1. Introduction
Species of the genus
Chlamydomonas Ehrenberg 1833 are an important component of the phytoplanktonic diversity in forest lakes in Sweden [
1]. They usually bloom in April–May [
2] and are observed in lower abundance during cooler and darker periods of the year [
1,
2]. Long-term monitoring provides a rich database of the variation of environmental parameters and organismic diversity and abundances (e.g., environmental databases from Länsstyrelsen and from Swedish Universities). It is of the utmost importance to investigate the biodiversity, organism dispersal, and the invasion of new species.
However, certain analyses remain quite constrained both spatially and taxonomically. On one hand, intensive monitoring provides meticulous descriptions of new taxa [
3], phytoplanktonic diversity, and trends [
4,
5] in few targeted lakes, often taking a life-time to achieve [
6]. On the other hand, large-scale monitoring using intensive sampling often lacks a description at the species level due to technical limitations (e.g., time allocated, expertise of the investigator for each taxon, and standardization of the monitoring method using preservative agents that impede the observation of organismal movement). For instance, a closer look at the Chlorophyceae does not grasp all the taxonomic levels of
Chlamydomonas-like species often reported as Chlamydomonadales,
Chlamydomonas sp., or green algae/dots (e.g.,
http://miljodata.slu.se/mvm/ (accessed on 4 May 2022); [
7,
8]). Deep organismic taxonomy requires diverse microscopic observations and a comparison with taxonomic keys, which is a time-consuming process that often-requires experts. Alternatively, genetic barcoding is employed to assess the diversity present in environmental samples or to rapidly identify organisms of interest. However, the technique remains limited by the number, diversity, and quality of available sequences in genetic databases [
9]. A solution is the combination of morphological and phylogenetic analyses [
10]. In addition, for most of the algal lineages, no universal barcode marker has been proposed.
Chlamydomonas is known to be a large polyphyletic genus comprising hundreds of species collected from different environments [
11]. Most of these have been described morphologically [
11,
12] and some have been characterized genetically (e.g., [
13,
14,
15] and see references therein). The genus was revised several times with the recognition of at least eight major monophyletic lineages [
13]. More recently, a study of the
Moewusii clade (
Moewusinia sensu Nakada et al. [
14]) showed substructures composed of five distinct lineages sustained by phenotypic traits and molecular phylogenetic analyses [
16]. It also reported a few unresolved species originally assigned as
Chlamydomonas, of which strains either occurred singly or regrouped within define lineages [
16].
The particularities of the novel species investigated here are its rather small body size, its peculiar whole-body pendulum motion, and its capacity to co-occur and interact with the potentially harmful Microcystis species. Due to their distinctive morphological traits, the species can be spotted quickly in live samples. Strains were collected in eutrophic lakes in which Chlamydomonas sp. were previously reported (miljodata.slu.se; 2018) but without being described. Moreover, its occurrence is of economic relevance as it is able to live in neutral-basic pH waters, commonly used for recreational purposes. In the present study, the morphology, cell ultrastructure, pendulum motion, and taxonomy of this taxon were investigated using diverse microscopic observations and phylogenetic analyses. In light of the results, a new name is proposed to characterize a novel genus within Moewusinia clade: Limnomonas gen. nov. with its type species L. gaiensis and L. spitsbergensis.
2. Materials and Methods
Thirty single cells of a small and ellipsoidal shaped
Chlamydomonas were isolated to established cultures from three eutrophic lakes in Sweden (
Table 1), i.e., 18 strains from Lake Ryssbysjön, 7 strains from Lake Skedviken, and 5 strains from Lake Västra Ringsjön. Their morphology was compared with the commonly studied round and larger
Chlamydomonas species known in Swedish lakes [
3]. Moreover, the peculiar whole-body pendulum motion caught our attention. Single-cell isolation was performed under an Olympus light microscope (CKX31SF, Olympus) using a micro-capillary glass pipette [
17]. Each individual was washed several times in clean droplets of MWC medium [
18], transferred into a volume of 200 µL of MWC medium, and grown at 20 °C under a light regime 12:12 light:dark with 30 µmol photons m
−2 s
−1. Environmental parameters from their natural habitat, such as average water temperature and pH, were retrieved from the Swedish University of Agricultural Science’s miljödatabas för mark-vatten-miljö (
http://miljodata.slu.se/mvm/ (accessed on 4 May 2022)) and Bergman [
19].
Additionally, Chlamydomonas strains were acquired from culture collections for morphological comparison. The freshwater strain CCAP 11/119 isolated from Blelham Tarn, Cumbria, England was ordered from the Culture Collection of Algae and Protozoa. The strain CCCryo 217-05 isolated from a persistent snow field in Doktorbreen, Nathorst Land, Spitsbergen, Svalbard, Norway was ordered from the Culture Collection of Cryophilic Algae.
The external morphology and the cell ultrastructure were investigated in four of the 30 established strains (VR66-07, VR66-10, R86-45, and R86-47). These strains were deposited at the Culture Collection of Algae (SAG), University of Göttingen, Germany, under the numbers SAG 2637, SAG 2636, SAG 2638, and SAG 2639, respectively.
The morphology and the phenotypic plasticity were documented in photographs and compared with the identification keys from Ettl [
11,
12], Ettl and Gärtner [
24], and the meticulous work of Skuja on
Chlamydomonas species in different habitats including Swedish lakes [
3,
4,
5,
6]. The individual pendulum motion and their distribution pattern were assessed under a light microscope (Eclipse Ts2, Nikon) using NIS-Elements BR (version 4.50, Nikon) software for image recording and ImageJ, version 1.51a [
25], for image analyses. The oscillation frequency (in Hertz) of the cell motion was inversely proportional to the number of frames necessary for a complete oscillation movement divided by the number frames per second of the registration (see the results section for details on the movement performed).
Cell ultrastructure was investigated using a transmission electron microscope (TEM) at the Microscopy Facility, Department of Biology, Lund University. A loose pellet of individual cells was fixated in 2% paraformaldehyde and 2.5% glutaraldehyde in 0.1 M cacodylic buffer (pH 7.4) for three hours at 4 °C and post-fixed in 1% osmium tetroxide in distilled water for an hour at 4 °C. The pellet was dehydrated in a graded ethanol series and embedded in epoxy resin (Agar 100, Resin kit, R1031) via acetone and polymerized for 48 h at 60 °C. Ultra-thin sections (50 nm) were cut using an ultramicrotome (EM UC7, Leica, Wetzlar, Germany) using a diamond knife. The sections were mounted on a pioloform-coated copper grid and stained with uranyl acetate (2%, 30 min) and lead citrate (4 min). The ultra-thin sections were examined at 100 kV using a transmission electron microscope (JEM-1400 PLUS, JEOL Ltd., Tokyo, Japan). Micrographs were recorded with a JEOL Matataki CMOS camera using TEM Centre for JEM1400 Plus software.
Complementary morphological information on chloroplast position and shape was inferred using an automated microscope with phase gradient contrast (Zeiss Cell discoverer 7) at the Lund university Bioimaging Center, Lund University. Cells were loaded in a µ-Plate 24-well black ibiTreat (ibidi GmbH, Gräfelfing, Germany) and an equivalent volume of carbonated mineral water was used to sedate the organisms. Chlorophyll autofluorescence signal was used to detect the chloroplast.
Genomic DNA was extracted following the CTAB extraction protocol described in Tesson et al. [
26]. The SSU rDNA of strains VR66-07, VR66-10, R86-45, and R86-47 were amplified by PCR reactions using the protocol described in Genitsaris et al. [
27] with 35 cycles and the primers EukA (5′-AAC CTG GTT GAT CCT GCC AGT-3′) and Euk1633rE (5′-GGG CGG TGT GTA CAA RGRG-3′). The ITS rDNA was amplified using the protocol of Goff and Moon [
28] with 30 cycles and the primers ITS-A (5′-GGG ATC CGT TTC CGT AGG TGA ACC TGC-3′) and ITS-B (5′-GGG ATC CAT ATG CTT AAG TTC AGC GGG-3′) [
29]. All PCR products were purified and sequenced as described by Lebret et al. [
30]. To identify which taxa are closely related to the isolated strains, we used the SSU rDNA sequences (ON124833-ON124836) and the ITS rDNA sequences (ON124901-ON124904) for BLAST N search [
31]. The results showed that these strains belong to the
Moewusii clade and the closest relative is an unidentified
Chlamydomonas species (uncultured clones AY2009D11 (HQ219443) and AY2009D32 (HQ219442)). In addition, the sequence of the strain CCAP 11/119 (FR865531) was absolutely identical in SSU (ON124833-ON124836) and ITS (ON124901-ON124904) to SAG 2636-2639.
The SSU rDNA sequences were aligned according to the secondary structures and included in a dataset of a total of 36 sequences (1782 bp) of representatives belonging to the
Moewusii clade of the Chlamydophyceae. Among these were the sequences of strains CCCryo 216-05 (GU117580) and CCCryo 217-05 (GU117581). To test which evolutionary model fitted best for the dataset, we calculated the log-likelihood values of 56 models using the automated model selection tool implemented in PAUP, version 4.0b169 [
32], and the best model according to the Akaike criterion by PAUP was chosen for the analyses. The settings of the best model are given in the figure legend. The following methods were used for the phylogenetic analyses: distance, maximum parsimony, maximum likelihood, and Bayesian inference. Programs used included PAUP version 4.0b169 [
32] and the PHASE package 2.0 [
33,
34,
35,
36,
37]. For the Bayesian calculations, the secondary structure models of SSU and ITS (RNA7D in PHASE) were also taken into account.
The secondary structures of ITS-2 sequences were folded using the computer programs
mfold [
38] and analyzed with the ITS-2/Compensatory Base Changes (CBC) approach [
39] described as follows: Those sequences were folded using the three programs mentioned above with the three constraints described as follows: (i) the last 25 bases of the 5.8S rRNA and the first 25 of the LSU rRNA must bind and form the 5.8S/LSU stem; (ii) the pyrimidine/pyrimidine mismatch (the first RNA processing site) in Helix II after the 5–7th base pair has to be present in the structure; and (iii) the second RNA processing site, the GGU motif characteristic for green algae, has to be at the 5′ site in Helix III (for details about the processing sites and constraints, see Coleman [
40] and Cote et al. [
41]). For the usage of ITS-2 as DNA Barcode, it is necessary to obtain ITS sequences free of ambiguities with the flanking regions of 5.8S and LSU rDNA to avoid analyzing pseudogenes. Only then is it possible to fold a unique secondary structure model of ITS-2 with the constraints mentioned above. The structures were visualized with the program PseudoViewer 3 [
42].
Considering this approach, we extracted the conserved region of ITS-2 from each strain as follows: (i) 16 base pairs of the 5.8S/LSU stem, (ii) five basal base pairs of Helix I, (iii) eleven base pairs of Helix II including the pyrimidine-pyrimidine mismatch, and (iv) all base pairs of Helix III. The resulting dataset was then aligned manually. These alignments were translated into base pair alignment using a number code for each base pair (1 = A-U; 2 = U-A; 3 = G-C; 4 = C-G; 5 = G•U; 6 = U•G). The resulting number barcodes are given in the diagnoses below. These barcodes of each species were compared to detect Compensatory Base Changes (CBCs), hemi-CBCs (HCBCs) and/or insertion/deletion, single, or unpaired bases.
3. Results
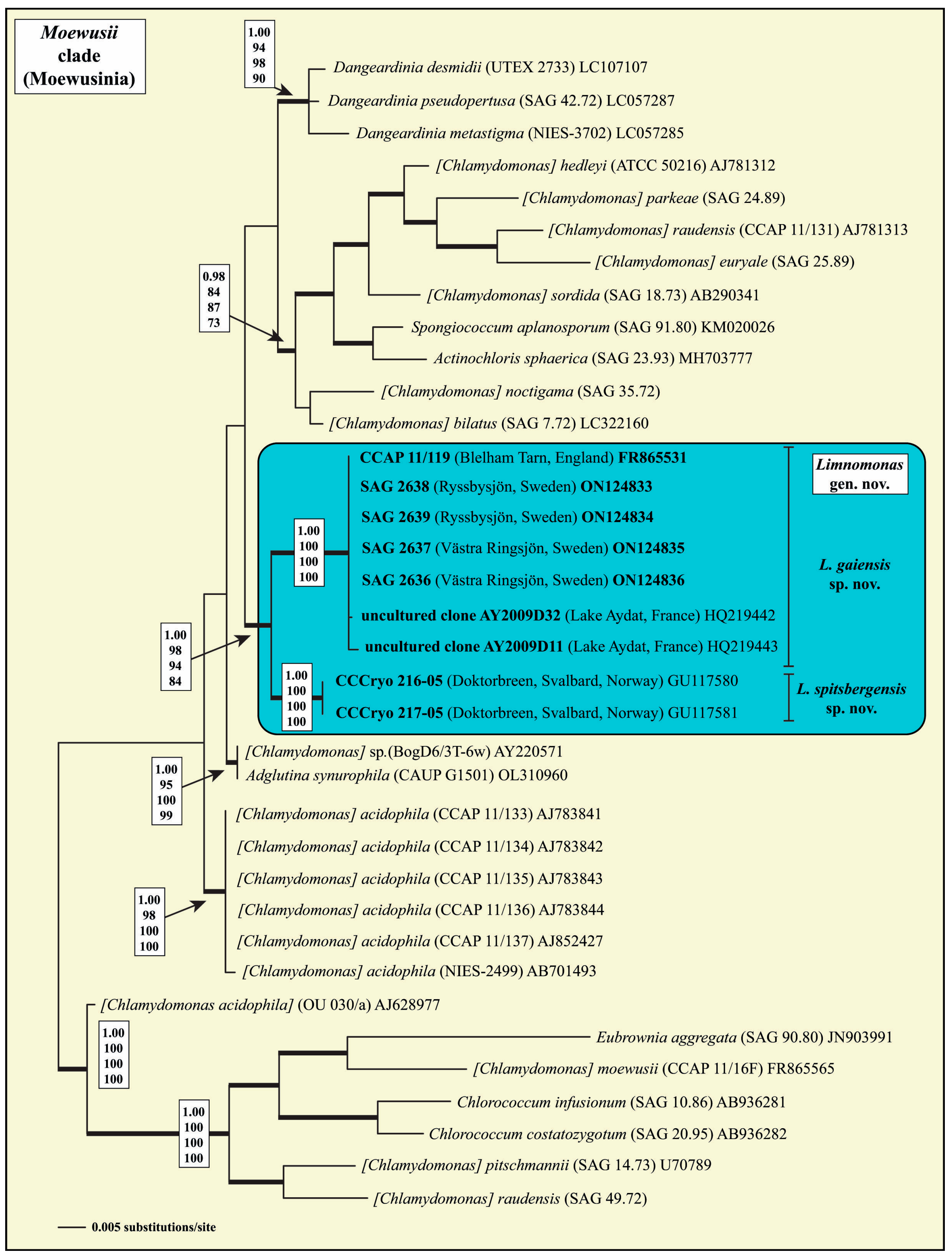
The four isolated strains from Swedish lakes belonged to the well-supported
Moewusii clade of the Chlamydophyceae Ettl (=clockwise-group of the Chlorophyta
sensu Mattox & Stewart). Phylogenetic analyses of the SSU rDNA revealed that these isolates formed together with the strain CCAP 11/119, the uncultured clones AY2009D32 and AY2009D11, and the strains CCCryo 216-05 and CCCryo 217-05, a monophyletic lineage within this clade, which is highly supported in all Bayesian and bootstrap analyses (
Figure 1). This monophyletic lineage is described below as the new genus
Limnomonas.
To determine if the two lineages within
Limnomonas represent different species, we analyzed the ITS-2 rDNA sequences and their secondary structures. As presented in
Figure 2, the two lineages differed in their ITS-2 secondary structures. The applied ITS-2/CBC approach revealed five compensatory base changes and four hemi- CBCs in the conserved regions of ITS-2 among the lineages (
Figure 3). This indicates that these lineages represent species. As a consequence of the phylogenetic results, we propose the new genus
Limnomonas with two species as follows.
Limnomonas S.V.M. Tesson & T. Pröschold gen. nov. (
Figure 4).
Description: Cells are unicellular, with motile and solitary vegetative stages, with two flagella, 1 to 2 times longer than the cell body. Chloroplast cup-shaped containing a single pyrenoid of type 1
sensu Watanabe & Lewis [
16], often ellipsoidal to spherical shape, surrounded by starch grains and penetrated by thylakoids. Two apical contractile vacuoles. The eyespot, often elliptic, part of the chloroplast and located in the anterior half of the cell. With or without frustoconical papilla.
Diagnosis: Differ from other biflagellated unicellular genera by SSU and ITS rDNA sequences.
Etymology: The generic name Limnomonas is a combination of two words—i.e., ‘Limnology’, which refers to the versatile inland waters from which available species were collected (e.g., lakes, mine drainage, snow) and ‘monas’ which refers to ‘unit’ in ancient Greek.
Type species (designated here): Limnomonas gaiensis S.V.M. Tesson & T. Pröschold sp. nov.
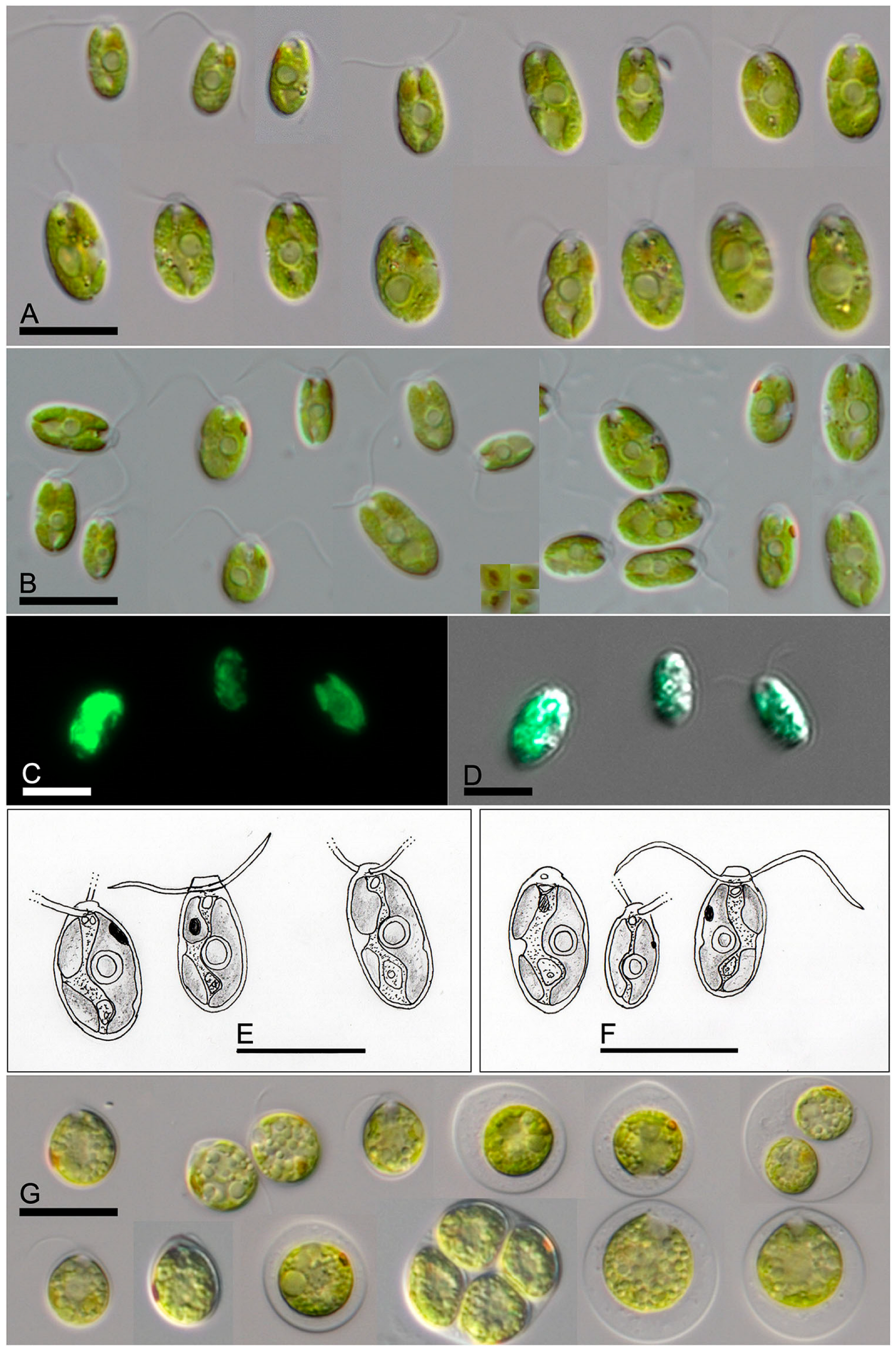
Limnomonas gaiensis S.V.M. Tesson & T. Pröschold sp. nov. (
Figure 4A–F).
Description: Individuals were distributed as single cells or in groups of cells. Each individual possessed a brown-red eyespot visible under light microscope. Their bodies were characterized by delicate walls, uniformly thick with no noticeable local thickening. The shape of the body was ellipsoidal, rounded in the base and truncated towards the front (head) by the presence of a frustoconical papilla, and no further visible extensions. The papilla base ranged from 0.7 to 1.1 µm (0.9 ± 0.1 µm) with a narrower length on one side of the organism. The body length was between 5.5 and 9.2 µm with an average of 6.8 ± 0.8 µm. The body width spanned from 2.1 and 4.5 µm with an average of 2.7 ± 0.5 µm. The organism exhibited two smooth flagella, close to each other. Each flagellum passed through one of the two apertures disposed oppositely on each side of the papilla. The flagella were of nearly equal size, of 1 to 1.5 times the organismal body length, spanning from 6.5 to 11 µm with a mean of 8.9 ± 1.3 µm. One basal nucleus was located in the first-half of the organism.
Diagnosis: Differs from other species of biflagellated chlorophytes by SSU-ITS (ON124833 and ON124901).
Etymology: Individuals were isolated from lakes, i.e., aquatic reservoirs surrounded by lands. The name of the species ‘gaiensis’ is a two-word combination of the root of the ancient Greek ‘gaia’ (signification: land/Earth) and the suffix ‘ensis’ indicating the origin.
Holotype (designated here): The strain SAG 2638 is permanently cryopreserved in a metabolically inactive state (cryopreserved in liquid nitrogen) in the SAG, University of Göttingen, Germany.
Type locality: Lake Ryssbysjön (WA77935742, [
22]), Sweden (57°41′44.9″ N 14°37′40.5″ E). Forest lake with eutrophic, basic to neutral pH waters.
Limnomonas spitsbergensis S.V.M. Tesson & T. Pröschold sp. nov. (
Figure 4G)
Description: Biflagellated cells 9.8 × 11.8 to 13.5 × 15.5 µm in size, broadly ellipsoidal, broadly ovoid, sometimes slightly dorsalventral in shape, with two flagella of cell body length. Cell wall 0.5–0.8 µm thick with small cone-like papilla (~1.2 µm). Chloroplast cup-shaped similar to Chlamydella type, with a single spherical pyrenoid surrounded by barely visible starch grains located towards the middle of the cell. Nucleus central. Elliptical eyespot rod-like positioned laterally, in the middle of the cell. After reaching a cell size of 15–16 µm, the cell wall started to become distant from the protoplast. Mature cells with distant cell walls 15.3 × 16.7 µm up to 20.1 × 21.0 µm in size. Asexual reproduction by 2–4 zoospores. Zoosporangia 21.9 × 22.4 to 23.5 × 25.3 µm in size. Sexual reproduction not observed.
Diagnosis: Deposited in GenBank and in the Culture Collection of Cryophilic Algae as Chlamydomonas cf. proboscigera Korshikov but differs from the original species description by chloroplast morphology (massive cup-shaped of the Euchlamydomonas type), cell shape (broadly ovoid), and ecology (plankton and soil of temperate zones around the world). From other species of biflagellated chlorophytes by SSU-ITS (GU117581).
Etymology: The name of the species originated from its origin.
Holotype (designated here): The strain CCCryo 217-05, collected from snow, is permanently cryopreserved in a metabolically inactive state (cryopreserved in liquid nitrogen) in the CCCryo, Fraunhofer Institute Potsdam-Golm, Germany.
Type locality: Doktorbreen, Nathorst Land, Spitsbergen, Svalbard, Norway (77°34′00.0″ N 16°54′00.0″ E).
The morphology of both species
L. gaiensis and
L. spitsbergensis showed a high phenotypic plasticity (
Figure 4). The cell shape and size, in addition to the position of the pyrenoid, varied. The ellipsoidal cell shape of vegetative cells was prolate spheroidal in
L. gaiensis strains (
Figure 4A–F) and round to oblate spheroidal in
L. spitsbergensis (
Figure 4G). Sporangia were observed in
L. spitsbergensis. In both
Limnomonas species, the ellipsoidal to spherical-shaped pyrenoid was located along the apical-basal axis in the mid region of the organisms.
All 30 strains of
L. gaiensis performed a pendulum movement characterized by a continuous and full-body length oscillation around the front-apical cellular axis, with an oscillation frequency of 3.1 ± 2.0 Hz, and an amplitude spanning from acute to straight angles along the basal-apical axis (
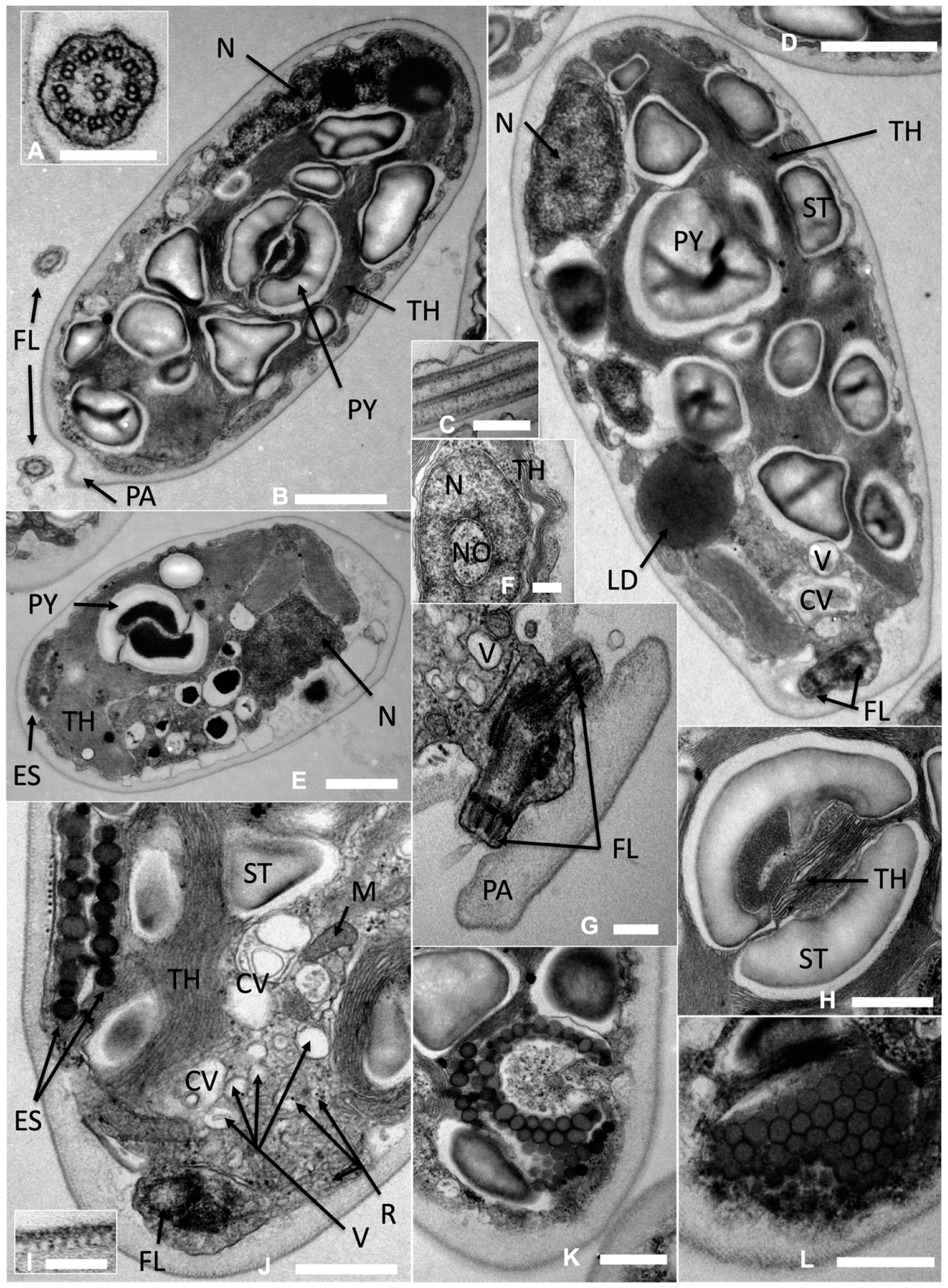
Video S1, Supplementary Materials). The ultrastructure of
Limnomonas gaiensis (
Figure 5) showed a single and large pyrenoid of ellipsoidal to round shape, surrounded by two starch plates looking like hourglass-shaped bowls (
Figure 5B,H), disposed laterally, halfway along the front-basal organism axis (
Figure 5B,D). A network of thylakoids passed through the structure bisecting the pyrenoid (
Figure 5H). The parietal bowl-shaped chloroplast was appended to one side of the organismal cell wall and smooth (
Figure 5E).
The basal body apparatus at apical position showed the typical 9 × 2 + 2 structure of flagellar axonemes in cross-section (
Figure 5A,B). The flagellar membrane and axoneme in longitudinal section are demonstrated in
Figure 5C. The two flagella were close to each other, going out on each side of the papilla (
Figure 5G). A bed of small vesicles and irregularly distributed larger vacuoles (
Figure 5H,J) and their fusion into two distinct contractile vacuoles (
Figure 5E,J) were visible near the flagella system. The cell wall was relatively thick and complex with several visible layers and a tubular-like structure beneath the outer membrane (
Figure 5I).
The nucleus in
L. gaiensis was located in the first-half of the organism towards the basal/posterior end (
Figure 5B,D), sometimes spanning along the apical-basal axis up to the pyrenoid location. The nucleus section showed nuclear pores, a central nucleolus, and patches of heterochromatin around the nuclear periphery and the nucleolus (
Figure 5F).
An ellipsoidal and curved eyespot was located in the anterior part of the cell (
Figure 5J,K). The tangential section of the eyespot showed a crystalline-like plate characterized by close-packing and regularly spaced hexagonally shaped granules (
Figure 5L). The cross-sections of the eyespot showed two layers of dense granules (
Figure 5J). The length of the outer-most layer of the eyespot was between 1.05 and 1.47 μm and contained an irregular size and arrangement of granules. The inner layer length spanned from 0.7 to 1.29 μm. The two layers formed a complex of 0.30–0.36 μm sandwiched between the organismal cell wall and a network of thylakoids (e.g.,
Figure 5J), which occupied one-sixth to one-ninth of the organismal cell width. The granules were of irregular size spanning from 70 to 190 nm in diameter.
Further ultrastructure features were visible, such as the presence of ribosomes and mitochondria (e.g.,
Figure 5J), and numerous inclusions of starch (e.g.,
Figure 5B,D).
Limnomonas gaiensis did not show dorso-ventral axis development, nor caudal extensions, but a regular ellipsoid shape without local thickening of the cell wall.
In environmental samples and mixed cultures, the co-occurrence of Limnomonas gaiensis and Microcystis sp. were monitored. It was possible to observe L. gaiensis organisms contacting both precipitates and colonies of Microcystis sp.
4. Discussion
As demonstrated in
Figure 1, the genus
Limnomonas, composed of two species, formed a highly supported lineage within the
Moewusii clade of the Chlamydophyceae. The two species of
Limnomonas vary in morphology and molecular phylogeny, and they also differ in their ecology. The first species,
L. gaiensis, seems to be widely distributed in plankton of cold-temperate lakes in Sweden and the UK. The second species,
L. spitsbergensis, represented by strains CCCryo 216-05 and CCCryo 217-05, is known, to date, only from snow fields (Spitsbergen, Svalbard, Norway). The two CCCryo strains were originally assigned to the taxon
Chlamydomonas cf.
proboscigera (
Figure 1). Unfortunately, no authentic strain of
C. proboscigera is available in public culture collections. However, comparing the habitats of all records (different water bodies and soils of temperate climate zones around the world) published for
C. proboscigera, it is clear that the two psychrophilic strains do not represent
C. proboscigera (see details in Ettl [
11]). In addition, the morphology of both strains also differs from the original description of Korshikov in Pascher [
43].
Limnomonas spitsbergensis has an ovoid, slightly elongated cell shape, a
Chlamydella-type cup-shaped chloroplast (not
Euchlamydomonas type), a single and round pyrenoid located in the middle of the cell (not basal), and an elliptical (not spotted) eyespot located laterally in the middle of the cells. As a consequence, we described both strains CCCryo 216-05 and CCCryo 217-05 as a new species,
Limnomonas spitsbergensis (see above).
The morphology of
L. gaiensis showed similarities to the descriptions of
Chlamydomonas pumilio and
C. sordida [
11,
12,
44,
45]. It has a regular ellipsoid shape, without local thickening of the cell wall. It also possesses a broader and straight-truncated papilla, a rather large eyespot, and no caudal extensions. As in
C. pumilio,
L. gaiensis showed delicate cell walls, flagella about the same length of the body, two apical contractile vacuoles, a lateral chloroplast, an ellipsoidal to spherical pyrenoid located half-height of the body and a nucleus located in the third of the cell towards the basal apex. Its elliptic eyespot is often in the front third of the cell. However, its body length was slightly bigger than expected in
C. pumilio (4.5–8 μm long, 1.5–3 μm wide). The differences with
C. pumilio var.
pumilio [
12,
24,
45] reside in both the presence of numerous starch reserves and two hourglass-shaped bowls of starch around the pyrenoid instead of the multiple starch plates drawn in the taxonomic key. However, this feature may be a minor issue, as several studies showed that pH and CO
2 conditions may affect the ultrastructural organization of both starch reserves and the pyrenoid starch sheath in
Chlamydomonas species [
46,
47]. Despite similar morphology, Nakada and Nozaki [
48] showed that
C. pumilio is a sister taxon to
C. pulsatilla and
C. subcaudata, belonging to the ‘
Polytoma’-clade
sensu Pröschold et al. [
13]. Considering our results, we therefore presented in this study
L. gaiensis as a new taxon of the ‘
Moewusii’-clade. Similarly, to Nakada and Nozaki [
48], the strain SAG 18.73 assigned as
C. sordida represented with high statistical support an own lineage within the ‘
Moewusii’-clade, and does not belong to
Limnomonas (
Figure 1).
The morphology varies between species of the
Limnomonas genus and with the two clades sharing a last common ancestor with
Limnomonas, i.e.,
Adglutina and
Chlamydomonas acidophila (strains CCAP 11/133-137, NIES-2499).
Limnomonas gaiensis and
C. acidophila possess comparable inner and outer morphology, characterized by a monad ellipsoidal body shape, a basal nucleus, two apical contractile vacuoles, a cup-shaped chloroplast, and an ellipsoidal stigma (eyespot) located towards the anterior half of the cell [
12,
16,
49]. However,
L. gaiensis misses the basal roundness and dorsoventral axis orientation described in both
C. acidophila and
C. proboscigera (Ettl [
12]) and in
L. spitsbergensis (this study). Contrary to the above-mentioned sister-clades,
Adglutina does not possess a pyrenoid and the position of several morphological features (eyespot and nucleus) are variable [
50]. The papilla in
Adglutina (low keel shape [
50]) and in
L. spitsbergensis (small cone-shape [this study]) are low, in the continuity of the cell, whereas
L. gaiensis presents a pronounced frustoconical papilla (this study). Moreover, the size of the eyespot is similar among species of
Limnomonas but differs in shape from that of
Adglutina (ellipsoidal somewhat triangular [
50]). Morphological similarities between
C. acidophila and
Limnomonas, and dissimilarities with
Adglutina, raise the question of the reciprocal impact of organism behavior and habitat on morphology.
Chlamydomonas-like taxa can exhibit cell adhesion to substrates. A study from Kreis et al. [
51] reveals that cell adhesion, investigated in the model species
Chlamydomonas reinhardhii, is independent from the nature of the substrate. Cell adhesion was reported in the above-mentioned sister-clades in
Adglutina synurophila to the eukaryotic microalga
Synura [
50], in
L. gaiensis to precipitates, abiotic surfaces, and the cyanobacteria
Microcystis (this study), and possibly in
C. acidophila NIES-2499 to microfilm [
49,
52]. The pronounced attachment of these taxa to substrate may fulfil similar ecological requirements (e.g., nutrition), but rise from different genetic and morphologic backgrounds. Further investigations are necessary to decipher the factors triggering cell adhesion in these species.
The natural environment in which these species live may be the cause of morphological variations reported above. Investigated strains were collected from a wide range of water acidity levels, e.g.,
C. acidophila from acidic (ca pH 3) river microfilm (NIES-2499 [
50,
53]) and water column (CCAP 11/133-137 [
53,
54,
55]),
A. synurophila from slightly acidic to neutral freshwater bodies (pH 6–7.9 [
50]),
L. spitsbergensis from acidic snow (pH 5.4, ‘Red-snow’,
’Moewusii’-clade/B in [
56]), and
L. gaiensis from slightly acidic to neutral freshwater lakes (pH 6.7–9.1, this study and [
57]). Water acidity can affect the ultrastructure and behavior in
Chlamydomonas species [
58]. For instance, it can increase vacuolization and auto-sporulation [
58] or the pyrenoid volume, cell wall thickness and the formation of palmelloid colonies in other species, possibly reducing the cell volume, the chloroplast volume, the starch reserves, and the cell motility [
49]. Acidification can also lead to flagella amputation [
59]. Intermediately, certain strains of
Chlamydomonas acidophila are not acidophilic
sensu stricto and are able to tolerate a wide range of pH values (1.5–8.4) [
55,
58]. Yet, the impact of water acidity on the size of the papilla remains to be demonstrated, which is one of the main discordant features between
C. acidophila and
L. gaiensis. Moreover, the impact of water acidity does not seem to explain the cell roundness observed in the sister-clades (
C. acidophila and
A. synurophila) and in the sister-species
L. spitzbergensis, which is absent in
L. gaiensis strains, nor the absence of pyrenoid in
A. synurophila only. Future genomic analyses of
L. gaiensis and its relatives would uncover valuable missing information in understanding the evolution of
Chlamydomonas-like organisms.