Catalyst structure
Various methods of analysis, such as
27Al and
29Si MAS NMR, show that “Maghnite” is a montmorillonite sheet silicate clay. The elementary analysis of the selected samples obtained using XRF and conversions obtained from the reaction of 1g of each sample with 10.75mol/l of THF, 5.67 mol/l of phthalic anhydride and 1.28 mol/l of acetic anhydride during 6 hours in a bulk polymerization at 40°C, are as settled in the following
Table 1.
It is necessary to report that the best value of conversion was obtained with “HMaghnite0.25M”, for this reason we kept this sample to study the effect of catalyst and acetic anhydride proportions on polymerization. Acid treatment of “Raw-Maghnite” cause reduction in octahedral content (Al2O3) resulted in an increase in the proportion of silica (SiO2) observed.
Table 1.
Elementary compositions of Protons exchanged samples “H-Maghnite”
Table 1.
Elementary compositions of Protons exchanged samples “H-Maghnite”
| | Compositions wt% | conver-sion % |
|---|
| sample | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | Na2O | K2O | TiO2 | SO3 | As | PF* |
|---|
| Raw-Maghnite | 69.39 | 14.67 | 1.16 | 0.30 | 1.07 | 0.50 | 0.79 | 0.16 | 0.91 | 0.05 | 11.0 | 0.0 |
| H-Mag0.05M | 70.75 | 14.67 | 1.05 | 0.30 | 1.01 | 0.49 | 0.78 | 0.16 | 0.75 | 0.04 | 10.0 | 1.0 |
| H-Mag0.10M | 71.00 | 14.60 | 1.00 | 0.30 | 0.98 | 0.39 | 0.78 | 0.16 | 0.55 | 0.04 | 10.2 | 2.51 |
| H-Mag0.15M | 71.58 | 14.45 | 0.95 | 0.29 | 0.91 | 0.35 | 0.77 | 0.15 | 0.42 | 0.03 | 10.1 | 9.38 |
| H-Mag0.20M | 71.65 | 14.20 | 0.80 | 0.28 | 0.85 | 0.30 | 0.77 | 0.15 | 0.39 | 0.01 | 10.6 | 12.27 |
| H-Mag0.25M | 71.70 | 14.03 | 0.71 | 0.28 | 0.80 | 0.21 | 0.77 | 0.15 | 0.34 | 0.01 | 11.0 | 28.32 |
| H-Mag0.30M | 73.20 | 13.85 | 0.70 | 0.27 | 0.78 | 0.20 | 0.76 | 0.13 | 0.31 | 0.02 | 9.78 | 20.57 |
| H-Mag0.35M | 75.31 | 13.52 | 0.71 | 0.26 | 0.78 | 0.18 | 0.75 | 0.13 | 0.32 | 0.01 | 8.03 | 17.26 |
The X-Ray powder diffraction profiles (
Figure 1 and
Table 2) exhibited the presence of other crystalline phases as quartz, feldspath and calcite in “raw-Maghnit” By treatment acid, all trace of calcite was removed in “H-Maghnite”.
The increase in basal spacing from 12.5 Å in “raw-Maghnite”, characteristic of one water layer between the sheets, to value of 15.02 Å in “H-Maghnite”, where there are two interlamellar water layers, subsequent to acid treatment reflects the change in interlayer cation and its associated hydration state [
6].
Figure 1.
X-ray Power Diffraction of (a) “Raw-Maghnite” and (b) “H-Maghnite 0.25M”
Figure 1.
X-ray Power Diffraction of (a) “Raw-Maghnite” and (b) “H-Maghnite 0.25M”
Table 2.
RX characteristic of Raw-Maghnite and H-Maghnite 0.25M.
Table 2.
RX characteristic of Raw-Maghnite and H-Maghnite 0.25M.
| Samples | dhkl (Å) | hkl | Naure of samples |
|---|
| Raw-Maghnite | 12.50 | 001 | Montmorillonite |
| 4.47 | 110 | Montmorillonite |
| 4.16 | ,, | Quartz |
| 3.35 | ,, | Quartz |
| 3.21 | ,, | Feldspath |
| 3.03 | ,, | Calcite |
| 2.55 | 200 | Montmorillonite |
| 1.68 | 009 | Montmorillonite |
| 1.49 | 060 | Montmorillonite |
| H-Maghnite 0.25M | 15.02 | 001 | Montmorillonite |
| 4.47 | 110 | Montmorillonite |
| 4.16 | ,, | Quartz |
| 3.35 | ,, | Quartz |
| 3.21 | ,, | Feldspath |
| 3.03 | ,, | Calcite |
| 2.55 | 200 | Montmorillonite |
| 1.68 | 009 | Montmorillonite |
| 1.49 | 060 | Montmorillonite |
The IR Spectra of Raw-Maghnite and H-Maghnite 0.25M (
Fig. 2) are in good agrement with literature [
7]. The characteristic vibrations of hydroxy groups, the silicate anion and the octahedral cations were observed in the IR spectra of both montmorillonites. The most intense band near 1040 Cm-1 is attributed to the Si-O stretching vibrations of the tetrahedral layer and the bands near 522 and 466 Cm-1 are assigned to Si-O bending vibrations of smectites. A low intensity band near 800 Cm-1, assigned to four-coordinated silica [
8], indicates the presence of amorphous silica. Comparison of Raw-Maghnite and H-Maghnite 0.25M spectra show a small increasing in the SiO
2 band intensity near 800 Cm-1 in H-Maghnite 0.25M, reflects alterations in the amount of amorphous silica [
9].
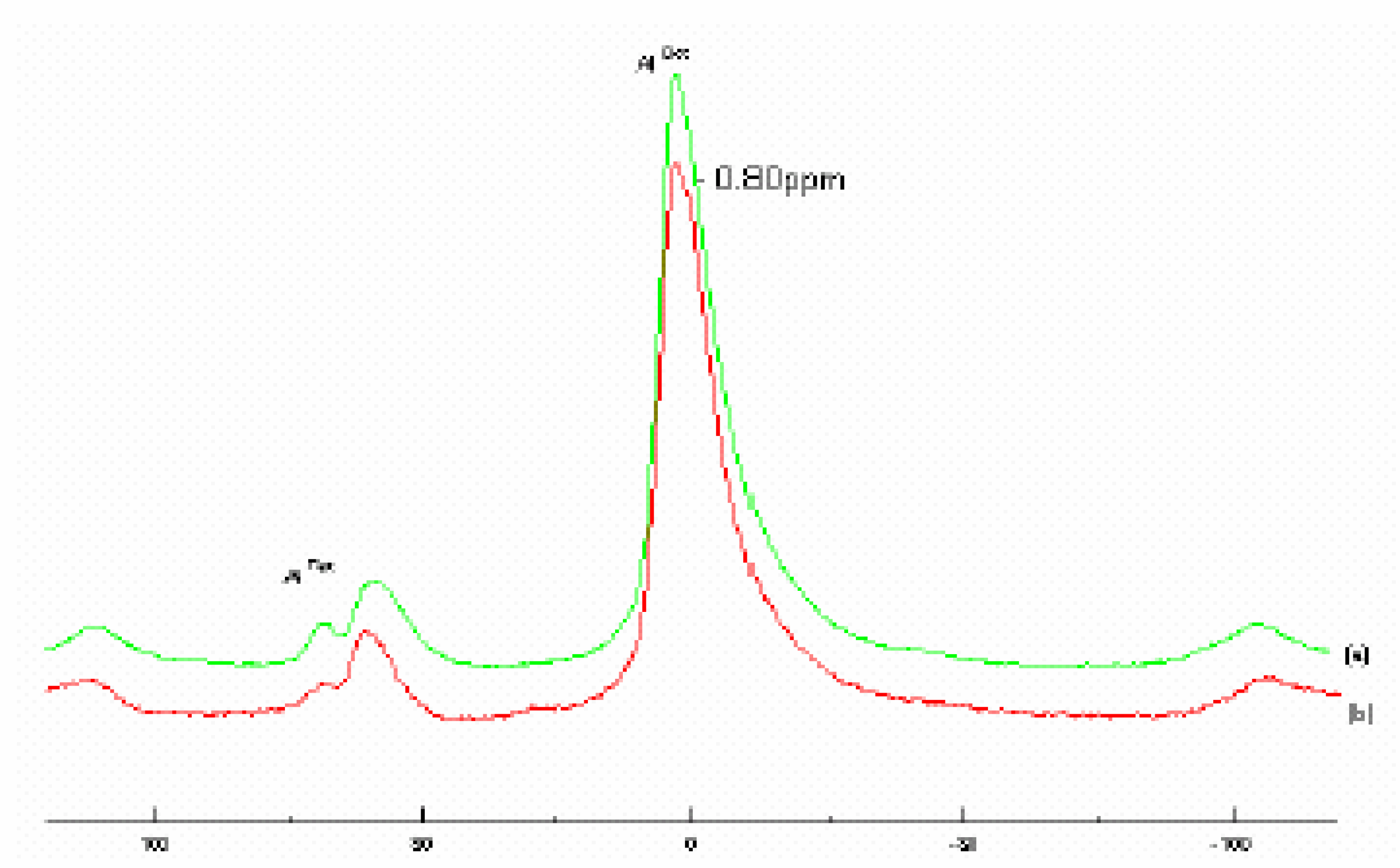
The
27Al MAS NMR spectra of both Raw–Maghnite and H-Maghnite 0.25M (
Fig. 3) show Aluminium in two tetrahedral environments with resonances centered at 60 and 68 ppm, in addition to the main contribution from the octahedral aluminium at 2.9 ppm. These values and assignments concur with those published in literature [
6,
10,
11].
The
29Si MAS NMR spectra for the Raw-Maghnite and H-Maghnite 0.25M are shown in
Fig. 4. The dominant resonance at – 93.5 ppm corresponds to Q
3(OAl) units, i.e SiO
4 groups crosslinked in the tetrahedral sheets with no aluminium in the neighbouring tetrahedral [
12]. The resonance at –112 ppm corresponds to three-dimensional (3D) silica with no aluminium present, designed Q
4 (OAl) [
6,
13].
Figure 2.
IR Spectra of (a) untreated Clay “Raw-Maghnite” and (b) Acid treated Clay “H-Maghnite0.25M”
Figure 2.
IR Spectra of (a) untreated Clay “Raw-Maghnite” and (b) Acid treated Clay “H-Maghnite0.25M”
Figure 3.
27Al MAS NMR spectra of (a) “Raw-Maghnite” and (b) “H-Maghnite 0.25M”
Figure 3.
27Al MAS NMR spectra of (a) “Raw-Maghnite” and (b) “H-Maghnite 0.25M”
Figure 4.
29Si MAS NMR spectra of (a) “Raw-Maghnite” and (b) “H-Maghnite 0.25M”
Figure 4.
29Si MAS NMR spectra of (a) “Raw-Maghnite” and (b) “H-Maghnite 0.25M”
Effect of “H-Maghnite 0.25M” proportion
We can see from
Table 3 that, the conversion increases with increasing “H-Maghnite 0.25 M” proportion (experiments 1,2). This lead was followed up by a serie of deeper experiments in which the conversion with the time has been observed at various amounts of catalyst.
Table 4 and
Figure 5 show that increasing the “H-Maghnite 0.25M” amounts yielded higher conversions. This phenomena is probably the result of number of “initiating active sites” responsible of inducing polymerization, this number is prorata to the catalyst amount used in reaction.
Table 4.
Conversions with time for 10.75 mol/l of THF, 5.67 mol/l of phthalic anhydride, 0.19 mol/l of acetic anhydride and the amounts of H-Maghnite 0.25M were: a) 2.5% , b)5% , c)10%
Table 4.
Conversions with time for 10.75 mol/l of THF, 5.67 mol/l of phthalic anhydride, 0.19 mol/l of acetic anhydride and the amounts of H-Maghnite 0.25M were: a) 2.5% , b)5% , c)10%
| Yield(%) | Time(hours) |
|---|
| 1 | 2 | 3 | 4 | 5 | 6 |
|---|
| a | 0.81 | 1.12 | 2.34 | 3.42 | 6.98 | 8.25 |
| b | 2.93 | 3.15 | 5.08 | 6.72 | 13.24 | 18.15 |
| c | 3.46 | 4.85 | 9.14 | 12.67 | 19.42 | 28.32 |
Figure 5.
Effect of catalyst proportion upon the conversion.
Figure 5.
Effect of catalyst proportion upon the conversion.
Effect of acetic anhydride proportion
The addition of acetic anhydride to the mixture creates ends of chains unable to take part in polyesterification, and consequently the problem of reproducibility of molecular weight does not exist, which will allow to have a stable polymer.
Data in
Table 3 show that the conversions increase with increasing acetic anhydride proportion (Experiments 2, 3). This result is due to multiplication of active centers. However the molecular weight increase with reduction acetic anhydride proportion .This result is due to increasing of methyl groups in extremities of chains who block chains of polymer in growth.
This lead was followed up by a series of deeper experiments in which the conversion with the time has been observed at two amounts of acetic anhydride.
Table 5 and
figure 6 show that increasing the acetic anhydride amounts yielded higher conversions.
Table 5.
Conversions with time for 10.75 mol/l of THF, 5.67 mol/l of phthalic anhydride, 2.5% of H-Maghnite 0.25M and the amounts of acetic anhydride were: a) 0.19 mol/l, b) 1.28 mol/l.
Table 5.
Conversions with time for 10.75 mol/l of THF, 5.67 mol/l of phthalic anhydride, 2.5% of H-Maghnite 0.25M and the amounts of acetic anhydride were: a) 0.19 mol/l, b) 1.28 mol/l.
| Yield(%) | Time |
|---|
| 1 | 2 | 3 | 4 | 5 | 6 |
|---|
| | 0.81 | 1.12 | 2.34 | 3.42 | 6.98 | 8.25 |
| | 2.85 | 3.92 | 5.17 | 9.76 | 13.12 | 17.23 |
Figure 6.
Effect of acetic anhydride proportion upon the conversion.
Figure 6.
Effect of acetic anhydride proportion upon the conversion.
Mechanism of the reaction
Polycondensations proceed by successive reactions between the functional groups of the reagents.According to the foregoing discussion and the results of product analysis, we may suggest the mechanism below for the resulting reaction of polymerization induced by “H-Maghnite 0.25M”.
Protons carried by montmorillonite sheets of “H-Maghnite 0.25M” induced the polymerization, these montmorillonite sheets take place as counter-anions.
The first stage is the protonation of acetic anhydride.
Then a molecule of THF attacks in a nucleophilic way protonic acetic anhydride.
The formed ions oxonium take place in the vicinity of the counter-anion carried by montmorillonite sheets, and then there is a nucleophilic attack of the oxygen of phthalic anhydride on the carbon located in alpha of oxygen charge carrier positive of the chains in growth.
The next stage is a nucleophilic attack of the oxygen of the THF on carbon-cation of the chains in growth.
Then we have successive additions of the phtalic anhydride suivue of the THF
![Ijms 04 00312 i007]()
The presence of the groupments acetate at the two ends of the chain was clearly identified by 1H NMR. Therefore, the last stage will be a nucleophilic attack of the oxygen of the acetic acid formed in the first stage which results from the protonation of acetic anhydride on the carbon located in alpha of oxygen charge carrier positive of the chains in growth.