A Pilot Study to Explore a Correlation between Inflammatory Markers and the Wound Healing Rate in Diabetic Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Treatment Protocol and Laboratory Examination
2.3. Measurement of Wound Characteristics including WHR
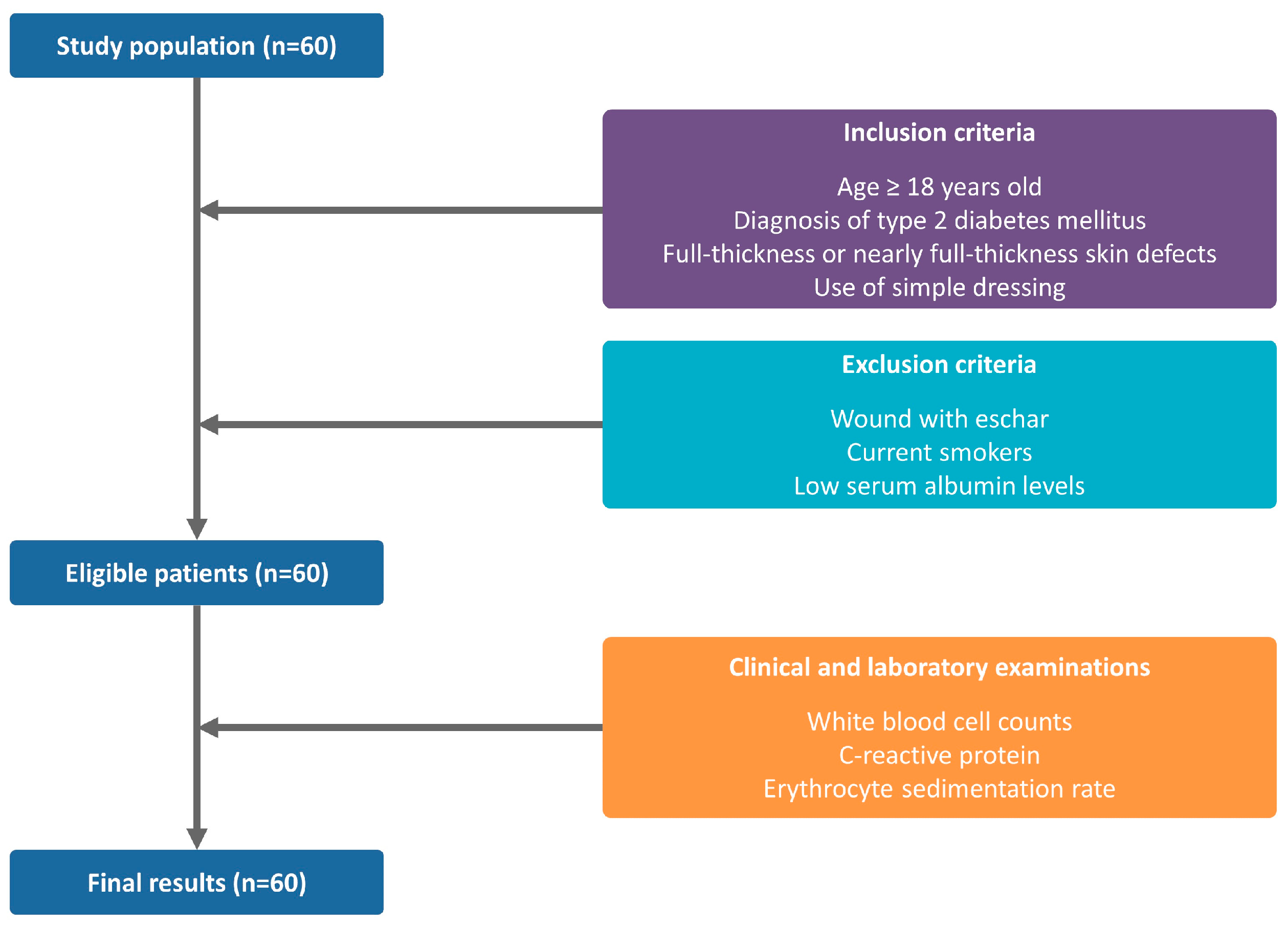
2.4. Patient Evaluation and Criteria
2.5. Statistical Analysis
3. Results
3.1. Baseline Characteristics of the Patients
3.2. Wound Measurements
3.3. Correlations between Inflammatory Markers and WHR
4. Discussion
- (1)
- We evaluated only patients who were hospitalized at a single, secondary medical institution.
- (2)
- We enrolled a small number of patients in the current study. We therefore cannot completely rule out the possibility of selection bias.
- (3)
- We failed to consider the possibility that factors such as aging, obesity or exercise might affect the wound healing process. It is well established that the normal WH process may be impaired in elderly or obese individuals; these patients are susceptible to infection, prolonged pain and other complications [45,46]. Moreover, recent studies have suggested that physical exercise may promote the WH process even in the presence of aging and obesity [47].
- (4)
- We failed to analyze the effects of confounding factors, such as hypertension, congestive heart failure or the location and shape of wounds, on WHR.
- (5)
- We failed to perform a wound fluid assay of inflammatory cytokines and proteases involved in WH; proteases such as matrix metalloproteinase-2 and -9 (MMP-2 and -9) as well as inflammatory markers such as IL-1 and tumor necrosis factor-α (TNF-α) are significantly elevated in chronic wounds as compared to their acute counterparts, which has been well described in the literature [48].
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wong, R.; Geyer, S.; Weninger, W.; Guimberteau, J.C.; Wong, J.K. The dynamic anatomy and patterning of skin. Exp. Dermatol. 2016, 25, 92–98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdallah, F.; Mijouin, L.; Pichon, C. Skin Immune Landscape: Inside and Outside the Organism. Mediators Inflamm. 2017, 2017, 5095293. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.A.; Sohail, M.; Khan, S.; Minhas, M.U.; de Matas, M.; Sikstone, V.; Hussain, Z.; Abbasi, M.; Kousar, M. Biopolymer-based biomaterials for accelerated diabetic wound healing: A critical review. Int. J. Biol. Macromol. 2019, 139, 975–993. [Google Scholar] [CrossRef] [PubMed]
- Sibbald, R.G.; Campbell, K.; Coutts, P.; Queen, D. Intact skin—An integrity not to be lost. Ostomy Wound Manag. 2003, 49, 27–28. [Google Scholar]
- El Ayadi, A.; Jay, J.W.; Prasai, A. Current Approaches Targeting the Wound Healing Phases to Attenuate Fibrosis and Scarring. Int. J. Mol. Sci. 2020, 21, 1105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chicharro-Alcántara, D.; Rubio-Zaragoza, M.; Damiá-Giménez, E.; Carrillo-Poveda, J.M.; Cuervo-Serrato, B.; Peláez-Gorrea, P.; Sopena-Juncosa, J.J. Platelet Rich Plasma: New Insights for Cutaneous Wound Healing Management. J. Funct. Biomater. 2018, 9, 10. [Google Scholar] [CrossRef] [Green Version]
- Ellis, S.; Lin, E.J.; Tartar, D. Immunology of Wound Healing. Curr. Dermatol. Rep. 2018, 7, 350–358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Liu, Y.; Deng, J.; Li, W.; Nie, X. Fibroblast Growth Factor in Diabetic Foot Ulcer: Progress and Therapeutic Prospects. Front. Endocrinol. 2021, 12, 744868. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.; Jackson, C.J. Extracellular Matrix Reorganization During Wound Healing and Its Impact on Abnormal Scarring. Adv. Wound Care 2015, 4, 119–136. [Google Scholar] [CrossRef] [Green Version]
- Gonzalez, A.C.; Costa, T.F.; Andrade, Z.A.; Medrado, A.R. Wound healing—A literature review. An. Bras. Dermatol. 2016, 91, 614–620. [Google Scholar] [CrossRef] [Green Version]
- Järbrink, K.; Ni, G.; Sönnergren, H.; Schmidtchen, A.; Pang, C.; Bajpai, R.; Car, J. The humanistic and economic burden of chronic wounds: A protocol for a systematic review. Syst. Rev. 2017, 6, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corbett, L.Q.; Ennis, W.J. What Do Patients Want? Patient Preference in Wound Care. Adv. Wound Care 2014, 3, 537–543. [Google Scholar] [CrossRef] [Green Version]
- Demidova-Rice, T.N.; Hamblin, M.R.; Herman, I.M. Acute and impaired wound healing: Pathophysiology and current methods for drug delivery, part 1: Normal and chronic wounds: Biology, causes, and approaches to care. Adv. Skin Wound Care 2012, 25, 304–314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Landén, N.X.; Li, D.; Ståhle, M. Transition from inflammation to proliferation: A critical step during wound healing. Cell. Mol. Life Sci. 2016, 73, 3861–3885. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grossmann, V.; Schmitt, V.H.; Zeller, T.; Panova-Noeva, M.; Schulz, A.; Laubert-Reh, D.; Juenger, C.; Schnabel, R.B.; Abt, T.G.; Laskowski, R.; et al. Profile of the Immune and Inflammatory Response in Individuals with Prediabetes and Type 2 Diabetes. Diabetes Care 2015, 38, 1356–1364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wysocki, A.B. Wound fluids and the pathogenesis of chronic wounds. J. Wound Ostomy Cont. Nurs. 1996, 23, 283–290. [Google Scholar]
- Alavi, A.; Sibbald, R.G.; Mayer, D.; Goodman, L.; Botros, M.; Armstrong, D.G.; Woo, K.; Boeni, T.; Ayello, E.A.; Kirsner, R.S. Diabetic foot ulcers: Part I. Pathophysiology and prevention. J. Am. Acad. Dermatol. 2014, 70, 1-e1. [Google Scholar] [CrossRef]
- Galkowska, H.; Wojewodzka, U.; Olszewski, W.L. Chemokines, cytokines, and growth factors in keratinocytes and dermal endothelial cells in the margin of chronic diabetic foot ulcers. Wound Repair Regen. 2006, 14, 558–565. [Google Scholar] [CrossRef]
- Patel, S.; Srivastava, S.; Singh, M.R.; Singh, D. Mechanistic insight into diabetic wounds: Pathogenesis, molecular targets and treatment strategies to pace wound healing. Biomed. Pharmacother. 2019, 112, 108615. [Google Scholar] [CrossRef] [PubMed]
- Cukjati, D.; Rebersek, S.; Miklavcic, D. A reliable method of determining wound healing rate. Med. Biol. Eng. Comput. 2001, 39, 263–271. [Google Scholar] [CrossRef]
- Goto, T.; Saligan, L.N. Wound Pain and Wound Healing Biomarkers from Wound Exudate: A Scoping Review. J. Wound Ostomy Cont. Nurs. 2020, 47, 559–568. [Google Scholar] [CrossRef]
- Santema, T.B.; Poyck, P.P.; Ubbink, D.T. Skin grafting and tissue replacement for treating foot ulcers in people with diabetes. Cochrane Database Syst. Rev. 2016, 2, CD011255. [Google Scholar] [CrossRef]
- Amin, N.; Doupis, J. Diabetic foot disease: From the evaluation of the “foot at risk” to the novel diabetic ulcer treatment modalities. World J. Diabetes 2016, 7, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.S.; Arulselvan, P.; Ng, S.F.; Mat Taib, C.N.; Sarian, M.N.; Fakurazi, S. Improvement of diabetic wound healing by topical application of Vicenin-2 hydrocolloid film on Sprague Dawley rats. BMC Complement. Altern. Med. 2019, 19, 20. [Google Scholar] [CrossRef]
- Choi, S.K.; Kim, C.K.; Jo, D.I.; Lee, M.C.; Kim, J.N.; Choi, H.G.; Shin, D.H.; Kim, S.H. Factors Associated with a Prolonged Length of Hospital Stay in Patients with Diabetic Foot: A Single-Center Retrospective Study. Arch. Plast. Surg. 2017, 44, 539–544. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sproston, N.R.; Ashworth, J.J. Role of C-Reactive Protein at Sites of Inflammation and Infection. Front. Immunol. 2018, 9, 754. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, M.F.; Khan, Z.A.; Park, S. Detection of C-Reactive Protein Using Histag-HRP Functionalized Nanoconjugate with Signal Amplified Immunoassay. Nanomaterials 2020, 10, 1240. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, R.; Majidi, A.; Motamed, H.; Amini, A.; Najari, F.; Tabatabaey, A. Erythrocyte Sedimentation Rate Measurement Using as a Rapid Alternative to the Westergren Method. Emergency 2015, 3, 50–53. [Google Scholar]
- Shetty, R.; Sreekar, H.; Lamba, S.; Gupta, A.K. A novel and accurate technique of photographic wound measurement. Indian J. Plast. Surg. 2012, 45, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Suarez-Arnedo, A.; Torres Figueroa, F.; Clavijo, C.; Arbeláez, P.; Cruz, J.C.; Muñoz-Camargo, C. An image J plugin for the high throughput image analysis of in vitro scratch wound healing assays. PLoS ONE 2020, 15, e0232565. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.C.; Dearman, B.; Greenwood, J.E. A comparison of wound area measurement techniques: Visitrak versus photography. Eplasty 2011, 11, e18. [Google Scholar] [PubMed]
- Jessup, R.L. What is the best method for assessing the rate of wound healing? A comparison of 3 mathematical formulas. Adv. Skin Wound Care 2006, 19, 138–147. [Google Scholar] [CrossRef]
- Mao, Z.; Wu, J.H.; Dong, T.; Wu, M.X. Additive enhancement of wound healing in diabetic mice by low level light and topical CoQ10. Sci. Rep. 2016, 6, 20084. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.; Zheng, Y.; Liu, H.; Li, T. Modulation of IGF2BP1 by long non-coding RNA HCG11 suppresses apoptosis of hepatocellular carcinoma cells via MAPK signaling transduction. Int. J. Oncol. 2017, 51, 791–800. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mast, B.A.; Schultz, G.S. Interactions of cytokines, growth factors, and proteases in acute and chronic wounds. Wound Repair Regen. 1996, 4, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Fivenson, D.P.; Faria, D.T.; Nickoloff, B.J.; Poverini, P.J.; Kunkel, S.; Burdick, M.; Strieter, R.M. Chemokine and inflammatory cytokine changes during chronic wound healing. Wound Repair Regen. 1997, 5, 310–322. [Google Scholar] [CrossRef] [Green Version]
- Werdin, F.; Tenenhaus, M.; Rennekampff, H.O. Chronic wound care. Lancet 2008, 372, 1860–1862. [Google Scholar] [CrossRef]
- Christman, A.L.; Selvin, E.; Margolis, D.J.; Lazarus, G.S.; Garza, L.A. Hemoglobin A1c predicts healing rate in diabetic wounds. J. Investig. Dermatol. 2011, 131, 2121–2127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Asten, S.A.; Jupiter, D.C.; Mithani, M.; La Fontaine, J.; Davis, K.E.; Lavery, L.A. Erythrocyte sedimentation rate and C-reactive protein to monitor treatment outcomes in diabetic foot osteomyelitis. Int. Wound J. 2017, 14, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Wang, J. Neutrophils in tissue injury and repair. Cell Tissue Res. 2018, 371, 531–539. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uçkay, I.; Aragón-Sánchez, J.; Lew, D.; Lipsky, B.A. Diabetic foot infections: What have we learned in the last 30 years? Int. J. Infect Dis. 2015, 40, 81–91. [Google Scholar] [CrossRef] [Green Version]
- Spampinato, S.F.; Caruso, G.I.; De Pasquale, R.; Sortino, M.A.; Merlo, S. The Treatment of Impaired Wound Healing in Diabetes: Looking among Old Drugs. Pharmaceuticals 2020, 13, 60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamza, R.Z.; Al-Baqami, N.M.; Khojah, E.; Mansour, A.M.A.E.; Al-Motaani, S.A.; Al-Salmi, F.; El-Megharbel, S.M. Possible Antioxidant and Antidiabetic Effects of Combretum Molle Extract in a Diabetes Mellitus Experimental Model in Male Rats. Nat. Prod. Commun. 2021, 16, 1934578X211043964. [Google Scholar] [CrossRef]
- Hamza, R.Z.; Al-Motaani, S.E.; Al-Talhi, T. Therapeutic and Ameliorative Effects of Active Compounds of Combretum molle in the Treatment and Relief from Wounds in a Diabetes Mellitus Experimental Model. Coatings 2021, 11, 324. [Google Scholar] [CrossRef]
- Pierpont, Y.N.; Dinh, T.P.; Salas, R.E.; Johnson, E.L.; Wright, T.G.; Robson, M.C.; Payne, W.G. Obesity and surgical wound healing: A current review. ISRN Obes. 2014, 2014, 638936. [Google Scholar] [CrossRef] [Green Version]
- Avishai, E.; Yeghiazaryan, K.; Golubnitschaja, O. Impaired wound healing: Facts and hypotheses for multi-professional considerations in predictive, preventive and personalised medicine. EPMA J. 2017, 8, 23–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eming, S.A.; Martin, P.; Tomic-Canic, M. Wound repair and regeneration: Mechanisms, signaling, and translation. Sci. Transl. Med. 2014, 6, 265sr6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gohel, M.S.; Windhaber, R.A.; Tarlton, J.F.; Whyman, M.R.; Poskitt, K.R. The relationship between cytokine concentrations and wound healing in chronic venous ulceration. J. Vasc. Surg. 2008, 48, 1272–1277. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| Variables | Values | p-Value | |
|---|---|---|---|
| Early WH Group (n = 27) | Late WH Group (n = 33) | ||
| Age (years) | 45.2 ± 19.3 | 51.4 ± 18.3 | 0.208 |
| Sex (male-to-female ratio) | 14:13 | 16:17 | 0.128 |
| Height (cm) | 167.2 ± 8.3 | 161.6 ± 7.1 | 0.067 |
| Weight (kg) | 68.3 ± 10.2 | 62.9 ± 8.4 | 0.091 |
| BMI (kg/m2) | 27.3 ± 4.0 | 21.1 ± 2.2 | 0.113 |
| BG (mg/dL) | 189.3 ± 86.3 | 237.1 ± 72.5 | <0.0001 |
| HbA1c (%) | 7.13 ± 2.5 | 9.22 ± 5.7 | |
| Underlying diseases | |||
| Hypertension | 12 (44.4%) | 19 (57.6%) | >0.05 |
| Hepatitis | 4 (14.8%) | 4 (12.1%) | |
| Tuberculosis | 2 (7.4%) | 3 (9.09%) | |
| Stroke | 4 (14.8%) | 3 (9.09%) | |
| Chronic renal failure | 3 (11.1%) | 2 (6.06%) | |
| Congestive heart failure | 1 (3.7%) | 2 (6.06%) | |
| Verruca | 1 (3.7%) | 0 (0%) | |
| Wound characteristics | |||
| Location | |||
| Upper extremity | 13 (48.1%) | 16 (48.5%) | >0.05 |
| Lower extremity | 13 (48.1%) | 15 (45.5%) | |
| Trunk | 9 (33.3%) | 11 (33.3%) | |
| Back | 3 (11.1%) | 3 (9.09%) | |
| Flank | 1 (3.7%) | 2 (6.06%) | |
| Chest | 1 (3.7%) | 1 (3.0%) | |
| Lower abdomen | 1 (3.7%) | 0 (0%) | |
| Shape | |||
| Trapezoid | 7 (25.9%) | 9 (27.3%) | >0.05 |
| Round | 8 (29.6%) | 8 (24.2%) | |
| Rod | 5 (18.5%) | 7 (21.2%) | |
| Irregular | 5 (18.5%) | 5 (15.2%) | |
| Triangle | 2 (7.4%) | 4 (12.1%) | |
| Surface area | |||
| ≥100 mm2 | 12 (44.4%) | 15 (45.5%) | >0.05 |
| <100 mm2 | 15 (55.6%) | 18 (54.5%) | |
| Bacterial infection | |||
| No growth | 25 (92.6%) | 30 (90.9%) | >0.05 |
| MRSA | 1 (3.7%) | 2 (6.0%) | |
| Staphylococcus epidermidis | 1 (3.7%) | 1 (3.0%) | |
| Variables | Values | p-Value | |
|---|---|---|---|
| Early WH Group (n = 27) | Late WH Group (n = 33) | ||
| WBC (×106/L) | 15,300 ± 6400 | 8000 ± 5300 | 0.027 * |
| CRP (mg/L) | 34.62 ± 13.75 | 146.28 ± 112.46 | 0.036 * |
| ESR (mm/h) | 16.34 ± 5.26 | 29.14 ± 6.88 | 0.043 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, Y.; Jo, Y.; Sohn, J.; Kim, R. A Pilot Study to Explore a Correlation between Inflammatory Markers and the Wound Healing Rate in Diabetic Patients. Medicina 2022, 58, 390. https://doi.org/10.3390/medicina58030390
Song Y, Jo Y, Sohn J, Kim R. A Pilot Study to Explore a Correlation between Inflammatory Markers and the Wound Healing Rate in Diabetic Patients. Medicina. 2022; 58(3):390. https://doi.org/10.3390/medicina58030390
Chicago/Turabian StyleSong, Yukwan, Yongkyu Jo, Jeongeun Sohn, and Robert Kim. 2022. "A Pilot Study to Explore a Correlation between Inflammatory Markers and the Wound Healing Rate in Diabetic Patients" Medicina 58, no. 3: 390. https://doi.org/10.3390/medicina58030390






