1. Introduction
Due to an abundance of coal reserves around the globe, coal will remain the main contributor to the energy supply chain, not only for power and heat generation, but also for fuel and chemical processing. The world’s coal reserves are estimated to be over 861 billion tonnes. Coal generates 42% of the world’s electricity and provides about 30% of the world’s primary energy [
1]. About 45% of the world’s coal has a high moisture or ash content. Many of the developing and developed countries, including China, Turkey, India, South Africa, Romania, etc., still rely heavily on coals to meet their power generation needs [
2]. To meet the energy demand and supply gap, low-rank coals can be an attractive option due to the availability of huge reserves for at least the next few decades [
3,
4]. Coal has the greatest capacity compared to any non-renewable fuel, accounting for 56% of global reserves and 89% of global capital [
5]. As coal is the most plentiful, efficient, and inexpensive fuel, it has the potential to become the most reliable and accessible energy source, contributing significantly to global energy security. The environmental effects of coal, both in processing and in use, are one of the most pressing issues faced by the power generation industry. The coal industry emits pollutants, such as SOx, NOx, Cox, and particulate matter of various sizes that significantly contribute towards global warming and climate change. Over the last few decades, various pollutant management systems have been developed and are still developing. Clean coal technologies are emerging technologies that make it easier to use coal in a more environmentally friendly way by significantly reducing pollutant emissions [
6].
Recent advancements in syngas, H
2, and technologies for energy production using natural gas and coal as fundamental fuels have emphasized meeting energy demands while lowering greenhouse gas emissions [
7]. Steam methane reforming (SMR), auto-thermal reforming (ATR), dry methane reforming (DMR), tri-methane reforming (TMR), Partial Oxidation (POX), and gasification are some of the industrial processes that can generate H
2 from fossil fuel [
8]. Different modes of gasification with multi-fuel and multi-products have been evaluated as one of the most effective strategies for future electricity and H
2 production with reduced carbon footprints. Furthermore, coal use has been on the rise for decades, with an estimated annual growth of 0.6% [
9]. When it comes to fossil fuel stocks, coal outnumbers natural gas by a large margin. Because of the plentiful and relatively low-cost coal reserves around the world, coal is to remain in the future a viable fossil fuel with primary power and hydrogen (H
2) applications.
Moreover, the diversity of products obtained from gasification, in particular H
2, has increased the popularity of the technology [
10,
11]. Furthermore, the versatility of the gasification process, allowing it to integrate with various technologies, reflects its long-term potential. The SMR method produces syngas with H
2 and CO at a ratio of 3:1, using natural gas and steam, which shows a more significant production potential. It is one of the most sophisticated, widely used, and cost-effective H
2 production technologies [
12]. Furthermore, the technology’s high efficiencies and low operating and production costs, as well as the readily available natural gas, reinforce it [
13]. Dry methane reforming (DMR), on the other hand, produces syngas from CO
2 and natural gas. Endothermic reactions are one of the key downsides of this technique, which, in comparison to SMR, increase operational and production costs. Furthermore, the carbon monoxide and hydrogen ratio is 1:1, indicating lower H
2 output than SMR [
14]. However, catalyst deactivation and energy consumption are some of the operation issues that need to be addressed [
15]. Similarly, POX is a non-catalytic method that produces syngas after burning with pure oxygen. The key disadvantages are the high cost of oxygen separation, the high operating temperature of 1300–1500 °C, and the pressure of 3–8 MPa needed to carry out reactions [
8]. Furthermore, syngas generated by the POX process gives a 1:1 ratio of hydrogen and carbon monoxide, which is significantly lower than that of SMR [
8,
16]. The major advantage of steam methane reforming and gasification over partial oxidation and dry methane reforming is that both SMR and gasification are more suitable for increasing H
2 yield with minimum energy requirements.
Process optimization [
17,
18], intensification [
19,
20], monitoring [
21,
22], and integration [
23] with current technologies can contribute to the development of more viable energy generation systems with carbon capture, thereby improving the economics of the process in terms of the cost of electricity (COE). Recently, there has been huge attention paid to integrating IGCC with natural gas reforming processes to increase the H
2/CO ratio in syngas. Most of the effort is focused on developing gasification processes incorporating CO
2 capture technologies using various feedstocks for the production of synthesis gas, H
2, energy, and polygeneration [
19,
24,
25,
26]. Recently, Alibrahim et al. [
27] developed different pathways for producing syngas by integrating gasification and DMR technologies followed by their comparative analyses for analyzing the sustainability of the process. Qian et al. [
28] proposed an integrative approach for the efficient use of coke oven gas and coal through gasification by a tri-reforming method to improve the H
2/CO ratio in syngas for methanol synthesis. Likewise, Yi et al. [
29] conducted a techno-economic analysis of connecting coal gasification and the DMR procedures to improve syngas synthesis, which may be used in a variety of polygeneration processes. Adams et al. [
30] investigated and analyzed several design paths for combining IGCC with methane reforming systems for process performance and economics. The combination of coal reforming and gasification processes has also been shown to have the potential to improve the robustness of the design and the sustainability of the process [
30].
The energy strategy requires developing the energy-mix and fuel-switching technology to reduce both greenhouse gas emissions and the CO
2 footprint. Using the energy-mix technologies to integrate with advanced technology leads to producing several products during the polygeneration process while minimizing reliance on a single fuel [
31,
32,
33]. The fuel switchover strategies of merging secondary and primary fuels and reducing greenhouse gas emissions have lately been examined by Bazzanella et al. [
34] and Rehfeldt et al. [
35]. Ahmed et al. [
36] undertook a study to develop a unique and commercially viable energy-efficient process, increasing methanol production capacity and reducing energy needs and process economies. This study integrates the coal gasification process with the gas reforming technology in a parallel design configuration to improve the syngas calorific value in order to synthesize methanol and the controlled emissions of GHG. The primary benefit of energy-mix and fuel-switch technology is that no large process adjustments are required. Recently, energy-mixed systems that use different fuels to create numerous chemicals have also been created. Chen et al. [
37] designed the process model to produce methanol in the CTM (coal to methanol) process by using different feedstocks. The proposed model reduced emissions of carbon dioxide by 14% and enhanced the production of methanol by 6.5%. Lv et al. [
38] showed a 55.8% process efficiency based on a process model of power, dual methanol based on natural gas, and charcoal feedstocks but did not disclose any information on the economics of the process. The study has demonstrated that the combining of gas and coal technologies can increase the potential of syngas with a higher level of hydrogen and carbon ratio. Due to inherent pollutants, the coal-based syngas composition is difficult to retain and therefore needs further treatment before it can be utilized in downstream operations. The coal-based syngas can be coupled with the natural gas-based syngas to optimize the HCR for the efficient generation of cleaner fuels [
39]. Models for merging coal gasification with natural gas reforming procedures for the production of hydrogen and power generation have been proposed by Ahmed et al. [
40,
41,
42]. Ahmed et al. [
43] also studied the techno-economic assessment of a process integration model to boost the potential of hydrogen production by using natural gas and coal feedstock. The studies have found that coal gasifiers can be connected with the gas reforming technologies using an efficient heat exchanger network without using additional power sources, thus increasing overall process performance.
Furthermore, more research is focused on the integration and intensification of the gasification process with the reforming technologies to improve the syngas yield. Cormos et al. [
44] investigated the techno-economics of flexible hydrogen and electricity generation by the gasification of coal. Ahmed et al. [
41] proposed to boost H
2 production by using steam and syngas-heat from the process of gasification by incorporating integrated models of coal gasification and SMR. The techno-economic analyses of both integrated coal gasification and conventional coal gasification and steam methane reforming processes are also carried out by Ahmed et al. [
40] concluded that the integrated model is the most effective method. On the other hand, previous processes of reforming units and the integration of gasification in series had technical problems in the reforming section because the synthesis gas obtained from coal has a higher sulfur content than the acceptable limits of the reforming catalyst.
The disadvantages of the harmonious blend of gasification and reforming technology insights show that the indirect use of gasification-derived syngas in the SMR process, employing a radiant cooler with no risks of catalytic deactivation, has become a realistic new incorporation approach. A simultaneous process integration network strategy will be used in the proposed integration to exploit the essential technological and economic benefits of several technologies, which will improve the total viability of the system without having to rely on one fuel. The primary aim of gasification technology is to produce syngas, which can be utilized in a wide range of applications, from fuel and chemical production to electricity and power generation. The Fischer–Tropsch (FT) process can be used to make gasoline, diesel, and other chemicals. In the presence of suitable catalysts, syngas can also be easily converted to methanol or dimethyl ether (DME), which can then be converted to gasoline. Synthetic natural gas (SNG) production is also another important application of syngas. Syngas is also used in the production of medium-BTU gas for turbine fuel and in advanced gasification [
45].
Coal gasification is an environmentally sustainable and effective solution for producing a range of chemicals, such as methanol, ammonia, hydrogen, and synthetic fuels. The gasification of low-ranked coals is even more tempting due to the low pricing of coal and the local availabilities or even the lack of other resources, such as natural gas and oil [
6]. Low-rank coals are extensively available but offer less power generation efficiencies and are usually ignored in power plants. Low-rank coals can be converted into syngas or hydrogen with better process feasibility instead of power generation. This study will focus on the production of syngas from low-rank coals, where the coal composition is found from experiments and then modelling is done in Aspen Plus to carry out the technical and economic analyses to evaluate the process feasibility.
2. Experimental and Simulation Approach
Coal samples were taken and crushed with a local roller crusher and disc mill and sieved in −60 mesh for performing the proximate and ultimate analyses. Standard procedure for proximate analysis (D3173), ash (D3174), volatile matter (D3175), sulfur (D3177), and gross calorific value (D5865) was adopted. The results of the proximate and ultimate analyses are shown in
Table 1.
After getting the coal composition from the experimental results, a simulation study is performed to analyze the syngas composition from the coal and to perform parametric analysis. The simulation model is developed in the commercial chemical and process engineering software, namely Aspen Plus V11
®. The Peng-Robinson equation of state was used as the thermodynamic package with the Boston Mathias function (PR-BM). The coal is an un-conventional component in Aspen Plus and is defined based on ultimate, proximate, sulfur, and heating value analyses obtained from experimental results. The RYield reactor model is used in Aspen Plus for converting the coal into its constituents, followed by its integration with the RGibbs reactor model to generate synthesis gas. The gasification process involves a series of complex endothermic and exothermic reactions to generate syngas, as shown in the following equations:
The RGibbs reactor model works based on the Gibbs free energy minimization principle to produce syngas at the specified pressure and temperature conditions.
Figure 1 represents the gasification model developed in Aspen Plus in which coal at the flow rate of 62.01 kg/s is fed to the RYield Reactor to get the yield of coal in terms of mainly C, H
2, N
2, O
2, S, and ash contents. The RYield model is specified to achieve 99% of the coal conversion into syngas and char. The separator is used to separate the volatiles from the un-converted char. The volatiles are then sent to the RGibbs reactor model to generate the reaction products at the gasifier operational conditions. Furthermore, the char is mixed with oxygen (95% pure) and steam to convert the elemental carbon into CO and CO
2, which is mixed with the volatiles and fed to the gasification unit to get the desired syngas composition at the pressure and temperature of 55 bar and 1165 °C, respectively. The syngas composition from the gasification model was compared with the literature and the results of the syngas composition are within the range of the reported data as represented in
Table 2.
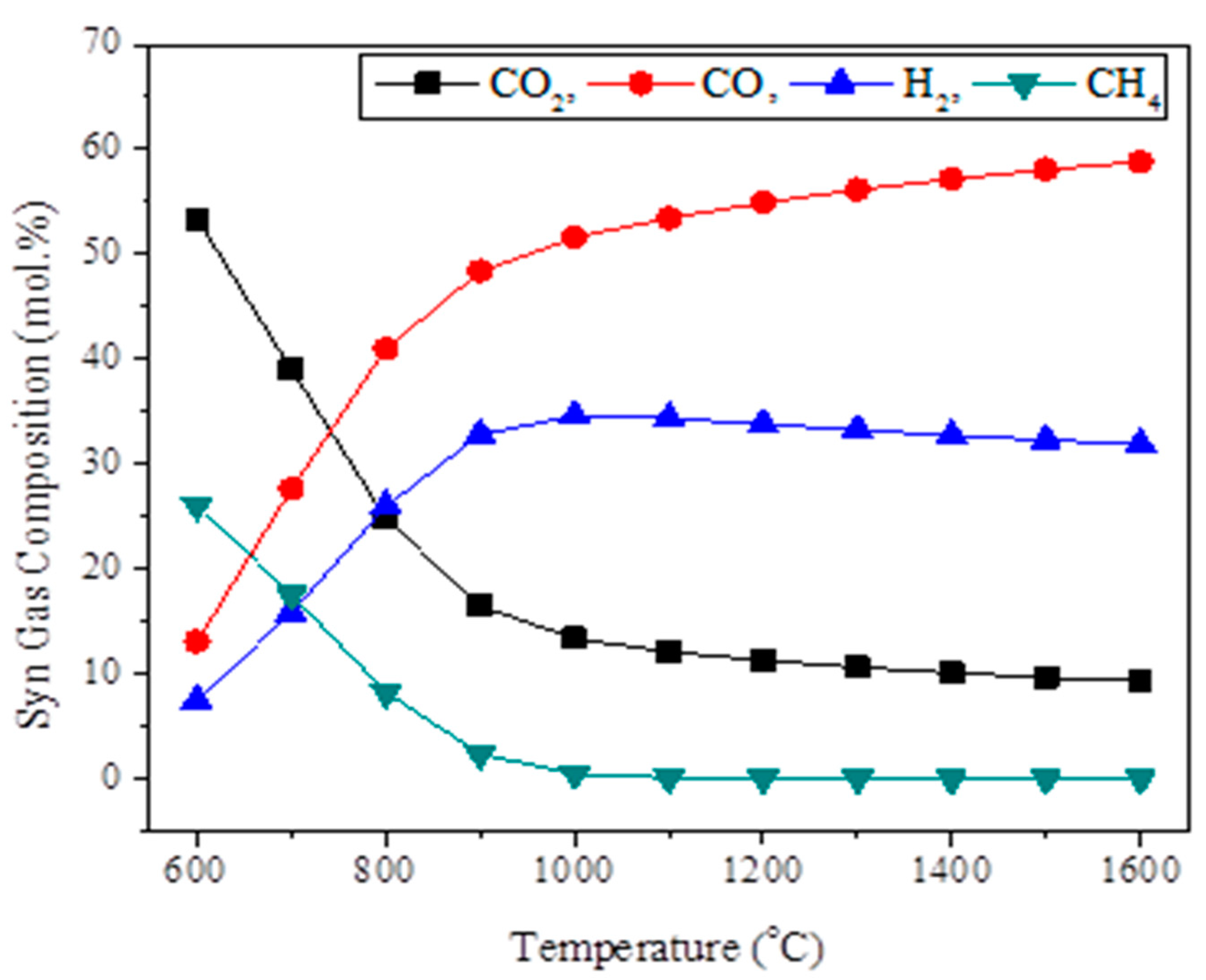
The sensitivity studies were also preformed to see the variation in the synthesis gas composition by varying the process parameters.