Goal-Setting in Multiple Sclerosis-Related Spasticity Treated with Botulinum Toxin: The GASEPTOX Study
Abstract
:1. Introduction
2. Results
2.1. Patient Characteristics
2.2. Description of Goal-Setting
2.3. Goals Distribution by Lower or Upper Limb
2.4. Goals Description According to Descriptive Data
2.5. BoNT-A Efficacy and Goal Attainment
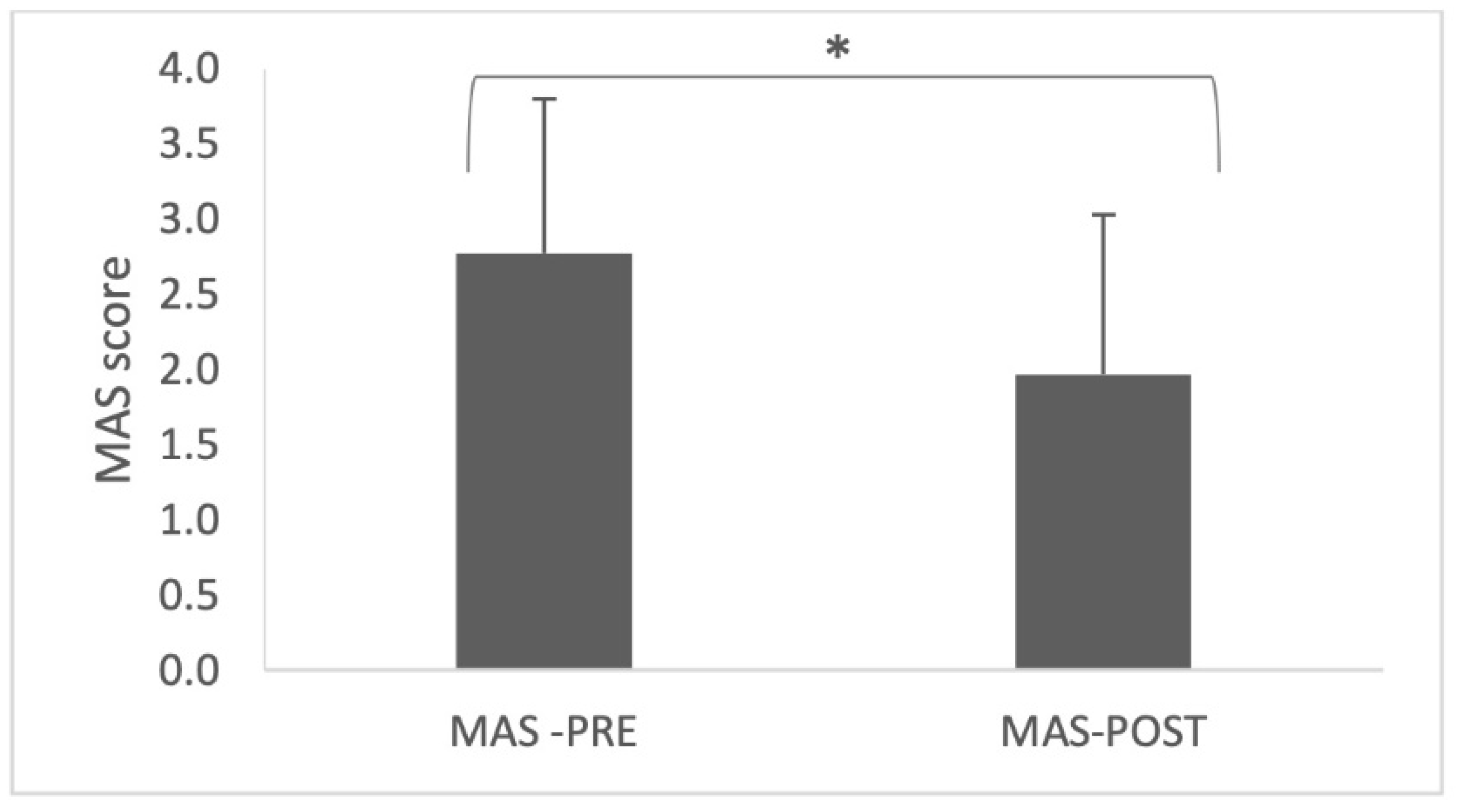
2.6. Pre-Post Spasticity Assessment (MAS)
3. Discussion
4. Conclusions
5. Future Perspectives
6. Methods
6.1. Study Design and Setting
6.2. Study Population
6.3. Defining Therapeutic Goals
6.4. Botulinum Toxin Injections and Follow-Up
6.5. Data Collection
6.6. Statistical Analysis
6.7. Ethics
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BoNT-A | botulinum neurotoxin type A |
| BTI | botulinum toxin injection |
| EDSS | expanded disability status scale |
| ICF | international classification of function |
| GAS | goal attainment scaling |
| IQ | interquartile |
| LL | lower limb |
| MAS | modified Ashworth scale |
| MR004 | méthodologie de référence 004 |
| MS | multiple sclerosis |
| PP | primary progressive |
| RR | relapsing remitting |
| SD | standard deviation |
| SMART | specific, measurable, achievable, relevant, timed |
| SP | secondary progressive |
| UL | upper limb |
References
- Kamm, C.P.; Uitdehaag, B.M.; Polman, C.H. Multiple sclerosis: Current knowledge and future outlook. Eur. Neurol. 2014, 72, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Compston, A.; Coles, A. Multiple sclerosis. Lancet 2008, 372, 1502–1517. [Google Scholar] [CrossRef]
- Strijbis, E.M.; Repovic, P.; Mostert, J.; Bowen, J.D.; Uitdehaag, B.M.; Cutter, G.; Koch, M.W. The MSIS-29 and SF-36 as outcomes in secondary progressive MS trials. Mult. Scler. 2022, 28, 1606–1619. [Google Scholar] [CrossRef] [PubMed]
- Lance, J.W. The Control of muscle tone, reflexes, and movement: Robert Wartenberg lecture. Neurology 1980, 30, 1303–1313. [Google Scholar] [CrossRef]
- Pandyan, A.; Gregoric, M.; Barnes, M.; Wood, D.; Wijck, F.V.; Burridge, J.; Hermens, H.; Johnson, G. Spasticity: Clinical perceptions, neurological realities and meaningful measurement. Disabil. Rehabil. 2005, 27, 2–6. [Google Scholar] [CrossRef]
- Rizzo, M.A.; Hadjimichael, O.C.; Preiningerova, J.; Vollmer, T.L. Prevalence and treatment of spasticity reported by multiple sclerosis patients. Mult. Scler. 2004, 10, 589–595. [Google Scholar] [CrossRef]
- Safarpour, Y.; Mousavi, T.; Jabbari, B. Botulinum toxin treatment in multiple sclerosis—A review. Curr. Treat. Options Neurol. 2017, 19, 33. [Google Scholar] [CrossRef]
- Sosnoff, J.J.; Gappmaier, E.; Frame, A.; Motl, R.W. Influence of spasticity on mobility and balance in persons with multiple sclerosis. J. Neurol. Phys. Ther. 2011, 35, 129–132. [Google Scholar] [CrossRef]
- Bensmail, D.; Vermersch, P. Epidemiology and clinical assessment of spasticity in multiple sclerosis. Rev. Neurol. 2012, 168 (Suppl. 3), S45–S50. [Google Scholar] [CrossRef]
- Food and Drug Administration. BOTOX (OnabotulinumtoxinA) for Injection, for Intramuscular, Intradetrusor, or Intradermal Use. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/103000s5318lbl.pdf (accessed on 7 September 2021).
- Snow, B.J.; Tsui, J.K.; Bhatt, M.H.; Varelas, M.; Hashimoto, S.A.; Calne, D.B. Treatment of spasticity with botulinum toxin: A double-blind study. Ann. Neurol 1990, 28, 512–515. [Google Scholar] [CrossRef]
- Grazko, M.A.; Polo, K.B.; Jabbari, B. Botulinum toxin A for spasticity, muscle spasms, and rigidity. Neurology 1995, 45, 712–717. [Google Scholar] [CrossRef]
- Hyman, N.; Barnes, M.; Bhakta, B.; Cozens, A.; Bakheit, M.; Kreczy-Kleedorfer, B.; Poewe, W.; Wissel, J.; Bain, P.; Glickman, S.; et al. Botulinum toxin (dysport) treatment of hip adductor spasticity in multiple sclerosis: A prospective, randomised, double blind, placebo controlled, dose ranging study. J. Neurol. Neurosurg. Psychiatr. 2000, 68, 707–712. [Google Scholar] [CrossRef]
- Paoloni, M.; Giovannelli, M.; Mangone, M.; Leonardi, L.; Tavernese, E.; Di Pangrazio, E.; Bernetti, A.; Santilli, V.; Pozzilli, C. Does giving segmental muscle vibration alter the response to botulinum toxin injections in the treatment of spasticity in people with multiple sclerosis? A single-blind randomized controlled trial. Clin. Rehabil. 2013, 27, 803–812. [Google Scholar] [CrossRef]
- Giovannelli, M.; Borriello, G.; Castri, P.; Prosperini, L.; Pozzilli, C. Early physiotherapy after injection of botulinum toxin increases the beneficial effects on spasticity in patients with multiple sclerosis. Clin. Rehabil. 2007, 21, 331–337. [Google Scholar] [CrossRef]
- Barnes, M.; Schnitzler, A.; Medeiros, L.; Aguilar, M.; Lehnert-Batar, A.; Minnasch, P. Efficacy and safety of NT 201 for upper limb spasticity of various etiologies—A randomized parallel-group study: NT 201 in upper limb spasticity. Acta Neurol. Scand. 2010, 122, 295–302. [Google Scholar] [CrossRef]
- Levy, J.; Molteni, F.; Cannaviello, G.; Lansaman, T.; Roche, N.; Bensmail, D. Does botulinum toxin treatment improve upper limb active function? Ann. Phys. Rehabil. Med. 2019, 62, 234–240. [Google Scholar] [CrossRef]
- Ashford, S.; Turner-Stokes, L. Management of shoulder and proximal upper limb spasticity using botulinum toxin and concurrent therapy interventions: A preliminary analysis of goals and outcomes. Disabil. Rehabil. 2009, 31, 220–226. [Google Scholar] [CrossRef]
- Turner-Stokes, L.; Fheodoroff, K.; Jacinto, J.; Maisonobe, P.; Zakine, B. Upper limb international spasticity study: Rationale and protocol for a large, international, multicentre prospective cohort study investigating management and goal attainment following treatment with botulinum toxin A in real-life clinical practice. BMJ Open 2013, 3, e002230. [Google Scholar] [CrossRef] [Green Version]
- Turner-Stokes, L.; Jacinto, J.; Fheodoroff, K.; Brashear, A.; Maisonobe, P.; Lysandropoulos, A.; Ashford, S. Upper limb international spasticity (ULIS-III) study group longitudinal goal attainment with integrated upper limb spasticity management including repeat injections of botulinum toxin A: Findings from the prospective, observational upper limb international spasticity (ULIS-III) cohort study. J. Rehabil. Med. 2021, 53, jrm00157. [Google Scholar] [CrossRef]
- Fheodoroff, K.; Ashford, S.; Jacinto, J.; Maisonobe, P.; Balcaitiene, J.; Turner-Stokes, L. Factors influencing goal attainment in patients with post-stroke upper limb spasticity following treatment with botulinum toxin A in real-life clinical practice: Sub-analyses from the upper limb international spasticity (ULIS)-II study. Toxins 2015, 7, 1192–1205. [Google Scholar] [CrossRef] [Green Version]
- Turner-Stokes, L.; Jacinto, J.; Fheodoroff, K.; Brashear, A.; Maisonobe, P.; Lysandropoulos, A.; Ashford, S. Upper limb international spasticity-III (ULIS-III) study group assessing the effectiveness of upper-limb spasticity management using a structured approach to goal-setting and outcome measurement: First cycle results from the ULIS-III study. J. Rehabil. Med. 2021, 53, jrm00133. [Google Scholar] [CrossRef] [PubMed]
- Ashford, S.; Turner-Stokes, L. Systematic review of upper-limb function measurement methods in botulinum toxin intervention for focal spasticity. Physiother. Res. Int. 2013, 18, 178–189. [Google Scholar] [CrossRef]
- Krasny-Pacini, A.; Hiebel, J.; Pauly, F.; Godon, S.; Chevignard, M. Goal attainment scaling in rehabilitation: A literature-based update. Ann. Phys. Rehabil. Med. 2013, 56, 212–230. [Google Scholar] [CrossRef] [PubMed]
- Kiresuk, T.J.; Sherman, R.E. Goal attainment scaling: A general method for evaluating comprehensive community mental health programs. Community Ment. Health J. 1968, 4, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Turner-Stokes, L. Goal attainment scaling (GAS) in rehabilitation: A practical guide. Clin. Rehabil. 2009, 23, 362–370. [Google Scholar] [CrossRef] [PubMed]
- Ashford, S.; Turner-Stokes, L. Goal attainment for spasticity management using botulinum toxin. Physiother. Res. Int. 2006, 11, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Krasny-Pacini, A.; Evans, J.; Sohlberg, M.M.; Chevignard, M. Proposed criteria for appraising goal attainment scales used as outcome measures in rehabilitation research. Arch. Phys. Med. Rehabil. 2016, 97, 157–170. [Google Scholar] [CrossRef] [Green Version]
- Krasny-Pacini, A.; Pauly, F.; Hiebel, J.; Godon, S.; Isner-Horobeti, M.-E.; Chevignard, M. Feasibility of a shorter goal attainment scaling method for a pediatric spasticity clinic—The 3-milestones GAS. Ann. Phys. Rehabil. Med. 2017, 60, 249–257. [Google Scholar] [CrossRef]
- Ashford, S.; Jackson, D.; Turner-Stokes, L. Goal setting, using goal attainment scaling, as a method to identify patient selected items for measuring arm function. Physiotherapy 2015, 101, 88–94. [Google Scholar] [CrossRef]
- Turner-Stokes, L.; Ashford, S.; Jacinto, J.; Maisonobe, P.; Balcaitiene, J.; Fheodoroff, K. Impact of integrated upper limb spasticity management including botulinum toxin A on patient-centred goal attainment: Rationale and protocol for an international prospective, longitudinal cohort study (ULIS-III). BMJ Open 2016, 6, e011157. [Google Scholar] [CrossRef] [Green Version]
- Hemmett, L.; Holmes, J.; Barnes, M.; Russell, N. What drives quality of life in multiple sclerosis? QJM An. Int. J. Med. 2004, 97, 671–676. [Google Scholar] [CrossRef]
- Stevens, V.; Goodman, K.; Rough, K.; Kraft, G.H. Gait impairment and optimizing mobility in multiple sclerosis. Phys. Med. Rehabil. Clin. N. Am. 2013, 24, 573–592. [Google Scholar] [CrossRef]
- Bensmail, D.; Ward, A.B.; Wissel, J.; Motta, F.; Saltuari, L.; Lissens, J.; Cros, S.; Beresniak, A. Cost-effectiveness modeling of intrathecal baclofen therapy versus other interventions for disabling spasticity. Neurorehabil. Neural. Repair 2009, 23, 546–552. [Google Scholar] [CrossRef]
- Jankovic, J. Botulinum toxin in clinical practice. J. Neurol. Neurosurg. Psychiatr. 2004, 75, 951–957. [Google Scholar] [CrossRef]
- Norbye, A.D.; Midgard, R.; Thrane, G. Spasticity, gait, and balance in patients with multiple sclerosis: A cross-sectional study. Physiother. Res. Int. 2020, 25, e1799. [Google Scholar] [CrossRef]
- Bovend’Eerdt, T.J.; Botell, R.E.; Wade, D.T. Writing SMART rehabilitation goals and achieving goal attainment scaling: A practical guide. Clin. Rehabil. 2009, 23, 352–361. [Google Scholar] [CrossRef]
- Turner-Stokes, L.; Ashford, S.; Esquenazi, A.; Wissel, J.; Ward, A.B.; Francisco, G.; Lains, J.; Suputtitada, A.; Serrano, S.; Baguley, I.J.; et al. A comprehensive person-centred approach to adult spastic paresis: A consensus-based framework. Eur. J. Phys. Rehabil. Med. 2017, 54, 605–617. [Google Scholar] [CrossRef]
- Khan, F.; Pallant, J. Use of International classification of functioning, disability and health (ICF) to describe patient-reported disability in multiple sclerosis and identification of relevant environmental factors. J. Rehabil. Med. 2007, 39, 63–70. [Google Scholar] [CrossRef] [Green Version]
- Khan, F.; Pallant, J.F.; Turner-Stokes, L. Use of goal attainment scaling in inpatient rehabilitation for persons with multiple sclerosis. Arch. Phys. Med. Rehabil. 2008, 89, 652–659. [Google Scholar] [CrossRef]
- Thompson, A.J.; Banwell, B.L.; Barkhof, F.; Carroll, W.M.; Coetzee, T.; Comi, G.; Correale, J.; Fazekas, F.; Filippi, M.; Freedman, M.S.; et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018, 17, 162–173. [Google Scholar] [CrossRef]
- Yelnik, A.P.; Simon, O.; Bensmail, D.; Chaleat-Valayer, E.; Decq, P.; Dehail, P.; Quentin, V.; Marque, P.; Parratte, B.; Pellas, F.; et al. Drug treatments for spasticity. Ann. Phys. Rehabil. Med. 2009, 52, 746–756. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kurtzke, J.F. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 1983, 33, 1444–1452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bohannon, R.W.; Smith, M.B. Interrater reliability of a modified ashworth scale of muscle spasticity. Phys. Ther. 1987, 67, 206–207. [Google Scholar] [CrossRef] [PubMed]
| General | Center 1 | Center 2 | ||
|---|---|---|---|---|
| N | 125 | 81 | 44 | |
| Sex | 36 male 85 female | 25 male 52 female | 11 male 33 female | |
| Age (year) | Mean ± SD | 54.9 ± 11.1 | 56.0 ± 10.4 | 53.0 ± 12.0 |
| Disease evolution (year) | Mean ± SD | 18.8 ± 9.7 | 18.8 ± 9.6 | 19 ±10.1 |
| EDSS | median | 6 | 6 | 6 |
| IQ | [4; 6.5] | [4.5; 6.5] | [3; 7] | |
| MS phenotype | SP: 73 PP: 22 RR: 30 | SP: 51 PP: 15 RR: 15 | SP: 22 PP: 7 RR: 15 |
| Goals | Percentage |
|---|---|
| Reduce the clinging of the ground | 9.87% |
| Improve stability during ground support | 9.87% |
| Reduce a toe claw to ease gait | 7.73% |
| Reduce an equinus disturbing gait | 6.67% |
| Improve walking perimeter | 4.00% |
| Reduce a knee recurvatum in the support phase | 3.47% |
| Other LL | 3.20% |
| Improve balance | 2.93% |
| Reduce a disturbing dystonia in hallux extension | 2.40% |
| Reduce an equinus varus disturbing gait | 2.40% |
| Prevent or limit retractions | 1.87% |
| Improve walking speed | 1.87% |
| Facilitate transfers | 1.87% |
| Reduce spontaneous pain or mobilization pain | 1.60% |
| Reduce knee flessum | 1.60% |
| Reduce painful toe claw | 1.33% |
| Facilitate the positioning of the feet on the wheelchair pallets | 1.07% |
| Reduce a hip adductum disturbing the toilet, the dressing… | 1.07% |
| Prevent or limit retractions | 1.07% |
| Reduce spontaneous or mobilization pain | 1.07% |
| Facilitate shoulder abduction | 0.80% |
| Facilitate the use of the hand as an auxiliary hand | 0.80% |
| Improve knee flexion in the oscillating phase | 0.80% |
| Other UL | 0.80% |
| Facilitate the passive opening of the hand | 0.53% |
| Facilitate dressing, hygiene, nursing | 0.53% |
| Reduce a disturbing attitude of the upper limb when walking or in other situations | 0.53% |
| Reduce a hip adductum disturbing gait | 0.53% |
| Facilitate the active opening of the hand to improve prehension | 0.27% |
| Reduce hand maceration | 0.27% |
| Reduce access to walking aids | 0.27% |
| Facilitate knee flexion in various situations such as sitting in the wheelchair | 0.27% |
| Facilitate shoeing | 0.27% |
| Facilitate the use of the hand in other activities (computer keyboard, tablets…) | 0.00% |
| Facilitate the wearing of a resting orthosis | 0.00% |
| Facilitate access to the perineum (probes, sexuality, toilet, hygiene…) | 0.00% |
| GAS Goal 1 Percentage | GAS Goal 2 Percentage | GAS Goal 3 Percentage | |
|---|---|---|---|
| Facilitate shoulder abduction | 1.6% | 0.8% | |
| Facilitate the active opening of the hand to improve prehension | 0.8% | ||
| Facilitate the use of the hand in other activities (computer keyboard, tablets…) | |||
| Facilitating the use of the hand as an auxiliary hand | 0.8% | 1.6% | |
| Facilitate the passive opening of the hand | 1.6% | ||
| Reduce hand maceration | 0.8% | ||
| Prevent or limit retractions | 2.4% | 2.4% | 0.8% |
| Facilitate the wearing of a resting orthosis | |||
| Facilitate dressing, hygiene, nursing | 0.8% | 0.8% | |
| Reduce spontaneous or mobilization pain | 1.6% | 1.6% | 1.6% |
| Reduce a disturbing attitude of the upper limb when walking or in other situations | 0.8% | 0.8% | |
| Reduce the clinging of the ground | 18.4% | 8.8% | 2.4% |
| To improve balance | 2.4% | 4.8% | 1.6% |
| To improve stability during ground support | 16.8% | 10.4% | 2.4% |
| Improve walking speed | 2.4% | 1.6% | 1.6% |
| Improve walking perimeter | 8.0% | 2.4% | 1.6% |
| Reduce access to walking aids | 0.8% | ||
| Reduce a toe claw to ease gait | 5.6% | 4.0% | 13.6% |
| Reduce painful toe claw | 1.6% | 0.8% | 1.6% |
| Reduce a disturbing dystonia in extension of the hallux | 3.2% | 1.6% | 2.4% |
| Reduce an equinus disturbing gait | 8.0% | 9.6% | 2.4% |
| Reduce an equinus varus disturbing gait | 2.4% | 3.2% | 1.6% |
| Facilitate the positioning of the feet on the wheelchair pallets | 1.6% | 1.6% | |
| Reduce a hip adductum disturbing gait | 1.6% | ||
| Reduce a hip adductum disturbing the toilet, dressing… | 2.4% | 0.8% | |
| Facilitate access to the perineum (probes, sexuality, toilet, hygiene…) | |||
| Reduce a knee recurvatum in the support phase | 2.4% | 6.4% | 1.6% |
| Improve knee flexion in the oscillating phase | 0.8% | 1.6% | |
| Facilitate knee flexion in various situations such as sitting in the wheelchair | 0.8% | ||
| Reduce knee flessum | 4.0% | 0.8% | |
| Facilitate shoeing | 0.8% | ||
| Facilitate transfers | 0.8% | 2.4% | 2.4% |
| Prevent or limit retractions | 0.8% | 1.6% | 0.8% |
| Reduce spontaneous or mobilization pain | 1.6% | 0.8% | 0.8% |
| Other UL | 2.4% | ||
| Other LL | 3.2% | 0.8% | 5.6% |
| LL | UL | ||
|---|---|---|---|
| General | 89.1% | 10.9% | |
| Center | Center 1 Center 2 | 88.8% 90.2% | 11.2% 9.8% |
| EDSS | [2; 5.5] [6; 7.5] [8; 8.5] | 100% 89.8% 44.1% | 0% 10.2% 55.9% |
| Sex | M F | 88.8% 88.6% | 11.2% 11.4% |
| MS phenotype | PP RR SP | 85.1% 100% 86.3% | 14.9% 0% 13.7% |
| Sex | Male | Female | |||||
|---|---|---|---|---|---|---|---|
| Reduce a toe claw to ease gait | 12.0% | Improve stability during ground support | 9.8% | ||||
| Reduce the clinging of the ground | 10.2% | Reduce the clinging of the ground | 9.0% | ||||
| Improve stability during ground support | 9.3% | Reduce an equinus disturbing gait | 6.7% | ||||
| Center | Center 1 | Center 2 | |||||
| Improve stability during ground support | 14.8% | Improve walking perimeter | 9.1% | ||||
| Reduce a toe claw to ease the gait | 11.9% | Reduce the clinging of the ground | 6.8% | ||||
| Reduce the clinging of the ground | 11.5% | Improve balance | 3.8% | ||||
| EDSS | [2; 5.5] | [6; 7.5] | [8; 8.5] | ||||
| Reduce the clinging of the ground | 16.1% | Improve stability when ground support | 11.3% | Prevent or limit retractions | 12.5% | ||
| Improve stability during ground support | 11.5% | Reduce a toe claw to ease gait | 9.3% | Other UL | 8.3% | ||
| Reduce a toe claw to ease the gait | 8.6% | Reduce an equinus disturbing gait | 8.7% | Facilitate shoulder abduction | 6.3% | ||
| MS Phenotype | PP | RR | SP | ||||
| Reduce the clinging of the ground | 16.4% | Reduce the clinging of the ground | 16.7% | Improve stability during ground support | 11.4% | ||
| Improve stability during ground support | 9.1% | Improve stability during ground support | 6.7% | Reduce a toe claw to ease the gait | 8.2% | ||
| Reduce a toe claw to ease the gait | 9.1% | Improve walking perimeter | 6.7% | Reduce the clinging of the ground | 6.8% | ||
| Goals | UL/LL | Number of Goal Selected Times | Number of Goal Achieved Times | Percentage |
|---|---|---|---|---|
| Improve walking perimeter | LL | 12 | 12 | 100% |
| Reduce an equinus varus disturbing gait | LL | 9 | 9 | 100% |
| Reduce a disturbing dystonia in hallux extension | LL | 8 | 8 | 100% |
| Reduce knee flessum | LL | 6 | 6 | 100% |
| Prevent or limit retractions | UL | 5 | 5 | 100% |
| Prevent or limit retractions | LL | 4 | 4 | 100% |
| Facilitate shoulder abduction | UL | 3 | 3 | 100% |
| other | UL | 3 | 3 | 100% |
| Facilitate dressing, hygiene, nursing | UL | 2 | 2 | 100% |
| Reduce a hip adductum disturbing gait | LL | 2 | 2 | 100% |
| Facilitate the active opening of the hand to improve prehension | UL | 1 | 1 | 100% |
| Facilitate the passive opening of the hand | UL | 1 | 1 | 100% |
| Reduce hand maceration | UL | 1 | 1 | 100% |
| Reduce access to walking aids | LL | 1 | 1 | 100% |
| Facilitate knee flexion in various situations such as sitting in the wheelchair | LL | 1 | 1 | 100% |
| Facilitate shoeing | LL | 1 | 1 | 100% |
| Improve stability during ground support | LL | 31 | 29 | 93.5% |
| other | LL | 11 | 10 | 90.9% |
| Reduce a toe claw to ease the gait | LL | 26 | 23 | 88.5% |
| Facilitate transfers | LL | 7 | 6 | 85.7% |
| Reduce an equinus disturbing gait | LL | 23 | 19 | 82.6% |
| Reduce painful toe claw | LL | 5 | 4 | 80% |
| Reduce the clinging of the ground | LL | 29 | 23 | 79.3% |
| Reduce spontaneous pain or mobilization pain | UL | 4 | 3 | 75% |
| Facilitate the positioning of the feet on the wheelchair pallets | LL | 4 | 3 | 75% |
| Reduce a hip adductum disturbing the toilet, the dressing… | LL | 4 | 3 | 75% |
| Improve walking speed | LL | 6 | 4 | 66.7% |
| Facilitate the use of the hand as an auxiliary hand | UL | 3 | 2 | 66.7% |
| Reduce spontaneous or mobilization pain | LL | 3 | 2 | 66.7% |
| Reduce a knee recurvatum in the support phase | LL | 11 | 7 | 63.6% |
| Improve balance | LL | 8 | 5 | 62.5% |
| Reduce a disturbing attitude of the upper limb when walking or in other situations | UL | 2 | 1 | 50% |
| Improve knee flexion in the oscillating phase | LL | 2 | 1 | 50% |
| Facilitate the use of the hand in other activities (computer keyboard, tablets…) | UL | 0 | 0 | 0% |
| Facilitate the wearing of a resting orthosis | UL | 0 | 0 | 0% |
| Facilitate access to the perineum (probes, sexuality, toilet, hygiene…) | LL | 0 | 0 | 0% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baccouche, I.; Bensmail, D.; Leblong, E.; Fraudet, B.; Aymard, C.; Quintaine, V.; Pottier, S.; Lansaman, T.; Malot, C.; Gallien, P.; et al. Goal-Setting in Multiple Sclerosis-Related Spasticity Treated with Botulinum Toxin: The GASEPTOX Study. Toxins 2022, 14, 582. https://doi.org/10.3390/toxins14090582
Baccouche I, Bensmail D, Leblong E, Fraudet B, Aymard C, Quintaine V, Pottier S, Lansaman T, Malot C, Gallien P, et al. Goal-Setting in Multiple Sclerosis-Related Spasticity Treated with Botulinum Toxin: The GASEPTOX Study. Toxins. 2022; 14(9):582. https://doi.org/10.3390/toxins14090582
Chicago/Turabian StyleBaccouche, Ines, Djamel Bensmail, Emilie Leblong, Bastien Fraudet, Claire Aymard, Victorine Quintaine, Sandra Pottier, Thibaud Lansaman, Claire Malot, Philippe Gallien, and et al. 2022. "Goal-Setting in Multiple Sclerosis-Related Spasticity Treated with Botulinum Toxin: The GASEPTOX Study" Toxins 14, no. 9: 582. https://doi.org/10.3390/toxins14090582





