Gemcitabine Combination Nano Therapies for Pancreatic Cancer
Abstract
:1. Introduction
2. Gemcitabine—A Gold Standard Chemotherapeutic Agent for Pancreatic Cancer
2.1. Gemzar
2.2. HPMA Copolymer-Based Gemcitabine Formulation
2.3. Gemlip
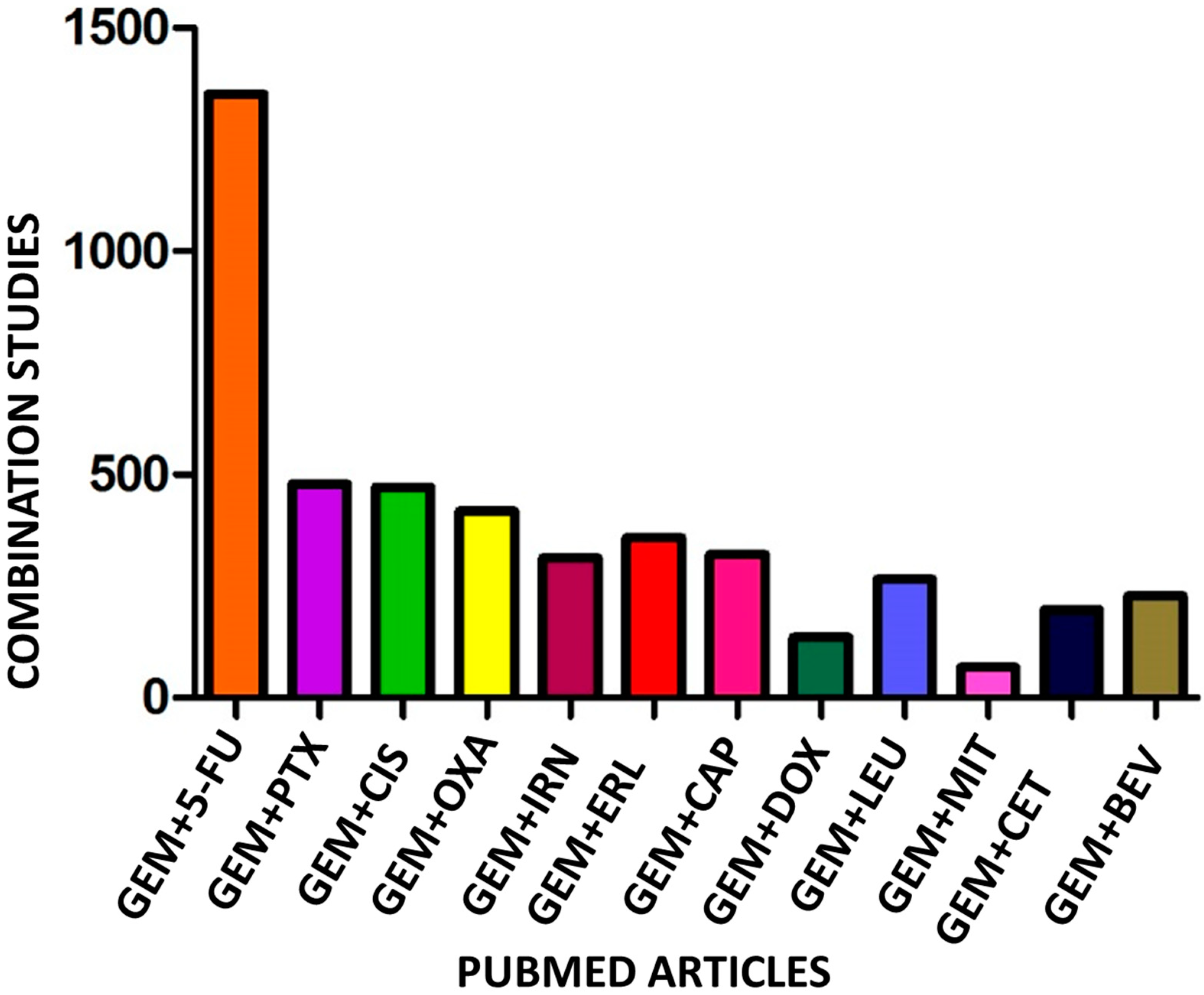
2.4. Co-Delivery of Gemcitabine
3. Nano Formulations Involved in Gemcitabine Co-Administration and Co-Delivery
3.1. Liposomes
3.2. Nanogels
3.3. Micelles
3.4. Albumin Nanoparticles
3.5. Multifunctional Nanoparticles
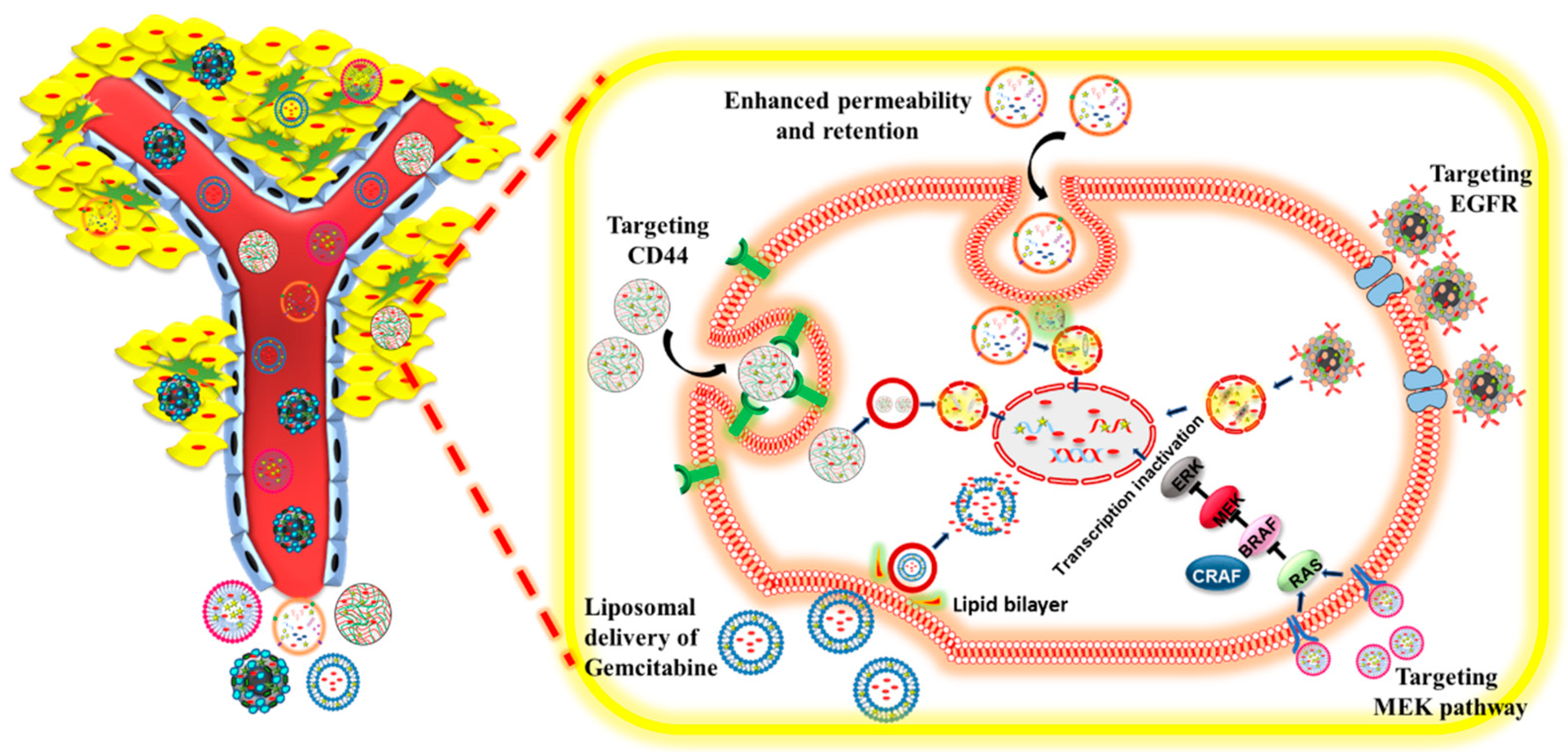
3.6. Targeted Nanoparticles
4. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| PanCa | Pancreatic cancer |
| PDAC | Pancreatic Ductal Adenocarcinoma |
| GEM | Gemcitabine |
| 5-FU | 5-Fluorouracil |
| PTX | Paclitaxel |
| CIS | Cisplatin |
| OXA | Oxaliplatin |
| IRN | Irinotecan |
| ERL | Erlotinib |
| CAP | Capecitabine |
| DOX | Doxorubicin |
| LEU | Leucovorin |
| MIT | Mitomycin |
| CET | Cetuximab |
| BEV | Bevacizumab |
| ABL | Ablifercept |
| SUN | Sunitinib |
| TRA | Tramitinib |
| PIM | Pimasertib |
| dFdCDP | Difluorodeoxycytidine Diphosphate |
| GEM-MOFs | Gemcitabine Loaded Metal Organic Frameworks |
References
- Rawla, P.; Sunkara, T.; Gaduputi, V. Epidemiology of Pancreatic Cancer: Global Trends, Etiology and Risk Factors. World J. Oncol. 2019, 10, 10–27. [Google Scholar] [CrossRef] [PubMed]
- Riva, G.; Pea, A.; Pilati, C.; Fiadone, G.; Lawlor, R.T.; Scarpa, A.; Luchini, C. Histo-molecular oncogenesis of pancreatic cancer: From precancerous lesions to invasive ductal adenocarcinoma. World J. Gastrointest. Oncol. 2018, 10, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J.; Kvederaviciute, K.; Meskinyte, I.; Meskinyte-Kausiliene, E.; Skeberdyte, A.; Cicenas, J. KRAS, TP53, CDKN2A, SMAD4, BRCA1, and BRCA2 Mutations in Pancreatic Cancer. Cancers 2017, 9, 42. [Google Scholar] [CrossRef] [PubMed]
- Rahman, F.A.U.; Ali, S.; Saif, M.W. Update on the role of nanoliposomal irinotecan in the treatment of metastatic pancreatic cancer. Ther. Adv. Gastroenterol. 2017, 10, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, J.; Guo, Y.; Wang, B.; Chu, H. Strategies targeting angiogenesis in advanced non-small cell lung cancer. Oncotarget 2017, 8, 53854–53872. [Google Scholar] [CrossRef] [Green Version]
- Rivera, F.; Benavides, M.; Gallego, J.; Guillen-Ponce, C.; Lopez-Martin, J.; Küng, M. Tumor treating fields in combination with gemcitabine or gemcitabine plus nab-paclitaxel in pancreatic cancer: Results of the PANOVA phase 2 study. Pancreatology 2019, 19, 64–72. [Google Scholar] [CrossRef]
- Plunkett, W.; Huang, P.; Xu, Y.Z.; Heinemann, V.; Grunewald, R.; Gandhi, V. Gemcitabine: Metabolism, mechanisms of action, and self-potentiation. Semin. Oncol. 1995, 22, 3–10. [Google Scholar]
- Heinemann, V.; Quietzsch, D.; Gieseler, F.; Gonnermann, M.; Schönekäs, H.; Rost, A.; Neuhaus, H.; Haag, C.; Clemens, M.; Heinrich, B.; et al. Randomized Phase III Trial of Gemcitabine Plus Cisplatin Compared with Gemcitabine Alone in Advanced Pancreatic Cancer. J. Clin. Oncol. 2006, 24, 3946–3952. [Google Scholar] [CrossRef]
- Huang, P.; Chubb, S.; Hertel, L.W.; Grindey, G.B.; Plunkett, W. Action of 2′,2′-difluorodeoxycytidine on DNA synthesis. Cancer Res. 1991, 51, 6110–6117. [Google Scholar]
- Poplin, E.A.; Corbett, T.; Flaherty, L.; Tarasoff, P.; Redman, B.G.; Valdivieso, M.; Baker, L. Difluorodeoxycytidine (dFdC)—Gemcitabine: A phase I study. Investig. New Drugs 1992, 10, 165–170. [Google Scholar] [CrossRef]
- Abbruzzese, J.L.; Grunewald, R.; Weeks, E.A.; Gravel, D.; Adams, T.; Nowak, B.; Mineishi, S.; Tarassoff, P.; Satterlee, W.; Raber, M.N. A phase I clinical, plasma, and cellular pharmacology study of gemcitabine. J. Clin. Oncol. 1991, 9, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Tonato, M.; Mosconi, A.M.; Martin, C. Safety profile of gemcitabine. Anticancer Drugs 1995, 6 (Suppl. 6), 27–32. [Google Scholar] [CrossRef] [PubMed]
- Celia, C.; Cosco, D.; Paolino, D.; Fresta, M. Gemcitabine-loaded innovative nanocarriers vs GEMZAR: Biodistribution, pharmacokinetic features and in vivo antitumor activity. Expert Opin. Drug Deliv. 2011, 8, 1609–1629. [Google Scholar] [CrossRef] [PubMed]
- Burris, H., III; Moore, M.J.; Andersen, J.; Green, M.R.; Rothenberg, M.L.; Modiano, M.R.; Christine Cripps, M.; Portenoy, R.K.; Storniolo, A.M.; Tarassoff, P.; et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: A randomized trial. J. Clin. Oncol. 1997, 15, 2403–2413. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Yang, J.; Sima, M.; Zhou, Y.; Kopeček, J. Sequential combination therapy of ovarian cancer with degradable N-(2-hydroxypropyl)methacrylamide copolymer paclitaxel and gemcitabine conjugates. Proc. Natl. Acad. Sci. USA 2014, 111, 12181–12186. [Google Scholar] [CrossRef]
- Lammers, T. Improving the efficacy of combined modality anticancer therapy using HPMA copolymer-based nanomedicine formulations. Adv. Drug Deliv. Rev. 2010, 62, 203–230. [Google Scholar] [CrossRef]
- Ciccolini, J.; Serdjebi, C.; Peters, G.J.; Giovannetti, E. Pharmacokinetics and pharmacogenetics of Gemcitabine as a mainstay in adult and pediatric oncology: An EORTC-PAMM perspective. Cancer Chemother. Pharmacol. 2016, 78, 1–12. [Google Scholar] [CrossRef]
- Bornmann, C.; Graeser, R.; Esser, N.; Ziroli, V.; Jantscheff, P.; Keck, T.; Unger, C.; Hopt, U.T.; Adam, U.; Schaechtele, C.; et al. A new liposomal formulation of Gemcitabine is active in an orthotopic mouse model of pancreatic cancer accessible to bioluminescence imaging. Cancer Chemother. Pharmacol. 2008, 61, 395–405. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, Y.; Wu, Y.; Hu, Y.-L.; Nan, K.; Nie, G.; Chen, H. Enhanced anti-tumor efficacy by co-delivery of doxorubicin and paclitaxel with amphiphilic methoxy PEG-PLGA copolymer nanoparticles. Biomaterials 2011, 32, 8281–8290. [Google Scholar] [CrossRef]
- Tan, M.; Luo, J.; Tian, Y. Delivering curcumin and gemcitabine in one nanoparticle platform for colon cancer therapy. RSC Adv. 2014, 4, 61948–61959. [Google Scholar] [CrossRef]
- Haydock, M.M.; Sigdel, S.; Pacioles, T. Gemcitabine-induced skin necrosis. SAGE Open Med. Case Rep. 2018, 6, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Guo, Y.; Duan, Y.; Li, Z.; Wang, C.; Niu, L.; Wang, N.; Ma, M.; Shi, Y.; Zhang, M. Co-delivery of paclitaxel and gemcitabine by methoxy poly(ethylene glycol)-poly(lactide-coglycolide)-polypeptide nanoparticles for effective breast cancer therapy. Anticancer Drugs 2018, 29, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Xiao, H.; Zheng, M.; Qi, R.; Yan, L.; Jing, X. A biodegradable polymer platform for co-delivery of clinically relevant oxaliplatin and gemcitabine. J. Mater. Chem. B 2014, 2, 6560–6570. [Google Scholar] [CrossRef]
- Krishnamurthy, S.; Ng, V.W.L.; Gao, S.; Tan, M.-H.; Hedrick, J.L.; Yang, Y.Y. Codelivery of dual drugs from polymeric micelles for simultaneous targeting of both cancer cells and cancer stem cells. Nanomedicine 2015, 10, 2819–2832. [Google Scholar] [CrossRef]
- Gaumet, M.; Vargas, A.; Gurny, R.; Delie, F. Nanoparticles for drug delivery: The need for precision in reporting particle size parameters. Eur. J. Pharm. Biopharm. 2008, 69, 1–9. [Google Scholar] [CrossRef]
- Blanco, E.; Shen, H.; Ferrari, M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 2015, 33, 941–951. [Google Scholar] [CrossRef]
- Matsumura, Y.; Maeda, H. A new concept for macromolecular therapeutics in cancer chemotherapy: Mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986, 46, 6387–6392. [Google Scholar]
- Maeda, H.; Wu, J.; Sawa, T.; Matsumura, Y.; Hori, K. Tumor vascular permeability and the EPR effect in macromolecular therapeutics: A review. J. Control. Release 2000, 65, 271–284. [Google Scholar] [CrossRef]
- Haley, B.; Frenkel, E. Nanoparticles for drug delivery in cancer treatment. Urol. Oncol. Semin. Orig. Investig. 2008, 26, 57–64. [Google Scholar] [CrossRef]
- Ventola, C.L. Progress in Nanomedicine: Approved and Investigational Nanodrugs. Pharm. Ther. 2017, 42, 742–755. [Google Scholar]
- Yameen, B.; Choi, W.I.; Vilos, C.; Swami, A.; Shi, J.; Farokhzad, O.C. Insight into nanoparticle cellular uptake and intracellular targeting. J. Control. Release 2014, 190, 485–499. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bertrand, N.; Leroux, J.-C. The journey of a drug-carrier in the body: An anatomo-physiological perspective. J. Control. Release 2012, 161, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Karavana, S.Y.; Şenyiğit, Z.A.; Çalışkan, C.; Sevin, G.; Özdemir, D.I.; Erzurumlu, Y.; Sen, S.; Baloğlu, E. Gemcitabine hydrochloride microspheres used for intravesical treatment of superficial bladder cancer: A comprehensive in vitro/ex vivo/in vivo evaluation. Drug Des. Dev. Ther. 2018, 12, 1959–1975. [Google Scholar] [CrossRef] [PubMed]
- Demidenko, Z.N.; Kalurupalle, S.; Hanko, C.; Lim, C.-U.; Broude, E.; Blagosklonny, M.V. Mechanism of G1-like arrest by low concentrations of paclitaxel: Next cell cycle p53-dependent arrest with sub G1 DNA content mediated by prolonged mitosis. Oncogene 2008, 27, 4402–4410. [Google Scholar] [CrossRef]
- Damaghi, M.; Wojtkowiak, J.W.; Gillies, R.J. pH sensing and regulation in cancer. Front. Physiol. 2013, 4, 370. [Google Scholar] [CrossRef] [Green Version]
- Chen, B.; Sun, Q.; Wang, X.; Gao, F.; Dai, Y.; Yin, Y.; Ding, J.; Gao, C.; Cheng, J.; Li, J.; et al. Reversal in multidrug resistance by magnetic nanoparticle of Fe3O4 loaded with adriamycin and tetrandrine in K562/A02 leukemic cells. Int. J. Nanomed. 2008, 3, 277–286. [Google Scholar]
- Urey, C.; Hilmersson, K.S.; Andersson, B.; Ansari, D.; Andersson, R. Development and In Vitro Characterization of a Gemcitabine-Loaded MUC4-Targeted Immunoliposome against Pancreatic Ductal Adenocarcinoma. Anticancer. Res. 2017, 37, 6031–6039. [Google Scholar]
- Oerlemans, C.; Bult, W.; Bos, M.; Storm, G.; Nijsen, J.F.W.; Hennink, W.E. Polymeric micelles in anticancer therapy: Targeting, imaging and triggered release. Pharm. Res. 2010, 27, 2569–2589. [Google Scholar] [CrossRef]
- Liang, T.-J.; Zhou, Z.-M.; Cao, Y.-Q.; Ma, M.-Z.; Wang, X.-J.; Jing, K. Gemcitabine-based polymer-drug conjugate for enhanced anticancer effect in colon cancer. Int. J. Pharm. 2016, 513, 564–571. [Google Scholar] [CrossRef]
- Mondal, G.; Almawash, S.; Chaudhary, A.K.; Mahato, R.I. EGFR-Targeted Cationic Polymeric Mixed Micelles for Codelivery of Gemcitabine and miR-205 for Treating Advanced Pancreatic Cancer. Mol. Pharm. 2017, 14, 3121–3133. [Google Scholar] [CrossRef]
- Zhu, S.; Lansakara-P., D.S.P.; Li, X.; Cui, Z. Lysosomal Delivery of a Lipophilic Gemcitabine Prodrug Using Novel Acid-Sensitive Micelles Improved Its Antitumor Activity. Bioconjug. Chem. 2012, 23, 966–980. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mondal, G.; Kumar, V.; Shukla, S.K.; Singh, P.K.; Mahato, R.I. EGFR-Targeted Polymeric Mixed Micelles Carrying Gemcitabine for Treating Pancreatic Cancer. Biomacromolecules 2016, 17, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Kattel, K.; Mondal, G.; Lin, F.; Kumar, V.; Mahato, R.I. Biodistribution of Self-Assembling Polymer–Gemcitabine Conjugate after Systemic Administration into Orthotopic Pancreatic Tumor Bearing Mice. Mol. Pharm. 2017, 14, 1365–1372. [Google Scholar] [CrossRef] [PubMed]
- Kushwah, V.; Katiyar, S.S.; Agrawal, A.K.; Gupta, R.C.; Jain, S. Co-delivery of docetaxel and gemcitabine using PEGylated self-assembled stealth nanoparticles for improved breast cancer therapy. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 1629–1641. [Google Scholar] [CrossRef]
- Lammers, T.; Subr, V.; Ulbrich, K.; Peschke, P.; Huber, P.E.; Hennink, W.E.; Storm, G. Simultaneous delivery of doxorubicin and gemcitabine to tumors in vivo using prototypic polymeric drug carriers. Biomaterials 2009, 30, 3466–3475. [Google Scholar] [CrossRef]
- Zhao, X.; Li, F.; Li, Y.; Wang, H.; Ren, H.; Chen, J.; Nie, G.; Hao, J. Co-delivery of HIF1α siRNA and gemcitabine via biocompatible lipid-polymer hybrid nanoparticles for effective treatment of pancreatic cancer. Biomaterials 2015, 46, 13–25. [Google Scholar] [CrossRef]
- Neutsch, L.; Wirth, E.-M.; Spijker, S.; Pichl, C.; Kählig, H.; Gabor, F.; Wirth, M. Synergistic targeting/prodrug strategies for intravesical drug delivery—Lectin-modified PLGA microparticles enhance cytotoxicity of stearoyl gemcitabine by contact-dependent transfer. J. Control. Release 2013, 169, 62–72. [Google Scholar] [CrossRef]
- Wang, F.; Wang, Y.; Ma, Q.; Cao, Y.; Yu, B. Development and characterization of folic acid-conjugated chitosan nanoparticles for targeted and controlled delivery of gemcitabinein lung cancer therapeutics. Artif. Cells Nanomed. Biotechnol. 2017, 45, 1530–1538. [Google Scholar] [CrossRef]
- Frese, K.K.; Neesse, A.; Cook, N.; Bapiro, T.E.; Lolkema, M.P.; Jodrell, D.I.; Tuveson, D.A. nab-Paclitaxel potentiates gemcitabine activity by reducing cytidine deaminase levels in a mouse model of pancreatic cancer. Cancer Discov. 2012, 2, 260–269. [Google Scholar] [CrossRef]
- Rodriguez-Ruiz, V.; Maksimenko, A.; Anand, R.; Monti, S.; Agostoni, V.; Couvreur, P.; Lampropoulou, M.; Yannakopoulou, K.; Gref, R.; Patrick, C. Efficient “green” encapsulation of a highly hydrophilic anticancer drug in metal–organic framework nanoparticles. J. Drug Target. 2015, 23, 759–767. [Google Scholar] [CrossRef]
- Aggad, D.; Jimenez, C.M.; Dib, S.; Croissant, J.G.; Lichon, L.; Laurencin, D.; Richeter, S.; Maynadier, M.; Alsaiari, S.K.; Boufatit, M.; et al. Gemcitabine Delivery and Photodynamic Therapy in Cancer Cells via Porphyrin-Ethylene-Based Periodic Mesoporous Organosilica Nanoparticles. ChemNanoMat 2017, 4, 46–51. [Google Scholar] [CrossRef] [Green Version]
- Croissant, J.G.; Zhang, D.; Alsaiari, S.; Lu, J.; Deng, L.; Tamanoi, F.; Almalik, A.M.; Zink, J.I.; Khashab, N.M. Protein-gold clusters-capped mesoporous silica nanoparticles for high drug loading, autonomous gemcitabine/doxorubicin co-delivery, and in-vivo tumor imaging. J. Control. Release 2016, 229, 183–191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meng, H.; Wang, M.; Liu, H.; Liu, X.; Situ, A.; Wu, B.; Ji, Z.; Chang, C.H.; Nel, A.E. Use of a Lipid-Coated Mesoporous Silica Nanoparticle Platform for Synergistic Gemcitabine and Paclitaxel Delivery to Human Pancreatic Cancer in Mice. ACS Nano 2015, 9, 3540–3557. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, C.; Zhang, G.; Liu, G.; Hu, J.; Liu, S. Photo- and thermo-responsive multicompartment hydrogels for synergistic delivery of gemcitabine and doxorubicin. J. Control. Release 2017, 259, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Allen, T.M.; Cullis, P.R. Liposomal drug delivery systems: From concept to clinical applications. Adv. Drug Deliv. Rev. 2013, 65, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Laouini, A.; Jaafar-Maalej, C.; Limayem-Blouza, I.; Sfar, S.; Charcosset, C.; Fessi, H. Preparation, Characterization and Applications of Liposomes: State of the Art. J. Colloid Sci. Biotechnol. 2012, 1, 147–168. [Google Scholar] [CrossRef]
- Chen, N.; Li, Y.; Ye, Y.; Palmisano, M.; Chopra, R.; Zhou, S. Pharmacokinetics and pharmacodynamics of nab-paclitaxel in patients with solid tumors: Disposition kinetics and pharmacology distinct from solvent-based paclitaxel. J. Clin. Pharmacol. 2014, 54, 1097–1107. [Google Scholar] [CrossRef]
- Moen, M.D.; Wellington, K. Gemcitabine. Am. J. Cancer 2005, 4, 327–333. [Google Scholar] [CrossRef]
- Liu, D.; Bimbo, L.M.; Mäkilä, E.; Villanova, F.; Kaasalainen, M.; Herranz-Blanco, B.; Caramella, C.M.; Lehto, V.-P.; Salonen, J.; Herzig, K.-H.; et al. Co-delivery of a hydrophobic small molecule and a hydrophilic peptide by porous silicon nanoparticles. J. Control. Release 2013, 170, 268–278. [Google Scholar] [CrossRef]
- Liu, Y.; Tamam, H.; Yeo, Y. Mixed Liposome Approach for Ratiometric and Sequential Delivery of Paclitaxel and Gemcitabine. AAPS PharmSciTech 2018, 19, 693–699. [Google Scholar] [CrossRef]
- Gallo, D.; Fruscella, E.; Ferlini, C.; Apollonio, P.; Mancuso, S.; Scambia, G. Preclinical in vivo activity of a combination gemcitabine/liposomal doxorubicin against cisplatin-resistant human ovarian cancer (A2780/CDDP). Int. J. Gynecol. Cancer 2006, 16, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Gaber, M.H.; Hong, K.; Huang, S.K.; Papahadjopoulos, D. Thermosensitive Sterically Stabilized Liposomes: Formulation and in Vitro Studies on Mechanism of Doxorubicin Release by Bovine Serum and Human Plasma. Pharm. Res. 1995, 12, 1407–1416. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Ru, Y.; Gao, Y.; Li, J.; Mao, S. Layer-by-layer nanoparticles co-loading gemcitabine and platinum(IV) prodrugs for synergistic combination therapy of lung cancer. Drug Des. Devel. Ther. 2017, 11, 2631–2642. [Google Scholar] [CrossRef] [PubMed]
- Emamzadeh, M.; Pasparakis, G. Dual Controlled Delivery of Gemcitabine and Cisplatin Using Polymer-Modified Thermosensitive Liposomes for Pancreatic Cancer. ACS Appl. Bio Mater. 2019, 2, 1298–1309. [Google Scholar] [CrossRef]
- Sercombe, L.; Veerati, T.; Moheimani, F.; Wu, S.Y.; Sood, A.K.; Hua, S. Advances and Challenges of Liposome Assisted Drug Delivery. Front. Pharmacol. 2015, 6, 286. [Google Scholar] [CrossRef] [Green Version]
- Jani, R.K.; Gohil, K.M. Liposomal Formulations in Cancer Therapy: Basic Concepts to Advanced Strategies. Int. J. Pharm. Sci. Drug Res. 2018, 10, 386–393. [Google Scholar] [CrossRef]
- Qian, K.; Qian, H.; Cai, J.; Yue, W.; Yu, X.; Liu, B. Evaluation of cisplatin-hydrogel for improving localized antitumor efficacy in gastric cancer. Pathol. Res. Pract. 2019, 215, 755–760. [Google Scholar] [CrossRef]
- Bastiancich, C.; Bozzato, E.; Luyten, U.; Danhier, F.; Bastiat, G.; Préat, V. Drug combination using an injectable nanomedicine hydrogel for glioblastoma treatment. Int. J. Pharm. 2019, 559, 220–227. [Google Scholar] [CrossRef]
- Li, L.; Neaves, W.B. Normal Stem Cells and Cancer Stem Cells: The Niche Matters. Cancer Res. 2006, 66, 4553–4557. [Google Scholar] [CrossRef]
- Berstein, L.M. Modern approach to metabolic rehabilitation of cancer patients: Biguanides (phenformin and metformin) and beyond. Future Oncol. 2010, 6, 1313–1323. [Google Scholar] [CrossRef]
- Hui, Y.F.; Reitz, J. Gemcitabine: A cytidine analogue active against solid tumors. Am. J. Health Pharm. 1997, 54, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Jain, K.K. Nanobiotechnology-based strategies for crossing the blood–brain barrier. Nanomedicine (London) 2012, 7, 1225–1233. [Google Scholar] [CrossRef] [PubMed]
- Gao, H. Perspectives on Dual Targeting Delivery Systems for Brain Tumors. J. Neuroimmune Pharmacol. 2017, 12, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Li, W.; Chen, T.; Yang, Q.; Huang, T.; Fu, Y.; Gong, T.; Zhang, Z. Hyaluronic Acid-Modified Micelles Encapsulating Gem-C12 and HNK for Glioblastoma Multiforme Chemotherapy. Mol. Pharm. 2018, 15, 1203–1214. [Google Scholar] [CrossRef]
- Tanbour, R.; Martins, A.M.; Pitt, W.G.; A Husseini, G. Drug Delivery Systems Based on Polymeric Micelles and Ultrasound: A Review. Curr. Pharm. Des. 2016, 22, 2796–2807. [Google Scholar] [CrossRef]
- Von Hoff, D.D.; Ramanathan, R.K.; Borad, M.J.; Laheru, D.A.; Smith, L.S.; Wood, T.E.; Korn, R.L.; Desai, N.; Trieu, V.; Iglesias, J.L.; et al. Gemcitabine plus nab-paclitaxel is an active regimen in patients with advanced pancreatic cancer: A phase I/II trial. J. Clin. Oncol. 2011, 29, 4548–4554. [Google Scholar] [CrossRef]
- Von Hoff, D.D.; Ervin, T.; Arena, F.P.; Chiorean, E.G.; Infante, J.; Moore, M.; Seay, T.; Tjulandin, S.A.; Ma, W.W.; Saleh, M.N.; et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N. Engl. J. Med. 2013, 369, 1691–1703. [Google Scholar] [CrossRef]
- Roesch, M.; Mueller-Huebenthal, B. Review: The role of hyperthermia in treating pancreatic tumors. Indian J. Surg. Oncol. 2015, 6, 75–81. [Google Scholar] [CrossRef]
- Diagaradjane, P.; Shetty, A.; Wang, J.C.; Elliott, A.M.; Schwartz, J.; Shentu, S.; Park, H.C.; Deorukhkar, A.; Stafford, R.J.; Cho, S.H.; et al. Modulation of in Vivo Tumor Radiation Response via Gold Nanoshell-Mediated Vascular-Focused Hyperthermia: Characterizing an Integrated Antihypoxic and Localized Vascular Disrupting Targeting Strategy. Nano Lett. 2008, 8, 1492–1500. [Google Scholar] [CrossRef] [Green Version]
- Kirui, D.K.; Celia, C.; Molinaro, R.; Bansal, S.S.; Cosco, D.; Fresta, M.; Shen, H.; Ferrari, M. Mild hyperthermia enhances transport of liposomal gemcitabine and improves in vivo therapeutic response. Adv. Health Mater. 2015, 4, 1092–1103. [Google Scholar] [CrossRef]
- Noble, S.; Goa, K.L. Gemcitabine. A review of its pharmacology and clinical potential in non-small cell lung cancer and pancreatic cancer. Drugs 1997, 54, 447–472. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.A.; Gadde, S.; Pfirschke, C.; Engblom, C.; Sprachman, M.M.; Kohler, R.H.; Yang, K.S.; Laughney, A.M.; Wojtkiewicz, G.; Kamaly, N.; et al. Predicting therapeutic nanomedicine efficacy using a companion magnetic resonance imaging nanoparticle. Sci. Transl. Med. 2015, 7, 314ra183. [Google Scholar] [CrossRef] [PubMed]
- Caravan, P.; Ellison, J.J.; McMurry, T.J.; Lauffer, R.B. Gadolinium(III) Chelates as MRI Contrast Agents: Structure, Dynamics, and Applications. Chem. Rev. 1999, 99, 2293–2352. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Tong, R.; Li, M.; Kohane, D.S. Self-assembled gemcitabine-gadolinium nanoparticles for magnetic resonance imaging and cancer therapy. Acta Biomater. 2016, 33, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Sanpui, P.; Paul, A.; Chattopadhyay, A. Theranostic potential of gold nanoparticle-protein agglomerates. Nanoscale 2015, 7, 18411–18423. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Park, J.; Ju, E.J.; Park, S.S.; Choi, J.; Lee, J.H.; Lee, K.J.; Shin, S.H.; Ko, E.J.; Park, I.; et al. Multifunctional hollow gold nanoparticles designed for triple combination therapy and CT imaging. J. Control. Release 2015, 207, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Kantoff, P.W.; Wooster, R.; Farokhzad, O.C. Cancer nanomedicine: Progress, challenges and opportunities. Nat. Rev. Cancer 2017, 17, 20–37. [Google Scholar] [CrossRef]
- Poon, C.; He, C.; Liu, D.; Lu, K.; Lin, W. Self-assembled nanoscale coordination polymers carrying oxaliplatin and gemcitabine for synergistic combination therapy of pancreatic cancer. J. Control. Release 2015, 201, 90–99. [Google Scholar] [CrossRef] [Green Version]
- Terada, T.; Ohta, T.; Sasaki, M.; Nakanuma, Y.; Kim, Y.S. Expression of MUC apomucins in normal pancreas and pancreatic tumours. J. Pathol. 1996, 180, 160–165. [Google Scholar] [CrossRef]
- Chauhan, S.C.; Ebeling, M.C.; Maher, D.M.; Koch, M.D.; Watanabe, A.; Aburatani, H.; Lio, Y.; Jaggi, M. MUC13 mucin augments pancreatic tumorigenesis. Mol. Cancer Ther. 2012, 11, 24–33. [Google Scholar] [CrossRef]
- Bafna, S.; Kaur, S.; Momi, N.; Batra, S.K. Pancreatic cancer cells resistance to gemcitabine: The role of MUC4 mucin. Br. J. Cancer 2009, 101, 1155–1161. [Google Scholar] [CrossRef] [PubMed]
- Noh, I.; Kim, H.-O.; Choi, J.; Choi, Y.; Lee, D.K.; Huh, Y.-M.; Haam, S. Co-delivery of paclitaxel and gemcitabine via CD44-targeting nanocarriers as a prodrug with synergistic antitumor activity against human biliary cancer. Biomaterials 2015, 53, 763–774. [Google Scholar] [CrossRef] [PubMed]
- Patra, C.R.; Bhattacharya, R.; Wang, E.; Katarya, A.; Lau, J.S.; Dutta, S.; Muders, M.; Wang, S.; Buhrow, S.A.; Safgren, S.L.; et al. Targeted delivery of gemcitabine to pancreatic adenocarcinoma using cetuximab as a targeting agent. Cancer Res. 2008, 68, 1970–1978. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Chen, Y.; Zeng, L.; Lian, G.; Chen, S.; Li, Y.; Yang, K.; Huang, K.; Jiajia, L.; Yinting, C.; et al. A Nanoparticle Carrier for Co-Delivery of Gemcitabine and Small Interfering RNA in Pancreatic Cancer Therapy. J. Biomed. Nanotechnol. 2016, 12, 1654–1666. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.Y.; Qian, W.P.; Wang, L.; Wang, Y.A.; Staley, C.A.; Satpathy, M.; Nie, S.; Mao, H.; Yang, L. Theranostic Nanoparticles with Controlled Release of Gemcitabine for Targeted Therapy and MRI of Pancreatic Cancer. ACS Nano 2013, 7, 2078–2089. [Google Scholar] [CrossRef] [PubMed]
- Viota, J.L.; Carazo, A.; Munoz-Gamez, J.A.; Rudzka, K.; Gómez-Sotomayor, R.; Ruiz-Extremera, A.; Salmerón, J.; Delgado, A.V. Functionalized magnetic nanoparticles as vehicles for the delivery of the antitumor drug gemcitabine to tumor cells. Physicochemical in vitro evaluation. Mater. Sci. Eng. C 2013, 33, 1183–1192. [Google Scholar] [CrossRef]
- Meng, H.; Zhao, Y.; Dong, J.; Xue, M.; Lin, Y.-S.; Ji, Z.; Mai, W.X.; Zhang, H.; Chang, C.H.; Brinker, C.J.; et al. Two-Wave Nanotherapy to Target the Stroma and Optimize Gemcitabine Delivery to a Human Pancreatic Cancer Model in Mice. ACS Nano 2013, 7, 10048–10065. [Google Scholar] [CrossRef]
- Zhang, Y.; Schwerbrock, N.M.; Rogers, A.B.; Kim, W.Y.; Huang, L. Codelivery of VEGF siRNA and Gemcitabine Monophosphate in a Single Nanoparticle Formulation for Effective Treatment of NSCLC. Mol. Ther. 2013, 21, 1559–1569. [Google Scholar] [CrossRef] [Green Version]


| Nano-Systems | Drug Conjugate | Characterization | Utilization |
|---|---|---|---|
| (Micelles) GEM-PL | GEM-methoxy poly(ethylene glycol)-poly(lactic acid) (GEM-PL) | Particle size 112.2 ± 1.86 nm, zeta potential, 5.2 ± 1.26, encapsulation efficiency 92.5 ± 3.26 loading efficiency of 14.6 ± 1.29 and PDI(Poly dispersity index) 0.118. | In-vitro study was done using human colon cancer cell line HT29 and in-vivo studies for anticancer efficacy was done by injecting HT29 (5 × 106 cells) subcutaneously into right flank per mice and when tumor volume reached 100 mm3, 5 mg/kg fixed drug dose was administered 3 times for 12 days. In-vivo studies depicted improved blood circulation time with greater accumulation of the drug in tumor site with significant tumor regression. Formulation killed HT29 cells at a time dependent manner [39]. This study provides a valid reason required for further clinical research. |
| (Micelles) C225—micelle-GEM-miR-205 | GEM-miR205-EGFR targeting Cetuximab antibody (C225) | Unmodified micelles containing GEM and miR-205 had the size of 76.6 ± 6 nm and zeta poteintial of 4.7 ± 1.65 mV. C225 conjugation was checked with standard BSA which was found to be 510 ug/mL C255 micelle 30% w/w for 10 gm polymer. | In-vitro studies were conducted using MIA PaCa-2 and in-vivo studies such as biodistribution and efficacy studies were done on NSC mice using an orthotopic tumor model where mice were implanted with GFP-transfected MIA PaCa-2 cells. In-vivo studies were conducted using a orthotopic pancreatic tumor model in 6 week old NSG mice injected with GFP transfected MIA PACA-2 cells [40]. |
| (Micelles) PHC-GEMC18 | poly(ethylene glycol) (PEG) conjugated hydrophobic stearic acid derivative (C18) bonded through acid sensitive hydrazine bond-GEM | PHC size of 21.6 ± 0.6 nm zeta of 3.3 ± 1.1 mV PHC3%(GEM) size of 9.6 ± 3.7 nm zeta of 2.7 ± 1.2 mV PHC5%(GEM) size of 5.0 ± 0.1 nm zeta of −2.0 ± 0.7 mV. PHC10%(GEM) size of 9.7 ± 1.3 nm zeta of −2.2 ± 0.9 mV. | B16-F10 tumor was grown on C57BL/6 mice by subcutaneous inoculation in the right flank followed by treatment after day 6 of inoculation. As compared to GEM alone which is because of the increase in GEM18 accumulation [41]. |
| (Micelle) GE11-PEG-PCD/mPEG-b-PCC-g-GEM-g-DC | G11 peptide of GEM conjugated with poly (ethylene glycol)-block-poly (2-methyl-2-carboxyl-propylenr carbonate-graft-Gemcitabine-graft-dodecanol micelle | Particle size of 26 ± 3 nm with a polydispersion index of 0.27 [42]. | MIA PaCa-2 cells have been used for the in-vivo studies and 6–8-week-old athymic nude mice were used for a orthotopic pancreatic cancer model for studying efficacy of the micelles after intraperitoneal injection of D-luciferin for bioluminescence studies. Increase in GEM delivery was seen up to 2.5 folds with an enhanced circulation half-life and EPR effect facilitating extravasation of micelle loaded drugs within the tumor microenvironment [43]. |
| (Micelle) DTX-PEG-GEM | Docetaxel-Polyethylene-glycol-GEM | Particle size of 124.2 ± 5.7 nm and PDI of 0.132 ± 0.03 with critial micelle concentration range of 5–10 × 10−3 mg/mL. | In-vitro studies were done using MCF-7 and MDA-MB-231 for checking cellular internalization and uptake, In-vivo study used female Sprague dolly rats for carrying forward the pharmacokinetic and toxicity studies. Clathrin mediated endocytosis with 4.8 fold higher AUC value as compared to Gemzar alone was observed with a noteworthy decrease in tumor volume, increase in total survival, and reduction in hepatic, nephron, and hemolytic toxicity when administered with DTX-PEG-GEM nanoparticles [44]. |
| (Micelle) P-GEM-DOX | Poly(HPMA-co-MA-GFLG-GEM co-MA-GFLG-DOX-co-MA-TyrNH2) | Molecular weight = 23.5 kDa, PD = 1.6, GEM = 6.4 wt %, Dox = 5.7 wt %, tyrNH2 = 1.0 mol %. | Dunning AT1rat prostate carcinoma cells were used for the in-vitro studies which involved drug release, cytotoxicity, and efficacy study of the formulation. In-vivo studies were conducted on male Copenhagen rats present with subcutaneous tumor. Enhanced circulation time with selectivity and localization at tumor-specific sites was seen with induction of apoptosis and inhibition of angiogenesis. In-vivo efficacy of P-GEM-DOX < free GEM, though co-conjugation enhanced in vitro efficacy [45]. |
| (Micelle) GEM-LEMPs-DNA | PEGylated lipid bilayer cationic ε poly lysine co-polymer with GEM-(si-HIF1α) | Particle size of 60 nm with a hydrodynamic diameter according to DLS study, size was 141.8 nm zeta potential of GEM-LEMP-DNA was −34 mV, encapsulation efficiency of 42%. | Serum stability, cytotoxicity, PCR, and immunohistology studies were done using Panc-1 and B-16 melanoma cells. The in-vivo studies were conducted on female BALB/c mice to check the antitumor activity of the formulation. Formulation caused effecting silencing of HIF1α via siRNA and reduced drug-related resistance. The lipid layer protects si-HIF1 α from degradation thereby maintaining the integrity of the particle and preventing leakage of GEM [46]. |
| (Micelle) GEM-C18-PLGA-MP | Stearoyl GEM incorporated within PLGA with surface functionalization with human serum albumin | GEMC18 content was found to be 488.9 ± 35.7 μg/mL with PLGA content of 1.71 ± 0.21 mg/mL corresponding to 285 ± 56 μg/mg or 28.6% ± 5.6% (w/w) of particle matrix, prodrug release of 13.2% ± 1.8% was seen after 5 days incubation. | SV-HUCC-1 normal urinary bladder cell was considered as control and urothelial cancer cell line 5637 and HT-1376 were used for a cell viability assay, studies for determining metabolic activity and for checking the cyto adhesive properties of formulation. GEMC18, when conjugated with PLGA microparticle, avoided intracellular drug activation thereby maintaining drug stability and covalent modification of the polymer with human serum albumin, led towards the enhanced binding capacity of the formulation with urothelial cells [47]. In-vivo studies using an animal model is still a requirement to confirm the pharmacokinetics of the formulation. |
| (Micelle) FA-PEG-GEM-NPs | Folic acid conjugated GEM loaded surface modified chitosan nanoparticle | Particle size determined to be 184.3 ± 12.47 nm with a PDI of 0.22 ± 0.07 zeta potential of 21.1 ± 1.18 mV and encapsulation efficiency of 37.2% ± 2.2%. | In-vivo cytotoxic studies were done using lung epithelial cancer cell line A549 for a cytotoxic assay, drug release and cellular uptake. Balb/c mice were used to conduct the pharmacokinetic study. Significant cytotoxicity showed while GEM being delivered through nano formulation when treated to A549 cells showed more effective cellular internalization than free GEM [48]. |
| (Metal-based) IONPs | GEM-siRNA-iron-oxide nanoparticles | Particle size of 80 nm. | Iron oxide was profoundly conjugated with CD44v6 targeted PanCa and GEM-siRNA conjugation with siBmi-1 oncogene to give multifunctional nanoparticle scFv-GEM-siBmi-1-NPs an in vivo anti-tumor synergistic activity [49]. |
| (Metal-based) MIL-100 Nano-MOFs | Metal-organic framework of iron III trimesate nanoparticles-phosphate GEM | Encapsulation efficiency of phosphated GEM = 30.7% ± 0.8% which was almost 98%. | GEM-MP loaded NanoMOFs were studied on PANC-1 cells in a phosphate devoid medium with 50% of encapsulated drug released within 1 min after administration which stayed for 20 h [50]. |
| (Metal-based) PS1-EPSMOs-GEM | Tetrasilylated porphyrin-ethylene periodic mesoporous organosilica nanoparticles | PS1-EPSMOs mean diameter (TEM) = 447 nm, zeta at pH 5.5 = −30 mV; zeta at pH 7.4 = −34 mV. | In-vivo delivery of drug was done on MCF-7 breast cancer cells. The porous structure provided high loading capacity and the addition of the porphyrin group provided photosensitivity to the nanoparticle [51]. In-vivo studies are yet to be done for confirming the significance of this formulation. |
| (Metal-based) AuNC@BSA-MSN-GEM-DOX | Gold nanocluster bovine serum albumin clustered with mesoporous silica added with 32% GEM and DOX combined with albumin, attached electrostatically to formulation | Nanoparticle size = 150 nm, gold-protein conjugate zeta potential of −38 ± 1 mV for MSN-AuNC@BSA + DOX + GEM with AuNC content of 2.10 ± 0.23 and BSA 15.90 ± 1.80. | In-vitro study for the formulation was done using A549 lung cancer cells and biodistribution of formulation was seen in nude mice being previously injected subcutaneously with MIA-PaCa-2. Dual loading of GEM + DOX was 72 wt % which was four times higher than previous reports with less than 4% leakage of the loaded drug after a week in blood serum [52]. |
| (Metal-based) PTX-GEM-LB-MSNNP | Lipid layer mesoporous-silica nanoparticle loaded with PTX and GEM | Hydrodynamic partilc size of 101 nm in saline and 112 nm with zeta potential of of −27.2 mV and −5.4 mV in saline plus 5% serum condition. | Cytotoxicity study, expression of cytidine deaminase and heme oxygenase via Western blot was done using PanC-1 cells and for the in-vitro studies, these cells were transfected with luciferase and implanted to grow subcutaneously within xenograft nude mice. Co-delivery of a dual drug caused enhanced phosphorylation with an increase in DNA-GEM interaction up to 13 fold and decreasing inactivated deaminated metabolite up to 4 folds producing synergistic codelivery of GEM and PTX [53]. |
| (Metal base) GEM-Au DENPs/miR-21-inh | Ultrasound targeted microbubble-based dendrimer entrapped gold particle-Gemcitabne-miR-21 inhibitor | Mean particle size obtained was 154–276 nm with a surface charge range of 11–33 mV. | SW1990 cells were used to check the effect of the formulation on cytotoxicity, female athymic Balb/c mice were used to check the antitumor activity of the formulation. Uptake and apoptosis. The apoptosis percent of GEM–Au DENPs/miR-21i group (20.87% ± 0.81%) and GEM–Au DENPs/miR-21i + U group (25.43% ± 0.60%) which came up to be much more than the free GEM group (10.50% ± 0.56%) [40]. |
| (Hydrogel) PNIPAM-b-PNAM-b-PNBOC | poly(N-isopropylacrylamide)-b-poly (4-acryloyl morpholine)-b-poly(2-(2-nitrobenzyl)oxy) carbonyl) amino)ethyl methacrylate)-GEM-Doxorubicin | Average hydrodynamic diameter was determined to be 68 nm. TEM observation gave spherical nanoparticles. | Extra micellar aqueous phase and the hydrophobic micellar core of formulation helped in the incorporation of a hydrophilic and hydrophobic drug [54]. This work showed the synthesis and characterization of formulation which needs follow up with in-vivo and in-vitro studies. |
| Formulation | Target/Ligand | Process of Conjugation | Outcome |
|---|---|---|---|
| MPDNCs Poly(l-lysine) carboxylate PTX(PLL-PTX) + Hyaluronic acid conjugated GEM | CD44 | Electrostatic attraction between PLL-PTX and conjugation between HA-Gem through hydrolysable linkers. | Biliary cancer cells HuCCT1 and SCK have been used to check the targeting efficiency and therapeutic efficacy of the formulation. Xenograft Balb/c nude mice were used for checking in vivo drug efficacy. Cellular uptake of MPDNCs induced synergistic apoptosis [92]. |
| GEM-AuNPs-C225 | EGFR | Incubation of AuNPs with 2 μg/mL C225 for 1 h at pH 7.8 proceeded by 1 h incubation with GEM 5 μg/mL. | Screening of PanCa cells such as PANC-1, AsPC-1, and MIA PACA-2 with EGFR expression enhanced the targeting efficiency of formulation with a significant reduction in cell proliferation and tumor growth in orthotopic nude mice injected with GFP transfected AsPC-1 cells [93]. |
| EM-gold nanoparticle | Plectin-1 | Pyrimidine group within GEM provides free NH2 group where Gem has an ability to bind with the gold nanoparticle via electrostatic force of interaction. | Surface modulated GNPs with peptides used for plectin 1 targeting and conjugation with GEM showed higher cytotoxicity in AsPC-1 and PANC-1 cell lines with a significant in vivo antitumor efficacy of formulation when given via tail vein in female Balb/c mice with xenografted pancreatic tumor [94]. |
| ATF-IONP-GEM (Amino terminal fragment-iron oxide nanoparticle-GEM) | Urokinase plasminogen activator receptor | Iron oxide nanoparticle are conjugated with amino terminal fragment peptide of uPA receptor domain through lysosomal cleavable tetra peptide linker. | In vitro studies to check drug cytotoxicity, drug targeting in PanCa cells was done using MIA PaCa-2. The anti-tumor activity was checked in-vivo with the help of MIA-PaCa-2 implanted xenograft model in nude mice. Drug dose given twice weekly. Endocytosis through receptor-mediated approach enabled the release of GEM within the cells which helped in intensifying MRI of the tumor and the presence of lysosomal cleavable bonds prevented the formulation from enzyme-based degradation [95]. |
| GEM-Chitosan-Carbopol-MNPs | Folate receptor | Surface conjugation of Poly acrylic acid polymer with chitosan forming multilayer shell with a surface conjugation with folic acid. | PLC-PRF-5, DLD-1, and MDA-231 cell line respective to different cancers were used to carry on the in-vitro studies to depict cytotoxic activity of the formulation. Additional studies were also conducted to check for folate expression within cell. Targeting of folate receptor increased the chance of particles to surpass the cell membrane and make GEM available at tumor sites [96]. This study requires animal work for further validation of the formulation. |
| Gem targeted TGFβi-MSNP | β-Kinase receptor | Co-precipitation method involved. PEI coating above the MSNP provide great number of non-complex hydrogens which gets attached to the nitrogen atom present in TGF B inhibitor LY364947 via hydrogen bonding. | Endothelial cells, human microvascular endothelial cells and human smooth muscle cells were used to mimic stromal environment and BxPC3 cancer cells were used for in-vitro studies and for implanting mice xenograft. Tumor bearing mice were injected IV. Formulation decreased vascular pericyte coverage by inhibiting TGF β pathway which is caused by LY364947 group of the formulation thereby enhancing efficient uptake of GEM-based IV liposomal formulation coming up as a two-way drug delivery approach [97]. |
| GEM-C18-PEG-DSPE/TPGS | EGF-receptor | Stearic acid conjugated with GEM further been incorporated within PEG-DSPE/TPGS micelles where GEMC18 loaded micelles where prepared using solvent evaporation. | Human BxPC-3 were used to check the proliferation and cellular uptake of the formulation. Antitumor and pharmacokinetic studies were performed on mice injected with BxPC-3 intraperitoneally in the right flank. Formulation avoided GEM deamination which was noticed in free GEM leading towards enhanced GEM circulation time and 3-fold increased GEM concentration in tumor cells [96]. |
| (GMP + VEGF)-LCP-AA | Sigma receptor | Phosphate group present in GEM interacts with calcium during the preparation of micro emulsion leading towards encapsulation of GEM and VEGF siRNA with a surface modification with PEG which increases particle retaining time within body. | 30–40% greater tumor inhibition with 8-fold reduced proliferation and decreased tumor microvessel density as compared to alone VEGF and GEM treatment was observed in H460 tumor induced mice with treatment given as IV injection, in vitro cytotoxic studies conducted using H460 non-small lung cancer cell. Multiple nucleic acid incorporation and targeting of sigma receptors found extensively on overexpressing cells [98]. |
| Folic acid conjugated GEM loaded chitosan nanoparticle | Folate receptor | Normal conjugation and centrifugation process were utilized in synthesis of FA-conjugated and PEGylated GEM-NPs. | Significant cytotoxicity showed while GEM being delivered through nanoformulation when treated to A549 cells showed effective cellular internalization than free GEM. Balb/c mice were used to conduct the pharmacokinetic study with formulation injected through lateral tail vein [48]. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samanta, K.; Setua, S.; Kumari, S.; Jaggi, M.; Yallapu, M.M.; Chauhan, S.C. Gemcitabine Combination Nano Therapies for Pancreatic Cancer. Pharmaceutics 2019, 11, 574. https://doi.org/10.3390/pharmaceutics11110574
Samanta K, Setua S, Kumari S, Jaggi M, Yallapu MM, Chauhan SC. Gemcitabine Combination Nano Therapies for Pancreatic Cancer. Pharmaceutics. 2019; 11(11):574. https://doi.org/10.3390/pharmaceutics11110574
Chicago/Turabian StyleSamanta, Kamalika, Saini Setua, Sonam Kumari, Meena Jaggi, Murali M. Yallapu, and Subhash C. Chauhan. 2019. "Gemcitabine Combination Nano Therapies for Pancreatic Cancer" Pharmaceutics 11, no. 11: 574. https://doi.org/10.3390/pharmaceutics11110574







