1. Introduction
Chronic liver diseases and liver cirrhosis are on the rise in Europe [
1]. Hepatic encephalopathy (HE) is one of the most debilitating complications of cirrhosis and is associated with increased morbidity and mortality [
2]. Although the pathogenesis is still incompletely understood, currently, it is assumed that protein and urea breakdown by colonic bacteria leads to ammonia release, and due to the reduced capacity of the liver to detoxify, ammonia accumulates and is shunted into the systemic circulation [
3,
4]. Ammonia accumulates in the brain and has neurotoxic effects, especially on astrocytes [
5,
6]. Available treatment modalities to lower ammonia include L-ornithine-L-aspartate (LOLA), nonabsorbable disaccharides (lactulose) and nonabsorbable antibiotics (rifaximin) [
7]. The amino acids L-ornithine and L-aspartate in LOLA dissociate readily and are consecutively absorbed.
L-ornithine serves as an intermediary in the urea cycle in periportal hepatocytes in the liver and as an activator of carbamoyl phosphate synthetase, and both amino acids also lower ammonia levels by transamination to glutamate via glutamine synthetase in perivenous hepatocytes as well as by skeletal muscle and the brain [
8]. Besides these direct effects, additional functions such as hepatoprotective effects or effects on restoring skeletal muscle proteostasis have been proposed but are not fully explained to date [
9]. Since oral LOLA comes into contact with the human gut microbiome and the intestinal barrier, effects on the gut microbiome and intestinal permeability may play a role in the clinical effects of LOLA. The gut microbiome is not only involved in ammonia generation and thereby in the pathogenesis of HE, but it is today also understood as a key driver of complications of cirrhosis [
10]. The gut microbiome is severely altered in liver cirrhosis in general, with a reduction in bacterial diversity and an increase in potential pathogens [
11]. Factors influencing the microbiome in cirrhosis are etiology and severity of liver disease, drug intake, nutritional status, and inflammation [
12]. In general, many human-targeted drugs can alter the composition of the gut microbiome [
13]. For LOLA thus far, no data are available on its association with gut microbiome composition and metabolomic changes in cirrhosis. Therefore, we aimed to investigate whether LOLA intake in patients with liver cirrhosis is associated with taxonomic and functional changes of the gut microbiome and the urinary, and/or serum and/or fecal metabolome.
2. Materials and Methods
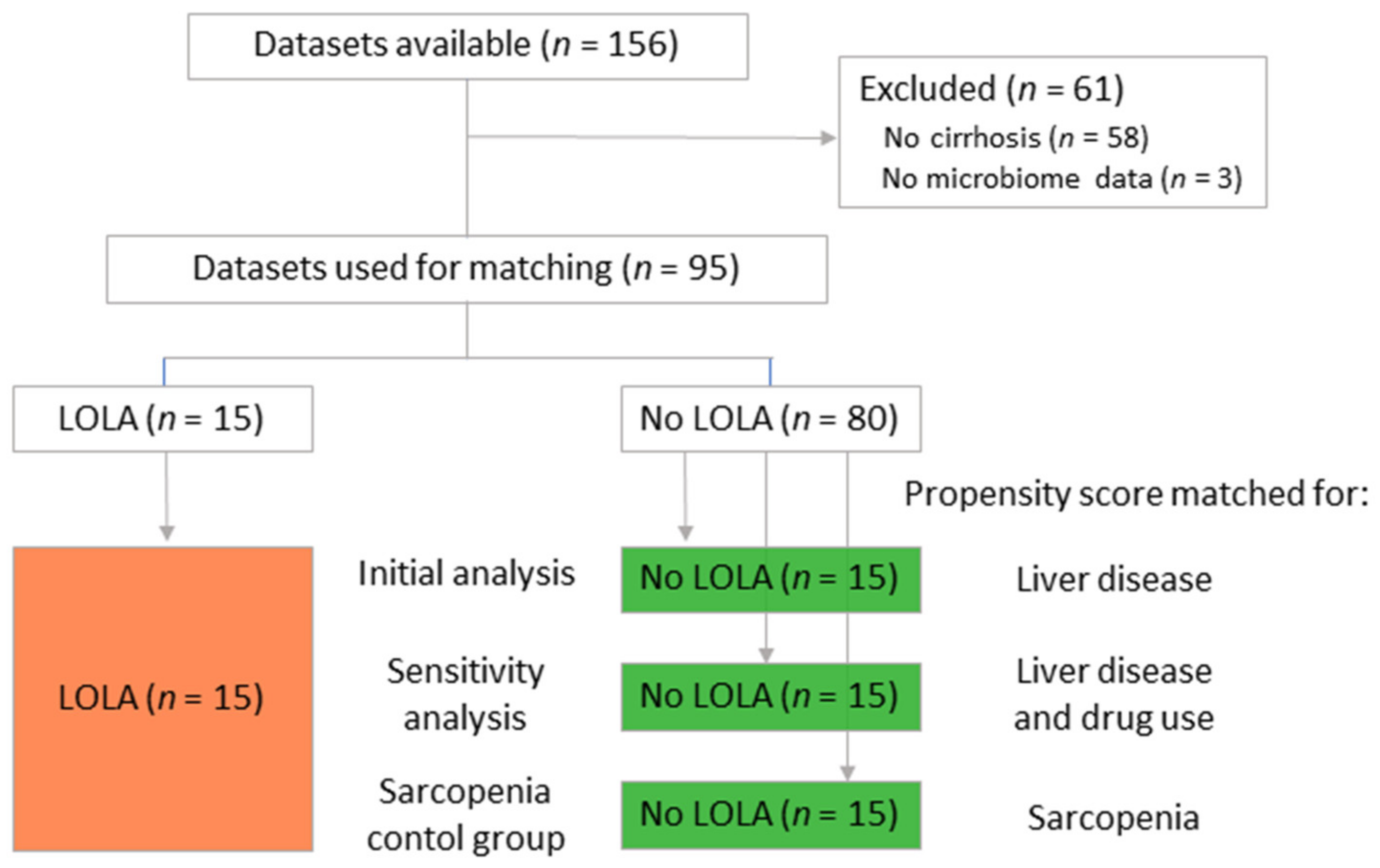
From an ongoing cohort study (NCT03080129, 29-280 ex 16/17), 15 patients with cirrhosis with oral LOLA intake for at least 1 month and 15 control cirrhotic patients were selected with nearest-neighbor propensity score matching. Propensity score matching was based on cirrhosis severity (Child-Pugh score and MELD-score), using the R package “MatchIt” [
14,
15]. To avoid overfitting and false positive results, results were validated by comparing the patients receiving LOLA to a second control group. This control group was selected by extending the regression model for nearest neighbor propensity score matching to include etiology of cirrhosis and proton pump inhibitor use in addition to disease severity. Due to this alteration, 9 out of 15 control patients (60%) were replaced. Sarcopenia is a potential confounder in the analysis too. To approximate its influence, LOLA patients were propensity score matched to a third control group of patients not receiving LOLA based on their sarcopenia status (non-sarcopenic, pre-sarcopenic, sarcopenic). To minimize confounding by potentially unbalanced parameters in regard to liver disease, this group comparison was only performed for sarcopenia related biomarkers with Mann–Whitney U tests.
Then, 16S rDNA sequencing data (for details regarding the method, see
Supplementary Methods) of these 30 patients were preprocessed with the QIIME 2 pipeline on a local Galaxy instance (
https://galaxy.medunigraz.at, accessed on 21 September 2021) [
16] and analyzed on the web-based platform “Calypso” V8.84 (
https://cgenome.net, accessed on 25 September 2021) [
17]. Taxonomic analysis was performed with a Bayesian classifier based on the SILVA V132 database. Low abundance filtering was applied to remove features that were present in only one sample or had less than 10 copies in total; cyanobacteria and chloroplasts were removed as likely contaminants. For alpha diversity approximations, a rarefied feature table with 8427 reads per sample was used to calculate richness, evenness, Chao-1 index and Shannon index. For beta diversity analysis and taxon comparisons, a full (not rarefied) feature table was subjected to Hellinger transformation (i.e., total sum scaling and square root transformation). Principal coordinate analysis (PCoA) with subsequent analysis of similarities (ANOSIM) based on Bray–Curtis dissimilarity as well as redundancy analysis (RDA) and non-parametric multidimensional scaling (NMDS) was used to identify similarities of microbiome structures between groups. LDA effect size (LEfSe) and analysis of compositions of microbiomes (ANCOM) identified differentially abundant taxa between the groups.
Nuclear magnetic resonance (NMR) metabolomic profiling was performed for urine, serum and stool samples using a well-established pipeline (for details regarding the method, see
Supplementary Methods) [
18,
19]. Metabolites were extracted using methanol, NMR spectra were recorded and processed in Matlab to obtain aligned and normalized datasets. Valid datasets were obtained from 29 patients (14 LOLA, 15 noLOLA), 27 patients (13 LOLA, 14 noLOLA) and 27 patients (13 LOLA, 14 noLOLA). The human metabolome database (HMDB) was used for metabolite annotation. Obtained concentrations were then normalized to the sum of all concentrations in a sample, square root transformed, mean centered and divided by the standard deviation of the respective feature for statistical analysis. Principal component analysis (PCA) was performed to visually assess the similarity of metabolome profiles between groups. To further discriminate groups, biomarker identification was performed based on area under the receiver operator characteristics curve (AUROC). For biomarker identification, the 20 ratios of metabolites with the lowest
p value in group comparison tests were included in the list of analytes. Analysis was performed in the web-based version of MetaboAnalyst 5.0 (
https://www.metaboanalyst.ca, accessed on 28 September 2021) [
20].
Targeted metabolomics were compared between groups, whereby routine and experimental parameters were selected to complement the NMR results. Parameters include routine biomarkers for liver function/injury and related information: alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (AP), gamma-glutamyltransferase (GGT), albumin, bilirubin, prothrombin time international normalized ratio (PZINR), C-reactive protein and total protein biomarkers for inflammation and gut permeability: fecal calprotectin, fecal zonulin, diamine oxidase (DAO), LPS binding protein and sCD14 indicators for sarcopenia: fibroblast growth factor (FGF) 21, irisin, myostatin and insulin-like growth factor (IGF)—1 biomarkers for neutrophil function: resting burst, priming and ROS production after
E. coli stimulation, all given as percentage of positive cells and geometric mean of fluorescence intensity (GMFI) [
20].
Biomarkers for liver function and injury as well as C-reactive protein were taken from the routine biochemistry report of the patients, fecal zonulin and fecal calprotectin were measured externally by Biovis’ Diagnostik MVZ GmbH (Limburg–Offenheim, Germany); neutrophil function was assessed by flow cytometry in heparinized whole blood using Phagoburst kits (Celonic, Basel, Switzerland) in house (for details, see
Supplementary Methods), the remaining biomarkers were assessed in house by ELISA (diamine oxidase and myostatin: Immundiagnostik, Bensheim, Germany; insulin-like growth factor-1 and fibroblast growth factor-21: Biotechne, Minneapolis, MN, USA; Irisin: Biovendor, Brno, Czeck Republic; LPS binding protein: Hycult Biotech, Uden, The Netherlands; sCD14: R&D systems, Minneapolis, MN, USA). All tests were performed according to manufacturers’ instructions. In addition, muscle function and muscle mass assessments (chair rise test, gait speed, midarm muscle circumference, hand grip strength and body mass index) were included in the analysis.
All analyses and visualizations, if not otherwise stated in the Method section, were performed with R (version 4.0.3) in R studio (version 1.4.1103) using the packages “tidyverse”, “readxl”, “writexl”, and “ggpubr” [
14,
21,
22,
23,
24].
4. Discussion
Our retrospective analysis of 15 patients with oral LOLA intake for at least one month and propensity-score-matched controls showed that patients with LOLA intake show a higher abundance of the genera Flavonifractor and Oscillospira in the intestinal microbiome, a reduced ratio of ethanol to acetic acid in urine, an increased ratio of propylene glycol to isopropyl alcohol in stool, increased ratio of propylene glycol to valeric acid in stool, decreased ratio of valeric acid to glycerol in stool, increased levels of ornithine and decreased levels of leucine and isoleucine as well as lower levels of IGF-1 in serum.
Flavonifractor and
Oscillospira are rather closely related bacterial taxa in the human intestinal microbiome. According to the Silva database, both genera are classified as Firmicutes (phylum), Clostridia (Class), Oscillospirales (order), Oscillospiraceae (family).
Flavonifractor that can be induced by green tea consumption and can exert anti-inflammatory properties in murine DSS colitis models [
25]. In humans, the role of the genus
Flavonifractor is still undetermined. Its abundance was previously associated with the consumption of Mediterranean diet [
26], which is generally regarded as beneficial for human health. In non-alcoholic liver disease,
Flavonifractor consistently showed low abundance compared to healthy controls [
27]. In addition, according to the GMrepo database,
Flavonifractor is more abundant in healthy volunteers than in cirrhosis, non-alcoholic fatty liver disease, obesity/adiposity, and diabetes mellitus (topic-related selection) [
28]. Conversely,
Flavonifractor has been associated with the microbiome of colorectal cancer patients in India [
29].
Oscillospira has previously been associated with low BMI, constipation, longer sleep time, higher HDL levels, lower uric acid and triglyceride levels [
30,
31]. In a previous study, we showed that lower HDL levels are predictive for the development of complications in patients with compensated cirrhosis and predictive for survival in cirrhotic patients with acute decompensation [
32,
33]. Taken together, the alterations in the microbiome of LOLA patients could generally be regarded as beneficial for patients with liver disease. However, due to the retrospective design and the low sample size, confounding factors cannot be ruled out, e.g., although not statistically significant, patients in the LOLA group more often also received rifaximin.
In the urine metabolome, patients in the LOLA group showed lower ethanol to acetic acid ratio. The ethanol to acetic acid ratio might be an indicator for the ethanol elimination process. When alcohol is consumed, it is converted to acetaldehyde—a toxic intermediate—and then quickly reduced to acetate. A reduced ratio of ethanol to acetic acid might indicate a more advanced state in the ethanol elimination process, either because of an earlier consumption time or accelerated conversion. Both patient groups reported similar amounts of alcohol intake and did not show any difference in serum or urine alcohol levels. An earlier starting point would therefore necessitate a higher level of intoxication in the LOLA group, which was not reported by the patients. However, in patients with present or past alcohol use disorder, alcohol consumption is notoriously hard to approximate, since some patients tend to embellish their consumption [
34]. Appropriately powered and designed studies are necessary to properly explain the reduced ethanol to acetic acid ratio in the urine metabolome of LOLA patients and to further explore the possible effects of LOLA on the ethanol elimination rate in cirrhotic patients. In the stool microbiome, the most prominent biomarker was the propylene glycol to isopropyl alcohol ratio. Propylene glycol is naturally occurring in mushrooms and sesame seed but is also ubiquitously used in the cosmetic, food and drug industries. Isopropyl alcohol is a common substance in various nutritious items, including apples and onions, which are a crucial part of the local cuisine of the patients´ catchment area. The relevance in health and disease is undetermined at the moment. An increase in this ratio in LOLA patients might indicate an increase in therapeutics and decrease in fresh food consumption. However, this exploratory study is not designed to answer this question. Valeric acid is a bacterial metabolite, among others produced by
Flavonifractor plautii as a product of flavonoid degradation [
35]. This short chain fatty acid can also be produced by odd-chain elongation of ethanol and propionate or by hydrolysis of valerate esters used as fruity tasting food additives [
36]. We observed low levels of valeric acid to glycerol ratio in the stool metabolome of LOLA patients, which is consistent with previous observations of low valerate levels in patients with recurring hepatic encephalopathy and might therefore reflect the disease rather than a therapy-related change in the metabolome [
37]. Similarly, serum marker reflect a typical decrease in branched chained amino acids in patients with hepatic encephalopathy [
38]. The lack of isoleucine probably would have been even more pronounced if the patients were not treated with LOLA, since LOLA supposedly increases isoleucine levels in serum [
39]. Moreover, the increased levels of ornithine confirm therapy adherence of the patients and adequate reabsorption of oral LOLA in these patients [
40].
IGF-1 was consistently lower in patients with LOLA, although we corrected for liver disease and sarcopenia in two separate matched cohort. In this pilot study, we could not discern the reason for this decrease nor could we find probable mechanistic indicators. It is likely that the pathophysiological changes in patients with hepatic encephalopathy, especially the high levels of ammonia, negatively influence muscle metabolism as it was previously shown in pufferfish and suggested in cirrhosis and hepatic encephalopathy [
41,
42].
In conclusion, patients with LOLA therapy for at least one month showed a potentially beneficial increase in Flavonifractor and Oscillospira in their microbiome compared to patients without LOLA therapy. Changes in the stool and serum metabolome mainly reflected pathophysiological changes of hepatic encephalopathy and identified possible therapeutic targets, such as valeric acid that is reduced in patients with hepatic encephalopathy but whose microbial production might be elevated by LOLA-associated changes in Flavonifractor abundance. Lastly, low levels of IGF-1 demonstrated a link between hepatic encephalopathy and muscle metabolism, a potential new avenue for LOLA use in liver cirrhotic patients.