L-Glutamate Regulates Npy via the mGluR4-Ca2+-ERK1/2 Signaling Pathway in Mandarin Fish (Siniperca chuatsi)
Abstract
:1. Introduction
2. Results
2.1. Synteny, Sequence Alignment, and Phylogenetic Analysis of mGluR4 Genes in Mandarin Fish
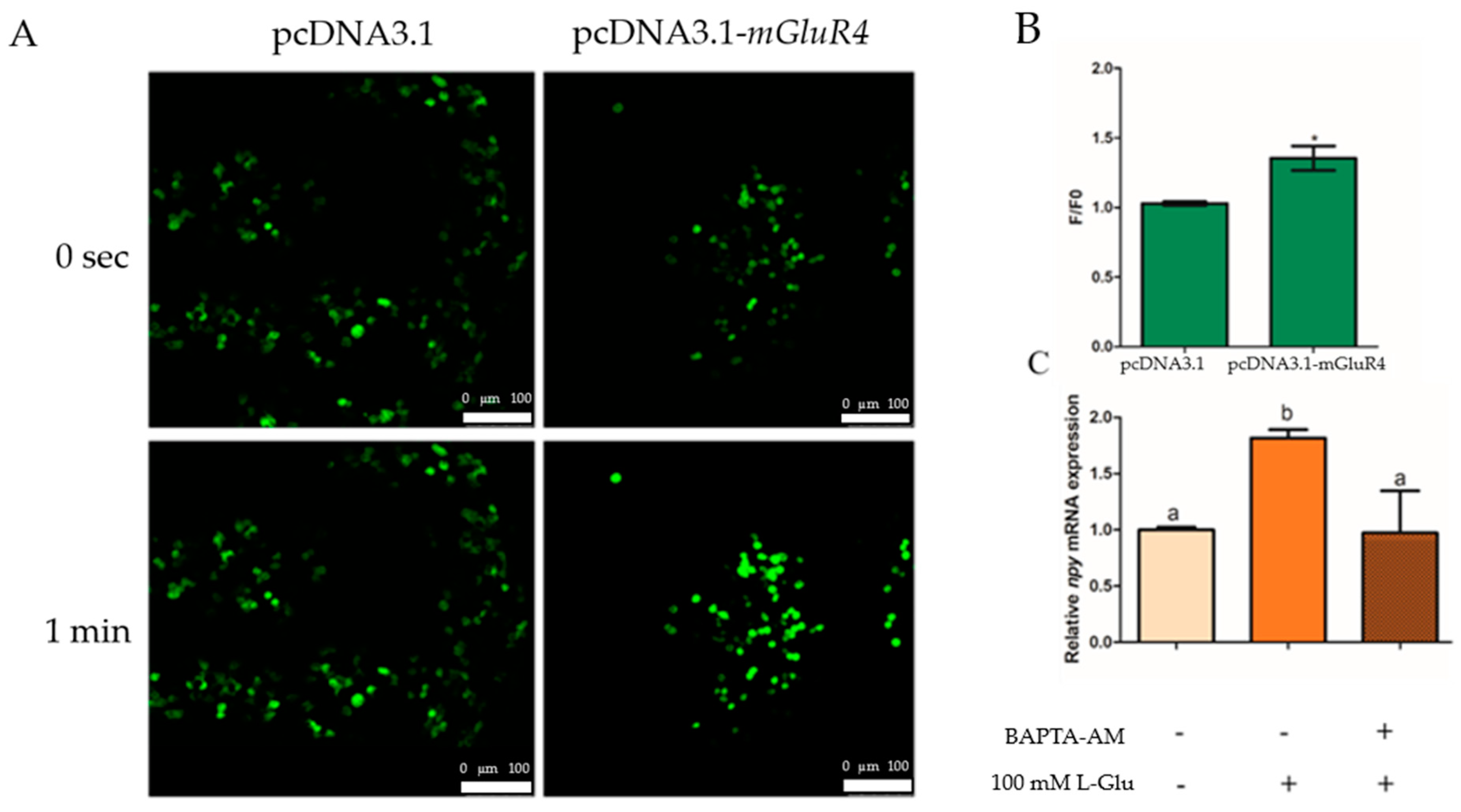
2.2. L-Glutamate Regulates Npy via mGluR4 in Mandarin Fish Brain Cells
2.3. Ca2+ Involvement in L-Glutamate Regulating Npy through mGluR4 in Mandarin Fish Brain Cells
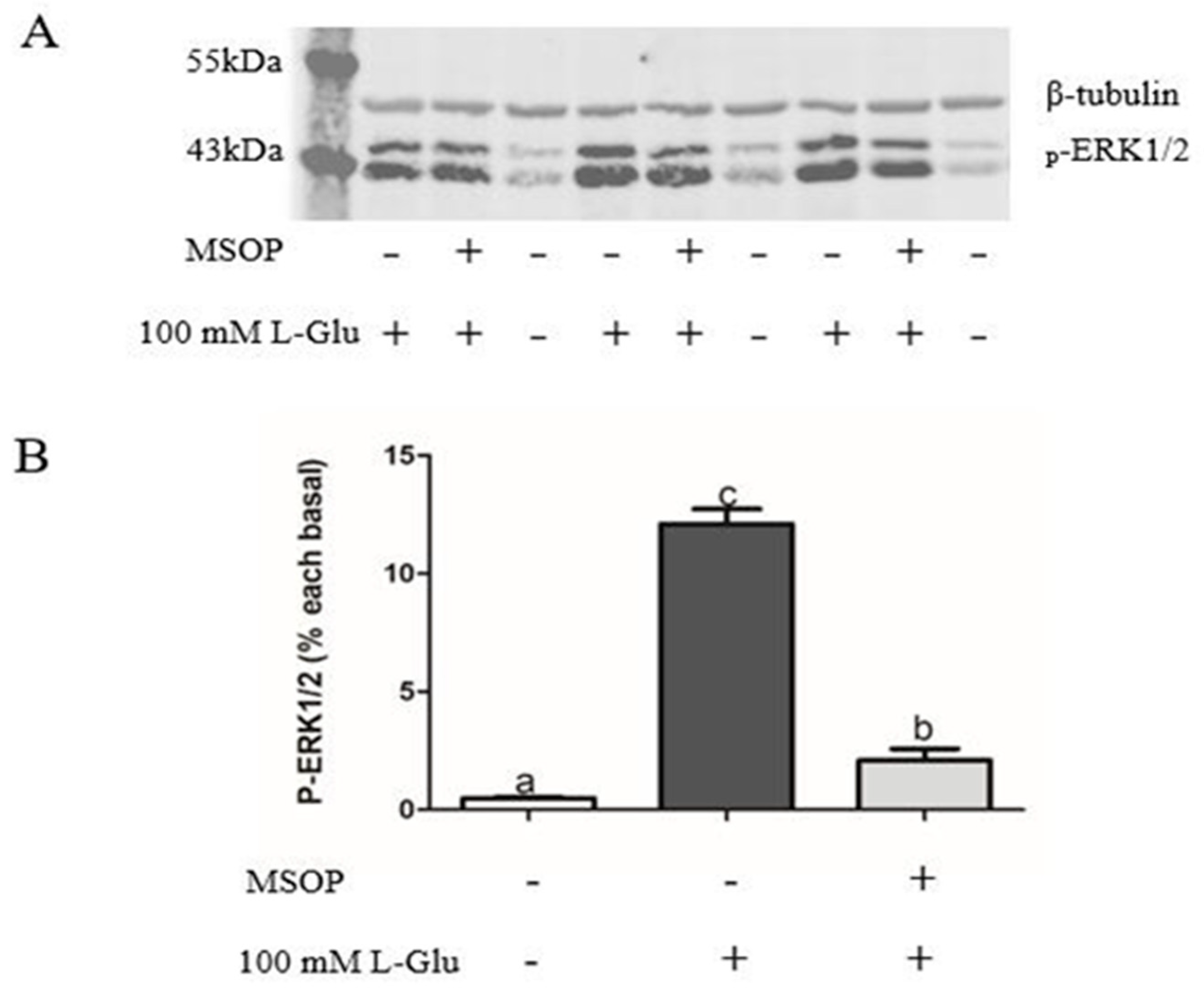
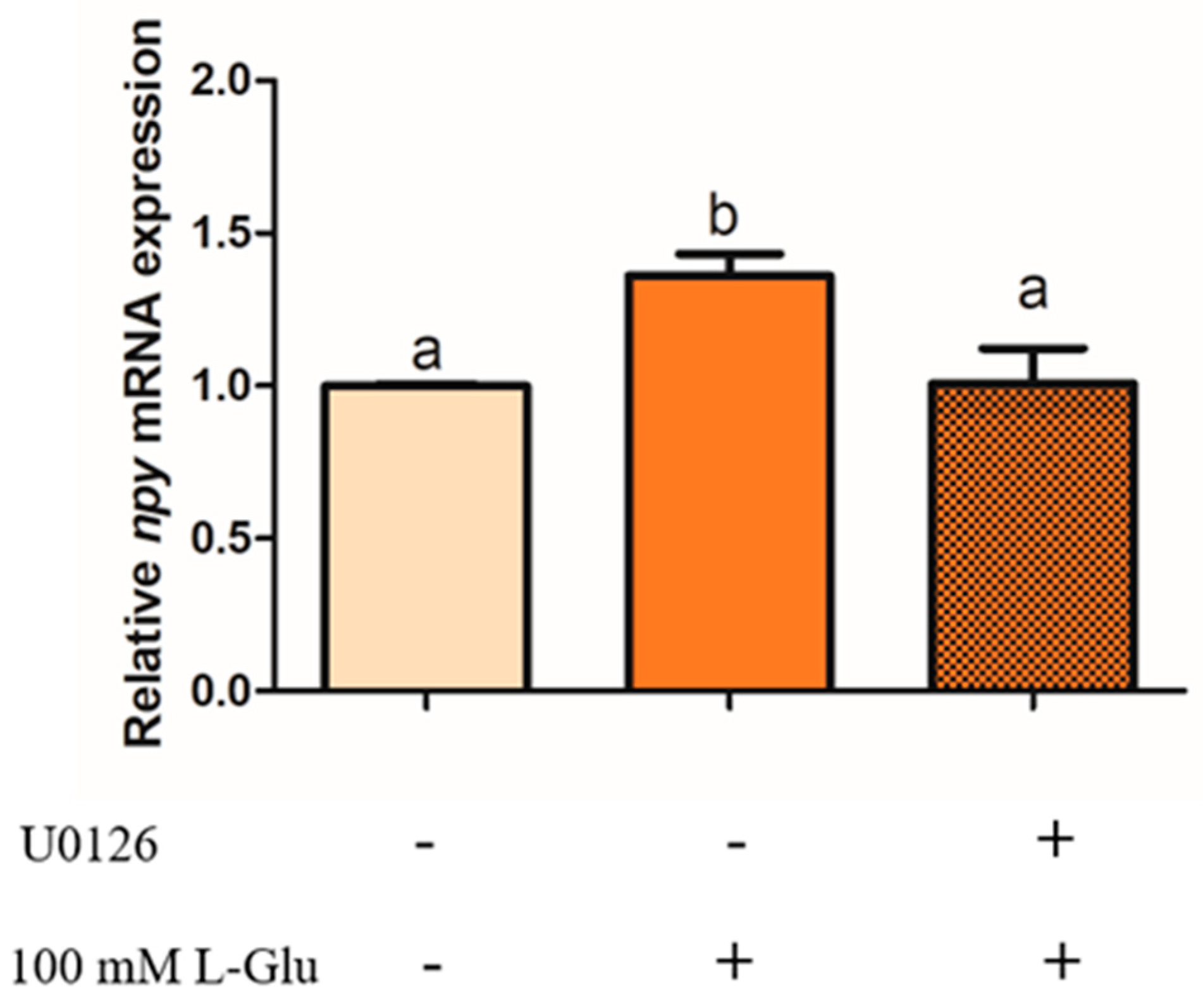
2.4. ERK1/2 Involvement in L-Glutamate Regulating Npy through mGluR4 in Mandarin Fish Brain Cells
3. Discussion
4. Materials and Methods
4.1. mGluR4 Sequence Analysis
4.2. Fish and Sample Collection
4.3. Mandarin Fish Brain Cell Culture and Treatment
4.4. Cell Counting Kit-8 (CCK-8)
4.5. Cloning mGluR4 Gene Sequences of Mandarin Fish
4.6. mGluR4 Functional Expression
4.7. Calcium Assays
4.8. RT-PCR
4.9. Western Blot
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Behrens, M.; Briand, L.; de March, C.A.; Matsunami, H.; Yamashita, A.; Meyerhof, W.; Weyand, S. Structure–Function Relationships of Olfactory and Taste Receptors. Chem. Senses 2018, 43, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Chandrashekar, J.; Hoon, M.A.; Ryba, N.J.P.; Zuker, C.S. The receptors and cells for mammalian taste. Nature 2006, 444, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Nei, M.; Niimura, Y.; Nozawa, M. The evolution of animal chemosensory receptor gene repertoires: Roles of chance and necessity. Nat. Rev. Genet. 2008, 9, 951–963. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Huang, Y.; Jiang, S.; Chen, G.; Liu, Y. Research on sensing characteristics of three human umami receptors via receptor-based biosensor. Flavour Fragr. J. 2020, 35, 695–702. [Google Scholar] [CrossRef]
- Kurihara, K. Umami the Fifth Basic Taste: History of Studies on Receptor Mechanisms and Role as a Food Flavor. BioMed Res. Int. 2015, 2015, 1–10. [Google Scholar] [CrossRef]
- Yasumatsu, K.; Ogiwara, Y.; Takai, S.; Yoshida, R.; Iwatsuki, K.; Torii, K.; Margolskee, R.F.; Ninomiya, Y. Umami taste in mice uses multiple receptors and transduction pathways. J. Physiol. 2012, 590, 1155–1170. [Google Scholar] [CrossRef]
- Chaudhari, N.; Yang, H.; Lamp, C.; Delay, E.; Cartford, C.; Than, T.; Roper, S. The taste of monosodium glutamate: Membrane receptors in taste buds. J. Neurosci. 1996, 16, 3817–3826. [Google Scholar] [CrossRef]
- Pal Choudhuri, S.; Delay, R.J.; Delay, E.R. Metabotropic glutamate receptors are involved in the detection of IMP and l-amino acids by mouse taste sensory cells. Neuroscience 2016, 316, 94–108. [Google Scholar] [CrossRef]
- Chavis, P.; Fagni, L.; Lansman, J.B.; Bockaert, J. Functional coupling between ryanodine receptors and L-type calcium channels in neurons. Nature 1996, 382, 719–722. [Google Scholar] [CrossRef]
- Toyono, T.; Seta, Y.; Kataoka, S.; Harada, H.; Morotomi, T.; Kawano, S.; Shigemoto, R.; Toyoshima, K. Expression of the metabotropic glutamate receptor, mGluR4a, in the taste hairs of taste buds in rat gustatory papillae. Arch. Histol. Cytol. 2002, 65, 91–96. [Google Scholar] [CrossRef]
- Yasumatsu, K.; Manabe, T.; Yoshida, R.; Iwatsuki, K.; Uneyama, H.; Takahashi, I.; Ninomiya, Y. Involvement of multiple taste receptors in umami taste: Analysis of gustatory nerve responses in metabotropic glutamate receptor 4 knockout mice. J. Physiol. 2015, 593, 1021–1034. [Google Scholar] [CrossRef] [PubMed]
- Finger, T.E.; Kinnamon, S.C. Taste isn’t just for taste buds anymore. F1000 Biol. Rep. 2011, 3, 20. [Google Scholar] [CrossRef] [PubMed]
- Risso Bradley, S.; Standaert, D.G.; Levey, A.I.; Conn, P.J. Distribution of Group III mGluRs in Rat Basal Ganglia with Subtype-Specific Antibodies. Ann. N. Y. Acad. Sci. 2006, 868, 531–534. [Google Scholar] [CrossRef] [PubMed]
- Müller, M.; Ryoo, M.C.-K.; Roura, E. Gut sensing of dietary amino acids, peptides and proteins, and feed-intake regulation in pigs. Anim. Prod. Sci. 2021, 62, 1147–1159. [Google Scholar] [CrossRef]
- Yao, J.-T.; Kong, C.; Hua, X.-M.; Yang, J.-F.; Liu, T.; Wang, G.; Shui, C.; Feng, Y.; Shi, Y.-H. T1R1 expression in obscure puffer (Takifugu fasciatus) is associated with effect of dietary soybean antigenic protein on intestinal health. Aquaculture 2019, 501, 202–212. [Google Scholar] [CrossRef]
- Kondoh, T.; Mallick, H.N.; Torii, K. Activation of the gut-brain axis by dietary glutamate and physiologic significance in energy homeostasis. Am. J. Clin. Nutr. 2009, 90, 832S–837S. [Google Scholar] [CrossRef]
- Yang, Z.; Huang, R.; Fu, X.; Wang, G.; Qi, W.; Mao, D.; Shi, Z.; Shen, W.L.; Wang, L. A post-ingestive amino acid sensor promotes food consumption in Drosophila. Cell Res. 2018, 28, 1013–1025. [Google Scholar] [CrossRef]
- Volkoff, H.; Canosa, L.F.; Unniappan, S.; Cerdá-Reverter, J.M.; Bernier, N.J.; Kelly, S.P.; Peter, R.E. Neuropeptides and the control of food intake in fish. Gen. Comp. Endocrinol. 2005, 142, 3–19. [Google Scholar] [CrossRef]
- Narnaware, Y.K.; Peter, R.E. Neuropeptide Y stimulates food consumption through multiple receptors in goldfish. Physiol. Behav. 2001, 74, 185–190. [Google Scholar] [CrossRef]
- Yokobori, E.; Azuma, M.; Nishiguchi, R.; Kang, K.S.; Kamijo, M.; Uchiyama, M.; Matsuda, K. Neuropeptide Y Stimulates Food Intake in the Zebrafish, Danio rerio. J. Neuroendocrinol. 2012, 24, 766–773. [Google Scholar] [CrossRef]
- Alhosaini, K.; Bahattab, O.; Qassam, H.; Challiss, R.A.J.; Willars, G.B. Ligand-Specific Signaling Profiles and Resensitization Mechanisms of the Neuromedin U2 Receptor. Mol. Pharmacol. 2018, 94, 674–688. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Ortega, A.J.; Buendia, I.; Mouhid, L.; Egea, J.; Lucea, S.; Ruiz-Nuño, A.; López, M.G.; Cano-Abad, M.F. CALHM1 and its polymorphism P86L differentially control Ca2+ homeostasis, mitogen-activated protein kinase signaling, and cell vulnerability upon exposure to amyloid β. Aging Cell 2015, 14, 1094–1102. [Google Scholar] [CrossRef] [PubMed]
- Fang, D.D.; Liu, Y.; Zhang, B.; Wen, X.; Zhang, S.; Wei, Y.; Lu, Q.; Liu, Z.; Wang, K.; Liu, F.; et al. Construction and characterization of a bacterial artificial chromosome library for Gossypium mustelinum. PLoS ONE 2018, 13, e0196847. [Google Scholar]
- Sato, J.J.; Wolsan, M. Loss or major reduction of umami taste sensation in pinnipeds. Naturwissenschaften 2012, 99, 655–659. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Yang, J.R.; Xu, H.; Zhang, J. Pseudogenization of the Umami Taste Receptor Gene Tas1r1 in the Giant Panda Coincided with its Dietary Switch to Bamboo. Mol. Biol. Evol. 2010, 27, 2669–2673. [Google Scholar] [CrossRef]
- Wolsan, M.; Sato, J.J. Role of feeding specialization in taste receptor loss: Insights from sweet and umami receptor evolution in Carnivora. Chem. Senses 2022, 47, bjac033. [Google Scholar] [CrossRef]
- Nunez-Salces, M.; Li, H.; Feinle-Bisset, C.; Young, R.L.; Page, A.J. Nutrient-sensing components of the mouse stomach and the gastric ghrelin cell. Neurogastroenterol. Motil. 2020, 32, e13944. [Google Scholar] [CrossRef]
- Roura, E.; Foster, S.R. Nutrient-Sensing Biology in Mammals and Birds. Annu. Rev. Anim. Biosci. 2018, 6, 197–225. [Google Scholar] [CrossRef]
- Tirassa, P.; Lundeberg, T.; Stenfors, C.; BracciLaudiero, L.; Theodorsson, E.; Aloe, L. Monosodium glutamate increases NGF and NPY concentrations in rat hypothalamus and pituitary. Neuroreport 1995, 6, 2450–2452. [Google Scholar] [CrossRef]
- Oehme, M.; Grammes, F.; Takle, H.; Zambonino-Infante, J.-L.; Refstie, S.; Thomassen, M.S.; Rørvik, K.-A.; Terjesen, B.F. Dietary supplementation of glutamate and arginine to Atlantic salmon (Salmo salar L.) increases growth during the first autumn in sea. Aquaculture 2010, 310, 156–163. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, T.-R.; Li, Q.; Feng, L.; Liu, Y.; Jiang, W.-D.; Wu, P.; Zhao, J.; Zhou, X.-Q.; Jiang, J. Effect of dietary L-glutamate levels on growth, digestive and absorptive capability, and intestinal physical barrier function in Jian carp (Cyprinus carpio var. Jian). Anim. Nutr. 2020, 6, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.B.; Khondowe, P.; Chen, S.F.; Yu, J.J.; Shu, G.; Zhu, X.T.; Wang, L.N.; Gao, P.; Xi, Q.Y.; Zhang, Y.L.; et al. Effects of “Bioactive” amino acids leucine, glutamate, arginine and tryptophan on feed intake and mRNA expression of relative neuropeptides in broiler chicks. J. Anim. Sci. Biotechnol. 2012, 3, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.-H.; Lin, M.; Zhang, B.; Yu, C.; Li, J.; Zhang, L.; Sun, H.; Gao, F.; Zhou, G. L-Glutamate Supplementation Improves Small Intestinal Architecture and Enhances the Expressions of Jejunal Mucosa Amino Acid Receptors and Transporters in Weaning Piglets. PLoS ONE 2014, 9, e111950. [Google Scholar]
- Cosgrove, K.E.; Meriney, S.D.; Barrionuevo, G. High affinity group III mGluRs regulate mossy fiber input to CA3 interneurons. Hippocampus 2010, 21, 1302–1317. [Google Scholar] [CrossRef] [PubMed]
- Wieroñska, J.M.; Szewczyk, B.; Palucha, A.; Brañski, P.; Ziêba, B.; Smialowska, M. Anxiolytic action of group II and III metabotropic glutamate receptors agonists involves neuropeptide Y in the amygdala. Pharmacol. Rep. 2005, 57, 734–743. [Google Scholar]
- Matsunami, H.; Iguchi, N.; Ohkuri, T.; Slack, J.P.; Zhong, P.; Huang, L. Sarco/Endoplasmic Reticulum Ca2+-ATPases (SERCA) Contribute to GPCR-Mediated Taste Perception. PLoS ONE 2011, 6, e23165. [Google Scholar]
- Wauson, E.M.; Zaganjor, E.; Lee, A.Y.; Guerra, M.L.; Ghosh, A.B.; Bookout, A.L.; Chambers, C.P.; Jivan, A.; McGlynn, K.; Hutchison, M.R.; et al. The G Protein-Coupled Taste Receptor T1R1/T1R3 Regulates mTORC1 and Autophagy. Mol. Cell 2012, 47, 851–862. [Google Scholar] [CrossRef]
- Zhou, Y.; Ren, J.; Song, T.; Peng, J.; Wei, H. Methionine Regulates mTORC1 via the T1R1/T1R3-PLCβ-Ca2+-ERK1/2 Signal Transduction Process in C2C12 Cells. Int. J. Mol. Sci. 2016, 17, 1684. [Google Scholar] [CrossRef]
- Tian, Y.; Jiang, C.; Pan, Y.; Guo, Z.; Wang, W.; Luo, X.; Cao, Z.; Zhang, B.; Yang, J.; Shi, Y.; et al. Bombyx neuropeptide G protein-coupled receptor A14 and A15 are two functional G protein-coupled receptors for CCHamide neuropeptides. Insect Biochem. Mol. Biol. 2021, 131, 103553. [Google Scholar] [CrossRef]
- Gemignani, A.; Marchese, S.; Fontana, G.; Raiteri, M. Neuropeptide Y release from cultured hippocampal neurons: Timulation by glutamate acting at n-methyl-d-aspartate and AMPA receptors. Neuroscience 1997, 81, 23–31. [Google Scholar] [CrossRef]
- Raman, M.; Chen, W.; Cobb, M.H. Differential regulation and properties of MAPKs. Oncogene 2007, 26, 3100–3112. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Gu, Z.L.; Zhang, G.Y.; Jing, G.Z. Diphosphorylation and involvement of extracellular signal-regulated kinases (ERK1/2) in glutamate-induced apoptotic-like death in cultured rat cortical neurons. Brain Res. 2000, 857, 71–77. [Google Scholar] [CrossRef] [PubMed]
- You, J.; Reilly, G.C.; Zhen, X.; Yellowley, C.E.; Chen, Q.; Donahue, H.J.; Jacobs, C.R. Osteopontin Gene Regulation by Oscillatory Fluid Flow via Intracellular Calcium Mobilization and Activation of Mitogen-activated Protein Kinase in MC3T3–E1 Osteoblasts. J. Biol. Chem. 2001, 276, 13365–13371. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.E.; Mouradian, M.M. Cytoprotective mechanisms of DJ-1 against oxidative stress through modulating ERK1/2 and ASK1 signal transduction. Redox Biol. 2018, 14, 211–217. [Google Scholar] [CrossRef]
- Duan, Z.; Zhang, X.; Zhu, G.; Gao, Y.; Xue, X. Activation of mGluR4 promotes proliferation of rat neural progenitor cells while mediating activation of ERK1/2 signaling pathway. Cell. Mol. Biol. 2013, 59, 1809–1817. [Google Scholar]
- Barnea, A.; Roberts, J.; Croll, S.D. Continuous exposure to brain-derived neurotrophic factor is required for persistent activation of TrkB receptor, the ERK signaling pathway, and the induction of neuropeptide Y production in cortical cultures. Brain Res. 2004, 1020, 106–117. [Google Scholar] [CrossRef]
- Olsson, A.-K.; Nånberg, E. A Functional Role for ERK in Gene Induction, but Not in Neurite Outgrowth in Differentiating Neuroblastoma Cells. Exp. Cell Res. 2001, 265, 21–30. [Google Scholar] [CrossRef]
- Barnea, A.; Roberts, J. Induction of functional and morphological expression of neuropeptide Y (NPY) in cortical cultures by brain-derived neurotrophic factor (BDNF): Evidence for a requirement for extracellular-regulated kinase (ERK)-dependent and ERK-independent mechanisms. Brain Res. 2001, 919, 57–69. [Google Scholar] [CrossRef]
- Sutton, G.M.; Patterson, L.M.; Berthoud, H.-R. Extracellular Signal-Regulated Kinase 1/2 Signaling Pathway in Solitary Nucleus Mediates Cholecystokinin-Induced Suppression of Food Intake in Rats. J. Neurosci. 2004, 24, 10240–10247. [Google Scholar] [CrossRef]

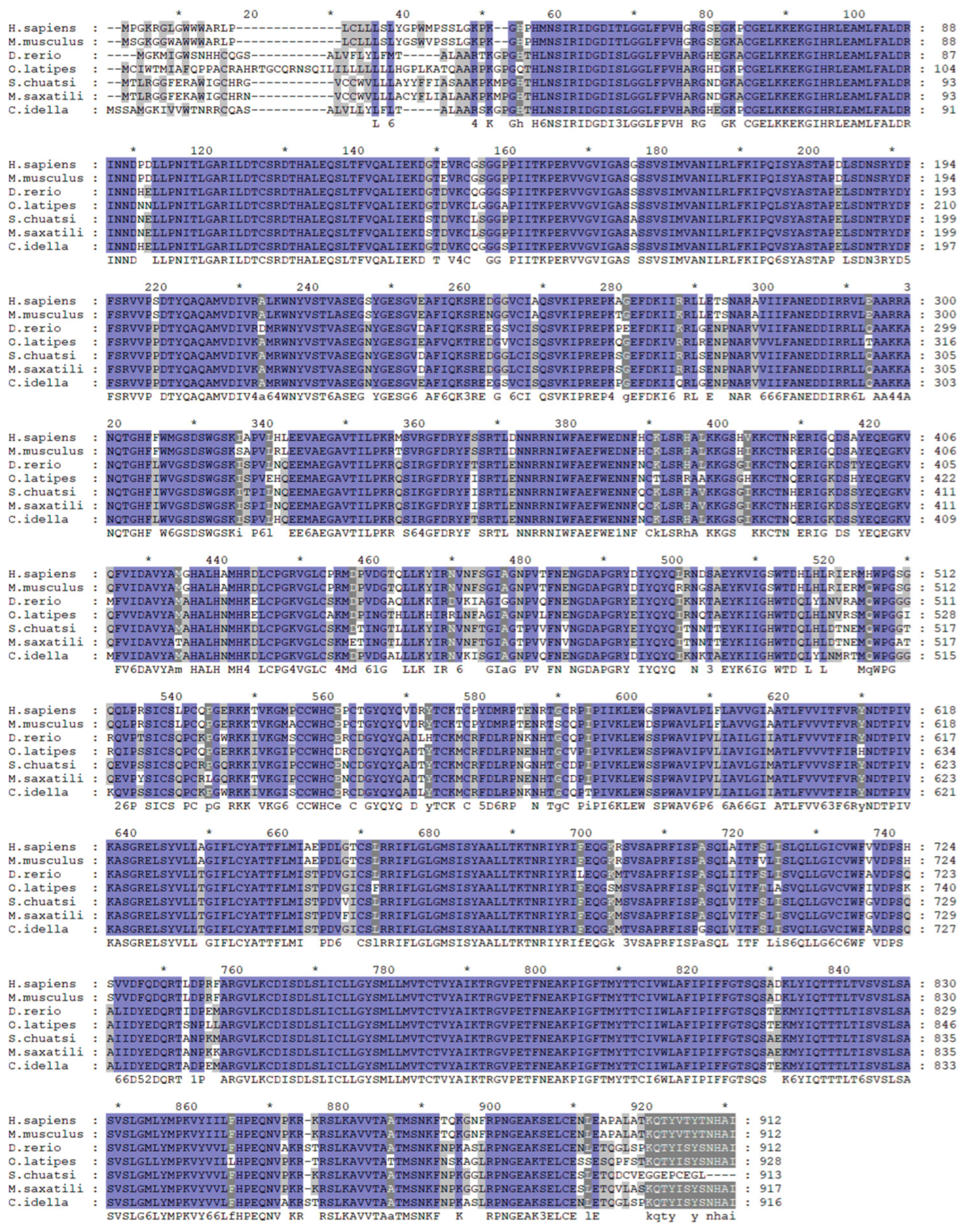
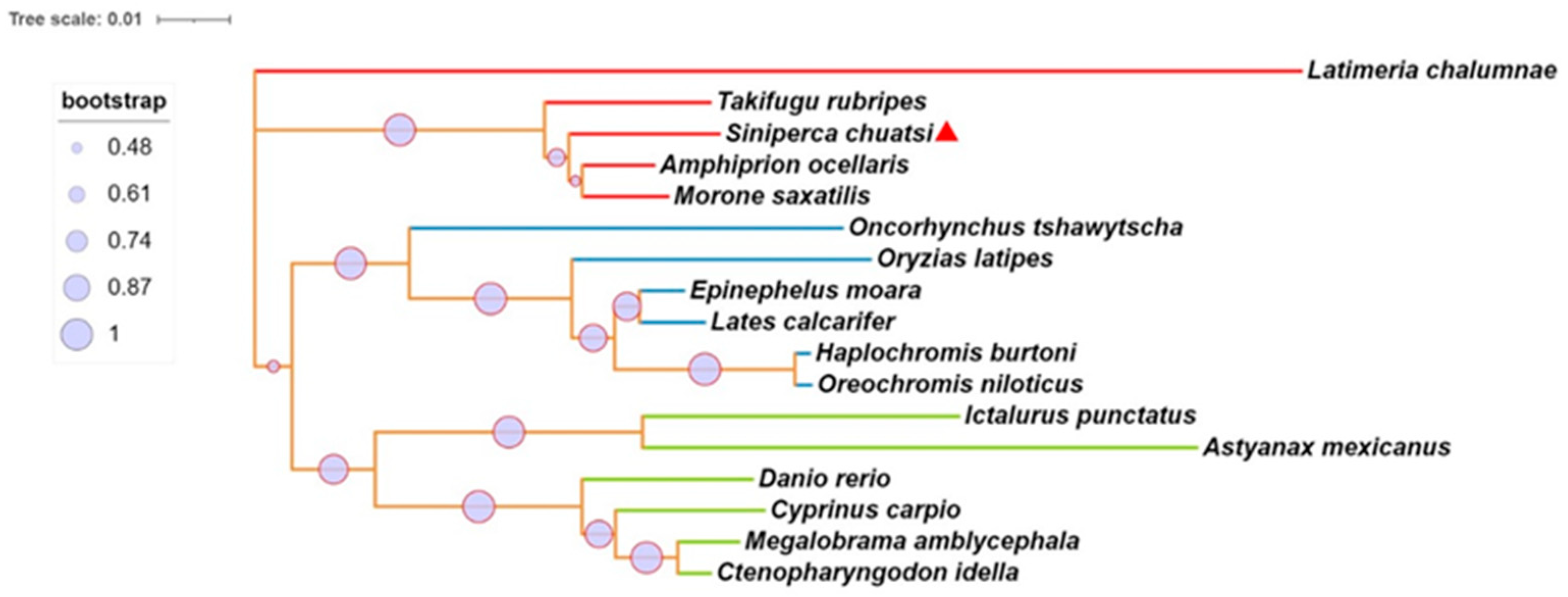

| Species | Siniperca chuatsi (%) |
|---|---|
| Homo sapiens | 79 |
| Mus musculus | 79 |
| Danio rerio | 85 |
| Oryzias latipes | 84 |
| Morone saxatilis | 96 |
| Ctenopharyngodon idella | 86 |
| Gene Name Primer | Sequence of Primer (5′ to 3′) | Tm (°C) |
|---|---|---|
| mGluR4-pcDNA3.1 | F TAGTCCAGTGTGGTGGAATTC ATGACTCTACGAGGAGGTTTTGA R GGTTTAAACGGGCCCTCTAGA TTAGATGGCATGGTTACTGTAGC | 58 |
| Gene Name (%) | Sequence of Primer (5′ to 3′) | Tm (°C) | Amplification Efficiency |
|---|---|---|---|
| rpl13a | F TATCCCCCCACCCTATGACA R ACGCCCAAGGAGAGCGAACT | 58.0 | 104.8 |
| mGluR4 | F GAGTGGTGGGAGTGATTGGTG R GGGAGAAGAAGTCATAGCGGG | 58.0 | 96.4 |
| npy | F GTTGAAGGAAAGCACAGACA R GCTCATAGAGGTAAAAGGGG | 60.0 | 92.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duan, J.; Wang, Q.; He, S.; Liang, X.-F.; Ding, L. L-Glutamate Regulates Npy via the mGluR4-Ca2+-ERK1/2 Signaling Pathway in Mandarin Fish (Siniperca chuatsi). Int. J. Mol. Sci. 2024, 25, 10035. https://doi.org/10.3390/ijms251810035
Duan J, Wang Q, He S, Liang X-F, Ding L. L-Glutamate Regulates Npy via the mGluR4-Ca2+-ERK1/2 Signaling Pathway in Mandarin Fish (Siniperca chuatsi). International Journal of Molecular Sciences. 2024; 25(18):10035. https://doi.org/10.3390/ijms251810035
Chicago/Turabian StyleDuan, Jiahui, Qiuling Wang, Shan He, Xu-Fang Liang, and Liyun Ding. 2024. "L-Glutamate Regulates Npy via the mGluR4-Ca2+-ERK1/2 Signaling Pathway in Mandarin Fish (Siniperca chuatsi)" International Journal of Molecular Sciences 25, no. 18: 10035. https://doi.org/10.3390/ijms251810035







