Acceptability and Feasibility of HPV Self-Sampling as an Alternative Primary Cervical Cancer Screening in Under-Screened Population Groups: A Cross-Sectional Study
Abstract
:1. Introduction
2. Methods
2.1. Study Designs
2.2. Study Population
2.3. HPV Self-Sampling
2.4. Laboratory Test for HPV Detection and Genotyping
2.5. Outcome Measure
2.6. Statistical Analysis
2.7. Ethical Consideration
3. Results
3.1. Demographic Characteristics
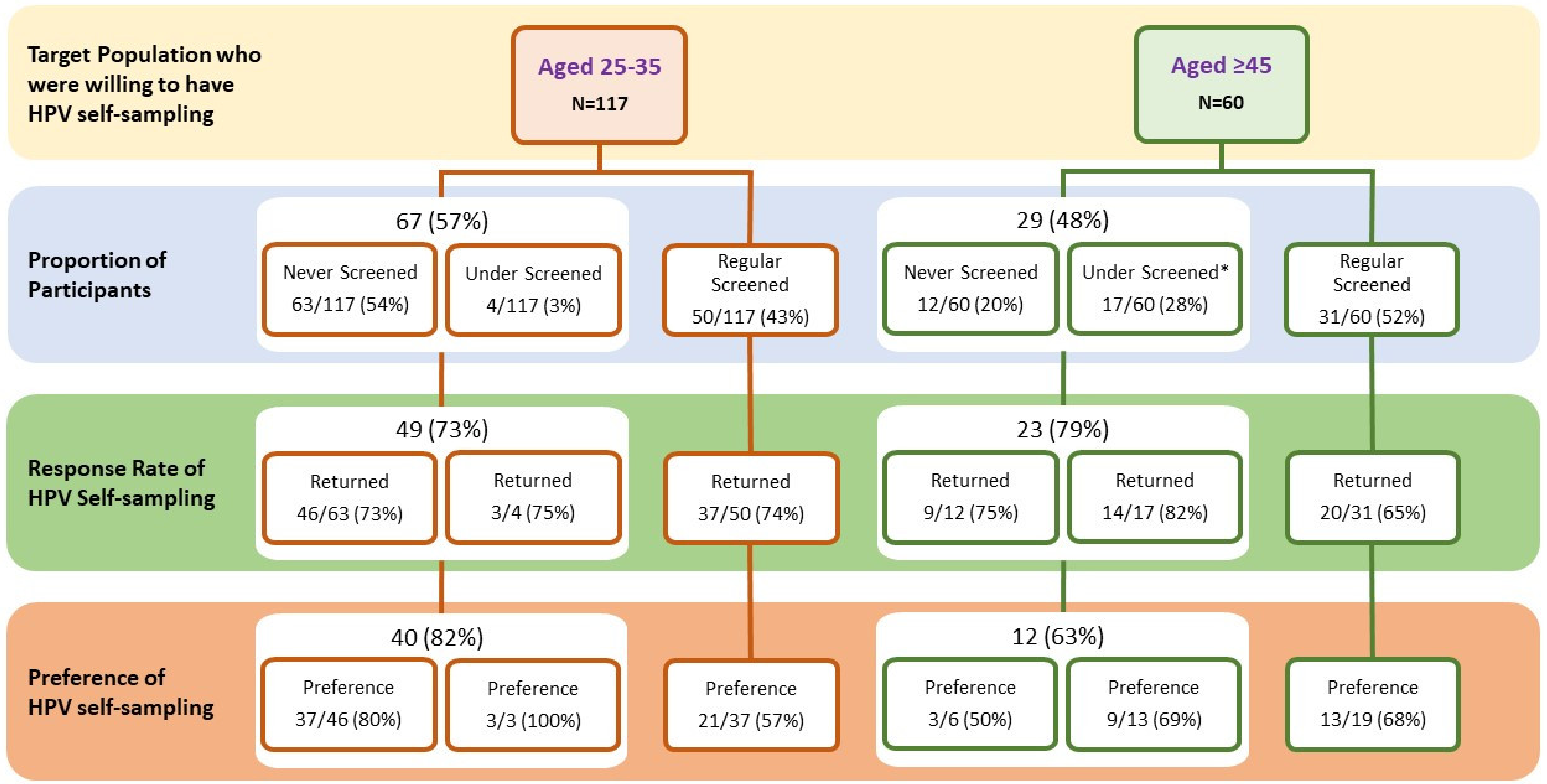
3.2. Response Rate of HPV Self-Sampling
3.3. Prevalence of HPV Infection
3.4. Factors Associated with the Uptake of HPV Self-Sampling
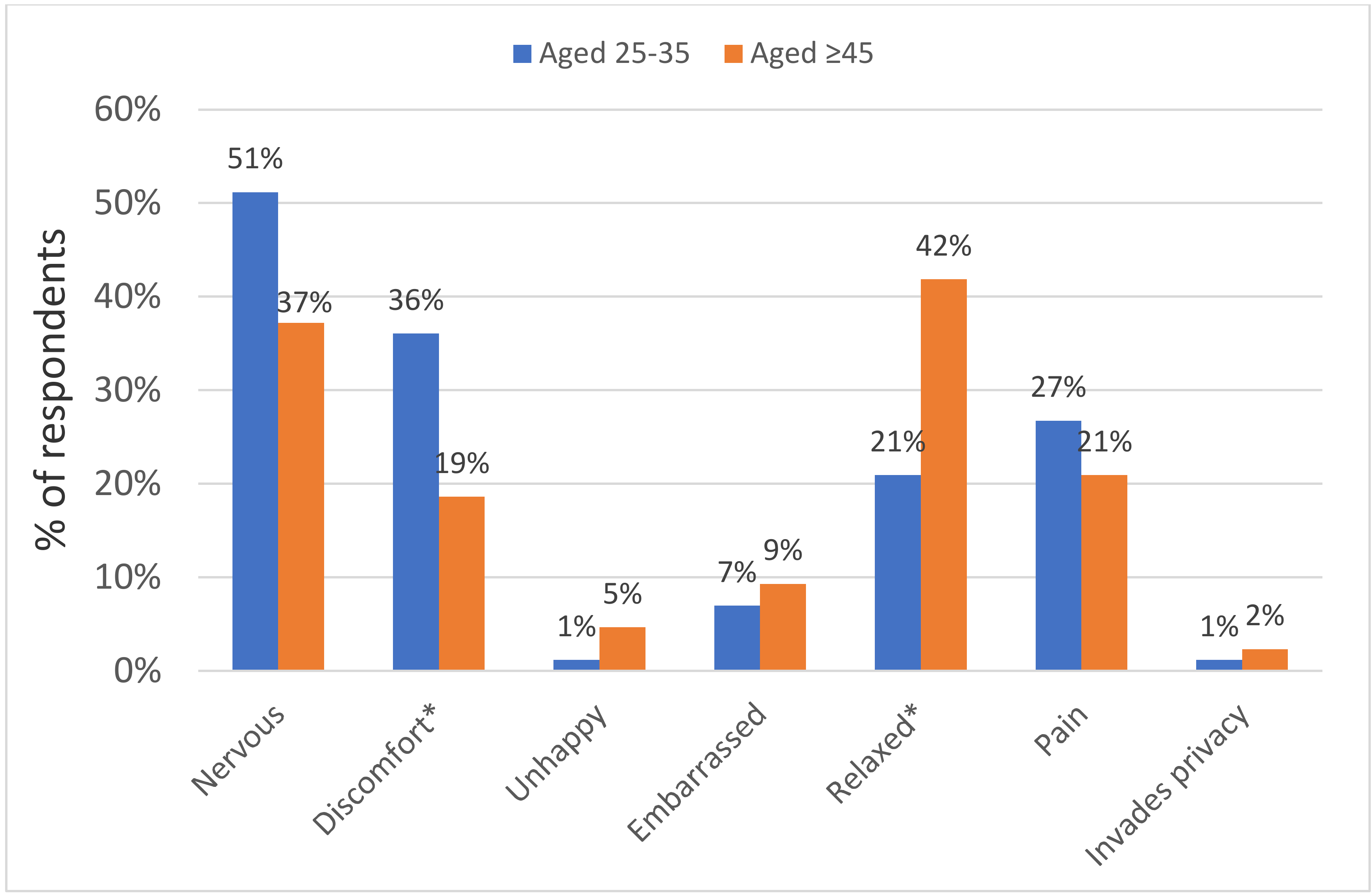
3.5. Acceptability Towards HPV Self-Sampling
3.6. Future Preference for HPV Self-Sampling as an Alternative Primary Screening for Cervical Cancer
4. Discussion
4.1. HPV Self-Sampling
4.2. Factors Associated with the Uptake of HPV Self-Sampling
4.3. Acceptability of HPV Self-Sampling
4.4. Barriers to Cervical Cancer Screening: Pap Smear vs. HPV Self-Sampling
4.5. Current Situation: HPV Self-Sampling as an Alternative Primary Screening
5. Strengths and Limitations of this Study
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. HPV and Cervical Cancer Fact Sheet. 2019. Available online: https://www.who.int/en/news-room/fact-sheets/detail/human-papillomavirus-(hpv)-and-cervical-cancer (accessed on 30 May 2019).
- Bao, H.; Zhang, D.; Wang, L.; Zhang, M.; Zhao, Z.; Fang, L.; Cong, S.; Zhou, M.; Wang, L. Significant variations in the cervical cancer screening rate in China by individual-level and geographical measures of socioeconomic status: A multilevel model analysis of a nationally representative survey dataset. Cancer Med. 2018, 7, 2089–2100. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer. Cancer screening in the European Union (2017). In Report on the Implementation of the Council Recommendation on Cancer Screening; IARC: Lyon, France, 2017. [Google Scholar]
- Canadian Cancer Society. Cancer Screening. 2018. Available online: https://www.statcan.gc.ca/eng/ (accessed on 12 December 2019).
- National Cancer Institute. Cancer Trends Progress Report. 2019. Available online: http://progressreport.cancer.gov (accessed on 1 January 2020).
- Department of Health. Report of Population Health Survey 2014/2015; Centre for Health Protection, DH: Hong Kong, China, 2017.
- Chorley, A.J.; Marlow, L.A.; Forster, A.S.; Haddrell, J.B.; Waller, J. Experiences of cervical screening and barriers to participation in the context of an organised programme: A systematic review and thematic synthesis. Psychooncology 2017, 26, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.; Bartoszek, M.; Marlow, L.; Wardie, J. Barriers to cervical cancer screening attendance in England: A population-based survey. J. Med. Screen 2009, 16, 199–204. [Google Scholar]
- Wong, E.L.Y.; Chan, P.K.S.; Chor, J.S.Y.; Cheung, A.W.L.; Huang, F.; Wong, S.Y.S. Evaluation of the impact of Huma Papillomavirus DNA self-sampling on the uptake of cervical cancer screening. Cancer Nurs. 2016, 39, E1–E11. [Google Scholar] [PubMed] [Green Version]
- Bulk, S.; van Kemenade, J.; Rozendaal, L.; Meijer, C.J. The Dutch CISOE-A framework for cytology reporting increases efficacy of screening upon standardisation since 1996. Clin. Pathol. 2004, 57, 388–393. [Google Scholar] [CrossRef] [Green Version]
- Cuzick, J.; Szarewski, A.; Terry, G.; Hanby, A.; Maddox, P.; Anderson, M.; Steele, S.; Guillebaud, J.; Kocjean, C. Human Papillomavirus testing in primary cervical screening. Lancet 1995, 345, 1533–1536. [Google Scholar] [CrossRef]
- Pengsaa, P.; Vatanasapt, V.; Sriamporn, S.; Sanchaisuriya, P.; Schelp, F.P.; Noda, S.; Kato, S.; Kongdee, W.; Kanchanawirojkul, N.; Aranyasen, O. A self-administered device for cervical cancer screening in northeast Thailand. Acta Cytol. 1997, 41, 749–754. [Google Scholar] [CrossRef] [PubMed]
- Madzima, T.R.; Vahabi, M.; Lofters, A. Emerging role of HPV self-sampling in cervical cancer screening for hard-to-reach women: Focused literature review. Can. Fam. Physician 2017, 63, 597–601. [Google Scholar] [PubMed]
- Arbyn, M.; Smith, S.B.; Temin, S.; Sultana, F.; Castle, P. Detecting cervical precancer and reaching underscreened women by using HPV testing on self samples: Updated meta-analysis. BMJ 2018, 363, k4823. [Google Scholar] [CrossRef] [Green Version]
- Aitken, C.A.; Agt, H.M.E.V.; Siebers, A.G.; Van Kemenade, F.J.; Niesters, H.G.; Melchers, W.J.G.; Vedder, J.E.M.; Schuurman, R.; Brule, A.J.C.V.D.; Van Der Linden, H.C.; et al. Introduction of primary screening using high-risk HPV DNA detection in the Dutch cervical cancer screening programme: A population-based cohort study. BMC Med. 2019, 17, 228. [Google Scholar] [CrossRef] [Green Version]
- Cancer Council Australia. National Cervical Screening Program: Guidelines for the Management of Screen-detected Abnormalities, Screening in Specific Populations and Investigation of Abnormal Vaginal Bleeding. 2019. Available online: https://wiki.cancer.org.au/australiawiki/index.php?oldid=207530 (accessed on 27 September 2019).
- Rose Foundation. Program Rose: Removing Obstacles to Cervical Screening. 2019. Available online: https://www.programrose.org/ (accessed on 27 September 2019).
- Finnish Medical Society. Current Care Guidelines are Independent, Evidence-Based Clinical Practice Guidelines. Available online: https://www.kaypahoito.fi/nix02431 (accessed on 27 September 2019).
- The Hong Kong College of Obstetricians and Gynaecologists. Guidelines for Cervical Cancer Prevention and Screening. Hong Kong Med. J. 2016, 4, 1–25. [Google Scholar]
- Qiagen-Sample and Assay Technologies. Available online: file:///C:/Users/MDPI/AppData/Local/Temp/080121_cheuvreux_js.pdf (accessed on 27 July 2020).
- Wong, M.C.S.; Vlantis, A.C.; Liang, M.; Wong, P.Y.; Ho, W.C.S.; Boon, S.S.; Sze, R.K.H.; Leung, C.; Chan, P.K.; Chen, Z. Prevalence and epidemiologi profile of oral infection with alpha, beta, and gamma papillomaviruses in an Asian-Chinese population. J. Infect. 2018, 218, 388–397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Illumina. MiSeq™ System Focused Power; Illumina, Inc.: San Diego, CA, USA, 2018. [Google Scholar]
- Statistical Package for Social Sciences. IBM SPSS Statistics for Windows, Version 24; IBM Corp.: Armonk, NY, USA, 2019. [Google Scholar]
- Zhao, Y.; Liao, Q.; Mi, X.; Li, M.Z.; Zhao, C.; Cui, S.H.; Li, J.R.; Wang, Y.; Wang, J.L.; Wei, L.H. Survey of the acceptance status of HPV self-sampling screening in female population for cervical cancer. Zhonghua Fu Chan Ke Za Zhi 2019, 54, 312–317. [Google Scholar] [PubMed]
- Chen, S.L.; Hsieh, P.C.; Chou, C.H.; Tzeng, Y.L. Determinants of women’s likelihood of vaginal self-sampling for human papillomavirus to screen for cervical cancer in Taiwan: A cross-sectional study. BMC Women’s Health 2014, 14, 139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, H.K.; Myong, J.-P.; Byun, S.W.; Lee, S.-J.; Lee, Y.S.; Lee, H.-N.; Lee, K.H.; Park, D.C.; Kim, C.J.; Hur, S.Y.; et al. Factors associated with participation in cervical cancer screening among young Koreans: A nationwide cross-sectional study. BMJ Open 2017, 7, e013868. [Google Scholar] [CrossRef]
- Nwabichie, C.C.; Manaf, R.A.; Ismail, S. Factors affecting uptake of cervical cancer screening among African women in Klang Valley, Malaysia. Asian Pac. J. Cancer Prev. 2018, 19, 825–831. [Google Scholar]
- Nelson, E.J.; Maynard, B.R.; Loux, T.; Fatla, J.; Gordon, R.; Arnold, L.D. The acceptability of self-sampled screening for HPV DNA: A systematic review and meta-analysis. Sex. Transm. Infect. 2016, 93, 56–61. [Google Scholar] [CrossRef]
- Oranratanaphan, S.; Termrungruanglert, W.; Khemapech, N. Acceptability of self-sampling HPV testing among Thai women for cervical cancer screening. Asian Pac. J. Cancer Prev. 2014, 15, 7437–7441. [Google Scholar] [CrossRef] [Green Version]
- Rosenbaum, A.J.; Gage, J.C.; Alfaro, K.M.; Ditzian, L.R.; Maza, M.; Scarinci, I.C.; Felix, J.C.; Castle, P.E.; Villalta, S.; Miranda, E.; et al. Acceptability of self-collected versus provider-collected sampling for HPV DNA testing among women in rural El Salvador. Int. J. Gynecol. Obstet. 2014, 126, 156–160. [Google Scholar] [CrossRef]
- Winer, R.L.; Gonzales, A.A.; Noonan, C.J.; Cherne, S.L.; Buchwald, D.S. Assessing acceptability of self-sampling kits, prevalence, and risk factors for human papillomavirus infection in American Indian women. J. Community Health 2016, 41, 1049–1061. [Google Scholar] [CrossRef] [Green Version]
- Penaranda, E.; Molokwu, J.; Flores, S.; Byrd, T.; Brown, L.; Shokar, N. Women’s attitudes towards cervico-vaginal self-sampling for high risk HPV infection on the U.S.-Mexico border. J. Low. Genit. Tract Dis. 2015, 19, 323–328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma’Som, M.; Bhoo-Pathy, N.; Nasir, N.H.; Bellinson, J.; Subramaniam, S.; Ma, Y.; Yap, S.-H.; Goh, P.-P.; Gravitt, P.; Woo, Y.L. Attitudes and factors affecting acceptability of self-administered cervicovaginal sampling for human papillomavirus (HPV) genotyping as an alternative to Pap testing among multiethnic Malaysian women. BMJ Open 2016, 6, e011022. [Google Scholar] [CrossRef] [PubMed]
- Lorenzi, N.P.C.; Termini, L.; Filho, A.L.; Tacla, M.; De Aguiar, L.M.; Beldi, M.C.; Ferreira-Filho, E.S.; Baracat, E.C.; Soares-Júnior, J.M. Age-related acceptability of vaginal self-sampling in cervical cancer screening at two university hospitals: A pilot cross-sectional study. BMC Public Health 2019, 19, 963. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anhang, R.; Nelson, J.A.; Telerant, R.; Chiasson, M.A.; Wright, T.C. Acceptability of self-collection of specimens for HPV DNA testing in an urban population. J. Women’s Health 2005, 14, 721–728. [Google Scholar] [CrossRef]
- Berner, A.; Ben Hassel, S.; Tebeu, P.-M.; Untiet, S.; Kengne-Fosso, G.; Navarria, I.; Boulvain, M.; Vassilakos, P.; Petignat, P. Human papillomavirus self-sampling in Cameroon: women’s uncertainties over the reliability of the method are barriers to acceptance. J. Low. Genit. Tract Dis. 2013, 17, 235–241. [Google Scholar] [CrossRef]
- Howard, M.; Lytwyn, A.; Lohfeld, L.; Redwood-Camphell, L.; Fowler, N.; Karwalajtys, T. Barriers to acceptance of self-sampling for Human Papillomavirus across Ethnolinguistic groups of women. Can. J. Public Health 2009, 100, 365–369. [Google Scholar] [CrossRef]
- Comprehensive Cervical Cancer Control: A Guide to Essential Practice. Available online: https://apps.who.int/iris/bitstream/handle/10665/144785/9789241548953_eng.pdf;jsessionid=B2336FF4E1B70CD7DA7ED8E6BD617ED0?sequence=1 (accessed on 27 July 2020).
- Anderson, C.; Breithaupt, L.; Marais, A.D.; Rastas, C.; Richman, A.; Barclay, L.; Brewer, N.T.; Smith, J.S. Acceptability and ease of use of mailed HPV self-collection among infrequently screened women in North Carolina. Sex. Transm. Infect. 2018, 94, 131–137. [Google Scholar] [CrossRef]
- Polman, N.J.; Snijders, P.; Kenter, G.; Berkhof, J.; Meijer, C. HPV-based cervical screening: Rationale, expectations and future perspectives of the new Dutch screening programme. Prev. Med. 2019, 119, 108–117. [Google Scholar] [CrossRef]
| Aged 25–35 N = 117 (%) | Aged ≥ 45 N = 60 (%) | Total N = 177 (%) | |
|---|---|---|---|
| Age | |||
| Mean {S.D.; Range} | 30 {1.8; 27–35} | 57 {5.8; 45–68} | 39 {13.1; 27–68} |
| Marital status | |||
| Single/divorced/widow | 70 (59.8) | 11 (18.3) | 81 (45.8) |
| Married/cohabited | 47 (40.2) | 49 (81.7) | 96 (54.2) |
| Highest Education Attainment | |||
| Primary or below | --- | 8 (13.3) | 8 (4.5) |
| Secondary | --- | 39 (65.0) | 39 (22.0) |
| Tertiary or above | 117 (100.0) | 13 (21.7) | 130 (73.5) |
| Religious Belief | |||
| Yes | 34 (29.1) | 23 (38.3) | 57 (32.2) |
| No | 83 (70.9) | 37 (61.7) | 120 (67.8) |
| Employment status | |||
| Full-time/part-time | 116 (99.1) | 22 (36.7) | 138 (78.0) |
| Unemployed | 0 (0.0) | 4 (6.7) | 4 (2.2) |
| Retired/Housewife | 1 (0.9) | 34 (56.7) | 35 (19.8) |
| Screening patterns (Pap smear) | |||
| Never screened | 63 (54.0) | 12 (20.0) | 75 (42.4) |
| Under-screened | 4 (3.0) | 17 (28.0) | 21 (11.9) |
| Regularly screened | 50 (43.0) | 31 (52.0) | 81 (45.7) |
| Returned HPV Self-Sampling Kit for Laboratory Test | ||||||
|---|---|---|---|---|---|---|
| Aged 25–35 N = 86 (Row%) | p-Value * | Aged ≥ 45 N = 43 (Row%) | p-Value * | Total N = 129 (Row%) | p-Value * | |
| Demographics | ||||||
| Age | ||||||
| Mean (S.D.) | 30.3 (1.8) | 0.734 | 56 (6.2) | 0.254 | 38.9 (12.9) | 0.609 |
| Marital status | ||||||
| Single/divorced/widow | 50 (70.4) | 0.348 | 8 (72.7) | 0.931 | 58 (70.7) | 0.550 |
| Married/cohabited | 36 (78.3) | 35 (71.4) | 71 (74.7) | |||
| Highest Education attainment | ||||||
| Primary or below | 0 (0.0) | N/A | 6 (75.0) | 0.913 | 6 (75.0) | 0.900 |
| Secondary | 0 (0.0) | 27 (69.2) | 27 (69.2) | |||
| Tertiary or above | 86 (73.5) | 10 (76.9) | 96 (73.8) | |||
| Religious Belief | ||||||
| Yes | 30 (88.2) | 0.022 # | 15 (65.2) | 0.382 | 45 (78.9) | 0.211 |
| No | 56 (67.5) | 28 (75.7) | 84 (70.0) | |||
| Employment status | ||||||
| Full-time/part-time | 86 (74.1) | 0.265 | 19 (86.4) | 0.135 | 105 (76.1) | 0.155 |
| Unemployed | 0 (0.0) | 3 (75.0) | 3 (75.0) | |||
| Retired/Housewife | 0 (0.0) | 21 (61.8) | 21 (60.0) | |||
| Lifestyle behaviour | ||||||
| Age at first sexual intercourse | ||||||
| ≤17 | 8 (88.9) | 0.680 | 4 (100.0) | 0.566 | 12 (92.3) | 0.300 |
| ≥18 | 66 (78.6) | 39 (70.9) | 105 (75.5) | |||
| Number of lifetime sexual partner | ||||||
| ≥2 | 31 (79.5) | 0.987 | 16 (80.0) | 0.539 | 47 (79.7) | 0.251 |
| ≤1 | 51 (72.9) | 27 (69.2) | 78 (71.6) | |||
| Frequent sexual activity & | ||||||
| Active | 54 (81.8) | 0.400 | 12 (66.7) | 0.446 | 66 (78.6) | 0.646 |
| Occasional | 20 (74.1) | 29 (76.3) | 49 (75.4) | |||
| Condom use & | ||||||
| Never/Sometimes | 31 (75.6) | 0.516 | 17 (65.4) | 0.534 | 48 (71.6) | 0.141 |
| Always/Often | 43 (82.7) | 9 (81.8) | 52 (82.5) | |||
| Post-coital bleeding & | ||||||
| Yes | 12 (100.0) | 0.117 | 2 (50.0) | 0.305 | 14 (87.5) | 0.522 |
| No | 62 (76.5) | 24 (75.0) | 86 (76.1) | |||
| Urethritis & | ||||||
| Yes | 10 (90.9) | 0.284 | 7 (77.8) | 0.720 | 17 (85.0) | 0.286 |
| No | 76 (71.7) | 36 (72.0) | 112 (71.8) | |||
| STD | ||||||
| Yes | 0 (0.0) | N/A | 3 (100.0) | 0.551 | 3 (100.0) | 0.564 |
| No | 86 (73.5) | 40 (70.2) | 126 (72.4) | |||
| Screening patterns (Pap) | ||||||
| Never/Under-screened | 48 (72.7) | 0.916 | 22 (78.6) | 0.234 | 71 (74.7) | 0.517 |
| Regularly screened | 37 (74.0) | 20 (64.5) | 57 (70.4) | |||
| Perception of Handling Self-Sampling | Aged 25–35 N = 86 (%) ^ | Aged ≥45 N = 39 (%) ^ | p-Value |
|---|---|---|---|
| Ease of use | |||
| Easy to perform | 57 (66.3) | 32 (82.1) | 0.071 |
| Easy to handle | 67 (77.9) | 33 (84.6) | 0.385 |
| Handling procedure | |||
| To collect self-samples correctly | 51 (59.3) | 26 (66.7) | 0.433 |
| Confidence in handling procedure | 69 (80.2) | 32 (82.1) | 0.811 |
| Beneficial to health | 81 (94.2) | 37 (94.9) | 0.877 |
| Convenience | 74 (86.0) | 37 (94.9) | 0.058 |
| Trusting the test results | 62 (72.1) | 29 (74.4) | 0.792 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wong, E.L.-Y.; Cheung, A.W.-L.; Wong, A.Y.-K.; Chan, P.K.-S. Acceptability and Feasibility of HPV Self-Sampling as an Alternative Primary Cervical Cancer Screening in Under-Screened Population Groups: A Cross-Sectional Study. Int. J. Environ. Res. Public Health 2020, 17, 6245. https://doi.org/10.3390/ijerph17176245
Wong EL-Y, Cheung AW-L, Wong AY-K, Chan PK-S. Acceptability and Feasibility of HPV Self-Sampling as an Alternative Primary Cervical Cancer Screening in Under-Screened Population Groups: A Cross-Sectional Study. International Journal of Environmental Research and Public Health. 2020; 17(17):6245. https://doi.org/10.3390/ijerph17176245
Chicago/Turabian StyleWong, Eliza Lai-Yi, Annie Wai-Ling Cheung, Amy Yuen-Kwan Wong, and Paul Kay-Sheung Chan. 2020. "Acceptability and Feasibility of HPV Self-Sampling as an Alternative Primary Cervical Cancer Screening in Under-Screened Population Groups: A Cross-Sectional Study" International Journal of Environmental Research and Public Health 17, no. 17: 6245. https://doi.org/10.3390/ijerph17176245





