Transcellular Transport of Heparin-coated Magnetic Iron Oxide Nanoparticles (Hep-MION) Under the Influence of an Applied Magnetic Field
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Synthesis of the Hep-MION
2.3. Physicochemical characterization of the Hep-MION
2.4. Transport of the Hep-MION across the polyester membrane

2.5. Transport of the Hep-MION across the cell monolayer
3. Results and Discussion
3.1. Physicochemical characterization of the Hep-MION nanoparticles

3.2. Stability of the Hep-MION nanoparticle dispersions

| Buffer | Water | HBSS + 10% FBS | HBSS + 1% FBS | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Incubation Time (h) | 0 h | 5 h | 24 h | 0 h | 5 h | 24 h | 0 h | 5 h | 24 h |
| Average Size (nm) | 61.37 | 57.93 | 59.50 | 75.47 | 76.70 | 76.37 | 1053.27 | 3515.53 | 17659.43 |
| S.D. (nm) | 6.07 | 9.10 | 6.38 | 6.31 | 7.33 | 7.46 | 66.82 | 260.23 | 1243.76 |
3.3. Transport of the Hep-MION across porous membranes
| Concentration of Hep-MION | HBSS with 10% FBS | HBSS with 1% FBS | ||
|---|---|---|---|---|
| Magnet (-) | Magnet (+) | Magnet (-) | Magnet (+) | |
| Peff (10-3 cm/sec) | Peff (10-3 cm/sec) | |||
| 0.206 mg/mL | 3.19 (0.425) | 7.21 (0.53) | 3.04 (0.449) | 6.77 (1.53) |
| 0.2575 mg/mL | 5.73 (0.272) | 8.47 (0.408) | 2.67 (0.544) | 6.67 (0.679) |
| 0.412 mg/mL | 6.72 (0.17) | 7.85 (0.849) | 2.45 (0.425) | 5.88 (0.736) |
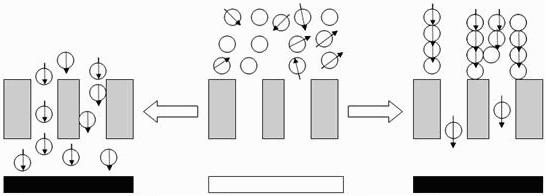
3.4. Transport of the Hep-MION across cell monolayers promoted by a magnetic field

| Peff (10-6 cm/sec) | dM/dt (10-7 mg/sec) | |||||||
|---|---|---|---|---|---|---|---|---|
| Concentration (mg/mL) | 0.2575 | 0.412 | 0.2575 | 0.412 | ||||
| Magnet | - | + | - | + | - | + | - | + |
| Average | 4.2 | 24 | 0.9 | 6.6 | 3.5 | 20 | 1.2 | 9 |
| S.D. | 0.8 | 3.7 | 0.5 | 0.7 | 0.7 | 3.2 | 0.7 | 1.0 |
| p-value | 5.81 × 10-3 | 3.94 × 10-4 | 5.81 × 10-3 | 3.94 × 10-4 | ||||

3.5. Accumulation of Hep-MION on cell monolayers induced by a magnetic field


4. Conclusions
Acknowledgements
References and Notes
- Bulte, J.W.M.; Douglas, T.; Witwer, B.; Zhang, S.C.; Strable, E.; Lewis, B.K.; Zywicke, H.; Miller, B.; van Gelderen, P.; Moskowitz, B.M.; Duncan, I.D.; Frank, J.A. Magnetodendrimers allow endosomal magnetic labeling and in vivo tracking of stem cells. Nat. Biotechnol. 2001, 19, 1141–1147. [Google Scholar] [CrossRef]
- Olsvik, O.; Popovic, T.; Skjerve, E.; Cudjoe, K.S.; Hornes, E.; Ugelstad, J.; Uhlen, M. Magnetic Separation Techniques in Diagnostic Microbiology. Clin. Microbiol. Rev. 1994, 7, 43–54. [Google Scholar]
- Yakub, G.P.; Stadterman-Knauer, K.L. Immunomagnetic separation of pathogenic organisms from environmental matrices. Methods Mol. Biol. 2004, 268, 189–197. [Google Scholar]
- Alexiou, C.; Arnold, W.; Klein, R.J.; Parak, F.G.; Hulin, P.; Bergemann, C.; Erhardt, W.; Wagenpfeil, S.; Lubbe, A.S. Locoregional cancer treatment with magnetic drug targeting. Cancer Res. 2000, 60, 6641–6648. [Google Scholar]
- Dobson, J. Gene therapy progress and prospects: magnetic nanoparticle-based gene delivery. Gene Ther. 2006, 13, 283–287. [Google Scholar] [CrossRef]
- McBain, S.C.; Griesenbach, U.; Xenariou, S.; Keramane, A.; Batich, C.D.; Alton, E.W.F.W.; Dobson, J. Magnetic nanoparticles as gene delivery agents: enhanced transfection in the presence of oscillating magnet arrays. Nanotechnology 2008, 19, 405102–405106. [Google Scholar]
- Moroz, P.; Jones, S.K.; Gray, B.N. The effect of tumour size on ferromagnetic embolization hyperthermia in a rabbit liver tumour model. Int. J. Hyperther. 2002, 18, 129–140. [Google Scholar] [CrossRef]
- Bulte, J.W.M. Magnetic nanoparticles as markers for cellular MR imaging. J. Magn. Magn. Mater. 2005, 289, 423–427. [Google Scholar] [CrossRef]
- Ferguson, R.M.; Minard, K.R.; Krishnan, K.M. Optimization of nanoparticle core size for magnetic particle imaging. J. Magn. Magn. Mater. 2009, 321, 1548–1551. [Google Scholar] [CrossRef]
- Wang, Y.X.J.; Hussain, S.M.; Krestin, G.P. Superparamagnetic iron oxide contrast agents: physicochemical characteristics and applications in MR imaging. Eur. Radiol. 2001, 11, 2319–2331. [Google Scholar] [CrossRef]
- Mondalek, F.G.; Zhang, Y.Y.; Kropp, B.; Kopke, R.D.; Ge, X.; Jackson, R.L.; Dormer, K.J. The permeability of SPION over an artificial three-layer membrane is enhanced by external magnetic field. J. Nanobiotechnol. 2006, 4, 1–9. [Google Scholar] [CrossRef]
- Pankhurst, Q.A.; Connolly, J.; Jones, S.K.; Dobson, J. Applications of magnetic nanoparticles in biomedicine. J. Phys. D. Appl. Phys. 2003, 36, R167–R181. [Google Scholar] [CrossRef]
- Chertok, B.; Moffat, B.A.; David, A.E.; Yu, F.Q.; Bergemann, C.; Ross, B.D.; Yang, V.C. Iron oxide nanoparticles as a drug delivery vehicle for MRI monitored magnetic targeting of brain tumors. Biomaterials 2008, 29, 487–496. [Google Scholar] [CrossRef]
- Jain, T.K.; Richey, J.; Strand, M.; Leslie-Pelecky, D.L.; Flask, C.A.; Labhasetwar, V. Magnetic nanoparticles with dual functional properties: Drug delivery and magnetic resonance imaging. Biomaterials 2008, 29, 4012–4021. [Google Scholar] [CrossRef]
- Polyak, B.; Friedman, G. Magnetic targeting for site-specific drug delivery: applications and clinical potential. Expert. Opin. Drug Del. 2009, 6, 53–70. [Google Scholar] [CrossRef]
- Balimane, P.V.; Chong, S. Cell culture-based models for intestinal permeability: a critique. Drug Discov. Today 2005, 10, 335–343. [Google Scholar] [CrossRef]
- Ward, P.D.; Tippin, T.K.; Thakker, D.R. Enhancing paracellular permeability by modulating epithelial tight junctions. Pharm. Sci. Technol. Today 2000, 3, 346–358. [Google Scholar] [CrossRef]
- Kitchens, K.M.; Kolhatkar, R.B.; Swaan, P.W.; Eddington, N.D.; Ghandehari, H. Transport of poly(amidoamine) dendrimers across Caco-2 cell monolayers: Influence of size, charge and fluorescent labeling. Pharm. Res. 2006, 23, 2818–2826. [Google Scholar] [CrossRef]
- Rojanasakul, Y.; Wang, L.Y.; Bhat, M.; Glover, D.D.; Malanga, C.J.; Ma, J.K. The transport barrier of epithelia: a comparative study on membrane permeability and charge selectivity in the rabbit. Pharm. Res. 1992, 9, 1029–1034. [Google Scholar] [CrossRef]
- Berry, C.C.; Curtis, A.S.G. Functionalisation of magnetic nanoparticles for applications in biomedicine. J. Phys. D. Appl. Phys. 2003, 36, R198–R206. [Google Scholar] [CrossRef]
- Ma, Y.J.; Gu, H.C. Study on the endocytosis and the internalization mechanism of aminosilane-coated Fe3O4 nanoparticles in vitro. J. Mater. Sci. Mater. Med. 2007, 18, 2145–2149. [Google Scholar] [CrossRef]
- Swaan, P.W. Recent advances in intestinal macromolecular drug delivery via receptor-mediated transport pathways. Pharm. Res. 1998, 15, 826–834. [Google Scholar] [CrossRef]
- Tartaj, P.; Morales, M.D.; Veintemillas-Verdaguer, S.; Gonzalez-Carreno, T.; Serna, C.J. The preparation of magnetic nanoparticles for applications in biomedicine. J. Phys. D. Appl. Phys. 2003, 36, R182–R197. [Google Scholar] [CrossRef]
- Calderon, F.L.; Stora, T.; Mondain Monval, O.; Poulin, P.; Bibette, J. Direct measurement of colloidal forces. Phys. Rev. Lett. 1994, 72, 2959–2962. [Google Scholar] [CrossRef]
- Gupta, A.K.; Gupta, M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar] [CrossRef]
- Kuhn, S.J.; Hallahan, D.E.; Giorgio, T.D. Characterization of superparamagnetic nanoparticle interactions with extracellular matrix in an in vitro system. Ann. Biomed. Eng. 2006, 34, 51–58. [Google Scholar] [CrossRef]
- Lemarchand, C.; Gref, R.; Couvreur, P. Polysaccharide-decorated nanoparticles. Eur. J. Pharm. Biopharm. 2004, 58, 327–341. [Google Scholar] [CrossRef]
- Gaush, C.R.; Hard, W.L.; Smith, T.F. Characterization of an Established Line of Canine Kidney Cells (Mdck). P. Soc. Exp. Biol. Med. 1966, 122, 931–935. [Google Scholar] [CrossRef]
- Hidalgo, I.J.; Raub, T.J.; Borchardt, R.T. Characterization of the Human-Colon Carcinoma Cell-Line (Caco-2) as a Model System for Intestinal Epithelial Permeability. Gastroenterology 1989, 96, 736–749. [Google Scholar]
- Irvine, J.D.; Takahashi, L.; Lockhart, K.; Cheong, J.; Tolan, J.W.; Selick, H.E.; Grove, J.R. MDCK (Madin-Darby canine kidney) cells: A tool for membrane permeability screening. J. Pharm. Sci. 1999, 88, 28–33. [Google Scholar] [CrossRef]
- Balimane, P.V.; Chong, S.H.; Morrison, R.A. Current methodologies used for evaluation of intestinal permeability and absorption. J. Pharmacol. Toxicol. 2000, 44, 301–312. [Google Scholar] [CrossRef]
- Kim, D.K.; Zhang, Y.; Voit, W.; Rao, K.V.; Muhammed, M. Synthesis and characterization of surfactant-coated superparamagnetic monodispersed iron oxide nanoparticles. J. Magn. Magn. Mater. 2001, 225, 30–36. [Google Scholar] [CrossRef]
- Bomsel, M.; Prydz, K.; Parton, R.G.; Gruenberg, J.; Simons, K. Endocytosis in filter-grown Madin-Darby canine kidney cells. J. Cell. Biol. 1989, 109, 3243–3258. [Google Scholar] [CrossRef] [Green Version]
- Yu, F.; Huang, Y.; Cole, A.J.; Yang, V.C. The artificial peroxidase activity of magnetic iron oxide nanoparticles and its application to glucose detection. Biomaterials 2009, 30, 4716–4722. [Google Scholar] [CrossRef]
- Yamaura, M.C., R.L.; Sampaio, L.C.; Macedo, M.A.; Nakamura, M.; Toma, H.E. Preparation and characterization of (3-aminopropyl) triethoxysilane-coated magnetite nanoparticles. J. Magn. Magn. Mater. 2004, 279, 210–217. [Google Scholar] [CrossRef]
- Kim, E.H.; Lee., H.S.; Kwak, B.K.; Kim, B.K. Synthesis of ferrofluid with magnetic nanoparticles by sonochemical method for MRI contrast agent. J. Magn. Magn. Mater. 2005, 289, 328–330. [Google Scholar] [CrossRef]
- Lee, Y.L.; Lee, J.; Bae, C.J.; Park, J.G.; Noh, H.J.; Park, J.H.; Hyeon, T. Large-Scale Synthesis of Uniform and Crystalline Magnetite Nanoparticles Using Reverse Micelles as Nanoreactors under Reflux Conditions. Adv. Funct. Mater. 2005, 15, 503–509. [Google Scholar] [CrossRef]
- Chatterjee, J.; Haik, Y.; Chen, C.J. Size dependent magnetic properties of iron oxide nanoparticles. J. Magn. Magn. Mater. 2003, 257, 113–118. [Google Scholar] [CrossRef]
- Caruso, F.; Susha, A.S.; Giersig, M.; Mohwald, H. Magnetic core-shell particles: Preparation of magnetite multilayers on polymer latex microspheres. Adv. Mater. 1999, 11, 950–953. [Google Scholar] [CrossRef]
- Yu, F.; Yang, V.C. Size-tunable synthesis of stable superparamagnetic iron oxide nanoparticles for potential biomedical applications. J. Biomed. Mater. Res. A 2010, 92, 1468–1475. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Min, K.A.; Yu, F.; Yang, V.C.; Zhang, X.; Rosania, G.R. Transcellular Transport of Heparin-coated Magnetic Iron Oxide Nanoparticles (Hep-MION) Under the Influence of an Applied Magnetic Field. Pharmaceutics 2010, 2, 119-135. https://doi.org/10.3390/pharmaceutics2020119
Min KA, Yu F, Yang VC, Zhang X, Rosania GR. Transcellular Transport of Heparin-coated Magnetic Iron Oxide Nanoparticles (Hep-MION) Under the Influence of an Applied Magnetic Field. Pharmaceutics. 2010; 2(2):119-135. https://doi.org/10.3390/pharmaceutics2020119
Chicago/Turabian StyleMin, Kyoung Ah, Faquan Yu, Victor C. Yang, Xinyuan Zhang, and Gus R. Rosania. 2010. "Transcellular Transport of Heparin-coated Magnetic Iron Oxide Nanoparticles (Hep-MION) Under the Influence of an Applied Magnetic Field" Pharmaceutics 2, no. 2: 119-135. https://doi.org/10.3390/pharmaceutics2020119