A Method for Evaluation the Fatigue Microcrack Propagation in Human Cortical Bone Using Differential X-ray Computed Tomography
Abstract
:1. Introduction
2. Material and Methods
2.1. Specimens
2.2. Mechanical Loading
2.3. Radiographical Imaging and Computed Tomography
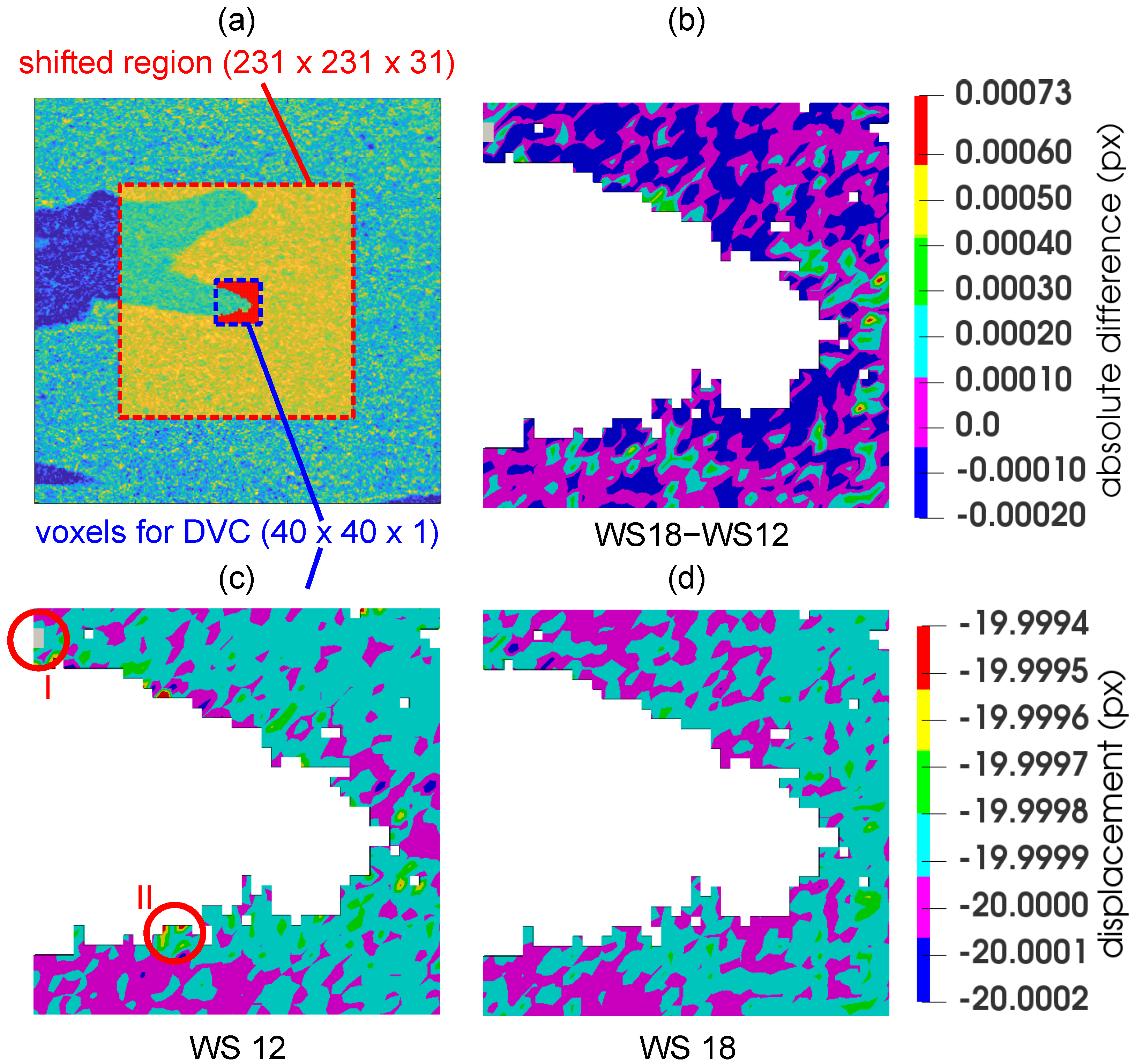
2.4. Digital Volume Correlation
2.4.1. Analysis of Optimal Correlation Window Size
3. Results
4. Discussion
- It was possible to identify fatigue microcracks and their propagation in the entire volume of the investigated sample using the laboratory X-ray scanner and custom post-processing procedures. The ability to identify the system of microcracks is essential to understand the bone failure mechanisms on the microscopic level, particularly regarding the fact that the microscopic damage of the bone and the rate of its accumulation plays a significant role in the reduction of the bone bearing capacity in the post-yield regime. The fully volumetric analysis, in this case, also brings advantages over an investigation based on 2D slices, both in terms of the microcrack identification and quantification, which is given by their complex arbitrary shape in the microstructure of the bone.
- In all the cases, it was found out, at the achieved voxel size, that the existing or newly formed microcracks either originate from the stress concentrators in the microstructure of the bone or interconnect them. In the investigated samples, the stress concentrators can be divided into three groups comprising the microcracks present in the intact state, the cracks on the surface of the specimen created during the manufacturing of the sample, and the Haversian canals. Although this is an expected result conforming to the fundamentals of fracture mechanics, it is an important finding that the regions of stress concentration with the presence of a system of microcracks were clearly apparent in the displacement field of the full-affine DVC procedure. With respect to the 2 micron resolution achieved in the reconstructed 3D images of the bone, the statistics from the two perpendicular planes, where the full-affine DVC was evaluated, shows approximately a 50% probability that a microcrack is present at the strain concentration location. This is still, however, a valuable result since the automated identification of the microcracks can be limited only to the regions of interest determined from the DVC with implications on computational costs and a time reduction. Conversely, no microcracks in the regions of the uniform strain distribution were identified at this scale level limited by the micrometric voxel size.
- It is possible to perform the in situ fatigue loading of a cylindrical human cortical bone sample using a laboratory CT scanner at a resolution given by the geometry of the loading device influencing the achievable source-to-object distance, and thus the achievable geometrical magnification of the projections. This enabled us to identify the microcracks in both the intact sample and after the loading procedure, with the width of at least 2 voxels, i.e., . Due to the nature of the CT imaging, regarding the conclusions, the probable presence of microcracks with dimensions under the resolution limit of the imaging instrumentation is questionable. However, this factor does not necessarily decrease the impact of this study as the aim was to show the methodology based upon a laboratory CT scanner, where the DVC using full-affine transformation serves as a tool for the identification of the damage accumulation in the bone represented by a system of microcracks.
- However, it is necessary to take appropriate measures with laboratory CT scanners to achieve the sufficient quality of the resulting reconstructed 3D images. Thus, the achieved value of the geometrical magnification of the projections is only one of the parameters determining the overall quality of the radiographical imaging. To reduce the influence of various tomographical artifacts including ring artifacts and the beam hardening effect, appropriate corrections of the imaging detector and X-ray source characteristics have to be performed to sharpen the reconstructed 3D image and enable the reliable identification of the microscopic features in the bone. It can be seen in the projections that the beam hardening effect consists of a higher intensity in the vicinity of the surface as no particular treatment was applied to its reduction. However, since the evaluation near the surface of the sample was omitted due to the damage induced by the sample preparation, the beam hardening itself has only a negligible influence on the acquired results. Furthermore, we have shown in the FWHM comparison with the SEM imaging that a combination of projection-level corrections with the focal spot drift correction leads to a quality of the reconstructed 3D images comparable to the SE microscopy in terms of the crack thickness calculation and void-material interface identification.
- Since the aim of the study was to show the methodology combining in situ fatigue loading using a simulated gait cycle with a DVC based evaluation and differential tomography for the identification of the microcracks present in the microstructure of the bone, only one sample was subjected to the full evaluation procedure comprised of the discussed methods. Nevertheless, in total, six samples were tested using the in situ loading procedure, but the acquired results were unsatisfactory due to several reasons. During two tests, problems were encountered with the mechanical response of the sample resulting in its sudden disintegration during the loading procedure, presumably due to the thermal and mechanical damage inflicted during the sample preparation. Generally, due to the variability in the mechanical response of the samples, it was difficult to determine the number of required cycles to generate microcracks, but prevent the destruction of the sample. Additionally, the behavior of the microstructure may also result in the closing of the microcracks, which corresponds to the behavior of the bone in the human body, but such a process precludes a study of the crack formation using time-lapse radiographical imaging. Additionally, the long-term stability of the detector used for imaging has an influence on the noise in the reconstructed 3D images and their sharpness, where the achieved geometrical resolution with a reduced reconstruction quality may also prevent the evaluation of the experiment.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| API | Application Programming Interface |
| CMOS | Complementary Metal Oxide Semiconductor |
| CT | Computed Tomography |
| DIC | Digital Image Correlation |
| DVC | Digital Volume Correlation |
| FBP | Filtered Back-Projection |
| FDK | Feldkamp, Davis, and Kress (CT reconstruction algorithm) |
| GOS | Gadolinium oxysulfide |
| FWHM | Full-Width Half-Maximum |
| MBE | Mean-Bias Error |
| NCC | Normalized Cross-Correlation |
| RMSE | Root-Mean Square Error |
| SEM | Scanning Electron Microscopy |
| VOI | Volume Of Interest |
| XCT | X-ray (micro) Computed Tomography |
References
- Hart, N.; Newton, R.; Tan, J.; Rantalainen, T.; Chivers, P.; Siafarikas, A.; Nimphius, S. Biological basis of bone strength: Anatomy, physiology and measurement. J. Musculoskelet. Neuronal Interact. 2020, 20, 347–371. [Google Scholar] [PubMed]
- O’Neill, M.; Taylor, D. Repair of microdamage caused by cyclic loading in insect cuticle. J. Exp. Zool. Part Ecol. Integr. Physiol. 2020, 333, 20–28. [Google Scholar] [CrossRef]
- Osterhoff, G.; Morgan, E.; Shefelbine, S.; Karim, L.; McNamara, L.; Augat, P. Bone mechanical properties and changes with osteoporosis. Injury 2016, 47, S11–S20. [Google Scholar] [CrossRef] [Green Version]
- Seref-Ferlengez, Z.; Kennedy, O.D.; Schaffler, M.B. Bone microdamage, remodeling and bone fragility: How much damage is too much damage? Bonekey Rep. 2015, 664. [Google Scholar] [CrossRef] [Green Version]
- Zysset, P.; Wolfram, U. A rate-independent continuum model for bone tissue with interaction of compressive and tensile micro-damage. J. Mech. Behav. Biomed. Mater. 2017, 74, 448–462. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, F.; Taylor, D.; Dickson, G.; Lee, T. Visualisation of three-dimensional microcracks in compact bone. J. Anat. 2000, 197, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Brock, G.; Kim, G.; Ingraffea, A.; Andrews, J.; Pianetta, P.; van der Meulen, M. Nanoscale Examination of Microdamage in Sheep Cortical Bone Using Synchrotron Radiation Transmission X-Ray Microscopy. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Voide, R.; Schneider, P.; Stauber, M.; Wyss, P.; Stampanoni, M.; Sennhauser, U.; van Lenthe, G.; Müller, R. Time-lapsed assessment of microcrack initiation and propagation in murine cortical bone at submicrometer resolution. Bone 2009, 45, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Pena Fernandez, M.; Kao, A.; Witte, F.; Arora, H.; Tozzi, G. Low-cycle full-field residual strains in cortical bone and their influence on tissue fracture evaluated via in situ stepwise and continuous X-ray computed tomography. J. Biomech. 2020, 113, 110105. [Google Scholar] [CrossRef] [PubMed]
- Kumpova, I.; Vavrik, D.; Fila, T.; Koudelka, P.; Jandejsek, I.; Jakubek, J.; Kytyr, D.; Zlamal, P.; Vopalensky, M.; Gantar, A. High resolution micro-CT of low attenuating organic materials using large area photon-counting detector. J. Instrum. 2016, 11. [Google Scholar] [CrossRef]
- Dudak, J. High-resolution X-ray imaging applications of hybrid-pixel photon counting detectors Timepix. Radiat. Meas. 2020, 137. [Google Scholar] [CrossRef]
- Akhter, M.; Recker, R. High resolution imaging in bone tissue research-review. Bone 2021, 143. [Google Scholar] [CrossRef] [PubMed]
- Khosravani, M.; Reinicke, T. On the Use of X-ray Computed Tomography in Assessment of 3D-Printed Components. J. Nondestruct. Eval. 2020, 39. [Google Scholar] [CrossRef]
- Senck, S.; Glinz, J.; Happl, M.; Scheerer, M.; Reiter, T.; Kastner, J. Quantification of surface-near porosity in additively manufactured aluminum brackets using X-ray microcomputed tomography. In Proceedings of the AIAA Scitech 2021 Forum, Online, 11–15 January 2021; pp. 1–7. [Google Scholar] [CrossRef]
- Qiu, Q.; Zhu, J.; Dai, J.G. In-situ X-ray microcomputed tomography monitoring of steel corrosion in engineered cementitious composite (ECC). Constr. Build. Mater. 2020, 262. [Google Scholar] [CrossRef]
- Vavrik, D.; Benes, P.; Fila, T.; Koudelka, P.; Kumpova, I.; Kytyr, D.; Vopalensky, M.; Vavro, M.; Vavro, L. Local fracture toughness testing of sandstone based on X-ray tomographic reconstruction. Int. J. Rock Mech. Min. Sci. 2021, 138. [Google Scholar] [CrossRef]
- Xu, F. Quantitative characterization of deformation and damage process by digital volume correlation: A review. Theor. Appl. Mech. Lett. 2018, 8, 83–96. [Google Scholar] [CrossRef]
- Ogierman, W.; Kokot, G. Determination of local strain distribution at the level of the constituents of particle reinforced composite: An experimental and numerical study. Materials 2020, 13, 3889. [Google Scholar] [CrossRef] [PubMed]
- Koudelka, P.; Fila, T.; Rada, V.; Zlamal, P.; Sleichrt, J.; Vopalensky, M.; Kumpova, I.; Benes, P.; Vavrik, D.; Vavro, L.; et al. In-situ X-ray differential micro-tomography for investigation of water-weakening in quasi-brittle materials subjected to four-point bending. Materials 2020, 13, 1405. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stefan, U.; Michael, B.; Werner, S. Effects of three different preservation methods on the mechanical properties of human and bovine cortical bone. Bone 2010, 47, 1048–1053. [Google Scholar] [CrossRef]
- Zhang, G.J.; Yang, J.; Guan, F.J.; Chen, D.; Li, N.; Cao, L.; Mao, H. Quantifying the Effects of Formalin Fixation on the Mechanical Properties of Cortical Bone Using Beam Theory and Optimization Methodology with Specimen-Specific Finite Element Models. J. Biomech. Eng. 2016, 138. [Google Scholar] [CrossRef] [PubMed]
- De Biase, P.; Biancalani, E.; Martinelli, D.; Cambiganu, A.; Bianco, S.; Buzzi, R. Subtrochanteric fractures: Two case reports of non-union treatment. Injury 2018, 49, S9–S15. [Google Scholar] [CrossRef]
- Singh, R.; Gupta, V.; Pandey, P.; Mridha, A. Effect of Drilling Techniques on Microcracks and Pull-Out Strength of Cortical Screw Fixed in Human Tibia: An In-Vitro Study. Ann. Biomed. Eng. 2021, 49, 382–393. [Google Scholar] [CrossRef] [PubMed]
- Fila, T.; Sleichrt, J.; Kytyr, D.; Kumpova, I.; Vopalensky, M.; Zlamal, P.; Rada, V.; Vavrik, D.; Koudelka, P.; Senck, S. Deformation analysis of the spongious sample in simulated physiological conditions based on in-situ compression, 4D computed tomography and fast readout detector. J. Instrum. 2018, 13. [Google Scholar] [CrossRef]
- Fila, T.; Koudelka, P.; Kumpova, I.; Vopalensky, M.; Sleichrt, J.; Rada, V.; Zlamal, P.; Tarasiuk, J.; Kytyr, D. Time-lapse micro-CT analysis of fatigue microcrack propagation in cortical bone. J. Instrum. 2020, 15. [Google Scholar] [CrossRef]
- Vavrik, D.; Kytyr, D.; Muhleder, S.; Vopalensky, M.; Benes, P.; Pichotka, M.; Kumpova, I.; Koudelka, P. High resolution X-ray micro-CT imaging of fibrin scaffold using large area single photon counting detector. J. Instrum. 2018, 13. [Google Scholar] [CrossRef]
- Bergmann, G.; Deuretzbacher, G.; Heller, M.; Graichen, F.; Rohlmann, A.; Strauss, J.; Duda, G. Hip contact forces and gait patterns from routine activities. J. Biomech. 2001, 34, 859–871. [Google Scholar] [CrossRef]
- Chun, B.; Jang, I. Determination of the representative static loads for cyclically repeated dynamic loads: A case study of bone remodeling simulation with gait loads. Comput. Methods Programs Biomed. 2021, 200. [Google Scholar] [CrossRef] [PubMed]
- X-RAY WorX. XWT-225-TC Microfocus X-ray Tube. Available online: https://www.x-ray-worx.com/index.php/en/microfocus-x-ray-tubes-overview/microfocus-transmission-tubes/product-line-tc (accessed on 1 March 2021).
- Varex. Dexela 1512 CMOS Flat Panel Detector. Available online: https://www.vareximaging.com/products/flat-panel-detectors/1512 (accessed on 1 March 2021).
- Vopalensky, V.; Vavrik, D.; Kumpova, I. Optimization of acquisition parameters in radiography and tomography. In Proceedings of the 7th Conference on Industrial Computed Tomography, Leuven, Belgium, 7–9 February 2017; Volume 3. [Google Scholar]
- Feldkamp, L.; Davis, L.; Kress, J. Practical cone-beam algorithm. J. Opt. Soc. Am. Opt. Image Sci. Vis. 1984, 1, 612–619. [Google Scholar] [CrossRef] [Green Version]
- Hoppe, S.; Hornegger, J.; Dennerlein, F.; Lauritsch, G.; Noo, F. Accurate image reconstruction using real C-arm data from a Circle-plus-arc trajectory. Int. J. Comput. Assist. Radiol. Surg. 2012, 7, 73–86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schofield, R.; King, L.; Tayal, U.; Castellano, I.; Stirrup, J.; Pontana, F.; Earls, J.; Nicol, E. Image reconstruction: Part 1—understanding filtered back projection, noise and image acquisition. J. Cardiovasc. Comput. Tomogr. 2020, 14, 219–225. [Google Scholar] [CrossRef]
- Cormack, A. Reconstruction of densities from their projections, with applications in radiological physics. Phys. Med. Biol. 1973, 18, 195–207. [Google Scholar] [CrossRef]
- Tescan. MIRA Scanning Electron Microscope. Available online: www.tescan.com/product/sem-for-materials-science-tescan-mira (accessed on 1 March 2021).
- Bay, B.; Smith, T.; Fyhrie, D.; Saad, M. Digital volume correlation: Three-dimensional strain mapping using X-ray tomography. Exp. Mech. 1999, 39, 217–226. [Google Scholar] [CrossRef]
- Lucas, B.D.; Kanade, T. Iterative image registration technique with an application to stereo vision. Proc. IJCAI 1981, 2, 674–679. [Google Scholar]
- Jirousek, O.; Jandejsek, I.; Vavrik, D. Evaluation of strain field in microstructures using micro-CT and digital volume correlation. J. Instrum. 2011, 6. [Google Scholar] [CrossRef]
- Tozzi, G.; Danesi, V.; Palanca, M.; Cristofolini, L. Elastic Full-Field Strain Analysis and Microdamage Progression in the Vertebral Body from Digital Volume Correlation. Strain 2016, 52, 446–455. [Google Scholar] [CrossRef] [Green Version]
- Tozzi, G.; Pena Fernandez, M.; Davis, S.; Karali, A.; Kao, A.P.; Blunn, G. Full-Field Strain Uncertainties and Residuals at the Cartilage-Bone Interface in Unstained Tissues Using Propagation-Based Phase-Contrast XCT and Digital Volume Correlation. Materials 2020, 13, 2579. [Google Scholar] [CrossRef] [PubMed]
- Willmott, C.J. Some comments on the evaluation of model performance. Bull. Am. Meteorol. Soc. 1982, 63, 1309–1315. [Google Scholar] [CrossRef] [Green Version]












| Window Size | Elapsed Time | NaN | Mean | MBE | RMSE | |
|---|---|---|---|---|---|---|
| ine [px] | [s] | [nodes] | [px] | [px] | [-] | [-] |
| ine 6 | 2674 | 159 | 24.38 | 3.9 | 3.9 | 6.73 |
| 8 | 2521 | 83 | 27.13 | 1.15 | 1.15 | 3.34 |
| 10 | 2627 | 47 | 27.92 | 0.37 | 0.37 | 1.72 |
| 12 | 3161 | 1 | 28.25 | 0.04 | 0.04 | 0.43 |
| 14 | 6915 | 0 | 28.28 | 0.15 | ||
| 16 | 6028 | 0 | 28.28 | |||
| 18 | 3423 | 0 | 28.28 | |||
| 20 | 4117 | 0 | 28.28 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koudelka, P.; Kytyr, D.; Fila, T.; Sleichrt, J.; Rada, V.; Zlamal, P.; Benes, P.; Bendova, V.; Kumpova, I.; Vopalensky, M. A Method for Evaluation the Fatigue Microcrack Propagation in Human Cortical Bone Using Differential X-ray Computed Tomography. Materials 2021, 14, 1370. https://doi.org/10.3390/ma14061370
Koudelka P, Kytyr D, Fila T, Sleichrt J, Rada V, Zlamal P, Benes P, Bendova V, Kumpova I, Vopalensky M. A Method for Evaluation the Fatigue Microcrack Propagation in Human Cortical Bone Using Differential X-ray Computed Tomography. Materials. 2021; 14(6):1370. https://doi.org/10.3390/ma14061370
Chicago/Turabian StyleKoudelka, Petr, Daniel Kytyr, Tomas Fila, Jan Sleichrt, Vaclav Rada, Petr Zlamal, Pavel Benes, Vendula Bendova, Ivana Kumpova, and Michal Vopalensky. 2021. "A Method for Evaluation the Fatigue Microcrack Propagation in Human Cortical Bone Using Differential X-ray Computed Tomography" Materials 14, no. 6: 1370. https://doi.org/10.3390/ma14061370








