Polysaccharide-Based Nanocomposites for Food Packaging Applications
Abstract
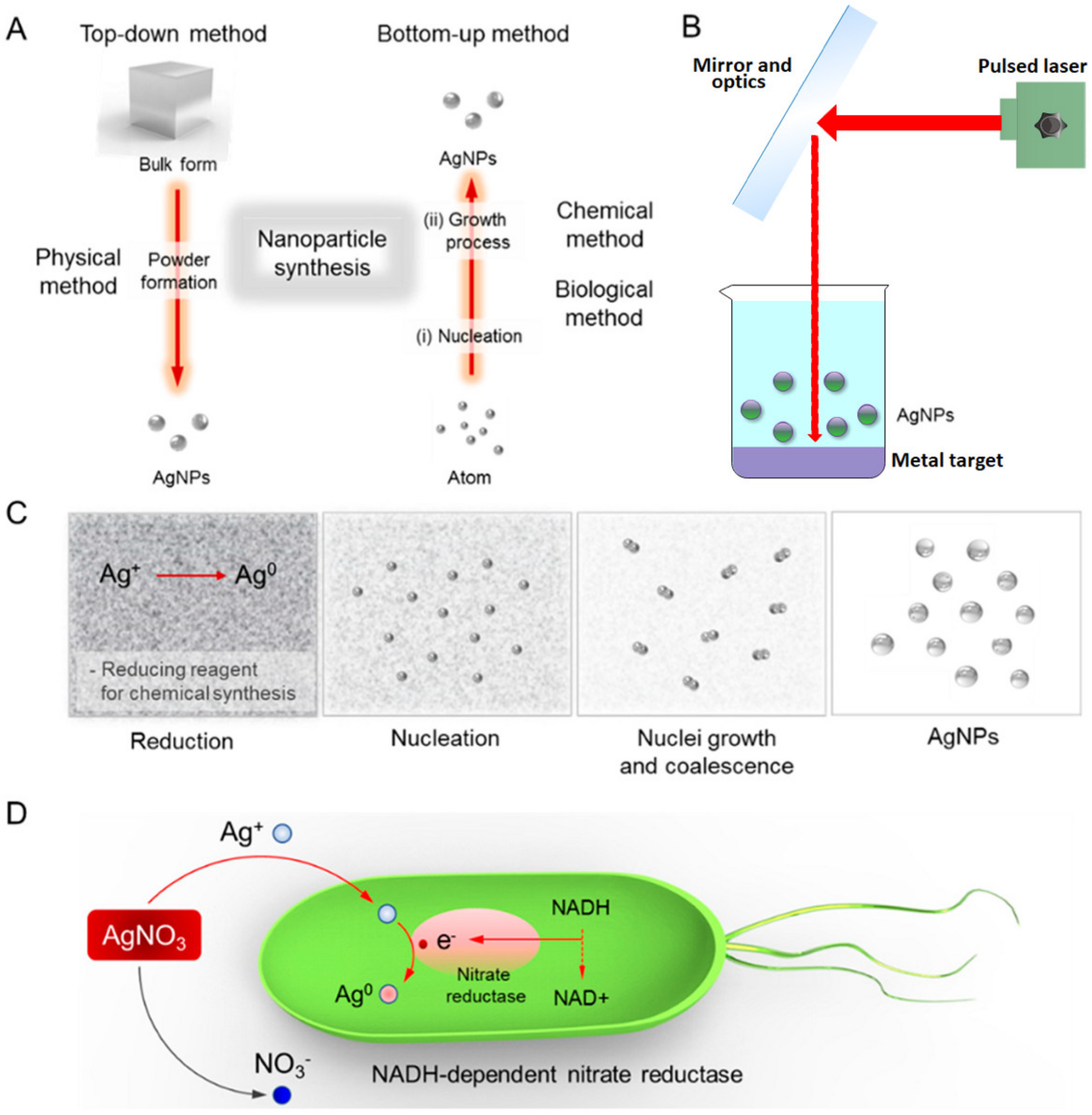
:1. Introduction
2. Scientometric Analysis
3. Chitosan-Based Nanocomposites
3.1. Chitosan and Silver Nanoparticle-Containing Food Packaging Systems
3.2. Chitosan and Cellulose-Based Nanostructure Containing Food Packaging Systems
3.3. Chitosan and Montmorillonite Nanoclay Containing Food Packaging Systems
4. Starch-Based Nanocomposites
4.1. Starch and Silver Nanoparticle-Containing Food Packaging Systems
4.2. Starch and Cellulose-Based Nanostructure Containing Food Packaging Systems
4.3. Starch and Montmorillonite Nanoclay Containing Food Packaging Systems
5. Cellulose-Based Nanocomposites
5.1. Cellulose and Silver Nanoparticle-Containing Food Packaging Systems
5.2. Cellulose and Cellulose-Based Nanostructure Containing Food Packaging Systems
5.3. Cellulose and Montmorillonite Nanoclay Containing Food Packaging Systems
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Dash, K.K.; Ali, N.A.; Das, D.; Mohanta, D. Thorough evaluation of sweet potato starch and lemon-waste pectin based-edible films with nano-titania inclusions for food packaging applications. Int. J. Biol. Macromol. 2019, 139, 449–458. [Google Scholar] [CrossRef]
- Czarnecka-Komorowska, D.; Wiszumirska, K. Sustainability design of plastic packaging for the Circular Economy. Polimery 2020, 65, 8–17. [Google Scholar] [CrossRef] [Green Version]
- Assman, K. Biodegradable polymers—Application examples. In Modern Polymer Materials and Their Processing; Part 3; Klepka, T., Ed.; Politechnika Lubelska: Lublin, Poland, 2017; pp. 51–70. ISBN 978-83-7947-300-7. (In Polish) [Google Scholar]
- Wojciechowska, P.; Kozak, W. Investigation of Transmission Oxygen Rate of Hybrid Materials Using Optical Method. Pol. J. Commod. Sci. 2016, 4, 81–88. [Google Scholar] [CrossRef]
- Kozak, W. Oxygen measurements in quality and safety assessment of products and services. Pol. J. Commod. Sci. 2020, 62, 9–25. [Google Scholar]
- Singh, A.; Shi, Y.; Magreault, P.; Kitts, D.D.; Jarzębski, M.; Siejak, P.; Pratap-Singh, A. A Rapid Gas-Chromatography/Mass-Spectrometry Technique for Determining Odour Activity Values of Volatile Compounds in Plant Proteins: Soy, and Allergen-Free Pea and Brown Rice Protein. Molecules 2021, 26, 4104. [Google Scholar] [CrossRef] [PubMed]
- Jamróz, E.; Kopel, P. Polysaccharide and Protein Films with Antimicrobial/Antioxidant Activity in the Food Industry: A Review. Polymers 2020, 12, 1289. [Google Scholar] [CrossRef]
- Guo, Q.; Liu, Y.; Cui, S.W. Structure, classification and modification of polysaccharides. In Comprehensive Glycoscience; Elsevier: Amsterdam, The Netherlands, 2021; pp. 204–219. [Google Scholar]
- Liu, X.; Xu, Y.; Zhan, X.; Xie, W.; Yang, X.; Cui, S.W.; Xia, W. Development and properties of new kojic acid and chitosan composite biodegradable films for active packaging materials. Int. J. Biol. Macromol. 2020, 144, 483–490. [Google Scholar] [CrossRef]
- Sothornvit, R.; Krochta, J.M. Plasticizers in edible films and coatings. Innov. Food Packag. 2005, 403–433. [Google Scholar] [CrossRef]
- Rodríguez-Núñez, J.R.; Madera-Santana, T.J.; Sánchez-Machado, D.I.; López-Cervantes, J.; Valdez, H.S. Chitosan/Hydrophilic Plasticizer-Based Films: Preparation, Physicochemical and Antimicrobial Properties. J. Polym. Environ. 2014, 22, 41–51. [Google Scholar] [CrossRef]
- Chan-Matú, D.I.; Toledo-López, V.M.; de Vargas, M.L.V.Y.; Rincón-Arriaga, S.; Rodríguez-Félix, A.; Madera-Santana, T.J. Preparation and characterization of chitosan-based bioactive films incorporating Moringa oleifera leaves extract. J. Food Meas. Charact. 2021, 1–12. [Google Scholar] [CrossRef]
- Zafar, R.; Zia, K.M.; Tabasum, S.; Jabeen, F.; Noreen, A.; Zuber, M. Polysaccharide based bionanocomposites, properties and applications: A review. Int. J. Biol. Macromol. 2016, 92, 1012–1024. [Google Scholar] [CrossRef]
- Xing, W.; Tang, Y. On mechanical properties of nanocomposite hydrogels: Searching for superior properties. Nano Mater. Sci. 2021, 1, 31–47. [Google Scholar] [CrossRef]
- Hallad, S.A.; Banapurmath, N.R.; Khan, T.M.Y.; Umarfarooq, M.A.; Soudagar, M.E.M.; Hunashyal, A.M.; Gujjar, S.V.; Yaradoddi, J.S.; Ganachari, S.V.; Elfasakhany, A.; et al. Statistical Analysis of Polymer Nanocomposites for Mechanical Properties. Molecules 2021, 26, 4135. [Google Scholar] [CrossRef]
- Lichade, K.M.; Jiang, Y.; Pan, Y. Hierarchical Nano/Micro-Structured Surfaces With High Surface Area/Volume Ratios. J. Manuf. Sci. Eng. 2021, 143, 1–36. [Google Scholar] [CrossRef]
- Brandelli, A.; Lopes, N.A. Nanocomposite antimicrobial films based on biopolymers. In Biopolymer-Based Nano Films; Elsevier: Amsterdam, The Netherlands, 2021; pp. 149–170. [Google Scholar]
- Sharma, R.; Jafari, S.M.; Sharma, S. Antimicrobial bio-nanocomposites and their potential applications in food packaging. Food Control 2020, 112, 107086. [Google Scholar] [CrossRef]
- Xia, G.-X.; Wu, Y.-M.; Bi, Y.-F.; Chen, K.; Zhang, W.-W.; Liu, S.-Q.; Zhang, W.-J.; Liu, R.-H. Antimicrobial Properties and Application of Polysaccharides and Their Derivatives. Chin. J. Polym. Sci. 2021, 39, 133–146. [Google Scholar] [CrossRef]
- Zheng, Y.; Monty, J.; Linhardt, R.J. Polysaccharide-based nanocomposites and their applications. Carbohydr. Res. 2015, 405, 23–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiao, L. Chitosan Application in Textile Processing. Curr. Trends Fash. Technol. Text. Eng. 2018, 4, 1–3. [Google Scholar] [CrossRef] [Green Version]
- Malerba, M.; Cerana, R. Recent Advances of Chitosan Applications in Plants. Polymers 2018, 10, 118. [Google Scholar] [CrossRef] [Green Version]
- Kabanov, V.L.; Novinyuk, L.V. Chitosan application in food technology: A review of rescent advances. Food Syst. 2020, 3, 10–15. [Google Scholar] [CrossRef] [Green Version]
- Kumar, M.N.R. A review of chitin and chitosan applications. React. Funct. Polym. 2000, 46, 1–27. [Google Scholar] [CrossRef]
- Irastorza, A.; Zarandona, I.; Andonegi, M.; Guerrero, P.; de la Caba, K. The versatility of collagen and chitosan: From food to biomedical applications. Food Hydrocoll. 2021, 116, 106633. [Google Scholar] [CrossRef]
- Schmitz, C.; Auza, L.G.; Koberidze, D.; Rasche, S.; Fischer, R.; Bortesi, L. Conversion of Chitin to Defined Chitosan Oligomers: Current Status and Future Prospects. Mar. Drugs 2019, 17, 452. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.; Sun, X. A Critical Review of the Abilities, Determinants, and Possible Molecular Mechanisms of Seaweed Polysaccharides Antioxidants. Int. J. Mol. Sci. 2020, 21, 7774. [Google Scholar] [CrossRef] [PubMed]
- Elfirta, R.R.; Saskiawan, I. The functional character of Auricularia auricula crude polysaccharides: Antioxidant and antibacterial activity. Ber. Biol. 2020, 19, 433–440. [Google Scholar] [CrossRef]
- Mo, X.; Liu, Y.; Li, T.; Peng, W.; Hu, M.; Wu, C. Extraction optimization and characterization of polysaccharide antioxidants from Pinellia ternata (Thunb) Breit rhizome. Trop. J. Pharm. Res. 2017, 16, 1129. [Google Scholar] [CrossRef] [Green Version]
- Mu, S.; Yang, W.; Huang, G. Antioxidant activities and mechanisms of polysaccharides. Chem. Biol. Drug Des. 2021, 97, 628–632. [Google Scholar] [CrossRef] [PubMed]
- Raafat, D.; Sahl, H.-G. Chitosan and its antimicrobial potential—A critical literature survey. Microb. Biotechnol. 2009, 2, 186–201. [Google Scholar] [CrossRef] [Green Version]
- Kittur, F.S.; Kumar, K.R.; Tharanathan, R.N. Functional packaging properties of chitosan films. Z. Lebensm. Forsch. A 1998, 206, 44–47. [Google Scholar] [CrossRef]
- Kurek, M.; Ščetar, M.; Voilley, A.; Galić, K.; Debeaufort, F. Barrier properties of chitosan coated polyethylene. J. Membr. Sci. 2012, 403–404, 162–168. [Google Scholar] [CrossRef]
- Darie-Niță, R.N.; Râpă, M.; Sivertsvik, M.; Rosnes, J.T.; Popa, E.E.; Dumitriu, R.P.; Marincaș, O.; Matei, E.; Predescu, C.; Vasile, C. PLA-Based Materials Containing Bio-Plasticizers and Chitosan Modified with Rosehip Seed Oil for Ecological Packaging. Polymers 2021, 13, 1610. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Mukherjee, A.; Dutta, J. Chitosan based nanocomposite films and coatings: Emerging antimicrobial food packaging alternatives. Trends Food Sci. Technol. 2020, 97, 196–209. [Google Scholar] [CrossRef]
- Fernandez, J.G.; Ingber, D.E. Unexpected Strength and Toughness in Chitosan-Fibroin Laminates Inspired by Insect Cuticle. Adv. Mater. 2011, 24, 480–484. [Google Scholar] [CrossRef]
- Da Silva Alves, D.C.; Healy, B.; de Pinto, L.A.A.; Cadaval, T.R.S.; Breslin, C.B. Recent Developments in Chitosan-Based Adsorbents for the Removal of Pollutants from Aqueous Environments. Molecules 2021, 26, 594. [Google Scholar] [CrossRef]
- Ke, C.-L.; Deng, F.-S.; Chuang, C.-Y.; Lin, C.-H. Antimicrobial Actions and Applications of Chitosan. Polymers 2021, 13, 904. [Google Scholar] [CrossRef]
- Tankhiwale, R.; Bajpai, S.K. Silver-nanoparticle-loaded chitosan lactate films with fair antibacterial properties. J. Appl. Polym. Sci. 2010, 115, 1894–1900. [Google Scholar] [CrossRef]
- Reidy, B.; Haase, A.; Luch, A.; Dawson, K.; Lynch, I. Mechanisms of Silver Nanoparticle Release, Transformation and Toxicity: A Critical Review of Current Knowledge and Recommendations for Future Studies and Applications. Materials 2013, 6, 2295–2350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmad, S.A.; Das, S.S.; Khatoon, A.; Ansari, M.T.; Afzal, M.; Hasnain, M.S.; Nayak, A.K. Bactericidal activity of silver nanoparticles: A mechanistic review. Mater. Sci. Energy Technol. 2020, 3, 756–769. [Google Scholar] [CrossRef]
- Kadam, D.; Momin, B.; Palamthodi, S.; Lele, S.S. Physicochemical and functional properties of chitosan-based nano-composite films incorporated with biogenic silver nanoparticles. Carbohydr. Polym. 2019, 211, 124–132. [Google Scholar] [CrossRef]
- Lee, S.H.; Jun, B.-H. Silver Nanoparticles: Synthesis and Application for Nanomedicine. Int. J. Mol. Sci. 2019, 20, 865. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anis, A.; Pal, K.; Al-Zahrani, S.A. Essential Oil-Containing Polysaccharide-Based Edible Films and Coatings for Food Security Applications. Polymers 2021, 13, 575. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Huang, X.; Li, Y.-C.; Xiao, H.; Wang, X. Novel chitosan films with laponite immobilized Ag nanoparticles for active food packaging. Carbohydr. Polym. 2018, 199, 210–218. [Google Scholar] [CrossRef]
- Gu, B.; Jiang, Q.; Luo, B.; Liu, C.; Ren, J.; Wang, X.; Wang, X. A sandwich-like chitosan-based antibacterial nanocomposite film with reduced graphene oxide immobilized silver nanoparticles. Carbohydr. Polym. 2021, 260, 117835. [Google Scholar] [CrossRef] [PubMed]
- Salleh, A.; Naomi, R.; Utami, N.D.; Mohammad, A.W.; Mahmoudi, E.; Mustafa, N.; Fauzi, M.B. The Potential of Silver Nanoparticles for Antiviral and Antibacterial Applications: A Mechanism of Action. Nanomaterials 2020, 10, 1566. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Shukla, A.; Baul, P.P.; Mitra, A.; Halder, D. Biodegradable hybrid nanocomposites of chitosan/gelatin and silver nanoparticles for active food packaging applications. Food Packag. Shelf Life 2018, 16, 178–184. [Google Scholar] [CrossRef]
- Qin, Y.; Liu, Y.; Yuan, L.; Yong, H.; Liu, J. Preparation and characterization of antioxidant, antimicrobial and pH-sensitive films based on chitosan, silver nanoparticles and purple corn extract. Food Hydrocoll. 2019, 96, 102–111. [Google Scholar] [CrossRef]
- Ossai, C.I.; Raghavan, N. Nanostructure and nanomaterial characterization, growth mechanisms, and applications. Nanotechnol. Rev. 2018, 7, 209–231. [Google Scholar] [CrossRef]
- Celebi, H.; Kurt, A. Effects of processing on the properties of chitosan/cellulose nanocrystal films. Carbohydr. Polym. 2015, 133, 284–293. [Google Scholar] [CrossRef]
- Yadav, M.; Behera, K.; Chang, Y.-H.; Chiu, F.-C. Cellulose Nanocrystal Reinforced Chitosan Based UV Barrier Composite Films for Sustainable Packaging. Polymers 2020, 12, 202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, Y.; Zhang, X.; Zhao, R.; Guo, D.; Zhang, J. Preparation and properties of chitosan/guar gum/nanocrystalline cellulose nanocomposite films. Carbohydr. Polym. 2018, 197, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Kusmono, K.; Wildan, M.W.; Lubis, F.I. Fabrication and Characterization of Chitosan/Cellulose Nanocrystal/Glycerol Bio-Composite Films. Polymers 2021, 13, 1096. [Google Scholar] [CrossRef]
- De Andrade, M.R.; Nery, T.B.R.; de Santana, T.I.D.; Leal, I.L.; Rodrigues, L.A.P.; de Oliveira Reis, J.H.D.O.; Druzian, J.I.; Machado, B.A.S.; Andrade, D. Effect of Cellulose Nanocrystals from Different Lignocellulosic Residues to Chitosan/Glycerol Films. Polymers 2019, 11, 658. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.; Willis, S.; Jordan, K.; Sismour, E. Chitosan nanocomposite films incorporating cellulose nanocrystals and grape pomace extracts. Packag. Technol. Sci. 2018, 31, 631–638. [Google Scholar] [CrossRef]
- Gan, P.G.; Sam, S.T.; Abdullah, M.F.; Omar, M.F.; Tan, W.K. Water resistance and biodegradation properties of conventionally-heated and microwave-cured cross-linked cellulose nanocrystal/chitosan composite films. Polym. Degrad. Stab. 2021, 188, 109563. [Google Scholar] [CrossRef]
- Rojas, J.; Bedoya, M.; Ciro, Y. Current trends in the production of cellulose nanoparticles and nanocomposites for biomedical applications. In Cellulose—Fundamental Aspects and Current Trends; BoD—Books on Demand: Norderstedt, Germany, 2015; pp. 193–228. [Google Scholar] [CrossRef] [Green Version]
- Salari, M.; Khiabani, M.S.; Mokarram, R.R.; Ghanbarzadeh, B.; Kafil, H.S. Development and evaluation of chitosan based active nanocomposite films containing bacterial cellulose nanocrystals and silver nanoparticles. Food Hydrocoll. 2018, 84, 414–423. [Google Scholar] [CrossRef]
- Thou, C.Z.; Khan, F.S.A.; Mubarak, N.M.; Ahmad, A.; Khalid, M.; Jagadish, P.; Walvekar, R.; Abdullah, E.C.; Khan, S.; Khan, M.; et al. Surface charge on chitosan/cellulose nanowhiskers composite via functionalized and untreated carbon nanotube. Arab. J. Chem. 2021, 14, 103022. [Google Scholar] [CrossRef]
- Deng, Z.; Jung, J.; Simonsen, J.; Zhao, Y. Cellulose nanocrystals Pickering emulsion incorporated chitosan coatings for improving storability of postharvest Bartlett pears (Pyrus communis) during long-term cold storage. Food Hydrocoll. 2018, 84, 229–237. [Google Scholar] [CrossRef]
- Adel, A.M.; Ibrahim, A.A.; El-Shafei, A.M.; Al-Shemy, M.T. Inclusion complex of clove oil with chitosan/β-cyclodextrin citrate/oxidized nanocellulose biocomposite for active food packaging. Food Packag. Shelf Life 2019, 20, 100307. [Google Scholar] [CrossRef]
- Mohammadi Sadati, S.M.; Shahgholian-Ghahfarrokhi, N.; Shahrousvand, E.; Mohammadi-Rovshandeh, J.; Shahrousvand, M. Edible chitosan/cellulose nanofiber nanocomposite films for potential use as food packaging. Mater. Technol. 2021, 1–13. [Google Scholar] [CrossRef]
- Souza, V.G.L.; Pires, J.R.A.; Rodrigues, P.F.; Lopes, A.A.S.; Fernandes, F.M.B.; Duarte, M.P.; Coelhoso, I.M.; Fernando, A.L. Bionanocomposites of chitosan/montmorillonite incorporated with Rosmarinus officinalis essential oil: Development and physical characterization. Food Packag. Shelf Life 2018, 16, 148–156. [Google Scholar] [CrossRef]
- Pires, J.; Paula, C.D.; de Souza, V.G.L.; Fernando, A.L.; Coelhoso, I. Understanding the Barrier and Mechanical Behavior of Different Nanofillers in Chitosan Films for Food Packaging. Polymers 2021, 13, 721. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Zhao, L.; He, B. Chitosan/Montmorillonite Coatings for the Fabrication of Food-Safe Greaseproof Paper. Polymers 2021, 13, 1607. [Google Scholar] [CrossRef]
- Pires, J.R.A.; de Souza, V.G.L.; Fernando, A.L. Chitosan/montmorillonite bionanocomposites incorporated with rosemary and ginger essential oil as packaging for fresh poultry meat. Food Packag. Shelf Life 2018, 17, 142–149. [Google Scholar] [CrossRef]
- Souza, V.G.L.; Pires, J.R.A.; Rodrigues, C.; Rodrigues, P.F.; Lopes, A.; Silva, R.J.; Caldeira, J.; Duarte, M.P.; Fernandes, F.B.; Coelhoso, I.M.; et al. Physical and Morphological Characterization of Chitosan/Montmorillonite Films Incorporated with Ginger Essential Oil. Coatings 2019, 9, 700. [Google Scholar] [CrossRef] [Green Version]
- Cui, R.; Yan, J.; Cao, J.; Qin, Y.; Yuan, M.; Li, L. Release properties of cinnamaldehyde loaded by montmorillonite in chitosan-based antibacterial food packaging. Int. J. Food Sci. Technol. 2021, 56, 3670–3681. [Google Scholar] [CrossRef]
- Romero-Bastida, C.A.; Velazquez, G.; Bautista-Baños, S. Effect of the preparation method on the properties of nanocomposites based on chitosan, montmorillonite and essential oils. Rev. Mex. Ing. Quím. 2020, 19, 1243–1253. [Google Scholar] [CrossRef]
- Müller, K.; Bugnicourt, E.; Latorre, M.; Jorda, M.; Echegoyen Sanz, Y.; Lagaron, J.; Miesbauer, O.; Bianchin, A.; Hankin, S.; Bölz, U.; et al. Review on the Processing and Properties of Polymer Nanocomposites and Nanocoatings and Their Applications in the Packaging, Automotive and Solar Energy Fields. Nanomaterials 2017, 7, 74. [Google Scholar] [CrossRef] [Green Version]
- Rodrigues, C.; de Mello, J.M.M.; Dalcanton, F.; Macuvele, D.L.P.; Padoin, N.; Fiori, M.A.; Soares, C.; Riella, H.G. Mechanical, Thermal and Antimicrobial Properties of Chitosan-Based-Nanocomposite with Potential Applications for Food Packaging. J. Polym. Environ. 2020, 28, 1216–1236. [Google Scholar] [CrossRef]
- Yan, W.; Chen, W.; Muhammad, U.; Zhang, J.; Zhuang, H.; Zhou, G. Preparation of α-tocopherol-chitosan nanoparticles/chitosan/montmorillonite film and the antioxidant efficiency on sliced dry-cured ham. Food Control 2019, 104, 132–138. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, H.; Jin, C.; Zhang, R.; Li, L.; Li, X.; Jiang, S. Sodium lactate loaded chitosan-polyvinyl alcohol/montmorillonite composite film towards active food packaging. Innov. Food Sci. Emerg. Technol. 2017, 42, 101–108. [Google Scholar] [CrossRef]
- Vianna, T.C.; Marinho, C.O.; Júnior, L.M.; Ibrahim, S.A.; Vieira, R.P. Essential oils as additives in active starch-based food packaging films: A review. Int. J. Biol. Macromol. 2021, 182, 1803–1819. [Google Scholar] [CrossRef]
- Cheng, H.; Chen, L.; McClements, D.J.; Yang, T.; Zhang, Z.; Ren, F.; Miao, M.; Tian, Y.; Jin, Z. Starch-based biodegradable packaging materials: A review of their preparation, characterization and diverse applications in the food industry. Trends Food Sci. Technol. 2021, 114, 70–82. [Google Scholar] [CrossRef]
- Mohammad, F.; Arfin, T.; Bwatanglang, I.B.; Al-lohedan, H.A. Starch-based nanocomposites: Types and industrial applications. In Bio-Based Polymers and Nanocomposites; Springer International Publishing: Cham, Switzerland, 2019; pp. 157–181. [Google Scholar]
- Othman, S.H.; Othman, N.F.L.; Shapi’i, R.A.; Ariffin, S.H.; Yunos, K.F.M. Corn Starch/Chitosan Nanoparticles/Thymol Bio-Nanocomposite Films for Potential Food Packaging Applications. Polymers 2021, 13, 390. [Google Scholar] [CrossRef]
- Yin, Z.; Zeng, J.; Wang, C.; Pan, Z. Preparation and Properties of Cross-Linked Starch Nanocrystals/Polylactic Acid Nanocomposites. Int. J. Polym. Sci. 2015, 2015, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Shah, N.; Mewada, R.K.; Mehta, T. Crosslinking of starch and its effect on viscosity behaviour. Rev. Chem. Eng. 2016, 32, 265–270. [Google Scholar] [CrossRef]
- Żołek-Tryznowska, Z.; Kałuża, A. The Influence of Starch Origin on the Properties of Starch Films: Packaging Performance. Materials 2021, 14, 1146. [Google Scholar] [CrossRef]
- Jung, J.; Raghavendra, G.M.; Kim, D.; Seo, J. One-step synthesis of starch-silver nanoparticle solution and its application to antibacterial paper coating. Int. J. Biol. Macromol. 2018, 107, 2285–2290. [Google Scholar] [CrossRef]
- Dang, C.; Yin, Y.; Xu, M.; Pu, J. Hydrophobic Noncrystalline Porous Starch (NCPS): Dispersed Silver Nanoparticle Suspension as an Antibacterial Coating for Packaging Paper. BioResources 2017, 13, 192–207. [Google Scholar] [CrossRef]
- Rozilah, A.; Jaafar, C.N.A.; Sapuan, S.M.; Zainol, I.; Ilyas, R.A. The Effects of Silver Nanoparticles Compositions on the Mechanical, Physiochemical, Antibacterial, and Morphology Properties of Sugar Palm Starch Biocomposites for Antibacterial Coating. Polymers 2020, 12, 2605. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.S.C.; Martim, S.R.; Gomes, D.M.D.; Prado, F.B.; Marinho, N.M.V.; de Silva, T.A.; Castillo, T.A.; de Rego, J.A.R.; do Seabra, A.B.; Durán, N.; et al. Amazonian tuber starch based films incorporated with silver nanoparticles for preservation of fruits. Res. Soc. Dev. 2021, 10, e23510615304. [Google Scholar] [CrossRef]
- Ortega, F.; Giannuzzi, L.; Arce, V.B.; García, M.A. Active composite starch films containing green synthetized silver nanoparticles. Food Hydrocoll. 2017, 70, 152–162. [Google Scholar] [CrossRef]
- Mohseni, M.S.; Khalilzadeh, M.A.; Mohseni, M.; Hargalani, F.Z.; Getso, M.I.; Raissi, V.; Raiesi, O. Green synthesis of Ag nanoparticles from pomegranate seeds extract and synthesis of Ag-Starch nanocomposite and characterization of mechanical properties of the films. Biocatal. Agric. Biotechnol. 2020, 25, 101569. [Google Scholar] [CrossRef]
- Ceballos, R.L.; von Bilderling, C.; Guz, L.; Bernal, C.; Famá, L. Effect of greenly synthetized silver nanoparticles on the properties of active starch films obtained by extrusion and compression molding. Carbohydr. Polym. 2021, 261, 117871. [Google Scholar] [CrossRef]
- Ortega, F.; Arce, V.B.; Garcia, M.A. Nanocomposite starch-based films containing silver nanoparticles synthesized with lemon juice as reducing and stabilizing agent. Carbohydr. Polym. 2021, 252, 117208. [Google Scholar] [CrossRef]
- Kumar, R.; Ghoshal, G.; Goyal, M. Development and characterization of corn starch based nanocomposite film with AgNPs and plant extract. Mater. Sci. Energy Technol. 2020, 3, 672–678. [Google Scholar] [CrossRef]
- Peighambardoust, S.J.; Peighambardoust, S.H.; Pournasir, N.; Mohammadzadeh Pakdel, P. Properties of active starch-based films incorporating a combination of Ag, ZnO and CuO nanoparticles for potential use in food packaging applications. Food Packag. Shelf Life 2019, 22, 100420. [Google Scholar] [CrossRef]
- Mahuwala, A.A.; Hemant, V.; Meharwade, S.D.; Deb, A.; Chakravorty, A.; Grace, A.N.; Raghavan, V. Synthesis and characterisation of starch/agar nanocomposite films for food packaging application. IET Nanobiotechnol. 2020, 14, 809–814. [Google Scholar] [CrossRef]
- Mathew, S.; Jayakumar, A.; Kumar, V.P.; Mathew, J.; Radhakrishnan, E.K. One-step synthesis of eco-friendly boiled rice starch blended polyvinyl alcohol bionanocomposite films decorated with in situ generated silver nanoparticles for food packaging purpose. Int. J. Biol. Macromol. 2019, 139, 475–485. [Google Scholar] [CrossRef]
- Srikhao, N.; Kasemsiri, P.; Ounkaew, A.; Lorwanishpaisarn, N.; Okhawilai, M.; Pongsa, U.; Hiziroglu, S.; Chindaprasirt, P. Bioactive Nanocomposite Film Based on Cassava Starch/Polyvinyl Alcohol Containing Green Synthesized Silver Nanoparticles. J. Polym. Environ. 2021, 29, 672–684. [Google Scholar] [CrossRef]
- Jeevahan, J.; Chandrasekaran, M. Influence of Nanocellulose Additive on the Film Properties of Native Rice Starch-based Edible Films for Food Packaging. Recent Pat. Nanotechnol. 2020, 13, 222–233. [Google Scholar] [CrossRef] [PubMed]
- Fazeli, M.; Keley, M.; Biazar, E. Preparation and characterization of starch-based composite films reinforced by cellulose nanofibers. Int. J. Biol. Macromol. 2018, 116, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.; Zeng, J.; Wang, B.; Cheng, Z.; Gao, W.; Xu, J.; Chen, K. Silver nanoparticles immobilized on cellulose nanofibrils for starch-based nanocomposites with high antibacterial, biocompatible, and mechanical properties. Cellulose 2021, 28, 855–869. [Google Scholar] [CrossRef]
- Ghoshal, G.; Singh, D. Synthesis and characterization of starch nanocellulosic films incorporated with Eucalyptus globulus leaf extract. Int. J. Food Microbiol. 2020, 332, 108765. [Google Scholar] [CrossRef]
- Ilyas, R.A.; Sapuan, S.M.; Ibrahim, R.; Abral, H.; Ishak, M.R.; Zainudin, E.S.; Atiqah, A.; Atikah, M.S.N.; Syafri, E.; Asrofi, M.; et al. Thermal, Biodegradability and Water Barrier Properties of Bio-Nanocomposites Based on Plasticised Sugar Palm Starch and Nanofibrillated Celluloses from Sugar Palm Fibres. J. Biobased Mater. Bioenergy 2020, 14, 234–248. [Google Scholar] [CrossRef]
- De Teixeira, E.M.; Pasquini, D.; Curvelo, A.A.S.; Corradini, E.; Belgacem, M.N.; Dufresne, A. Cassava bagasse cellulose nanofibrils reinforced thermoplastic cassava starch. Carbohydr. Polym. 2009, 78, 422–431. [Google Scholar] [CrossRef]
- Kaushik, A.; Kaur, R.; Tripathi, S.K.; Dharamvir, K.; Kumar, R.; Saini, G.S.S. Thermal Behaviour of Nanocomposites based on Glycerol Plasticized Thermoplastic Starch and Cellulose Nanocrystallites. AIP Conf. Proc. 2011, 1393, 353–354. [Google Scholar]
- Ilyas, R.A.; Sapuan, S.M.; Ishak, M.R.; Zainudin, E.S. Development and characterization of sugar palm nanocrystalline cellulose reinforced sugar palm starch bionanocomposites. Carbohydr. Polym. 2018, 202, 186–202. [Google Scholar] [CrossRef]
- Bagde, P.; Nadanathangam, V. Mechanical, antibacterial and biodegradable properties of starch film containing bacteriocin immobilized crystalline nanocellulose. Carbohydr. Polym. 2019, 222, 115021. [Google Scholar] [CrossRef]
- Fabra, M.J.; López-Rubio, A.; Ambrosio-Martín, J.; Lagaron, J.M. Improving the barrier properties of thermoplastic corn starch-based films containing bacterial cellulose nanowhiskers by means of PHA electrospun coatings of interest in food packaging. Food Hydrocoll. 2016, 61, 261–268. [Google Scholar] [CrossRef] [Green Version]
- Oleyaei, S.A.; Moayedi, A.A.; Ghanbarzadeh, B. The Effect of Montmorillonite (MMT) on Structural, Thermal and Optical Properties of Iranian Potato Starch Based Nanobiocomposite Films. Innov. Food Technol. 2017, 4, 89–105. [Google Scholar] [CrossRef]
- Vaezi, K.; Asadpour, G.; Sharifi, H. Effect of ZnO nanoparticles on the mechanical, barrier and optical properties of thermoplastic cationic starch/montmorillonite biodegradable films. Int. J. Biol. Macromol. 2019, 124, 519–529. [Google Scholar] [CrossRef] [PubMed]
- De Souza, A.G.; dos Santos, N.M.A.; da Silva Torin, R.F.; dos Santos Rosa, D. Synergic antimicrobial properties of Carvacrol essential oil and montmorillonite in biodegradable starch films. Int. J. Biol. Macromol. 2020, 164, 1737–1747. [Google Scholar] [CrossRef]
- Campos-Requena, V.H.; Rivas, B.L.; Pérez, M.A.; Figueroa, C.R.; Figueroa, N.E.; Sanfuentes, E.A. Thermoplastic starch/clay nanocomposites loaded with essential oil constituents as packaging for strawberries—In vivo antimicrobial synergy over Botrytis cinerea. Postharvest Biol. Technol. 2017, 129, 29–36. [Google Scholar] [CrossRef]
- Piñeros-Guerrero, N.; Marsiglia-Fuentes, R.; Ortega-Toro, R. Improvement of the physicochemical properties of composite materials based on cassavastarch and polycaprolactone reinforced with sodium montmorillonite. Rev. Mex. Ing. Quím. 2021, 20, Alim2416. [Google Scholar]
- Kumar, M.; Panjagari, N.R.; Kanade, P.P.; Singh, A.K.; Badola, R.; Ganguly, S.; Behare, P.V.; Sharma, R.; Alam, T. Sodium caseinate-starch-modified montmorillonite based biodegradable film: Laboratory food extruder assisted exfoliation and characterization. Food Packag. Shelf Life 2018, 15, 17–27. [Google Scholar] [CrossRef]
- Yousefi, A.R.; Savadkoohi, B.; Zahedi, Y.; Hatami, M.; Ako, K. Fabrication and characterization of hybrid sodium montmorillonite/TiO2 reinforced cross-linked wheat starch-based nanocomposites. Int. J. Biol. Macromol. 2019, 131, 253–263. [Google Scholar] [CrossRef]
- Mathew, S.; Snigdha, S.; Mathew, J.; Radhakrishnan, E.K. Poly(vinyl alcohol): Montmorillonite: Boiled rice water (starch) blend film reinforced with silver nanoparticles; characterization and antibacterial properties. Appl. Clay Sci. 2018, 161, 464–473. [Google Scholar] [CrossRef]
- Tayeb, A.; Amini, E.; Ghasemi, S.; Tajvidi, M. Cellulose Nanomaterials—Binding Properties and Applications: A Review. Molecules 2018, 23, 2684. [Google Scholar] [CrossRef] [Green Version]
- Wahid, F.; Zhong, C. Production and applications of bacterial cellulose. In Biomass, Biofuels, Biochemicals; Elsevier: Amsterdam, The Netherlands, 2021; pp. 359–390. [Google Scholar]
- Feng, Y.H.; Li, J.C.; Lin, Q.; Pang, S.J.; Wang, X.B.; Wu, Z.X. The Characterizations of Bacterial Cellulose and Dialdehyde Celluloses from Bacterial Cellulose. Key Eng. Mater. 2007, 361–363, 563–566. [Google Scholar] [CrossRef]
- Betlej, I.; Zakaria, S.; Krajewski, K.J.; Boruszewski, P. Bacterial Cellulose—Properties and Its Potential Application. Sains Malays. 2021, 50, 493–505. [Google Scholar] [CrossRef]
- Chen, Q.-Y.; Xiao, S.-L.; Shi, S.Q.; Cai, L.-P. A One-Pot Synthesis and Characterization of Antibacterial Silver Nanoparticle–Cellulose Film. Polymers 2020, 12, 440. [Google Scholar] [CrossRef] [Green Version]
- Adepu, S.; Khandelwal, M. Broad-spectrum antimicrobial activity of bacterial cellulose silver nanocomposites with sustained release. J. Mater. Sci. 2018, 53, 1596–1609. [Google Scholar] [CrossRef]
- Gu, R.; Yun, H.; Chen, L.; Wang, Q.; Huang, X. Regenerated Cellulose Films with Amino-Terminated Hyperbranched Polyamic Anchored Nanosilver for Active Food Packaging. ACS Appl. Bio Mater. 2020, 3, 602–610. [Google Scholar] [CrossRef] [Green Version]
- Marrez, D.A.; Abdelhamid, A.E.; Darwesh, O.M. Eco-friendly cellulose acetate green synthesized silver nano-composite as antibacterial packaging system for food safety. Food Packag. Shelf Life 2019, 20, 100302. [Google Scholar] [CrossRef]
- Yu, Z.; Wang, W.; Kong, F.; Lin, M.; Mustapha, A. Cellulose nanofibril/silver nanoparticle composite as an active food packaging system and its toxicity to human colon cells. Int. J. Biol. Macromol. 2019, 129, 887–894. [Google Scholar] [CrossRef]
- Yu, Z.; Wang, W.; Dhital, R.; Kong, F.; Lin, M.; Mustapha, A. Antimicrobial effect and toxicity of cellulose nanofibril/silver nanoparticle nanocomposites prepared by an ultraviolet irradiation method. Colloids Surf. B Biointerfaces 2019, 180, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Saenjaiban, A.; Singtisan, T.; Suppakul, P.; Jantanasakulwong, K.; Punyodom, W.; Rachtanapun, P. Novel Color Change Film as a Time–Temperature Indicator Using Polydiacetylene/Silver Nanoparticles Embedded in Carboxymethyl Cellulose. Polymers 2020, 12, 2306. [Google Scholar] [CrossRef]
- Kwon, G.; Lee, K.; Kim, D.; Jeon, Y.; Kim, U.-J.; You, J. Cellulose nanocrystal-coated TEMPO-oxidized cellulose nanofiber films for high performance all-cellulose nanocomposites. J. Hazard. Mater. 2020, 398, 123100. [Google Scholar] [CrossRef]
- Sun, X.; Wu, Q.; Zhang, X.; Ren, S.; Lei, T.; Li, W.; Xu, G.; Zhang, Q. Nanocellulose films with combined cellulose nanofibers and nanocrystals: Tailored thermal, optical and mechanical properties. Cellulose 2018, 25, 1103–1115. [Google Scholar] [CrossRef]
- Sá, N.M.S.M.; Mattos, A.L.A.; Silva, L.M.A.; Brito, E.S.; Rosa, M.F.; Azeredo, H.M.C. From cashew byproducts to biodegradable active materials: Bacterial cellulose-lignin-cellulose nanocrystal nanocomposite films. Int. J. Biol. Macromol. 2020, 161, 1337–1345. [Google Scholar] [CrossRef]
- Roy, S.; Kim, H.-J.; Rhim, J.-W. Synthesis of Carboxymethyl Cellulose and Agar-Based Multifunctional Films Reinforced with Cellulose Nanocrystals and Shikonin. ACS Appl. Polym. Mater. 2021, 3, 1060–1069. [Google Scholar] [CrossRef]
- Ghaderi, M.; Mousavi, M.; Yousefi, H.; Labbafi, M. All-cellulose nanocomposite film made from bagasse cellulose nanofibers for food packaging application. Carbohydr. Polym. 2014, 104, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, V.; Ghanbarzadeh, B.; Ayaseh, A.; Ostadrahimi, A.; Ehsani, A.; Alizadeh-Sani, M.; Adun, P.A. The optimization of physico-mechanical properties of bionanocomposite films based on gluten/ carboxymethyl cellulose/ cellulose nanofiber using response surface methodology. Polym. Test. 2019, 78, 105989. [Google Scholar] [CrossRef]
- Zabihollahi, N.; Alizadeh, A.; Almasi, H.; Hanifian, S.; Hamishekar, H. Development and characterization of carboxymethyl cellulose based probiotic nanocomposite film containing cellulose nanofiber and inulin for chicken fillet shelf life extension. Int. J. Biol. Macromol. 2020, 160, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Lao, T.L.; Pengson, L.T.; Placido, J.; Diaz, L.J. Synthesis of Montmorillonite Nanoclay Reinforced Chitin-cellulose Nanocomposite Film. IOP Conf. Ser. Mater. Sci. Eng. 2019, 540, 012010. [Google Scholar] [CrossRef] [Green Version]
- Demircan, D.; Ilk, S.; Zhang, B. Cellulose-Organic Montmorillonite Nanocomposites as Biomacromolecular Quorum-Sensing Inhibitor. Biomacromolecules 2017, 18, 3439–3446. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Fei, X.; Li, H. Carboxymethyl cellulose-based nanocomposites reinforced with montmorillonite and ε-poly-l-lysine for antimicrobial active food packaging. J. Appl. Polym. Sci. 2020, 137, 48782. [Google Scholar] [CrossRef]
- Peighambardoust, S.J.; Zahed-Karkaj, S.; Peighambardoust, S.H.; Ebrahimi, Y.; Peressini, D. Characterization of carboxymethyl cellulose-based active films incorporating non-modified and Ag or Cu-modified Cloisite 30B and montmorillonite nanoclays. Iran. Polym. J. 2020, 29, 1087–1097. [Google Scholar] [CrossRef]
- Achachlouei, B.F.; Zahedi, Y. Fabrication and characterization of CMC-based nanocomposites reinforced with sodium montmorillonite and TiO2 nanomaterials. Carbohydr. Polym. 2018, 199, 415–425. [Google Scholar] [CrossRef]
- Sommer, A.; Staroszczyk, H.; Sinkiewicz, I.; Bruździak, P. Preparation and Characterization of Films Based on Disintegrated Bacterial Cellulose and Montmorillonite. J. Polym. Environ. 2021, 29, 1526–1541. [Google Scholar] [CrossRef]
- Salarbashi, D.; Tafaghodi, M.; Bazzaz, B.S.F. Soluble soybean polysaccharide/TiO2 bionanocomposite film for food application. Carbohydr. Polym. 2018, 186, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, C.; Zheng, X.; Chen, M.; Tang, K. Soluble soybean polysaccharide/nano zinc oxide antimicrobial nanocomposite films reinforced with microfibrillated cellulose. Int. J. Biol. Macromol. 2020, 159, 793–803. [Google Scholar] [CrossRef]
- Ghasemzadeh, H.; Afraz, S.; Moradi, M.; Hassanpour, S. Antimicrobial chitosan-agarose full polysaccharide silver nanocomposite films. Int. J. Biol. Macromol. 2021, 179, 532–541. [Google Scholar] [CrossRef] [PubMed]
- Jin, B.; Li, X.; Zhou, X.; Xu, X.; Jian, H.; Li, M.; Guo, K.; Guan, J.; Yan, S. Fabrication and characterization of nanocomposite film made from a jackfruit filum polysaccharide incorporating TiO2 nanoparticles by photocatalysis. RSC Adv. 2017, 7, 16931–16937. [Google Scholar] [CrossRef] [Green Version]
- Salarbashi, D.; Tafaghodi, M.; Bazzaz, B.S.F.; Birjand, S.M.A.; Bazeli, J. Characterization of a green nanocomposite prepared from soluble soy bean polysaccharide/Cloisite 30B and evaluation of its toxicity. Int. J. Biol. Macromol. 2018, 120, 109–118. [Google Scholar] [CrossRef]
- Salarbashi, D.; Tafaghodi, M.; Bazzaz, B.S.F.; Mohammad Aboutorabzade, S.; Fathi, M. pH-sensitive soluble soybean polysaccharide/SiO 2 incorporated with curcumin for intelligent packaging applications. Food Sci. Nutr. 2021, 9, 2169–2179. [Google Scholar] [CrossRef]
- He, Q.; Huang, Y.; Lin, B.; Wang, S. A nanocomposite film fabricated with simultaneously extracted protein-polysaccharide from a marine alga and TiO2 nanoparticles. J. Appl. Phycol. 2017, 29, 1541–1552. [Google Scholar] [CrossRef]
- Dai, L.; Xi, X.; Li, X.; Li, W.; Du, Y.; Lv, Y.; Wang, W.; Ni, Y. Self-assembled all-polysaccharide hydrogel film for versatile paper-based food packaging. Carbohydr. Polym. 2021, 271, 118425. [Google Scholar] [CrossRef]
- Salarbashi, D.; Noghabi, M.S.; Bazzaz, B.S.F.; Shahabi-Ghahfarrokhi, I.; Jafari, B.; Ahmadi, R. Eco-friendly soluble soybean polysaccharide/nanoclay Na+ bionanocomposite: Properties and characterization. Carbohydr. Polym. 2017, 169, 524–532. [Google Scholar] [CrossRef]
- Venkateshaiah, A.; Cheong, J.Y.; Habel, C.; Wacławek, S.; Lederer, T.; Černík, M.; Kim, I.-D.; Padil, V.V.T.; Agarwal, S. Tree Gum-Graphene Oxide Nanocomposite Films as Gas Barriers. ACS Appl. Nano Mater. 2020, 3, 633–640. [Google Scholar] [CrossRef] [Green Version]
- Salarbashi, D.; Tafaghodi, M.; Bazzaz, B.S.F.; Jafari, B. Characterization of soluble soybean (SSPS) polysaccharide and development of eco-friendly SSPS/TiO2 nanoparticle bionanocomposites. Int. J. Biol. Macromol. 2018, 112, 852–861. [Google Scholar] [CrossRef]
- Cai, Z.; Dai, Q.; Guo, Y.; Wei, Y.; Wu, M.; Zhang, H. Glycyrrhiza polysaccharide-mediated synthesis of silver nanoparticles and their use for the preparation of nanocomposite curdlan antibacterial film. Int. J. Biol. Macromol. 2019, 141, 422–430. [Google Scholar] [CrossRef]
- González, K.; Iturriaga, L.; González, A.; Eceiza, A.; Gabilondo, N. Improving mechanical and barrier properties of thermoplastic starch and polysaccharide nanocrystals nanocomposites. Eur. Polym. J. 2020, 123, 109415. [Google Scholar] [CrossRef]
- Zhao, K.; Wang, W.; Teng, A.; Zhang, K.; Ma, Y.; Duan, S.; Li, S.; Guo, Y. Using cellulose nanofibers to reinforce polysaccharide films: Blending vs layer-by-layer casting. Carbohydr. Polym. 2020, 227, 115264. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Lin, L.; Guo, Y.; Long, J.; Mu, R.-J.; Pang, J. Enhanced functional properties of nanocomposite film incorporated with EGCG-loaded dialdehyde glucomannan/gelatin matrix for food packaging. Food Hydrocoll. 2020, 108, 105863. [Google Scholar] [CrossRef]
- Shahabi-Ghahfarrokhi, I.; Babaei-Ghazvini, A. Using photo-modification to compatibilize nano-ZnO in development of starch-kefiran-ZnO green nanocomposite as food packaging material. Int. J. Biol. Macromol. 2019, 124, 922–930. [Google Scholar] [CrossRef]
- Makwana, D.; Castaño, J.; Somani, R.S.; Bajaj, H.C. Characterization of Agar-CMC/Ag-MMT nanocomposite and evaluation of antibacterial and mechanical properties for packaging applications. Arab. J. Chem. 2020, 13, 3092–3099. [Google Scholar] [CrossRef]
- Goudarzi, V.; Shahabi-Ghahfarrokhi, I. Development of photo-modified starch/kefiran/TiO2 bio-nanocomposite as an environmentally-friendly food packaging material. Int. J. Biol. Macromol. 2018, 116, 1082–1088. [Google Scholar] [CrossRef]
- Ni, Y.; Sun, J.; Wang, J. Enhanced antimicrobial activity of konjac glucomannan nanocomposite films for food packaging. Carbohydr. Polym. 2021, 267, 118215. [Google Scholar] [CrossRef]
- Dairi, N.; Ferfera-Harrar, H.; Ramos, M.; Garrigós, M.C. Cellulose acetate/AgNPs-organoclay and/or thymol nano-biocomposite films with combined antimicrobial/antioxidant properties for active food packaging use. Int. J. Biol. Macromol. 2019, 121, 508–523. [Google Scholar] [CrossRef] [Green Version]
- Hou, X.; Xue, Z.; Xia, Y.; Qin, Y.; Zhang, G.; Liu, H.; Li, K. Effect of SiO2 nanoparticle on the physical and chemical properties of eco-friendly agar/sodium alginate nanocomposite film. Int. J. Biol. Macromol. 2019, 125, 1289–1298. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Li, Y.; Du, Y.; Wang, L.; Tong, C.; Hu, Y.; Pang, J.; Yan, Z. Preparation and characterization of konjac glucomannan-based bionanocomposite film for active food packaging. Food Hydrocoll. 2019, 89, 682–690. [Google Scholar] [CrossRef]
- Ma, Z.; Liu, J.; Liu, Y.; Zheng, X.; Tang, K. Green synthesis of silver nanoparticles using soluble soybean polysaccharide and their application in antibacterial coatings. Int. J. Biol. Macromol. 2021, 166, 567–577. [Google Scholar] [CrossRef] [PubMed]
- Davachi, S.M.; Shekarabi, A.S. Preparation and characterization of antibacterial, eco-friendly edible nanocomposite films containing Salvia macrosiphon and nanoclay. Int. J. Biol. Macromol. 2018, 113, 66–72. [Google Scholar] [CrossRef] [PubMed]














| Polysaccharide | Reinforcing Agent | Antimicrobial Activity | Food Tested | Reference | |
|---|---|---|---|---|---|
| 1. | Chitosan lactate | Silver nanoparticle | E. coli | -- | [38] |
| 2. | Chitosan | Biogenic silver nanoparticle | E. coli, P. aeruginosa, B. subtilis, S. aureus | -- | [41] |
| 3. | Chitosan | Silver nanoparticle within graphene oxide | E. coli and S. aureus | -- | [45] |
| 4. | Chitosan/gelatin | Silver nanoparticle | -- | Green grapes | [47] |
| 5. | Chitosan | Silver nanoparticle | E. coli, Salmonella, S. aureus, L. monocytogenes | -- | [48] |
| 6. | Chitosan | Cellulose nanocrystal | -- | -- | [51] |
| 7. | Chitosan/guar gum | Cellulose nanocrystal | -- | -- | [52] |
| 8. | Chitosan | Cellulose nanocrystal | -- | -- | [53] |
| 9. | Chitosan | Cellulose nanocrystal | -- | -- | [54] |
| 10. | Chitosan | Cellulose nanocrystal | -- | -- | [55] |
| 11. | Chitosan | Montmorillonite | -- | -- | [65] |
| 12. | Chitosan | Montmorillonite | Coliforms | Fresh poultry meat | [66] |
| 13. | Chitosan | Montmorillonite | -- | -- | [67] |
| 14. | Chitosan | Acidified modified montmorillonite | S. aureus, E. coli | -- | [68] |
| 15. | Chitosan nanoparticles/ chitosan | Montmorillonite | -- | Sliced dry-cured ham | [72] |
| Polysaccharide | Reinforcing Agent | Antimicrobial Activity | Food Tested | Reference | |
|---|---|---|---|---|---|
| 1. | Starch | Silver nanoparticle | -- | -- | [81] |
| 2. | Hydrophobic Noncrystalline Porous Starch | Silver nanoparticle | E. coli, S, aureus | -- | [82] |
| 3. | Sugar palm starch | Silver nanoparticle | E. coli, S, aureus, S. cholerasuis | -- | [83] |
| 4. | Amazonian tuber starch | Silver nanoparticle | E. coli, S, aureus | -- | [84] |
| 5. | Starch | Silver nanoparticle | E. coli, Salmonella spp. | Fresh cheese | [85] |
| 6. | Native rice starch | Nanocellulose extracted from banana pseudostems | -- | -- | [94] |
| 7. | Thermoplastic starch | Cellulose nanofiber | -- | -- | [95] |
| 8. | Starch | Silver nanoparticles immobilized on cellulose nanofibrils | E. coli, S, aureus | -- | [96] |
| 9. | Starch | Carrot cellulose nano fibers | E. coli, L. monocytogenes, S. typhimurium, Penicillium spp. | Grapes | [97] |
| 10. | Plasticised sugar palm starch | Nanofibrillated celluloses from sugar palm fibres | -- | -- | [98] |
| 11. | Iranian potato starch | Montmorillonite | -- | -- | [104] |
| 12. | Cationic starch | Nanoclay montmorillonite and ZnO nanopowders | -- | -- | [105] |
| 13. | Starch | Montmorillonite | E. coli | -- | [106] |
| 14. | Thermoplastic starch | Montmorillonite | Botrytis cinerea | Strawberries | [107] |
| 15. | Cassava starch and polycaprolactone | Sodium montmorillonite | -- | -- | [108] |
| Polysaccharide | Reinforcing Agent | Antimicrobial Activity | Food Tested | Reference | |
|---|---|---|---|---|---|
| 1. | Cellulose | Silver nanoparticle | S. aureus, E. coli | -- | [116] |
| 2. | Bacterial cellulose | Silver nanoparticle | -- | -- | [117] |
| 3. | Regenerated cellulose | Amino-terminated hyperbranched polyamic anchored nanosilver | S. aureus, E. coli | -- | [118] |
| 4. | Cellulose acetate | Silver nanoparticle | S. aureus, B. cereus, S. typhi, E. coli, K. pneumoniae, Pseudomonase spp. | -- | [119] |
| 5. | Cellulose nanofibrils | Silver nanoparticle | E. coli, L. monocytogenes | -- | [120] |
| 6. | Cellulose nanofibers | Cellulose nanocrystals | -- | -- | [124] |
| 7. | Bacterial cellulose | Lignin and cellulose nanocrystals from cashew tree pruning fiber | S. aureus, E. Coli, S. cerevisiae | -- | [125] |
| 8. | Carboxymethyl cellulose/agar | Cellulose nanocrystals | -- | -- | [126] |
| 9. | Sugarcane bagasse nanofibers | -- | -- | -- | [127] |
| 10. | Gluten/ carboxymethyl cellulose | Cellulose nanofiber | -- | -- | [128] |
| 11. | Chitin/cellulose | Montmorillonite nanoclay | -- | -- | [130] |
| 12. | Cellulose | Octadecylamine-modified montmorillonite | C. violaceum | -- | [131] |
| 13. | Carboxymethyl cellulose | Montmorillonite and ε-poly-l-lysine | S. aureus, E. Coli, B. Cinerea, R. oligosporus | Strawberries | [132] |
| 14. | Carboxymethyl cellulose | Cloisite 30B, montmorillonite | S. aureus, E. Coli | -- | [133] |
| 15. | Carboxymethyl cellulose | sodium montmorillonite, titanium dioxide | -- | -- | [134] |
| Polysaccharide | Reinforcing Agent | Key Properties | Antimicrobial Activity | Food Tested | Reference | |
|---|---|---|---|---|---|---|
| 1. | Soluble soybean polysaccharide | Titanium dioxide | Antimicrobial activity; neither anti-cancerous nor pro-cancerous | S. aureus PTCC 1431 | -- | [136] |
| 2. | Soluble soybean polysaccharide | Nano zinc oxide, microfibrillated cellulose | Antimicrobial activity | E. coli, B. subtlis | -- | [137] |
| 3. | Chitosan/agarose | Silver | Antibacterial activity | P. aeruginosa, E. coli, S. aureus | -- | [138] |
| 4. | Jackfruit filum polysaccharide | Titanium dioxide | Biodegradable; decreased the transparency, moisture content, and total soluble matter | E. coli, S. aureus | -- | [139] |
| 5. | Soluble soy bean polysaccharide | Cloisite 30B | Antibacterial activity; only packaging of solids foods such as bread | S. typhi, S. epidermis, L. monocytogenes, A. niger | Bread | [140] |
| 6. | Soluble soy bean polysaccharide | Silica nanoparticles | Antibacterial activity; water solubility, and water vapor permeability decreased; mechanical performance increased | S. aureus, B. subtilis, S. epidermidis | Shrimp | [141] |
| 7. | Protein/polysaccharide from the marine red alga (Gracilaria lemaneiformis) | Titanium dioxide | Antibacterial activity; nutrition retention and quality improvement of food products | E. coli, S. aureus | Cherry tomatoes | [142] |
| 8. | Cationic guar gum | TEMPO-oxidized cellulose nanofibers | Maintained mooncake’s freshness | -- | Mooncakes | [143] |
| 9. | Soluble soybean polysaccharide | Montmorillonite | Antimicrobial activity against bacteria and mold; storage modulus of nanocomposites increased | B. cereus, S. aureus, E. coli, P. expansum | -- | [144] |
| 10. | Gum arabic, gum karaya, Kondagogu gum | Graphene oxide | Oxygen gas barriers; freestanding film with substantial mechanical strength | -- | -- | [145] |
| 11. | Soluble soybean polysaccharide | Titanium dioxide | Antimicrobial activity; decreased water solubility, moisture content, and water-vapor permeability | S. epidermidis, S. epidermis, L. monocytogenes, P. expansum | -- | [146] |
| 12. | Curdlan | Glycyrrhiza polysaccharides-stabilized silver nanoparticles | Antimicrobial activity | E. coli | -- | [147] |
| 13. | Thermoplastic starch | Waxy starch nanocrystals | Effective interfacial hydrogen bonding between the polymer and the reinforcing agent | -- | -- | [148] |
| 14. | Alginate, chitosan | Cellulose nanofiber | Higher tensile strength and better water barrier property | -- | -- | [149] |
| 15. | Konjac glucomannan/gelatin | Rutin functionalized cellulose nanocrystal | Antibacterial activity; higher mechanical and barrier properties | S. aureus, E. coli | -- | [150] |
| 16. | Starch/kefiran | Zinc oxide nanoparticles | Antibacterial activity; higher mechanical properties; decreased water-related barrier properties | -- | -- | [151] |
| 17. | Agar/carboxymethyl cellulose | Silver modified montmorillonite | Antibacterial activity | B. subtilis, E. coli | -- | [152] |
| 18. | Starch/kefiran | Titanium dioxide | Higher tensile strength; decreased water-related barrier properties | -- | -- | [153] |
| 19. | Konjac glucomannan | Graphite carbon nitride nanosheets/ molybdenum disulfide nanodots | Sustained and efficient antimicrobial activity; Hemo- and cytocompatible | S. aureus, E. coli | Cherry tomatoes | [154] |
| 20. | Cellulose acetate | Silver nanoparticles /gelatin-modified montmorillonite nanofiller | Antimicrobial and antioxidant properties | E. coli | -- | [155] |
| 21. | Agar/sodium alginate | Nano-silica | Enhanced mechanical properties, water resistance, and thermal stability | -- | -- | [156] |
| 22. | Konjac glucomannan | Chitosan/gallic acid nanoparticles | Enhanced mechanical and barrier properties; Excellent mechanical properties | S. aureus, E. coli | -- | [157] |
| 23. | Soluble soybean polysaccharide | Silver nanoparticles | Excellent antibacterial property | E. coli, S. aureus, P. aeruginosa | -- | [158] |
| 24. | Salvia macrosiphon seed mucilage | Nanoclay | Increased hydrophobicity; antimicrobial property | S. aureus, E. coli | -- | [159] |
| 25. | Sweet potato starch/ lemon-waste pectin | Nano-titania | Biodegradable packaging material; increased thermal stability | -- | -- | [160] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pal, K.; Sarkar, P.; Anis, A.; Wiszumirska, K.; Jarzębski, M. Polysaccharide-Based Nanocomposites for Food Packaging Applications. Materials 2021, 14, 5549. https://doi.org/10.3390/ma14195549
Pal K, Sarkar P, Anis A, Wiszumirska K, Jarzębski M. Polysaccharide-Based Nanocomposites for Food Packaging Applications. Materials. 2021; 14(19):5549. https://doi.org/10.3390/ma14195549
Chicago/Turabian StylePal, Kunal, Preetam Sarkar, Arfat Anis, Karolina Wiszumirska, and Maciej Jarzębski. 2021. "Polysaccharide-Based Nanocomposites for Food Packaging Applications" Materials 14, no. 19: 5549. https://doi.org/10.3390/ma14195549










