Biocompatibility of a HA/β-TCP/C Scaffold as a Pulp-Capping Agent for Vital Pulp Treatment: An In Vivo Study in Rat Molars
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals and Surgical Procedure
2.2. Experimental Groups
2.3. Microscope Observation
2.4. Histological Evaluation
2.5. Statistical Analysis
3. Results
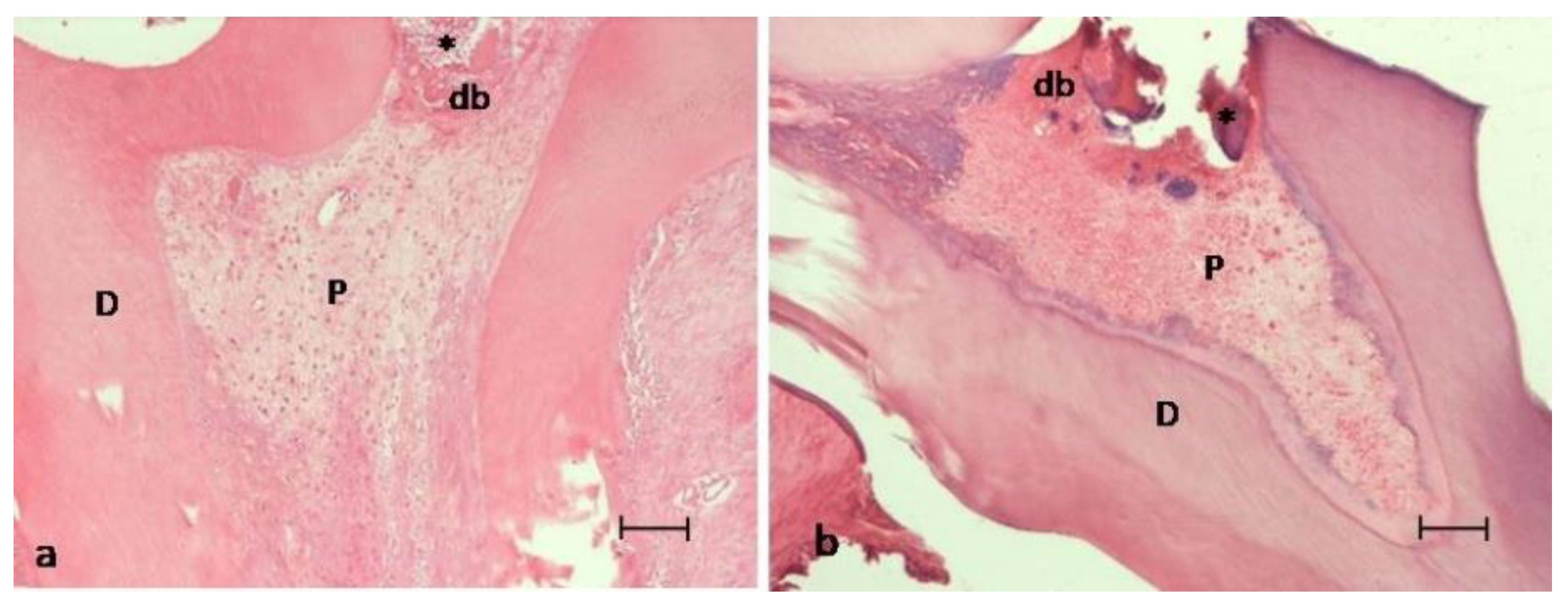
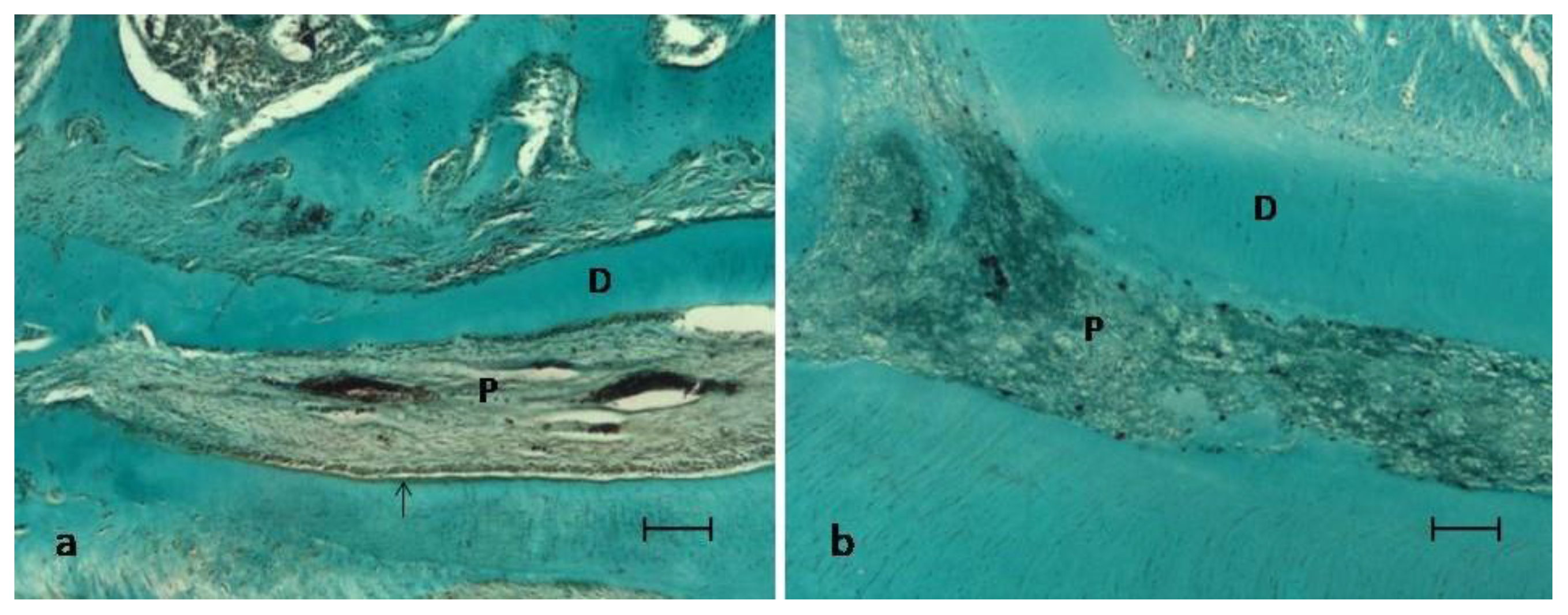
Histological Evaluation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| β-TCP | Beta-tricalcium phosphate |
| HA | Hydroxyapatite |
| C | Collagen |
| MTA | Mineral Trioxide Aggregate |
| HA/β-TCP/C | triphasic calcium phosphate ceramic with Hidoxiapatite, beta-tricalciumphosphate and Collagen |
| TCP | Tricalcium Phosphate |
| BCP | Biphasic calcium phosphate ceramic |
| HA/β-TCP | biphasic calcium phosphate ceramic with Hidoxiapatite and beta-tricalciumphosphate |
| DSPP | dentin sialophosphoprotein |
| db | dentinal bridge |
| P | viable pulp without necrosis |
| D | dentin |
| VEGF | vascular endothelial growth factor |
| VPT | vital pulp treatment |
References
- Bucholz, R.W. Nonallograft osteoconductive bone graft substitutes. Clin. Orthop. Relat. Res. 2002, 44–52. [Google Scholar] [CrossRef]
- Su, Y.F.; Lin, C.C.; Huang, T.H.; Chou, M.Y.; Yang, J.J.; Shie, M.Y. Osteogenesis and angiogenesis properties of dental pulp cell on novel injectable tricalcium phosphate cement by silica doped. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 42, 672–680. [Google Scholar] [CrossRef] [PubMed]
- Kao, C.T.; Huang, T.H.; Chen, Y.J.; Hung, C.J.; Lin, C.C.; Shie, M.Y. Using calcium silicate to regulate the physicochemical and biological properties when using β-tricalcium phosphate as bone cement. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 43, 126–134. [Google Scholar] [CrossRef]
- Kouhestani, F.; Dehabadi, F.; Hasan Shahriari, M.; Motamedian, S.R. Allogenic vs. synthetic granules for bone tissue engineering: An in vitro study. Prog. Biomater. 2018, 7, 133–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ogose, A.; Kondo, N.; Umezu, H.; Hotta, T.; Kawashima, H.; Tokunaga, K.; Ito, T.; Kudo, N.; Hoshino, M.; Gu, W.; et al. Histological assessment in grafts of highly purified beta-tricalcium phosphate (OSferion) in human bones. Biomaterials 2006, 27, 1542–1549. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Scott, M.; Rubio, A. α- and β-tricalcium phosphate: A density functional study. Phys. Rev. B 2003, 68, 205205. [Google Scholar] [CrossRef]
- Camargo, C.H.R.; Gomes, L.C.L.; França, M.C.M.; Bittencourt, T.S.; Valera, M.C.; Camargo, S.E.A.; Bottino, M.C. Incorporating N-acetylcysteine and tricalcium phosphate into epoxy resin-based sealer improved its biocompatibility and adhesiveness to radicular dentine. Dent. Mater. 2019, 35, 1750–1756. [Google Scholar] [CrossRef]
- Jitaru, S.; Hodisan, I.; Timis, L.; Lucian, A.; Bud, M. The use of bioceramics in endodontics—Literature review. Clujul Med. 2016, 89, 470–473. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.B.; Park, S.J.; Kim, H.H.; Kwon, Y.S.; Lee, K.W.; Min, K.S. Physical properties and biological/odontogenic effects of an experimentally developed fast-setting α-tricalcium phosphate-based pulp capping material. BMC Oral Health 2014, 14, 87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shayegan, A.; Petein, M.; Vanden Abbeele, A. The use of beta-tricalcium phosphate, white MTA, white Portland cement and calcium hydroxide for direct pulp capping of primary pig teeth. Dent. Traumatol. 2009, 25, 413–419. [Google Scholar] [CrossRef]
- Zaen El-Din, A.M.; Hamama, H.H.; Abo El-Elaa, M.A.; Grawish, M.E.; Mahmoud, S.H.; Neelakantan, P. The effect of four materials on direct pulp capping: An animal study. Aust. Endod. J. 2020, 46, 249–256. [Google Scholar] [CrossRef]
- Tonomura, A.; Mizuno, D.; Hisada, A.; Kuno, N.; Ando, Y.; Sumita, Y.; Honda, M.J.; Satomura, K.; Sakurai, H.; Ueda, M.; et al. Differential effect of scaffold shape on dentin regeneration. Ann. Biomed. Eng. 2010, 38, 1664–1671. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, Y.M.; Lim, Y.J.; Kim, B. Ridge Augmentation Using β-Tricalcium Phosphate and Biphasic Calcium Phosphate Sphere with Collagen Membrane in Chronic Pathologic Extraction Sockets with Dehiscence Defect: A Pilot Study in Beagle Dogs. Materials 2020, 13, 1452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lobo, S.E.; Glickman, R.; da Silva, W.N.; Arinzeh, T.L.; Kerkis, I. Response of stem cells from different origins to biphasic calcium phosphate bioceramics. Cell Tissue Res. 2015, 361, 477–495. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pek, Y.S.; Gao, S.; Arshad, M.S.; Leck, K.J.; Ying, J.Y. Porous collagen-apatite nanocomposite foams as bone regeneration scaffolds. Biomaterials 2008, 29, 4300–4305. [Google Scholar] [CrossRef]
- Cao, S.; Han, J.; Sharma, N.; Msallem, B.; Jeong, W.; Son, J.; Kunz, C.; Kang, H.W.; Thieringer, F.M. In Vitro Mechanical and Biological Properties of 3D Printed Polymer Composite and β-Tricalcium Phosphate Scaffold on Human Dental Pulp Stem Cells. Materials 2020, 13, 3057. [Google Scholar] [CrossRef] [PubMed]
- Fahimipour, F.; Dashtimoghadam, E.; Rasoulianboroujeni, M.; Yazdimamaghani, M.; Khoshroo, K.; Tahriri, M.; Yadegari, A.; Gonzalez, J.A.; Vashaee, D.; Lobner, D.C.; et al. Collagenous matrix supported by a 3D-printed scaffold for osteogenic differentiation of dental pulp cells. Dent. Mater. 2018, 34, 209–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Wang, L.; Su, Q.; Ye, L.; Zhou, X.; Song, D.; Huang, D. Highly Proliferative Immortalized Human Dental Pulp Cells Retain the Odontogenic Phenotype when Combined with a Beta-Tricalcium Phosphate Scaffold and BMP2. Stem Cells Int. 2020, 2020, 4534128. [Google Scholar] [CrossRef]
- Fei, L.; Wang, C.; Xue, Y.; Lin, K.; Chang, J.; Sun, J. Osteogenic differentiation of osteoblasts induced by calcium silicate and calcium silicate/β-tricalcium phosphate composite bioceramics. J. Biomed. Mater. Res. B Appl. Biomater. 2012, 100, 1237–1244. [Google Scholar] [CrossRef]
- Miyamoto, S.; Shinmyouzu, K.; Miyamoto, I.; Takeshita, K.; Terada, T.; Takahashi, T. Histomorphometric and immunohistochemical analysis of human maxillary sinus-floor augmentation using porous β-tricalcium phosphate for dental implant treatment. Clin. Oral. Implant. Res. 2013, 24 (Suppl. A100), 134–138. [Google Scholar] [CrossRef] [Green Version]
- Musu, D.; Shemesh, H.; Boccuzzi, M.; Dettori, C.; Cotti, E. Correction to: The effectiveness of ultrasound examination to assess the healing process of bone lesions of the jaws: A systematic review. Clin. Oral. Investig. 2020, 24, 4663. [Google Scholar] [CrossRef] [PubMed]
- Da Rosa, W.L.O.; Cocco, A.R.; Silva, T.M.D.; Mesquita, L.C.; Galarca, A.D.; Silva, A.F.D.; Piva, E. Current trends and future perspectives of dental pulp capping materials: A systematic review. J. Biomed. Mater. Res. B Appl. Biomater. 2018, 106, 1358–1368. [Google Scholar] [CrossRef] [PubMed]
- Hosoya, N.; Takigawa, T.; Horie, T.; Maeda, H.; Yamamoto, Y.; Momoi, Y.; Yamamoto, K.; Okiji, T. A review of the literature on the efficacy of mineral trioxide aggregate in conservative dentistry. Dent. Mater. J. 2019, 38, 693–700. [Google Scholar] [CrossRef] [Green Version]
- Zhu, C.; Ju, B.; Ni, R. Clinical outcome of direct pulp capping with MTA or calcium hydroxide: A systematic review and meta-analysis. Int. J. Clin. Exp. Med. 2015, 8, 17055–17060. [Google Scholar]
- Youssef, A.R.; Emara, R.; Taher, M.M.; Al-Allaf, F.A.; Almalki, M.; Almasri, M.A.; Siddiqui, S.S. Effects of mineral trioxide aggregate, calcium hydroxide, biodentine and Emdogain on osteogenesis, Odontogenesis, angiogenesis and cell viability of dental pulp stem cells. BMC Oral Health 2019, 19, 133. [Google Scholar] [CrossRef] [PubMed]
- Ferracane, J.L.; Cooper, P.R.; Smith, A.J. Can interaction of materials with the dentin-pulp complex contribute to dentin regeneration? Odontology 2010, 98, 2–14. [Google Scholar] [CrossRef]
- Bjørndal, L.; Simon, S.; Tomson, P.L.; Duncan, H.F. Management of deep caries and the exposed pulp. Int. Endod. J. 2019, 52, 949–973. [Google Scholar] [CrossRef]
- Anthonappa, R.P.; King, N.M.; Martens, L.C. Is there sufficient evidence to support the long-term efficacy of mineral trioxide aggregate (MTA) for endodontic therapy in primary teeth? Int. Endod. J. 2013, 46, 198–204. [Google Scholar] [CrossRef] [Green Version]
- Cvikl, B.; Hess, S.C.; Miron, R.J.; Agis, H.; Bosshardt, D.; Attin, T.; Schmidlin, P.R.; Lussi, A. Response of human dental pulp cells to a silver-containing PLGA/TCP-nanofabric as a potential antibacterial regenerative pulp-capping material. BMC Oral Health 2017, 17, 57. [Google Scholar] [CrossRef] [Green Version]
- Gronthos, S.; Brahim, J.; Li, W.; Fisher, L.W.; Cherman, N.; Boyde, A.; DenBesten, P.; Robey, P.G.; Shi, S. Stem cell properties of human dental pulp stem cells. J. Dent. Res. 2002, 81, 531–535. [Google Scholar] [CrossRef]
- Zhang, W.; Walboomers, X.F.; van Osch, G.J.; van den Dolder, J.; Jansen, J.A. Hard tissue formation in a porous HA/TCP ceramic scaffold loaded with stromal cells derived from dental pulp and bone marrow. Tissue Eng. Part. A 2008, 14, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Girones, J.; Alcaina-Lorente, A.; Ortiz-Ruiz, C.; Ortiz-Ruiz, E.; Pecci-Lloret, M.P.; Rodriguez-Lozano, F.J.; Martinez, C.M.; Ortiz-Ruiz, A.J. Melatonin as an Agent for Direct Pulp-Capping Treatment. Int. J. Env. Res. Public Health 2020, 17, 1043. [Google Scholar] [CrossRef] [Green Version]
- Cai, J.; Zeng, D. Sample size/power calculation for case-cohort studies. Biometrics 2004, 60, 1015–1024. [Google Scholar] [CrossRef]
- Hørsted, P.; El Attar, K.; Langeland, K. Capping of monkey pulps with Dycal and a Ca-eugenol cement. Oral Surg. Oral Med. Oral Pathol. 1981, 52, 531–553. [Google Scholar] [CrossRef]
- Fuks, A.B.; Jones, P.C.; Michaeli, Y.; Bimstein, E. Pulp response to collagen and glutaraldehyde in pulpotomized primary teeth of baboons. Pediatr. Dent. 1991, 13, 142–150. [Google Scholar]
- Dammaschke, T. Rat molar teeth as a study model for direct pulp capping research in dentistry. Lab. Anim. 2010, 44, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Bal, C.; Oztas, N.; Cincik, M.; Baris, E. Immunolocalization of fibronectin during reparative dentinogenesis in rat molor teeth after pulp capping with mineral trioxide aggregate or calcium hydroxide. N. Y. State Dent. J. 2011, 77, 36–42. [Google Scholar] [PubMed]
- Morimoto, S.; Anada, T.; Honda, Y.; Suzuki, O. Comparative study on in vitro biocompatibility of synthetic octacalcium phosphate and calcium phosphate ceramics used clinically. Biomed. Mater. 2012, 7, 45020. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.J.; Murray, P.E.; Sloan, A.J.; Matthews, J.B.; Zhao, S. Trans-dentinal stimulation of tertiary dentinogenesis. Adv. Dent. Res. 2001, 15, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Paranjpe, A.; Smoot, T.; Zhang, H.; Johnson, J.D. Direct contact with mineral trioxide aggregate activates and differentiates human dental pulp cells. J. Endod. 2011, 37, 1691–1695. [Google Scholar] [CrossRef] [Green Version]
- Tran, X.V.; Salehi, H.; Truong, M.T.; Sandra, M.; Sadoine, J.; Jacquot, B.; Cuisinier, F.; Chaussain, C.; Boukpessi, T. Reparative Mineralized Tissue Characterization after Direct Pulp Capping with Calcium-Silicate-Based Cements. Materials 2019, 12, 2102. [Google Scholar] [CrossRef] [Green Version]
- Masuda-Murakami, Y.; Kobayashi, M.; Wang, X.; Yamada, Y.; Kimura, Y.; Hossain, M.; Matsumoto, K. Effects of mineral trioxide aggregate on the differentiation of rat dental pulp cells. Acta Histochem. 2010, 112, 452–458. [Google Scholar] [CrossRef]
- Atalayin, C.; Tezel, H.; Dagci, T.; Karabay Yavasoglu, N.U.; Oktem, G.; Kose, T. In vivo performance of different scaffolds for dental pulp stem cells induced for odontogenic differentiation. Braz. Oral Res. 2016, 30, e120. [Google Scholar] [CrossRef] [Green Version]
- Ando, Y.; Honda, M.J.; Ohshima, H.; Tonomura, A.; Ohara, T.; Itaya, T.; Kagami, H.; Ueda, M. The induction of dentin bridge-like structures by constructs of subcultured dental pulp-derived cells and porous HA/TCP in porcine teeth. Nagoya J. Med. Sci. 2009, 71, 51–62. [Google Scholar] [PubMed]
- Gu, Y.; Xie, X.; Zhuang, R.; Weir, M.D.; Oates, T.W.; Bai, Y.; Zhao, L.; Xu, H.H.K. A Biphasic Calcium Phosphate Cement Enhances Dentin Regeneration by Dental Pulp Stem Cells and Promotes Macrophages M2 Phenotype In Vitro. Tissue Eng. Part A 2021. [Google Scholar] [CrossRef] [PubMed]
- Higashi, T.; Okamoto, H. Influence of particle size of calcium phosphate ceramics as a capping agent on the formation of a hard tissue barrier in amputated dental pulp. J. Endod. 1996, 22, 281–283. [Google Scholar] [CrossRef]
- AbdulQader, S.T.; Rahman, I.A.; Thirumulu, K.P.; Ismail, H.; Mahmood, Z. Effect of biphasic calcium phosphate scaffold porosities on odontogenic differentiation of human dental pulp cells. J. Biomater. Appl. 2016, 30, 1300–1311. [Google Scholar] [CrossRef] [PubMed]
- Dioguardi, M.; Di Gioia, G.; Illuzzi, G.; Arena, C.; Caponio, V.C.A.; Caloro, G.A.; Zhurakivska, K.; Adipietro, I.; Troiano, G.; Lo Muzio, L. Inspection of the Microbiota in Endodontic Lesions. Dent. J. 2019, 7, 47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Pulp Inflammation | 0 | Absent of inflammation |
| 1 | Mild inflammation | |
| 2 | Moderate inflammation | |
| 3 | Severe inflammation | |
| 4 | Abscess | |
| Pulp Necrosis | 0 | Absence |
| 1 | Presence | |
| Dentinal bridge and reparative dentin formation | 0 | Presence |
| 1 | Absence | |
| Odontoblastic layer | 0 | Regular |
| 1 | Irregular | |
| 2 | Absence | |
| Fibrotic tissue | 0 | Absence |
| 1 | Presence |
| Criteria | Degree | MTA | HA/β-TCP/C | p Value |
|---|---|---|---|---|
| Pulp Inflammation | 0 | 100% | 100% | p = 0.124 |
| 1 | ||||
| 2 | ||||
| 3 | ||||
| 4 | ||||
| Pulp Necrosis | 0 | 100% | 100% | p = 0.124 |
| 1 | ||||
| Dentinal bridge and reparative dentin formation | 0 | 87.5% | 25% | p = 0.0005 * |
| 1 | 12.5% | 75% | ||
| Odontoblastic layer | 0 | 100% | 56.25% | p = 0.003 * |
| 1 | ||||
| 2 | 43.75% | |||
| Fibrotic tissue | 0 | 25% | p = 0.03 * | |
| 1 | 100% | 75% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guerrero-Gironés, J.; Alcaina-Lorente, A.; Ortiz-Ruiz, C.; Ortiz-Ruiz, E.; Pecci-Lloret, M.P.; Ortiz-Ruiz, A.J.; Rodríguez-Lozano, F.J.; Pecci-Lloret, M.R. Biocompatibility of a HA/β-TCP/C Scaffold as a Pulp-Capping Agent for Vital Pulp Treatment: An In Vivo Study in Rat Molars. Int. J. Environ. Res. Public Health 2021, 18, 3936. https://doi.org/10.3390/ijerph18083936
Guerrero-Gironés J, Alcaina-Lorente A, Ortiz-Ruiz C, Ortiz-Ruiz E, Pecci-Lloret MP, Ortiz-Ruiz AJ, Rodríguez-Lozano FJ, Pecci-Lloret MR. Biocompatibility of a HA/β-TCP/C Scaffold as a Pulp-Capping Agent for Vital Pulp Treatment: An In Vivo Study in Rat Molars. International Journal of Environmental Research and Public Health. 2021; 18(8):3936. https://doi.org/10.3390/ijerph18083936
Chicago/Turabian StyleGuerrero-Gironés, Julia, Antonia Alcaina-Lorente, Clara Ortiz-Ruiz, Eduardo Ortiz-Ruiz, María P. Pecci-Lloret, Antonio José Ortiz-Ruiz, Francisco Javier Rodríguez-Lozano, and Miguel R. Pecci-Lloret. 2021. "Biocompatibility of a HA/β-TCP/C Scaffold as a Pulp-Capping Agent for Vital Pulp Treatment: An In Vivo Study in Rat Molars" International Journal of Environmental Research and Public Health 18, no. 8: 3936. https://doi.org/10.3390/ijerph18083936







