Experimentally Measured Thermal Masses of Adsorption Heat Exchangers
Abstract
:1. Introduction
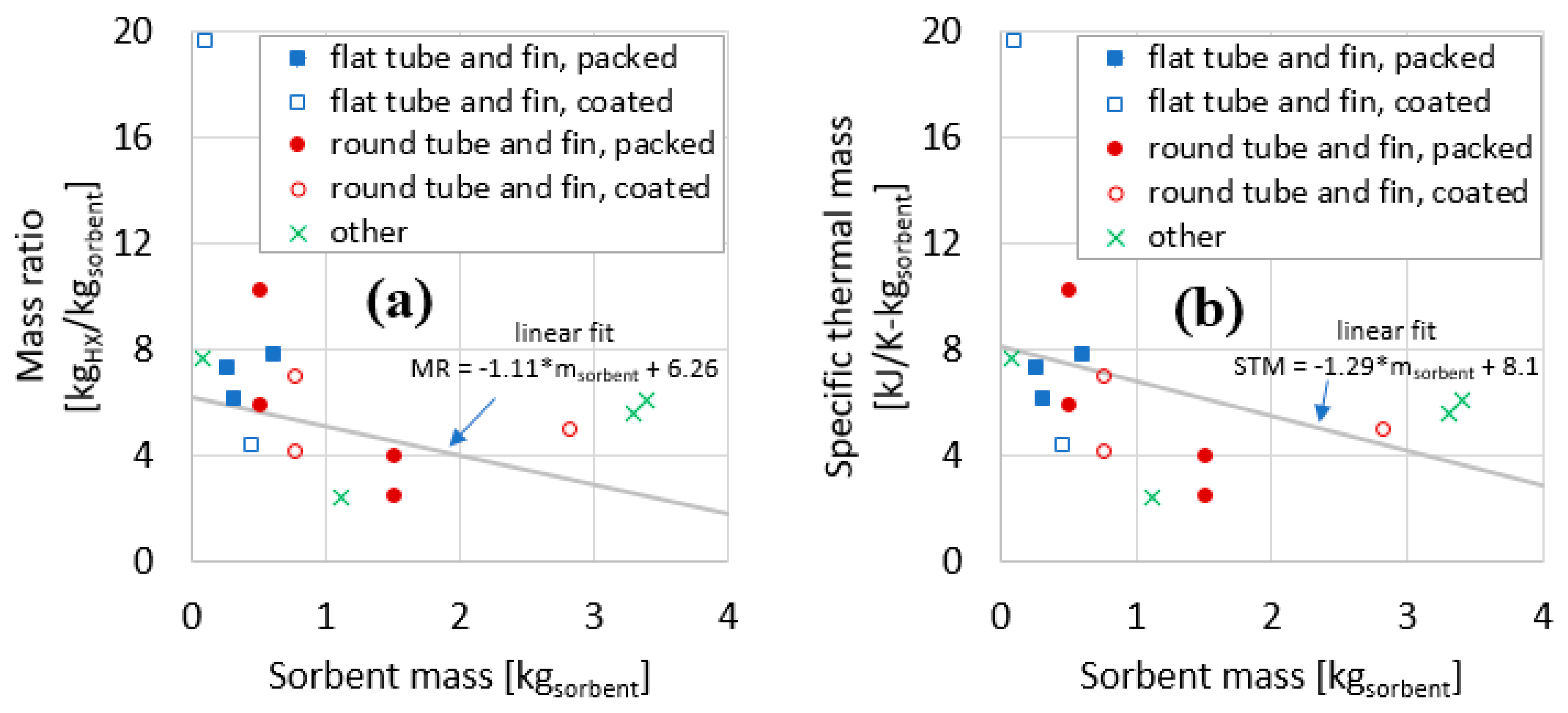
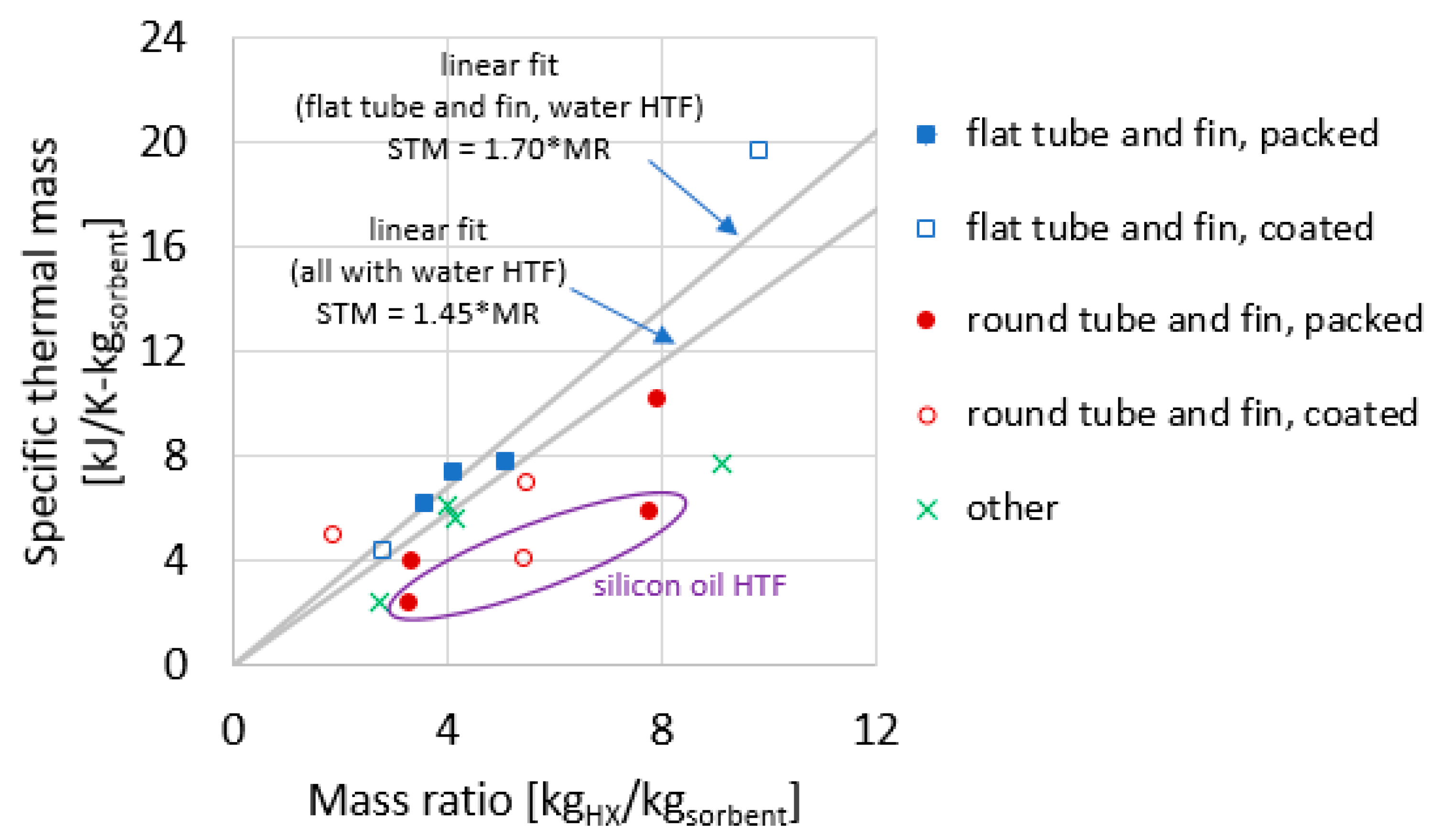
1.1. Mass Ratios of Heat Exchangers
1.2. Thermal Masses of Heat Exchangers
2. Methodology
2.1. The Definition of Heat Exchanger Thermal Mass
2.2. Definition of a Relevant Control Volume
2.3. The Role of Heat Transfer Fluid
2.4. The Role of Heat Exchanger Enclosures
3. Experimental Results for Specific Thermal Mass
3.1. Flat Tube–Fin—Packed (Water as Refrigerant)
3.2. Flat Tube–Fin—Packed (Ethanol as Refrigerant)
3.3. Flat Tube–Fin—Coated
3.4. Flat Tube–Fin—Coated
3.5. Round Tube–Corrugated Fin—Coated and Packed (Water as Refrigerant)
3.6. Modular Finned Tube
3.7. Shell–Tube
3.8. Plate–Shell
3.9. Fiber Heat Exchangers
3.10. Summary of Experimental Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Nomenclature
| A | area (m2) |
| AC | activated carbon |
| AT | approach temperature (K) |
| c | specific heat capacity (kJ1kg−1K−1) |
| ceffective | effective specific heat |
| HTS | high temperature salt |
| HX | heat exchanger |
| LTS | low temperature salt |
| m | mass (kg) |
| MR | mass ratio (kgHX/kgsorbent) |
| STM | specific thermal mass |
| TM | thermal mass (kJ/kg) |
| V | volume (m3) |
| Y | ratio of refrigerant (sorbate) mass to sorbent mass (kgref/kgsorbent) |
| Greek | |
| density (kg/m3) | |
| ω | humidity ratio (kgw/kgda) |
| Subscripts | |
| 0 | initial |
| HTF | heat transfer fluid |
| liq | liquid |
| mat | material (non-HTF, non-sorbent), typically metals, comprising the heat exchanger |
| total | total (thermal mass) |
| ref | refrigerant (sorbate) |
| sorb | sorbent |
References
- Ziegler, F. State of the art in sorption heat pumping and cooling technologies. Int. J. Refrig. 2002, 25, 450–459. [Google Scholar] [CrossRef]
- Wittstadt, U.; Füldner, G.; Vasta, S.; Volmer, R.; Bendix, P.; Schnabel, L.; Mittelbach, W. Adsorption heat pumps and chillers: Recent developments for materials and components. In Proceedings of the 12th International Energy Agency Heat Pump Conference, Rotterdam, The Netherlands, 15–18 May 2017. [Google Scholar]
- Critoph, R.E.; Metcalf, S.J. Specific cooling power intensification limits in ammonia–carbon adsorption refrigeration systems. Appl. Therm. Eng. 2004, 24, 661–678. [Google Scholar] [CrossRef]
- Goetz, V.; Elie, F.; Spinner, B. The structure and performance of single effect solid-gas chemical heat pumps. Heat Recover. Syst. CHP 1993, 13, 79–96. [Google Scholar] [CrossRef]
- Neveu, P. Performances d’un pilote de pompe à chaleur chimique solide/gaz de 20/50 Kw : analyse des flux engendrés par la thermicité de telles réactions. Ph.D. Thesis, Université Perpignan, Perpignan, France, 1990. [Google Scholar]
- Douss, N.; Meunier, F.E.; Sun, L.M. Predictive model and experimental results for a two-adsorber solid adsorption heat pump. Ind. Eng. Chem. Res. 1988, 27, 310–316. [Google Scholar] [CrossRef]
- Li, T.X.; Wang, R.Z.; Kiplagat, J.K.; Wang, L.W.; Oliveira, R.G. A conceptual design and performance analysis of a triple-effect solid-gas thermochemical sorption refrigeration system with internal heat recovery. Chem. Eng. Sci. 2009, 64, 3376–3384. [Google Scholar] [CrossRef]
- Demir, H.; Mobedi, M.; Ülkü, S. A review on adsorption heat pump: Problems and solutions. Renew. Sustain. Energy Rev. 2008, 12, 2381–2403. [Google Scholar] [CrossRef] [Green Version]
- Paul, B.K.; Lee, K.; Wang, H. Improving the Energy Efficiency of Adsorption Chillers by Intensifying Thermal Management Systems in Sorbent Beds. J. Manuf. Sci. Eng. Trans. ASME 2018, 140. [Google Scholar] [CrossRef]
- Gluesenkamp, K.R. Development and Analysis of Micro Polygeneration Systems and Adsorption Chillers. Ph.D. Thesis, University of Maryland, College Park, MD, USA, 2012. [Google Scholar]
- Zhu, C.; Gluesenkamp, K.R.; Yang, Z.; Blackman, C. Unified thermodynamic model to calculate COP of diverse sorption heat pump cycles: Adsorption, absorption, resorption, and multistep crystalline reactions. Int. J. Refrig. 2019, 99, 382–392. [Google Scholar] [CrossRef]
- Sharafian, A.; Mahdi, S.; Mehr, N.; Thimmaiah, C.; Huttema, W.; Bahrami, M. Effects of adsorbent mass and number of adsorber beds on the performance of a waste heat-driven adsorption cooling system for vehicle air conditioning applications. Energy 2016. [Google Scholar] [CrossRef]
- Sharafian, A.; Bahrami, M. Assessment of adsorber bed designs in waste-heat driven adsorption cooling systems for vehicle air conditioning and refrigeration. Renew. Sustain. Energy Rev. 2014, 30, 440–451. [Google Scholar] [CrossRef]
- Qian, S.; Gluesenkamp, K.; Hwang, Y.; Radermacher, R.; Chun, H.-H. Cyclic steady state performance of adsorption chiller with low regeneration temperature zeolite. Energy 2013, 60. [Google Scholar] [CrossRef]
- Rouhani, M. Sorption Thermal Energy Storage for Sustainable Heating and Cooling. Ph.D. Thesis, Simon Fraser University, Vancouver, Canada, 2019. [Google Scholar]
- Xu, J.; Oliveira, R.G.; Wang, R.Z. Resorption system with simultaneous heat and cold production. Int. J. Refrig. 2011, 34, 1262–1267. [Google Scholar] [CrossRef]
- Bao, H.S.; Oliveira, R.G.; Wang, R.Z.; Wang, L.W.; Ma, Z.W. Working pairs for resorption refrigerator. Appl. Therm. Eng. 2011, 31, 3015–3021. [Google Scholar] [CrossRef]
- Lepinasse, E.; Goetz, V.; Crosat, G. Modelling and experimental investigation of a new type of thermochemical transformer based on the coupling of two solid-gas reactions. Chem. Eng. Process. 1994, 33, 125–134. [Google Scholar] [CrossRef]
- Tamainot-Telto, Z.; Critoph, R.E. Thermophysical properties of monolithic carbon. Int. J. Heat Mass Transf. 2000, 43, 2053–2058. [Google Scholar] [CrossRef]
- Perry, R.H.; Green, D.W.; Maloney, J.O. Perry’s Chemical Engineers’; Handbook; McGraw-Hill: New York, NY, USA, 1997. [Google Scholar]
- Ullmann, F. Ullmann’s Encyclopedia of Industrial Chemistry, 6th ed.; Wiley: Hoboken, NJ, USA, 2001. [Google Scholar]
- Füldner, G. Stofftransport und Adsorptionskinetik in Porösen Adsorbenskompositen für Wärmetransformationsanwendungen. Ph.D. Thesis, University of Freiburg, Breisgau, Germany, 2015. [Google Scholar]
- Domalski, E.S.; Hearing, E.D. Condensed Phase Heat Capacity Data. In NIST Chemistry WebBook, NIST Standard Reference Database Number 69; Linstrom, P.J., Mallard, W.G., Eds.; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Santori, G.; Frazzica, A.; Freni, A.; Galieni, M.; Bonaccorsi, L.; Polonara, F.; Restuccia, G. Optimization and testing on an adsorption dishwasher. Energy 2013, 50, 170–176. [Google Scholar] [CrossRef] [Green Version]
- Kakiuchi, H.; Iwade, M.; Shimooka, S.; Ooshima, K.; Yamazaki, M.; Takewaki, T. Novel zeolite adsorbents and their application for AHP and Desiccant system. lEA-Annex 2004, 17, 6. [Google Scholar]
- Julabo. The Thermal Bath Fluids; Julabo: Seelbach, Germany, 2015. [Google Scholar]
- Brancato, V.; Frazzica, A.; Sapienza, A.; Gordeeva, L.; Freni, A. Ethanol adsorption onto carbonaceous and composite adsorbents for adsorptive cooling system. Energy 2015, 84, 177–185. [Google Scholar] [CrossRef]
- Freni, A.; Bonaccorsi, L.; Calabrese, L.; Caprì, A.; Frazzica, A.; Sapienza, A. SAPO-34 coated adsorbent heat exchanger for adsorption chillers. Appl. Therm. Eng. 2016, 82, 1–7. [Google Scholar] [CrossRef]
- Kim, C.S. Thermophysical Properties of Stainless SteelsArgonne National Laboratory No. ANL-75-55Argonne National Laboratory: Lemont, IL, USA, 1975.
- Sapienza, A.; Santamaria, S.; Frazzica, A.; Freni, A. Influence of the management strategy and operating conditions on the performance of an adsorption chiller. Energy 2011, 36, 5532–5538. [Google Scholar] [CrossRef]
- Frazzica, A.; Palomba, V.; Dawoud, B.; Gullì, G.; Brancato, V.; Sapienza, A.; Vasta, S.; Freni, A.; Costa, F.; Restuccia, G. Design, realization and testing of an adsorption refrigerator based on activated carbon/ethanol working pair. Appl. Energy 2016, 174, 15–24. [Google Scholar] [CrossRef]
- Palomba, V.; Dawoud, B.; Sapienza, A.; Vasta, S.; Frazzica, A. On the impact of different management strategies on the performance of a two-bed activated carbon/ethanol refrigerator: An experimental study. Energy Convers. Manag. 2017, 142, 322–333. [Google Scholar] [CrossRef]
- Bendix, P.; Füldner, G.; Möllers, M.; Kummer, H.; Schnabel, L.; Henninger, S.; Henning, H.M. Optimization of power density and metal-to-adsorbent weight ratio in coated adsorbers for adsorptive heat transformation applications. Appl. Therm. Eng. 2017, 124, 83–90. [Google Scholar] [CrossRef]
- Tamainot-Telto, Z.; Critoph, R.E. Advanced solid sorption air conditioning modules using monolithic carbon–ammonia pair. Appl. Therm. Eng. 2003, 23, 659–674. [Google Scholar] [CrossRef]
- Rivero-Pacho, A.M.; Critoph, R.E.; Metcalf, S.J. Alternative monolithic/composite carbons for adsorption generators and simulation for optimal performance. Appl. Therm. Eng. 2017, 126, 350–357. [Google Scholar] [CrossRef]
- Blackman, C.; Bales, C.; Thorin, E. Experimental evaluation and concept demonstration of a novel modular gas-driven sorption heat pump. Proceedings of 12th IEA Heat Pump Conference, Rotterdam, The Netherlands, 15–18 May 2017. [Google Scholar]
- Bauer, J.; Herrmann, R.; Mittelbach, W.; Schwieger, W. Zeolite/aluminum composite adsorbents for application in adsorption refrigeration. Int. J. Energy Res. 2009, 33, 1233–1249. [Google Scholar] [CrossRef]
- Fink, M.; Andersen, O.; Seidel, T.; Schlott, A. Strongly Orthotropic Open Cell Porous Metal Structures for Heat Transfer Applications. Metals 2018, 8, 554. [Google Scholar] [CrossRef] [Green Version]
- Wittstadt, U.; Füldner, G.; Andersen, O.; Herrmann, R.; Schmidt, F. A new adsorbent composite material based on metal fiber technology and its application in adsorption heat exchangers. Energies 2015, 8, 8431–8446. [Google Scholar] [CrossRef]
- Wittstadt, U.; Füldner, G.; Laurenz, E.; Warlo, A.; Große, A.; Herrmann, R.; Schnabel, L.; Mittelbach, W. A novel adsorption module with fiber heat exchangers: Performance analysis based on driving temperature differences. Renew. Energy 2017, 110, 154–161. [Google Scholar] [CrossRef]

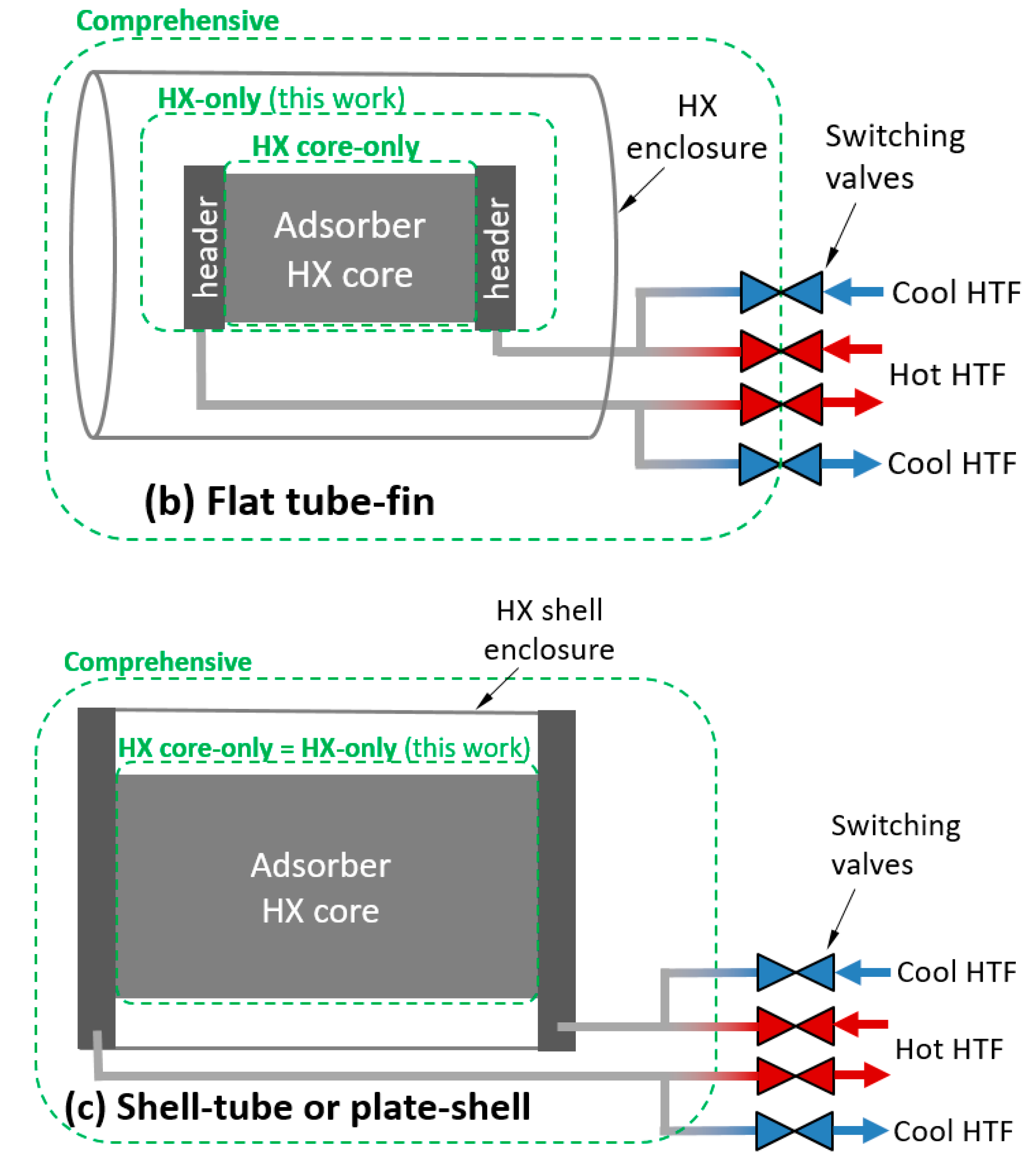









| Sorbent Material Mass (kg) | Non-Sorbent Material Mass (kg) |
|---|---|
| Live mass | Dead mass |
| Active mass | Inactive mass |
| Active mass | Inert mass |
| Host or active mass | Guest material |
| System and Working Pair | HX | HTF | Specific Thermal Mass: TM/msorb (kJ1K−1kgsorb−1) | ceffective:TM/mHX (kJ1K−1kgHX−1) | Reference |
|---|---|---|---|---|---|
| Adsorption: water/FAM-Z02 | Coated round tube–plain fin | Water | 5.62* | 1.64* | [10,14] |
| Adsorption: water/FAM-Z02 | Coated round tube–corrugated fin | Silicon oil | 4.17* | 0.77* | [15] |
| Water | 7.01* | 1.28* | |||
| Adsorption: water/FAM-Z02 | Packed round tube–corrugated fin | Silicon oil | 5.96*1 2.53*2 | 0.77*1 0.78*2 | [12,15] |
| Water | 10.30*1 3.98*2 | 1.31*1 1.21*2 | |||
| Resorption: ammonia/MnCl2 (HTS)/BaCl2 (LTS) | Packed shell–tube | Not reported | 2.34† (HTS) 2.18† (LTS) | 0.49† (HTS) 0.47† (LTS) | [18] |
| Resorption: ammonia/MnCl2 (HTS)/NH4Cl (LTS) | Coated annular tube | Not reported | 4.77† (HTS) 3.85† (LTS) | 0.47† (HTS) 0.48† (LTS) | [16] |
| Resorption: ammonia/MnCl2 (HTS)/NaBr (LTS) | Packed fin–tube | Not reported | 4.55† (HTS) 5.00† (LTS) | 0.47† (HTS) 0.47† (LTS) | [17] |
| Material | Specific Heat Capacity (kJ1kg−1K−1) | Density (kg1m−³) | Reference |
|---|---|---|---|
| Activated carbon (monolithic) | 1.05 | 750 | [19] |
| Aluminum (purity > 99%) | 0.91 | 2700 | [20] |
| Ammonia (liquid) | 4.60 | 639 | [21] |
| Binder SilRes MP50 E | 1.27 | Not used | Measured |
| TiAPSO SCT-323 | 0.90 | Not used | Estimated value |
| Adsorbent SAPO-34 directly crystallized | 0.90 | 1500 | [22] |
| Copper | 0.385 | 8.96 | [23] |
| Water | 4.19 | 1,000 | [20] |
| AQSOA FAM Z02 grains | 0.69 | Not used | [24] |
| AQSOA FAM Z02 powder | 0.822 | 600–700 | [24,25] |
| Silicon oil (HL80, Julabo) | 1.726 | 910 | [26] |
| Siogel | 0.62 | Not used | [24] |
| Activated carbon | 1.1 | Not used | [27] |
| Silane binder | 1.3 | Not used | [28] |
| Stainless steel (316) | 0.49 | 7954 | [29] |
| MnCl2–graphite 1:2 mixture | 1.24 | 470 | [16] |
| NH4Cl–graphite 1:2 mixture | 0.611 | 470 | [16] |
| MnCl2–graphite 13:7 mixture | 0.61 | 495 | [17,18] |
| BaCl2–graphite 13:7 mixture | 0.51 | 507 | [17,18] |
| MnCl2–graphite 13:7 mixture | 0.611 | 310 | [17] |
| NaBr–graphite 13:7 mixture | 0.541 | 300 | [17] |
| Component (Section) | Working Pair (refr./Sorbent) | HTF | TMtotal (kJ1K−1) | Sorbent Mass msorb (kgsorb) | HX Mass*mHX (kgHX) | MR, mHX/msorb (kgHX1 kgsorb−1) | STM, TM/msorb (kJ1K−1kgsorb−1) | ceffective TM/mHX (kJ1K−1 kgHX−1) |
|---|---|---|---|---|---|---|---|---|
| Flat tube–fin, packed (Section 3.1) | Water/silica gel | Water | 1.92 | 0.31 | 1.12 | 3.58 | 6.10 | 1.70 |
| Flat tube–fin, packed (Section 3.1) | Water/zeolite | Water | 1.90 | 0.26 | 1.07 | 4.12 | 7.31 | 1.78 |
| Flat tube–fin, packed (Section 3.2) | Ethanol/AC | Water | 4.66 | 0.60 | 3.05 | 5.08 | 7.77 | 1.53 |
| Flat tube–fin, coated (Section 3.3) | Water/zeolite | Water | 1.80 | 0.092 | 0.90 | 9.80 | 19.58 | 2.00 |
| Flat tube–fin, coated (Section 3.4) | Water/zeolite | Water | 1.97 | 0.45 | 1.24 | 2.77 | 4.40 | 1.59 |
| Round tube–fin, packed (Section 3.5) | Water/FAM-Z02 | Silicon oil | 2.98 | 0.5 | 3.86 | 7.72 | 5.96 | 0.77 |
| 3.80 | 1.5 | 4.86 | 3.24 | 2.53 | 0.78 | |||
| Round tube–fin, packed (Section 3.5) | Water/FAM-Z02 | Water | 5.15 | 0.5 | 3.94 | 7.87 | 10.30 | 1.31 |
| 5.97 | 1.5 | 4.94 | 3.29 | 3.98 | 1.21 | |||
| Round tube–fin, coated (Section 3.5) | Water/FAM-Z02 | Silicon oil | 3.20 | 0.766 | 4.12 | 5.39 | 4.17 | 0.77 |
| Round tube–fin, coated (Section 3.5) | Water/FAM-Z02 | Water | 5.37 | 0.766 | 4.20 | 5.49 | 7.01 | 1.28 |
| Round tube–fin, coated (1.2) | Water/zeolite | Water | 14.1 | 2.82 | 9.68 | 1.84 | 5.00 | 1.46 |
| Modular finned tube (Section 3.6) | NH3/A | Water | 0.6 | 0.078 | 0.71 | 9.1 | 7.7 | 0.85 |
| Shell–tube (Section 3.7) | NH3/AC | Water | 2.75 | 1.12 | 3.05 | 2.72 | 2.46 | 0.9 |
| Plate–shell (Section 3.8) | NH3/nanocoated halide salt | Water | 20.8 | 3.4 | 13.56 | 3.99 | 6.12 | 1.53 |
| Flat tube–fiber (Section 3.9) | Water/zeolite | Water | 18.58 | 3.30 | 13.69 | 4.15 | 5.63 | 1.36 |
| Component (Section) | Masses | Thermal Masses | ||||
|---|---|---|---|---|---|---|
| HTF (kg) | Metal (kg) | Sorbentmsorb (kgsorb) | HTF (kJ1K−1) | Metal (kJ1K−1) | Sorbent (kJ1K−1) | |
| Flat tube–fin, packed (Section 3.1) | 0.3 (water) | 0.51 | 0.31 | 1.25 | 0.46 | 0.226 |
| Flat tube–fin, packed (Section 3.1) | 0.3 (water) | 0.51 | 0.26 | 1.25 | 0.46 | 0.195 |
| Flat tube–fin, packed (Section 3.2) | 0.55 (water) | 1.9 | 0.60 | 2.30 | 1.73 | 0.66 |
| Flat tube–fin, coated (Section 3.3) | 0.3 (water) | 0.51 | 0.084 (sorbent) 0.009 (binder) | 1.25 | 0.46 | 0.074 |
| Flat tube–fin, coated (Section 3.4) | 0.25 (water) | 0.47 (Al) | 0.45 | 1.05 | 0.42 | 0.501 |
| Round tube–fin, packed (Section 3.5) | 0.76 (silicon oil) 0.83 (water) | 2.1 (copper) 0.5 (Al) | 0.5 1.5 | 1.31 3.49 | 1.25 | 0.41 1.23 |
| Round tube–fin, coated (Section 3.5) | 0.76 (silicon oil) 0.83 (water) | 2.1 (copper) 0.5 (Al) | 0.766 | 1.31 3.49 | 1.25 | 0.63 |
| Round tube–fin, coated (1.2) | 1.66 (water) | 5.20 | 2.82 | 6.96 | 4.68 | 4.23 |
| Modular finned tube (Section 3.6) | 5e-4 (air) | 0.636 | 0.078 | 5e-4 | 0.53 | 0.07 |
| Shell–tube (Section 3.7) | 0.222 (water) | 1.71 | 1.12 | 0.92 | 0.86 | 1 |
| Plate–shell (Section 3.8) | 2.36 (water) | 7.8 (stainless steel) | 3.4 | 9.86 | 3.82 | 5.44 |
| Fiber heat exchanger (Section 3.9) | 1.9 (water) | 8.5 (Al) | 3.3 | 7.94 | 7.65 | 2.97 |
| Component (Section) | Header Mass (Mass Difference between HX-Only and HX Core-Only) | Header Thermal Mass (Difference between HX-Only and HX Core-Only) | Thermal Mass, HX Core-Only | |
|---|---|---|---|---|
| HTF (kg) | Metal (kg) | (kJ1K−1) | (kJ1K−1) | |
| Adsorber, flat tube–fin, coated (Section 3.4) | 0.15 | 0.13 | 0.76 | 1.21 |
| Adsorber, round tube–fin, packed (Section 3.5) 1 HTF: silicon oil | 0.30 | 1.49 | 1.08 | 1.90 (0.5 kg packed) 2.12 (0.766 kg coated) |
| Adsorber, round tube–fin, packed (Section 3.5) 1 HTF: water | 0.33 | 1.49 | 1.93 | 3.22 (0.5 kg packed) 3.44 (0.766 kg coated) |
| Adsorber, shell–tube (Section 3.7) | 0.1 | 0.25 | 0.52 | 2.23 |
| Adsorber, fiber heat exchanger (Section 3.9) | 0.83 | 2.23 | 5.48 | 13.08 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gluesenkamp, K.R.; Frazzica, A.; Velte, A.; Metcalf, S.; Yang, Z.; Rouhani, M.; Blackman, C.; Qu, M.; Laurenz, E.; Rivero-Pacho, A.; et al. Experimentally Measured Thermal Masses of Adsorption Heat Exchangers. Energies 2020, 13, 1150. https://doi.org/10.3390/en13051150
Gluesenkamp KR, Frazzica A, Velte A, Metcalf S, Yang Z, Rouhani M, Blackman C, Qu M, Laurenz E, Rivero-Pacho A, et al. Experimentally Measured Thermal Masses of Adsorption Heat Exchangers. Energies. 2020; 13(5):1150. https://doi.org/10.3390/en13051150
Chicago/Turabian StyleGluesenkamp, Kyle R., Andrea Frazzica, Andreas Velte, Steven Metcalf, Zhiyao Yang, Mina Rouhani, Corey Blackman, Ming Qu, Eric Laurenz, Angeles Rivero-Pacho, and et al. 2020. "Experimentally Measured Thermal Masses of Adsorption Heat Exchangers" Energies 13, no. 5: 1150. https://doi.org/10.3390/en13051150










