Influence of Oxygen Content in the Protective Gas on Pitting Corrosion Resistance of a 316L Stainless Steel Weld Joint
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Influence of Oxygen Content on Discoloration
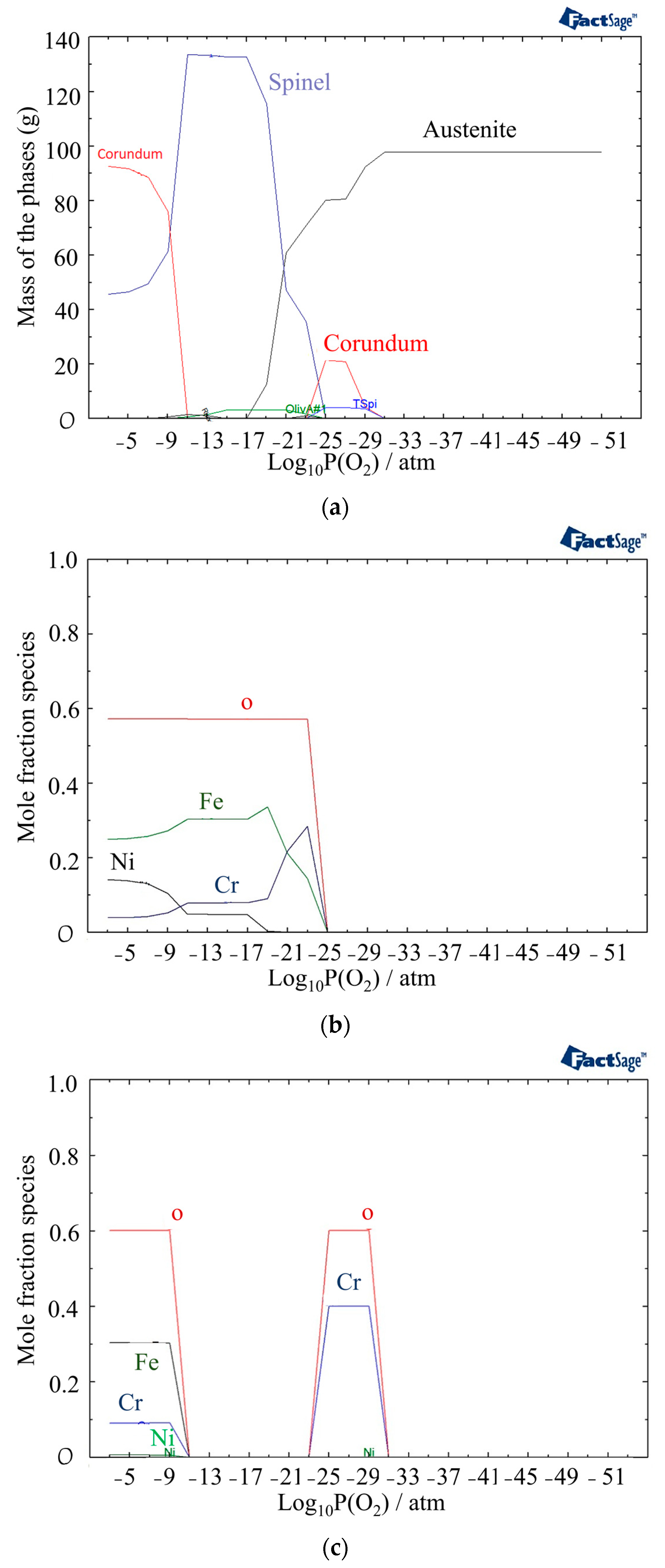
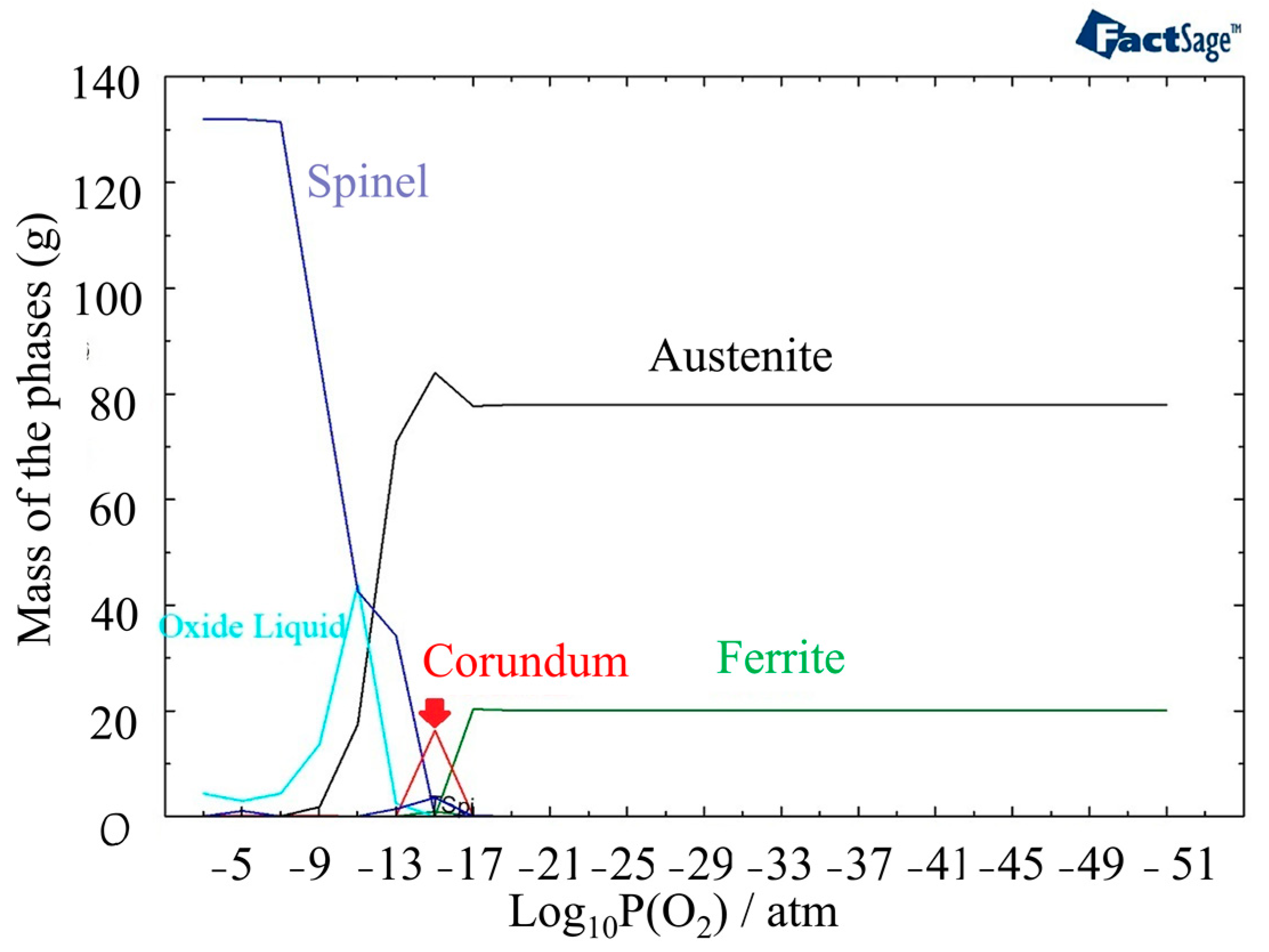
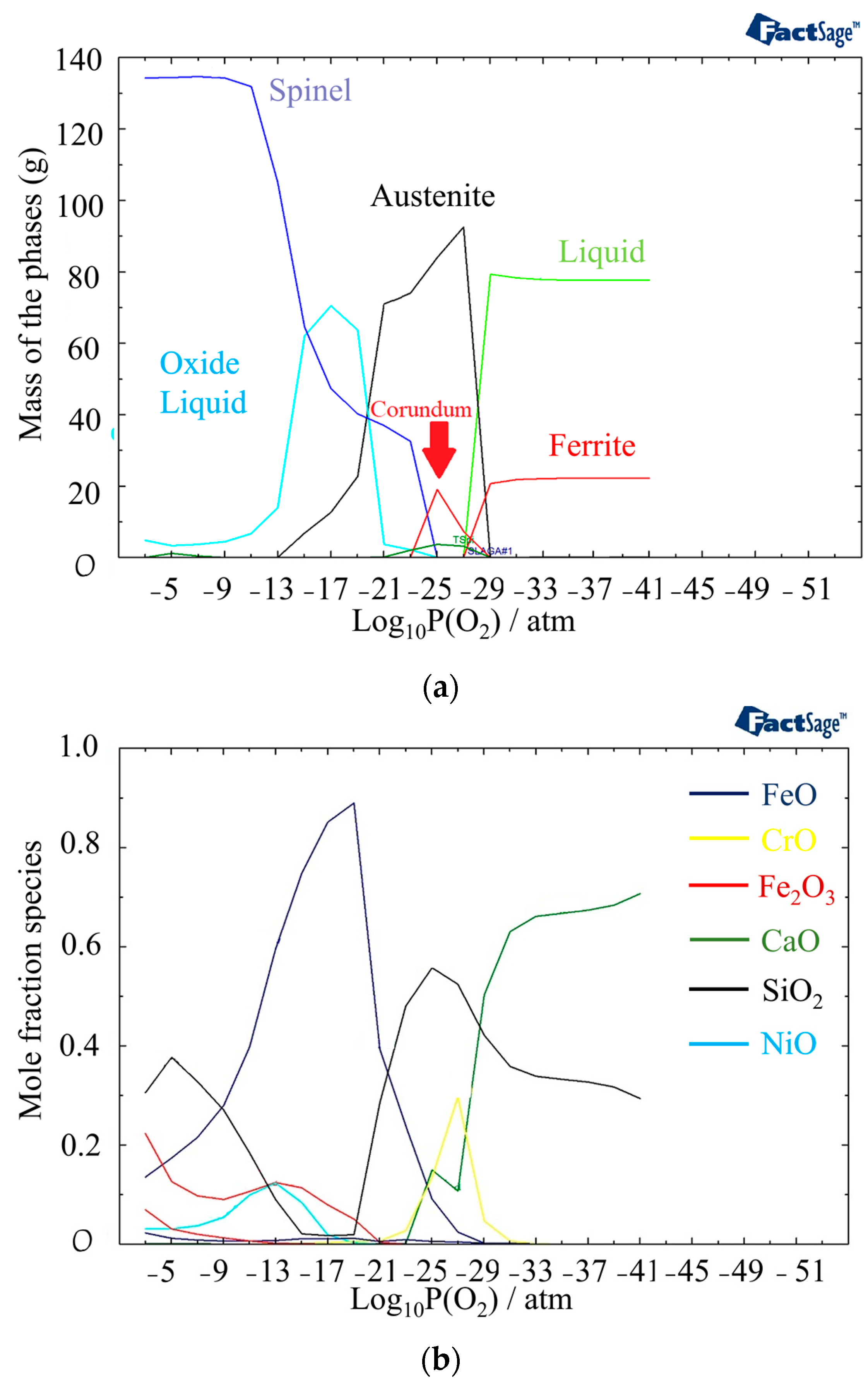
3.2. Thermodynamic Calculation
3.3. Oxide Geometrical Characteristics
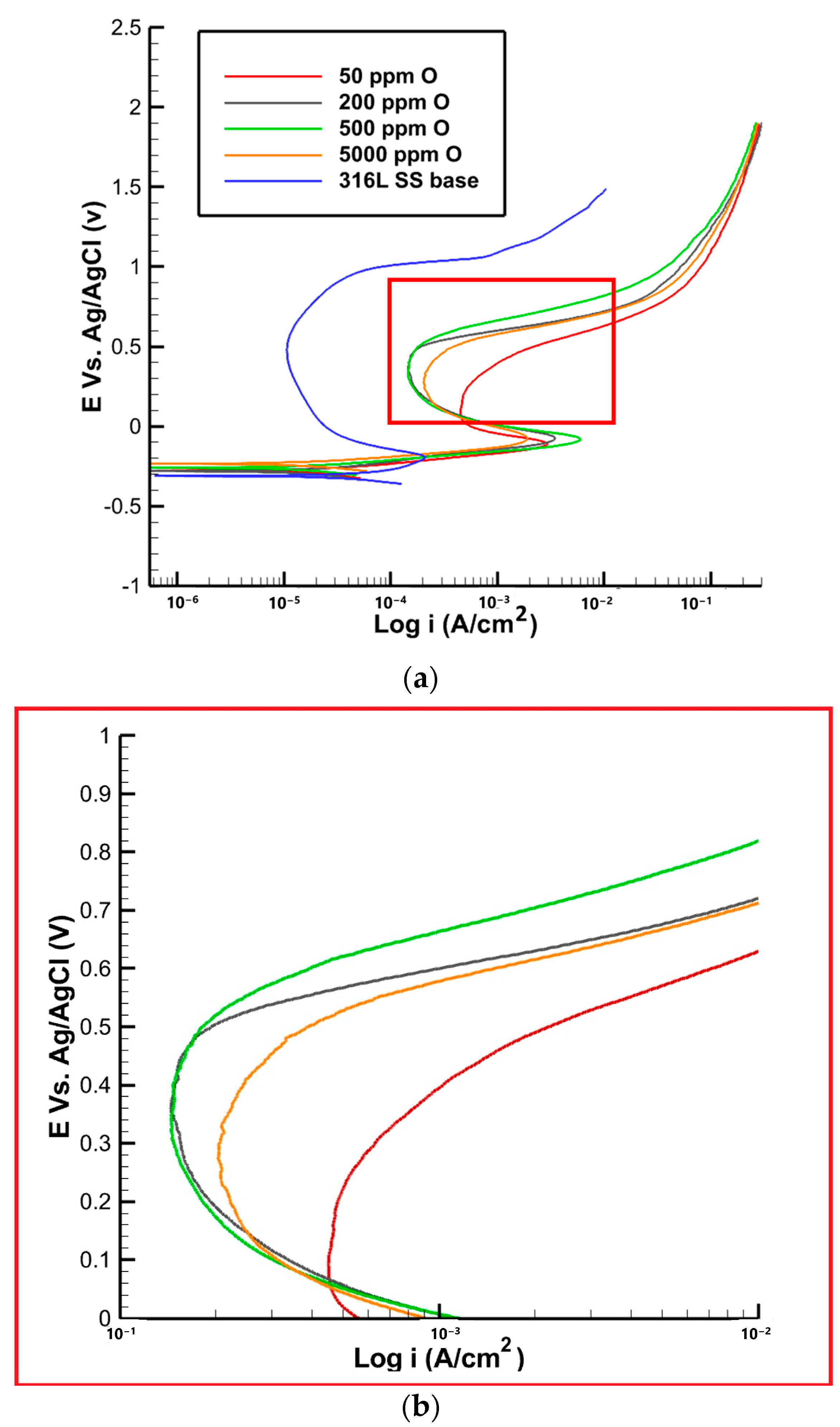
3.4. Pitting Corrosion Tests
4. Conclusions
- The width of discoloration increased with increasing oxygen content, and notable differences in color were observed between the samples with 5000 ppm and 500 ppm/200 ppm of oxygen. In particular, the 5000 ppm sample exhibited an opaque zone located behind the welding line, indicating distinct characteristics in the oxide formation process at high oxygen concentrations.
- The results indicate that the oxide thickness increases with an increase in the oxygen content of the purging gas, when it ranges from 50 to 5000 ppm. This suggests that a higher concentration of oxygen promotes the formation of a thicker oxide layer on the surface of the material. The observed trend in oxide thickness provides valuable insights into the influence of oxygen content on the oxide formation process and its subsequent impact on the corrosion resistance of the material.
- The oxide structure was found to be a mixture of Spinel and Corundum phases, with the presence of an oxide liquid phase at high temperatures. The oxide liquid phase exhibited a higher concentration of FeO, which resulted in a porous structure.
- The pitting corrosion resistance showed an increasing trend up to a 500 ppm oxygen content, followed by a decrease at 5000 ppm. The sample with 500 ppm exhibited the highest pit potential, suggesting the formation of a more stable passive film that is less susceptible to localized corrosion. The presence of a porous oxide structure near the fusion line in the 5000 ppm sample may have contributed to its lower pitting corrosion resistance.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Revie, R.W. Uhlig’s Corrosion Handbook; John Wiley & Sons: Hoboken, NJ, USA, 2011; Volume 51. [Google Scholar]
- Dai, P.; Wang, Y.; Li, S.; Lu, S.; Feng, G.; Deng, D. FEM analysis of residual stress induced by repair welding in SUS304 stainless steel pipe butt-welded joint. J. Manuf. Process. 2020, 58, 975–983. [Google Scholar] [CrossRef]
- Paulraj, P.; Garg, R. Effect of welding parameters on pitting behavior of GTAW of DSS and super DSS weldments. Eng. Sci. Technol. Int. J. 2016, 19, 1076–1083. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, Z.; Tan, H.; Hong, J.; Jiang, Y.; Jiang, L.; Li, J. Effect of a brief post-weld heat treatment on the microstructure evolution and pitting corrosion of laser beam welded UNS S31803 duplex stainless steel. Corros. Sci. 2012, 65, 472–480. [Google Scholar] [CrossRef]
- Chuaiphan, W.; Srijaroenpramong, L. Effect of welding speed on microstructures, mechanical properties and corrosion behavior of GTA-welded AISI 201 stainless steel sheets. J. Mater. Process. Technol. 2014, 214, 402–408. [Google Scholar] [CrossRef]
- Mohammed, G.R.; Ishak, M.; Aqida, S.N.; Abdulhadi, H.A. Effects of heat input on microstructure, corrosion and mechanical characteristics of welded austenitic and duplex stainless steels: A review. Metals 2017, 7, 39. [Google Scholar] [CrossRef]
- Bergquist, E.-L.; Huhtala, T.; Karlsson, L. The effect of purging gas on 308L TIG root pass ferrite content. Weld. World 2011, 55, 57–64. [Google Scholar] [CrossRef]
- Taban, E.; Kaluc, E.; Aykan, T. Effect of the purging gas on properties of 304H GTA welds. Weld. J. 2014, 93, 124s–130s. [Google Scholar]
- Shirwaikar, C.; Reddy, G. Purging with nitrogen in the welding of austenitic stainless steels. Weld. J. 1975, 12s–15s. Available online: http://files.aws.org/wj/supplement/WJ_1975_01_s12.pdf (accessed on 2 August 2023).
- Trigwell, S.; Selvaduray, G. Effects of welding on the passive oxide film of electropolished 316L stainless steel. J. Mater. Process. Technol. 2005, 166, 30–43. [Google Scholar] [CrossRef]
- Perez, N. Corrosivity and passivity. In Electrochemistry and Corrosion Science; Springer: Cham, Switzerland, 2016; pp. 199–264. [Google Scholar] [CrossRef]
- Ling, L.; Liu, T.; Lu, Y.; Guo, P. Investigation of the oxides film on 304L base metal produced during welding process without inert gas shielding. Appl. Surf. Sci. 2019, 465, 780–786. [Google Scholar] [CrossRef]
- Huang, X.; Xiao, K.; Fang, X.; Xiong, Z.; Wei, L.; Zhu, P.; Li, X. Oxidation behavior of 316L austenitic stainless steel in high temperature air with long-term exposure. Mater. Res. Express 2020, 7, 066517. [Google Scholar] [CrossRef]
- Xie, B.; Sun, M.; Xu, B.; Wang, C.; Jiang, H.; Li, D.; Li, Y. Oxidation of stainless steel in vacuum and evolution of surface oxide scales during hot-compression bonding. Corros. Sci. 2019, 147, 41–52. [Google Scholar] [CrossRef]
- Kimbrel, K. Determining Acceptable Levels of Weld Discoloration on Mechanically Polished and Electropolished Stainless Steel Surface. Pharm. Eng. 2011, 31, 1–7. [Google Scholar]
- Macleod, H.A. Thin-Film Optical Filters; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Pedrotti, L.S. Basic physical optics. Fundam. Photonics 2008, 1, 152–154. [Google Scholar] [CrossRef]
- Khafaji, N.Y.; Demir, A.G.; Vitali, L.; Fustinoni, D.; Niro, A.; Previtali, B.; Taha, Z.A. Optical characterization of laser coloured titanium under different processing atmospheres. Surf. Coat. Technol. 2017, 321, 156–163. [Google Scholar] [CrossRef]
- Bale, C.; Bélisle, E.; Chartrand, P.; Decterov, S.; Eriksson, G.; Hack, K.; Jung, I.-H.; Kang, Y.-B.; Melançon, J.; Pelton, A.; et al. FactSage thermochemical software and databases. Calphad 2002, 26, 189–228. [Google Scholar] [CrossRef]
- ASTM G5-14; Standard Reference Test Method for Making Potentiodynamic Anodic Polarization Measurements. ASTM: West Conshohocken, PA, USA, 2014.
- Kumar, S.; Shahi, A.S.; Sharma, V.; Malhotra, D. Effect of welding heat input and post-weld thermal aging on the sensitization and pitting corrosion behavior of AISI 304L stainless steel butt welds. J. Mater. Eng. Perform. 2021, 30, 1619–1640. [Google Scholar] [CrossRef]
- Bellezze, T.; Giuliani, G.; Roventi, G. Study of stainless steels corrosion in a strong acid mixture. Part 1: Cyclic potentiodynamic polarization curves examined by means of an analytical method. Corros. Sci. 2018, 130, 113–125. [Google Scholar] [CrossRef]
- Comotti, I.M.; Trueba, M.; Trasatti, S.P. The pit transition potential in the repassivation of aluminium alloys. Surf. Interface Anal. 2013, 45, 1575–1584. [Google Scholar] [CrossRef]
- Finšgar, M.; Milošev, I. Corrosion behaviour of stainless steels in aqueous solutions of methanesulfonic acid. Corros. Sci. 2010, 52, 2430–2438. [Google Scholar] [CrossRef]
- AWS D18.2:2020; Guide to Weld Discoloration Levels on Inside of Austenitic Stainless Steel Tube. American Welding Society: Doral, FL, USA, 2020.
- Geffroy, P.-M.; Guironnet, L.; Bouwmeester, H.; Chartier, T.; Grenier, J.-C.; Bassat, J.-M. Influence of oxygen partial pressure on the oxygen diffusion and surface exchange coefficients in mixed conductors. J. Eur. Ceram. Soc. 2019, 39, 59–65. [Google Scholar] [CrossRef]
- Laidler, K.J. The development of the Arrhenius equation. J. Chem. Educ. 1984, 61, 494. [Google Scholar] [CrossRef]
- Li, J.; Li, H.; Zhao, G.; Zhou, C.; Ma, L.; Liu, H. Insights into the High-Temperature Oxidation Behavior of Austenitic Stainless Steel and Influence of Copper on It. J. Mater. Eng. Perform. 2020, 29, 3661–3669. [Google Scholar] [CrossRef]
- Habib, K.A.; Damra, M.S.; Saura, J.J.; Cervera, I.; Bellés, J. Breakdown and evolution of the protective oxide scales of AISI 304 and AISI 316 stainless steels under high-temperature oxidation. Int. J. Corros. 2011, 2011, 824676. [Google Scholar] [CrossRef]
- Prado, L.H.; Anastasiou, E.; Virtanen, S. Corrosion Behavior of Anodic Self-Ordered Porous Oxide Layers on Stainless Steel. J. Electrochem. Soc. 2021, 168, 021507. [Google Scholar] [CrossRef]
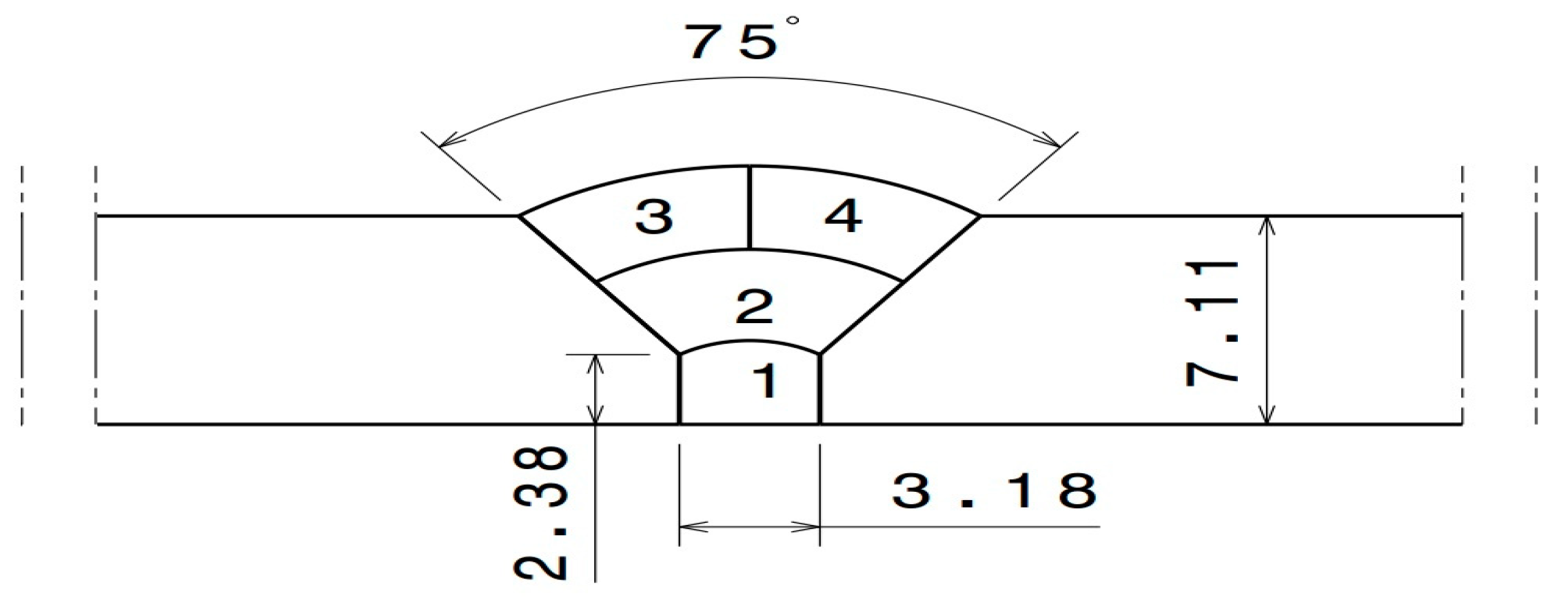
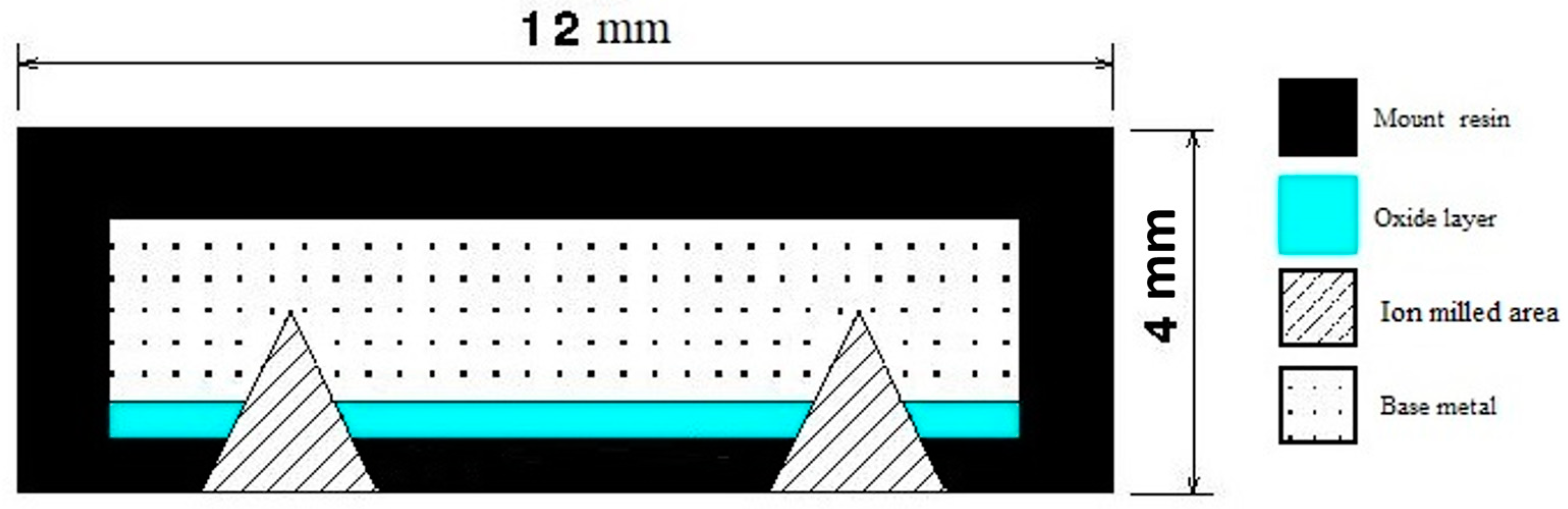







| Element | C | Mn | Si | P | S | Mo | Cr |
| Amount | 0.02 | 0.97 | 0.43 | 0.28 | 0.001 | 2.05 | 16.47 |
| Element | Ni | Al | Co | Cu | W | N | Other |
| Amount | 11.37 | 0.0162 | 0.22 | 0.25 | 0.073 | 0.093 | 0.13 |
| Process | GTAW | GTAW | GTAW | GTAW |
|---|---|---|---|---|
| Pass | 1 | 2 | 3 | 4 |
| Shielding gas | Ar | Ar | Ar | Ar |
| Purge gas flow rate (CFH) | 40 | 40 | 40 | 40 |
| Current (A) | 125–202 | 166–215 | 200–223 | 208–243 |
| Voltage (V) | 10.6–14 | 11.8–15.7 | 12.8–16.4 | 13.8–16.4 |
| Tube to work distance (mm) | 9.52 | 9.52 | 9.52 | 9.52 |
| Heat In Put (kJ.mm−1) | 1.31 | 1.17 | 1.28 | 1.44 |
| Filer material | ER316L (Exocor) | ER316L (Exocor) | ER316L (Exocor) | ER316L (Exocor) |
| Oxygen Content (PPM) | 50 | 200 | 500 | 5000 |
|---|---|---|---|---|
| Component | Ar + 0.005% O | Ar + 0.02% O | Ar + 0.05% O | Ar + 0.5% O |
| Zone | Phase |
|---|---|
| 1 | Liquid |
| 2 | Liquid + MeS |
| 3 | Liquid + alfa Ca2SiO4 |
| 4 | Liquid + Oxide Liquid |
| 5 | Liquid + Oxide Liquid + Spinel |
| 6 | Liquid + Oxide Liquid + Corundum + SiO2 + TSpinel |
| 7 | Delta ferrite + Austenite + MeS |
| 8 | Austenite + MeS |
| 9 | Oxide Liquid + Austenite + Spinel + TSpinel |
| 10 | Austenite + MeS + Corundum + SiO2 + TSpinel + Ca3Cr2Si3O12 |
| 11 | Austenite + MeS + Wollastonite |
| 12 | Austenite + MeS |
| 13 | Austenite + MeS + Corundum + SiO2 + TSpinel + Wollastonite |
| 14 | Spinel + Austenite + Olivine + Beta Ni3S2 |
| 15 | Spinel + Olivine + Rhodonite + Pyrrhotite |
| 16 | Oxide Liquid + Spinel |
| 17 | Spinel + Corundum + SiO2 + MnSO4 + CaSO4 |
| 18 | Austenite + MeS + Spinel + Rhodonite + TSpinel + Wollastonite |
| 19 | Spinel + Olivine + Rhodonite + MnSO4 + CaSO4 |
| 20 | Spinel + Corundum + Olivine + CaSO4 + MnSO4 |
| 21 | Spinel + Corundum + SiO2 + MnSO4 + CaSO4 + MnO2 + CrO3 |
| 22 | Spinel + Olivine |
| 23 | Delta ferrite + Austenite + MeS + Liquid |
| 24 | Delta ferrite + MeS + Liquid |
| Oxygen Content (PPM) | 50 | 200 | 500 | 5000 | 316L SS Base |
|---|---|---|---|---|---|
| ipassive (µA) | 463 ± 10 | 210 ± 10 | 230 ± 20 | 290 ± 30 | 198 ± 30 |
| ipit (µA) | 463 ± 10 | 200 ± 10 | 210 ± 10 | 260 ± 20 | 297 ± 30 |
| Epassive (V) | 0 ± 0.01 | 0.05 ± 0.03 | 0.07 ± 0.03 | 0.09 ± 0.01 | −0.04 ± 0.01 |
| Epit (V) | 0.42 ± 0.06 | 0.53 ± 0.01 | 0.54 ± 0.01 | 0.49 ± 0.02 | 0.96 ± 0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maroufkhani, M.; Hakimian, S.; Khodabandeh, A.; Radu, I.; Hof, L.A.; Jahazi, M. Influence of Oxygen Content in the Protective Gas on Pitting Corrosion Resistance of a 316L Stainless Steel Weld Joint. Materials 2023, 16, 5968. https://doi.org/10.3390/ma16175968
Maroufkhani M, Hakimian S, Khodabandeh A, Radu I, Hof LA, Jahazi M. Influence of Oxygen Content in the Protective Gas on Pitting Corrosion Resistance of a 316L Stainless Steel Weld Joint. Materials. 2023; 16(17):5968. https://doi.org/10.3390/ma16175968
Chicago/Turabian StyleMaroufkhani, Mohammad, Soroosh Hakimian, Alireza Khodabandeh, Iulian Radu, Lucas A. Hof, and Mohammad Jahazi. 2023. "Influence of Oxygen Content in the Protective Gas on Pitting Corrosion Resistance of a 316L Stainless Steel Weld Joint" Materials 16, no. 17: 5968. https://doi.org/10.3390/ma16175968








