Impact of a Gender-Neutral HPV Vaccination Program in Men Who Have Sex with Men (MSM)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Computational Network Model Building
- The global frequencies of the intercourses per age group and time step.
- The average recovery time from HPV 6/11 infection.
- Two parameters determining if the HPV 6/11 infection is transmitted from a man or woman to his/her partner (force of infection).
2.2. Scenarios to Be Simulated
- Scenario 1: Only girls were vaccinated with a coverage of 70% (current vaccination program in Spain).
- Scenario 2: 70% of both girls and boys were vaccinated.
- Scenario 3: 70% of girls were vaccinated and 20% of MSM 18 to 45 years starting 15 years after the girls’ program (to resemble most of the programs).
2.3. Sensitivity Analysis
3. Results
3.1. Community Effect in the Heterosexual Population
3.2. Impact on Men Who Have Sex with Men
3.3. Averted HPV 6/11 Infections
3.4. Sensitivity Analysis
- Case base. This is the current situation, with the percentage of MSM equal to 3.88%, and the current program to vaccinate young girls with a coverage of 70%.
- Case 1. It is the case base increasing the percentage of MSM up to 10%.
4. Discussion
Limitations of the Model
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boehmer, U.; Cooley, T.P.; Clark, M.A. Cancer and men who have sex with men: A systematic review. Lancet Oncol. 2012, 13, e545–e553. [Google Scholar] [CrossRef]
- World Health Organization, Director-General. Accelerating Cervical Cancer Elimination Cervical Cancer: A Global Public Health Priority; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Serrano, B.; Brotons, M.; Bosch, F.X.; Bruni, L. Epidemiology and burden of HPV-related disease. Best Pract. Res. Clin. Obstet. Gynaecol. 2018, 47, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, A.R.; Anic, G.; Nyitray, A.G. Epidemiology and pathology of HPV disease in males. Gynecol. Oncol. 2010, 117, S15–S19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mirghani, H.; Sturgis, E.M.; Aupérin, A.; Monsonego, J.; Blanchard, P. Is there an increased risk of cancer among spouses of patients with an HPV-related cancer: A systematic review. Oral Oncol. 2017, 67, 138–145. [Google Scholar] [CrossRef]
- Brisson, M.; Bénard, É.; Drolet, M.; Bogaards, J.A.; Baussano, I.; Vänskä, S.; Jit, M.; Boily, M.C.; Smith, M.A.; Berkhof, J.; et al. Population-level impact, herd immunity, and elimination after human papillomavirus vaccination: A systematic review and meta-analysis of predictions from transmission-dynamic models. Lancet Public Health 2016, 1, e8–e17. [Google Scholar] [CrossRef] [Green Version]
- Datta, S.; Pink, J.; Medley, G.F.; Petrou, S.; Staniszewska, S.; Underwood, M.; Sonnenberg, P.; Keeling, M.J. Assessing the cost-effectiveness of HPV vaccination strategies for adolescent girls and boys in the UK. BMC Infect. Dis. 2019, 19, 552. [Google Scholar] [CrossRef] [Green Version]
- Elam-Evans, L.D.; Yankey, D.; Singleton, J.A.; Sterrett, N.; Markowitz, L.E.; Williams, C.L.; Fredua, B.; McNamara, L.; Stokley, S. National, Regional, State, and Selected Local Area Vaccination Coverage Among Adolescents Aged 13–17 Years—United States, 2019. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 1109–1116. [Google Scholar] [CrossRef]
- Marty, R.; Roze, S.; Bresse, X.; Largeron, N.; Smith-Palmer, J. Estimating the clinical benefits of vaccinating boys and girls against HPV-related diseases in Europe. BMC Cancer 2013, 13, 10. [Google Scholar] [CrossRef] [Green Version]
- Pasmans, H.; Hoes, J.; Tymchenko, L.; de Melker, H.E.; van der Klis, F.R.M. Changes in HPV Seroprevalence from an Unvaccinated Toward a Girls-Only Vaccinated Population in the Netherlands. Cancer Epidemiol. Biomarkers Prev. 2020, 29, 2243–2254. [Google Scholar] [CrossRef]
- Barrera, J.; Greene, S.; Petyak, E.; Kenneson, S.; McGill, E.; Howell, H.; Billing, D.; Taylor, S.; Ewing, A.; Cull, J. Reported rationales for HPV vaccination vs. Non-vaccination among undergraduate and medical students in South Carolina. J. Am. Coll. Health 2019, 1–5. [Google Scholar] [CrossRef]
- Patel, C.; Brotherton, J.M.; Pillsbury, A.; Jayasinghe, S.; Donovan, B.; Macartney, K.; Marshall, H. The impact of 10 years of human papillomavirus (HPV) vaccination in Australia: What additional disease burden will a nonavalent vaccine prevent? Euro Surveill. 2018, 23, 1700737. [Google Scholar] [CrossRef]
- Drolet, M.; Bénard, É.; Boily, M.C.; Ali, H.; Baandrup, L.; Bauer, H.; Beddows, S.; Brisson, J.; Brotherton, J.M.L.; Cummings, T.; et al. Population-level impact and herd effects following human papillomavirus vaccination programmes: A systematic review and meta-analysis. Lancet Infect. Dis. 2015, 15, 565–580. [Google Scholar] [CrossRef] [Green Version]
- De La Fuente, J.; Hernandez Aguado, J.J.; San Martín, M.; Ramirez Boix, P.; Cedillo Gómez, S.; López, N. Estimating the epidemiological impact and cost-effectiveness profile of a nonavalent HPV vaccine in Spain. Hum. Vaccines Immunother. 2019, 15, 1949–1961. [Google Scholar] [CrossRef] [PubMed]
- Boiron, L.; Joura, E.; Largeron, N.; Prager, B.; Uhart, M. Estimating the cost-effectiveness profile of a universal vaccination programme with a nine-valent HPV vaccine in Austria. BMC Infect. Dis. 2016, 16, 153. [Google Scholar] [CrossRef] [Green Version]
- Burger, E.A.; Sy, S.; Nygård, M.; Kristiansen, I.S.; Kim, J.J. Prevention of HPV-related cancers in Norway: Cost-effectiveness of expanding the HPV vaccination program to include pre-adolescent boys. PLoS ONE 2014, 9, e89974. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daling, J.R.; Madeleine, M.M.; Johnson, L.G.; Schwartz, S.M.; Shera, K.A.; Wurscher, M.A.; Carter, J.J.; Porter, P.L.; Galloway, D.A.; McDougall, J.K. Human papillomavirus, smoking, and sexual practices in the etiology of anal cancer. Cancer 2004, 101, 270–280. [Google Scholar] [CrossRef]
- Machalek, D.A.; Poynten, M.; Jin, F.; Fairley, C.K.; Farnsworth, A.; Garland, S.M.; Hillman, R.J.; Petoumenos, K.; Roberts, J.; Tabrizi, S.N.; et al. Anal human papillomavirus infection and associated neoplastic lesions in men who have sex with men: A systematic review and meta-analysis. Lancet Oncol. 2012, 13, 487–500. [Google Scholar] [CrossRef]
- Machalek, D.A.; Jin, F.; Poynten, I.M.; Hillman, R.J.; Templeton, D.J.; Law, C.; Roberts, J.M.; Tabrizi, S.N.; Garland, S.M.; Farnsworth, A.; et al. Prevalence and risk factors associated with high-grade anal squamous intraepithelial lesions (HSIL)-AIN2 and HSIL-AIN3 in homosexual men. Papillomavirus Res. 2016, 2, 97–105. [Google Scholar] [CrossRef] [Green Version]
- Duncan, K.C.; Chan, K.J.; Chiu, C.G.; Montaner, J.S.G.; Coldman, A.J.; Cescon, A.; Au-Yeung, C.G.; Wiseman, S.M.; Hogg, R.S.; Press, N.M. HAART slows progression to anal cancer in HIV-infected MSM. AIDS 2015, 29, 305–311. [Google Scholar] [CrossRef]
- Chiu, C.G.; Smith, D.; Salters, K.A.; Zhang, W.; Kanters, S.; Milan, D.; Montaner, J.S.G.; Coldman, A.; Hogg, R.S.; Wiseman, S.M. Overview of cancer incidence and mortality among people living with HIV/AIDS in British Columbia, Canada: Implications for HAART use and NADM development. BMC Cancer 2017, 17, 270. [Google Scholar] [CrossRef] [Green Version]
- Parka, L.S.; Hernandez-Ramirez, R.U.; Silverberg, M.J.; Crothers, K.; Dubrow, R. Prevalence of non-HIV cancer risk factors in persons living with HIV/AIDS: A meta-analysis. AIDS 2016, 30, 273–291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Heer, E.; Hackl, M.; Ferlitsch, M.; Waldhoer, T.; Yang, L. Trends in incidence of anal cancer in Austria, 1983–2016. Wien. Klin. Wochenschr. 2020, 132, 438–443. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaturvedi, A.K.; Engels, E.A.; Pfeiffer, R.M.; Hernandez, B.Y.; Xiao, W.; Kim, E.; Jiang, B.; Goodman, M.T.; Sibug-Saber, M.; Cozen, W.; et al. Human papillomavirus and rising oropharyngeal cancer incidence in the United States. J. Clin. Oncol. 2011, 29, 4294–4301. [Google Scholar] [CrossRef]
- Rietbergen, M.; Leemans, C.; Bloemena, E.; Heideman, D.; Braakhuis, B.; Hesselink, A.; Witte, B.; Baatenburg de Jong, R.; Meijer, C.; Snijders, P.; et al. Increasing prevalence rates of HPV attributable oropharyngeal squamous cell carcinomas in the Netherlands as assessed by a validated test algorithm. Int J. Cancer. 2013, 132. [Google Scholar] [CrossRef]
- McDonald, S.A.; Qendri, V.; Berkhof, J.; de Melker, H.E.; Bogaards, J.A. Disease burden of human papillomavirus infection in the Netherlands, 1989–2014: The gap between females and males is diminishing. Cancer Causes Control 2017, 28, 203–214. [Google Scholar] [CrossRef]
- Pulte, D.; Brenner, H. Changes in Survival in Head and Neck Cancers in the Late 20th and Early 21st Century: A Period Analysis. Oncologist 2010, 15, 994–1001. [Google Scholar] [CrossRef] [Green Version]
- Baussano, I.; Lazzarato, F.; Brisson, M.; Franceschi, S. Human papillomavirus vaccination at a time of changing sexual behavior. Emerg. Infect. Dis. 2016, 22, 18–23. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Regan, D.G.; Ong, J.J.; Gambhir, M.; Chow, E.P.F.; Zou, H.; Law, M.; Hocking, J.; Fairley, C.K. Targeted human papillomavirus vaccination for young men who have sex with men in Australia yields significant population benefits and is cost-effective. Vaccine 2017, 35, 4923–4929. [Google Scholar] [CrossRef] [Green Version]
- Freidl, G.S.; Sonder, G.J.; Bovée, L.P.; Friesema, I.H.; van Rijckevorsel, G.G.; Ruijs, W.L.; van Schie, F.; Siedenburg, E.C.; Yang, J.; Vennema, H. Hepatitis A outbreak among men who have sex with men (MSM) predominantly linked with the EuroPride, the Netherlands, July 2016 to February 2017. Euro Surveill. 2017, 22. [Google Scholar] [CrossRef]
- Choi, Y.H.; Jit, M.; Gay, N.; Cox, A.; Garnett, G.P.; Edmunds, W.J. Transmission dynamic modelling of the impact of human papillomavirus vaccination in the United Kingdom. Vaccine 2010, 28, 4091–4102. [Google Scholar] [CrossRef] [PubMed]
- Qendri, V.; Bogaards, J.A.; Berkhof, J. Who Will Benefit from Expanding HPV Vaccination Programs to Boys? JNCI Cancer Spectr. 2018, 2, 76. [Google Scholar] [CrossRef] [PubMed]
- Instituto Nacional de Estadistica (INE) Encuesta de Salud y Hábitos Sexuales 2003 (Health and Sexual Habits Survey). Published 27 July 2004. Available online: https://www.ine.es/dyngs/INEbase/es/operacion.htm?c=Estadistica_C&cid=1254736176785&menu=resultados&idp=1254735573175#!tabs-1254736194731 (accessed on 1 November 2020).
- Díez-Domingo, J.; Sánchez-Alonso, V.; Villanueva, R.J.; Acedo, L.; Moraño, J.A.; Villanueva-Oller, J. Random network models to predict the long-term impact of HPV vaccination on genital warts. Viruses 2017, 9, 300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villanueva, R.J.; Hidalgo, J.I.; Cervigón, C.; Villanueva-Oller, J.; Cortés, J.C. Calibration of an agent-based simulation model to the data of women infected by Human Papillomavirus with uncertainty. Appl. Soft Comput. J. 2019, 80, 546–556. [Google Scholar] [CrossRef]
- Durex. Federación Estatal de Lesbianas Gays Transexuales y Bisexuales (FELGTB); Estudio Conducta Sexual Entre Homosexuales. Available online: http://www.sidastudi.org/es/registro/2c9391e41fb402cc011fb442355a4176 (accessed on 1 November 2020).
- Castellsagué, X.; Iftner, T.; Roura, E.; Vidart, J.A.; Kjaer, S.K.; Bosch, F.X.; Muñoz, N.; Palacios, S.; Rodriguez, M.S.M.; Serradell, L.; et al. Prevalence and genotype distribution of human papillomavirus infection of the cervix in Spain: The CLEOPATRE study. J. Med. Virol. 2012, 84, 947–956. [Google Scholar] [CrossRef]
- Garland, S.M.; Kjaer, S.K.; Muñoz, N.; Block, S.L.; Brown, D.R.; Dinubile, M.J.; Lindsay, B.R.; Kuter, B.J.; Perez, G.; Dominiak-Felden, G.; et al. Impact and effectiveness of the quadrivalent human papillomavirus vaccine: A systematic review of 10 years of real-world experience. Clin. Infect. Dis. 2016, 63, 519–527. [Google Scholar] [CrossRef]
- Acedo, L.; Cortés, J.; Díez-Domingo, J.; Sánchez-Alonso, V.; Tuells, J.; Villanueva, R. Uncertainty and sensitivity of the sexual behavior changes to the current human papillomavirus vaccination campaign in Spain. Math. Methods Appl. Sci. 2020. [Google Scholar] [CrossRef]
- Acedo, L.; Moraño, J.A.; Villanueva, R.J.; Villanueva-Oller, J.; Díez-Domingo, J. Using random networks to study the dynamics of respiratory syncytial virus (RSV) in the Spanish region of Valencia. Math. Comput. Model. 2011, 54, 1650–1654. [Google Scholar] [CrossRef] [Green Version]
- Murray, J.D. Mathematical Biology, 3rd ed.; Springer: New York, NY, USA, 2002. [Google Scholar]
- Bogaards, J.A.; Mooij, S.H.; Xiridou, M.; Schim Van Derloeff, M.F. Potential effectiveness of prophylactic hpv immunization for men who have sex with men in the Netherlands: A multi-modelapproach. PLoS Med. 2019, 16, e1002756. [Google Scholar] [CrossRef]
- Gao, S.; Martcheva, M.; Miao, H.; Rong, L. A Dynamic Model to Assess Human Papillomavirus Vaccination Strategies in a Heterosexual Population Combined with Men Who have Sex with Men. Bull. Math. Biol. 2021, 83. [Google Scholar] [CrossRef]
- Sonnenberg, P.; Tanton, C.; Mesher, D.; King, E.; Beddows, S.; Field, N.; Mercer, C.H.; Soldan, K.; Johnson, A.M. Epidemiology of genital warts in the British population: Implications for HPV vaccination programmes. Sex. Transm. Infect. 2019, 95, 386–390. [Google Scholar] [CrossRef] [PubMed]


| Males | ||||||
| Age | 0 LSP | 1 LSP | 2 LSP | 3–4 LSP | 5–9 LSP | 10 or more LSP |
| 14–29 | 0.107 | 0.207 | 0.131 | 0.225 | 0.168 | 0.162 |
| 30–39 | 0.027 | 0.225 | 0.128 | 0.21 | 0.17 | 0.24 |
| 40–65 | 0.019 | 0.268 | 0.14 | 0.193 | 0.163 | 0.217 |
| Females | ||||||
| Age | 0 LSP | 1 LSP | 2 LSP | 3–4 LSP | 5–9 LSP | 10 or more LSP |
| 14–29 | 0.138 | 0.43 | 0.186 | 0.158 | 0.056 | 0.032 |
| 30–39 | 0.029 | 0.501 | 0.168 | 0.177 | 0.077 | 0.048 |
| 40–65 | 0.017 | 0.652 | 0.138 | 0.118 | 0.039 | 0.036 |
| Years | |||||
|---|---|---|---|---|---|
| 10 | 20 | 30 | 40 | 50 | |
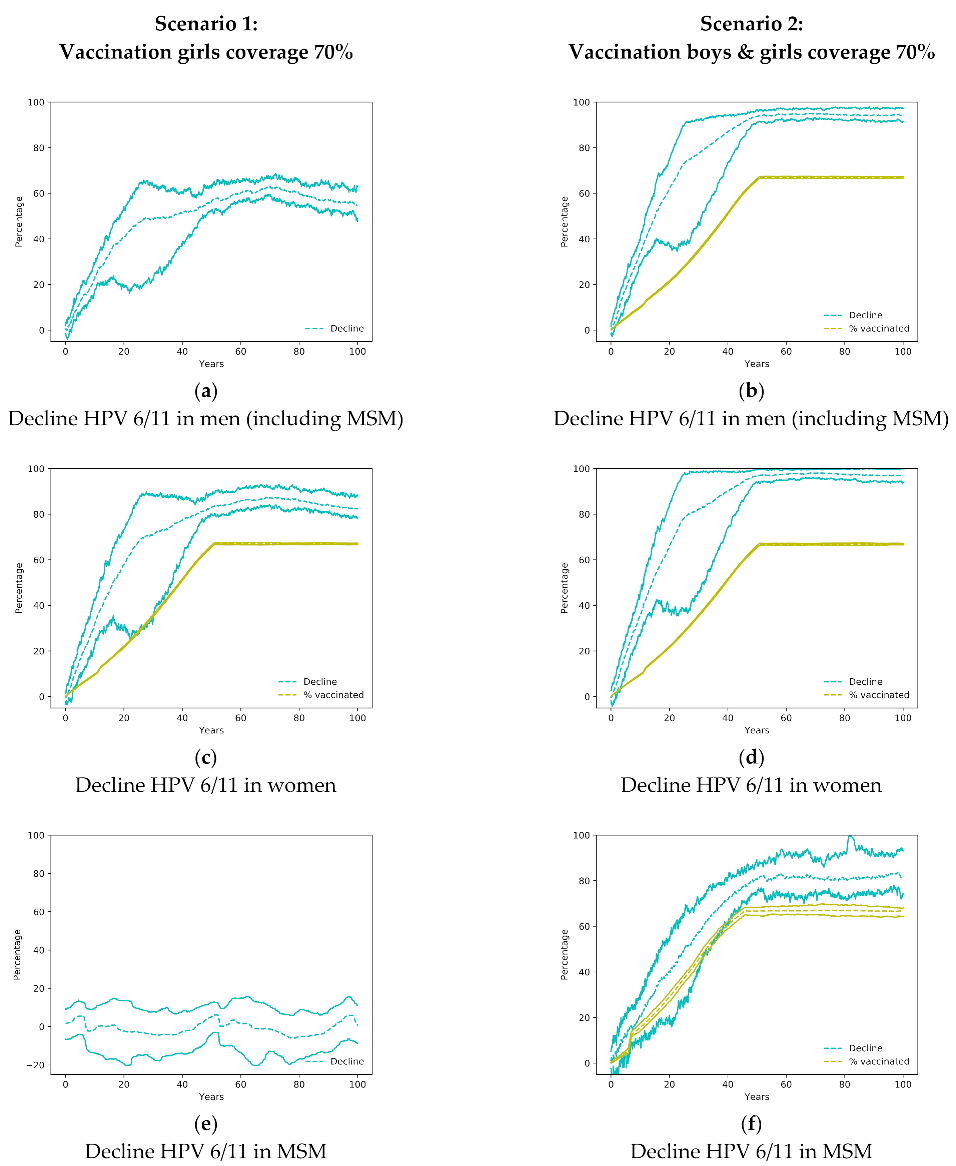
| Men * (Scenario 1) | 9% | 26% | 41% | 48% | 55% |
| Men * (Scenario 2) | 16% | 49% | 73% | 86% | 93% |
| MSM (Scenario 2) | 10% | 38% | 57% | 72% | 79% |
| Women (Scenario 1) | 14% | 45% | 66% | 75% | 82% |
| Women (Scenario 2) | 18% | 54% | 80% | 89% | 97% |
| % of vaccinated women (both scenarios) | 9.5% | 21.6% | 35.3% | 51% | 65.7% |
| % of vaccinated men (Scenario 2) | 10% | 21% | 34.6% | 51% | 65.7% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Díez-Domingo, J.; Sánchez-Alonso, V.; Villanueva, R.-J.; Acedo, L.; Tuells, J. Impact of a Gender-Neutral HPV Vaccination Program in Men Who Have Sex with Men (MSM). Int. J. Environ. Res. Public Health 2021, 18, 963. https://doi.org/10.3390/ijerph18030963
Díez-Domingo J, Sánchez-Alonso V, Villanueva R-J, Acedo L, Tuells J. Impact of a Gender-Neutral HPV Vaccination Program in Men Who Have Sex with Men (MSM). International Journal of Environmental Research and Public Health. 2021; 18(3):963. https://doi.org/10.3390/ijerph18030963
Chicago/Turabian StyleDíez-Domingo, Javier, Víctor Sánchez-Alonso, Rafael-J. Villanueva, Luis Acedo, and José Tuells. 2021. "Impact of a Gender-Neutral HPV Vaccination Program in Men Who Have Sex with Men (MSM)" International Journal of Environmental Research and Public Health 18, no. 3: 963. https://doi.org/10.3390/ijerph18030963









