1. Introduction
Waterborne emulsion paints are widely used as exterior coatings in the construction industry. The properties and applications of waterborne emulsion paints have been well documented [
1,
2,
3,
4]. They show excellent performance when it comes to durability, water resistance, and flexibility. They are also simple, single-component products that are ready to use. However, the properties of waterborne emulsion paints continue to be improved [
5,
6,
7,
8].
Most manufacturers now offer waterborne emulsion paints in a wide range of colors. Colorants are usually added in the form of pigment pastes instead of dry pigments. This method ensures that the pigments have a minimal impact on the coating properties, provided they are not used excessively. However, such interference does not necessarily diminish the properties of the paint. It can maintain or even improve its performance in some areas. There are many different types of colorants, which affect more than just the color of the paint [
9,
10,
11].
Most research focuses only on the properties that are supposed to be changed by the pigment (or other additive). Some studies also consider the ability of cool pigments to reduce surface temperature [
12,
13], the properties of organic pigments [
14,
15,
16] and spinel pigments [
17,
18,
19], color changes of thermochromic pigments under temperature [
20,
21,
22], or color fading [
23]. However, there are few studies testing the impact of pigments on the functional and processing properties of waterborne emulsion paints or coatings [
24,
25,
26]. Mentioned studies provide little research on the overall impact that additives can have on the product.
This work presents the impacts and possible benefits of including dry pigments in coating formulas by testing modified paint for all important application and processing properties. These effects and slight improvements are not usually investigated while testing the properties of new pigments. In the first part of the research, several pigments (green earth, spinel, iron oxide) were added to a premade base of acrylic–silicone paint. Following tests on the liquid paint and hardened coating, one pigment was selected as the most suitable, and a new paint was formulated with the pigment included as a core ingredient. Subsequently, further tests were conducted to determine the properties of the new paint.
Pigments for tests were chosen from different groups, with various reasons to use them in paint. Iron oxide red is a well-known, UV-stable red pigment; green earths are chosen as more economical alternatives to popular cobalt greens; and zinc–iron bronze is a spinel-type pigment (iron and zinc oxide mineral), with potential to have additional positive effects on the product. Green earths have been used as pigments for thousands years [
27]. They offer a less expensive alternative to other green pigments. The most popular green pigments—cobalt greens—are among most expensive, reaching prices of €150–200 per kg. Possible alternatives such as Nicosia Green can be purchased for €16 per kg [
28]. Spinels have been shown to have beneficial effects in different applications; for example, reducing metallical defects in ceramics [
29], or in anti-corrosive, heat-resistant coatings for metal applications [
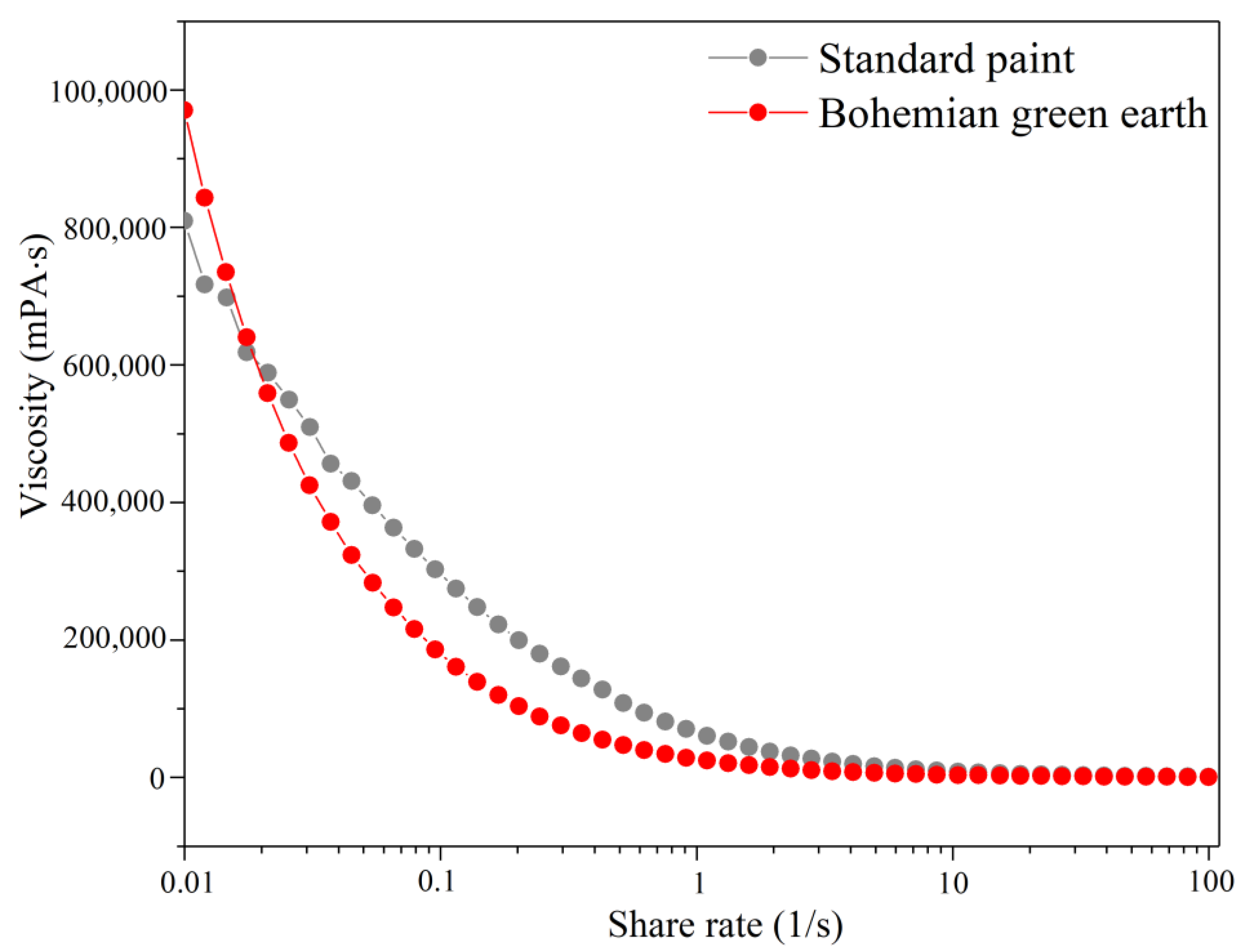
30]. In the present work, it has been shown that Bohemian Green Earth pigment may be successfully used as an effective coloring additive for paint formulation, while it had no major impact on the characteristics of the original paint, including its viscosity, wet-scrub resistance, and rheological properties. This work highlights the importance of extended product testing after any modification, as even minor changes in recipe can lead to differences in product properties.
2. Materials and Methods
2.1. Raw Materials
A standard waterborne acrylic-silicone facade paint (Atlas Sp. Z o. o., Łódź, Poland) was used as a carrier for the tested pigments. The following pigments were chosen for testing:
Zinc–iron bronze Fe2O3ZnO (Kremer Pigmente GmbH & Co., Aischstetten, Germany)—a spinel pigment obtained under high temperature from iron oxides (II) and (III) and zinc oxide, characterized by high heat resistance, high UV resistance, and low absorption of solar radiation through the entire spectrum. It is classified as a “cool” pigment, able to resist surface heating under sunlight.
Iron red (Kremer Pigmente GmbH & Co., Aischstetten, Germany)—iron oxide α-Fe2O3, a red pigment, known by the color index PR101.
Nicosia green (Kremer Pigmente GmbH & Co., Aischstetten, Germany)—a mixture of earth pigments containing compounds of iron, aluminum, magnesium, and potassium, known by the color index PG23.
Bohemian earth (Kremer Pigmente GmbH & Co., Aischstetten, Germany)—PG23 excavated at a different location, containing additional silicon and calcium compounds (Fe, Al, Si, K, Mg, Ca). The shade and hue of PG23 may vary due to composition differences, depending on the excavation site.
The following commercial pigment pastes were used only as a reference for color aging tests (they were not taken into consideration when testing properties):
Commercial green GX11—a pigment paste based on cobalt green pigment Co2TiO4. PG50—a spinel pigment obtained under high temperature from cobalt (II) oxide and titanium (IV) oxide.
Commercial red RX10—iron red, PR101, in the form of a pigment paste.
Commercial red RH6—organic red pigment, known by the color index PR254.
The following material was used as reference filler. It was added to the reference sample to maintain equal dry component concentration through the samples:
2.2. Sample Preparation
Samples were prepared by the addition of 5% w/w of each pigment to the base paint. The pigments were gradually incorporated to the samples using a rotary agitator at a speed of 1000 rpm. Each sample was placed in a sealed container until needed for research. P60 is a neutral paint filler used in the reference sample to equalize the pigment concentration with the other samples.
2.3. Test Methods
The following test methods were applied to determine the effects of each pigment on the material:
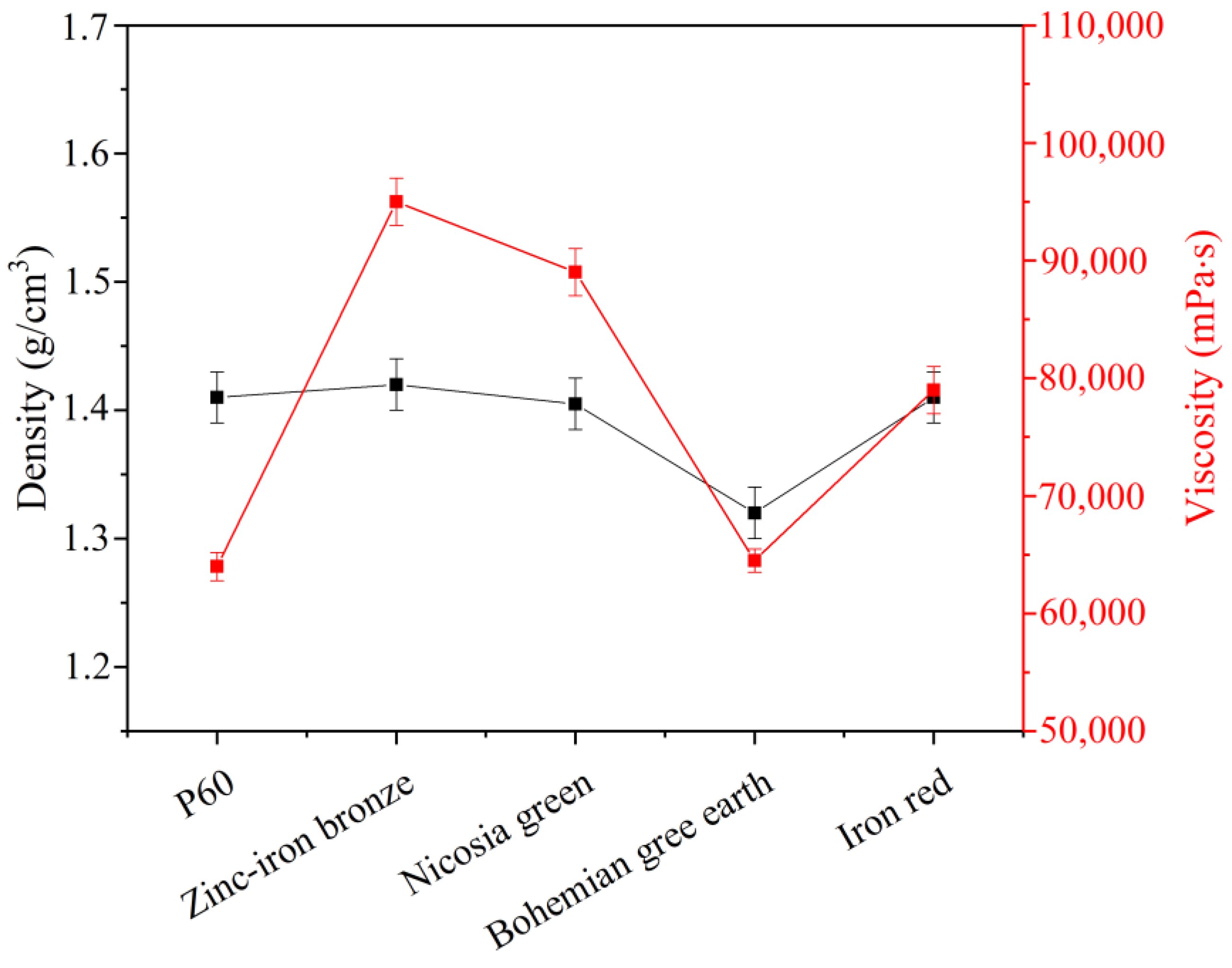
Density measurements were performed using a density cup with a volume of 100.088 cm
3 to measure the weight of each sample, followed by density calculations according to the formula given in PN-EN ISO 2811-1:2012 [
31]:
where
m is sample mass and
V is volume. Each sample was measured three times. The results were averaged.
Viscosity measurements were performed according to PN-EN ISO 2555:2011 [
32] using a Brookfield (Middleboro, MA, USA) DV-II+ Pro viscometer at 20 rpm with a spindle for highly viscous liquids. Each sample was measured immediately after incorporating the pigment into the paint.
Gloss measurement was performed using a GLS glossmeter (Gdańsk, Poland) at angles of 60° and 85°, since the base paint is supposed to be matt, according to PN-EN ISO 2813:2014-11 [
33].
The rub-out test was performed by gently rubbing the surface of the applied paint with a fingertip after short time periods (1, 5, and 10 min after application) [
34]. The rub-out test was used to analyze the compatibility of the pigment with the material by observing the occurrence of flocculation.
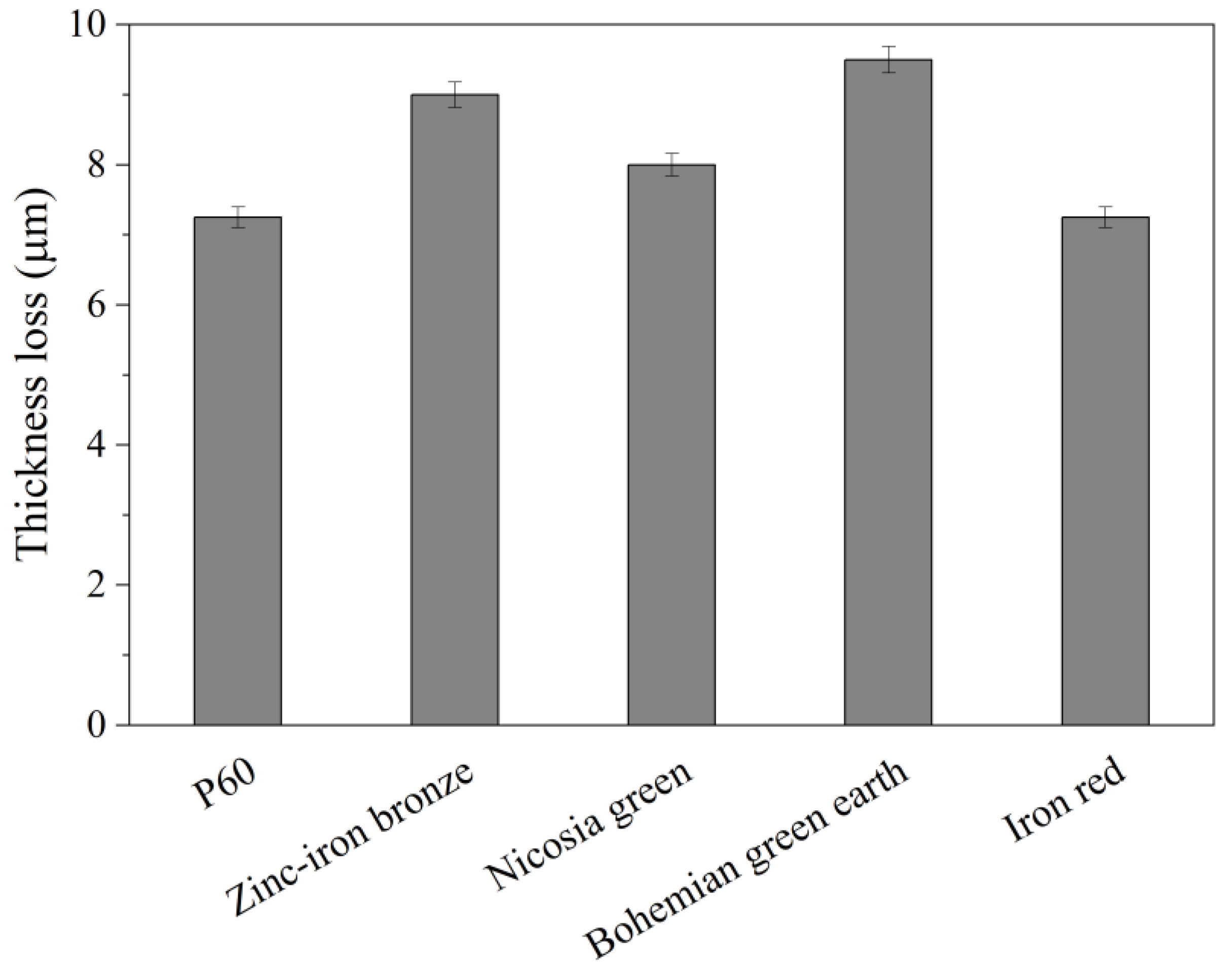
The wet-scrub resistance test was performed for 200 cycles of scrub on an Elcometer 1720 (Manchester, UK) using a sponge and a 5% solution of sodium dodecylbenzenesulfonate (ABSNa) as a wetting agent. The samples were prepared by the application of a 300 µm thick coating of each paint on a dedicated scrub test substrate. The coatings were conditioned for 28 days at room temperature to ensure complete drying. The dry samples were measured using a thickness meter and placed in the scrub resistance tester. After the test, the samples were rinsed with water and placed in an air dryer at temperature of 50 °C for 7 days. Subsequently, the thicknesses of the coatings was measured again. Scrub resistance class was determined based on loss of coating thickness (PN-EN 13300:2002 standard) [
35].
Adhesion to the substrate was measured using the TQC cross-cut adhesion test (Hadley, UK). Samples were applied to cardboard using a 300-µm applicator and left for 28 days at room temperature to dry out. Next, the surface of each sample was cut vertically and horizontally, with an angle of 90° between cuts. Adhesion to the substrate was determined based on chipping at the incision edges.
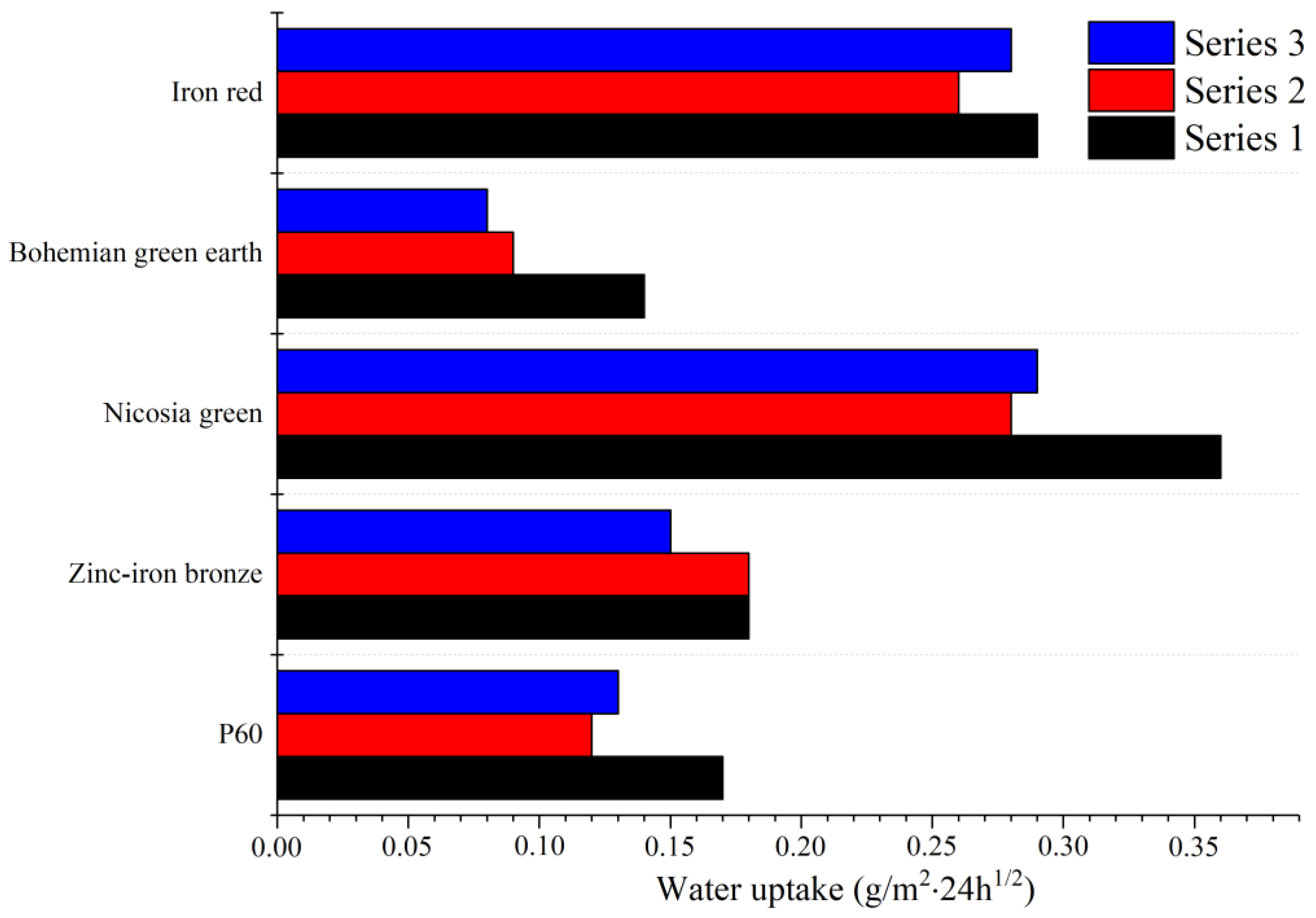
Water absorption tests were conducted according to the PN-C-81521:1976 standard. Two equal layers of paint were applied to a limestone substrate with a 24-h wait between layers. The dimensions of the limestone substrate were 11.5 × 11 × 2.5 cm. The samples were left to dry out for 28 days at room temperature. The edges and sides of the limestone were covered with silicone to seal any areas not covered with paint. Two samples were prepared for each pigment. The samples were conditioned by dipping in water for 72 h, followed by drying in an air dryer in 50 °C for 72 h and resting for 24 h in room temperature. The samples were then weighed and placed in a container filled with water up to half the height of the sides of the samples. The samples were placed on a piece of sponge with the surface covered with paint facing down, to ensure contact with water throughout the test. After 24 h, the samples were weighed again and left to dry at 50 °C for another 24 h. The process was repeated three times. The water uptake factor was calculated according to the formula [
36].
where
W24 is the water absorbance factor after 24 h,
m24 is the sample mass after 24 h in water,
m0 is the sample mass before dipping in water and
S is the surface of the sample covered with paint.
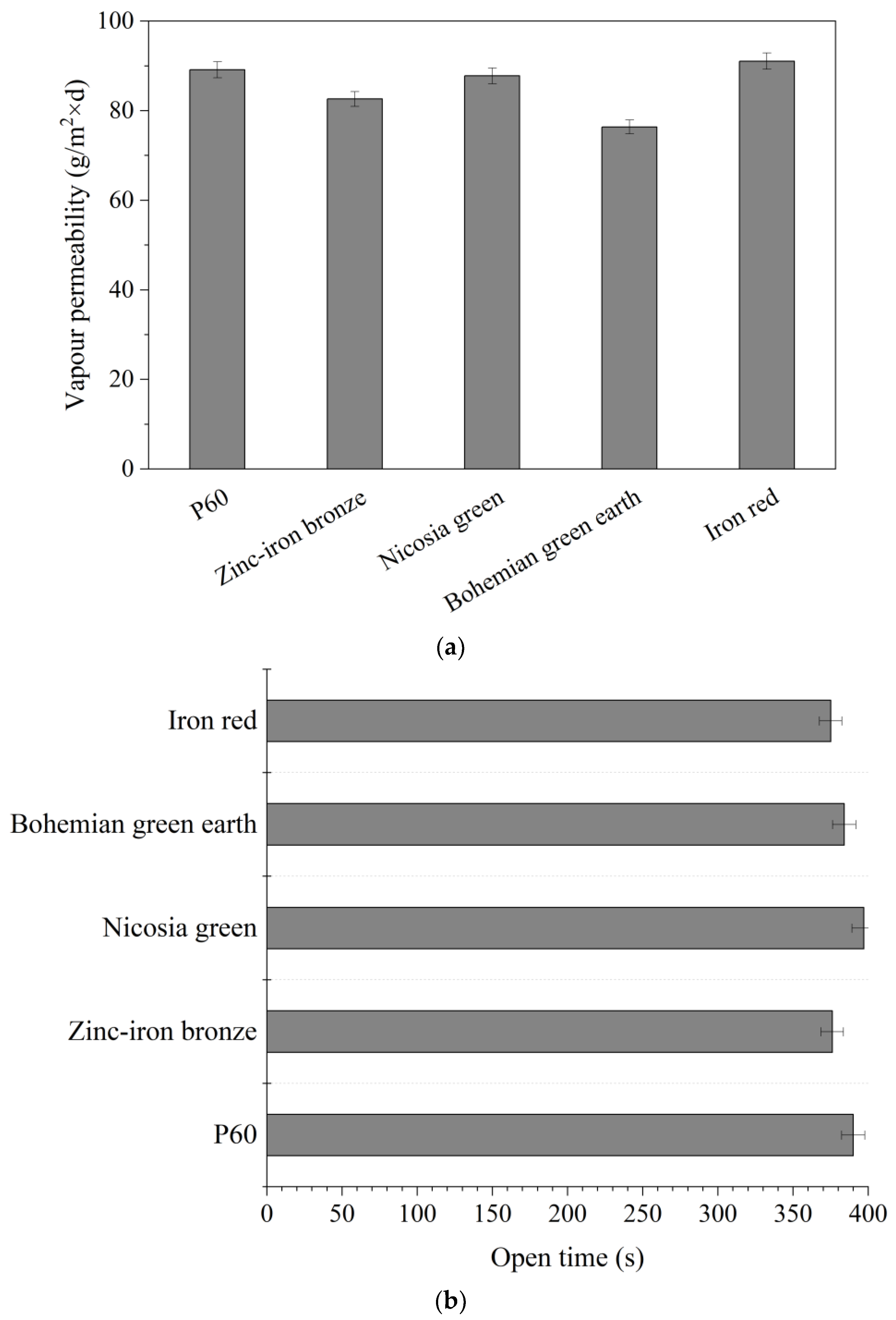
Vapor permeability was measured using the wet-cup method, according to PN-EN ISO 7783:2018-11 [
37]. Samples were prepared by applying two equal layers of paint on an oval substrate, with a 24-h wait between layers. The samples were left to dry out for 28 days. Measuring cups were filled with a saturated solution of anhydrous ammonium dihydrogen phosphate, leaving space of about 30 mm between the surface of the solution and the sample. The samples were placed on top of the measuring cups, sealed, and weighed. The edges of each sample were sealed with rubber bands to ensure vapor circulation exclusively through the sample. The cups were placed in a chamber with forced air circulation and weighed every 24 h for 4 days. The linearity of the graph shows that the test has been correctly executed.
Open time measurements were conducted using a Q-Lab open time tester (Westlake, OH, USA) equipped with a rod for scratching the surface of the sample and a timescale alongside the sample. As the scratching rod moves steadily over the surface, the timescale makes it possible to monitor the time passing. Coatings were applied with a 200-µm applicator on a glass plate. The measurements lasted for 15 min, during which the scratching rod moved through the sample to the point where it no longer damaged the forming film. Each sample was measured separately immediately after application [
34].
Contrast measurements (mating) were conducted using an X-Rite Color i5 (Grand Rapids, MI, USA) spectrophotometer according to PN-EN ISO 2814:2006 [
38]. Samples were prepared by applying a 300-µm film of paint on black-white contrast cards. The samples were conditioned for 28 days at room temperature. Mating ability was determined by measuring Lab values over white and black fields and calculating the ΔE values.
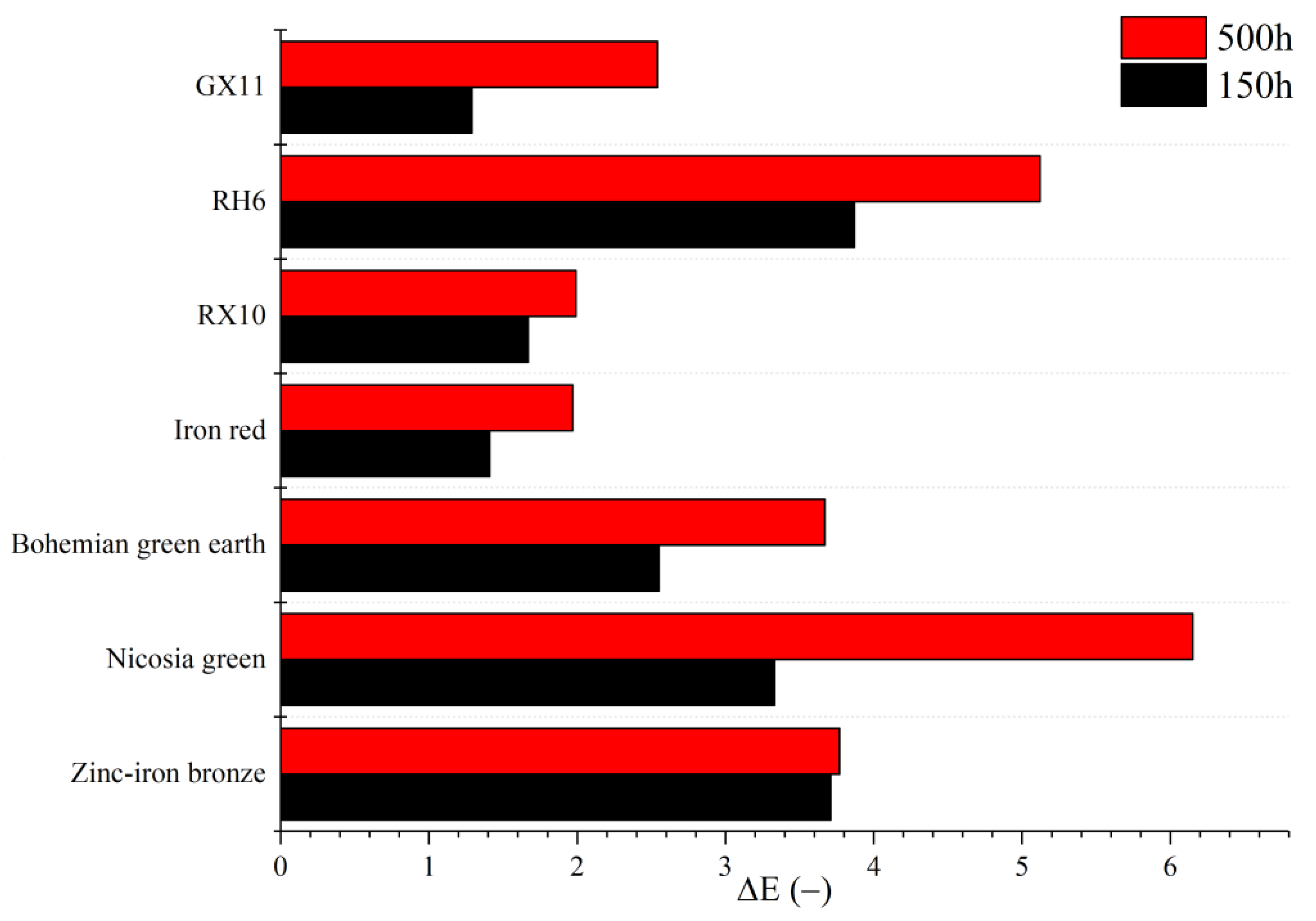
UV color stability was measured using a X-Rite Color i5 (Grand Rapids, MI, USA) spectrophotometer. The lab values were compared with reference samples using ΔE, according to PN-EN ISO 16474-3:2014_02 [
39]. The samples were conditioned using a Q-Lab xenotest chamber (Westlake, OH, USA), where they were exposed to xenon arc lamps imitating full spectrum solar irradiance for 28 days at room temperature. Two series of measurements were conducted: ageing for 150 h and aging for 500 h. After that time, the lab values of the samples were measured
Particle size measurements were conducted using the light scattering method with a Zetasizer device (Malvern, UK). Samples were prepared by mixing 0.1 g of the pigment in 100 g of water. Ultrasound was used to maximize pigment dispersal. The samples were placed in measuring cuvettes and transferred to the Zetasizer device.
4. Conclusions
This work investigated the possible use of dry pigments as replacements for currently used colorants, even without first being liquified into pigment pastes. The best results were obtained with the addition of Bohemian green earth pigment, which had no major impact on the rheological characteristics of the original paint, meaning it can be used without being made into a pigment paste. This is a huge advantage, as green pigments tend to be the most expensive additives. Nonetheless, any change in the formulas of commercial paints, especially related to the color, brings additional costs. Each new addition to the formula should be subjected to tests to determine its impact on important properties, and its possible benefits for paint quality.