binds to miR-29c-3p and reduces ADAM12 expression in hepatocellular carcinoma. Dig. Liver Dis. 2023, 55, 412–421. [Google Scholar] [CrossRef]
- Wu, Y.-H.; Yu, B.; Zhou, J.-M.; Shen, X.-H.; Chen, W.-X.; Ai, X.; Leng, C.; Liang, B.-Y.; Shao, Y.-J. MicroRNA-188-5p inhibits hepatocellular carcinoma proliferation and migration by targeting forkhead box N2. BMC Cancer 2023, 23, 511. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Liu, Z.; Jin, L.; Zhang, F.; Peng, X.; Xiao, Y.; Wang, X.; Lyu, Q.; Cai, X. lncRNA TUG1-mediated Mir-142-3p downregulation contributes to metastasis and the epithelial-to-mesenchymal transition of hepatocellular carcinoma by targeting ZEB1. Cell. Physiol. Biochem. 2018, 48, 1928–1941. [Google Scholar] [CrossRef]
- Yu, Q.; Xiang, L.; Yin, L.; Liu, X.; Yang, D.; Zhou, J. Loss-of-function of miR-142 by hypermethylation promotes TGF-β-mediated tumour growth and metastasis in hepatocellular carcinoma. Cell Prolif. 2017, 50, e12384. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Sun, L.-Q.; Huang, Y.; Quan, J.; Hu, X.; Tang, D.; Kang, R.; Li, N.; Fan, X.-G. miR-142-3p Inhibits the Metastasis of Hepatocellular Carcinoma Cells by Regulating HMGB1 Gene Expression. Curr. Mol. Med. 2018, 18, 135–141. [Google Scholar] [CrossRef]
- Kim, H.S.; Kim, J.S.; Park, N.R.; Nam, H.; Sung, P.S.; Bae, S.H.; Choi, J.Y.; Yoon, S.K.; Hur, W.; Jang, J.W. Exosomal miR-125b Exerts Anti-Metastatic Properties and Predicts Early Metastasis of Hepatocellular Carcinoma. Front. Oncol. 2021, 11, 637247. [Google Scholar] [CrossRef]
- Liu, X.; Wang, H.; Yang, M.; Hou, Y.; Chen, Y.; Bie, P. Exosomal miR-29b from cancer-associated fibroblasts inhibits the migration and invasion of hepatocellular carcinoma cells. Transl. Cancer Res. 2020, 9, 2576. [Google Scholar] [CrossRef] [PubMed]
- Shen, D.; Zhao, H.Y.; Gu, A.D.; Wu, Y.W.; Weng, Y.H.; Li, S.J.; Song, J.Y.; Gu, X.F.; Qiu, J.; Zhao, W. miRNA-10a-5p inhibits cell metastasis in hepatocellular carcinoma via targeting SKA1. Kaohsiung J. Med. Sci. 2021, 37, 784–794. [Google Scholar] [CrossRef]
- Deng, L.; Tang, J.; Yang, H.; Cheng, C.; Lu, S.; Jiang, R.; Sun, B. MTA1 modulated by miR-30e contributes to epithelial-to-mesenchymal transition in hepatocellular carcinoma through an ErbB2-dependent pathway. Oncogene 2017, 36, 3976–3985. [Google Scholar] [CrossRef]
- Ma, L.; Tao, C.; Zhang, Y. MicroRNA-517c Functions as a Tumor Suppressor in Hepatocellular Carcinoma via Downregulation of KPNA2 and Inhibition of PI3K/AKT Pathway. J. Healthc. Eng. 2022, 2022, 7026174. [Google Scholar] [CrossRef]
- Liu, Y.; Xiao, X.; Wang, J.; Wang, Y.; Yu, Y. Silencing CircEIF3I/miR-526b-5p Axis Epigenetically Targets HGF/c-Met Signal to Hinder the Malignant Growth, Metastasis and Angiogenesis of Hepatocellular Carcinoma. Biochem. Genet. 2023, 61, 48–68. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, C.-f.; Sun, L.-b.; Li, Y.-c. microRNA-4270-5p inhibits cancer cell proliferation and metastasis in hepatocellular carcinoma by targeting SATB2. Hum. Cell 2020, 33, 1155–1164. [Google Scholar] [CrossRef]
- Majid, A.; Wang, J.; Nawaz, M.; Abdul, S.; Ayesha, M.; Guo, C.; Liu, Q.; Liu, S.; Sun, M.-Z. miR-124-3p suppresses the invasiveness and metastasis of hepatocarcinoma cells via targeting CRKL. Front. Mol. Biosci. 2020, 7, 223. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Huang, J.; Yang, Z.; Zhang, J.; Wang, X.; Xu, N.; Meng, H.; Wu, J.; Huang, Q.; Yang, X. miR-449a is related to short-term recurrence of hepatocellular carcinoma and inhibits migration and invasion by targeting notch1. OncoTargets Ther. 2019, 12, 10975. [Google Scholar] [CrossRef]
- Huang, Z.; Wen, J.; Yu, J.; Liao, J.; Liu, S.; Cai, N.; Liang, H.; Chen, X.; Ding, Z.; Zhang, B. MicroRNA-148a-3p inhibits progression of hepatocelluar carcimoma by repressing SMAD2 expression in an Ago2 dependent manner. J. Exp. Clin. Cancer Res. 2020, 39, 150. [Google Scholar] [CrossRef] [PubMed]
- Cao, N.; Mu, L.; Yang, W.; Liu, L.; Liang, L.; Zhang, H. MicroRNA-298 represses hepatocellular carcinoma progression by inhibiting CTNND1-mediated Wnt/β-catenin signaling. Biomed. Pharmacother. 2018, 106, 483–490. [Google Scholar] [CrossRef]
- Chang, J.-H.; Xu, B.-W.; Shen, D.; Zhao, W.; Wang, Y.; Liu, J.-L.; Meng, G.-X.; Li, G.-Z.; Zhang, Z.-L. BRF2 is mediated by microRNA-409-3p and promotes invasion and metastasis of HCC through the Wnt/β-catenin pathway. Cancer Cell Int. 2023, 23, 46. [Google Scholar] [CrossRef]
- Jia, C.; Tang, D.; Sun, C.; Yao, L.; Li, F.; Hu, Y.; Zhang, X.; Wu, D. MicroRNA-466 inhibits the aggressive behaviors of hepatocellular carcinoma by directly targeting metadherin. Oncol. Rep. 2018, 40, 3890–3898. [Google Scholar] [CrossRef]
- Yang, G.; Guo, S.; Liu, H.T.; Yang, G. MiR-138-5p predicts negative prognosis and exhibits suppressive activities in hepatocellular carcinoma HCC by targeting FOXC1. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 8788–8800. [Google Scholar]
- Hu, Y.; Yang, Z.; Bao, D.; Ni, J.-S.; Lou, J. miR-455-5p suppresses hepatocellular carcinoma cell growth and invasion via IGF-1R/AKT/GLUT1 pathway by targeting IGF-1R. Pathol. Res. Pract. 2019, 215, 152674. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; You, S.; Fan, R.; Shan, X. UCK2 regulated by miR-139-3p regulates the progression of hepatocellular carcinoma cells. Future Oncol. 2022, 18, 979–990. [Google Scholar] [CrossRef]
- Ahmed, F.; Onwumeh-Okwundu, J.; Yukselen, Z.; Endaya Coronel, M.-K.; Zaidi, M.; Guntipalli, P.; Garimella, V.; Gudapati, S.; Mezidor, M.D.; Andrews, K.; et al. Atezolizumab plus bevacizumab versus sorafenib or atezolizumab alone for unresectable hepatocellular carcinoma: A systematic review. World J. Gastrointest. Oncol. 2021, 13, 1813–1832. [Google Scholar] [CrossRef]
- Li, L.-M.; Chen, C.; Ran, R.-X.; Huang, J.-T.; Sun, H.-L.; Zeng, C.; Zhang, Z.; Zhang, W.; Liu, S.-M. Corrigendum: Loss of TARBP2 drives the progression of hepatocellular carcinoma via miR-145-SERPINE1 axis. Front. Oncol. 2021, 11, 620912. [Google Scholar] [CrossRef]
- Ni, J.-S.; Zheng, H.; Ou, Y.-L.; Tao, Y.-P.; Wang, Z.-G.; Song, L.-H.; Yan, H.-L.; Zhou, W.-P. miR-515–5p suppresses HCC migration and invasion via targeting IL6/JAK/STAT3 pathway. Surg. Oncol. 2020, 34, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhou, T.; Cheng, X.; Li, D.; Zhao, M.; Zheng, W.V. microRNA-378a-3p regulates the progression of hepatocellular carcinoma by regulating PD-L1 and STAT3. Bioengineered 2022, 13, 4730–4743. [Google Scholar] [CrossRef]
- Zhan, Y.; Zheng, N.; Teng, F.; Bao, L.; Liu, F.; Zhang, M.; Guo, M.; Guo, W.; Ding, G.; Wang, Q. MiR-199a/b-5p inhibits hepatocellular carcinoma progression by post-transcriptionally suppressing ROCK1. Oncotarget 2017, 8, 67169. [Google Scholar] [CrossRef] [PubMed]
- Zou, H.; Yang, L. miR-378a-5p improved the prognosis and suppressed the progression of hepatocellular carcinoma by targeting the VEGF pathway. Transl. Cancer Res. 2020, 9, 1558–1566. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Zhuang, J.; Wang, G.; He, S.; Zhang, S.; Wang, G.; Ni, J.; Wang, J.; Xia, W. MicroRNA-495 suppresses cell proliferation and invasion of hepatocellular carcinoma by directly targeting insulin-like growth factor receptor-1. Exp. Ther. Med. 2018, 15, 1150–1158. [Google Scholar] [PubMed]
- Liu, B.; Tian, Y.; Chen, M.; Shen, H.; Xia, J.; Nan, J.; Yan, T.; Wang, Y.; Shi, L.; Shen, B. CircUBAP2 promotes MMP9-mediated oncogenic effect via sponging miR-194-3p in hepatocellular carcinoma. Front. Cell Dev. Biol. 2021, 9, 1634. [Google Scholar] [CrossRef]
- Lin, Y.; Liu, J.; Huang, Y.; Liu, D.; Zhang, G.; Kan, H. microRNA-489 plays an anti-metastatic role in human hepatocellular carcinoma by targeting matrix metalloproteinase-7. Transl. Oncol. 2017, 10, 211–220. [Google Scholar] [CrossRef]
- Hong, H.; Sui, C.; Qian, T.; Xu, X.; Zhu, X.; Fei, Q.; Yang, J.; Xu, M. Long noncoding RNA LINC00460 conduces to tumor growth and metastasis of hepatocellular carcinoma through miR-342-3p-dependent AGR2 up-regulation. Aging 2020, 12, 10544–10555. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Dai, S.-J.; Dong, L.; Li, H. Long noncoding RNA RP11-909N17. 2 promotes proliferation, invasion, and migration of hepatocellular carcinoma by regulating microRNA-767-3p. Biochem. Cell Biol. 2020, 98, 709–718. [Google Scholar] [CrossRef]
- He, H.; Wang, Y.; Ye, P.; Yi, D.; Cheng, Y.; Tang, H.; Zhu, Z.; Wang, X.; Jin, S. Long noncoding RNA ZFPM2-AS1 acts as a miRNA sponge and promotes cell invasion through regulation of miR-139/GDF10 in hepatocellular carcinoma. J. Exp. Clin. Cancer Res. 2020, 39, 159. [Google Scholar] [CrossRef]
- Sandbothe, M.; Buurman, R.; Reich, N.; Greiwe, L.; Vajen, B.; Gürlevik, E.; Schäffer, V.; Eilers, M.; Kühnel, F.; Vaquero, A.; et al. The microRNA-449 family inhibits TGF-β-mediated liver cancer cell migration by targeting SOX4. J. Hepatol. 2017, 66, 1012–1021. [Google Scholar] [CrossRef]
- Li, Q.; Li, S.; Wu, Y.; Gao, F. miRNA-708 functions as a tumour suppressor in hepatocellular carcinoma by targeting SMAD3. Oncol. Lett. 2017, 14, 2552–2558. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Guo, Y.; Li, Y.; Yang, J.; Liu, J.; Wu, Q.; Wang, R. miR-300 regulates tumor proliferation and metastasis by targeting lymphoid enhancer-binding factor 1 in hepatocellular carcinoma. Int. J. Oncol. 2019, 54, 1282–1294. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Sun, P.; Zhang, C.; Li, Z.; Zhou, W. MiR-98 suppresses the effects of tumor-associated macrophages on promoting migration and invasion of hepatocellular carcinoma cells by regulating IL-10. Biochimie 2018, 150, 23–30. [Google Scholar] [CrossRef]
- Zhang, J.-J.; Chen, J.-T.; Hua, L.; Yao, K.-H.; Wang, C.-Y. miR-98 inhibits hepatocellular carcinoma cell proliferation via targeting EZH2 and suppressing Wnt/β-catenin signaling pathway. Biomed. Pharmacother. 2017, 85, 472–478. [Google Scholar] [CrossRef]
- Zhang, Q.; Huang, F.; Yao, Y.; Wang, J.; Wei, J.; Wu, Q.; Xiang, S.; Xu, L. Interaction of transforming growth factor-β-Smads/microRNA-362-3p/CD82 mediated by M2 macrophages promotes the process of epithelial-mesenchymal transition in hepatocellular carcinoma cells. Cancer Sci. 2019, 110, 2507–2519. [Google Scholar] [CrossRef]
- Wang, J.; Zhu, Y.; Ai, X.; Wan, H.; Jia, W.; Chu, J.; Xu, B.; Kong, X.; Kong, L. Long noncoding RNA 02027 inhibits proliferation, migration and invasion of hepatocellular carcinoma via miR-625-3p/PDLIM5 pathway. J. Gene Med. 2023, 25, e3485. [Google Scholar] [CrossRef]
- Yang, B.; Feng, X.; Liu, H.; Tong, R.; Wu, J.; Li, C.; Yu, H.; Chen, Y.; Cheng, Q.; Chen, J.; et al. High-metastatic cancer cells derived exosomal miR92a-3p promotes epithelial-mesenchymal transition and metastasis of low-metastatic cancer cells by regulating PTEN/Akt pathway in hepatocellular carcinoma. Oncogene 2020, 39, 6529–6543. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Cui, M.; Qu, F.; Cheng, D.; Yu, J.; Tang, Z.; Cheng, L.; Wei, Y.; Wu, X.; Liu, X. MiR-92a-3p promotes the malignant progression of hepatocellular carcinoma by mediating the PI3K/AKT/mTOR signaling pathway. Curr. Pharm. Des. 2021, 27, 3244–3250. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Zhou, Y.; Huan, L.; Xu, L.; Shen, M.; Huang, S.; Liang, L. LncRNA MIR22HG inhibits growth, migration and invasion through regulating the miR-10a-5p/NCOR2 axis in hepatocellular carcinoma cells. Cancer Sci. 2019, 110, 973–984. [Google Scholar] [CrossRef]
- Guo, L.; Gao, R.; Gan, J.; Zhu, Y.; Ma, J.; Lv, P.; Zhang, Y.; Li, S.; Tang, H. Downregulation of TNFRSF19 and RAB43 by a novel miRNA, miR-HCC3, promotes proliferation and epithelial–mesenchymal transition in hepatocellular carcinoma cells. Biochem. Biophys. Res. Commun. 2020, 525, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Wang, P.; Lin, J.; Zheng, X.; Yang, F.; Zhang, G.; Chen, D.; Xie, J.; Gao, Z.; Peng, L. MicroRNA-197 promotes metastasis of hepatocellular carcinoma by activating Wnt/β-catenin signaling. Cell. Physiol. Biochem. 2018, 51, 470–486. [Google Scholar]
- Hong, Y.; Ye, M.; Wang, F.; Fang, J.; Wang, C.; Luo, J.; Liu, J.; Liu, J.; Liu, L.; Zhao, Q. MiR-21-3p promotes hepatocellular carcinoma progression via SMAD7/YAP1 regulation. Front. Oncol. 2021, 11, 642030. [Google Scholar] [CrossRef]
- Du, W.; Zhang, X.; Wan, Z. miR-3691-5p promotes hepatocellular carcinoma cell migration and invasion through activating PI3K/Akt signaling by targeting PTEN. OncoTargets Ther. 2019, 12, 4897–4906. [Google Scholar] [CrossRef]
- Qian, Y.; Chen, H.; Chen, L.; Ge, C.; Zhu, D.; Zhou, D. Suppression of hepatocellular carcinoma progression by long noncoding RNA apolipoprotein C1 pseudogene via the regulation of the microRNA-106b-PTEN axis. Transl. Cancer Res. 2023, 12, 3752–3763. [Google Scholar] [CrossRef]
- Cao, M.-Q.; You, A.B.; Zhu, X.-D.; Zhang, W.; Zhang, Y.-Y.; Zhang, S.-Z.; Zhang, K.-w.; Cai, H.; Shi, W.-K.; Li, X.-L. miR-182-5p promotes hepatocellular carcinoma progression by repressing FOXO3a. J. Hematol. Oncol. 2018, 11, 12. [Google Scholar] [CrossRef]
- Xian, Y.; Wang, L.; Yao, B.; Yang, W.; Mo, H.; Zhang, L.; Tu, K. MicroRNA-769-5p contributes to the proliferation, migration and invasion of hepatocellular carcinoma cells by attenuating RYBP. Biomed. Pharmacother. 2019, 118, 109343. [Google Scholar] [CrossRef]
- Han, S.; Wang, L.; Sun, L.; Wang, Y.; Yao, B.; Chen, T.; Liu, R.; Liu, Q. MicroRNA-1251-5p promotes tumor growth and metastasis of hepatocellular carcinoma by targeting AKAP12. Biomed. Pharmacother. 2020, 122, 109754. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Chen, Y.; Feng, S.; Yi, T.; Liu, X.; Li, Q.; Liu, Z.; Zhu, C.; Hu, J.; Yu, X. MiR-483-5p promotes IGF-II transcription and is associated with poor prognosis of hepatocellular carcinoma. Oncotarget 2017, 8, 99871. [Google Scholar] [CrossRef]
- Liu, Y.-M.; Cao, Y.; Zhao, P.-S.; Wu, L.-Y.; Lu, Y.-M.; Wang, Y.-L.; Zhao, J.-F.; Liu, X.-G. CircCCNB1 silencing acting as a miR-106b-5p sponge inhibited GPM6A expression to promote HCC progression by enhancing DYNC1I1 expression and activating the AKT/ERK signaling pathway. Int. J. Biol. Sci. 2022, 18, 637. [Google Scholar] [CrossRef]
- Fu, X.; Tang, Y.; Wu, W.; Ouyang, Y.; Tan, D.; Huang, Y. Exosomal microRNA-25 released from cancer cells targets SIK1 to promote hepatocellular carcinoma tumorigenesis. Dig. Liver Dis. 2021, 54, 954–963. [Google Scholar] [CrossRef]
- Liu, H.; Chen, W.; Zhi, X.; Chen, E.-J.; Wei, T.; Zhang, J.; Shen, J.; Hu, L.-Q.; Zhao, B.; Feng, X.-H.; et al. Tumor-derived exosomes promote tumor self-seeding in hepatocellular carcinoma by transferring miRNA-25-5p to enhance cell motility. Oncogene 2018, 37, 4964–4978. [Google Scholar] [CrossRef]
- Zhou, Y.; Ren, H.; Dai, B.; Li, J.; Shang, L.; Huang, J.; Shi, X. Hepatocellular carcinoma-derived exosomal miRNA-21 contributes to tumor progression by converting hepatocyte stellate cells to cancer-associated fibroblasts. J. Exp. Clin. Cancer Res. 2018, 37, 324. [Google Scholar] [CrossRef]
- Yu, Y.; Min, Z.; Zhou, Z.; Linhong, M.; Tao, R.; Yan, L.; Song, H. Hypoxia-induced exosomes promote hepatocellular carcinoma proliferation and metastasis via miR-1273f transfer. Exp. Cell Res. 2019, 385, 111649. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Xu, H.-F.; Liu, M.-Y.; Xu, Y.-J.; He, J.-C.; Zhou, Y.; Cang, S.-D. Mechanism of exosomal microRNA-224 in development of hepatocellular carcinoma and its diagnostic and prognostic value. World J. Gastroenterol. 2019, 25, 1890–1898. [Google Scholar] [CrossRef]
- Yang, Y.; Mao, F.; Guo, L.; Shi, J.; Wu, M.; Cheng, S.; Guo, W. Tumor cells derived-extracellular vesicles transfer miR-3129 to promote hepatocellular carcinoma metastasis by targeting TXNIP. Dig. Liver Dis. 2021, 53, 474–485. [Google Scholar] [CrossRef]
- Fang, T.; Lv, H.; Lv, G.; Li, T.; Wang, C.; Han, Q.; Yu, L.; Su, B.; Guo, L.; Huang, S. Tumor-derived exosomal miR-1247-3p induces cancer-associated fibroblast activation to foster lung metastasis of liver cancer. Nat. Commun. 2018, 9, 191. [Google Scholar] [CrossRef]
- Zhuyan, J.; Chen, M.; Zhu, T.; Bao, X.; Zhen, T.; Xing, K.; Wang, Q.; Zhu, S. Critical steps to tumor metastasis: Alterations of tumor microenvironment and extracellular matrix in the formation of pre-metastatic and metastatic niche. Cell Biosci. 2020, 10, 89. [Google Scholar] [CrossRef] [PubMed]
- Anderson, N.M.; Simon, M.C. The tumor microenvironment. Curr. Biol. 2020, 30, R921–R925. [Google Scholar] [CrossRef] [PubMed]
- Brassart-Pasco, S.; Brézillon, S.; Brassart, B.; Ramont, L.; Oudart, J.-B.; Monboisse, J.C. Tumor Microenvironment: Extracellular Matrix Alterations Influence Tumor Progression. Front. Oncol. 2020, 10, 397. [Google Scholar] [CrossRef]
- Yu, S.; Cao, H.; Shen, B.; Feng, J. Tumor-derived exosomes in cancer progression and treatment failure. Oncotarget 2015, 6, 37151–37168. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhou, T.; Chen, J.; Li, R.; Chen, H.; Luo, S.; Chen, D.; Cai, C.; Li, W. The role of Exosomal miRNAs in cancer. J. Transl. Med. 2022, 20, 6. [Google Scholar] [CrossRef]
- Li, S.; Yao, J.; Xie, M.; Liu, Y.; Zheng, M. Exosomal miRNAs in hepatocellular carcinoma development and clinical responses. J. Hematol. Oncol. 2018, 11, 54. [Google Scholar] [CrossRef]
- Semenza, G.L. Hypoxia-inducible factors: Mediators of cancer progression and targets for cancer therapy. Trends Pharmacol. Sci. 2012, 33, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Lu, Q.; Liu, Y.; Zhao, J.; Zhang, Q.; Hu, L.; Shi, Z.; Tu, Y.; Xiao, Z.; Xu, Q.; et al. Effect of the Hypoxia Inducible Factor on Sorafenib Resistance of Hepatocellular Carcinoma. Front. Oncol. 2021, 11, 641522. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.P.; Wang, C.Y.; Jin, X.H.; Li, M.; Wang, F.W.; Huang, W.J.; Yun, J.P.; Xu, R.H.; Cai, Q.Q.; Xie, D. Acidic Microenvironment Up-Regulates Exosomal miR-21 and miR-10b in Early-Stage Hepatocellular Carcinoma to Promote Cancer Cell Proliferation and Metastasis. Theranostics 2019, 9, 1965–1979. [Google Scholar] [CrossRef]
- Kim, M.-Y.; Oskarsson, T.; Acharyya, S.; Nguyen, D.X.; Zhang, X.H.F.; Norton, L.; Massagué, J. Tumor self-seeding by circulating cancer cells. Cell 2009, 139, 1315–1326. [Google Scholar] [CrossRef]
- Comen, E.; Norton, L.; Massagué, J. Clinical implications of cancer self-seeding. Nat. Rev. Clin. Oncol. 2011, 8, 369–377. [Google Scholar] [CrossRef]
- Jayatilaka, H.; Phillip, J.M. Targeting metastasis through the inhibition of interleukin 6 and 8. Future Med. 2019, 8, BMT20. [Google Scholar] [CrossRef]
- Hughey, C.C.; James, F.D.; Wang, Z.; Goelzer, M.; Wasserman, D.H. Dysregulated transmethylation leading to hepatocellular carcinoma compromises redox homeostasis and glucose formation. Mol. Metab. 2019, 23, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.; Tian, Y.; Niu, G.; Cao, C. Role of microRNAs in remodeling the tumor microenvironment. Int. J. Oncol. 2020, 56, 407–416. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Guo, H.; Wang, X.; Zhu, X.; Yan, M.; Wang, X.; Xu, Q.; Shi, J.; Lu, E.; Chen, W. Exosomal miR-196a derived from cancer-associated fibroblasts confers cisplatin resistance in head and neck cancer through targeting CDKN1B and ING5. Genome Biol. 2019, 20, 12. [Google Scholar] [CrossRef]
- Qin, X.; Yan, M.; Zhang, J.; Wang, X.; Shen, Z.; Lv, Z.; Li, Z.; Wei, W.; Chen, W. TGFβ3-mediated induction of Periostin facilitates head and neck cancer growth and is associated with metastasis. Sci. Rep. 2016, 6, 20587. [Google Scholar] [CrossRef]
- Linares, J.; Marín-Jiménez, J.A.; Badia-Ramentol, J.; Calon, A. Determinants and Functions of CAFs Secretome During Cancer Progression and Therapy. Front. Cell Dev. Biol. 2021, 8, 621070. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.; Liang, S.; Lin, W.; Li, S.; Yuan, J.; Jin, M.; Nie, S.; Zhang, Y.; Zhai, X.; Zhou, L.; et al. Hypoxia-induced exosomes facilitate lung pre-metastatic niche formation in hepatocellular carcinoma through the miR-4508-RFX1-IL17A-p38 MAPK-NF-κB pathway. Int. J. Biol. Sci. 2023, 19, 4744–4762. [Google Scholar] [CrossRef]
- Yugawa, K.; Yoshizumi, T.; Mano, Y.; Itoh, S.; Harada, N.; Ikegami, T.; Kohashi, K.; Oda, Y.; Mori, M. Cancer-associated fibroblasts promote hepatocellular carcinoma progression through downregulation of exosomal miR-150-3p. Eur. J. Surg. Oncol. 2021, 47, 384–393. [Google Scholar] [CrossRef]
- Eun, J.W.; Ahn, H.R.; Baek, G.O.; Yoon, M.G.; Son, J.A.; Weon, J.H.; Yoon, J.H.; Kim, H.S.; Han, J.E.; Kim, S.S.; et al. Aberrantly Expressed MicroRNAs in Cancer-Associated Fibroblasts and Their Target Oncogenic Signatures in Hepatocellular Carcinoma. Int. J. Mol. Sci. 2023, 24, 4272. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wu, T.; Zheng, B.; Chen, L. Individualized precision treatment: Targeting TAM in HCC. Cancer Lett. 2019, 458, 86–91. [Google Scholar] [CrossRef]
- Zhu, Y.; Yang, J.; Xu, D.; Gao, X.-M.; Zhang, Z.; Hsu, J.L.; Li, C.-W.; Lim, S.-O.; Sheng, Y.-Y.; Zhang, Y. Disruption of tumour-associated macrophage trafficking by the osteopontin-induced colony-stimulating factor-1 signalling sensitises hepatocellular carcinoma to anti-PD-L1 blockade. Gut 2019, 68, 1653–1666. [Google Scholar] [CrossRef] [PubMed]
- Nakano, T.; Chen, C.-L.; Chen, I.-H.; Tseng, H.-P.; Chiang, K.-C.; Lai, C.-Y.; Hsu, L.-W.; Goto, S.; Lin, C.-C.; Cheng, Y.-F. Overexpression of miR-4669 Enhances Tumor Aggressiveness and Generates an Immunosuppressive Tumor Microenvironment in Hepatocellular Carcinoma: Its Clinical Value as a Predictive Biomarker. Int. J. Mol. Sci. 2023, 24, 7908. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Chen, J.; Zhao, Y.; Yan, C.; Wang, Y.; Zhu, J.; Li, L. Exosomal miR-452-5p Induce M2 Macrophage Polarization to Accelerate Hepatocellular Carcinoma Progression by Targeting TIMP3. J. Immunol. Res. 2022, 2022, 1032106. [Google Scholar] [CrossRef]
- Zhao, J.; Li, H.; Zhao, S.; Wang, E.; Zhu, J.; Feng, D.; Zhu, Y.; Dou, W.; Fan, Q.; Hu, J. Epigenetic silencing of miR-144/451a cluster contributes to HCC progression via paracrine HGF/MIF-mediated TAM remodeling. Mol. Cancer 2021, 20, 46. [Google Scholar] [CrossRef]
- Ke, M.; Zhang, Z.; Cong, L.; Zhao, S.; Li, Y.; Wang, X.; Lv, Y.; Zhu, Y.; Dong, J. MicroRNA-148b-colony-stimulating factor-1 signaling-induced tumor-associated macrophage infiltration promotes hepatocellular carcinoma metastasis. Biomed. Pharmacother. 2019, 120, 109523. [Google Scholar] [CrossRef]
- Zhou, S.L.; Hu, Z.Q.; Zhou, Z.J.; Dai, Z.; Wang, Z.; Cao, Y.; Fan, J.; Huang, X.W.; Zhou, J. miR-28-5p-IL-34-macrophage feedback loop modulates hepatocellular carcinoma metastasis. Hepatology 2016, 63, 1560–1575. [Google Scholar] [CrossRef]
- Xing, Y.; Wang, Z.; Lu, Z.; Xia, J.; Xie, Z.; Jiao, M.; Liu, R.; Chu, Y. MicroRNAs: Immune modulators in cancer immunotherapy. Immunother. Adv. 2021, 1, ltab006. [Google Scholar] [CrossRef]
- Han, Y.; Liu, D.; Li, L. PD-1/PD-L1 pathway: Current researches in cancer. Am. J. Cancer Res. 2020, 10, 727. [Google Scholar]
- Fu, Y.; Mackowiak, B.; Feng, D.; Lu, H.; Guan, Y.; Lehner, T.; Pan, H.; Wang, X.W.; He, Y.; Gao, B. MicroRNA-223 attenuates hepatocarcinogenesis by blocking hypoxia-driven angiogenesis and immunosuppression. Gut 2023, 72, 1942–1958. [Google Scholar] [CrossRef]
- Wang, Y.; Cao, K. KDM1A Promotes Immunosuppression in Hepatocellular Carcinoma by Regulating PD-L1 through Demethylating MEF2D. J. Immunol. Res. 2021, 2021, 9965099. [Google Scholar] [CrossRef]
- Sun, C.; Lan, P.; Han, Q.; Huang, M.; Zhang, Z.; Xu, G.; Song, J.; Wang, J.; Wei, H.; Zhang, J. Oncofetal gene SALL4 reactivation by hepatitis B virus counteracts miR-200c in PD-L1-induced T cell exhaustion. Nat. Commun. 2018, 9, 1241. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Setayesh, T.; Vaziri, F.; Wu, X.; Hwang, S.T.; Chen, X.; Wan, Y.-J.Y. miR-22 gene therapy treats HCC by promoting anti-tumor immunity and enhancing metabolism. Mol. Ther. 2023, 31, 1829–1845. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Guo, S.; Stiles, J.K. The emerging role of CXCL10 in cancer (Review). Oncol. Lett. 2011, 2, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Decalf, J.; Tarbell, K.V.; Casrouge, A.; Price, J.D.; Linder, G.; Mottez, E.; Sultanik, P.; Mallet, V.; Pol, S.; Duffy, D.; et al. Inhibition of DPP4 activity in humans establishes its in vivo role in CXCL10 post-translational modification: Prospective placebo-controlled clinical studies. EMBO Mol. Med. 2016, 8, 679–683. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.-Y.; Zhang, P.-F.; Wei, C.-Y.; Peng, R.; Lu, J.-C.; Gao, C.; Cai, J.-B.; Yang, X.; Fan, J.; Ke, A.-W. Circular RNA circMET drives immunosuppression and anti-PD1 therapy resistance in hepatocellular carcinoma via the miR-30-5p/snail/DPP4 axis. Mol. Cancer 2020, 19, 92. [Google Scholar] [CrossRef]
- Hai, Y.; Hong, Y.; Yang, Y. miR-1258 Enhances the Anti-Tumor Effect of Liver Cancer Natural Killer (NK) Cells by Stimulating Toll-Liker Receptor (TLR) 7/8 to Promote Natural Killer (NK)-Dendritic Cell (DC) Interaction. J. Biomater. Tissue Eng. 2022, 12, 1241–1246. [Google Scholar] [CrossRef]
- Xie, H.; Zhang, Q.; Zhou, H.; Zhou, J.; Zhang, J.; Jiang, Y.; Wang, J.; Meng, X.; Zeng, L.; Jiang, X. microRNA-889 is downregulated by histone deacetylase inhibitors and confers resistance to natural killer cytotoxicity in hepatocellular carcinoma cells. Cytotechnology 2018, 70, 513–521. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, X.; Sun, W.; Yue, S.; Yang, J.; Li, J.; Ma, B.; Wang, J.; Yang, X.; Pu, M. Loss of exosomal miR-320a from cancer-associated fibroblasts contributes to HCC proliferation and metastasis. Cancer Lett. 2017, 397, 33–42. [Google Scholar] [CrossRef]
- Chai, Z.-T.; Zhu, X.-D.; Ao, J.-Y.; Wang, W.-Q.; Gao, D.-M.; Kong, J.; Zhang, N.; Zhang, Y.-Y.; Ye, B.-G.; Ma, D.-N.; et al. microRNA-26a suppresses recruitment of macrophages by down-regulating macrophage colony-stimulating factor expression through the PI3K/Akt pathway in hepatocellular carcinoma. J. Hematol. Oncol. 2015, 8, 56. [Google Scholar] [CrossRef]
- Wang, L.; Yi, X.; Xiao, X.; Zheng, Q.; Ma, L.; Li, B. miR-628-5p from M1 polarized macrophages hinders m6A modification of circFUT8 to suppress hepatocellular carcinoma progression. Cell. Mol. Biol. Lett. 2022, 27, 106. [Google Scholar] [CrossRef]
- Chen, L.; Gibbons, D.L.; Goswami, S.; Cortez, M.A.; Ahn, Y.H.; Byers, L.A.; Zhang, X.; Yi, X.; Dwyer, D.; Lin, W.; et al. Metastasis is regulated via microRNA-200/ZEB1 axis control of tumor cell PD-L1 expression and intratumoral immunosuppression. Nat. Commun. 2015, 5, 5241. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Zhang, P.; Liang, H.; Xu, Y.; Shen, J.; Wang, W.; Li, M.; Huang, J.; Ni, C.; Zhang, X.; et al. Circular RNA hsa_circ_0003288 induces EMT and invasion by regulating hsa_circ_0003288/miR-145/PD-L1 axis in hepatocellular carcinoma. Cancer Cell Int. 2021, 21, 212. [Google Scholar] [CrossRef] [PubMed]
- Fan, F.; Chen, K.; Lu, X.; Li, A.; Liu, C.; Wu, B. Dual targeting of PD-L1 and PD-L2 by PCED1B-AS1 via sponging hsa-miR-194-5p induces immunosuppression in hepatocellular carcinoma. Hepatol. Int. 2021, 15, 444–458. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Liu, S.; Su, L.; Su, Q.; Lin, J.; Huang, X.; Wang, C. miR-570 Inhibits Proliferation, Angiogenesis, and Immune Escape of Hepatocellular Carcinoma. Cancer Biother. Radiopharm. 2018, 33, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Wang, B.; Zeng, J.; Sang, S.; Lei, J.; Lu, Y. MicroRNA-374b inhibits liver cancer progression via down regulating programmed cell death-1 expression on cytokine-induced killer cells. Oncol. Lett. 2018, 15, 4797–4804. [Google Scholar] [CrossRef]
- Xu, Y.; Luan, G.; Liu, F.; Zhang, Y.; Li, Z.; Liu, Z.; Yang, T. Exosomal miR-200b-3p induce macrophage polarization by regulating transcriptional repressor ZEB1 in hepatocellular carcinoma. Hepatol. Int. 2023, 17, 889–903. [Google Scholar] [CrossRef]
- Liu, G.; Ouyang, X.; Sun, Y.; Xiao, Y.; You, B.; Gao, Y.; Yeh, S.; Li, Y.; Chang, C. The miR-92a-2-5p in exosomes from macrophages increases liver cancer cells invasion via altering the AR/PHLPP/p-AKT/β-catenin signaling. Cell Death Differ. 2020, 27, 3258–3272. [Google Scholar] [CrossRef]
- Li, W.; Xin, X.; Li, X.; Geng, J.; Sun, Y. Exosomes secreted by M2 macrophages promote cancer stemness of hepatocellular carcinoma via the miR-27a-3p/TXNIP pathways. Int. Immunopharmacol. 2021, 101, 107585. [Google Scholar] [CrossRef]
- Tian, B.; Zhou, L.; Wang, J.; Yang, P. miR-660-5p-loaded M2 macrophages-derived exosomes augment hepatocellular carcinoma development through regulating KLF3. Int. Immunopharmacol. 2021, 101, 108157. [Google Scholar] [CrossRef]
- Herbert, S.P.; Stainier, D.Y.R. Molecular control of endothelial cell behaviour during blood vessel morphogenesis. Nat. Rev. Mol. Cell Biol. 2011, 12, 551–564. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Folkman, J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 1996, 86, 353–364. [Google Scholar] [CrossRef]
- Ghosh, A.; Dasgupta, D.; Ghosh, A.; Roychoudhury, S.; Kumar, D.; Gorain, M.; Butti, R.; Datta, S.; Agarwal, S.; Gupta, S. MiRNA199a-3p suppresses tumor growth, migration, invasion and angiogenesis in hepatocellular carcinoma by targeting VEGFA, VEGFR1, VEGFR2, HGF and MMP2. Cell Death Dis. 2017, 8, e2706. [Google Scholar] [CrossRef] [PubMed]
- Contarelli, S.; Fedele, V.; Melisi, D. HOX Genes Family and Cancer: A Novel Role for Homeobox B9 in the Resistance to Anti-Angiogenic Therapies. Cancers 2020, 12, 3299. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Tong, D.; Guo, Q.; Wang, X.; Wu, F.; Li, Q.; Yang, J.; Zhao, L.; Qin, Y.; Liu, Y. HOXD3 targeted by miR-203a suppresses cell metastasis and angiogenesis through VEGFR in human hepatocellular carcinoma cells. Sci. Rep. 2018, 8, 2431. [Google Scholar] [CrossRef] [PubMed]
- Hou, M.; Lai, Y.; He, S.; He, W.; Shen, H.; Ke, Z. SGK3 (CISK) may induce tumor angiogenesis (Hypothesis). Oncol. Lett. 2015, 10, 23–26. [Google Scholar] [CrossRef]
- Wu, M.; Huang, C.; Huang, X.; Liang, R.; Feng, Y.; Luo, X. MicroRNA-144-3p suppresses tumor growth and angiogenesis by targeting SGK3 in hepatocellular carcinoma. Oncol. Rep. 2017, 38, 2173–2181. [Google Scholar] [CrossRef]
- Shah, A.V.; Birdsey, G.M.; Randi, A.M. Regulation of endothelial homeostasis, vascular development and angiogenesis by the transcription factor ERG. Vasc. Pharmacol. 2016, 86, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Moh-Moh-Aung, A.; Fujisawa, M.; Ito, S.; Katayama, H.; Ohara, T.; Ota, Y.; Yoshimura, T.; Matsukawa, A. Decreased miR-200b-3p in cancer cells leads to angiogenesis in HCC by enhancing endothelial ERG expression. Sci. Rep. 2020, 10, 10418. [Google Scholar] [CrossRef]
- Zhang, P.; Ha, M.; Li, L.; Huang, X.; Liu, C. MicroRNA-3064-5p sponged by MALAT1 suppresses angiogenesis in human hepatocellular carcinoma by targeting the FOXA1/CD24/Src pathway. FASEB J. 2020, 34, 66–81. [Google Scholar] [CrossRef]
- Orso, F.; Quirico, L.; Dettori, D.; Coppo, R.; Virga, F.; Ferreira, L.C.; Paoletti, C.; Baruffaldi, D.; Penna, E.; Taverna, D. Role of miRNAs in tumor and endothelial cell interactions during tumor progression. Semin. Cancer Biol. 2020, 60, 214–224. [Google Scholar] [CrossRef]
- Muz, B.; de la Puente, P.; Azab, F.; Azab, A.K. The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia 2015, 3, 83–92. [Google Scholar] [CrossRef]
- Matsuura, Y.; Wada, H.; Eguchi, H.; Gotoh, K.; Kobayashi, S.; Kinoshita, M.; Kubo, M.; Hayashi, K.; Iwagami, Y.; Yamada, D. Exosomal miR-155 derived from hepatocellular carcinoma cells under hypoxia promotes angiogenesis in endothelial cells. Dig. Dis. Sci. 2019, 64, 792–802. [Google Scholar] [CrossRef] [PubMed]
- Rhoads, K.; Arderiu, G.; Charboneau, A.; Hansen, S.L.; Hoffman, W.; Boudreau, N. A role for Hox A5 in regulating angiogenesis and vascular patterning. Lymphat. Res. Biol. 2005, 3, 240–252. [Google Scholar] [CrossRef]
- Liao, Y.; Wang, C.; Yang, Z.; Liu, W.; Yuan, Y.; Li, K.; Zhang, Y.; Wang, Y.; Shi, Y.; Qiu, Y. Dysregulated Sp1/miR-130b-3p/HOXA5 axis contributes to tumor angiogenesis and progression of hepatocellular carcinoma. Theranostics 2020, 10, 5209. [Google Scholar] [CrossRef]
- Schwarte-Waldhoff, I.; Schmiegel, W. Smad4 transcriptional pathways and angiogenesis. Int. J. Gastrointest. Cancer 2002, 31, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.-J.; Fang, J.-H.; Yang, X.-J.; Zhang, C.; Yuan, Y.; Zheng, L.; Zhuang, S.-M. Hepatocellular carcinoma cell-secreted exosomal microRNA-210 promotes angiogenesis in vitro and in vivo. Mol. Ther. Nucleic Acids 2018, 11, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhang, X.-F.; Lu, X.; Jia, H.-L.; Liang, L.; Dong, Q.-Z.; Ye, Q.-H.; Qin, L.-X. MicroRNA-26a Suppresses Angiogenesis in Human Hepatocellular Carcinoma by Targeting Hepatocyte Growth Factor-cMet Pathway. Hepatology 2014, 59, 1874–1885. [Google Scholar] [CrossRef]
- Li, D.; Wang, T.; Sun, F.-F.; Feng, J.-Q.; Peng, J.-J.; Li, H.; Wang, C.; Wang, D.; Liu, Y.; Bai, Y.-D. MicroRNA-375 represses tumor angiogenesis and reverses resistance to sorafenib in hepatocarcinoma. Cancer Gene Ther. 2021, 28, 126–140. [Google Scholar] [CrossRef]
- Liu, Y.; Tang, H.; Zhang, Y.; Wang, Q.; Li, S.; Wang, Z.; Shi, X. Circular RNA hsa_circ_0000519 contributes to angiogenesis and tumor progression in hepatocellular carcinoma through the miR-1296/E2F7 axis. Hum. Cell 2023, 36, 738–751. [Google Scholar] [CrossRef]
- Yang, H.D.; Kim, H.S.; Kim, S.Y.; Na, M.J.; Yang, G.; Eun, J.W.; Wang, H.J.; Cheong, J.Y.; Park, W.S.; Nam, S.W. HDAC6 suppresses Let-7i-5p to elicit TSP1/CD47-mediated anti-tumorigenesis and phagocytosis of hepatocellular carcinoma. Hepatology 2019, 70, 1262–1279. [Google Scholar] [CrossRef] [PubMed]
- Özkan, A.; Stolley, D.L.; Cressman, E.N.K.; McMillin, M.; DeMorrow, S.; Yankeelov, T.E.; Rylander, M.N. Tumor Microenvironment Alters Chemoresistance of Hepatocellular Carcinoma Through CYP3A4 Metabolic Activity. Front. Oncol. 2021, 11, 662135. [Google Scholar] [CrossRef]
- Duan, B.; Huang, C.; Bai, J.; Zhang, Y.L.; Wang, X.; Yang, J.; Li, J. Multidrug Resistance in Hepatocellular Carcinoma. In Hepatocellular Carcinoma; Tirnitz-Parker, J.E.E., Ed.; Codon Publications: Brisbane, Australia, 2019. [Google Scholar] [CrossRef]
- Liang, Y.; Liang, Q.; Qiao, L.; Xiao, F. MicroRNAs modulate drug resistance-related mechanisms in hepatocellular carcinoma. Front. Oncol. 2020, 10, 920. [Google Scholar] [CrossRef]
- Wei, X.; Zhao, L.; Ren, R.; Ji, F.; Xue, S.; Zhang, J.; Liu, Z.; Ma, Z.; Wang, X.W.; Wong, L. MiR-125b Loss Activated HIF1α/pAKT Loop, Leading to Transarterial Chemoembolization Resistance in Hepatocellular Carcinoma. Hepatology 2021, 73, 1381–1398. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Chen, E.; Tang, Y.; Mao, J.; Shen, J.; Zheng, X.; Xie, S.; Zhang, S.; Wu, Y.; Liu, H. miR-223 overexpression inhibits doxorubicin-induced autophagy by targeting FOXO3a and reverses chemoresistance in hepatocellular carcinoma cells. Cell Death Dis. 2019, 10, 843. [Google Scholar] [CrossRef] [PubMed]
- Yuan, P.; Cao, W.; Zang, Q.; Li, G.; Guo, X.; Fan, J. The HIF-2α-MALAT1-miR-216b axis regulates multi-drug resistance of hepatocellular carcinoma cells via modulating autophagy. Biochem. Biophys. Res. Commun. 2016, 478, 1067–1073. [Google Scholar] [CrossRef] [PubMed]
- Ren, Q.; Xiao, X.; Leng, X.; Zhang, Q.; Zhou, X.; Ren, Z.; Xiao, H. MicroRNA-361-5p induces hepatocellular carcinoma cell apoptosis and enhances drug sensitivity by targeting MAP3K9. Exp. Ther. Med. 2021, 21, 574. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wang, H.; Gao, W. MiRNA-610 acts as a tumour suppressor to depress the cisplatin resistance in hepatocellular carcinoma through targeted silencing of hepatoma-derived growth factor. Arch. Med. Sci. AMS 2020, 16, 1394. [Google Scholar] [CrossRef] [PubMed]
- Forouzanfar, M.; Lachinani, L.; Dormiani, K.; Nasr-Esfahani, M.H.; Gure, A.O.; Ghaedi, K. Intracellular functions of RNA-binding protein, Musashi1, in stem and cancer cells. Stem Cell Res. Ther. 2020, 11, 193. [Google Scholar] [CrossRef]
- Gao, J.; Dai, C.; Yu, X.; Yin, X.B.; Zhou, F. Long noncoding RNA LEF1-AS1 acts as a microRNA-10a-5p regulator to enhance MSI1 expression and promote chemoresistance in hepatocellular carcinoma cells through activating AKT signaling pathway. J. Cell. Biochem. 2021, 122, 86–99. [Google Scholar] [CrossRef]
- Ignatius, M.S.; Hayes, M.N.; Lobbardi, R.; Chen, E.Y.; McCarthy, K.M.; Sreenivas, P.; Motala, Z.; Durbin, A.D.; Molodtsov, A.; Reeder, S.; et al. The NOTCH1/SNAIL1/MEF2C Pathway Regulates Growth and Self-Renewal in Embryonal Rhabdomyosarcoma. Cell Rep. 2017, 19, 2304–2318. [Google Scholar] [CrossRef]
- Li, H.; Radford, J.C.; Ragusa, M.J.; Shea, K.L.; McKercher, S.R.; Zaremba, J.D.; Soussou, W.; Nie, Z.; Kang, Y.J.; Nakanishi, N.; et al. Transcription factor MEF2C influences neural stem/progenitor cell differentiation and maturation in vivo. Proc. Natl. Acad. Sci. USA 2008, 105, 9397–9402. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.; Kim, C.; Ji, E.; Ahn, S.; Jung, M.; Hong, Y.; Kim, W.; Lee, E.K. The MicroRNA-551a/MEF2C axis regulates the survival and sphere formation of cancer cells in response to 5-fluorouracil. Mol. Cells 2019, 42, 175. [Google Scholar]
- Qin, J.; Luo, M.; Qian, H.; Chen, W. Upregulated miR-182 increases drug resistance in cisplatin-treated HCC cell by regulating TP53INP1. Gene 2014, 538, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.-P.; Liao, J.-Z.; Lu, Y.-Q.; Tian, D.-A.; Ye, F.; Zhao, P.-X.; Xiang, G.-Y.; Tang, W.-X.; He, X.-X. MiR-375 and doxorubicin co-delivered by liposomes for combination therapy of hepatocellular carcinoma. Mol. Ther. Nucleic Acids 2017, 7, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Zheng, Z.-M.; Li, X.-N.; Li, Z.-F.; Wang, Y.; Geng, Y.-F.; Bai, L.; Zhang, X.-B. MiR-223 modulates multidrug resistance via downregulation of ABCB1 in hepatocellular carcinoma cells. Exp. Biol. Med. 2013, 238, 1024–1032. [Google Scholar] [CrossRef] [PubMed]
- Tu, C.; Chen, W.; Wang, S.; Tan, W.; Guo, J.; Shao, C.; Wang, W. MicroRNA-383 inhibits doxorubicin resistance in hepatocellular carcinoma by targeting eukaryotic translation initiation factor 5A2. J. Cell. Mol. Med. 2019, 23, 7190–7199. [Google Scholar] [CrossRef]
- Gao, X.; Jiang, Y.; Li, Y. Inhibitory effect of miR-140-5p on doxorubicin resistance of hepatocellular carcinoma. Exp. Ther. Med. 2021, 21, 507. [Google Scholar] [CrossRef]
- Chen, M.; Wu, L.; Tu, J.; Zhao, Z.; Fan, X.; Mao, J.; Weng, Q.; Wu, X.; Huang, L.; Xu, M. miR-590-5p suppresses hepatocellular carcinoma chemoresistance by targeting YAP1 expression. EBioMedicine 2018, 35, 142–154. [Google Scholar] [CrossRef]
- Yahya, S.M.M.; Fathy, S.A.; El-Khayat, Z.A.; El-Toukhy, S.E.; Hamed, A.R.; Hegazy, M.G.A.; Nabih, H.K. Possible Role of microRNA-122 in Modulating Multidrug Resistance of Hepatocellular Carcinoma. Indian J. Clin. Biochem. 2018, 33, 21–30. [Google Scholar] [CrossRef]
- Pan, C.; Wang, X.; Shi, K.; Zheng, Y.; Li, J.; Chen, Y.; Jin, L.; Pan, Z. MiR-122 Reverses the Doxorubicin-Resistance in Hepatocellular Carcinoma Cells through Regulating the Tumor Metabolism. PLoS ONE 2016, 11, e0152090. [Google Scholar] [CrossRef]
- Tian, T.; Fu, X.; Lu, J.; Ruan, Z.; Nan, K.; Yao, Y.; Yang, Y. MicroRNA-760 inhibits doxorubicin resistance in hepatocellular carcinoma through regulating Notch1/Hes1-PTEN/Akt signaling pathway. J. Biochem. Mol. Toxicol. 2018, 32, e22167. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; He, J.; Ren, X.; Zhao, H.; Zhao, H. Circ_0003998 enhances doxorubicin resistance in hepatocellular carcinoma by regulating miR-218-5p/EIF5A2 pathway. Diagn. Pathol. 2020, 15, 141. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Ma, T.; Huang, C.; Zhang, L.; Lv, X.; Xu, T.; Hu, T.; Li, J. MiR-27a modulates the MDR1/P-glycoprotein expression by inhibiting FZD7/β-catenin pathway in hepatocellular carcinoma cells. Cell Signal. 2013, 25, 2693–2701. [Google Scholar] [CrossRef]
- Qin, Y.-F.; Zhou, Z.-Y.; Fu, H.-W.; Lin, H.-M.; Xu, L.-B.; Wu, W.-R.; Liu, C.; Xu, X.-L.; Zhang, R. Hepatitis B Virus Surface Antigen Promotes Stemness of Hepatocellular Carcinoma through Regulating MicroRNA-203a. J. Clin. Transl. Hepatol. 2023, 11, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.-X.; Gao, S.; Pan, Y.-Z.; Yu, C.; Sun, C.-Y. Overexpression of microRNA-125b sensitizes human hepatocellular carcinoma cells to 5-fluorouracil through inhibition of glycolysis by targeting hexokinase II. Mol. Med. Rep. 2014, 10, 995–1002. [Google Scholar] [CrossRef]
- Zheng, R.-P.; Ma, D.-K.; Li, Z.; Zhang, H.-F. MiR-145 Regulates the Chemoresistance of Hepatic Carcinoma Cells Against 5-Fluorouracil by Targeting Toll-Like Receptor 4. Cancer Manag. Res. 2020, 12, 6165–6175. [Google Scholar] [CrossRef] [PubMed]
- Bai, B.; Liu, Y.; Fu, X.-M.; Qing, H.-Y.; Li, G.-K.; Wang, H.-C.; Sun, S.-L. Dysregulation of EZH2/miR-138-5p Axis Contributes to Radiosensitivity in Hepatocellular Carcinoma Cell by Downregulating Hypoxia-Inducible Factor 1 Alpha (HIF-1α). Oxidative Med. Cell. Longev. 2022, 2022, 7608712. [Google Scholar] [CrossRef]
- Fu, X.; Liu, M.; Qu, S.; Ma, J.; Zhang, Y.; Shi, T.; Wen, H.; Yang, Y.; Wang, S.; Wang, J. Exosomal microRNA-32-5p induces multidrug resistance in hepatocellular carcinoma via the PI3K/Akt pathway. J. Exp. Clin. Cancer Res. 2018, 37, 52. [Google Scholar] [CrossRef]
- Jin, X.; Cai, L.; Wang, C.; Deng, X.; Yi, S.; Lei, Z.; Xiao, Q.; Xu, H.; Luo, H.; Sun, J. CASC2/miR-24/miR-221 modulates the TRAIL resistance of hepatocellular carcinoma cell through caspase-8/caspase-3. Cell Death Dis. 2018, 9, 318. [Google Scholar] [CrossRef]
- Simpson, D.; Keating, G.M. Sorafenib. Drugs 2008, 68, 251–258. [Google Scholar] [CrossRef]
- Berretta, M.; Rinaldi, L.; Di Benedetto, F.; Lleshi, A.; De Re, V.; Facchini, G.; De Paoli, P.; Di Francia, R. Angiogenesis Inhibitors for the Treatment of Hepatocellular Carcinoma. Front. Pharmacol. 2016, 7, 428. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Montal, R.; Sia, D.; Finn, R.S. Molecular therapies and precision medicine for hepatocellular carcinoma. Nat. Rev. Clin. Oncol. 2018, 15, 599–616. [Google Scholar] [CrossRef]
- Nair, A.; Reece, K.; Donoghue, M.B.; Yuan, W.V.; Rodriguez, L.; Keegan, P.; Pazdur, R. FDA Supplemental Approval Summary: Lenvatinib for the Treatment of Unresectable Hepatocellular Carcinoma. Oncologist 2021, 26, e484–e491. [Google Scholar] [CrossRef]
- Capozzi, M.; De Divitiis, C.; Ottaiano, A.; von Arx, C.; Scala, S.; Tatangelo, F.; Delrio, P.; Tafuto, S. Lenvatinib, a molecule with versatile application: From preclinical evidence to future development in anti-cancer treatment. Cancer Manag. Res. 2019, 11, 3847–3860. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Zhu, H.; Shen, Y.; Zhang, X.; He, X.; Xu, X. The role of non-coding RNAs in the sorafenib resistance of hepatocellular carcinoma. Front. Oncol. 2021, 11, 696705. [Google Scholar] [CrossRef] [PubMed]
- Kabir, T.D.; Ganda, C.; Brown, R.M.; Beveridge, D.J.; Richardson, K.L.; Chaturvedi, V.; Candy, P.; Epis, M.; Wintle, L.; Kalinowski, F.; et al. A microRNA-7/growth arrest specific 6/TYRO3 axis regulates the growth and invasiveness of sorafenib-resistant cells in human hepatocellular carcinoma. Hepatology 2018, 67, 216–231. [Google Scholar] [CrossRef]
- Reinkens, T.; Stalke, A.; Huge, N.; Vajen, B.; Eilers, M.; Schäffer, V.; Dittrich-Breiholz, O.; Schlegelberger, B.; Illig, T.; Skawran, B. Ago-RIP Sequencing Identifies New MicroRNA-449a-5p Target Genes Increasing Sorafenib Efficacy in Hepatocellular Carcinoma. J. Cancer 2022, 13, 62. [Google Scholar] [CrossRef]
- He, X.; Sun, H.; Jiang, Q.; Chai, Y.; Li, X.; Wang, Z.; Zhu, B.; You, S.; Li, B.; Hao, J. Hsa-miR-4277 Decelerates the Metabolism or Clearance of Sorafenib in HCC Cells and Enhances the Sensitivity of HCC Cells to Sorafenib by Targeting Cyp3a4. Front. Oncol. 2021, 11, 735447. [Google Scholar] [CrossRef]
- Sun, T.; Liu, H.; Ming, L. Multiple Roles of Autophagy in the Sorafenib Resistance of Hepatocellular Carcinoma. Cell. Physiol. Biochem. 2017, 44, 716–727. [Google Scholar] [CrossRef]
- Chen, M.-Y.; Yadav, V.K.; Chu, Y.C.; Ong, J.R.; Huang, T.-Y.; Lee, K.-F.; Lee, K.-H.; Yeh, C.-T.; Lee, W.-H. Hydroxychloroquine (HCQ) modulates autophagy and oxidative DNA damage stress in hepatocellular carcinoma to overcome sorafenib resistance via TLR9/SOD1/hsa-miR-30a-5p/Beclin-1 axis. Cancers 2021, 13, 3227. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Tan, X.; Luo, J.; Yao, H.; Si, Z.; Tong, J.-S. The miR-30a-5p/CLCF1 axis regulates sorafenib resistance and aerobic glycolysis in hepatocellular carcinoma. Cell Death Dis. 2020, 11, 902. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhou, Y.; Yang, L.; Ma, Y.; Peng, X.; Yang, S.; Li, H.; Liu, J. LncRNA NEAT1 promotes autophagy via regulating miR-204/ATG3 and enhanced cell resistance to sorafenib in hepatocellular carcinoma. J. Cell. Physiol. 2020, 235, 3402–3413. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Chen, J.; Zhou, H.; Chen, Y.; Zhi, Y.; Zhang, B.; Chen, L.; Chu, X.; Wang, R.; Zhang, C. PU. 1/microRNA-142-3p targets ATG5/ATG16L1 to inactivate autophagy and sensitize hepatocellular carcinoma cells to sorafenib. Cell Death Dis. 2018, 9, 312. [Google Scholar] [CrossRef]
- Wang, D.; Yang, J. MiR-375 attenuates sorafenib resistance of hepatocellular carcinoma cells by inhibiting cell autophagy. Acta Biochim. Pol. 2023, 70, 239–246. [Google Scholar] [CrossRef]
- Lin, Z.; Xia, S.; Liang, Y.; Ji, L.; Pan, Y.; Jiang, S.; Wan, Z.; Tao, L.; Chen, J.; Lin, C. LXR activation potentiates sorafenib sensitivity in HCC by activating microRNA-378a transcription. Theranostics 2020, 10, 8834. [Google Scholar] [CrossRef]
- Kliewer, S.A. Nuclear receptor PXR: Discovery of a pharmaceutical anti-target. J. Clin. Investig. 2015, 125, 1388–1389. [Google Scholar] [CrossRef]
- Feng, F.; Jiang, Q.; Cao, S.; Cao, Y.; Li, R.; Shen, L.; Zhu, H.; Wang, T.; Sun, L.; Liang, E. Pregnane X receptor mediates sorafenib resistance in advanced hepatocellular carcinoma. Biochim. Biophys. Acta BBA Gen. Subj. 2018, 1862, 1017–1030. [Google Scholar] [CrossRef]
- Li, J.; Zhao, J.; Wang, H.; Li, X.; Liu, A.; Qin, Q.; Li, B. MicroRNA-140-3p enhances the sensitivity of hepatocellular carcinoma cells to sorafenib by targeting pregnenolone X receptor. OncoTargets Ther. 2018, 11, 5885. [Google Scholar] [CrossRef]
- Li, B.; Feng, F.; Jia, H.; Jiang, Q.; Cao, S.; Wei, L.; Zhang, Y.; Lu, J. Rhamnetin decelerates the elimination and enhances the antitumor effect of the molecular-targeting agent sorafenib in hepatocellular carcinoma cells via the miR-148a/PXR axis. Food Funct. 2021, 12, 2404–2417. [Google Scholar] [CrossRef]
- Dong, Z.-B.; Wu, H.-M.; He, Y.-C.; Huang, Z.-T.; Weng, Y.-H.; Li, H.; Liang, C.; Yu, W.-M.; Chen, W. MiRNA-124-3p. 1 sensitizes hepatocellular carcinoma cells to sorafenib by regulating FOXO3a by targeting AKT2 and SIRT1. Cell Death Dis. 2022, 13, 35. [Google Scholar] [CrossRef]
- Li, T.-T.; Mou, J.; Pan, Y.-J.; Huo, F.-C.; Du, W.-Q.; Liang, J.; Wang, Y.; Zhang, L.-S.; Pei, D.-S. MicroRNA-138-1-3p sensitizes sorafenib to hepatocellular carcinoma by targeting PAK5 mediated β-catenin/ABCB1 signaling pathway. J. Biomed. Sci. 2021, 28, 56. [Google Scholar] [CrossRef] [PubMed]
- Bozkulak, E.C.; Weinmaster, G. Selective use of ADAM10 and ADAM17 in activation of Notch1 signaling. Mol. Cell. Biol. 2009, 29, 5679–5695. [Google Scholar] [CrossRef] [PubMed]
- Ruan, Y.; Chen, T.; Zheng, L.; Cai, J.; Zhao, H.; Wang, Y.; Tao, L.; Xu, J.; Ji, L.; Cai, X. cDCBLD2 mediates sorafenib resistance in hepatocellular carcinoma by sponging miR-345-5p binding to the TOP2A coding sequence. Int. J. Biol. Sci. 2023, 19, 4608–4626. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Chan, Y.-T.; Wu, J.; Feng, Z.; Yuan, H.; Li, Q.; Xing, T.; Xu, L.; Zhang, C.; Tan, H.-Y.; et al. CRISPR/Cas9 screens unravel miR-3689a-3p regulating sorafenib resistance in hepatocellular carcinoma via suppressing CCS/SOD1-dependent mitochondrial oxidative stress. Drug Resist. Updat. 2023, 71, 318. [Google Scholar] [CrossRef]
- Lu, Y.; Chan, Y.-T.; Tan, H.-Y.; Zhang, C.; Guo, W.; Xu, Y.; Sharma, R.; Chen, Z.-S.; Zheng, Y.-C.; Wang, N. Epigenetic regulation of ferroptosis via ETS1/miR-23a-3p/ACSL4 axis mediates sorafenib resistance in human hepatocellular carcinoma. J. Exp. Clin. Cancer Res. 2022, 41, 3. [Google Scholar] [CrossRef] [PubMed]
- Doll, S.; Proneth, B.; Tyurina, Y.Y.; Panzilius, E.; Kobayashi, S.; Ingold, I.; Irmler, M.; Beckers, J.; Aichler, M.; Walch, A.; et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition. Nat. Chem. Biol. 2017, 13, 91–98. [Google Scholar] [CrossRef]
- Hirao, A.; Sato, Y.; Tanaka, H.; Nishida, K.; Tomonari, T.; Hirata, M.; Bando, M.; Kida, Y.; Tanaka, T.; Kawaguchi, T. MiR-125b-5p Is Involved in Sorafenib Resistance through Ataxin-1-Mediated Epithelial-Mesenchymal Transition in Hepatocellular Carcinoma. Cancers 2021, 13, 4917. [Google Scholar] [CrossRef]
- Bergamini, C.; Leoni, I.; Rizzardi, N.; Melli, M.; Galvani, G.; Coada, C.A.; Giovannini, C.; Monti, E.; Liparulo, I.; Valenti, F.; et al. MiR-494 induces metabolic changes through G6pc targeting and modulates sorafenib response in hepatocellular carcinoma. J. Exp. Clin. Cancer Res. 2023, 42, 145. [Google Scholar] [CrossRef]
- Hu, Z.; Zhao, Y.; Mang, Y.; Zhu, J.; Yu, L.; Li, L.; Ran, J. MiR-21-5p promotes sorafenib resistance and hepatocellular carcinoma progression by regulating SIRT7 ubiquitination through USP24. Life Sci. 2023, 15, 121773. [Google Scholar] [CrossRef]
- Yu, T.; Yu, J.; Lu, L.; Zhang, Y.; Zhou, Y.; Zhou, Y.; Huang, F.; Sun, L.; Guo, Z.; Hou, G.; et al. MT1JP-mediated miR-24-3p/BCL2L2 axis promotes Lenvatinib resistance in hepatocellular carcinoma cells by inhibiting apoptosis. Cell. Oncol. 2021, 44, 821–834. [Google Scholar] [CrossRef]
- Gu, D.; Tong, M.; Wang, J.; Zhang, B.; Liu, J.; Song, G.; Zhu, B. Overexpression of the lncRNA HOTAIRM1 promotes lenvatinib resistance by downregulating miR-34a and activating autophagy in hepatocellular carcinoma. Discov. Oncol. 2023, 14, 66. [Google Scholar] [CrossRef] [PubMed]
- Han, T.; Zheng, H.; Zhang, J.; Yang, P.; Li, H.; Cheng, Z.; Xiang, D.; Wang, R. Downregulation of MUC15 by miR-183-5p.1 promotes liver tumor-initiating cells properties and tumorigenesis via regulating c-MET/PI3K/AKT/SOX2 axis. Cell Death Dis. 2022, 13, 200. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Liu, W.; Chen, X.; Yin, J.; Ma, L.; Liu, M.; Zhou, X.; Xian, L.; Li, P.; Tan, X.; et al. circKCNN2 suppresses the recurrence of hepatocellular carcinoma at least partially via regulating miR-520c-3p/methyl-DNA-binding domain protein 2 axis. Clin. Transl. Med. 2022, 12, e662. [Google Scholar] [CrossRef]
- Wei, Y.; Wei, L.; Han, T.; Ding, S. miR-3154 promotes hepatocellular carcinoma progression via suppressing HNF4α. Carcinogenesis 2022, 43, 1002–1014. [Google Scholar] [CrossRef]
- Wang, G.; Zhao, W.; Wang, H.; Qiu, G.; Jiang, Z.; Wei, G.; Li, X. Exosomal MiR-744 inhibits proliferation and sorafenib chemoresistance in hepatocellular carcinoma by targeting PAX2. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2019, 25, 7209. [Google Scholar] [CrossRef]
- Feng, Y.; Jiang, W.; Zhao, W.; Lu, Z.; Gu, Y.; Dong, Y. miR-124 regulates liver cancer stem cells expansion and sorafenib resistance. Exp. Cell Res. 2020, 394, 112162. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Sun, W.; Liu, J.; Pu, H.; Li, Y. Circ_RBM23 knockdown suppresses chemoresistance, proliferation, migration and invasion of sorafenib-resistant HCC cells through miR-338–3p/RAB1B axis. Pathol. Res. Pract. 2023, 245, 154435. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Huang, J.; Ma, L.; Shan, J.; Shen, J.; Yang, Z.; Liu, L.; Luo, Y.; Yao, C.; Qian, C. MicroRNA-122 confers sorafenib resistance to hepatocellular carcinoma cells by targeting IGF-1R to regulate RAS/RAF/ERK signaling pathways. Cancer Lett. 2016, 371, 171–181. [Google Scholar] [CrossRef]
- Turato, C.; Fornari, F.; Pollutri, D.; Fassan, M.; Quarta, S.; Villano, G.; Ruvoletto, M.; Bolondi, L.; Gramantieri, L.; Pontisso, P. MiR-122 targets SerpinB3 and is involved in sorafenib resistance in hepatocellular carcinoma. J. Clin. Med. 2019, 8, 171. [Google Scholar] [CrossRef]
- Chen, B.W.; Zhou, Y.; Wei, T.; Wen, L.; Zhang, Y.b.; Shen, S.c.; Zhang, J.; Ma, T.; Chen, W.; Ni, L. lncRNA-POIR promotes epithelial–mesenchymal transition and suppresses sorafenib sensitivity simultaneously in hepatocellular carcinoma by sponging miR-182-5p. J. Cell. Biochem. 2021, 122, 130–142. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.F.; Wang, F.; Wu, J.; Wu, Y.; Huang, W.; Liu, D.; Huang, X.Y.; Zhang, X.M.; Ke, A.W. LncRNA SNHG3 induces EMT and sorafenib resistance by modulating the miR-128/CD151 pathway in hepatocellular carcinoma. J. Cell. Physiol. 2019, 234, 2788–2794. [Google Scholar] [CrossRef]
- Qin, X.; Wang, S. LncASAP1-IT1 promotes hepatocellular carcinoma progression through the regulation of the miR-1294/TGFBR1 pathway in vitro and in vivo. J. Gastrointest. Oncol. 2023, 14, 1451–1461. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Huang, Y.; Dai, T.; Hua, Z.; Xu, J.; Lin, Y.; Han, L.; Yue, X.; Ho, L.; Lu, J. LncRNA TTN-AS1 intensifies sorafenib resistance in hepatocellular carcinoma by sponging miR-16-5p and upregulation of cyclin E1. Biomed. Pharmacother. 2021, 133, 111030. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, H.; Hong, H.; Zhang, Z. MiR-374b re-sensitizes hepatocellular carcinoma cells to sorafenib therapy by antagonizing PKM2 mediated glycolysis pathway. Am. J. Cancer Res. 2019, 9, 765–778. [Google Scholar]
- Yang, S.; Wang, M.; Yang, L.; Li, Y.; Ma, Y.; Peng, X.; Li, X.; Li, B.; Jin, H.; Li, H. MicroRNA-375 Targets ATG14 to Inhibit Autophagy and Sensitize Hepatocellular Carcinoma Cells to Sorafenib. OncoTargets Ther. 2020, 13, 3557–3570. [Google Scholar] [CrossRef]
- Ji, L.; Lin, Z.; Wan, Z.; Xia, S.; Jiang, S.; Cen, D.; Cai, L.; Xu, J.; Cai, X. miR-486-3p mediates hepatocellular carcinoma sorafenib resistance by targeting FGFR4 and EGFR. Cell Death Dis. 2020, 11, 250. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Wang, C.; Xie, H.; Wang, Y.; Huang, J.; Rong, Y.; Zhang, H.; Kong, H.; Yang, Y.; Lu, Y. MicroRNA-3163 targets ADAM-17 and enhances the sensitivity of hepatocellular carcinoma cells to molecular targeted agents. Cell Death Dis. 2019, 10, 784. [Google Scholar] [CrossRef] [PubMed]
- Niture, S.; Gadi, S.; Qi, Q.; Gyamfi, M.A.; Varghese, R.S.; Rios-Colon, L.; Chimeh, U.; Vandana; Ressom, H.W.; Kumar, D. MicroRNA-483-5p Inhibits Hepatocellular Carcinoma Cell Proliferation, Cell Steatosis, and Fibrosis by Targeting PPARα and TIMP2. Cancers 2023, 15, 1715. [Google Scholar] [CrossRef]
- Song, M.; Ma, L.; Shen, C.; Liu, W.; Zhang, P.; Bi, R.; Zhao, C. FGD5-AS1/miR-5590-3p/PINK1 induces Lenvatinib resistance in hepatocellular carcinoma. Cell. Signal. 2023, 111, 110828. [Google Scholar] [CrossRef]
- Tan, W.; Lin, Z.; Chen, X.; Li, W.; Zhu, S.; Wei, Y.; Huo, L.; Chen, Y.; Shang, C. miR-126-3p contributes to sorafenib resistance in hepatocellular carcinoma via downregulating SPRED1. Ann. Transl. Med. 2021, 9, 38. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Zhang, J.; Li, H.; Peng, H.; Gu, M.; Wang, H. miR-96 regulates liver tumor-initiating cells expansion by targeting TP53INP1 and predicts Sorafenib resistance. J. Cancer 2020, 11, 6545–6555. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Zhou, J.; Cheng, F.; Li, H.; Quan, Y. MiR-3677-3p promotes development and sorafenib resistance of hepatitis B-related hepatocellular carcinoma by inhibiting FOXM1 ubiquitination. Hum. Cell 2023, 36, 1773–1789. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Yang, W.; Shu, Z.; Shen, X.; Zhang, W.; Cen, C.; Cao, L.; Zhang, M.; Zheng, S.; Yu, J. MicroRNA-223 promotes hepatocellular carcinoma cell resistance to sorafenib by targeting FBW7. Oncol. Rep. 2019, 41, 1231–1237. [Google Scholar] [CrossRef]
- Elhefnawi, M.; Salah, Z.; Soliman, B. The promise of miRNA replacement therapy for hepatocellular carcinoma. Curr. Gene Ther. 2019, 19, 290–304. [Google Scholar] [CrossRef] [PubMed]
- Moshiri, F.; Callegari, E.; D’Abundo, L.; Corrà, F.; Lupini, L.; Sabbioni, S.; Negrini, M. Inhibiting the oncogenic mir-221 by microRNA sponge: Toward microRNA-based therapeutics for hepatocellular carcinoma. Gastroenterol. Hepatol. Bed Bench 2014, 7, 43. [Google Scholar]
- Kim, T.; Croce, C.M. MicroRNA: Trends in clinical trials of cancer diagnosis and therapy strategies. Exp. Mol. Med. 2023, 55, 1314–1321. [Google Scholar] [CrossRef]
- Reda El Sayed, S.; Cristante, J.; Guyon, L.; Denis, J.; Chabre, O.; Cherradi, N. MicroRNA Therapeutics in Cancer: Current Advances and Challenges. Cancers 2021, 13, 2680. [Google Scholar] [CrossRef]
- Kalfert, D.; Ludvikova, M.; Pesta, M.; Ludvik, J.; Dostalova, L.; Kholová, I. Multifunctional Roles of miR-34a in Cancer: A Review with the Emphasis on Head and Neck Squamous Cell Carcinoma and Thyroid Cancer with Clinical Implications. Diagnostics 2020, 10, 563. [Google Scholar] [CrossRef]
- Bader, A.G. miR-34—A microRNA replacement therapy is headed to the clinic. Front. Genet. 2012, 3, 120. [Google Scholar] [CrossRef]
- Beg, M.S.; Brenner, A.J.; Sachdev, J.; Borad, M.; Kang, Y.K.; Stoudemire, J.; Smith, S.; Bader, A.G.; Kim, S.; Hong, D.S. Phase I study of MRX34, a liposomal miR-34a mimic, administered twice weekly in patients with advanced solid tumors. Investig. New Drugs 2017, 35, 180–188. [Google Scholar] [CrossRef]
- Jivrajani, M.; Nivsarkar, M. Ligand-targeted bacterial minicells: Futuristic nano-sized drug delivery system for the efficient and cost effective delivery of shRNA to cancer cells. Nanomedicine 2016, 12, 2485–2498. [Google Scholar] [CrossRef] [PubMed]
- Khordadmehr, M.; Shahbazi, R.; Sadreddini, S.; Baradaran, B. miR-193: A new weapon against cancer. J. Cell. Physiol. 2019, 234, 16861–16872. [Google Scholar] [CrossRef] [PubMed]
- Telford, B.J.; Yahyanejad, S.; de Gunst, T.; den Boer, H.C.; Vos, R.M.; Stegink, M.; van den Bosch, M.T.; Alemdehy, M.F.; van Pinxteren, L.A.; Schaapveld, R.Q. Multi-modal effects of 1B3, a novel synthetic miR-193a-3p mimic, support strong potential for therapeutic intervention in oncology. Oncotarget 2021, 12, 422. [Google Scholar] [CrossRef] [PubMed]
- van den Bosch, M.T.; Yahyanejad, S.; Alemdehy, M.F.; Telford, B.J.; de Gunst, T.; den Boer, H.C.; Vos, R.M.; Stegink, M.; van Pinxteren, L.A.; Schaapveld, R.Q. Transcriptome-wide analysis reveals insight into tumor suppressor functions of 1B3, a novel synthetic miR-193a-3p mimic. Mol. Ther. Nucleic Acids 2021, 23, 1161–1171. [Google Scholar] [CrossRef]
- Hsu, S.H.; Wang, B.; Kota, J.; Yu, J.; Costinean, S.; Kutay, H.; Yu, L.; Bai, S.; La Perle, K.; Chivukula, R.R.; et al. Essential metabolic, anti-inflammatory, and anti-tumorigenic functions of miR-122 in liver. J. Clin. Investig. 2012, 122, 2871–2883. [Google Scholar] [CrossRef]
- Bai, S.; Nasser, M.W.; Wang, B.; Hsu, S.H.; Datta, J.; Kutay, H.; Yadav, A.; Nuovo, G.; Kumar, P.; Ghoshal, K. MicroRNA-122 inhibits tumorigenic properties of hepatocellular carcinoma cells and sensitizes these cells to sorafenib. J. Biol. Chem. 2009, 284, 32015–32027. [Google Scholar] [CrossRef] [PubMed]
- Tsai, W.C.; Hsu, P.W.; Lai, T.C.; Chau, G.Y.; Lin, C.W.; Chen, C.M.; Lin, C.D.; Liao, Y.L.; Wang, J.L.; Chau, Y.P.; et al. MicroRNA-122, a tumor suppressor microRNA that regulates intrahepatic metastasis of hepatocellular carcinoma. Hepatology 2009, 49, 1571–1582. [Google Scholar] [CrossRef]
- Hsu, S.H.; Yu, B.; Wang, X.; Lu, Y.; Schmidt, C.R.; Lee, R.J.; Lee, L.J.; Jacob, S.T.; Ghoshal, K. Cationic lipid nanoparticles for therapeutic delivery of siRNA and miRNA to murine liver tumor. Nanomedicine 2013, 9, 1169–1180. [Google Scholar] [CrossRef]
- Kota, J.; Chivukula, R.R.; O’Donnell, K.A.; Wentzel, E.A.; Montgomery, C.L.; Hwang, H.W.; Chang, T.C.; Vivekanandan, P.; Torbenson, M.; Clark, K.R.; et al. Therapeutic microRNA delivery suppresses tumorigenesis in a murine liver cancer model. Cell 2009, 137, 1005–1017. [Google Scholar] [CrossRef]
- Querfeld, C.; Foss, F.M.; Kim, Y.H.; Pinter-Brown, L.; William, B.M.; Porcu, P.; Pacheco, T.; Haverkos, B.M.; DeSimone, J.; Guitart, J.; et al. Phase 1 Trial of Cobomarsen, an Inhibitor of Mir-155, in Cutaneous T Cell Lymphoma. Blood 2018, 132, 2903. [Google Scholar] [CrossRef]
- James, A.; Ruckman, J.; Pestano, L.; Hopkins, R.; Rodgers, R.; Marshall, W.; Rubin, P.; Escolar, D. SOLAR: A phase 2, global, randomized, active comparator study to investigate the efficacy and safety of cobomarsen in subjects with mycosis fungoides (MF). Hematol. Oncol. 2019, 37, 562–563. [Google Scholar] [CrossRef]
- Dhuri, K.; Pradeep, S.P.; Shi, J.; Anastasiadou, E.; Slack, F.J.; Gupta, A.; Zhong, X.B.; Bahal, R. Simultaneous Targeting of Multiple oncomiRs with Phosphorothioate or PNA-Based Anti-miRs in Lymphoma Cell Lines. Pharm. Res. 2022, 39, 2709–2720. [Google Scholar] [CrossRef]
- Zhang, M.; Zhou, X.; Wang, B.; Yung, B.C.; Lee, L.J.; Ghoshal, K.; Lee, R.J. Lactosylated gramicidin-based lipid nanoparticles (Lac-GLN) for targeted delivery of anti-miR-155 to hepatocellular carcinoma. J. Control Release 2013, 168, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Shao, S.; Hu, Q.; Wu, W.; Wang, M.; Huang, J.; Zhao, X.; Tang, G.; Liang, T. Tumor-triggered personalized microRNA cocktail therapy for hepatocellular carcinoma. Biomater. Sci. 2020, 8, 6579–6591. [Google Scholar] [CrossRef] [PubMed]
- Sheedy, P.; Medarova, Z. The fundamental role of miR-10b in metastatic cancer. Am. J. Cancer Res. 2018, 8, 1674–1688. [Google Scholar] [PubMed]
- Liang, G.; Zhu, Y.; Ali, D.J.; Tian, T.; Xu, H.; Si, K.; Sun, B.; Chen, B.; Xiao, Z. Engineered exosomes for targeted co-delivery of miR-21 inhibitor and chemotherapeutics to reverse drug resistance in colon cancer. J. Nanobiotechnol. 2020, 18, 10. [Google Scholar] [CrossRef]
- Iacomino, G.; Siani, A. Role of microRNAs in obesity and obesity-related diseases. Genes Nutr. 2017, 12, 23. [Google Scholar] [CrossRef]
- Gebert, L.F.; Rebhan, M.A.; Crivelli, S.E.; Denzler, R.; Stoffel, M.; Hall, J. Miravirsen (SPC3649) can inhibit the biogenesis of miR-122. Nucleic Acids Res. 2014, 42, 609–621. [Google Scholar] [CrossRef]
- van der Ree, M.H.; de Vree, J.M.; Stelma, F.; Willemse, S.; van der Valk, M.; Rietdijk, S.; Molenkamp, R.; Schinkel, J.; Van Nuenen, A.C.; Beuers, U. Safety, tolerability, and antiviral effect of RG-101 in patients with chronic hepatitis C: A phase 1B, double-blind, randomised controlled trial. Lancet 2017, 389, 709–717. [Google Scholar] [CrossRef]
- Deng, Y.; Campbell, F.; Han, K.; Theodore, D.; Deeg, M.; Huang, M.; Hamatake, R.; Lahiri, S.; Chen, S.; Horvath, G.; et al. Randomized clinical trials towards a single-visit cure for chronic hepatitis C: Oral GSK2878175 and injectable RG-101 in chronic hepatitis C patients and long-acting injectable GSK2878175 in healthy participants. J. Viral Hepat. 2020, 27, 699–708. [Google Scholar] [CrossRef]
- Hong, D.S.; Kang, Y.-K.; Borad, M.; Sachdev, J.; Ejadi, S.; Lim, H.Y.; Brenner, A.J.; Park, K.; Lee, J.-L.; Kim, T.-Y. Phase 1 study of MRX34, a liposomal miR-34a mimic, in patients with advanced solid tumours. Br. J. Cancer 2020, 122, 1630–1637. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Liu, N.; Wei, Y.; Zhou, D.; Lin, R.; Wang, X.; Shi, B. Anticancer effects of miR-124 delivered by BM-MSC derived exosomes on cell proliferation, epithelial mesenchymal transition, and chemotherapy sensitivity of pancreatic cancer cells. Aging 2020, 12, 19660–19676. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Lazarus, D.; Robertson, N.; Crosier, E.; Liu, P.; Medarova, Z. Abstract 548: The microRNA-10b targeted therapeutic, TTX-MC138, is effective in preclinical pancreatic adenocarcinoma. Cancer Res. 2023, 83, 548. [Google Scholar] [CrossRef]
- Preethi, K.A.; Selvakumar, S.C.; Ross, K.; Jayaraman, S.; Tusubira, D.; Sekar, D. Liquid biopsy: Exosomal microRNAs as novel diagnostic and prognostic biomarkers in cancer. Mol. Cancer 2022, 21, 54. [Google Scholar] [CrossRef]
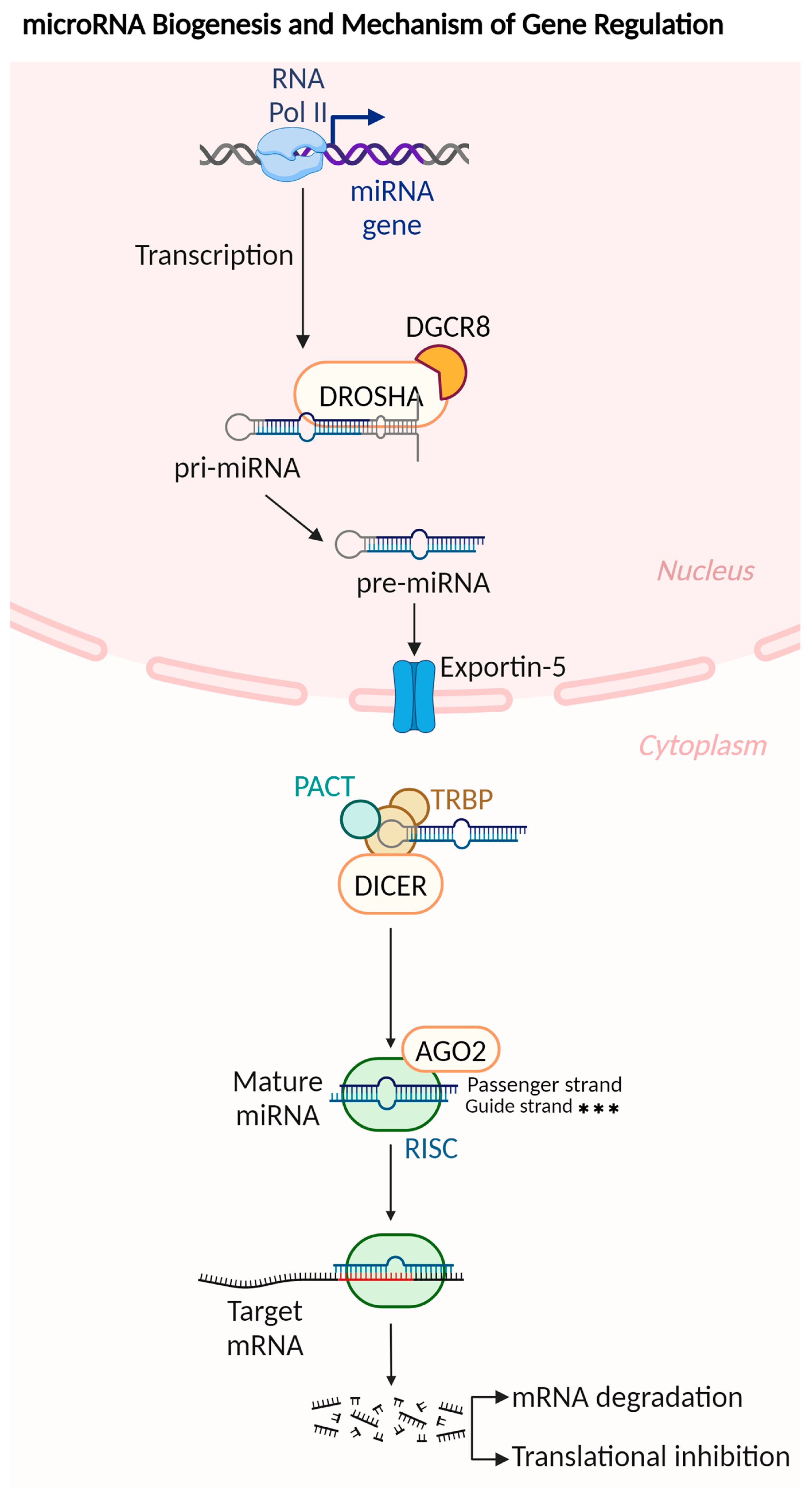
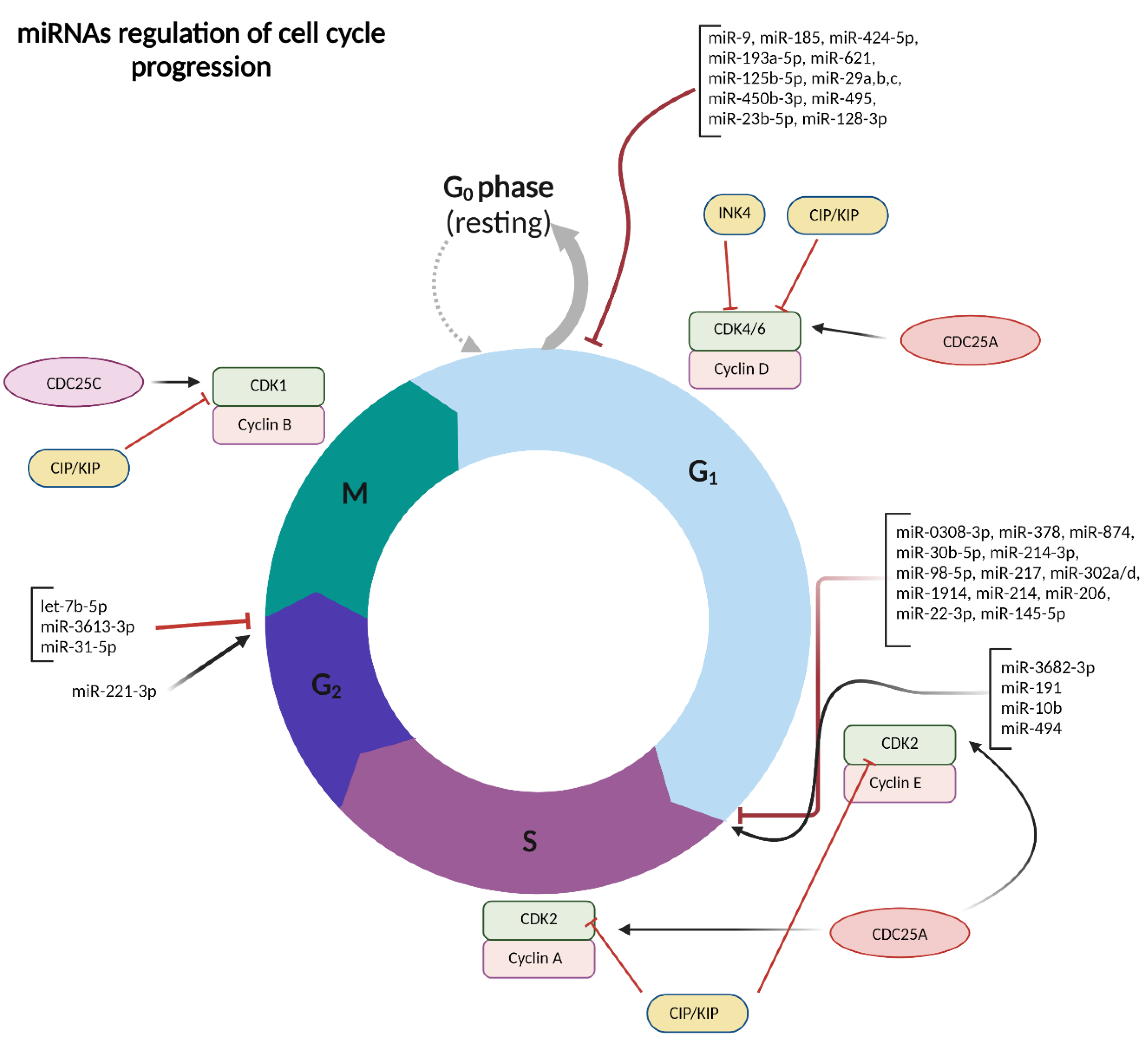
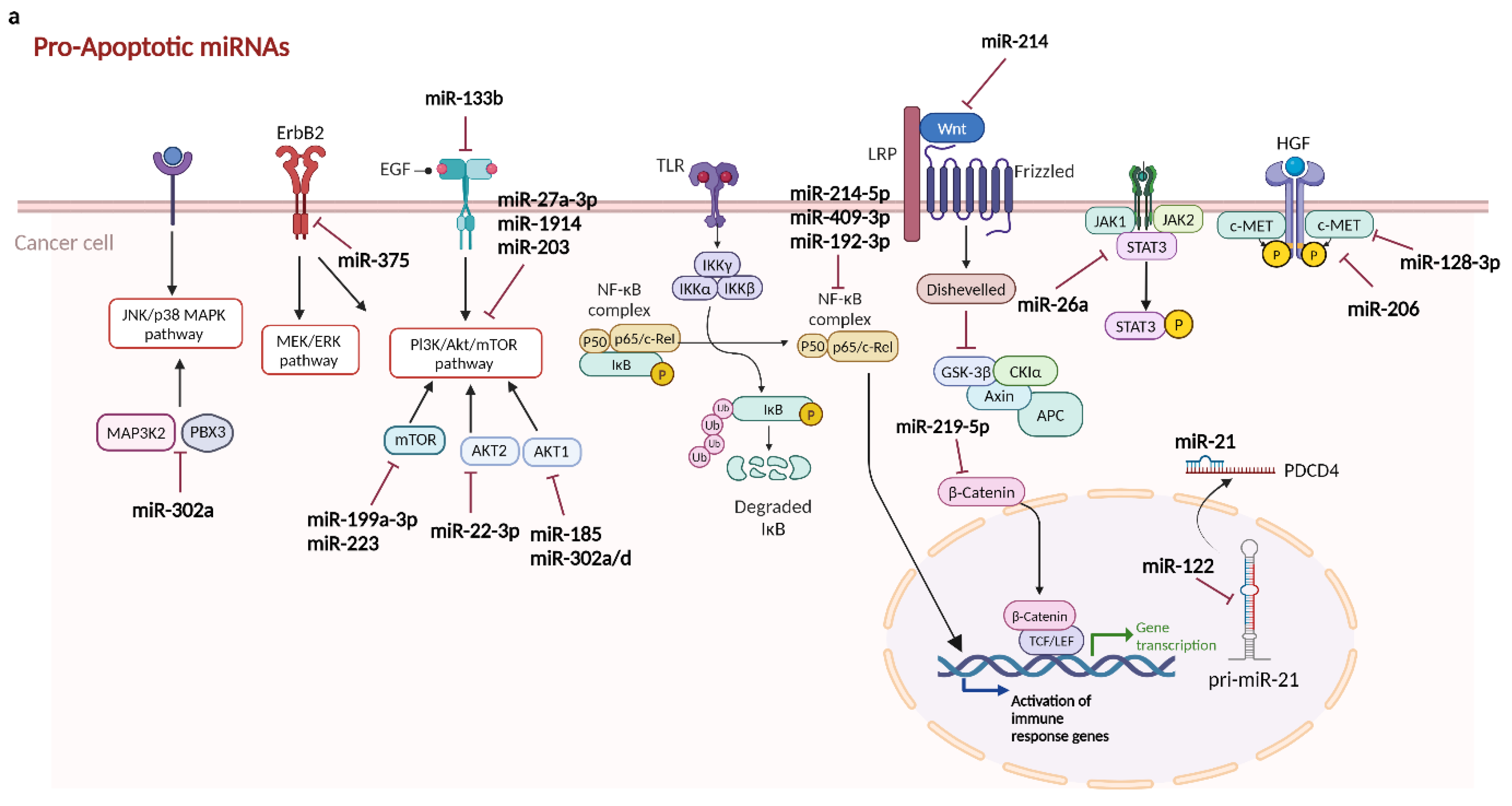
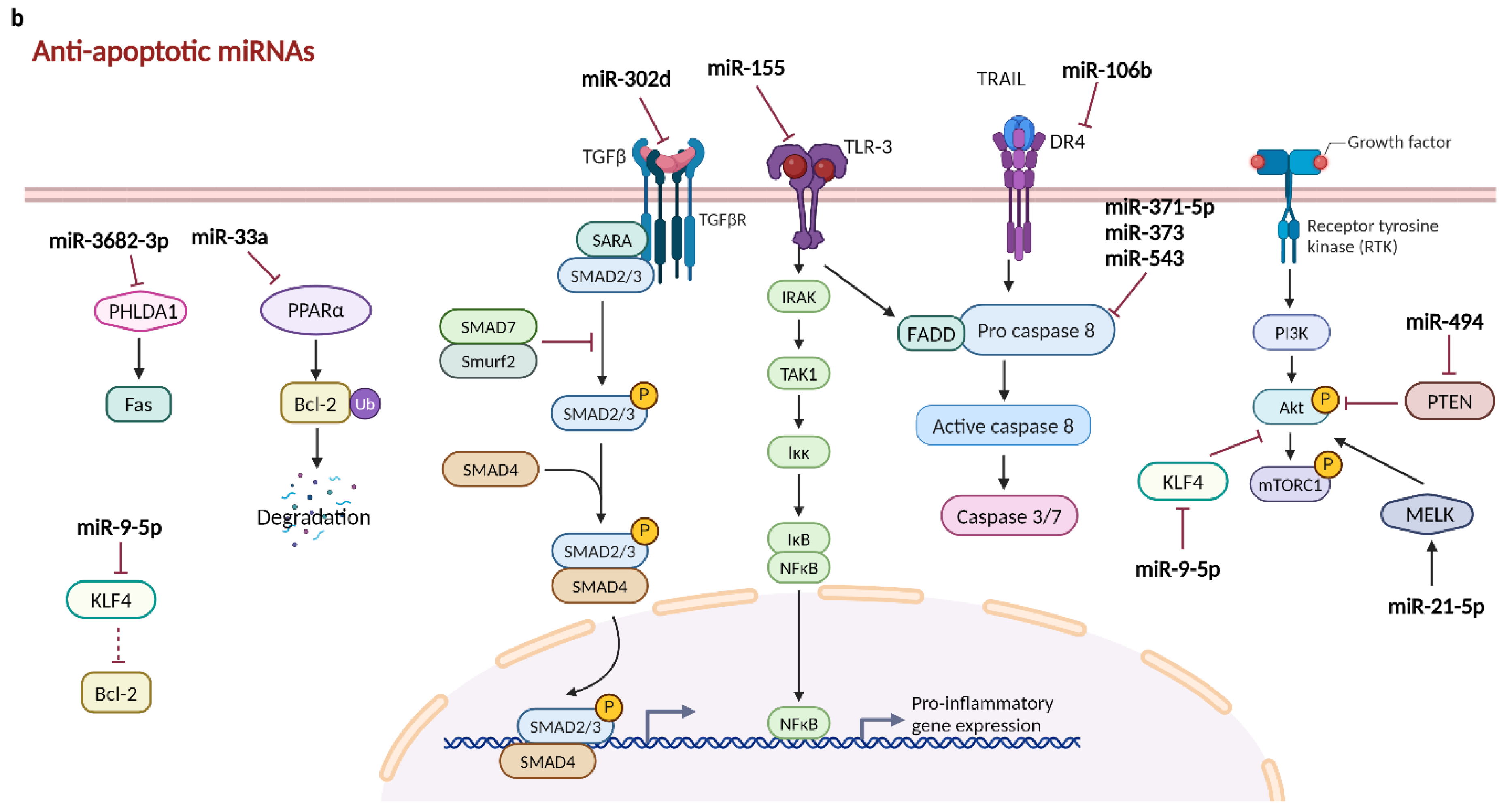

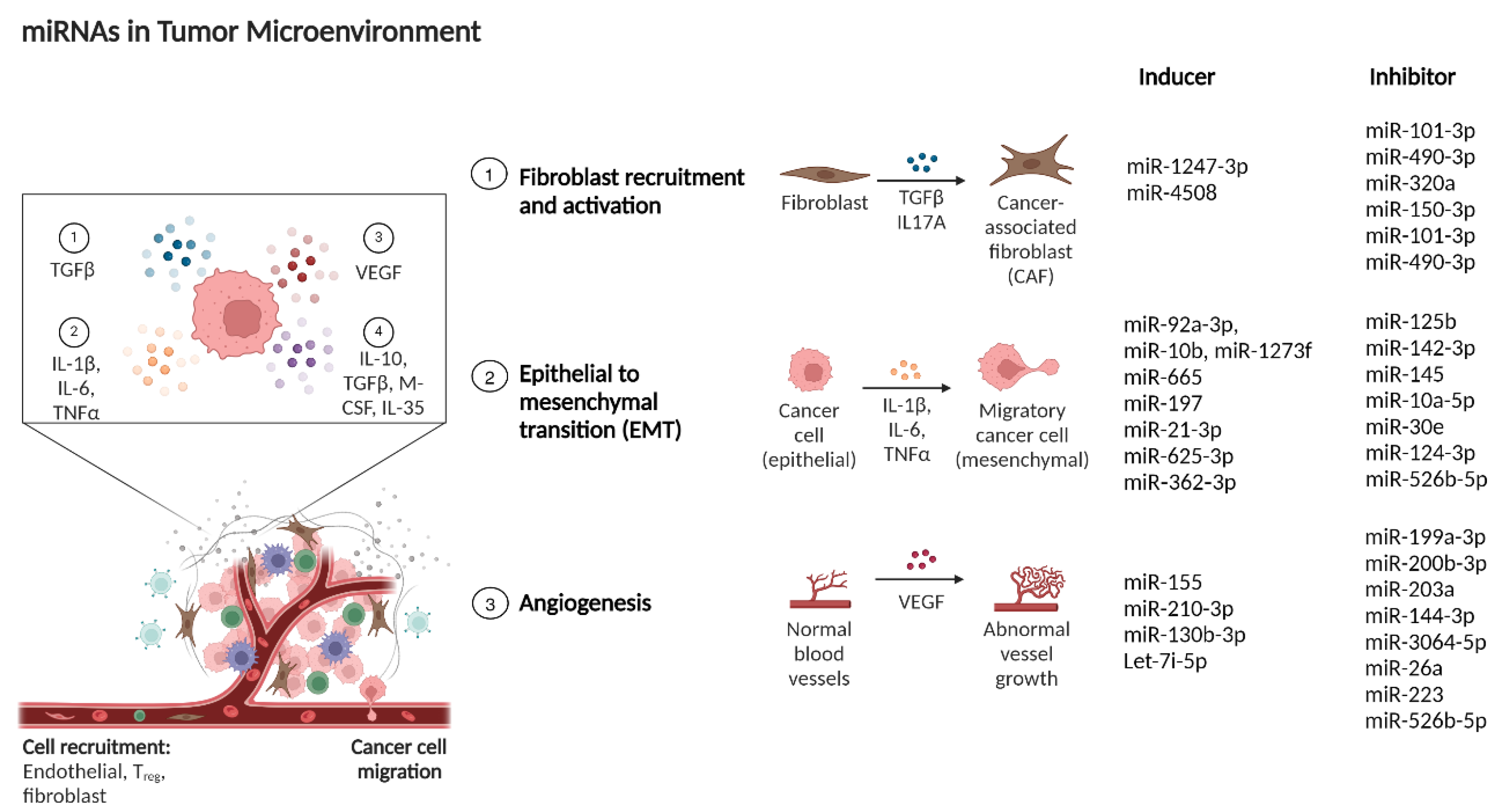

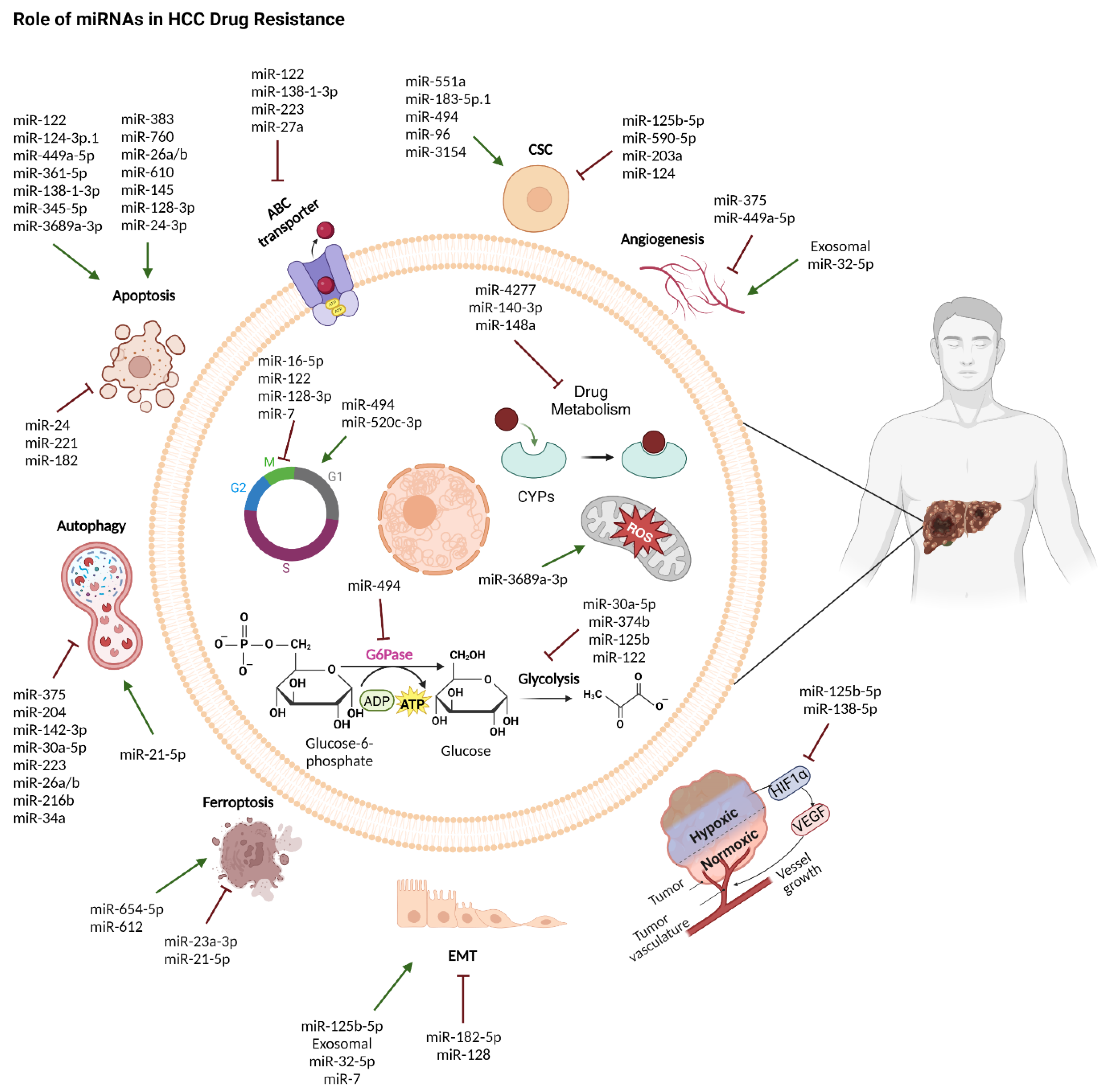
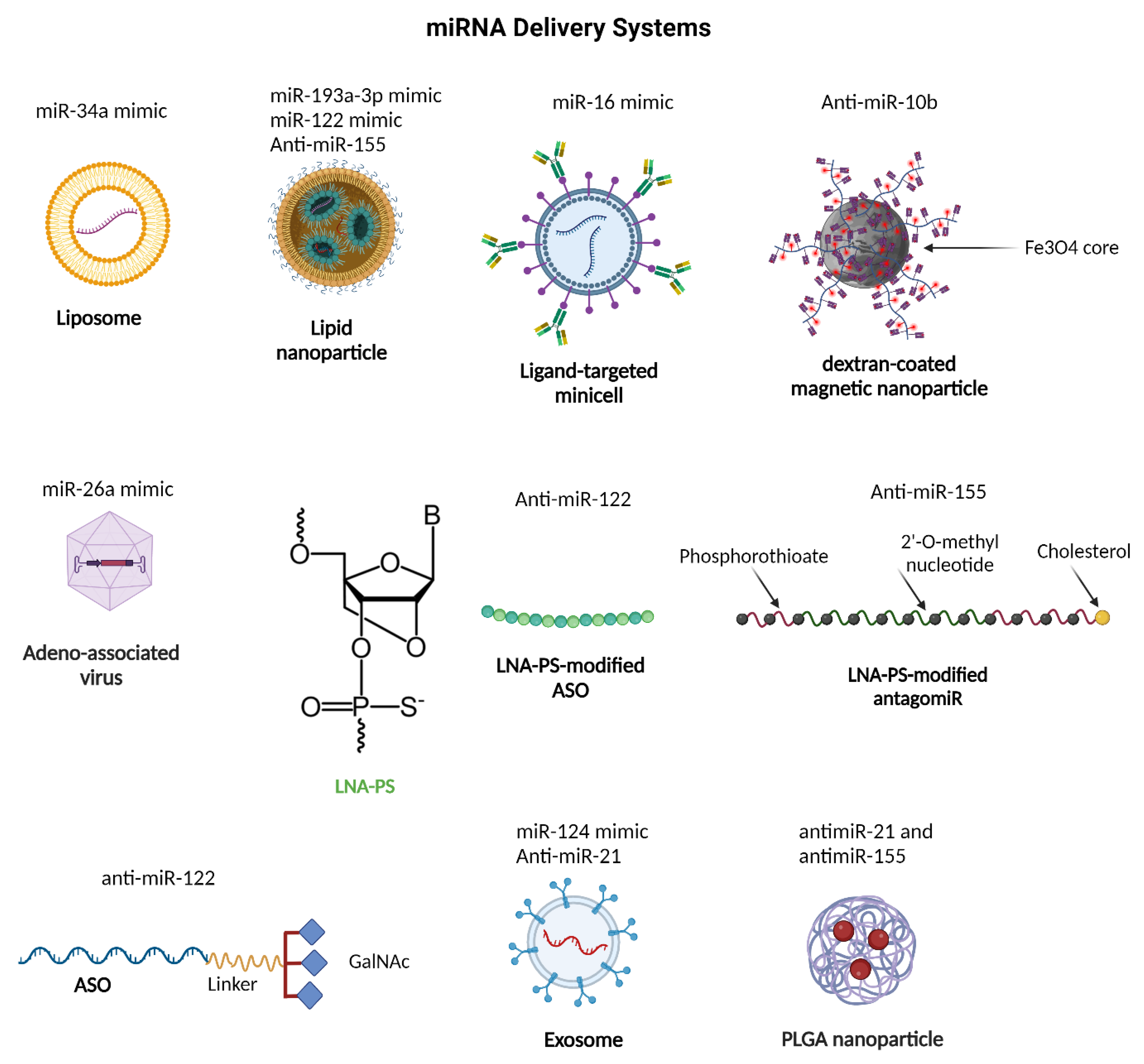
| miRNA | Expression in Liver | Target Genes | Pathway | Cellular Process | Refs. |
|---|---|---|---|---|---|
| Tumor suppressor miRNAs | |||||
| miR-204-5p | Down | RGS20 | N/A | Proliferation (−), Apoptosis (+), Cell cycle arrest (G0/G1) | [101] |
| miR-497-5p | Down | ANXA11 | N/A | Proliferation (−), Apoptosis (+), Cell cycle arrest (G0/G1) | [102] |
| miR-377-3p | Down | RNF38 CPT1C | CPT1C-mediated fatty acid oxidation | Proliferation (−), Apoptosis (+) | [103,104] |
| miR-559 | Down | PARD3 GP73 | N/A | Proliferation (−), Autophagy (−) | [105,106] |
| miR-638 | Down | EZH2 | N/A | Proliferation (−), Apoptosis (+), Autophagy (+) | [107] |
| miR-199a-3p | Down | mTOR, c-Met upregulation of ZHX1 and PUMA | mTOR pathway, ZHX1/PUMA signaling | Proliferation (−), Apoptosis (+), Cell cycle arrest (G0/G1) | [108,109] |
| miR-27a-3p | Down | N/A | PI3K/Akt signaling | Proliferation (−), Apoptosis (+), Cell cycle arrest (G0/G1) | [110] |
| miR-9 | Down | HMGA2 | N/A | Proliferation (−), Cell cycle arrest (G0/G1) | [32] |
| miR-185 | Down | RHEB, RICTOR, and AKT1 | AKT1 pathway | Cell cycle arrest (G0/G1), Apoptosis (+), Autophagy (+) | [94] |
| miR-424-5p | Down | E2F7 | N/A | Proliferation (−), Cell cycle arrest (G0/G1) | [33] |
| miR-193a-5p | Down | NUSAP1 | N/A | Proliferation (−), Cell cycle arrest (G0/G1), Apoptosis (+) | [42] |
| miR-621 | Down | CAPRIN1 | CAPRIN1/CCND2/c-MYC axis | Proliferation (−), Cell cycle arrest (G0/G1) | [34] |
| miR-125b-5p | Down | KIAA1522 | cyclinD1, CDK6, cyclin E and CDK2, and p21 | Proliferation (−), Cell cycle arrest (G0/G1), Apoptosis (+) | [35] |
| miR-29 family (miR-29a, b, c) | Down | RPS15A | cyclin A and cyclin D1, p21 | Proliferation (−), Cell cycle arrest (G0/G1), Apoptosis (+) | [37] |
| miR-450b-3p | Down | PGK1 | AKT signaling | Proliferation (−), Cell cycle arrest (G0/G1) | [38] |
| miR-495 | Down | CTRP3 | N/A | Proliferation (−), Cell cycle arrest (G0/G1) | [111] |
| miR-23b-5p | Down | FOXM1 | c-MYC and cyclin D1 axis | Proliferation (−), Cell cycle arrest (G0/G1) | [112] |
| miR-128-3p | Down | c-Met | c-Met signaling | Proliferation (−), Cell cycle arrest (G0/G1), Apoptosis (+) | [113] |
| miR-875-5p | Down | AEG-1 | N/A | Proliferation (−), Cell cycle arrest (G1/S) | [114] |
| miR-0308-3p | Down | CDK6/Cyclin D1 | N/A | Proliferation (−), Cell cycle arrest (G1/S) | [45] |
| miR-378 | Down | Cyclin D1, Bcl-2, Akt, β-catenin and Survivin | N/A | Proliferation (−), Cell cycle arrest (G1/S), Apoptosis (+) | [59] |
| miR-874 | Down | DOR | DOR/EGFR/ERK pathway | Proliferation (−), Cell cycle arrest (G1/S) | [115] |
| miR-30b-5p | Down | DNMT3A, USP37 | N/A | Proliferation (−), Cell cycle arrest (G1/S) | [116] |
| miR-214-3p | Down | MELK | N/A | Proliferation (−), Cell cycle arrest (G1/S), Apoptosis (+) | [44] |
| miR-214 | Down | Wnt3a | Wnt/β-catenin pathway | Proliferation (−), Cell cycle arrest (G1/S) | [43] |
| miR-214-5p | Down | E2F2 | NF-κB pathway | Proliferation (−), Apoptosis (+) | [117] |
| miR-409-3p | Down | RAB10 | NF-κB pathway | Proliferation (−), Apoptosis (+) | [117] |
| miR-98-5p | Down | IGF2BP1 | N/A | Proliferation (−), Cell cycle arrest (G1/S), Apoptosis (+) | [118] |
| miR-217 | Down | MTDH, EZH2, cyclin-D1, KLF5 | N/A | Proliferation (−), Cell cycle arrest (G1/S), Apoptosis (+) | [46,47,48] |
| miR-302a/d | Down | E2F7 | AKT/βcatenin/CCND1 pathway AKT1-cyclin D1 pathway | Proliferation (−), Cell cycle arrest (G1/S), Apoptosis (+) | [119] |
| miR-1914 | Down | GPR39 | PI3K/AKT/mTOR pathway | Proliferation (−), Cell cycle arrest (G1/S), Apoptosis (+) | [120] |
| miR-206 | Down | cMET, CCND1, and CDK6 | cMet signaling | Proliferation (−), Cell cycle arrest (G1/S), Apoptosis (+) | [65] |
| miR-22-3p | Down | AKT2 | AKT/PI3K pathway | Proliferation (−), Cell cycle arrest (G1/S), Apoptosis (+) | [64] |
| miR-145-5p | Down | SPATS2, p21 and p27 | N/A | Proliferation (−), Cell cycle arrest (G1/S), Apoptosis (+) | [121] |
| miR-203 | Down | MAT2A, MAT2B, NRas | AKT/PI3K pathway RAS/MAPK signaling | Proliferation (−), Cell cycle arrest (S/G2), Apoptosis (+) | [122] |
| let-7b-5p | Down | p21, CDC25B, HMGA2 | CDC25B/CDK1 axis | Proliferation (−) Cell cycle arrest (G2/M) | [49,123] |
| miR-3613-3p | Down | BIRC5, CDK1, NUF2, ZWINT, and SPC24 | N/A | Proliferation (−) Cell cycle arrest (G2/M) | [51] |
| miR-31-5p | Down | SP1 | SP1/cyclin D1 | Proliferation (−) Cell cycle arrest (G2/M) | [50] |
| miR-217 | Down | HMGA2 | N/A | Proliferation (−) Cell cycle arrest (G2/M) | [124] |
| miR-448 | Down | BCL-2 | N/A | Proliferation (−), Apoptosis (+) | [61] |
| miR-223 | Down | NLRP3, Rab1 | NLRP3 inflammasome pathway, mTOR pathway | Proliferation (−), Apoptosis (+) | [125] |
| miR-133b | Down | EGFR, SF3B4 | EGFR/PI3K/Akt/mTOR pathway SF3B4/KLF4/KIP1/SNAI2 axis | Proliferation (−) Apoptosis (+) | [62,126] |
| miR-181a-5p | Down | ATG7 | N/A | Proliferation (−), Autophagy (−) | [95] |
| miR-7 | Down | mTOR, ATG5 | Autophagy | Proliferation (−), Autophagy (+) | [98,127] |
| miR-192-3p | Down | XIAP | NF-κB signaling | Apoptosis (+), Autophagy (−) | [128] |
| miR-519d | Down | Rab10 | AMPK Signaling Pathway | Apoptosis (+), Autophagy (+) | [93] |
| miR-219-5p | Down | NEK6 | β-catenin/c-Myc pathway | Proliferation (−) | [129] |
| miR-375 | Down | ErbB2 | N/A | Proliferation (−) Apoptosis (+) | [130] |
| miR-122 | Down | miR-21 | miR-21–PDCD4 pathway | Proliferation (−) Apoptosis (+) | [131] |
| miR-4651 | Down | FOXP4 | N/A | Proliferation (−) Apoptosis (+) | [132] |
| Let-7b | Down | IGF-1R | N/A | Proliferation (−) Apoptosis (+) | [133] |
| miR-26a | Down | JAK1 | JAK1-STAT3 signaling | Proliferation (−) Apoptosis (+) | [67] |
| miR-26a/b | Down | ULK1 | N/A | Apoptosis (+), Autophagy (−) | [100] |
| miR-342-3p | Down | MCT1 | N/A | Proliferation (−) Apoptosis (+), Necrosis (+) | [134] |
| miR-654-5p | Down | HSPB1 | N/A | Proliferation (−), Ferroptosis (+) | [135] |
| miR-188-3p | Down | GPX4 | N/A | Proliferation (−), Ferroptosis (+) | [136] |
| miR-612 | Down | CoQ10 | HADHA-mediated MVA pathway | Proliferation (−), Ferroptosis (+) | [137] |
| Oncogenic miRNAs | |||||
| miR-183-5p | Up | PDCD4 | N/A | Proliferation (+), Apoptosis (−) | [138] |
| miR-494 | Up | p27, PUMA, and PTEN | mTOR pathway | Proliferation (+), Cell cycle (induces G1/S transition), Apoptosis (−) | [52] |
| miR-3682-3p | Up | PHLDA1 | PHLDA1-Fas pathway | Proliferation (+), Cell cycle (induces G1/S transition), Apoptosis (−) | [54] |
| miR-191 | Up | KLF6 | N/A | Proliferation (+), Cell cycle (induces G1/S transition) | [53] |
| miR-10b | Up | CSMD1 | N/A | Proliferation (+), Cell cycle (induces G1/S transition), Apoptosis (−) | [55] |
| miR-221-3p | Up | MGMT | N/A | Proliferation (+), Cell cycle (induces G2/M transition), Apoptosis (−) | [56] |
| miR-21-5p | Up | MELK | AKT/mTOR signaling | Proliferation (+), Ferroptosis (−) | [139] |
| miR-338-5p | Up | RTN4 | N/A | Proliferation (+), Apoptosis (−) | [140] |
| miR-9-5p | Up | Klf4 | AKT/mTOR signaling | Proliferation (+), Apoptosis (−) | [141] |
| miR-302d | Up | TGFBR2 | TGF-β signaling | Proliferation (+), Apoptosis (−) | [142] |
| miR-371-5p, miR-373 and miR-543 | Up | Casp-8 | N/A | Necrosis (+) | [85] |
| miR-106b | Up | DR4 | TRAIL pathway | Proliferation (+), Apoptosis (−) | [81] |
| miR-3174 | Up | FOXO1 | N/A | Proliferation (+), Apoptosis (−) | [143] |
| miR-33a | Up | PPARα | N/A | Proliferation (+), Apoptosis (−) | [75] |
| miR-155 | Up | TLR3 | TLR3-NF-kB pathway | Proliferation (+), Apoptosis (−) | [80] |
| miR-93-5p | Up | ERBB4 | TETs dependent DNA demethylation | Proliferation (+), Apoptosis (−) | [144] |
| miRNA | Target Genes | Pathway | Stemness | Ref. |
|---|---|---|---|---|
| Tumor suppressor miRNAs | ||||
| miR-6838-5p | CBX4 | ERK signaling | (−) | [154] |
| miR-148b | NRP1 | ACVR1-BMP-Wnt axis | (−) | [157] |
| miR-148a | ACVR1 | Brg1/Sall4 axis | (−) | [156] |
| miR-296-5p | Brg1 | AKT/βcatenin/CCND1 signaling | (−) | [158] |
| miR-302a/d | E2F7 | N/A | (−) | [119] |
| miR-589-5p | MAP3K8 | SOX9 signaling | (−) | [155] |
| miR-125a/b | CD90 | N/A | (−) | [163] |
| miR-613 | SOX9 | PI3K/AKT/mTOR pathway | (−) | [164] |
| miR-100 and miR-125b | IGF2 | hexosamine pathway | (−) | [165] |
| miR-325-3p | DPAGT1 | N/A | (−) | [166] |
| miR-217 | HMGA2 | N/A | (−) | [124] |
| miR-448 | MAGEA | AMPK signaling | (−) | [167] |
| Oncogenic miRNAs | ||||
| miR-106b-5p | PTEN | PTEN/PI3K/AKT pathway | (+) | [162] |
| miR-1246 | AXIN2, GSK3β | Wnt/β-catenin pathway | (+) | [160] |
| miR-5188 | FOXO1 | β-catenin/Wnt signaling | (+) | [161] |
| miR-454-3p | CPEB1 | N/A | (+) | [168] |
| miRNA | Target Genes | Pathway | Cellular Process | Refs. |
|---|---|---|---|---|
| Anti-metastatic miRNAs | ||||
| miR-320a | HMGB1, c-Myc | N/A | N/A | [202,203] |
| miR-29c-3p | ADAM12 | N/A | N/A | [204] |
| miR-188-5p | FOXN2 | N/A | N/A | [205] |
| miR-142-3p | ZEB1, TGF-β1, HMGB1 | TGF-β pathway | EMT, Angiogenesis | [206,207,208] |
| Exosomal miR-125b | SMAD2 | TGFβ1/SMAD signaling | EMT | [209] |
| Exosomal miR-29b | DNMT3b | N/A | EMT | [210] |
| miR-10a-5p | SKA1 | N/A | EMT | [211] |
| miR-2113 | WDR66 | N/A | EMT | [195] |
| miR-30e | MTA1 | MTA1/ErbB2 axis | EMT | [212] |
| miR-517c | KPNA2 | PI3K/AKT pathway | EMT | [213] |
| miR-130a-3p | AR | N/A | EMT, Angiogenesis | [176] |
| miR-526b-5p | HGF | HGF/c-Met pathway | EMT, Angiogenesis | [214] |
| miR-4270-5p | SATB2 | N/A | EMT | [215] |
| miR-124-3p | CRKL, Sp1 | RAF/MEK/ERK1/2 pathways | EMT | [177,216] |
| miR-875-5p | AEG1/MTDH | N/A | EMT | [114] |
| miR-296-5p | NRG1 | NRG1/ERBB2/ERBB3/RAS/MAPK/Fra-2 signaling | EMT | [159] |
| miR-449a | Notch1 | Notch pathway | EMT | [217] |
| miR-30a | Beclin 1, Atg5 | Autophagy pathway | Autophagy | [99] |
| miR-7 | FAK | Akt pathway | N/A | [190] |
| miR-148a-3p | SMAD2 | TGF-β signaling | N/A | [218] |
| miR-298 | CTNND1 | Wnt/β-catenin signaling | N/A | [219] |
| miR-409-3p | BRF2 | Wnt/β-catenin signaling | N/A | [220] |
| miR-466 | MTDH/AEG-1 | N/A | N/A | [221] |
| miR-138-5p | FOXC1 | N/A | N/A | [222] |
| miR-455-5p | IGF-1R | IGF-1R-AKT-GLUT1 axis | N/A | [223] |
| miR-139-3p | UCK2 | N/A | N/A | [224] |
| miR--219-5p | NEK6 | β-catenin/c-Myc pathway | N/A | [225] |
| miR-145 | SERPINE1 | HIF-1 signaling pathway | N/A | [226] |
| miR-515–5p | IL6 | IL-6/JNK/STAT3 signaling | N/A | [227] |
| miR-378a-3p | PD-L1 | STAT3 signaling | Immune response | [228] |
| miR-199a/b-5p | ROCK1 | ROCK1/MLC and PI3K/Akt signaling | N/A | [229] |
| miR-378a-5p | VEGF | VEGF pathway | Angiogenesis | [230] |
| miR-495 | IGF1R | N/A | N/A | [231] |
| miR-194-3p | MMP9 | N/A | ECM degradation | [232] |
| miR-489 | MMP7 | N/A | ECM degradation | [233] |
| miR-211-5p | ACSL4 | N/A | N/A | [175] |
| miR-342-3p | AGR2 | N/A | Apoptosis, cell cycle, ECM degradation | [234] |
| miR-767-3p | SMIM7 | N/A | N/A | [235] |
| miR-139 | GDF10 | N/A | N/A | [236] |
| miR-15a-3p | HMOX1 | N/A | N/A | [179] |
| miR-449 family | SOX4 | TGF-β pathway | N/A | [237] |
| miR-708 | SMAD3 | TGF-β pathway | N/A | [238] |
| miR-300 | LEF-1 | N/A | N/A | [239] |
| miR-98 | EZH2, IL-10 | Wnt/β-catenin pathway | N/A | [240,241] |
| Metastatic miRNAs | ||||
| miR-362-3p | CD82 | TGF-β signaling | EMT | [242] |
| miR-625-3p | PDLIM5 | N/A | EMT | [243] |
| miR-92a-3p | PTEN | Akt/Snail pathway, PI3K/AKT/mTOR Signaling | EMT | [244,245] |
| miR-93-5p | directly upregulates MAP3K2, inhibits ERBB4 and TETs | MAP3K2/ p38-JNK/p21 signaling pathway | N/A | [144,201] |
| miR-10a-5p | NCOR2 | Wnt/β-catenin pathway | EMT | [246] |
| miR-HCC3 | TNFRSF19, RAB43 | N/A | EMT | [247] |
| miR-197 | Axin-2, NKD1, DKK2 | Wnt/β-catenin pathway | EMT | [248] |
| miR-21-3p | SMAD7 | SMAD7/YAP1 axis | EMT | [249] |
| miR-3691-5p | PTEN | PI3K/Akt signaling | N/A | [250] |
| miR-106b | PTEN | N/A | N/A | [251] |
| miR-18a | KLF4, Bcl2L10 | N/A | N/A | [196,197] |
| miR-106b-5p | FOG2 | N/A | N/A | [199] |
| miR-376c-3p | ARID2 | N/A | N/A | [200] |
| miR-182-5p | FOXO3a | AKT/FOXO3a pathway, Wnt/β-catenin signaling | N/A | [252] |
| miR-769-5p | RYBP | N/A | N/A | [253] |
| miR-1251-5p | AKAP12 | N/A | N/A | [254] |
| miR-483-5p | Positive regulator of IGF-II | N/A | N/A | [255] |
| miR-106b-5p | GPM6A | AKT/ERK signaling | N/A | [256] |
| Exosomal miR-25-5p | SIK1 Fbxw7, LRRC7 | Wnt/β-catenin signaling | N/A | [198,257,258] |
| Exosomal miR-21 | PTEN | PDK1/AKT signalling | N/A | [259] |
| Exosomal miR-1273f | LHX6 | Wnt/β-catenin signaling | EMT | [260] |
| Exosomal miR-92a-3p | PTEN | Akt/Snail signaling | N/A | [244] |
| Exosomal miR-224 | GNMT | N/A | N/A | [261] |
| Extracellular vesicle (EV)-miR-3129 | TXNIP | N/A | N/A | [262] |
| Exosomal miR-1247-3p | B4GALT3 | β1-integrin/NF-κB signaling | N/A | [263] |
| miRNA | Source of Cell Type | Target Gene | Pathway | Function | Ref. |
|---|---|---|---|---|---|
| Tumor suppressor miRNAs | |||||
| Exosomal miR-320a | CAFs | PBX3 | MAPK pathway | Inhibits HCC proliferation and metastasis | [302] |
| Exosomal miR-150-3p | CAFs | N/A | N/A | Inhibits HCC invasion | [282] |
| miR-101-3p/miR-490-3p | CAFs | TGFBR1 | N/A | Induce infiltration of myeloid-derived suppressor cells, regulatory T cells, and M2 macrophages | [283] |
| miR-26a | HCC cells | M-CSF | PI3K/Akt pathway | Inhibits macrophage recruitment and M2 polarization | [303] |
| Exosomal miR-628-5p | M1 macrophages | METTL14 | circFUT8/miR-552-3p/CHMP4B pathway | Inhibits HCC progression | [304] |
| miR-144/miR-451a | HCC cells | HGF, MIF | N/A | Induce macrophage M1 polarization | [288] |
| Exosomal miR-125a/b | M2 macrophages | CD90 | N/A | Inhibit cell proliferation and stem cell properties | [163] |
| miR-148b | HCC cells | CSF1 | CSF1/CSF1R pathway | Inhibits TAM infiltration, HCC growth and metastasis | [289] |
| miR-28-5p | HCC cells | IL-34 | TGFβ1 signaling | Inhibits TAM infiltration, HCC growth and metastasis | [290] |
| miR-200 | HCC cells | PD-L1 | N/A | Induces CD8+ T cells viability, inhibits metastasis | [305] |
| miR-145 | HCC cells | PD-L1 | PI3K/AKT signaling | Inhibits tumor growth, EMT, and metastasis | [306] |
| miR-194-5p | HCC cells | PD-L1 and PD-L2 | N/A | Reduces cytotoxic T cells apoptosis | [307] |
| miR-223 | HCC cells | HIF1α | CD39/CD73-adenosine pathway | Inhibits infiltration of PD-1+ T cells and PD-L1+ macrophages, inhibits angiogenesis | [293] |
| miR-329-3p | HCC cells | KDM1A | N/A | Inhibits the expression of PD-L1 in HCC cells via increasing MEF2D methylation, inhibits tumor growth | [294] |
| miR-200c | HBV+ HCC cells | CD274 | HBV-pSTAT3-SALL4-miR-200c-PD-L1 axis | Inhibits HBV-mediated PD-L1 expression and CD8+ T cell exhaustion | [295] |
| miR-22 | HCC cells | HIF1α | Retinoic acid signaling | Inhibits IL17 signaling, expands cytotoxic T cells and reduces Treg | [296] |
| miR-30-5p | HCC cells | Snail | Snail-DDP4- CXCL10 axis | Induces CD8+ T cell infiltration | [299] |
| miR-570 | HCC cells | CD31 and VEGF | N/A | Increases CD8+IFN-γ+ T cells, induces apoptosis, inhibits angiogenesis | [308] |
| miR-374b | CIK cells | PD-1 | N/A | Induces cytotoxicity of cytokine-induced killer cells | [309] |
| miR-1258 | NK cells and DC cells | N/A | N/A | Stimulates TLR7/8 expression, activates NKs and promotes DCs maturation, inhibits tumor growth and metastasis | [300] |
| Oncogenic miRNAs | |||||
| Exosomal miR-1247-3p | HCC cells | B4GALT3 | β1-integrin–NF-κB signaling | Induces CAFs activation and lung metastasis | [263] |
| Exosomal miR-4508 | HCC cells | RFX1 | RFX1-IL17A-p38 MAPK-NF-κB pathway | Activates lung fibroblasts and induces lung metastasis | [281] |
| Exosomal miR-21 | HCC cells | PTEN | PDK1/AKT signaling | Converts normal HSCs to CAFs, induces angiogenesis | [259] |
| Exosomal miR-200b-3p | HCC cells | ZEB1 | JAK/STAT signaling | Induces macrophage M2 polarization | [310] |
| Exosomal miR-4669 | HCC cells | N/A | N/A | Induces M2 macrophage polarization, migration ability, and sorafenib resistance | [286] |
| Exosoma miR-92a-2-5p | M2 macrophages | AR | AR/PHLPP/p-AKT/β-catenin signaling | Induces HCC cells invasion | [311] |
| Exosomal miR-27a-3p | M2 macrophages | TXNIP | N/A | Induces HCC cells stemness, proliferation, drug resistance, migration, invasion, and tumorigenicity | [312] |
| Exosomal miR-660-5p | M2 macrophages | KLF3 | N/A | Induces EMT | [313] |
| Exosomal miR-452-5p | HCC cells | TIMP3 | N/A | Induces M2 macrophage polarization, | [287] |
| miR-889 | HCC cells | MICB | N/A | Reduces NK cell-mediated cytotoxicity | [301] |
| miR-561-5p | HCC cells | CX3CL1 | STAT3 signaling | Reduces CX3CR1+ NK cell infiltration, induces tumor growth and lung metastasis | [4] |
| miRNA | Target Genes | Pathway | Ref. |
|---|---|---|---|
| Anti-angiogenic miRNAs | |||
| miR-26a | HGF | HGF/c-Met pathway | [331] |
| miR-526b-5p | HGF | HGF/c-Met pathway | [214] |
| miR-200b | Transcription factor ERG | N/A | [322] |
| miR-203a | HOXD3 | N/A | [318] |
| miR-144-3p | SGK3 | N/A | [320] |
| miR-375 | PDGFC, AEG-1 | N/A | [332] |
| miR-3064-5p | FOXA1 | FOXA1/CD24/Src pathway | [323] |
| miR-199a-3p | MMP2, HGF, VEGFA, VEGFR1 | N/A | [316] |
| miR-378a-5p | VEGF | VEGF pathway | [230] |
| miR-223 | HIF1α | CD39/CD73-adenosine pathway | [293] |
| miR-1296 | E2F7 | N/A | [333] |
| Pro-angiogenic miRNAs | |||
| Let-7i-5p | TSP1 | N/A | [334] |
| miR-210-3p | SMAD4, STAT6 | N/A | [330] |
| miR-130b-3p | HOXA5 | N/A | [328] |
| miRNA | Target Genes | Drug Response | Cellular Process | Pathway | Ref. |
|---|---|---|---|---|---|
| miRNAs improving drug sensitivity | |||||
| miR-375 | MDR1, AEG1, YAP1, and ATG7 | doxorubicin | Proliferation, autophagy | N/A | [349] |
| miR-223 | ABCB1 | doxorubicin | N/A | N/A | [350] |
| miR-125b | HIF1A, YBX1, PDGFRB | doxorubicin | stemness | HIF1α/PDGFβ/pAKT | [338] |
| miR-383 | EIF5A2 | doxorubicin | Proliferation, apoptosis | N/A | [351] |
| miR-140-5p | PIN1 | doxorubicin | Proliferation | N/A | [352] |
| miR-590-5p | YAP1 | doxorubicin | Proliferation, stemness | Hippo signaling | [353] |
| miR-122 | ABCB1 and ABCF2 | doxorubicin | Cell cycle | N/A | [354] |
| miR-122 | PKM2 | doxorubicin | Glycolysis | N/A | [355] |
| miR-760 | Notch1 | doxorubicin | Proliferation, apoptosis | Notch1/Hes1-PTEN/Akt Signaling | [356] |
| miR-26a/b | ULK1 | doxorubicin | Autophagy, apoptosis | N/A | [100] |
| miR-218-5p | EIF5A2 | doxorubicin | N/A | N/A | [357] |
| miR-325-3p | DPAGT1 | doxorubicin | N/A | Hexosamine pathway | [166] |
| miR-223 | FOXO3a | doxorubicin | Autophagy | N/A | [339] |
| miR 361 5p | MAP3K9 | cisplatin | Apoptosis | N/A | [341] |
| miR-610 | HDGF | cisplatin | Proliferation and apoptosis | N/A | [342] |
| miR-10a-5p | MSI1 | cisplatin | Proliferation and apoptosis | AKT signaling | [344] |
| miR-27a-3p | ABCB1 | 5-fluorouracil cisplatin | Proliferation and apoptosis | PI3K/Akt pathway | [110,358] |
| miR-203a | BMI1 | 5-fluorouracil | Proliferation, stemness | N/A | [359] |
| miR-125b | HK II | 5-fluorouracil | Glycolysis | N/A | [360] |
| miR-216b | MALAT1 | 5-fluorouracil | Autophagy | N/A | [340] |
| miR-145 | TLR4 | 5-fluorouracil | Apoptosis | N/A | [361] |
| miR-138-5p | HIF-1α | Radiosensitivity | N/A | Migration/invasion, EMT | [362] |
| miRNAs inducing drug resistance | |||||
| miR-182 | TP53INP1 | cisplatin | Viability | N/A | [348] |
| miR-551a | MEF2C | 5-fluorouracil | Viability and sphere formation | N/A | [347] |
| Exosomal miR-32-5p | PTEN | 5-fluorouracil | Angiogenesis, EMT | PI3K/Akt pathway | [363] |
| miR-24 and miR-221 | caspase 8/3 | TRAIL | Angiogenesis | N/A | [364] |
| miRNA | Target Genes | Targeted Therapy Agent | Cellular Process | Pathway | Refs. |
|---|---|---|---|---|---|
| miRNAs improving drug sensitivity | |||||
| Exosomal miR-744 | PAX2 | Sorafenib | N/A | N/A | [400] |
| miR-3689a-3p | CCS | Sorafenib | mitochondrial oxidative stress, Apoptosis | CCS/SOD1 axis | [389] |
| miR-124 | CAV1 | Sorafenib | Stemness | N/A | [401] |
| miR-338-3p | RAB1B | Sorafenib | Apoptosis, invasion | N/A | [402] |
| miR-122 | IGF-1R | Sorafenib | Apoptosis | RAS/RAF/ERK signaling | [403] |
| miR-345-5p | TOP2A | Sorafenib | Apoptosis | N/A | [388] |
| miR-122 | SerpinB3 | Sorafenib | Apoptosis | N/A | [404] |
| miR-138-1-3p | PAK5 | Sorafenib | Apoptosis | β-catenin/ABCB1 signaling | [386] |
| miR-654-5p | HSPB1 | Sorafenib | Ferroptosis | N/A | [135] |
| miR-182-5p | N/A | Sorafenib | EMT | N/A | [405] |
| miR-128 | CD151 | Sorafenib | EMT | N/A | [406] |
| miR-1294 | TGFβR1 | Sorafenib | EMT | N/A | [407] |
| miR-16-5p | cyclin E1 | Sorafenib | Cell cycle | PTEN/Akt signaling | [408] |
| miR-449a-5p | PEA15, PPP1CA, TUFT1 | Sorafenib | N/A | AKT and ERK signaling | [372] |
| miR-4277 | CYP3A4 | Sorafenib | Drug metabolism | N/A | [373] |
| miR-374b | hnRNPA1 | Sorafenib | Aerobic glycolysis | PKM2 | [409] |
| miR-30a-5p | ATG5, Beclin-1, CLCF1 | Sorafenib | Autophagy, aerobic glycolysis | PI3K/AKT signaling | [375,376] |
| miR-204 | ATG3 | Sorafenib | Autophagy | N/A | [377] |
| miR-378a-3p | IGF1R | Sorafenib | N/A | ERK/PI3K signaling | [380] |
| miR-140-3p | PXR | Sorafenib | Drug clearance | N/A | [383] |
| miR-375 | PDGFC, AEG-1, ATG14 | Sorafenib | Angiogenesis, autophagy | N/A | [332,410] |
| miR-124-3p.1 | AKT2, SIRT1 | Sorafenib | N/A | FOXO3a pathway | [385] |
| miR-142-3p | ATG5, ATG16L1 | Sorafenib | Autophagy | N/A | [378] |
| miR-148a | PXR | Sorafenib | Drug clearance | N/A | [384] |
| miR-486-3p | FGFR4, EGFR | Sorafenib | N/A | N/A | [411] |
| miR-3163 | ADAM-17 | Sorafenib | N/A | Notch signaling | [412] |
| miR-483-5p | PPARα, TIMP2 | Sorafenib | Apoptosis, steatosis, fibrosis | Notch signaling | [413] |
| miR-3163 | ADAM17 | Sorafenib | N/A | Notch signaling | [412] |
| miR-5590-3p | PINK1 | Lenvatinib | Apoptosis | N/A | [414] |
| miR-128-3p | c-Met | Lenvatinib | Apoptosis, cell cycle | c-Met pathway | [113] |
| miR-24-3p | BCL2L2 | Lenvatinib | Apoptosis | N/A | [395] |
| miR-34a | Beclin-1 | Lenvatinib | Autophagy | N/A | [396] |
| miRNAs inducing drug resistance | |||||
| miR-21-5p | USP24 | Sorafenib | Autophagy | USP24-SIRT7 axis | [394] |
| miR-23a-3p | ACSL4 | Sorafenib | Ferroptosis | N/A | [390] |
| miR-125b-5p | Ataxin-1 | Sorafenib | EMT | N/A | [392] |
| miR-494 | p27, PUMA, PTEN, G6pc | Sorafenib | Cell cycle, Survival, invasion, stemness, glycogenolysis, gluconeogenesis | mTOR pathway | [52] |
| miR-126-3p | SPRED1 | Sorafenib | N/A | ERK signaling pathway | [415] |
| miR-96 | TP53INP1 | Sorafenib | promotes liver T-ICs expansion | N/A | [416] |
| miR-3677-3p | FBXO31 | Sorafenib | Proliferation, invasion, stemness, apoptosis | N/A | [417] |
| miR-4669 | SIRT1 | Sorafenib | Immunosuppressive TME | N/A | [286] |
| miR-223 | FBW7 | Sorafenib | N/A | N/A | [418] |
| miR-183-5p.1 | MUC15 | Lenvatinib | Proliferation, apoptosis, stemness | c-MET/PI3K/AKT/SOX2 signaling | [397] |
| miR-520c-3p | MBD2 | Lenvatinib | Proliferation, cell cycle | GF19/FGFR4/FRS2 signaling | [398] |
| miR-3154 | HNF4α | Lenvatinib | Proliferation, apoptosis, stemness | N/A | [399] |
| Therapeutic Molecule | Target miRNA | Disease | Phase | Delivery Platform | Route of Administration | Ref. |
|---|---|---|---|---|---|---|
| miRNA replacement therapy | ||||||
| MRX34 | miR-34a | Advanced solid tumors | Phase I | Liposomal | IV | [446] |
| TargomiR | miR-16 | MPM, NSCLC | Phase I | EGFR targeting minicell | IV | [86] |
| INT-1B3 | miR-193a-3p | Advanced solid tumors | Phase I | LNP | IV | [428] |
| N/A | miR-124 | Pancreatic cancer | Preclinical | Exosome | SC | [447] |
| N/A | miR-122 | HCC | Preclinical | LNP | IV | [433] |
| N/A | miR-26a | HCC | Preclinical | MSCV-derived retroviral construct | IV | [434] |
| N/A | miR-22 | HCC | Preclinical | AAV | IV | [402] |
| miRNA inhibition therapy | ||||||
| MRG-106 (cobomarsen) | miR-155 | CTCL, CLL, DLBCL, ATLL | Phase II | LNA | IT, SC, IV | [436] |
| Miravirsen | miR-122 | Chronic HCV | Phase II | LNA-PS-modified ASO | SC | [443] |
| RG-101 | miR-122 | Chronic HCV | Phase II | GalNAc-ASO | SC | [445] |
| TTX-MC138 | miR-10b | PAC | Preclinical | NP | IV | [448] |
| N/A | miR-155 | HCC | Preclinical | LNP | IV | [438] |
| N/A | miR-21 | CRC | Preclinical | Exosome | IV | [441] |
| N/A | miR-21 and miR-155 | Lymphoma | N/A | NP containing PS and PNA | N/A | [437] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahboobnia, K.; Beveridge, D.J.; Yeoh, G.C.; Kabir, T.D.; Leedman, P.J. MicroRNAs in Hepatocellular Carcinoma Pathogenesis: Insights into Mechanisms and Therapeutic Opportunities. Int. J. Mol. Sci. 2024, 25, 9393. https://doi.org/10.3390/ijms25179393
Mahboobnia K, Beveridge DJ, Yeoh GC, Kabir TD, Leedman PJ. MicroRNAs in Hepatocellular Carcinoma Pathogenesis: Insights into Mechanisms and Therapeutic Opportunities. International Journal of Molecular Sciences. 2024; 25(17):9393. https://doi.org/10.3390/ijms25179393
Chicago/Turabian StyleMahboobnia, Khadijeh, Dianne J. Beveridge, George C. Yeoh, Tasnuva D. Kabir, and Peter J. Leedman. 2024. "MicroRNAs in Hepatocellular Carcinoma Pathogenesis: Insights into Mechanisms and Therapeutic Opportunities" International Journal of Molecular Sciences 25, no. 17: 9393. https://doi.org/10.3390/ijms25179393






