In the next stage, a comprehensive physicochemical analysis of the obtained binary systems was carried out to determine their crystallographic and morphological parameters, surface composition, and thermal stability.
3.2.1. Crystal Structure
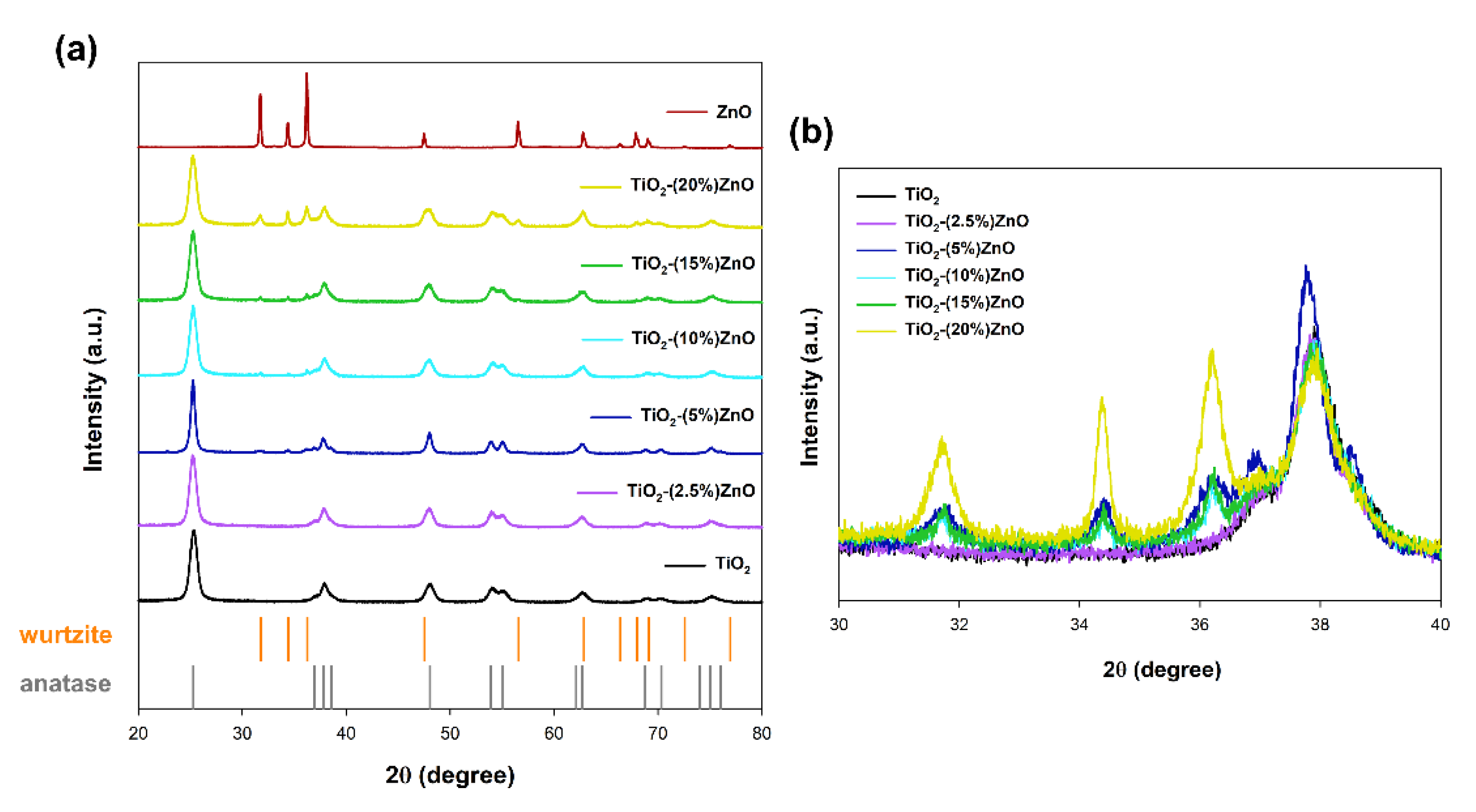
In the case of materials based on metal oxides, one of the most critical parameters is the crystal structure, which determines the subsequent application properties. The obtained XRD patterns are presented in
Figure 4.
For both reference samples of titanium dioxide and zinc oxide, the crystal structure of tetragonal anatase (space group
I41/amd, no. 141; databased card no. 9009086) and hexagonal wurtzite (space group
P63mc no. 186; databased card no. 2300112) was recorded, respectively. Many reports show the anatase and wurtzite structures for samples synthesized with hydrothermal treatment [
39]. Similar observations were also demonstrated in our earlier works on the one-step hydrothermal-assisted synthesis of TiO
2-ZnO binary systems [
40].
In the next stage, the obtained XRD patterns for TiO
2-ZnO systems were analyzed. Based on the obtained results, it was shown that the addition of ZnO ≤ 5 wt.% caused the crystallization of the wurtzite structure, as confirmed by the diffraction bands at 2θ = 31.6, 34.3, as well as 36.1. On the other hand, only the anatase structure was observed for the material TiO
2-(2.5%)ZnO. Additionally, no shift on the XRD pattern was noted for the above-mentioned two-component material compared to the reference titania sample (
Figure 4b). Therefore, it should be supposed that the crystallization of ZnO can be inhibited by further growth of TiO
2 in the next step of the hydrothermal treatment.
The average crystallite size and phase composition were determined in the next step of the crystallographic analysis (see
Table 2).
By analyzing the presented crystallinity results, it was shown that the crystallite size of 10 nm characterized the reference titania material. In contrast, for zinc oxide, an average crystallite size of 45 nm was determined. According to the previously presented data, no crystalline phase of wurtzite was observed for the material containing 2.5 wt.% of ZnO (TiO
2-(2.5%)ZnO). However, for the sample mentioned above, an increase of the crystallites size of anatase (the largest crystallite size of all analyzed materials) was confirmed. Considering the possible reason for the lack of ZnO crystallinity in the discussed material, it is necessary to quote Ludi and Niederberger’s [
41] work. The authors indicate that the crystallization process can be inhibited if the amount of locking agent is high. Moreover, only single ZnO particles were observed for these materials. Additionally, Garcia and Samancik [
42] found that many modification agents can determine the growth of ZnO crystal in different directions. Scientists have shown, among others, the addition of citrate ions inhibits axial growth. However, the increase in crystal width could imply that mentioned ions accelerate the equatorial growth of ZnO crystal. Likewise, the modification agents might suppress nucleation, allowing higher supersaturation in the solution, forming larger crystal dimensions overall. However, the modifying agent used in our work, TiO
2 modified with triethylamine, due to amine groups, attaches the non-polar facets of the zincite crystal, thereby cutting off the access of Zn
2+ ions to them, leaving only the polar (001) face for epitaxial growth [
20,
43]. For this reason, at a low concentration of Zn
2+ ions, the formation of the ZnO structure may be inhibited. In the case of other synthesized TiO
2-ZnO systems, the presence of both tetragonal anatase and hexagonal wurtzite phases was noted—in a similar ratio as assumed. On the other hand, when analyzing the values of the average size of crystallites, attention should be paid to the significant size of ZnO crystallites in the range of 43–45 nm, which may also confirm the previous considerations wurtzite crystallization process. It was noted that the TiO
2-based systems containing from 2.5–10 wt.% of ZnO were characterized by a larger size of anatase crystallites, which is related to their growth during the secondary hydrothermal treatment.
In the next stage of research on the crystal structure, determining the direction of crystal growth and defining the main planes influencing their growth was evaluated. Thus, the HR-TEM analysis (
Figure 5) was carried out, considering the growth of metal oxide crystals during hydrothermal treatment.
According to the available scientific literature, the orientation of the growth of crystalline zinc oxide under certain heat treatment conditions is well defined [
44]. Therefore, HR-TEM imaging focused mainly on TiO
2 particles for which the synthesis conditions have a determining the influence on the crystal growth. In HR-TEM and FFT images (
Figure 5), TiO
2 particles in TiO
2-ZnO systems are correctly oriented with respect to the impinging electron beam showing lattice fringes 0.23 nm apart (d
004 = 0.2378 nm) [
25]. Thus, they correspond to {001} surfaces. Based on the presented HR-TEM data, as well as the available scientific literature, including Mino et al. [
45], it was confirmed that the planes (101), (004), and (200) had a determining influence on the formation of anatase nanocrystals. Therefore, the crystallite size was presented in the next step for the planes mentioned above (
Table 3).
Based on the data presented in
Table 3, it can be assumed, above all, that the crystallite sizes for the crucial crystalline planes are similar to each other. Additionally, it was noted that the crystallites of planes (004) and (200) slightly decreased with increasing zinc oxide addition, which may indicate the influence of ZnO on the anatase crystal structure.
Based on the XRD results obtained, it was shown that the proposed two-stage synthesis methodology based on the modification of titania and then the synthesis of TiO
2-ZnO systems resulted in preparation materials with two crystalline phases—tetragonal anatase and hexagonal wurtzite. In addition, it was noted that no crystalline phase of wurtzite was observed in the case of materials containing a small addition of ZnO (≥5%). This can be caused by the high amount of capping agent (in this case, nanocrystalline titanium dioxide), leading to the formation of single ZnO particles. Furthermore, attention should be paid to the refined lattice parameters for TiO
2-ZnO systems that are in good agreement with other literature reports [
46,
47].
3.2.2. Morphology and Surface Composition
The shape and size of nanoparticles can determine their application properties, e.g., defective structure or different access to individual planes. Therefore, a detailed analysis of morphology and microstructure is one of the critical parameters in materials science. For the obtained TiO
2-ZnO systems, transmission electron microscopy was performed, and the results are presented in
Figure 6.
Single spherical and cubic nanoparticles with a size of about 10 nm were observed for the reference titanium dioxide sample (
Figure 5a). Based on the above-described crystallinity results, the average size of crystallites for titania was determined to be 10 nm, which is a similar value to the size of a single particle. Thus, it was proved that TiO
2 was in the form of single nanocrystalline particles. For the sample containing 2.5 wt.% of ZnO (TiO
2-(2.5%) ZnO) (
Figure 5b), only the single nanoparticles (about 10 nm in size) were noted, which is similar to the morphology observed for the reference titania material. The increase of ZnO addition to 5–10 wt.% (TiO
2-(5%)ZnO and TiO
2-(10%)ZnO) (
Figure 5c,d) resulted in the presence of a new morphological structure. ZnO nanowires with a 10–20 nm diameter were observed for the materials mentioned above, and the structure of nanocrystalline TiO
2 particles was also found. A further increase in ZnO content (TiO
2-(15%)ZnO and TiO
2-(20%)ZnO) (
Figure 5e,f) resulted in an increase in the diameter of ZnO nanowires (diameter about 50 nm). The change of ZnO addition did not affect the morphology of the TiO
2 phase. On the other hand, for the ZnO reference sample, pyramidal and rod structures were approximately 2 µm (
Figure 5g). The structures mentioned above are described in the scientific literature for ZnO materials synthesized in the presence of amines in a basic medium [
20,
43]. It should be noted that both pyramidal and rod-like structures were not observed for the TiO
2-ZnO systems which only ZnO nanowires were found. The main reason for the formation of ZnO nanowires is the use of TiO
2 modified with triethylamine.
The high-resolution transmission electron microscopy with fast Fourier transformation was carried out (
Figure 7) to confirm the existence of a surface junction between titanium dioxide and zinc oxide.
Based on the presented HRTEM and FFT images, the high crystallinity of the analyzed systems was confirmed due to observed crystallographic spacing. It was found that selected TiO
2-ZnO samples have spacing characteristics for a plane (101) and (004) of anatase [
43] and (002) of wurtzite [
48]. Attention was also paid to the crossing of lattice fringes of titania and zinc oxide. Such connections have been described as a heterojunction which is widely known in the scientific literature [
31,
49].
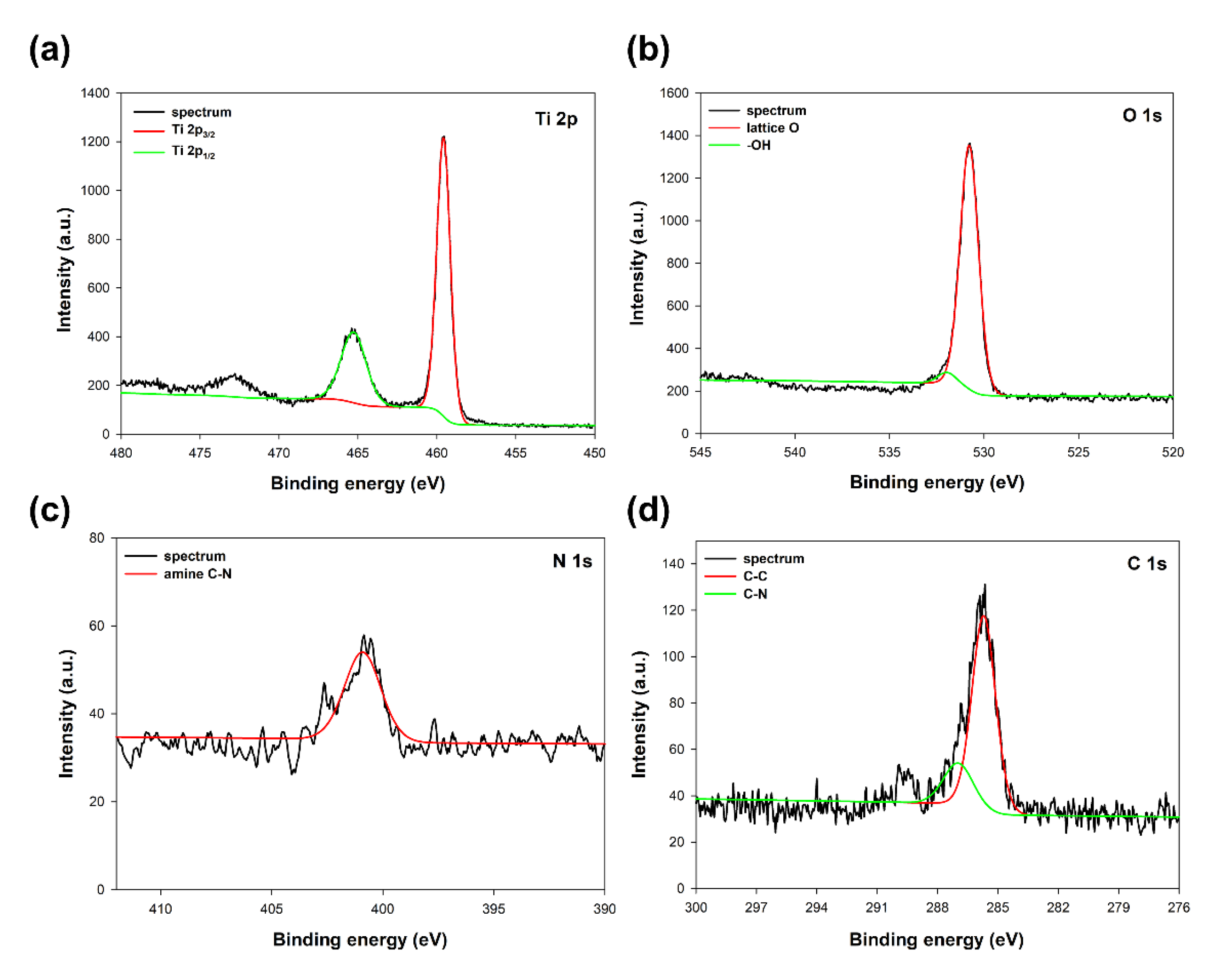
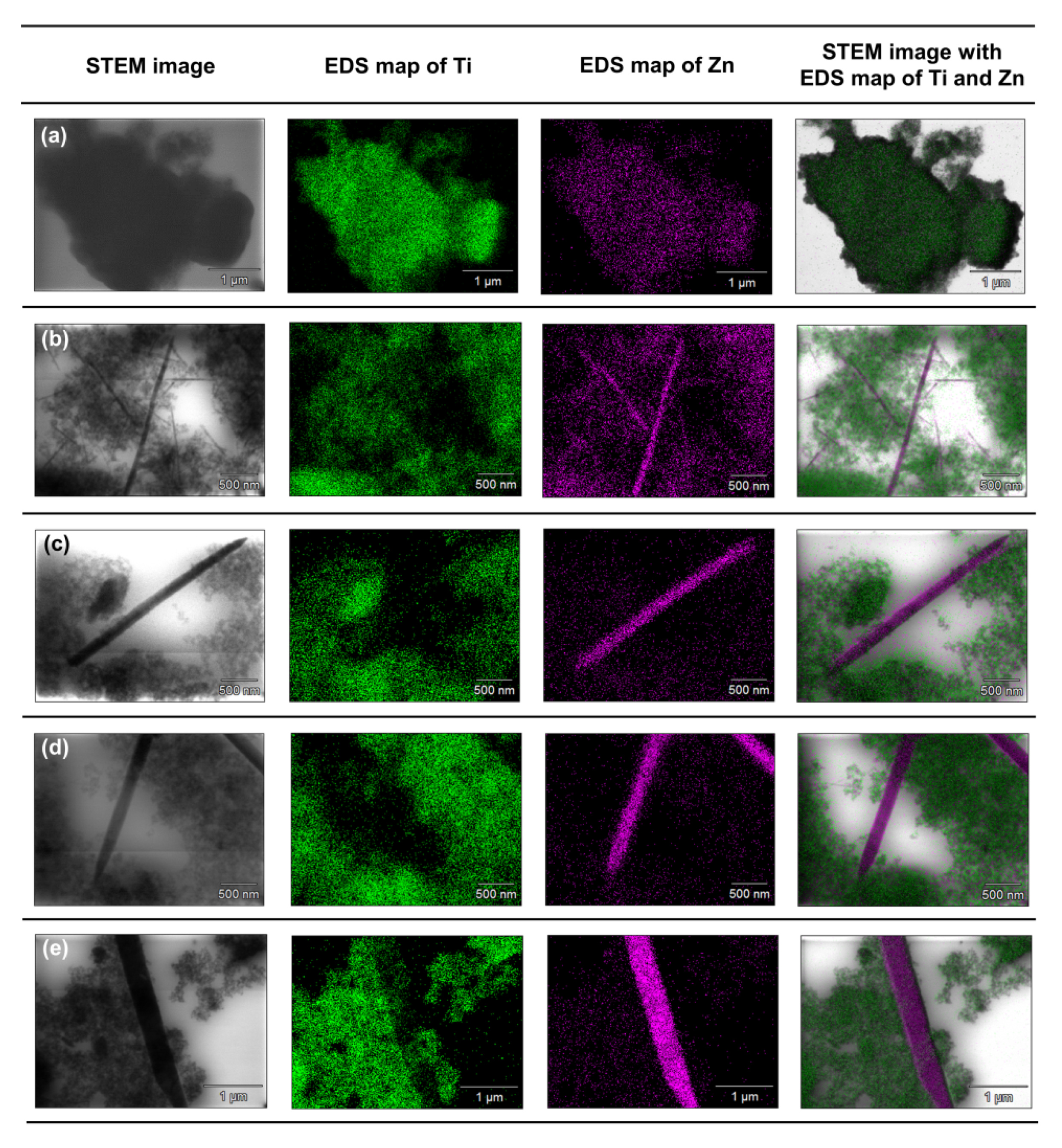
To accurately determine the position of individual components in the synthesized systems, an analysis of the EDX mapping (
Figure 8) and EDXRF (
Table 4) was performed.
Based on the presented EDX data, it can be concluded that for the TiO
2-(2.5%)ZnO sample, aggregates of titania particles were observed, while ZnO is present only on single nanoparticles. The described observations confirm that with a low concentration of ZnO additive, titanium oxide can play a role as a capping agent, which inhibits ZnO crystallization and precludes obtaining morphological structures known from the literature [
5,
50]. In the case of the TiO
2-(5%)ZnO sample, apart from TiO
2 nanoparticle aggregates, there are also single particles and ZnO nanowires of various lengths. The presence of both nanowires and individual ZnO nanoparticles indicates that the increase in ZnO addition to 5 wt.% results in the crystallization of wurtzite and the formation of crystalline nanowires. However, some of the particles are still blocked by titanium dioxide, which is consistent with the XRD data obtained for the material mentioned above. The content of the crystalline phase of wurtzite (3.8%) was determined as expected. For the remaining analyzed materials (ZnO = 10, 15, and 20 wt.%) apart from the phase of nanocrystalline TiO
2 particles, the structure of ZnO nanowires without the presence of single particles was also noted, which may indicate that with the ZnO content equal to 10 wt.%, titanium dioxide ceases to be an inhibition of crystallization growth of ZnO. In addition, based on the results of EDS mapping (
Figure 8), it was indicated that TiO
2 nanoparticle aggregates are observed near crystalline ZnO nanowires. Such a result may also confirm the proposed nanofiber growth mechanism based on interactions of the modified titania sample with the non-polar facets of the zincite crystal.
On the other hand, attention should be paid to the results of the EDXRF analysis (
Table 4). It was found that the determined percentages of titanium dioxide and zinc oxide are similar to the theoretical values assumed during synthesis. Therefore, it seems that the observed single ZnO particles in TiO
2 nanocrystalline aggregates are not incorporated in the titanium structure, but are amorphous ZnO particles whose crystal growth has been inhibited. Additional confirmation of the absence of ZnO particles incorporation was also showed by XRD data (crystal lattice parameters), which were close to the literature values [
51,
52].
There are many reports in the available scientific literature about the connections between titanium oxide and zinc oxide. Among others, Siwińska-Stefańska et al. [
53] and Perez-Gonzalez et al. [
54] confirmed that zinc oxide could inhibit the growth of zinc oxide during one-step synthesis anatase structure. For this reason, the advantageous solution seems to be the synthesis in two stages carry out, as presented, among others, by Sartori et al. [
55] and Cheng et al. [
56], who used commercial titanium dioxide (P25) for synthesis. However, the use of P25 is also associated with certain disadvantages related to its homogeneity, which was pointed out by Ohtani [
57]. At the same time, the homogeneity of the material is one of the crucial parameters that influence physicochemical properties. For this reason, it seems advantageous not only to synthesize a homogeneous and comprehensively characterized titanium dioxide, but also to modify it to obtain the designed oxide systems. The
in-situ modification method used in our work made it possible to influence the next stage of synthesis and obtain the determined crystalline ZnO nanowires.
3.2.6. Photocatalytic Activity
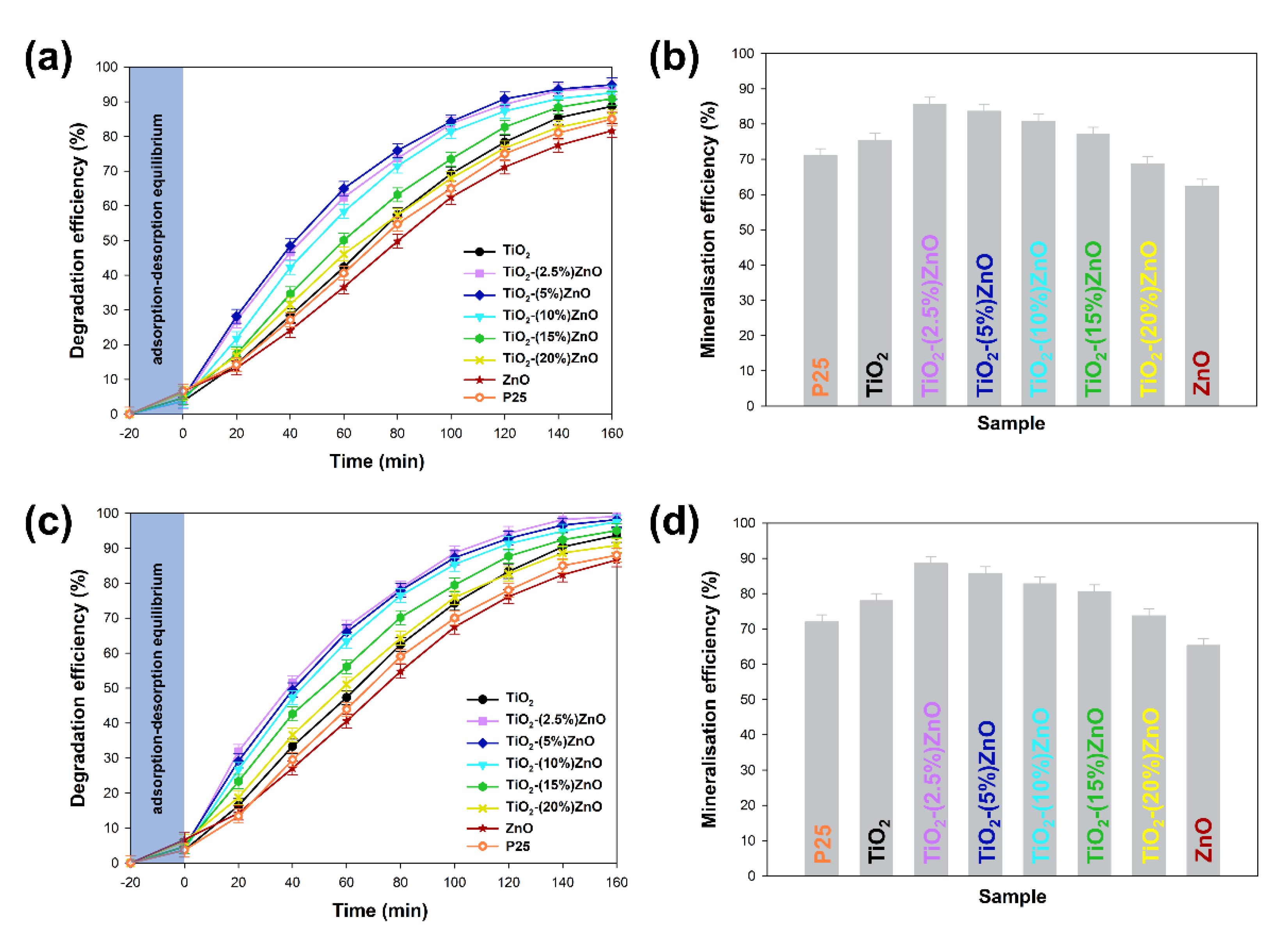
The obtained two-component systems were tested as active photocatalysts in the degradation process of 4-chlorophenol, bearing in mind the widely described photocatalytic properties of both titanium dioxide and zinc oxide. The critical element of the conducted photo-oxidation tests was applying low-power UV-LED lamps as a light source. Moreover, the influence of lamp power on the degradation process of the tested organic pollutant was determined. The obtained degradation curves, as well as the mineralization efficiency, are summarised in
Figure 12.
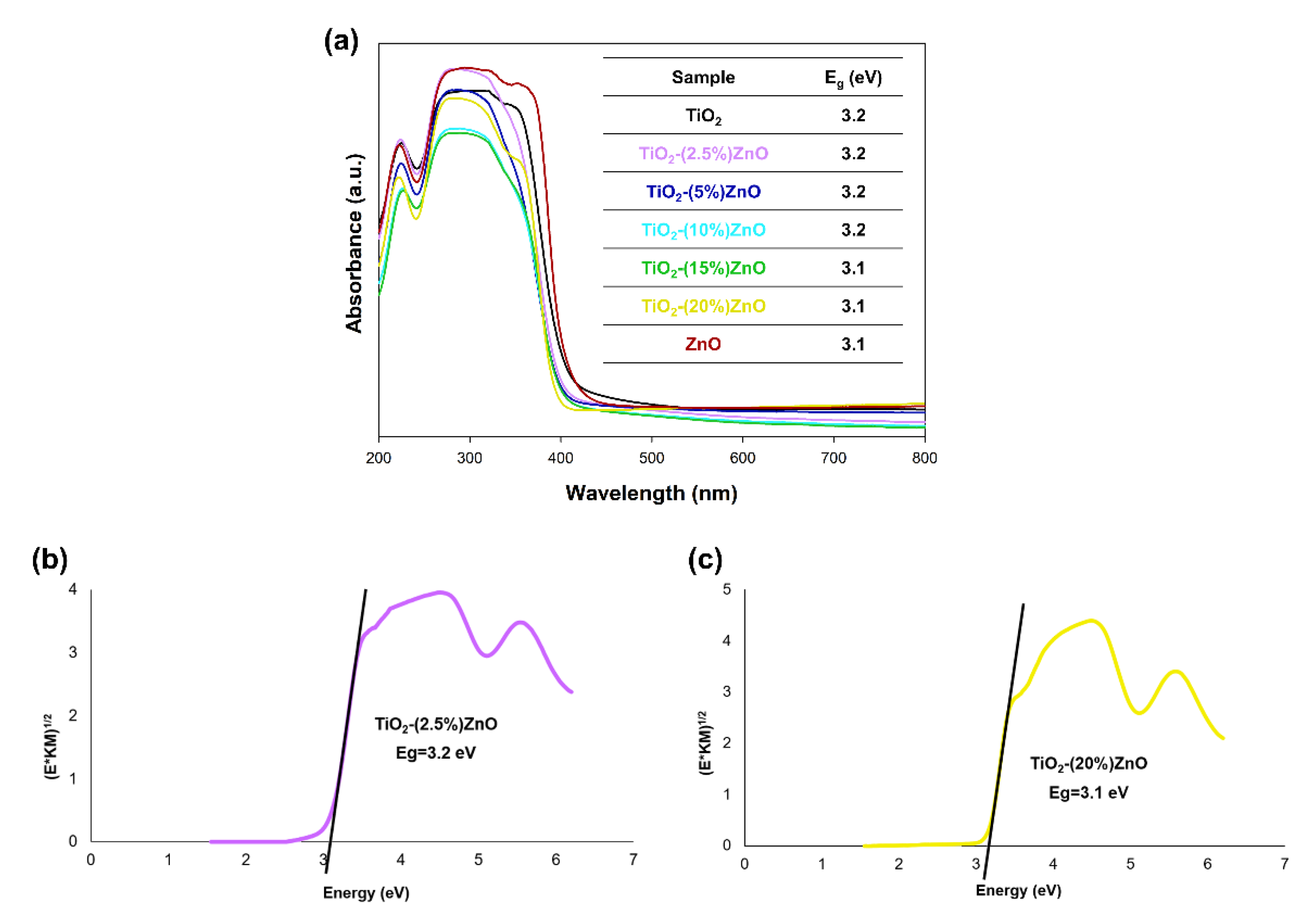
At the outset, it should be noted that regardless of the power of the light source, the analyzed materials were characterized with high photoactivity. By applying a 20 W lamp, about 3–5% higher removal efficiency was observed than when 10 W light source was used. Based on the conducted photo-oxidation tests, it was shown that the TiO2 and ZnO reference samples were characterized by correspondingly the degradation efficiency—88 and 81%, as well as mineralization—75% and 62% of the tested impurity. In case the two-component materials containing from 2.5 wt.% to 15 wt.% of ZnO, an increase in photocatalytic activity (both in the case of degradation and mineralization) was noted in relation to the reference titanium dioxide sample. For TiO2-(2.5%)ZnO, TiO2-(5%)ZnO, and TiO2-(10%)ZnO photocatalysts, a similar photodegradation curve was recorded, which may be related to the similar value of the band gap energy of the above materials. The mentioned materials showed degradation of 4-chlorophenol equal to about 94%, while the mineralization efficiency was in the range of 85–80%. Increasing the ZnO addition to 15 wt.% (TiO2-(15%)ZnO sample) resulted in a slight deterioration in the degradation efficiency (91%) of 4-chlorophenol as compared to the materials described above. For the TiO2-(20%)ZnO sample, a lower degradation (85%) and mineralization (68%) yield of the organic pollutant among the tested TiO2-ZnO systems were noted. The obtained results of photo-oxidation tests were compared with the commercially available photocatalyst—P25. The observed degradation efficiency reached 88%, while the mineralization efficiency was 72% regardless of the light source used. This allows indicating that the obtained TiO2-ZnO systems are characterized by a higher degree of removal of the tested pollutant, which may be related to a different photocatalytic mechanism and different absorption of radiation emitted by UV-LED diodes.
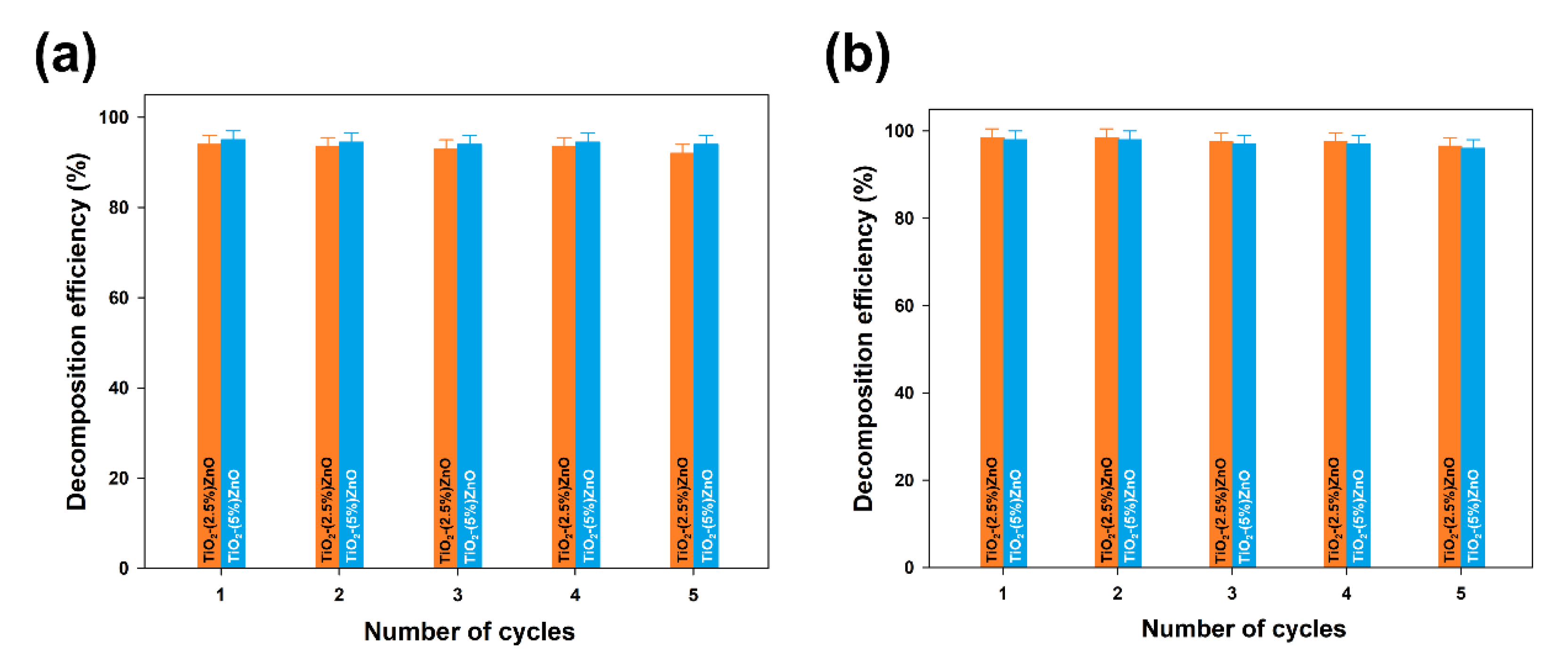
Furthermore, the TiO
2-ZnO systems with the highest removal efficiency (TiO
2-(2.5%)ZnO and TiO
2-(5%)ZnO samples) were selected for reusability studies. Five successive cycles were carried out to evaluate the effectiveness of the photocatalysts after their recovery. The data are shown in
Figure 13.
At the end of the first run of photo-oxidation tests, the TiO2-ZnO samples were separated from the aqueous solution by filtration. The separated materials were then reused without any purification. In each case, the efficiency of 4-chlorophenol removal using TiO2-ZnO systems was above 90%, even after 5 catalytic cycles, regardless of the UV-LED light source used. Such a result confirms that the synthesized systems can be used many times in photo-oxidation processes.
Based on the obtained results of the photo-oxidation test, it was found that the high removal of 4-chlorophenol characterizes the obtained TiO
2-ZnO systems. However, analyzing the obtained results, attention should be paid to the current standards of 4-chlorophenol concentration in the environment. According to the World Health Organization, the presence of chlorophenols in drinking water is equal to 2 µg/dm
3 [
61]. Hence, the obtained in the presented study mineralization efficiencies lead to the significant reduction of the concentration of 4-chlorophenol content in the aquatic environment and the fulfillment of the current WHO standards regarding the chlorophenols content in drinking water.
One of the fundamental observations resulting from the performed photo-oxidation tests was the influence of ZnO addition on the obtained photodegradation efficiency. It is well known that zinc oxide is a frequently used “second oxide” in combination with titanium dioxide, thereby improving the photocatalytic performance of the resulting binary oxide system. For example, Upadhyay et al. [
62] indicated that TiO
2-ZnO material is one of the most popular semiconductors couplings. However, the synthesized (molar/mass ratio) composition is one of the most important parameters, because it directly impacts the crystal structure and morphology. Xu et al. [
63] pointed out that the improvement of photocatalytic properties compared to pure TiO
2 can occur at the atomic ratio Ti/Zn = 3/1. Additionally, Fu et al. [
64] found that the photo-oxidation activity increases with increasing the TiO
2 doping content until the TiO
2-ZnO ratio is more than 5% ZnO, which can explain the observed increase in photodegradation efficiency for TiO
2-(2.5%)ZnO and TiO
2-(5%)ZnO materials. On the other hand, the high photoactivity of the TiO
2-(10%)ZnO system can be explained by using a light source with a specific wavelength and the value of the energy band gap similar to the previously described materials. Moreover, for the rest, materials containing 10 wt.% ZnO can be a highly efficient photocatalyst, which was highlighted by Siwińska-Stefańska et al. [
40]. The decrease of the photocatalytic activity in samples containing 15% and 20% ZnO results, among others, from the increase of carriers charge recombination effect [
65]. Furthermore, the higher zinc oxide content can cause an increase in opacity and light scattering, adversely influencing photon absorption. Based on the obtained results, it was shown that the addition of ZnO ranging from 2.5 wt.% to 10 wt.% of ZnO significantly improves the photocatalytic degradation of 4-chlorophenol in comparison to the reference TiO
2 sample. Additionally, it was observed that to improve the photocatalytic activity of the systems, ZnO does not have to be in a crystalline form—wurtzite. It seems that ZnO nanoparticles dispersed in TiO
2 aggregates can also effectively improve the photocatalytic performance in UV-LED light.
Another important conclusion is the lack of a decisive influence of the power of the used light source on the obtained degradation efficiency of the tested impurity. In our research, photocatalytic LED reactors based on strips of light-emitting diodes were used. Despite using SMD diodes, the discussed technological solution made it possible to use a uniform beam of light supplied to the entire reactor volume, which resulted in high degradation efficiency. To the best of our knowledge, in the available scientific literature, work concerning the use of LEDs in photocatalytic processes focuses on the use of COB modules [
66] or LED boards [
17,
18]. The main problem with the solutions mentioned above is the limited light flux, which decreases with the distance of the LED light source from the photo-reaction system, as described in detail by Casado et al. [
17]. For this reason, the solution presented by us seems to be interesting, which allows for even illumination of the reaction system using a low power of the light source. An additional advantage is using SMD LEDs, which are commonly used in commercially produced lighting fixtures despite their inferior parameters to COB systems. Therefore, they seem to be a more beneficial and cheaper solution at the current stage of LED technology development. However, the technological solution used should also consider the lack of visible influence of the power of the light source on the achieved degradation efficiency. According to the assumptions, the LED strip generates about 10 W per meter of the strip (60 LEDs). Therefore, in the case of a lamp with a higher power, it was necessary to use 120 LEDs to obtain complex radiation power. On the other hand, an increase in the number of diodes is also associated with an increase in light scattering and its partial reflection. Additionally, the light source used in our experiment had a specific wavelength of 395 nm. Consequently, to achieve maximum radiation absorption, it was possible to use a lamp with lower power. Nevertheless, it seems that the further development of photocatalysis based on LED light sources is another step in integrating photodegradation processes into environmental protection strategy.
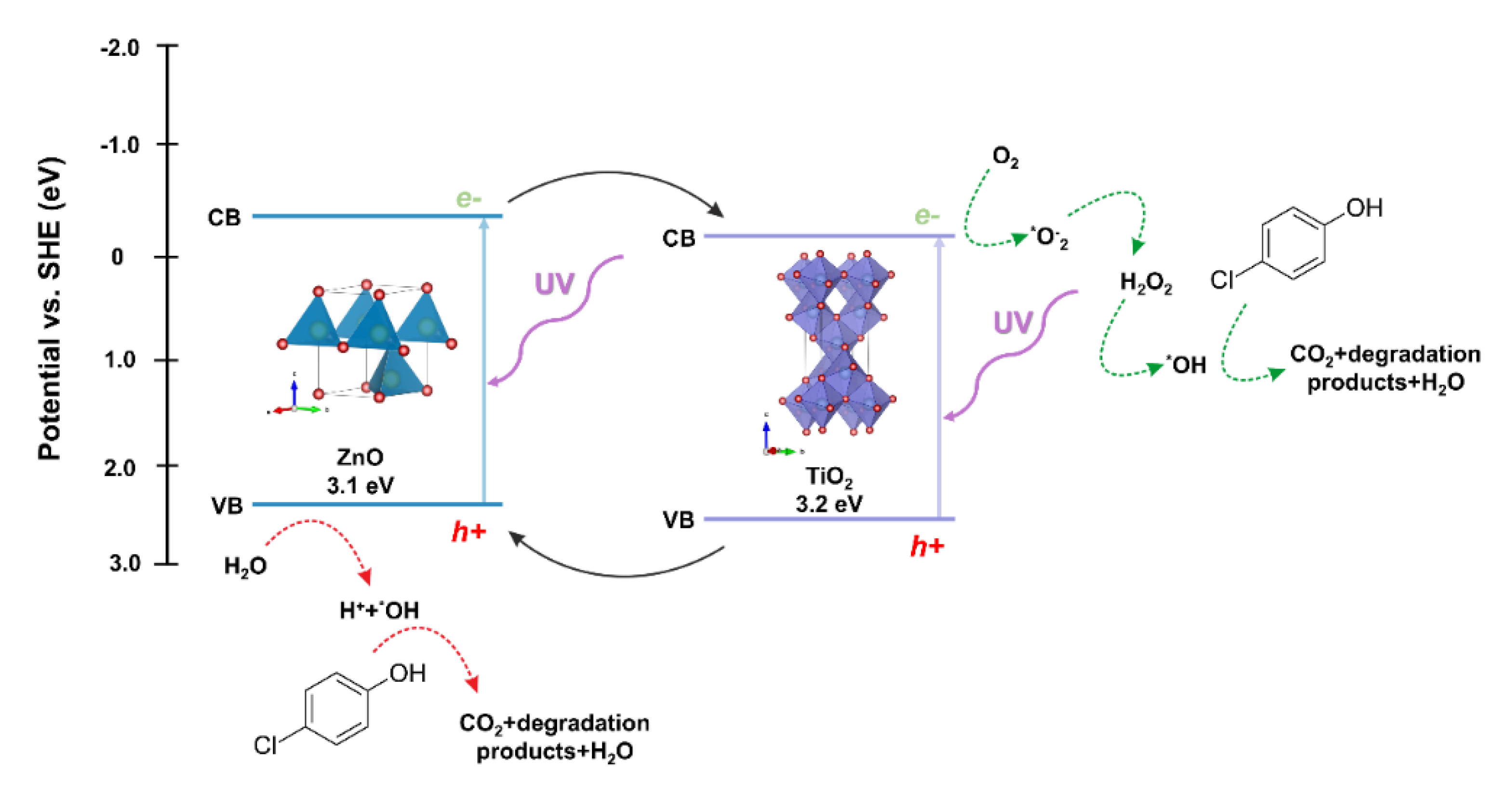
The next step in evaluating the photocatalytic properties of the synthesized two-component oxide systems was to propose a possible photodegradation mechanism. Due to the location of the conduction (CB) and valence bands (VB) of the semiconductors included in the described oxide systems, titanium dioxide and zinc oxide, the probable mechanism of the photocatalytic reaction is the heterojunction type II [
67]. The photo-excited electrons are transferred from the ZnO conduction band to CB TiO
2. This transfer can occur directly between the semiconductors due to the favorable energetics of the conduction bands’ position [
68]. The holes are transferred simultaneously from the titanium dioxide VB to the zinc oxide VB, reducing the likelihood of recombination and increasing the lifetime of the charge carriers. The oxidation process occurs in the zinc oxide valence band by reacting the holes with adsorbed water or surface hydroxyl groups, resulting in the formation of hydroxyl radicals. The reduction process occurs in the conductivity band of titanium dioxide, where radicals of superoxide anions are formed, which can then be transformed into active oxygen species. Based on the scientific knowledge, Stafford et al. [
69] found that the critical parameter influencing the photooxidative of 4-chlorophenol was the presence of the hydroxyl radical (*OH), which is described as the primary oxidant in the impurity mentioned above. Furthermore, the scientists indicated that holes (h
+) were responsible for the degradation process to a lesser extent [
70]. On the other hand, Li et al. [
71] showed that the major degradation road for 4-chlorophenol is the hydroquinone pathway. Hydroquinone is mainly oxidized to 1,2,4-benzenetriol, and most of the ring cleavage comes from this compound. However, a modest amount of tetraol and two additional acyclic six-carbon compounds can also be observed. The authors also indicated that the presented photo-oxidation pathway is suchlike to other aromatic compounds, including quinoline, naphthalene, and pyridine. On the other hand, after opening the ring, further decomposition into shorter organic compounds occurs, and consequently their mineralization. Finally, the main reactions in the photocatalytic removal of 4-chlorophenol are proposed as follows hydroxylation, dihydroxylation, hydrations, and decarboxylation.
The proposed mechanism of degradation of the model dye solution in the presence of the TiO
2-ZnO oxide system is shown in
Figure 14.
Phenol and its derivatives, such as chloro- and nitrophenols or bis-phenols, are recognized by the World Health Organization (WHO) as significant impurities posing a threat to aquatic organisms [
72]. For this reason, many research teams are working on the degradation of the pollutants mentioned above. An overview of the available scientific knowledge on the photocatalytic degradation of 4-chlorophenol is summarized in
Table 5.
According to the presented review of the available scientific literature on the photodegradation of 4-chlorophenol, various photoactive materials are used, including oxides such as TiO
2, ZrO
2, ZnO, and SnO
2. The results of photodegradation efficiency obtained by us were better or similar to those presented in the literature. However, the main difference is the UV light source used. According to the presented data, scientists used high-power mercury or xenon lamps (125–500 W) in most of the mentioned works. Apart from xenon and mercury lamps’ high operating costs, they are also characterized by having a short lifetime of 10,000 h and 500–1500 h, respectively [
78]. On the other hand, for the LEDs, the lifetime can reach 100,000 h [
79], where the minimum lighting time is 30,000 h [
18]. An additional problem with the use of high-power mercury or xenon lamps is the high energy loss in the form of heat. At the same time, in LED lamps, some electricity is converted into heat, but much less, and the cooling system is limited to a passive heat sink. Finally, LEDs are primarily widespread technology what makes them cheaper and less energy-consuming than other solutions such as Hg or Xe lamps [
78], and an additional advantage of LEDs is the possibility of choosing an LED well-matched to the spectrum of the target photocatalyst. For this reason, obtaining high photodegradation efficiency with the use of a low-power UV-LED lamp shows that in the case of a properly selected photocatalyst (in our case, TiO
2-ZnO systems), LEDs can be an alternative to conventional UV light sources, and most importantly, with due to their advantages such as long life time, low energy consumption as well as high light efficiency, they are environmentally friendly.
Based on the obtained results, it was confirmed that using a UV-LED lamp and the fast and efficient hydrothermal synthesis of TiO2-ZnO systems. It is possible to obtain particles that exhibited enhanced photocatalytic performance. Thus, these promising TiO2-ZnO photocatalysts may also be applied in the removal of other organic impurities.
3.2.7. Photovoltaic Properties
The most active ZnO containing photocatalysts, i.e., TiO
2-(2.5%)ZnO, TiO
2-(5%)ZnO, and TiO
2-(10%)ZnO, and reference TiO
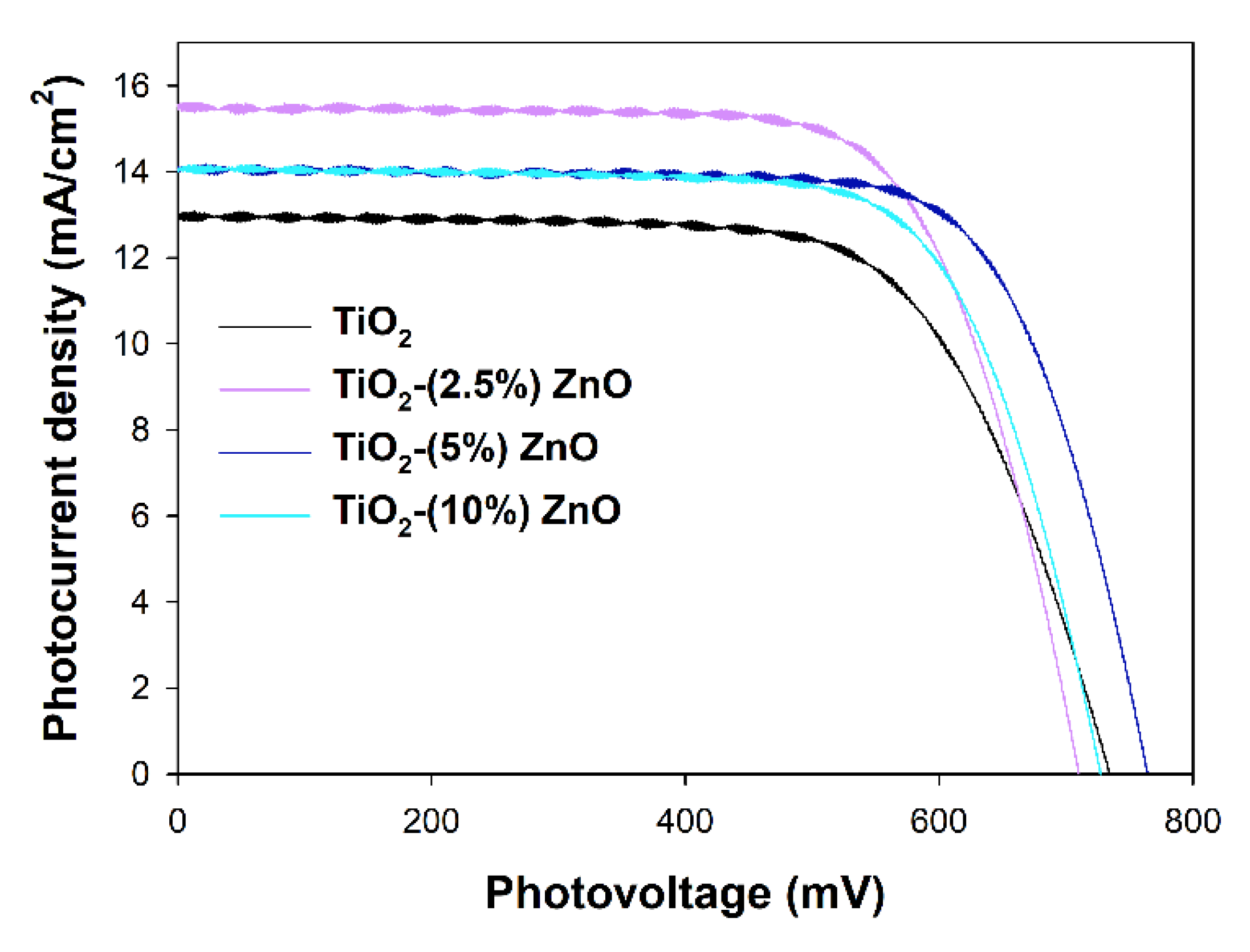
2 material have been selected to investigate their properties as the semiconducting layer of the working electrodes in DSSCs. Assembled DSSCs have been characterized with
I–V curves presented in
Figure 15, and the parameters of the device red from the
I–V curves have been collected in
Table 6. The overall photon-to-current efficiencies (
η) of all DSSCs utilizing ZnO-containing materials have been improved compared to TiO
2 cells. The
η increase is the direct result of the increase in two other parameters, i.e., fill factors (
FF), which is the energy loss related to inherent resistance in the photovoltaic cells [
80], and photocurrent densities (
JSC). The growth in
FF values may suggest that the electron transport abilities of the ZnO-containing materials have been increased and/or the recombination rate has been suppressed. Both effects mentioned above also affect the
JSC values, but additionally,
JSC depends on the number of dye molecules adsorbed on the working electrode surface. The amounts of the adsorbed dye molecules on the investigated working electrodes have been determined, and the results have been collected in
Table 7 (see
Ndye value). Surprisingly, the higher the ZnO content in the semiconducting material, the lower the adsorbed dye amount. The decrease in the
Ndye amount simultaneously with the improvement of the
JSC registered for ZnO-containing cells suggests that the recombination rate and electron transport abilities effects have prime importance in the efficiencies of investigated cells.
The third value that influences
η is the open circuit photovoltage (
VOC), mainly depending on the energy level difference between the Fermi level of the semiconductor and the present in the electrolyte solution redox mediator’s Nernst potential [
81]. The
VOC values are pretty random in the presented devices, and no visible tendency may be observed. As mentioned above, the band gaps of investigated materials selected for the photovoltaic experiments do not differ, and all are equal to 3.2 eV. As in all investigated DSSCs, the same electrolyte solution has been used, and nothing suggests the shift of the Fermi levels of the utilized semiconductors. The observed situation makes some difficulties in interpreting the obtained results and leads to a significant decrease of the
η value of TiO
2-(2.5%)ZnO cell even if it is characterized with still high
FF and the highest
JSC values.
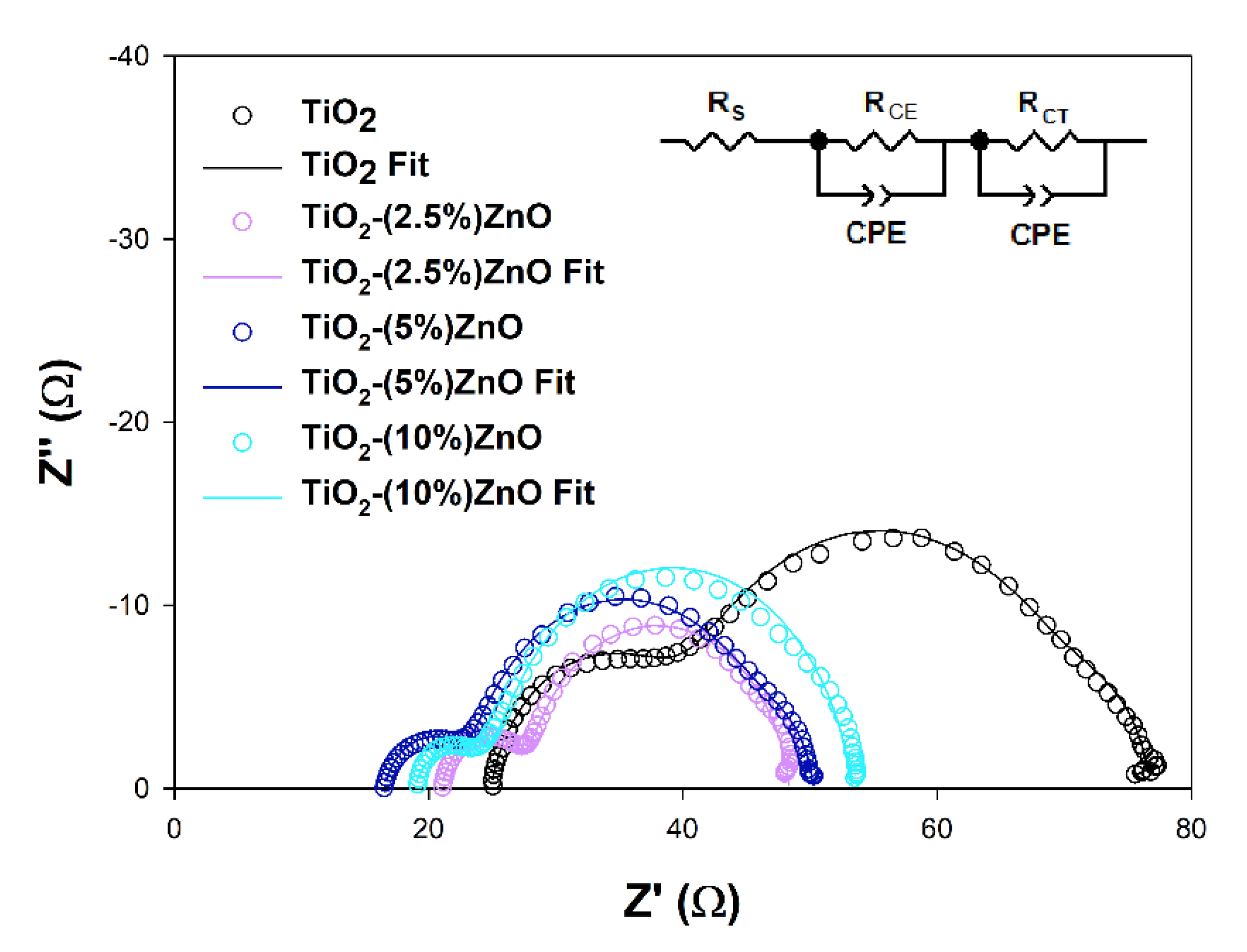
Electrochemical impedance spectroscopy (EIS) is a valuable tool to explain deeper the reasons for some DSSCs’ behaviors. The EIS results obtained for investigated cells are presented in
Figure 16 and summarized in
Table 7.
The
RS and
RCE values, which are the series resistance, mainly depend on the parameters of the measuring system used and the resistance of the counter electrode used to build the cells, respectively. As it can be seen, they do not differ significantly between cells tested, do not depend on the semiconductor materials used, and do not considerably impact differences in the efficiency of the devices discussed [
80]. The effect may cause the increase in the
RCE parameter observed for the TiO
2 cells resulted from the increase of the
RCT parameter in these cells or the minor differences in the Pt film thickness or discontinuities in the Pt layer [
82,
83]. More interesting for this study is the
RCT value, which may be understood as the semiconductor/dye/electrolyte interface resistance. In general, the higher the registered
RCT value, the lower the
JSC of the cell, except for the 5 and 10% ZnO cells pair, where the
JSC values are equal to each other, and the
RCT values are different. The above observation suggests that additional effect has to be involved in the photovoltaic process of investigated cells. The electron lifetimes (
τ) have been estimated from the highest frequency point (
f) of the
RCT semicircle using equation
τ = (2
πf)
−1 to get a deeper insight into electron processes occurring in the cells presented. It is well known that the electron lifetime depends on the semiconducting material’s trapping abilities, but intense trapping disturbing the electron transport and may cause a decrease in cell efficiency [
81,
84]. The fact that the electron trapping process is too effective may be why the higher
RCT value has been observed in TiO
2-(10%)ZnO cells. The observed
FF values may also support the above interpretation. Some authors state that the electron lifetime may also affect the
VOC value of the DSSC by the relaxation of the injected electrons trapped into the semiconductor [
85]. Based on the above literature findings, one may say that the slightly higher
τ value estimated for TiO
2-(2.5%)ZnO cells may lead to
VOC loss. However, the intense electron trapping in the TiO
2-(10%)ZnO material may suppress the relaxation and support relatively high VOC keeping.