Effect of Docosahexaenoic Acid on Ca2+ Signaling Pathways in Cerulein-Treated Pancreatic Acinar Cells, Determined by RNA-Sequencing Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Line and Treatments
2.2. Preparation of Total RNA Extracts and Library Construction
2.3. RNA-Sequencing and Bioinformatics Analysis
2.4. Validation of RNA-Seq Profiles by Real-time Polymerase Chain Reaction (PCR)
2.5. Statistical Analysis
3. Results
3.1. DHA Inhibits Cerulein-Induced Changes in the Transcriptomic Profile of AR42J Cells
3.2. DHA Suppresses Cerulein-Induced Alteration in the Calcium Signaling Pathway, Determined by Functional Annotation and Pathway Analysis of DEGs
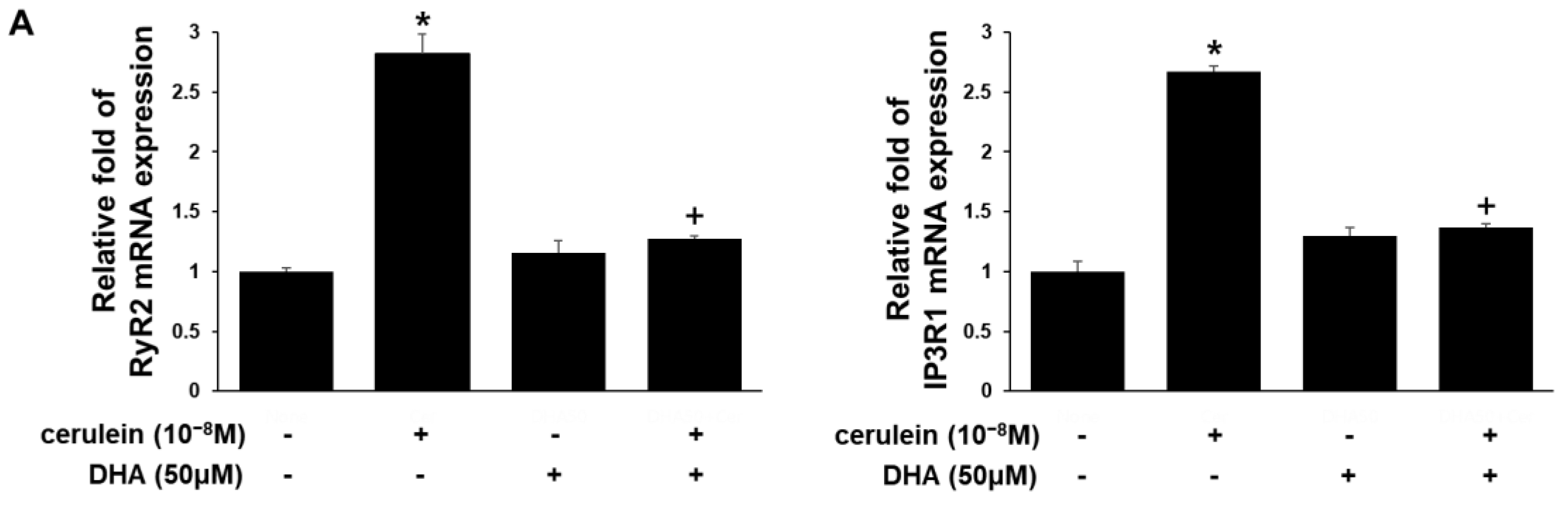
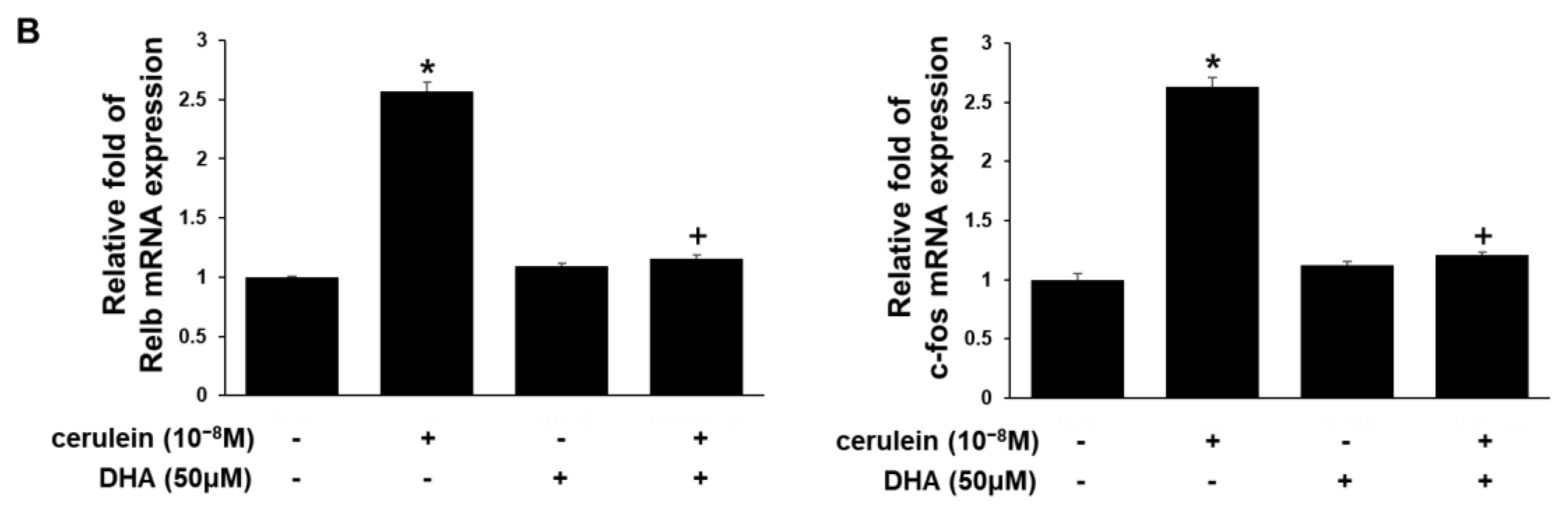
3.3. DHA Inhibits Expression of RyR2, IP3RI, Relb, and c-fos Genes in Cerulein-Treated AR42J Cells, Determined by Real-Time PCR
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bhatia, M.; Wong, F.L.; Cao, Y.; Lau, H.Y.; Huang, J.; Puneet, P.; Chevali, L. Pathophysiology of acute pancreatitis. Pancreatology 2005, 5, 132–144. [Google Scholar] [CrossRef] [PubMed]
- Frossard, J.L.; Hadengue, A.; Pastor, C.M. New serum markers for the detection of severe acute pancreatitis in humans. Am. J. Respir. Crit. Care Med. 2001, 164, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, J.A.; Cash, N.; Soares, P.M.; Souza, M.H.; Sutton, R.; Criddle, D.N. Oxidative stress in acute pancreatitis: Lost in translation? Free Radic. Res. 2013, 47, 917–933. [Google Scholar] [CrossRef] [PubMed]
- Ward, J.B.; Jenkins, S.A.; Sutton, R.; Petersen, O.H. Is an elevated concentration of acinar cytosolic free ionised calcium the trigger for acute pancreatitis? Lancet 1995, 346, 1016–1019. [Google Scholar] [CrossRef]
- Ward, J.B.; Sutton, R.; Jenkins, S.A.; Petersen, O.H. Progressive disruption of acinar cell calcium signaling is an early feature of cerulein-induced pancreatitis in mice. Gastroenterology 1996, 111, 481–491. [Google Scholar] [CrossRef] [PubMed]
- Kruger, B.; Albrecht, E.; Lerch, M.M. The role of intracellular calcium signaling in premature protease activation and the onset of pancreatitis. Am. J. Pathol. 2000, 157, 43–50. [Google Scholar] [CrossRef]
- Ederle, A.; Vantini, I.; Harvey, R.F.; Cavallini, G.; Piubello, W.; Benini, L.; Scuro, L.A. Fasting serum cholecystokinin immunoreactivity in chronic relapsing pancreatitis. Ric. Clin. Lab. 1978, 8, 199–206. [Google Scholar]
- Shirohara, H.; Otsuki, M. Plasma cholecystokinin levels in acute pancreatitis. Pancreas 1997, 14, 249–254. [Google Scholar] [CrossRef]
- Atozaki, T.; Williams, J.A. Multiple sources of 1, 2-diacylglycerol in isolated rat pancreatic acini stimulated by cholecystokinin. Involvement of phosphatidylinositol bisphosphate and phosphatidylcholine hydrolysis. J. Biol. Chem. 1989, 264, 14729–14734. [Google Scholar]
- Trimble, E.R.; Bruzzone, R.; Meehan, C.J.; Biden, T.J. Rapid increases in inositol 1, 4, 5-trisphosphate, inositol 1, 3, 4, 5-tetrakisphosphate and cytosolic free Ca2+ in agonist-stimulated pancreatic acini of the rat. Effect of carbachol, caerulein and secretin. Biochem. J. 1987, 242, 289–292. [Google Scholar] [CrossRef]
- Pérez, S.; Pereda, J.; Sabater, L.; Sastre, J. Redox signaling in acute pancreatitis. Redox Biol. 2015, 5, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Voronina, S.; Longbottom, R.; Sutton, R.; Petersen, O.H.; Tepikin, A. Bile acids induce calcium signals in mouse pancreatic acinar cells: Implications for bile-induced pancreatic pathology. J. Physiol. 2002, 540, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Gerasimenko, J.V.; Lur, G.; Sherwood, M.W.; Ebisui, E.; Tepikin, A.V.; Mikoshiba, K.; Gersimenko, O.V.; Petersen, O.H. Pancreatic protease activation by alcohol metabolite depends on Ca2+ release via acid store IP3 receptors. Proc. Natl. Acad. Sci. USA 2009, 106, 10758–10763. [Google Scholar] [CrossRef]
- Orabi, A.I.; Shah, A.U.; Ahmad, M.U.; Choo-Wing, R.; Parness, J.; Jain, D.; Bhandari, V.; Husain, S.Z. Dantrolene mitigates caerulein-induced pancreatitis in vivo in mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 299, G196–G204. [Google Scholar] [CrossRef] [Green Version]
- Booth, D.M.; Mukherjee, R.; Sutton, R.; Criddle, D.N. Calcium and reactive oxygen species in acute pancreatitis: Friend or foe? Antioxid. Redox Signaling 2011, 15, 2683–2698. [Google Scholar] [CrossRef] [PubMed]
- Gerasimenko, J.V.; Gryshchenko, O.; Ferdek, P.E.; Stapleton, E.; Hébert, T.O.; Bychkova, S.; Peng, S.; Begg, M.; Gerasimenko, O.V.; Petersen, O.H. Ca2+ release-activated Ca2+ channel blockade as a potential tool in antipancreatitis therapy. Proc. Natl. Acad. Sci. USA 2013, 110, 13186–13191. [Google Scholar] [CrossRef] [PubMed]
- Husain, S.; Thrower, E. Molecular and cellular regulation of pancreatic acinar cell Function. Curr. Opin. Gastroenterol. 2009, 25, 466–471. [Google Scholar] [CrossRef] [PubMed]
- Nathanson, M.H.; Fallon, M.B.; Padfield, P.J.; Maranto, A.R. Localization of the type 3 inositol 1, 4, 5-trisphosphate receptor in the Ca2+ wave trigger zone of pancreatic acinar cells. J. Biol. Chem. 1994, 269, 4693–4696. [Google Scholar]
- Hofbauer, B.; Saluja, A.K.; Lerch, M.M.; Bhagat, L.; Bhatia, M.; Lee, H.S.; Steer, M.L. Intra-acinar cell activation of trypsinogen during caerulein-induced pancreatitis in rats. Am. J. Physiol. Gastrointest. Liver Physiol. 1998, 275, G352–G362. [Google Scholar] [CrossRef]
- Nathanson, M.H.; Padfield, P.J.; O’Sullivan, A.J.; Burgstahler, A.D.; Jamieson, J.D. Mechanism of Ca2+ wave propagation in pancreatic acinar cells. J. Biol. Chem. 1992, 267, 18118–18121. [Google Scholar]
- Leite, M.F.; Burgstahler, A.D.; Nathanson, M.H. Ca2+ waves require sequential activation of inositol trisphosphate receptors and ryanodine receptors in pancreatic acini. Gastroenterology 2002, 122, 415–427. [Google Scholar] [CrossRef] [PubMed]
- Lerch, M.M.; Adler, G. Experiemental animal models of acute pancreatitis. Int. J. Pancreatol. 1994, 15, 159–170. [Google Scholar] [PubMed]
- Dolmetsch, R.E.; Xu, K.; Lewis, R.S. Calcium oscillations increase the efficiency and specificity of gene expression. Nature 1998, 392, 933–936. [Google Scholar] [CrossRef] [PubMed]
- Manalo, R.V.; Medina, P.M. The endoplasmic reticulum stress response in disease pathogenesis and pathophysiology. Egypt J. Med. Hum. Genet. 2018, 19, 59–68. [Google Scholar] [CrossRef]
- Ratner, A.J.; Bryan, R.; Weber, A.; Nguyen, S.; Barnes, D.; Pitt, A.; Gelber, S.; Cheung, A.; Prince, A. Cystic fibrosis pathogens activate Ca2+-dependent mitogen-activated protein kinase signaling pathways in airway epithelial cells. J. Biol. Chem. 2001, 276, 19267–19275. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Chen, A.; Lai, X.; Liu, J.; Wu, J.; Kang, Y.; Wang, X.; Shao, L. Neuroinflammation is induced by tongue-instilled ZnO nanoparticles via the Ca 2+-dependent NF-κB and MAPK pathways. Part. Fibre Toxicol. 2018, 15, 39. [Google Scholar] [CrossRef]
- Brown, D.M.; Donaldson, K.; Borm, P.J.; Schins, R.P.; Dehnhardt, M.; Gilmour, P.; Jimenez, L.A.; Stone, V. Calcium and ROS-mediated activation of transcription factors and TNF-α cytokine gene expression in macrophages exposed to ultrafine particles. Am. J. Physiol. Lung Cell Mol. Physiol. 2004, 286, L344–L353. [Google Scholar] [CrossRef]
- Calder, P.C. n-3 Polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am. J. Clin. Nutr. 2006, 83, 1505S–1519S. [Google Scholar] [CrossRef]
- Song, E.A.; Lim, J.W.; Kim, H. Docosahexaenoic acid inhibits IL-6 expression via PPARγ-mediated expression of catalase in cerulein-stimulated pancreatic acinar cells. Int. J. Biochem. Cell Biol. 2017, 88, 60–68. [Google Scholar] [CrossRef]
- Ju, K.D.; Lim, J.W.; Kim, K.H.; Kim, H. Potential role of NADPH oxidase-mediated activation of Jak2/Stat3 and mitogen-activated protein kinases and expression of TGF-β1 in the pathophysiology of acute pancreatitis. Inflamm. Res. 2011, 60, 791–800. [Google Scholar] [CrossRef]
- Yu, J.H.; Lim, J.W.; Kim, H.; Kim, K.H. NADPH oxidase mediates interleukin-6 expression in cerulein-stimulated pancreatic acinar cells. Int. J. Biochem. Cell Biol. 2005, 37, 1458–1469. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, H.J.; Chang, K.C.; Baek, J.C.; Park, J.K.; Shin, J.K.; Choi, W.J.; Lee, J.H.; Paik, W.Y. DHA and EPA down-regulate COX-2 expression through suppression of NF-κB activity in LPS-treated human umbilical vein endothelial cells. Korean J. Physiol. Pharmacol. 2009, 13, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.Y.; Jin, J.Y.; Choi, J.I.; Choi, I.S.; Kim, S.J. DHA suppresses Prevotella intermedia lipopolysaccharide-induced production of proinflammatory mediators in murine macrophages. Br. J. Nutr. 2014, 111, 1221–1230. [Google Scholar] [CrossRef] [PubMed]
- Park, K.S.; Lim, J.W.; Kim, H. Inhibitory mechanism of omega-3 fatty acids in pancreatic inflammation and apoptosis. Ann. N. Y. Acad. Sci. 2009, 1171, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Lim, J.W.; Kim, H. Docoxahexaenoic acid induces apoptosis of pancreatic cancer cells by suppressing activation of STAT3 and NF-κB. Nutrients 2018, 10, E1621. [Google Scholar] [CrossRef] [PubMed]
- Criddle, D.N. Reactive oxygen species, Ca2+ stores and acute pancreatitis; a step closer to therapy? Cell Calcium 2016, 60, 180–189. [Google Scholar] [CrossRef] [Green Version]
- Brookes, P.S.; Yoon, Y.; Robotham, J.L.; Anders, M.W.; Sheu, S.S. Calcium, ATP, and ROS: A mitochondrial love-hate triangle. Am. J. Physiol. Cell Physiol. 2004, 287, C817–C833. [Google Scholar] [CrossRef]
- Gerasimenko, J.V.; Peng, S.; Tsugorka, T.; Gerasimenko, O.V. Ca2+ signalling underlying pancreatitis. Cell Calcium. 2018, 70, 95–101. [Google Scholar] [CrossRef]
- Gerasimenko, J.V.; Flowerdew, S.E.; Voronina, S.G.; Sukhomlin, T.K.; Tepikin, A.V.; Petersen, O.H.; Gerasimenko, O.V. Bile acids induce Ca2+ release from both the endoplasmic reticulum and acidic intracellular calcium stores through activation of inositol trisphosphate receptors and ryanodine receptors. J. Biol. Chem. 2006, 281, 40154–40163. [Google Scholar] [CrossRef]
- Gerasimenko, J.V.; Gerasimenko, O.V.; Petersen, O.H. The role of Ca2+ in the pathophysiology of pancreatitis. J. Physiol. 2014, 592, 269–280. [Google Scholar] [CrossRef]
- Straub, S.V.; Giovannucci, D.R.; Yule, D.I. Calcium wave propagation in pancreatic acinar cells: Functional interaction of inositol 1, 4, 5-trisphosphate receptors, ryanodine receptors, and mitochondria. J. Gen. Physiol. 2000, 116, 547–560. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Logsdon, C.D. CCK stimulates mob-1 expression and NF-κB activation via protein kinase C and intracellular Ca2+. Am. J. Physiol. Cell Physiol. 2000, 278, C344–C351. [Google Scholar] [CrossRef] [PubMed]
- Tando, Y.; Algul, H.; Wagner, M.; Weidenbach, H.; Adler, G.; Schmid, R.M. Caerulein-induced NF-κB/Rel activation requires both Ca2+ and protein kinase C as messengers. Am. J. Physiol. Gastrointest. Liver Physiol. 1999, 277, G678–G686. [Google Scholar] [CrossRef] [PubMed]
- Hietaranta, A.J.; Singh, V.P.; Bhagat, L.; Van Acker, G.J.; Song, A.M.; Mykoniatis, A.; Steer, M.L.; Saluja, A.K. Water immersion stress prevents caerulein-induced pancreatic acinar cell NF-κB activation by attenuating caerulein-induced intracellular Ca2+ changes. J. Biol. Chem. 2001, 276, 18742–18747. [Google Scholar] [CrossRef] [PubMed]
- Orlichenko, L.S.; Behari, J.; Yeh, T.H.; Liu, S.; Stolz, D.B.; Saluja, A.K.; Singh, V.P. Transcriptional regulation of CXC-ELR chemokines KC and MIP-2 in mouse pancreatic acini. Am. J. Physiol. Heart Circ. Physiol. 2010, 299, G867–G876. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turner, D.J.; Cowles, R.A.; Segura, B.J.; Mulholland, M.W. Cholinergic stimulation of rat acinar cells increases c-fos and c-jun expression via a mitogen-activated protein kinase-dependent pathway. J. Gastrointest. Surg. 2001, 5, 661–672. [Google Scholar] [CrossRef]
- Ramnath, R.D.; Sun, J.; Bhatia, M. Role of calcium in substance P-induced chemokine synthesis in mouse pancreatic acinar cells. Br. J. Pharmacol. 2008, 154, 1339–1348. [Google Scholar] [CrossRef] [Green Version]
- Begum, G.; Harvey, L.; Dixon, C.E.; Sun, D. ER stress and effects of DHA as an ER stress inhibitor. Transl. Stroke Res. 2013, 4, 635–642. [Google Scholar] [CrossRef]
- Begum, G.; Kintner, D.; Liu, Y.; Cramer, S.W.; Sun, D. DHA inhibits ER Ca2+ release and ER stress in astrocytes following in vitro ischemia. J. Neurochem. 2012, 120, 622–630. [Google Scholar] [CrossRef]
- Rodrigo, G.C.; Dhanapala, S.; Macknight, A.D.C. Effects of eicosapentaenoic acid on the contraction of intact, and spontaneous contraction of chemically permeabilized mammalian ventricular myocytes. J. Mol. Cell. Cardiol. 1999, 31, 733–743. [Google Scholar] [CrossRef]
- Honen, B.N.; Saint, D.A.; Laver, D.R. Suppression of calcium sparks in rat ventricular myocytes and direct inhibition of sheep cardiac RyR channels by EPA, DHA and oleic acid. J. Membr. Biol. 2003, 196, 95–103. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Liu, W.; Shi, M.; Yang, Z.; Zhang, X.; Gong, P. Docosahexaenoic acid attenuates LPS-stimulated inflammatory response by regulating the PPARγ/NF-κB pathways in primary bovine mammary epithelial cells. Res. Vet. Sci. 2017, 112, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Q.; Chen, M.T.; Zhang, R.; Zhang, Y.; Li, W.; Li, Y.G. Docosahexaenoic acid attenuates doxorubicin-induced cytotoxicity and inflammation by suppressing NF-κB/iNOS/NO signaling pathway activation in H9C2 cardiac cells. J. Cardiovasc. Pharmacol. 2016, 67, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.Y.; Kuan, Y.H.; Li, J.R.; Chen, W.Y.; Ou, Y.C.; Pan, H.C.; Lia, S.L.; Raung, S.L.; Chang, C.J.; Chen, C.J. Docosahexaenoic acid reduces cellular inflammatory response following permanent focal cerebral ischemia in rats. J. Nutr. Biochem. 2013, 24, 2127–2137. [Google Scholar] [CrossRef] [PubMed]
- Sheng, M.; Greenberg, M.E. The regulation and function of c-fos and other immediate early genes in the nervous system. Neuron 1990, 4, 477–485. [Google Scholar] [CrossRef]
- Sadoshima, J.; Izumo, S. Signal transduction pathways of angiotensin II--induced c-fos gene expression in cardiac myocytes in vitro. Roles of phospholipid-derived second messengers. Circ. Res. 1993, 73, 424–438. [Google Scholar] [CrossRef] [PubMed]
- Serkkola, E.; Hurme, M. Synergism between protein-kinase C and cAMP-dependent pathways in the expression of the interleukin-1β gene is mediated via the activator-protein-1 (AP-1) enhancer activity. Eur. J. Biochem. 1993, 213, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Silvers, A.L.; Bachelor, M.A.; Bowden, G.T. The role of JNK and p38 MAPK activities in UVA-induced signaling pathways leading to AP-1 activation and c-Fos expression. Neoplasia 2003, 5, 319–329. [Google Scholar] [CrossRef]
- Cavigelli, M.; Dolfi, F.; Claret, F.X.; Karin, M. Induction of c-fos expression through JNK-mediated TCF/Elk-1 phosphorylation. EMBO J. 1995, 14, 5957–5964. [Google Scholar] [CrossRef]
- Guma, M.; Stepniak, D.; Shaked, H.; Spehlmann, M.E.; Shenouda, S.; Cheroutre, H.; Vincente-Suarez, I.; Eckmann, L.; Kagnoff, M.F.; Karin, M. Constitutive intestinal NF-κB does not trigger destructive inflammation unless accompanied by MAPK activation. J. Exp. Med. 2001, 208, 1889–1900. [Google Scholar] [CrossRef]
- Jakkampudi, A.; Jangala, R.; Reddy, B.R.; Mitnala, S.; Reddy, D.N.; Talukdar, R. NF-κB in acute pancreatitis: Mechanisms and therapeutic potential. Pancreatology 2016, 16, 477–488. [Google Scholar] [CrossRef] [PubMed]


| Gene Name | None Mapped Read 1 | Cerulein Mapped Read 1 | DHA Mapped Read 1 | Cerulein+DHA Mapped Read 1 |
|---|---|---|---|---|
| Fosb | 49 | 242 | 55 | 113 |
| Relb | 38 | 194 | 62 | 156 |
| Pde2a | 997 | 2143 | 919 | 1402 |
| Rras2 | 2058 | 4369 | 1837 | 3879 |
| Npas4 | 10 | 210 | 7 | 112 |
| Pnlip | 3165 | 9804 | 2821 | 6068 |
| Pdzd2 | 35 | 124 | 47 | 55 |
| Trpc4 | 44 | 117 | 32 | 62 |
| Ankrd34a | 38 | 261 | 26 | 178 |
| Dkk2 | 97 | 275 | 53 | 194 |
| Nr4a2 | 77 | 363 | 60 | 204 |
| Gad1 | 237 | 763 | 182 | 483 |
| Itga4 | 214 | 500 | 112 | 280 |
| F2 | 121 | 293 | 140 | 188 |
| Mylk2 | 1651 | 4457 | 1293 | 2648 |
| Nfs1 | 205 | 1109 | 156 | 67 |
| Rims4 | 118 | 567 | 125 | 401 |
| Chrna4 | 414 | 1059 | 381 | 803 |
| Pdk4 | 81 | 287 | 43 | 198 |
| Reg3b | 12 | 2873 | 2 | 892 |
| Reg1a | 11,048 | 26,879 | 9104 | 12,476 |
| Itpr1 | 2398 | 6178 | 2656 | 4547 |
| Bid | 109 | 317 | 105 | 213 |
| M6pr | 421 | 1352 | 426 | 485 |
| C1s | 51 | 276 | 45 | 139 |
| Gucy2c | 138 | 2830 | 65 | 1420 |
| Akap2 | 54 | 151 | 110 | 102 |
| Cela3b | 810 | 5573 | 915 | 3610 |
| Lbh | 652 | 2070 | 572 | 1366 |
| Dock4 | 99 | 331 | 135 | 198 |
| Fos | 14 | 226 | 34 | 164 |
| Tshr | 70 | 556 | 80 | 496 |
| Moap1 | 372 | 832 | 444 | 684 |
| Pros1 | 54 | 122 | 52 | 100 |
| Timp3 | 102 | 260 | 116 | 146 |
| Matn2 | 111 | 268 | 159 | 234 |
| Abcd2 | 146 | 436 | 101 | 346 |
| Rgl3 | 84 | 168 | 76 | 133 |
| Chrnb4 | 157 | 382 | 81 | 312 |
| Plcl1 | 738 | 1457 | 332 | 1357 |
| Ihh | 3020 | 10,806 | 1511 | 6831 |
| Per1 | 146 | 436 | 101 | 346 |
| Tmem130 | 465 | 1162 | 550 | 899 |
| Hpd | 24 | 139 | 24 | 88 |
| Sgsm1 | 666 | 1706 | 560 | 1286 |
| Igsf8 | 2961 | 9669 | 3829 | 7782 |
| Kcnj9 | 119 | 290 | 115 | 209 |
| Gabra2 | 361 | 922 | 209 | 558 |
| Klf2 | 61 | 302 | 117 | 185 |
| Pdlim3 | 574 | 1796 | 294 | 886 |
| RyR2 | 32 | 154 | 32 | 96 |
| Impad1 | 129 | 447 | 117 | 326 |
| Egr1 | 138 | 752 | 286 | 478 |
| Mapk4 | 645 | 1750 | 508 | 1098 |
| Junb | 32 | 154 | 32 | 96 |
| Col18a1 | 277 | 1286 | 243 | 818 |
| Armcx1 | 80 | 203 | 17 | 102 |
| Metabolic Pathways | Gad1, Hpd, Hpse, Impad1, Nfs1, Pnlip |
|---|---|
| Neuroactive ligand-receptor interaction | Chrna4, Chrnb4, F2, Gabra2, Tshr |
| Oxytocin signaling pathway | Fos, Itpr1, Kcnj9, Mylk2, Ryr2 |
| Apelin signaling pathway | Itpr1, Klf2, Mylk2, Rras2, Ryr2 |
| Proteoglycans in cancer | Hpse, Ihh, Itpr1, Rras2, Timp3 |
| Pancreatic secretion | Cela3b, Itpr1, Pnlip, Ryr2 |
| Regulation of actin cytoskeleton | F2, Itga4, Mylk2, Rras2 |
| cAMP signaling pathway | Fos, Rras2, Ryr2, Tshr |
| MAPK signaling pathway | Fos, Nr4a1, Relb, Rras2 |
| Calcium signaling pathway | Itpr1, Mylk2, Ryr2 |
| cGMP-PKG signaling pathway | Itpr1, Mylk2, Pde2a |
| Apoptosis | Bid, Fos, Itpr1 |
| Autophagy | Itpr1, Rab7b, Rras2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.H.; Park, Y.; Lim, J.W.; Kim, H. Effect of Docosahexaenoic Acid on Ca2+ Signaling Pathways in Cerulein-Treated Pancreatic Acinar Cells, Determined by RNA-Sequencing Analysis. Nutrients 2019, 11, 1445. https://doi.org/10.3390/nu11071445
Kim SH, Park Y, Lim JW, Kim H. Effect of Docosahexaenoic Acid on Ca2+ Signaling Pathways in Cerulein-Treated Pancreatic Acinar Cells, Determined by RNA-Sequencing Analysis. Nutrients. 2019; 11(7):1445. https://doi.org/10.3390/nu11071445
Chicago/Turabian StyleKim, Suhn Hyung, Yeeun Park, Joo Weon Lim, and Hyeyoung Kim. 2019. "Effect of Docosahexaenoic Acid on Ca2+ Signaling Pathways in Cerulein-Treated Pancreatic Acinar Cells, Determined by RNA-Sequencing Analysis" Nutrients 11, no. 7: 1445. https://doi.org/10.3390/nu11071445






