Effect of Cr on Aqueous and Atmospheric Corrosion of Automotive Carbon Steel
Abstract
:1. Introduction
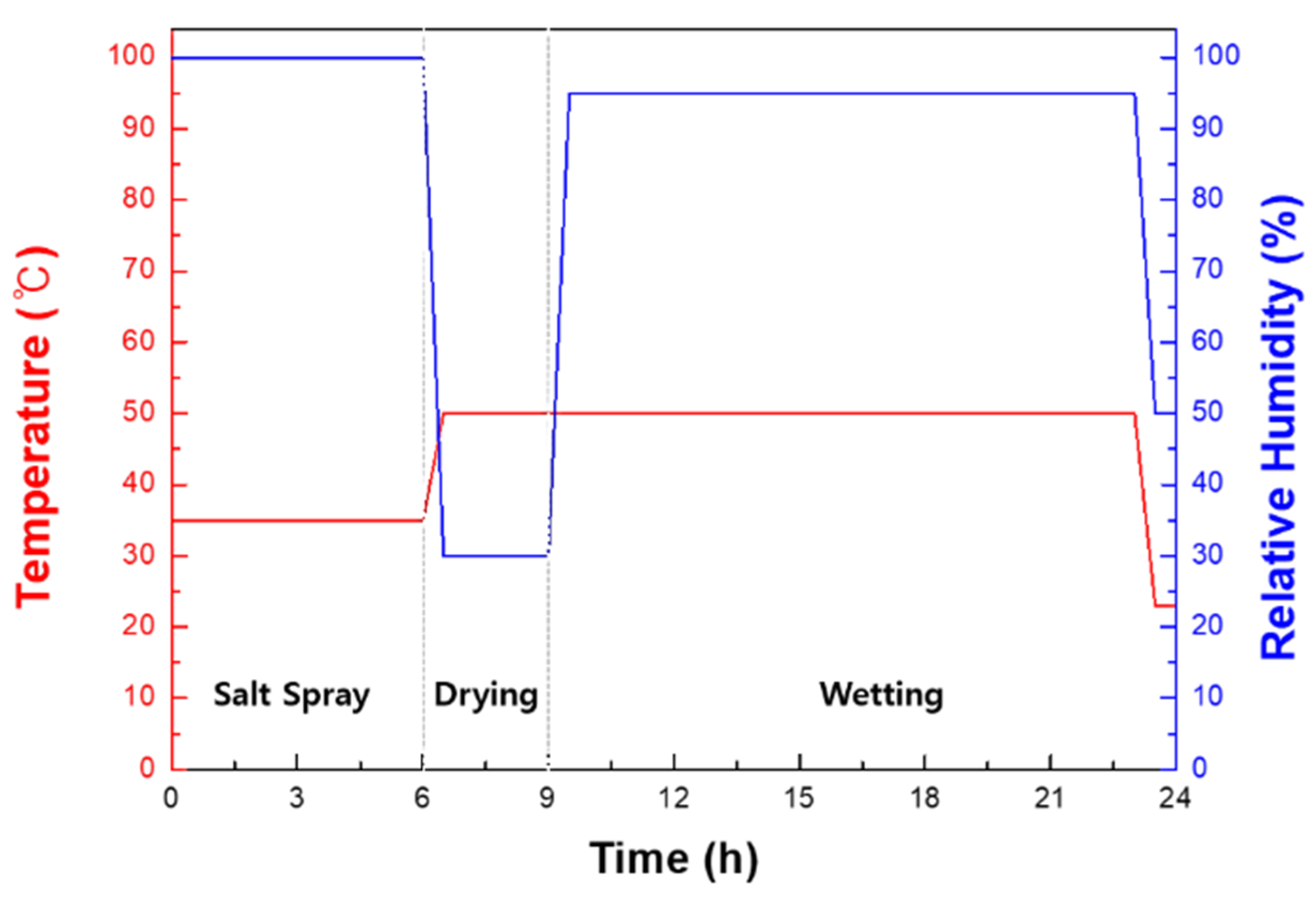
2. Materials and Methods
3. Results and Discussion
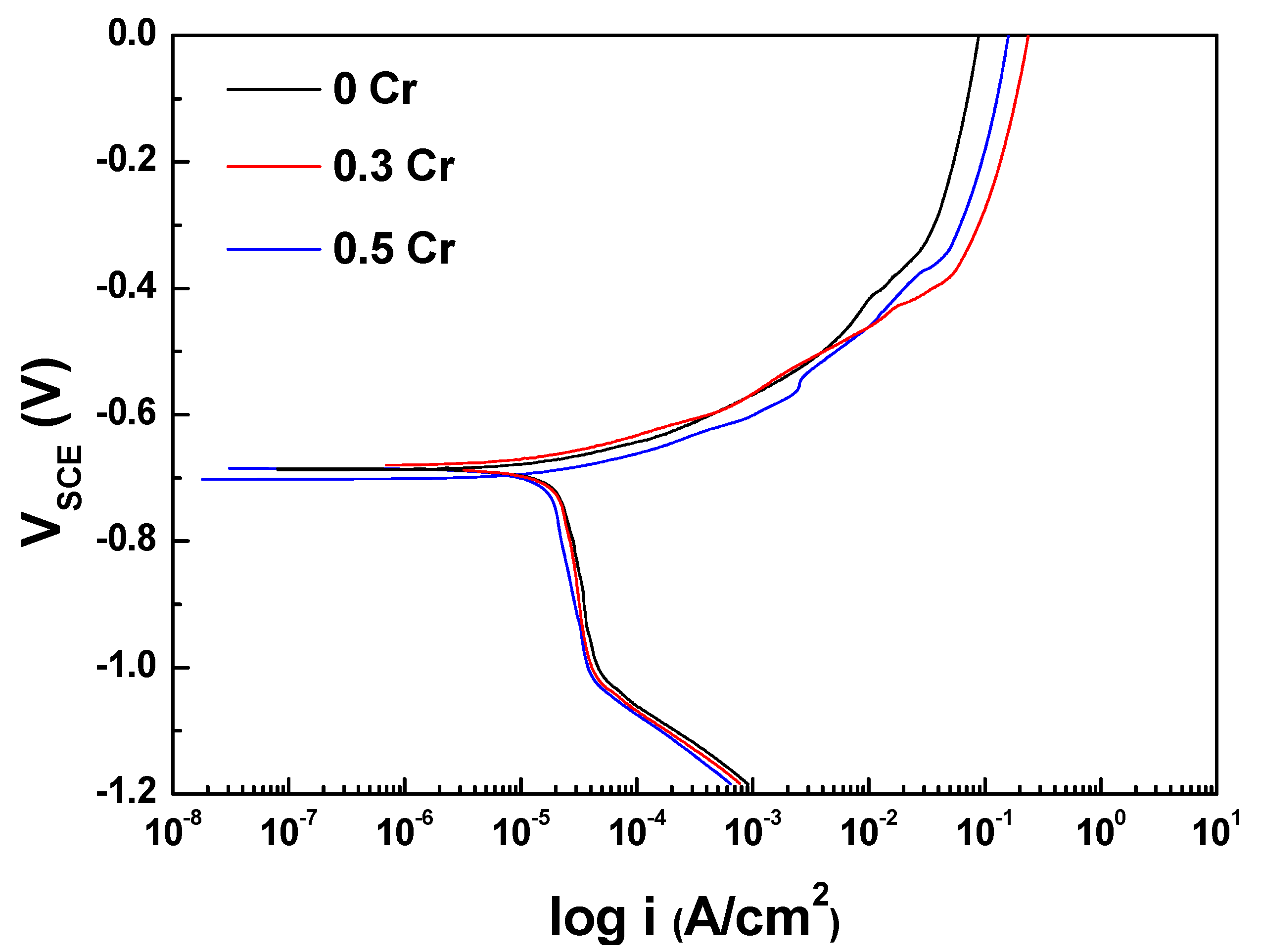
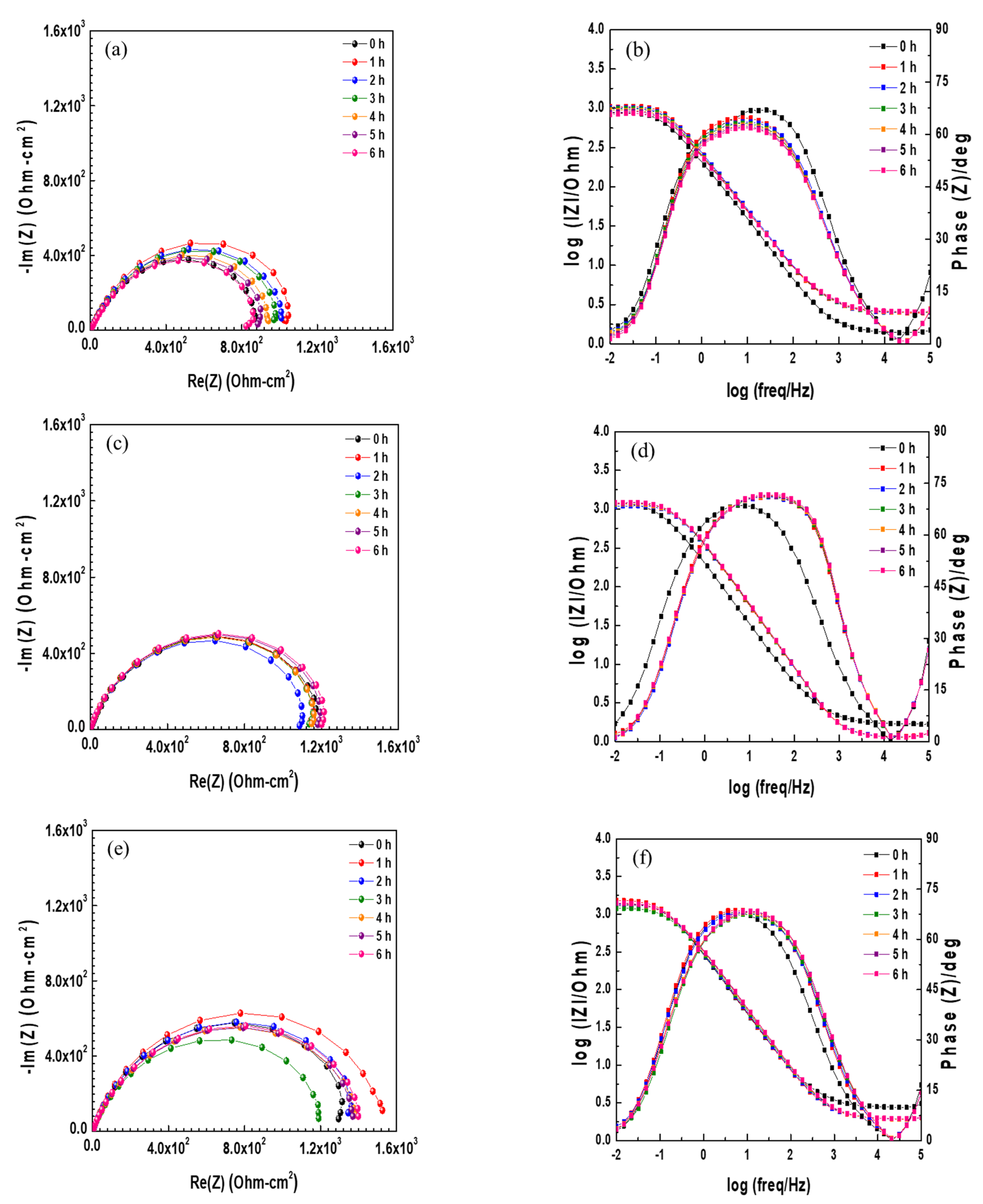
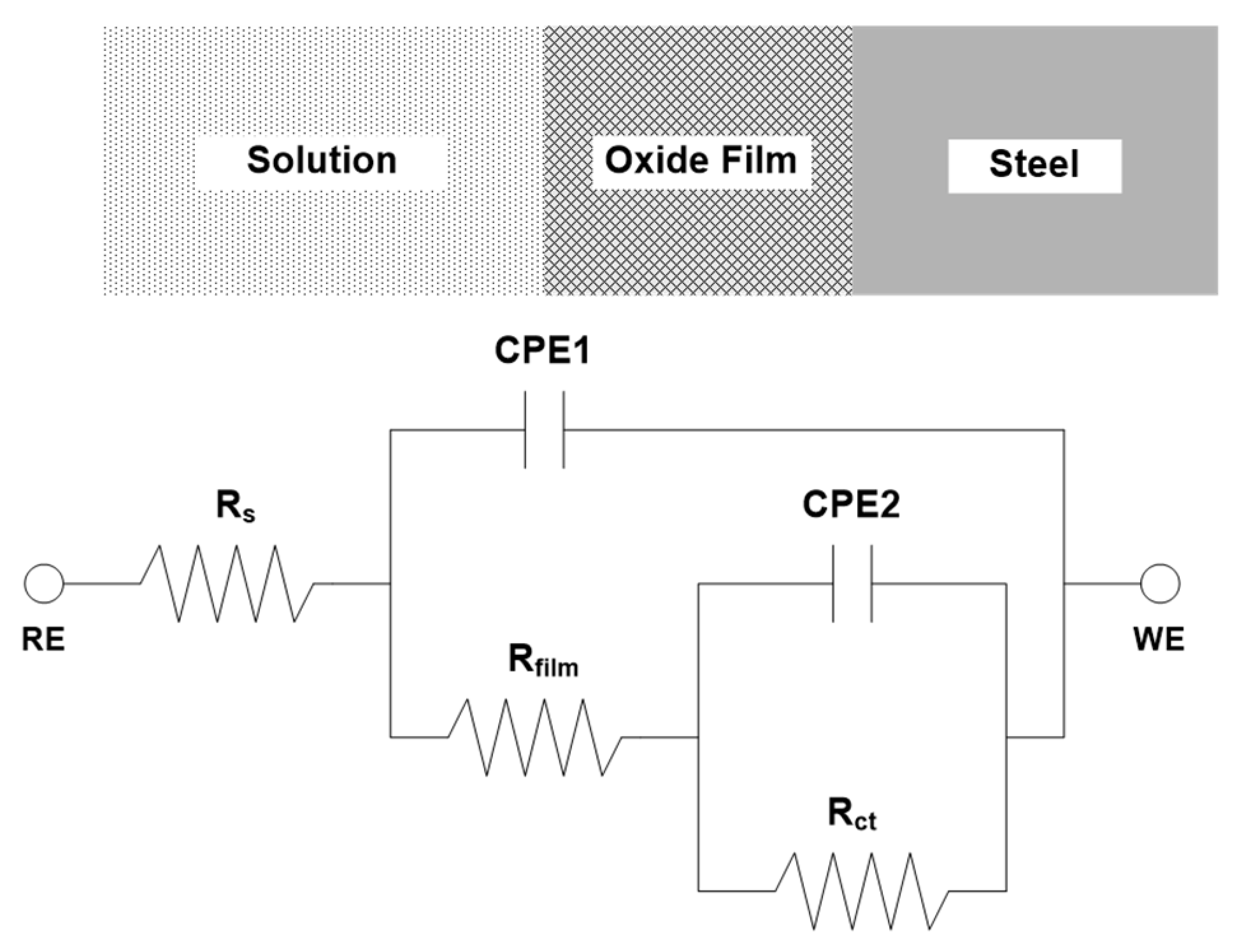
3.1. Electrochemical Measurement
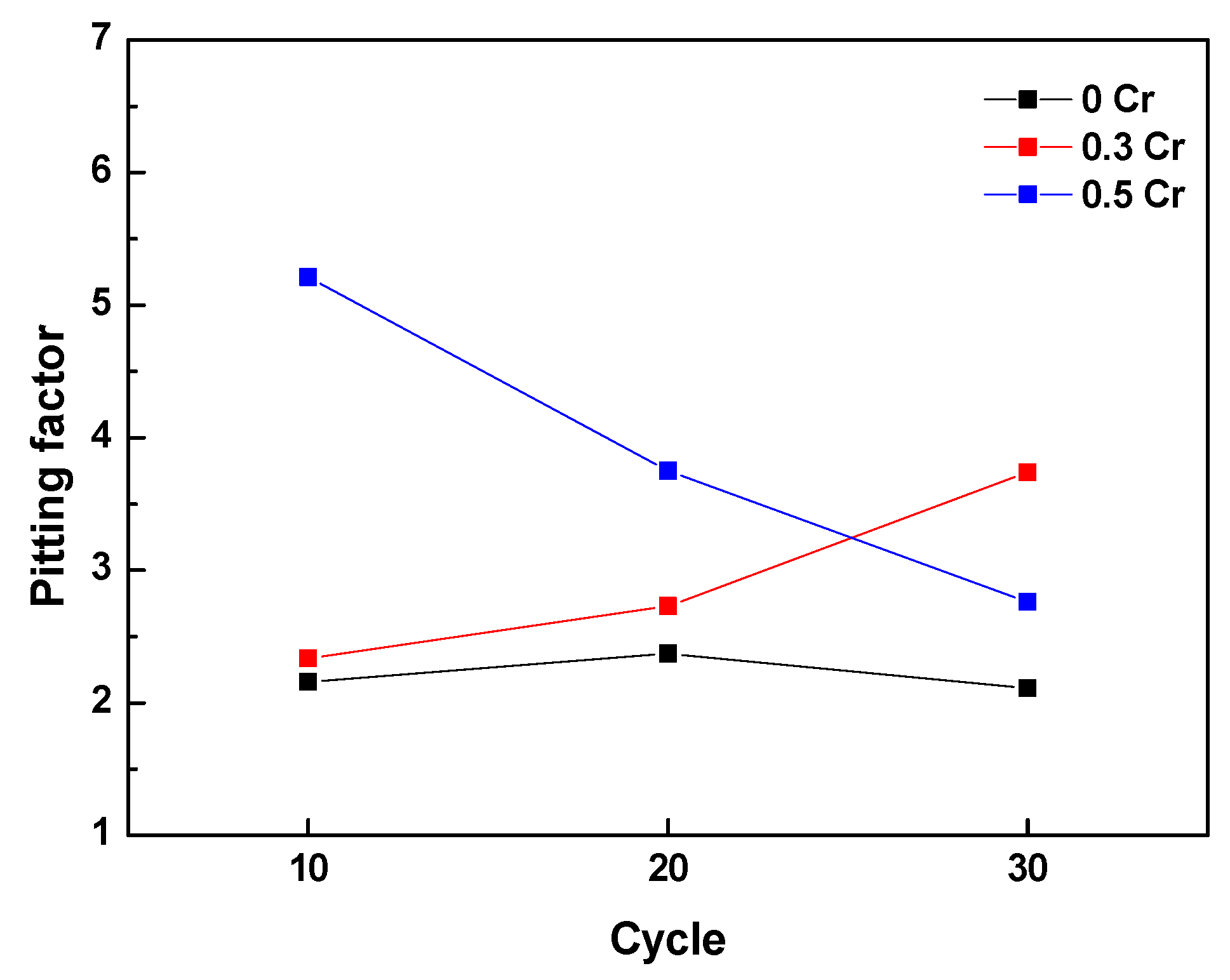
3.2. Cyclic Corrosion Test Results
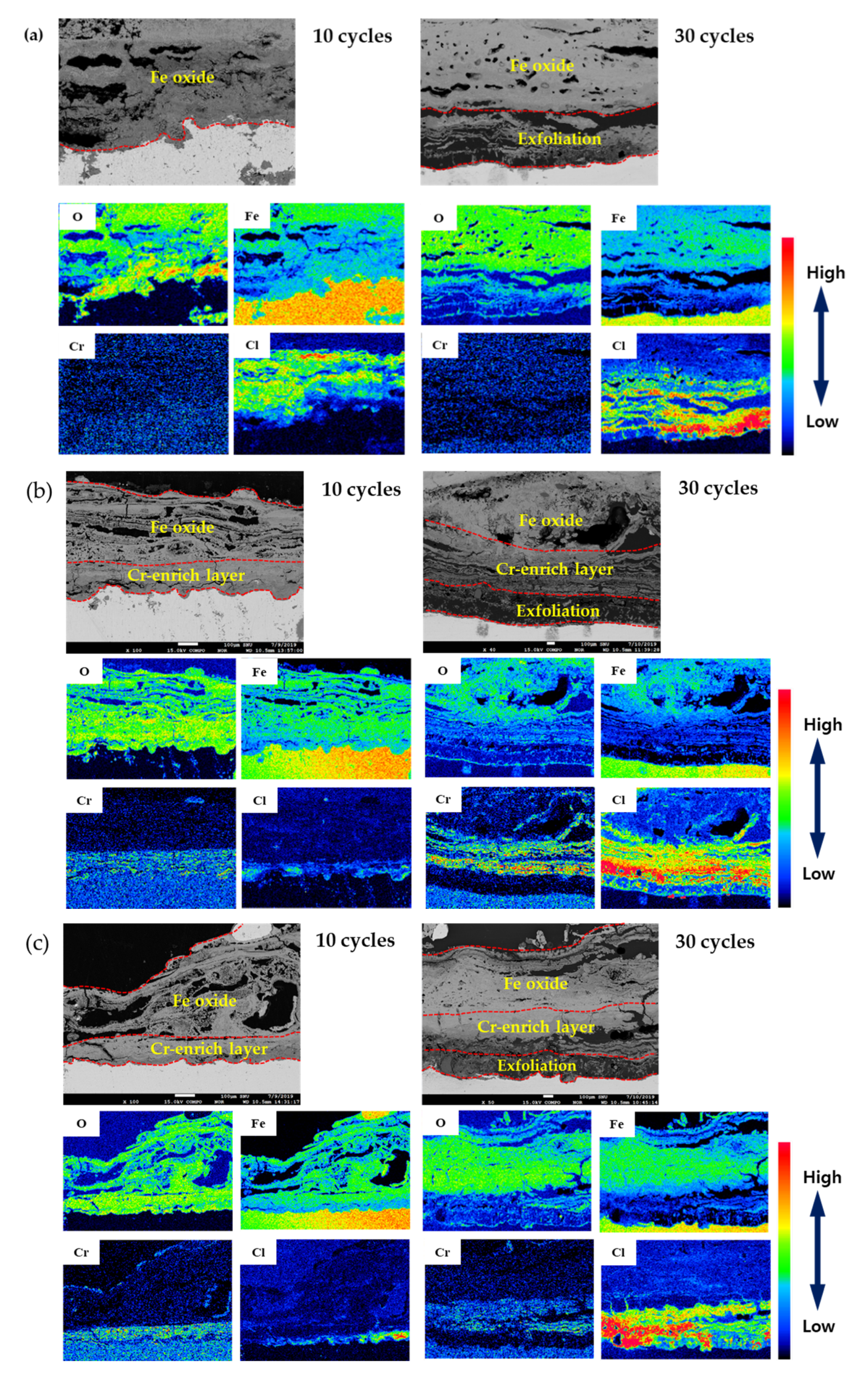
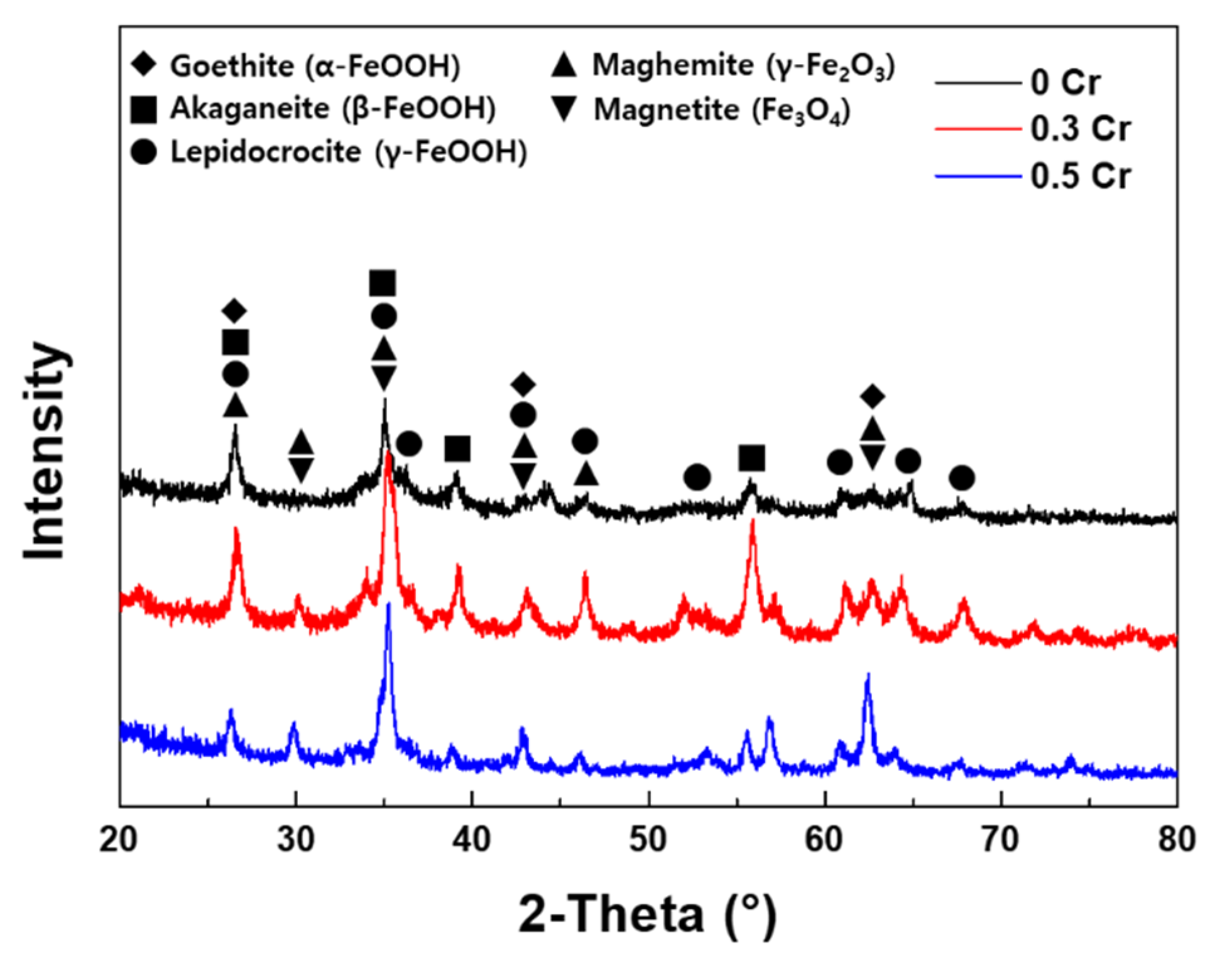
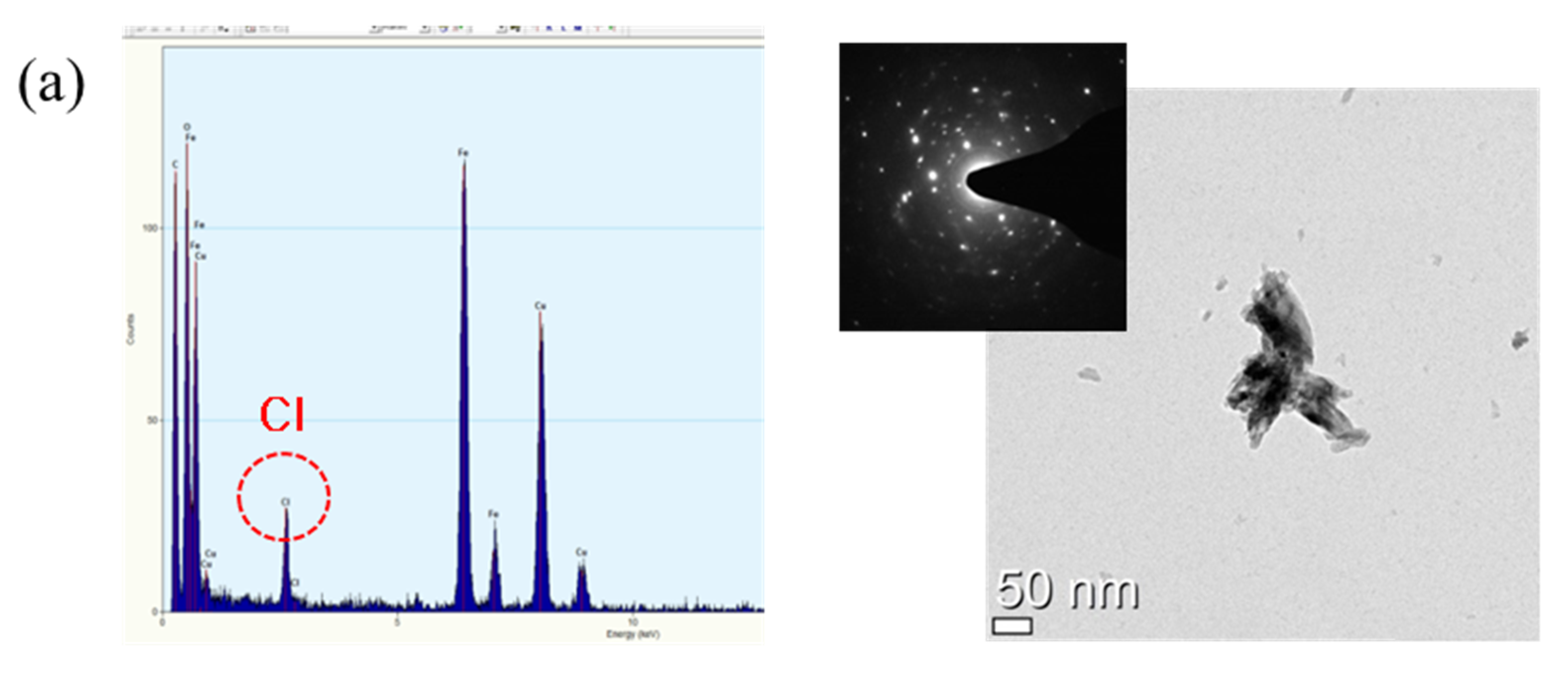
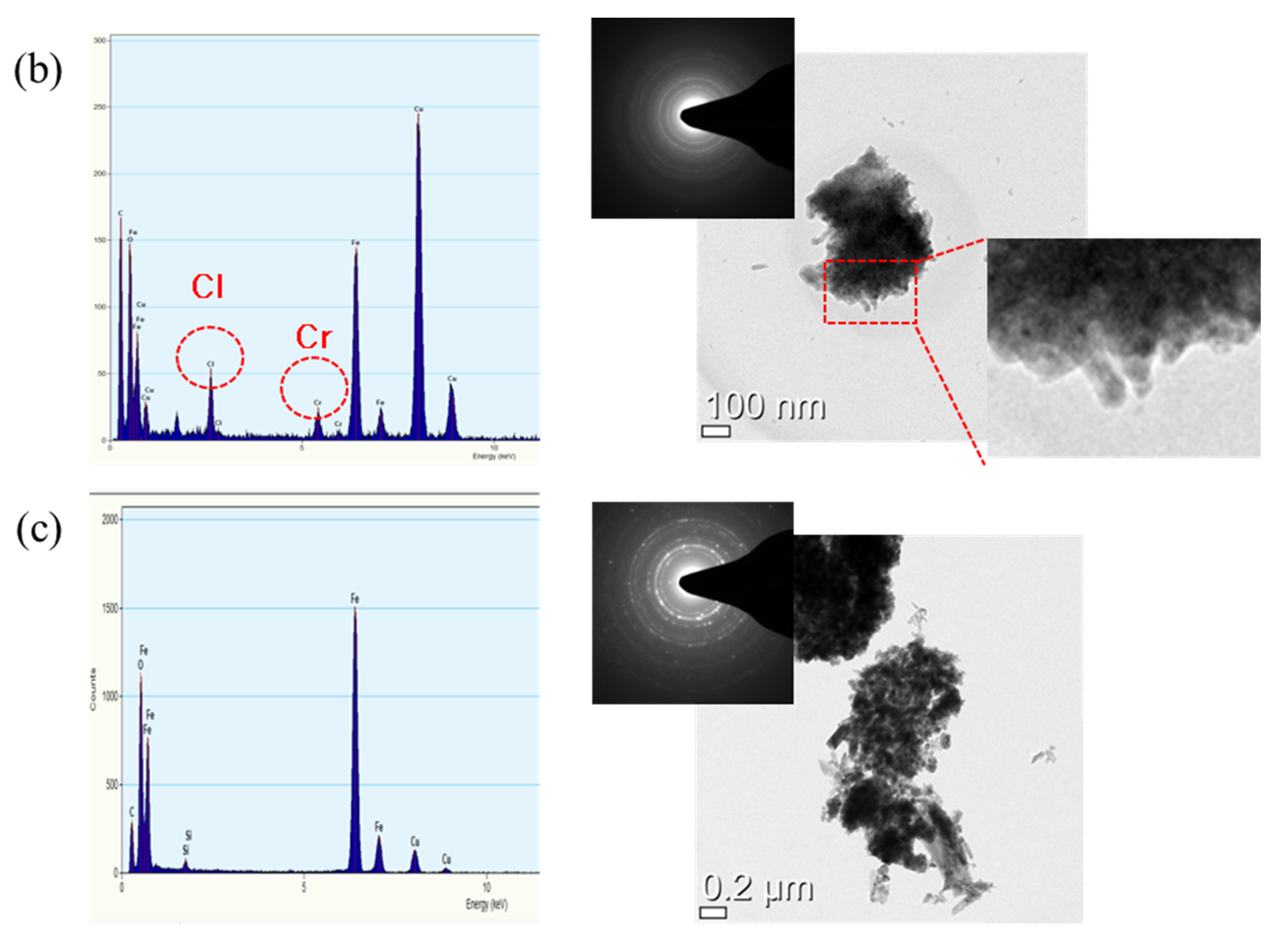
3.3. Rust Constituent Analysis
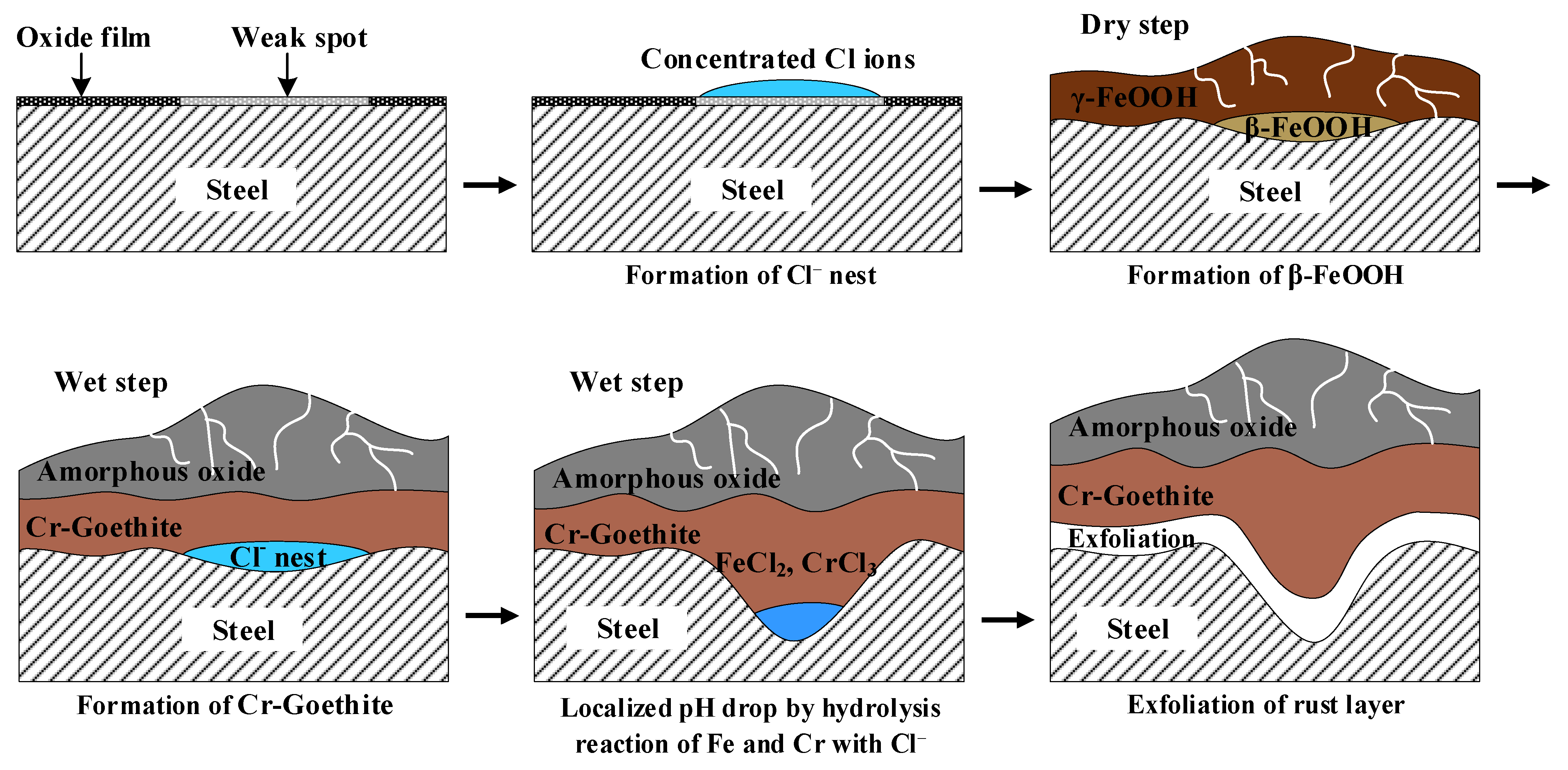
3.4. Localized Corrosion Mechanism of Cr-Added Steel under Wet/Dry Conditions
4. Conclusions
- In the electrochemical measurement results, the Cr alloying element improves the corrosion resistance of the ACS that was immersed in the Cl-containing aqueous solution.
- Cl is concentrated at the metal/rust interface in all of the specimens regardless of Cr content after the CCT. The Cl is uniformly concentrated and distributed on the 0 Cr steel, whereas Cl is localized and non-uniformly concentrated on the Cr-added steels. The PF of the Cr-added steels is higher than that of the 0 Cr steel during the CCT.
- The inner rust layer consists of Cl-containing akaganeite and Cr-goethite, while the outer rust layer is composed of amorphous iron oxyhydroxide mixed with various types of rust.
- FeCl2 and CrCl3 are formed from the Cl nest developed in the early stage, and the pitting at CrCl3-formed regions is locally accelerated because Cr is strongly hydrolyzed to a very low pH.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Lee, C.; Kang, B.; Choi, B.-H.; Lee, J.; Lee, K. Observation and characterization of squeak noises of polymeric materials for automotive interior parts under field-degradation. Trans. KSAE 2017, 25, 257–265. [Google Scholar] [CrossRef]
- Adikari, A.; Munasinghe, R.D.S.; Jayatileke, S. Prediction of atmospheric corrosion—A Review. Engineer 2014, 47, 75–83. [Google Scholar] [CrossRef]
- Alcántara, J.; Chico, B.; Simancas, J.; Díaz, I.; Morcillo, M. Marine atmospheric corrosion of carbon steel: A review. Materials 2017, 10, 406. [Google Scholar] [CrossRef] [Green Version]
- Kamimura, T.; Stratmann, M. The influence of chromium on the atmospheric corrosion of steel. Corros. Sci. 2001, 43, 429–447. [Google Scholar] [CrossRef]
- Asami, K.; Kikuchi, M. Characterization of rust layers on weathering steels air-exposed for a long period. Mater. Trans. 2002, 43, 2818–2825. [Google Scholar] [CrossRef] [Green Version]
- Yamashita, M.; Shimizu, T.; Konishi, H.; Mizuki, J.; Uchida, H. Structure and protective performance of atmospheric corrosion product of Fe–Cr alloy film analyzed by Mössbauer spectroscopy and with synchrotron radiation X-rays. Corros. Sci. 2003, 45, 381–394. [Google Scholar] [CrossRef]
- Yamashita, M.; Miyuki, H.; Matsuda, Y.; Nagano, H.; Misawa, T. The long term growth of the protective rust layer formed on weathering steel by atmospheric corrosion during a quarter of a century. Corros. Sci. 1994, 36, 283–299. [Google Scholar] [CrossRef]
- Zhao, Q.-H.; Liu, W.; Zhu, Y.-C.; Zhang, B.-L.; Li, S.-Z.; Lu, M.-X. Effect of small content of chromium on wet–dry acid corrosion behavior of low alloy steel. Acta Metall. Sin-Engl. 2017, 30, 164–175. [Google Scholar] [CrossRef]
- Park, S.-A.; Kim, J.-G.; Lee, B.-H.; Yoon, J.-B. Development of sulfuric and hydrochloric acid dew-point corrosion-resistant steels: 1. Effect of alloying elements on the corrosion resistance of low-alloy steels. Korean J. Met. Mater. 2014, 52, 837–855. [Google Scholar]
- Kim, S.-H.; Lee, J.-H.; Kim, J.-G.; Kim, W.-C. Effect of the crevice former on the corrosion behavior of 316L stainless steel in chloride-containing synthetic tap water. Met. Mater. Int. 2018, 24, 516–524. [Google Scholar] [CrossRef]
- Cheng, Q.; Chen, Z. The cause analysis of the incomplete semi-circle observed in high frequency region of EIS obtained from TEL-covered pure copper. Int. J. Electrochem. Sci 2013, 8, 8282–8290. [Google Scholar]
- Keddam, M.; Mottos, O.R.; Takenouti, H. Reaction model for iron dissolution studied by electrode impedance: I. Experimental results and reaction model. J. Electrochem. Soc. 1981, 128, 257–266. [Google Scholar] [CrossRef]
- Liu, W.; Dou, J.; Lu, S.; Zhang, P.; Zhao, Q. Effect of silty sand in formation water on CO2 corrosion behavior of carbon steel. Appl. Surf. Sci. 2016, 367, 438–448. [Google Scholar] [CrossRef]
- Zeng, L.; Zhang, G.; Guo, X. Erosion–corrosion at different locations of X65 carbon steel elbow. Corros. Sci. 2014, 85, 318–330. [Google Scholar] [CrossRef]
- Srinivasan, A.; Blawert, C.; Huang, Y.; Mendis, C.; Kainer, K.; Hort, N. Corrosion behavior of Mg–Gd–Zn based alloys in aqueous NaCl solution. J. Magnes. Alloy 2014, 2, 245–256. [Google Scholar] [CrossRef] [Green Version]
- Kim, M.; Jang, S.; Woo, S.; Kim, J.; Kim, Y. Corrosion resistance of ferritic stainless steel in exhaust condensed water containing aluminum cations. Corrosion 2015, 71, 285–291. [Google Scholar] [CrossRef]
- Cho, S.; An, J.-H.; Lee, S.-H.; Kim, J.-G. Effect of pH on the passive film characteristics of lean duplex stainless steel in chloride-containing synthetic tap water. Int. J. Electrochem. Sci 2020, 15, 4406–4420. [Google Scholar] [CrossRef]
- Chen, Y.; Hong, T.; Gopal, M.; Jepson, W. EIS studies of a corrosion inhibitor behavior under multiphase flow conditions. Corros. Sci. 2000, 42, 979–990. [Google Scholar] [CrossRef]
- Hamdy, A.S.; El-Shenawy, E.; El-Bitar, T. Electrochemical impedance spectroscopy study of the corrosion behavior of some niobium bearing stainless steels in 3.5% NaCl. Int. J. Electrochem. Sci. 2006, 1, 171–180. [Google Scholar]
- Bentiss, F.; Lebrini, M.; Vezin, H.; Chai, F.; Traisnel, M.; Lagrené, M. Enhanced corrosion resistance of carbon steel in normal sulfuric acid medium by some macrocyclic polyether compounds containing a 1,3,4-thiadiazole moiety: AC impedance and computational studies. Corros. Sci. 2009, 51, 2165–2173. [Google Scholar] [CrossRef]
- Lopez, D.A.; Simison, S.; De Sanchez, S. The influence of steel microstructure on CO2 corrosion. EIS studies on the inhibition efficiency of benzimidazole. Electrochim. Acta 2003, 48, 845–854. [Google Scholar] [CrossRef]
- Mansfeld, F. Recording and analysis of AC impedance data for corrosion studies. Corrosion 1981, 37, 301–307. [Google Scholar] [CrossRef]
- Ralston, K.D.; Birbilis, N.; Davies, C.H.J. Revealing the relationship between grain size and corrosion rate of metals. Scripta Mater. 2010, 63.12, 1201–1204. [Google Scholar] [CrossRef]
- Xiao, H.; Ye, W.; Song, X.; Ma, Y.; Li, Y. Evolution of akaganeite in rust layers formed on steel submitted to wet/dry cyclic tests. Materials 2017, 10, 1262–1275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asami, K.; Kikuchi, M. In-depth distribution of rusts on a plain carbon steel and weathering steels exposed to coastal–industrial atmosphere for 17 years. Corros. Sci. 2003, 45, 2671–2688. [Google Scholar] [CrossRef]
- Alcántara, J.; Chico, B.; Díaz, I.; De la Fuente, D.; Morcillo, M. Airborne chloride deposit and its effect on marine atmospheric corrosion of mild steel. Corros. Sci. 2015, 97, 74–88. [Google Scholar] [CrossRef] [Green Version]
- Senthilnathan, A.; Dissanayake, D.; Chandrakumara, G.; Mantilaka, M.; Rajapakse, R.; Pitawala, H.; Nalin de Silva, K. Akaganeite nanorices deposited muscovite mica surfaces as sunlight active green photocatalyst. R. Soc. Open Sci. 2019, 6, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verma, S.; Baig, R.N.; Nadagouda, M.N.; Varma, R.S. Oxidative CH activation of amines using protuberant lychee-like goethite. Sci. Rep. 2018, 8, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kimura, M.; Kihira, H. Nanoscopic mechanism of protective-rusts formation on weathering steel surfaces. SHINNITTETSU GIHO 2004, 91, 77–81. [Google Scholar]
- Xu, Q.; Gao, K.; Lv, W.; Pang, X. Effects of alloyed Cr and Cu on the corrosion behavior of low-alloy steel in a simulated groundwater solution. Corros. Sci. 2016, 102, 114–124. [Google Scholar] [CrossRef]
- Melchers, R.E. Effect of small compositional changes on marine immersion corrosion of low alloy steels. Corros. Sci. 2004, 46, 1669–1691. [Google Scholar] [CrossRef]
- Henriksen, J. The distribution of NaCl on Fe during atmospheric corrosion. Corros. Sci. 1969, 9, 573–576. [Google Scholar] [CrossRef]
- Ma, Y.; Li, Y.; Wang, F. Corrosion of low carbon steel in atmospheric environments of different chloride content. Corros. Sci. 2009, 51, 997–1006. [Google Scholar] [CrossRef]
- Ma, Y.; Li, Y.; Wang, F. The effect of β-FeOOH on the corrosion behavior of low carbon steel exposed in tropic marine environment. Mater. Chem. Phys. 2008, 112, 844–852. [Google Scholar] [CrossRef]
- Misawa, T.; Hashimoto, K.; Shimodaira, S. The mechanism of formation of iron oxide and oxyhydroxides in aqueous solutions at room temperature. Corros. Sci. 1974, 14, 131–149. [Google Scholar] [CrossRef]
- Yang, W.; Ni, R.-C.; Hua, H.-Z.; Pourbaix, A. The behavior of chromium and molybdenum in the propagation process of localized corrosion of steels. Corros. Sci. 1984, 24, 691–707. [Google Scholar] [CrossRef]
- SA, P.; DP, L. Alloying effect of chromium on the corrosion behavior of low-alloy steels. Meter. Trans. 2013, 54, 1770–1778. [Google Scholar]
- Jones, D.A. Principles and Prevention of Corrosion, 2nd ed.; Prentice Hall, Inc.: Upper Saddle River, NJ, USA, 1996; p. 217. [Google Scholar]


| Steels | Cr | C | Si | Mn | Fe |
|---|---|---|---|---|---|
| 0 Cr | 0.01 | 0.10 | 0.52 | 2.49 | Bal. |
| 0.3 Cr | 0.32 | 0.10 | 0.52 | 2.49 | Bal. |
| 0.5 Cr | 0.50 | 0.10 | 0.52 | 2.49 | Bal. |
| Parameter | 0 Cr | 0.3 Cr | 0.5 Cr |
|---|---|---|---|
| Ecorr (mVSCE) | −686.5 ± 1.4 | −683.6 ± 5.4 | −693.2 ± 12.6 |
| Icorr (μA/cm2) | 21.3 | 20.2 | 17.4 |
| Steel | Immersion Time | Rs (Ω·cm−2) | CPE1 | Rfilm (Ω·cm−2) | CPE2 | Rct (Ω·cm−2) | Rp (Ω·cm−2) | ||
|---|---|---|---|---|---|---|---|---|---|
| Qfilm (Ω−1 cm−2·sn) | n1 | Qct (Ω−1· cm−2·sn) | n1 | ||||||
| 0 Cr | 0 h | 1.424 | 7.52 × 10−4 | 0.8355 | 29.9 | 3.97 × 10−4 | 0.9671 | 636.5 | 665.7 |
| 1 h | 2.531 | 6.11 × 10−4 | 0.8146 | 192.6 | 1.29 × 10−4 | 0.9516 | 951.6 | 1144.2 | |
| 2 h | 2.522 | 6.36 × 10−4 | 0.8037 | 294 | 1.48 × 10−4 | 0.9883 | 801.2 | 1095.2 | |
| 3 h | 2.542 | 6.65 × 10−4 | 0.801 | 239.9 | 1.55 × 10−4 | 0.9724 | 831.3 | 1071.2 | |
| 4 h | 2.549 | 6.84 × 10−4 | 0.796 | 267.4 | 1.61 × 10−4 | 0.997 | 752.1 | 1019.5 | |
| 5 h | 2.574 | 6.72 × 10−4 | 0.7959 | 217.5 | 1.73 × 10−4 | 0.9593 | 766.3 | 983.8 | |
| 6 h | 2.589 | 6.81 × 10−4 | 0.7953 | 203.7 | 1.85 × 10−4 | 0.9509 | 733.1 | 936.8 | |
| 0.3 Cr | 0 h | 1.773 | 1.71 × 10−4 | 1 | 3 | 9.06 × 10−4 | 0.7621 | 1304 | 1307 |
| 1 h | 1.227 | 2.05 × 10−4 | 0.9653 | 32.3 | 3.76 × 10−4 | 0.7516 | 1210 | 1242.3 | |
| 2 h | 1.22 | 1.92 × 10−4 | 0.9714 | 29.2 | 3.79 × 10−4 | 0.7541 | 1151 | 1180.2 | |
| 3 h | 1.212 | 1.83 × 10−4 | 0.9759 | 26.5 | 3.90 × 10−4 | 0.7497 | 1215 | 1241.5 | |
| 4 h | 1.212 | 1.69 × 10−4 | 0.9843 | 24.3 | 4.00 × 10−4 | 0.7475 | 1223 | 1247.3 | |
| 5 h | 1.217 | 1.59 × 10−4 | 0.9914 | 23.7 | 4.10 × 10−4 | 0.7442 | 1266 | 1289.7 | |
| 6 h | 1.215 | 1.57 × 10−4 | 0.993 | 23.1 | 4.08 × 10−4 | 0.7424 | 1285 | 1308.1 | |
| 0.5 Cr | 0 h | 2.809 | 4.71 × 10−4 | 0.8449 | 21.6 | 2.14 × 10−4 | 0.8069 | 1414 | 1435.6 |
| 1 h | 1.986 | 5.09 × 10−4 | 0.8474 | 59 | 1.63 × 10−4 | 0.7983 | 1543 | 1602 | |
| 2 h | 1.978 | 5.18 × 10−4 | 0.8405 | 63.5 | 1.67 × 10−4 | 0.7818 | 1419 | 1482.5 | |
| 3 h | 1.964 | 3.86 × 10−4 | 0.8637 | 20.6 | 2.95 × 10−4 | 0.7261 | 1270 | 1290.6 | |
| 4 h | 1.963 | 3.24 × 10−4 | 0.8782 | 14.9 | 3.39 × 10−4 | 0.7095 | 1489 | 1503.9 | |
| 5 h | 2.001 | 1.18 × 10−4 | 0.9798 | 4.5 | 5.31 × 10−4 | 0.7418 | 1502 | 1506.5 | |
| 6 h | 2.013 | 9.42 × 10−4 | 0.9999 | 3.6 | 5.44 × 10−4 | 0.748 | 1542 | 1545.6 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cho, S.-w.; Ko, S.-J.; Yoo, J.-S.; Yoo, Y.-H.; Song, Y.-K.; Kim, J.-G. Effect of Cr on Aqueous and Atmospheric Corrosion of Automotive Carbon Steel. Materials 2021, 14, 2444. https://doi.org/10.3390/ma14092444
Cho S-w, Ko S-J, Yoo J-S, Yoo Y-H, Song Y-K, Kim J-G. Effect of Cr on Aqueous and Atmospheric Corrosion of Automotive Carbon Steel. Materials. 2021; 14(9):2444. https://doi.org/10.3390/ma14092444
Chicago/Turabian StyleCho, Sang-won, Sang-Jin Ko, Jin-Seok Yoo, Yun-Ha Yoo, Yon-Kyun Song, and Jung-Gu Kim. 2021. "Effect of Cr on Aqueous and Atmospheric Corrosion of Automotive Carbon Steel" Materials 14, no. 9: 2444. https://doi.org/10.3390/ma14092444







