Mycotoxins in Cereal-Based Products and Their Impacts on the Health of Humans, Livestock Animals and Pets
Abstract
:1. Introduction
2. Common Mycotoxins in Cereal Grains and Their Producing Fungi
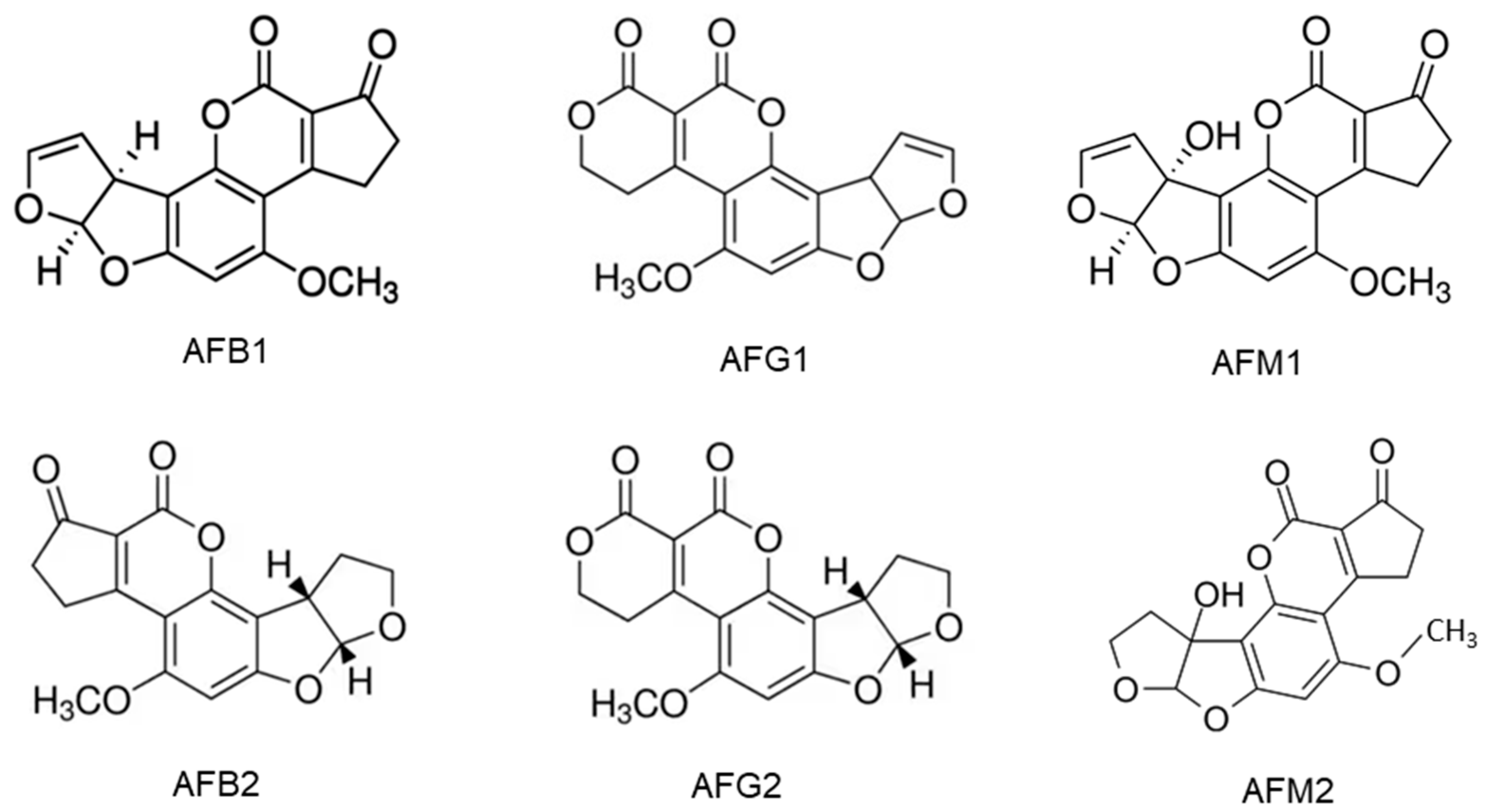
2.1. Aflatoxins and Their Producing Fungi
2.2. Ochratoxins and Their Producing Fungi
2.3. Zearalenone and Its Producing Fungi
2.4. Fumonisins and Their Producing Fungi
2.5. Trichothecenes and Their Producing Fungi
3. Occurrence and Co-Occurrence of Mycotoxins in Unprocessed Cereal Grains
3.1. Occurrence of Mycotoxins in Unprocessed Cereal Grains
3.2. Co-Occurrence of Mycotoxins in Unprocessed Cereal Grains
4. Occurrence and Co-Occurrence of Mycotoxins in Cereal-Based Products
4.1. Mycotoxin Contamination of Cereal-Based Foods for Human Consumption
4.1.1. Aflatoxins in Cereal-Based Foods
4.1.2. Ochratoxin A in Cereal-Based Foods
4.1.3. Fumonisins in Cereal-Based Foods
4.1.4. Trichothecenes in Cereal-Based Foods
4.1.5. Zearalenone in Cereal-Based Foods
4.2. Mycotoxin Contamination of Feed Products
5. Evidence of Mycotoxin Impacts on Human Health
5.1. Acute Mycotoxicosis in Human
5.1.1. Acute aflatoxicosis in human
5.1.2. Acute Mycotoxicoses in Human-Caused by Fusarium Mycotoxins
5.1.3. Acute Ochratoxicosis in Humans
5.2. Chronic Health Impact of Mycotoxins in Human
5.2.1. Chronic Health Impact of Aflatoxins
5.2.2. Chronic Health Impact of Ochratoxin A
5.2.3. Chronic Health Impact of Fumonisins
5.2.4. Chronic Health Impact of Trichothecene Mycotoxin
5.2.5. Chronic Health Impact of Zearalenone
6. Impact of Mycotoxins on Livestock Animals
6.1. Ruminants
6.1.1. Effects of Aflatoxins on Ruminants
6.1.2. Effects of OTA on Ruminants
6.1.3. Effects of Trichothecenes on Ruminants
6.2. Pigs
6.2.1. Effects of Aflatoxin on Pigs
6.2.2. Effects of OTA on Pigs
6.2.3. Effects of Fumonisins on Pigs
6.2.4. Effects of DON and ZEA on Pigs
6.3. Poultry
6.3.1. Effects of Aflatoxin on Poultry
6.3.2. Effects of OTA on Poultry
6.3.3. Effects of Fusarium Mycotoxins on Poultry
6.4. Effects of Mycotoxins on Pets
6.4.1. Mycotoxin Contamination of Pet Foods
6.4.2. Outbreaks of Pets Mycotoxicoses
6.4.3. Recent Recalls of Dry Pet Foods Due to High Aflatoxin Content
7. Efforts to Ensure the Safety of Food and Feed Supplies
7.1. Prevention of Mycotoxins Contamination before and after Harvest
7.2. Detoxification of Mycotoxin-contaminated Cereal Grains
7.3. Regulations of Mycotoxins Levels in Cereal Grains
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Malachová, A.; Stránská, M.; Václavíková, M.; Elliott, C.T.; Black, C.; Meneely, J.; Hajšlová, J.; Ezekiel, C.N.; Schuhmacher, R.; Krska, R. Advanced LC–MS-based methods to study the co-occurrence and metabolization of multiple mycotoxins in cereals and cereal-based food. Anal. Bioanal. Chem. 2018, 410, 801–825. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Freire, L.; Sant’ana, A.S. Modified mycotoxins: An updated review on their formation, detection, occurrence, and toxic effects. Food Chem. Toxicol. 2018, 111, 189–205. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.; Taniwaki, M.H.; Cole, M. Mycotoxin production in major crops as influenced by growing, harvesting, storage and processing, with emphasis on the achievement of Food Safety Objectives. Food Control 2013, 32, 205–215. [Google Scholar] [CrossRef]
- Patriarca, A.; Pinto, V.F. Prevalence of mycotoxins in foods and decontamination. Curr. Opin. Food Sci. 2017, 14, 50–60. [Google Scholar] [CrossRef]
- Zain, M.E. Impact of mycotoxins on humans and animals. J. Saudi Chem. Soc. 2011, 15, 129–144. [Google Scholar] [CrossRef] [Green Version]
- Yogendrarajah, P.; Jacxsens, L.; De Saeger, S.; De Meulenaer, B. Co-occurrence of multiple mycotoxins in dry chilli (Capsicum annum L.) samples from the markets of Sri Lanka and Belgium. Food Control 2014, 46, 26–34. [Google Scholar] [CrossRef]
- Neme, K.; Mohammed, A. Mycotoxin occurrence in grains and the role of postharvest management as a mitigation strategies. A review. Food Control 2017, 78, 412–425. [Google Scholar] [CrossRef]
- De Ruyck, K.; De Boevre, M.; Huybrechts, I.; De Saeger, S. Dietary mycotoxins, co-exposure, and carcinogenesis in humans: Short review. Mutat. Res.-Rev. Mutat. Res. 2015, 766, 32–41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reddy, B.N.; Raghavender, C.R. Outbreaks of Aflatoxicoses in India. Afr. J. Food Agric. Nutr. Dev. 2007, 7, 1–15. [Google Scholar] [CrossRef]
- Obura, A. Aflatoxicosis: Evidence from Kenya. In Aflatoxins: Finding Solutions for Improved Food Safety; Unnevehr, L.J., Grace, D., Eds.; International Food Policy Research Institute: Washington, DC, USA, 2013. [Google Scholar] [CrossRef] [Green Version]
- Probst, C.; Njapau, H.; Cotty, P.J. Outbreak of an Acute Aflatoxicosis in Kenya in 2004: Identification of the Causal Agent. Appl. Environ. Microbiol. 2007, 73, 2762–2764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamala, A.; Shirima, C.; Jani, B.; Bakari, M.; Sillo, H.; Rusibamayila, N.; De Saeger, S.; Kimanya, M.; Gong, Y.; Simba, A.; et al. Outbreak of an acute aflatoxicosis in Tanzania during 2016. World Mycotoxin J. 2018, 11, 311–320. [Google Scholar] [CrossRef]
- Marasas, W.F.; Kellerman, T.S.; Gelderblom, W.C.; Coetzer, J.A.; Thiel, P.G.; Van Der Lugt, J.J. Leukoencephalomalacia in a horse induced by fumonisin B1 isolated from Fusarium moniliforme. Onderstepoort J. Veter.-Res. 1988, 55, 197–203. [Google Scholar]
- Wu, Q.; Dohnal, V.; Kuca, K.; Yuan, Z. Trichothecenes: Structure-Toxic Activity Relationships. Curr. Drug Metab. 2013, 14, 641–660. [Google Scholar] [CrossRef] [PubMed]
- Rheeder, J.; Marasas, W.; Theil, P.; Sydenham, E.; Shephard, G.; Van Schalkwyk, D. Fusarium moniliformeand Fumonisins in Corn in Relation to Human Esophageal Cancer in Transkei. Phytopathology 1992, 82, 353–357. [Google Scholar] [CrossRef]
- Xue, K.S.; Tang, L.; Sun, G.; Wang, S.; Hu, X.; Wang, J.-S. Mycotoxin exposure is associated with increased risk of esophageal squamous cell carcinoma in Huaian area, China. BMC Cancer 2019, 19, 12–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, C.; Riley, R.T.; Wu, F. Dietary Fumonisin and Growth Impairment in Children and Animals: A Review. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1448–1464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, G.; Wang, S.; Hu, X.; Su, J.; Huang, T.; Yu, J.; Tang, L.; Gao, W.; Wang, J.S. Fumonisin B1 contamination of home-grown corn in high-risk areas for esophageal and liver cancer in China. Food Addit. Contam. 2007, 24, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.; Wang, S.; Hu, X.; Su, J.; Zhang, Y.; Xie, Y.; Zhang, H.; Tang, L.; Wang, J.S. Co-contamination of aflatoxin B1 and fumonisin B1 in food and human dietary exposure in three areas of China. Food Addit. Contam. 2011, 28, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Marasas, W.F.O. Fumonisins: Their implications for human and animal health. Nat. Toxins 1995, 3, 193–198. [Google Scholar] [CrossRef]
- Gelderblom, W.C.A.; Semple, E.; Marasas, W.F.O.; Farber, E. The cancer-initiating potential of the fumonisin B mycotoxins. Carcinogenesis 1992, 13, 433–437. [Google Scholar] [CrossRef]
- Chu, F.S.; Guo, Y. Simultaneous occurrence of Fumonisin B1 and other mycotoxins in moldy corn collected from the People’s Republic of China in regions with high incidences of esophageal cancer. Appl. Environ. Microbiol. 1994, 60, 847–852. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alizadeh, A.M.; Roshandel, G.; Roudbarmohammadi, S.; Roudbary, M.; Sohanaki, H.; Ghiasian, S.A.; Taherkhani, A.; Semnani, S.; Aghasi, M. Fumonisin B1 Contamination of Cereals and Risk of Esophageal Cancer in a High Risk Area in Northeastern Iran. Asian Pac. J. Cancer Prev. 2012, 13, 2625–2628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- IARC. Monographs on the evaluation of carcinogenic risks to humans: Chemical agents and related occupations. In A Review of Human Carcinogens; International Agency for Research on Cancer: Lyon, France, 2012; Volume 100F, pp. 224–248. [Google Scholar]
- Wild, C.P. Aflatoxin Exposure in Developing Countries: The Critical Interface of Agriculture and Health. Food Nutr. Bull. 2007, 28, S372–S380. [Google Scholar] [CrossRef] [Green Version]
- Ropejko, K.; Twarużek, M. Zearalenone and Its Metabolites—General Overview, Occurrence, and Toxicity. Toxins 2021, 13, 35. [Google Scholar] [CrossRef]
- Ferrigo, D.; Raiola, A.; Causin, R. Fusarium Toxins in Cereals: Occurrence, Legislation, Factors Promoting the Appearance and Their Management. Molecules 2016, 21, 627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dohlman, E. Mycotoxin hazards and regulations: Impacts on food and animal feed crop trade. In International Trade and Food Safety/AER-828; Jean, C., Buzby, J.C., Eds.; Agricultural Economic Report No. 828; US Department of Agriculture: Washington, DC, USA, 2003; pp. 97–108. Available online: https://www.ers.usda.gov/webdocs/publications/41603/15644_aer828_1_.pdf?v=42055 (accessed on 14 December 2021).
- van Veen, T.W.S. International trade and food safety in developing countries. Food Control 2005, 16, 491–496. [Google Scholar] [CrossRef]
- Kumar, P.; Mahato, D.K.; Kamle, M.; Mohanta, T.K.; Kang, S.G. Aflatoxins: A Global Concern for Food Safety, Human Health and Their Management. Front. Microbiol. 2017, 7, 2170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmale, D.G.; Munkvold, G.P. Mycotoxins in Crops: A Threat to Human and Domestic Animal Health. Plant Health Instr. 2009, 1, 10. [Google Scholar] [CrossRef]
- Ülger, T.G.; Uçar, A.; Çakıroğlu, F.P.; Yilmaz, S. Genotoxic effects of mycotoxins. Toxicon 2020, 185, 104–113. [Google Scholar] [CrossRef]
- Theumer, M.; Henneb, Y.; Khoury, L.; Snini, S.; Tadrist, S.; Canlet, C.; Puel, O.; Oswald, I.; Audebert, M. Genotoxicity of aflatoxins and their precursors in human cells. Toxicol. Lett. 2018, 287, 100–107. [Google Scholar] [CrossRef]
- Kuiper-Goodman, T.; Scott, P.M. Risk assessment of the mycotoxin ochratoxin A. Biomed. Environ. Sci. BES 1989, 2, 179–248. [Google Scholar] [PubMed]
- Heussner, A.H.; Bingle, L.E.H. Comparative Ochratoxin Toxicity: A Review of the Available Data. Toxins 2015, 7, 4253–4282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kőszegi, T.; Poór, M.; Manderville, R.A.; Pfohl-Leszkowicz, A. Ochratoxin A: Molecular Interactions, Mechanisms of Toxicity and Prevention at the Molecular Level. Toxins 2016, 8, 111. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, R.; Peraica, M. Ochratoxin A in human kidney diseases. Food Addit. Contam. 2005, 22, 53–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agriopoulou, S.; Stamatelopoulou, E.; Varzakas, T. Advances in Occurrence, Importance, and Mycotoxin Control Strategies: Prevention and Detoxification in Foods. Foods 2020, 9, 137. [Google Scholar] [CrossRef]
- Gupta, R.C.; Srivastava, A.; Lall, R. Chapter 72—Ochratoxins and Citrinin. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Academic Press: Cambridge, MA, USA, 2018; pp. 1019–1027. [Google Scholar] [CrossRef]
- Longobardi, C.; Ferrara, G.; Andretta, E.; Montagnaro, S.; Damiano, S.; Ciarcia, R. Ochratoxin A and Kidney Oxidative Stress: The Role of Nutraceuticals in Veterinary Medicine—A Review. Toxins 2022, 14, 398. [Google Scholar] [CrossRef]
- Lonkar, P.; Dedon, P.C. Reactive species and DNA damage in chronic inflammation: Reconciling chemical mechanisms and biological fates. Int. J. Cancer 2010, 128, 1999–2009. [Google Scholar] [CrossRef] [Green Version]
- Zhang, G.-L.; Feng, Y.-L.; Song, J.-L.; Zhou, X.-S. Zearalenone: A Mycotoxin with Different Toxic Effect in Domestic and Laboratory Animals’ Granulosa Cells. Front. Genet. 2018, 9, 667. [Google Scholar] [CrossRef] [Green Version]
- Rogowska, A.; Pomastowski, P.; Sagandykova, G.; Buszewski, B. Zearalenone and its metabolites: Effect on human health, metabolism and neutralisation methods. Toxicon 2019, 162, 46–56. [Google Scholar] [CrossRef]
- Shier, W.T.; Shier, A.C.; Xie, W.; Mirocha, C.J. Structure-activity relationships for human estrogenic activity in zearalenone mycotoxins. Toxicon Off. J. Int. Soc. Toxinol. 2001, 39, 1435–1438. [Google Scholar] [CrossRef]
- WHO. World Health Organization Food Safety Digest—Fumonisins. 2018. Available online: https://www.who.int/foodsafety/FSDigest_Fumonisins_EN.pdf (accessed on 11 December 2021).
- Persson, E.C.; Sewram, V.; Evans, A.A.; London, W.T.; Volkwyn, Y.; Shen, Y.-J.; Van Zyl, J.A.; Chen, G.; Lin, W.; Shephard, G.S.; et al. Fumonisin B1 and risk of hepatocellular carcinoma in two Chinese cohorts. Food Chem. Toxicol. 2012, 50, 679–683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCormick, S.P.; Stanley, A.M.; Stover, N.A.; Alexander, N.J. Trichothecenes: From Simple to Complex Mycotoxins. Toxins 2011, 3, 802–814. [Google Scholar] [CrossRef] [PubMed]
- Janik, E.; Niemcewicz, M.; Podogrocki, M.; Ceremuga, M.; Stela, M.; Bijak, M. T-2 Toxin—The Most Toxic Trichothecene Mycotoxin: Metabolism, Toxicity, and Decontamination Strategies. Molecules 2021, 26, 6868. [Google Scholar] [CrossRef] [PubMed]
- Jakovac-Strajn, B.; Tavčar-Kalcher, G. A Method Using Gas Chromatography—Mass Spectrometry for the Detection of Mycotoxins from Trichothecene Groups A and B in Grains. In Gas Chromatography in Plant Science, Wine Technology, Toxicology and Some Specific Applications; IntechOpen: London, UK, 2012; pp. 225–244. [Google Scholar]
- Polak-Śliwińska, M.; Paszczyk, B. Trichothecenes in Food and Feed, Relevance to Human and Animal Health and Methods of Detection: A Systematic Review. Molecules 2021, 26, 454. [Google Scholar] [CrossRef]
- Audenaert, K.; Vanheule, A.; Höfte, M.; Haesaert, G. Deoxynivalenol: A Major Player in the Multifaceted Response of Fusarium to Its Environment. Toxins 2014, 6, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Foroud, N.A.; Baines, D.; Gagkaeva, T.Y.; Thakor, N.; Badea, A.; Steiner, B.; Bürstmayr, M.; Bürstmayr, H. Trichothecenes in Cereal Grains—An Update. Toxins 2019, 11, 634. [Google Scholar] [CrossRef] [Green Version]
- Broekaert, N.; Devreese, M.; Demeyere, K.; Berthiller, F.; Michlmayr, H.; Varga, E.; Adam, G.; Meyer, E.; Croubels, S. Comparative in vitro cytotoxicity of modified deoxynivalenol on porcine intestinal epithelial cells. Food Chem. Toxicol. 2016, 95, 103–109. [Google Scholar] [CrossRef]
- Paterson, R.R.M.; Lima, N. How will climate change affect mycotoxins in food? Food Res. Int. 2010, 43, 1902–1914. [Google Scholar] [CrossRef] [Green Version]
- Streit, E.; Naehrer, K.; Rodrigues, I.; Schatzmayr, G. Mycotoxin occurrence in feed and feed raw materials worldwide: Long-term analysis with special focus on Europe and Asia. J. Sci. Food Agric. 2013, 93, 2892–2899. [Google Scholar] [CrossRef]
- CAST. Mycotoxins: Economics and Health Risks; Task Force Report No. 116; Council for Agricultural Science and Technology: Ames, IA, USA, 1989. [Google Scholar]
- Eskola, M.; Kos, G.; Elliott, C.T.; Hajslova, J.; Mayar, S.; Krska, R. Worldwide contamination of food-crops with mycotoxins: Validity of the widely cited ‘FAO estimate’ of 25%. Crit. Rev. Food Sci. Nutr. 2020, 60, 2773–2789. [Google Scholar] [CrossRef]
- Biomin. Biomin World Mycotoxin Survey 2020, Annual Report No. 17. 2020. Available online: https://www.biomin.net/science-hub/world-mycotoxin-survey-impact-2021/ (accessed on 20 September 2021).
- Bhat, R.; Rai, R.V.; Karim, A. Mycotoxins in Food and Feed: Present Status and Future Concerns. Compr. Rev. Food Sci. Food Saf. 2010, 9, 57–81. [Google Scholar] [CrossRef] [PubMed]
- Ostry, V.; Malir, F.; Toman, J.; Grosse, Y. Mycotoxins as human carcinogens—The IARC Monographs classification. Mycotoxin Res. 2017, 33, 65–73. [Google Scholar] [CrossRef]
- FAO/JECFA. Safety Evaluation of Certain Contaminants in Food; Who Food Additives Series; 74 FAO JECFA Monographs 19 bis; FAO/JECFA: Rome, Italy, 2016; pp. 895–925. [Google Scholar]
- WHO. Evaluation of Certain Contaminants in Food: Eighty-Third Report of the Joint FAO/WHO Expert Committee on Food Additives, 83rd ed.; WHO Technical Report Series; World Health Organization & Joint FAO/WHO Expert Committee on Food Additives: Geneva, Switzerland, 2017. Available online: https://apps.who.int/iris/handle/10665/254893 (accessed on 27 September 2022).
- Sangare-Tigori, B.; Moukha, S.; Kouadio, H.J.; Betbeder, A.-M.; Dano, D.S.; Creppy, E.E. Co-occurrence of aflatoxin B1, fumonisin B1, ochratoxin A and zearalenone in cereals and peanuts from Côte d’Ivoire. Food Addit. Contam. 2007, 23, 1000–1007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weaver, A.C.; King, W.D.; Verax, M.; Fox, U.; Kudupoje, M.B.; Mathis, G.; Lumpkins, B.; Yiannikouris, A. Impact of Chronic Levels of Naturally Multi-Contaminated Feed with Fusarium Mycotoxins on Broiler Chickens and Evaluation of the Mitigation Properties of Different Titers of Yeast Cell Wall Extract. Toxins 2020, 12, 636. [Google Scholar] [CrossRef] [PubMed]
- Fusilier, K.; Chilvers, M.I.; Limay-Rios, V.; Singh, M.P. Mycotoxin Co-Occurrence in Michigan Harvested Maize Grain. Toxins 2022, 14, 431. [Google Scholar] [CrossRef]
- Almeida, M.I.; Almeida, N.G.; Carvalho, K.L.; Gonçalves, G.A.A.; Silva, C.N.; Santos, E.A.; Garcia, J.C.; Vargas, E.A. Co-occurrence of aflatoxins B1, B2, G1and G2, ochratoxin A, zearalenone, deoxynivalenol, and citreoviridin in rice in Brazil. Food Addit. Contam. Part A 2012, 29, 694–703. [Google Scholar] [CrossRef]
- CFIA. Ochratoxin A in Wheat Products, Oat Products, Rice Products and Other Grain Products—1 April 2018 to 31 March 2019. Canadian Food Inspection Agency. 2020. Available online: https://inspection.canada.ca/food-safety-for-industry/food-chemistry-and-microbiology/food-safety-testing-bulletin-and-reports (accessed on 16 April 2023).
- Wang, Y.; Dong, Y.J.; Yue, H.; Li, Z.M.; Chen, Y.B.; Wang, Y.T.; Deng, L.G.; Zhao, S.C. Investigation and analysis on mycotoxins contamination of maize in Shandong Province. Sci. Technol. Cereals Oils Foods 2016, 24, 69–73. (In Chinese) [Google Scholar]
- Sun, X.; Su, P.; Shan, H. Mycotoxin contamination of maize in China. Compr. Rev. Food Sci. Food Saf. 2017, 16, 835–849. [Google Scholar] [CrossRef]
- Shi, H.; Schwab, W.; Yu, P. Natural Occurrence and Co-Contamination of Twelve Mycotoxins in Industry-Submitted Cool-Season Cereal Grains Grown under a Low Heat Unit Climate Condition. Toxins 2019, 11, 160. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.; Cheng, T.; Xu, W.; Han, X.; Zhang, J.; Zhang, H.; Wang, C.; Fanning, S.; Li, F. Natural co-occurrence of multi-mycotoxins in unprocessed wheat grains from China. Food Control 2021, 130, 108321. [Google Scholar] [CrossRef]
- Joubrane, K.; Mnayer, D.; El Khoury, A.; El Khoury, A.; Awad, E. Co-Occurrence of Aflatoxin B1 and Ochratoxin A in Lebanese Stored Wheat. J. Food Prot. 2020, 83, 1547–1552. [Google Scholar] [CrossRef]
- Kim, D.-H.; Hong, S.-Y.; Kang, J.W.; Cho, S.M.; Lee, K.R.; An, T.K.; Lee, C.; Chung, S.H. Simultaneous Determination of Multi-Mycotoxins in Cereal Grains Collected from South Korea by LC/MS/MS. Toxins 2017, 9, 106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Makun, H.A.; Dutton, M.F.; Njobeh, P.B.; Mwanza, M.; Kabiru, A.Y. Natural multi-occurrence of mycotoxins in rice from Niger State, Nigeria. Mycotoxin Res. 2011, 27, 97–104. [Google Scholar] [CrossRef] [Green Version]
- Mudili, V.; Siddaih, C.N.; Nagesh, M.; Garapati, P.; Kumar, K.N.; Murali, H.S.; Yli-Mattila, T.; Batra, H.V. Mould incidence and mycotoxin contamination in freshly harvested maize kernels originated from India. J. Sci. Food Agric. 2014, 94, 2674–2683. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, S.Z.; Asi, M.R.; Hanif, U.; Zuber, M.; Jinap, S. The presence of aflatoxins and ochratoxin A in rice and rice products; and evaluation of dietary intake. Food Chem. 2016, 210, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Wajih Ul Hassan, S.; Sadef, Y.; Hussain, S.; Asi, M.R.; Ashraf, M.Y.; Anwar, S.; Malik, A. Unusual pattern of aflatoxins and ochratoxin in commercially grown maize varieties of Pakistan. Toxicon 2020, 182, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Gruber-Dorninger, C.; Jenkins, T.; Schatzmayr, G. Multi-mycotoxin screening of feed and feed raw materials from Africa. World Mycotoxin J. 2018, 11, 369–383. [Google Scholar] [CrossRef]
- Kortei, N.K.; Annan, T.; Kyei-Baffour, V.; Essuman, E.K.; Okyere, H.; Tettey, C.O. Exposure and risk characterizations of ochratoxins A and aflatoxins through maize (Zea mays) consumed in different agro-ecological zones of Ghana. Sci. Rep. 2021, 11, 23339. [Google Scholar] [CrossRef]
- Lewis, L.; Onsongo, M.; Njapau, H.; Schurz-Rogers, H.; Luber, G.; Kieszak, S.; Nyamongo, J.; Backer, L.; Dahiye, A.M.; Misore, A.; et al. Aflatoxin Contamination of Commercial Maize Products during an Outbreak of Acute Aflatoxicosis in Eastern and Central Kenya. Environ. Health Perspect. 2005, 113, 1763–1767. [Google Scholar] [CrossRef]
- Ngure, F.; Ngure, C.; Achieng, G.; Munga, F.; Moran, Z.; Stafstrom, W.; Nelson, R. Mycotoxins contamination of market maize and the potential of density sorting in reducing exposure in unregulated food systems in Kenya. World Mycotoxin J. 2021, 14, 165–178. [Google Scholar] [CrossRef]
- Mutiga, S.K.; Mutuku, J.M.; Koskei, V.; Gitau, J.K.; Ng’ang’a, F.; Musyoka, J.; Chemining’wa, G.N.; Murori, R. Multiple Mycotoxins in Kenyan Rice. Toxins 2021, 13, 203. [Google Scholar] [CrossRef] [PubMed]
- Echodu, R.; Malinga, G.M.; Kaducu, J.M.; Ovuga, E.; Haesaert, G. Prevalence of aflatoxin, ochratoxin and deoxynivalenol in cereal grains in northern Uganda: Implication for food safety and health. Toxicol. Rep. 2019, 6, 1012–1017. [Google Scholar] [CrossRef] [PubMed]
- Karlovsky, P.; Suman, M.; Berthiller, F.; De Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016, 32, 179–205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Habschied, K.; Šarić, G.K.; Krstanović, V.; Mastanjević, K. Mycotoxins—Biomonitoring and Human Exposure. Toxins 2021, 13, 113. [Google Scholar] [CrossRef] [PubMed]
- Kolakowski, B.; O’Rourke, S.M.; Bietlot, H.P.; Kurz, K.; Aweryn, B. Ochratoxin A Concentrations in a Variety of Grain-Based and Non–Grain-Based Foods on the Canadian Retail Market from 2009 to 2014. J. Food Prot. 2016, 79, 2143–2159. [Google Scholar] [CrossRef]
- Cappozzo, J.; Jackson, L.; Lee, H.J.; Zhou, W.; Al-Taher, F.; Zweigenbaum, J.; Ryu, D. Occurrence of Ochratoxin A in Infant Foods in the United States. J. Food Prot. 2017, 80, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.T.N.; Ryu, D. Concentration of ochratoxin A in breakfast cereals and snacks consumed in the United States. Food Control 2014, 40, 140–144. [Google Scholar] [CrossRef]
- Cerveró, M.C.; Castillo, M.A.; Montes, R.; Hernández, E. Determination of trichothecenes, zearalenone and zearalenols in commercially available corn-based foods in Spain. Rev. Iberoam. Micol. 2007, 24, 52–55. [Google Scholar] [PubMed]
- Cano-Sancho, G.; Marin, S.; Ramos, A.; Sanchis, V. Occurrence of zearalenone, an oestrogenic mycotoxin, in Catalonia (Spain) and exposure assessment. Food Chem. Toxicol. 2012, 50, 835–839. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Xiao, Y.; Wang, W.; Lyu, W.; Wang, X.; Li, Y.; Deng, T.; Yang, H. Mycotoxins in cereal-based infant foods marketed in China: Occurrence and risk assessment. Food Control 2022, 138, 108998. [Google Scholar] [CrossRef]
- Martins, C.; Assunção, R.; Cunha, S.C.; Fernandes, J.O.; Jager, A.; Petta, T.; Oliveira, C.A.; Alvito, P. Assessment of multiple mycotoxins in breakfast cereals available in the Portuguese market. Food Chem. 2018, 239, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Cano-Sancho, G.; Sanchis, V.; Marín, S.; Ramos, A. Occurrence and exposure assessment of aflatoxins in Catalonia (Spain). Food Chem. Toxicol. 2013, 51, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Stanciu, O.; Juan, C.; Miere, D.; Berrada, H.; Loghin, F.; Mañes, J. First study on trichothecene and zearalenone exposure of the Romanian population through wheat-based products consumption. Food Chem. Toxicol. 2018, 121, 336–342. [Google Scholar] [CrossRef]
- Andrade, P.D.; Dias, J.V.; Souza, D.M.; Brito, A.P.; van Donkersgoed, G.; Pizzutti, I.R.; Caldas, E.D. Mycotoxins in cereals and cereal-based products: Incidence and probabilistic dietary risk assessment for the Brazilian population. Food Chem. Toxicol. 2020, 143, 111572. [Google Scholar] [CrossRef]
- Kimanya, M.E.; Shirima, C.P.; Magoha, H.; Shewiyo, D.H.; De Meulenaer, B.; Kolsteren, P.; Gong, Y.Y. Co-exposures of aflatoxins with deoxynivalenol and fumonisins from maize based complementary foods in Rombo, Northern Tanzania. Food Control 2014, 41, 76–81. [Google Scholar] [CrossRef]
- Blankson, G.K.; Mill-Robertson, F.C. Aflatoxin contamination and exposure in processed cereal-based complementary foods for infants and young children in greater Accra, Ghana. Food Control 2016, 64, 212–217. [Google Scholar] [CrossRef]
- Kortei, N.K.; Agyekum, A.A.; Akuamoa, F.; Baffour, V.K.; Alidu, H.W. Risk assessment and exposure to levels of naturally occurring aflatoxins in some packaged cereals and cereal based foods consumed in Accra, Ghana. Toxicol. Rep. 2019, 6, 34–41. [Google Scholar] [CrossRef]
- Bashiry, M.; Yazdanpanah, H.; Sadeghi, E.; Shokri, S.; Mirmoghtadaie, L.; Mortazavian, A.M.; Mohammadi, A.; Nematollahi, A.; Hejazi, E.; Hosseini, H. Occurrence of Aflatoxins in Commercial Cereal-based Baby Foods in Iran: A Probabilistic Risk Assessment to Health. Iran J. Pharm. Res. 2021, 20, 31–45. [Google Scholar] [CrossRef] [PubMed]
- Mottaghianpour, E.; Nazari, F.; Mehrasbi, M.R.; Hosseini, M.-J. Occurrence of aflatoxin B1in baby foods marketed in Iran. J. Sci. Food Agric. 2017, 97, 2690–2694. [Google Scholar] [CrossRef] [PubMed]
- Bouafifssa, Y.; Manyes, L.; Rahouti, M.; Mañes, J.; Berrada, H.; Zinedine, A.; Fernández-Franzón, M. Multi-Occurrence of Twenty Mycotoxinsin Pasta and a Risk Assessment in the Moroccan Population. Toxins 2018, 10, 432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adekoya, I.; Obadina, A.; Adaku, C.C.; De Boevre, M.; Okoth, S.; De Saeger, S.; Njobeh, P. Mycobiota and co-occurrence of mycotoxins in South African maize-based opaque beer. Int. J. Food Microbiol. 2018, 270, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Nafuka, S.N.; Misihairabgwi, J.M.; Bock, R.; Ishola, A.; Sulyok, M.; Krska, R. Variation of Fungal Metabolites in Sorghum Malts Used to Prepare Namibian Traditional Fermented Beverages Omalodu and Otombo. Toxins 2019, 11, 165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mushtaq, M.; Sultana, B.; Anwar, F.; Khan, M.Z.; Ashrafuzzaman, M. Occurrence of Aflatoxins in Selected Processed Foods from Pakistan. Int. J. Mol. Sci. 2012, 13, 8324–8337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iqbal, S.Z.; Rabbani, T.; Asi, M.R.; Jinap, S. Assessment of aflatoxins, ochratoxin A and zearalenone in breakfast cereals. Food Chem. 2014, 157, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, S.Z.; Asi, M.R.; Jinap, S.; Rashid, U. Detection of aflatoxins and zearalenone contamination in wheat derived products. Food Control 2014, 35, 223–226. [Google Scholar] [CrossRef]
- Luo, S.; Du, H.; Kebede, H.; Liu, Y.; Xing, F. Contamination status of major mycotoxins in agricultural product and food stuff in Europe. Food Control 2021, 127, 108120. [Google Scholar] [CrossRef]
- Li, X.; Ma, W.; Ma, Z.; Zhang, Q.; Li, H. The Occurrence and Contamination Level of Ochratoxin A in Plant and Animal-Derived Food Commodities. Molecules 2021, 26, 6928. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Ryu, D. Significance of Ochratoxin a in Breakfast Cereals from the United States. J. Agric. Food Chem. 2015, 63, 9404–9409. [Google Scholar] [CrossRef]
- Mitchell, N.J.; Chen, C.; Palumbo, J.D.; Bianchini, A.; Cappozzo, J.; Stratton, J.; Ryu, D.; Wu, F. A risk assessment of dietary Ochratoxin a in the United States. Food Chem. Toxicol. 2017, 100, 265–273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bakker, M.; Pieters, M.N. Risk Assessment of Ochratoxin A in the Netherlands; RIVM report 388802025; Rijksinstituut voor Volksgezondheid en Milieu RIVM: Utrecht, The Netherlands, 2022.
- Cruz, J.V.d.S. Ocorrência de Aflatoxinas e Fumonisinas em Produtos à Base de Milho e Milho Utilizado Como Ingrediente de Ração Para Animais de Companhia, Comercializados na Região de Pirassununga. Estado de São Paulo. Ph.D. Thesis, São Paulo University, Pirassununga, Brazil, 2010. [Google Scholar]
- Cano-Sancho, G.; Valle-Algarra, F.; Jiménez, M.; Burdaspal, P.; Legarda, T.; Ramos, A.; Sanchis, V.; Marín, S. Presence of trichothecenes and co-occurrence in cereal-based food from Catalonia (Spain). Food Control 2011, 22, 490–495. [Google Scholar] [CrossRef]
- Khaneghah, A.M.; Farhadi, A.; Nematollahi, A.; Vasseghian, Y.; Fakhri, Y. A systematic review and meta-analysis to investigate the concentration and prevalence of trichothecenes in the cereal-based food. Trends Food Sci. Technol. 2020, 102, 193–202. [Google Scholar] [CrossRef]
- Zhang, K.; Flannery, B.M.; Oles, C.J.; Adeuya, A. Mycotoxins in infant/toddler foods and breakfast cereals in the US retail market. Food Addit. Contam. Part B 2018, 11, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Khaneghah, A.M.; Fakhri, Y.; Raeisi, S.; Armoon, B.; Sant’Ana, A.S. Prevalence and concentration of ochratoxin A, zearalenone, deoxynivalenol and total aflatoxin in cereal-based products: A systematic review and meta-analysis. Food Chem. Toxicol. 2018, 118, 830–848. [Google Scholar] [CrossRef]
- Maragos, C.M. Zearalenone occurrence and human exposure. World Mycotoxin J. 2010, 3, 369–383. [Google Scholar] [CrossRef]
- Schiavone, A.; Cavallero, C.; Girotto, L.; Pozzo, L.; Antoniazzi, S.; Cavallarin, L. A survey on the occurrence of ochratoxin a in feeds and sera collected in conventional and organic poultry farms in Northern Italy. Ital. J. Anim. Sci. 2008, 7, 495–503. [Google Scholar] [CrossRef]
- Santos Pereira, C.; Cunha, S.C.; Fernandes, J.O. Prevalent Mycotoxins in Animal Feed: Occurrence and Analytical Methods. Toxins 2019, 11, 290. [Google Scholar] [CrossRef] [Green Version]
- Kovalsky, P.; Kos, G.; Nährer, K.; Schwab, C.; Jenkins, T.; Schatzmayr, G.; Sulyok, M.; Krska, R. Co-Occurrence of Regulated, Masked and Emerging Mycotoxins and Secondary Metabolites in Finished Feed and Maize—An Extensive Survey. Toxins 2016, 8, 363. [Google Scholar] [CrossRef] [Green Version]
- Zhao, L.; Zhang, L.; Xu, Z.; Liu, X.; Chen, L.; Dai, J.; Karrow, N.A.; Sun, L. Occurrence of Aflatoxin B1, deoxynivalenol and zearalenone in feeds in China during 2018–2020. J. Anim. Sci. Biotechnol. 2021, 12, 1. [Google Scholar] [CrossRef] [PubMed]
- Gruber-Dorninger, C.; Jenkins, T.; Schatzmayr, G. Global Mycotoxin Occurrence in Feed: A Ten-Year Survey. Toxins 2019, 11, 375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mwanda, O.; Otieno, C.; Omonge, E. Acute aflatoxicosis: Case report. East Afr. Med. J. 2005, 82, 320–324. [Google Scholar] [CrossRef]
- Pitt, J.I. An Introduction to Mycotoxins, in Mycotoxin Prevention and Control in Food Grains; Semple, R.L., Frio, A.S., Hicks, P.A., Lozare, J.V., Eds.; Food and Agriculture Organization: Rome, Italy, 1989. [Google Scholar]
- Lye, M.S.; Ghazali, A.A.; Mohan, J.; Alwin, N.; Nair, R.C. An outbreak of acute hepatic encephalopathy due to severe aflatoxicosis in Malaysia. Am. J. Trop. Med. Hyg. 1995, 53, 68–72. [Google Scholar] [PubMed]
- Ekwomadu, T.I.; Akinola, S.A.; Mwanza, M. Fusarium Mycotoxins, Their Metabolites (Free, Emerging, and Masked), Food Safety Concerns, and Health Impacts. Int. J. Environ. Res. Public Health 2021, 18, 11741. [Google Scholar] [CrossRef] [PubMed]
- Dey, D.K.; Kang, J.I.; Bajpai, V.K.; Kim, K.; Lee, H.; Sonwal, S.; Simal-Gandara, J.; Xiao, J.; Ali, S.; Huh, Y.S.; et al. Mycotoxins in food and feed: Toxicity, preventive challenges, and advanced detection techniques for associated diseases. Crit. Rev. Food Sci. Nutr. 2022, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wen, J.; Tang, Y.; Shi, J.; Mu, G.; Yan, R.; Cai, J.; Long, M. Research Progress on Fumonisin B1 Contamination and Toxicity: A Review. Molecules 2021, 26, 5238. [Google Scholar] [CrossRef] [PubMed]
- McKean, C.; Tang, L.; Tang, M.; Billam, M.; Wang, Z.; Theodorakis, C.; Kendall, R.; Wang, J.-S. Comparative acute and combinative toxicity of aflatoxin B1 and fumonisin B1 in animals and human cells. Food Chem. Toxicol. 2006, 44, 868–876. [Google Scholar] [CrossRef]
- Forsell, J.; Jensen, R.; Tai, J.-H.; Witt, M.; Lin, W.; Pestka, J. Comparison of acute toxicities of deoxynivalenol (vomitoxin) and 15-acetyldeoxynivalenol in the B6C3F1 mouse. Food Chem. Toxicol. 1987, 25, 155–162. [Google Scholar] [CrossRef]
- Zinedine, A.; Soriano, J.M.; Moltó, J.C.; Mañes, J. Review on the toxicity, occurrence, metabolism, detoxification, regulations and intake of zearalenone: An oestrogenic mycotoxin. Food Chem. Toxicol. 2007, 45, 1–18. [Google Scholar] [CrossRef]
- Wen, J.; Mu, P.; Deng, Y. Mycotoxins: Cytotoxicity and biotransformation in animal cells. Toxicol. Res. 2016, 5, 377–387. [Google Scholar] [CrossRef] [Green Version]
- Creppy, E.E.; Chiarappa, P.; Baudrimont, I.; Borracci, P.; Moukha, S.; Carratù, M. Synergistic effects of fumonisin B1 and ochratoxin a: Are in vitro cytotoxicity data predictive of in vivo acute toxicity? Toxicology 2004, 201, 115–123. [Google Scholar] [CrossRef]
- Wentzel, J.F.; Lombard, M.J.; Du Plessis, L.H.; Zandberg, L. Evaluation of the cytotoxic properties, gene expression profiles and secondary signalling responses of cultured cells exposed to fumonisin B1, deoxynivalenol and zearalenone mycotoxins. Arch. Toxicol. 2017, 91, 2265–2282. [Google Scholar] [CrossRef]
- Alvito, P.; Pereira-Da-Silva, L. Mycotoxin Exposure during the First 1000 Days of Life and Its Impact on Children’s Health: A Clinical Overview. Toxins 2022, 14, 189. [Google Scholar] [CrossRef] [PubMed]
- Atkins, D.; Norman, J. Mycotoxins and food safety. Nutr. Food Sci. 1998, 98, 260–266. [Google Scholar] [CrossRef]
- Christopher, P.; Wild, J.; Miller, D.; John, D.; Groopman, J.D. Mycotoxin Control in Low and Middle-Income Countries; IARC Working Group Reports, No. 9; International Agency for Research on Cancer: Lyon, France, 2015; pp. 13–16.
- Wagacha, J.M.; Muthomi, J. Mycotoxin problem in Africa: Current status, implications to food safety and health and possible management strategies. Int. J. Food Microbiol. 2008, 124, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wu, F. Global Burden of Aflatoxin-Induced Hepatocellular Carcinoma: A Risk Assessment. Environ. Health Perspect. 2010, 118, 818–824. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.-G.; Egner, P.A.; Ng, D.; Jacobson, L.P.; Muñoz, A.; Zhu, Y.-R.; Qian, G.-S.; Wu, F.; Yuan, J.-M.; Groopman, J.D.; et al. Reduced Aflatoxin Exposure Presages Decline in Liver Cancer Mortality in an Endemic Region of China. Cancer Prev. Res. 2013, 6, 1038–1045. [Google Scholar] [CrossRef] [Green Version]
- Claeys, L.; Romano, C.; De Ruyck, K.; Wilson, H.; Fervers, B.; Korenjak, M.; Zavadil, J.; Gunter, M.J.; De Saeger, S.; De Boevre, M.; et al. Mycotoxin exposure and human cancer risk: A systematic review of epidemiological studies. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1449–1464. [Google Scholar] [CrossRef]
- Omotayo, O.P.; Omotayo, A.O.; Mwanza, M.; Babalola, O.O. Prevalence of Mycotoxins and Their Consequences on Human Health. Toxicol. Res. 2019, 35, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Gong, Y.Y.; Watson, S.; Routledge, M.N. Aflatoxin Exposure and Associated Human Health Effects, a Review of Epidemiological Studies. Food Saf. 2016, 4, 14–27. [Google Scholar] [CrossRef] [Green Version]
- Malir, F.; Ostry, V.; Pfohl-Leszkowicz, A.; Novotna, E. Ochratoxin A: Developmental and Reproductive Toxicity—An Overview. Birth Defects Res. B Dev. Reprod. Toxicol. 2014, 98, 493–502. [Google Scholar] [CrossRef]
- Abid, S.; Hassen, W.; Achour, A.; Skhiri, H.; Maaroufi, K.; Ellouz, F.; Creppy, E.; Bacha, H. Ochratoxin a and human chronic nephropathy in Tunisia: Is the situation endemic? Hum. Exp. Toxicol. 2003, 22, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Stefanovic, V.; Toncheva, D.; Atanasova, S.; Polenakovic, M. Etiology of Balkan Endemic Nephropathy and Associated Urothelial Cancer. Am. J. Nephrol. 2006, 26, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Krogh, P.; Hald, B.; Pleština, R.; Čeović, S. Balkan (endemic) nephropathy and foodborn ochratoxin a: Preliminary results of a survey of foodstuffs. Acta Pathol. Microbiol. Scand. Sect. B Microbiol. 1977, 85B, 238–240. [Google Scholar] [CrossRef] [PubMed]
- Pavlović, M.; Plestina, R.; Krogh, P. Ochratoxin a Contamination of Foodstuffs in an Area with Balkan (Endemic) Nephropathy. Acta Pathol. Microbiol. Scand. Sect. B Microbiol. 1979, 87B, 243–246. [Google Scholar] [CrossRef] [PubMed]
- Bui-Klimke, T.R.; Wu, F. Ochratoxin A and Human Health Risk: A Review of the Evidence. Crit. Rev. Food Sci. Nutr. 2015, 55, 1860–1869. [Google Scholar] [CrossRef] [Green Version]
- Pavlović, N.M. Balkan endemic nephropathy—Current status and future perspectives. Clin. Kidney J. 2013, 6, 257–265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scott, P.M. Biomarkers of human exposure to ochratoxin A. Food Addit. Contam. 2005, 22 (Suppl. S1), 99–107. [Google Scholar] [CrossRef]
- Wafa, E.W.; Yahya, R.S.; Sobh, M.A.; Eraky, I.; el-Baz, M.; el-Gayar, H.A.; Betbeder, A.M.; Creppy, E.E. Human ochratoxicosis and nephropathy in Egypt: A preliminary study. Hum. Exp. Toxicol. 1998, 17, 124–129. [Google Scholar] [CrossRef]
- Studer-Rohr, J.; Schlatter, J.; Dietrich, D.R. Intraindividual variation in plasma levels and kinetic parameters of ochratoxin a in humans. Arch. Toxicol. 2000, 74, 499–510. [Google Scholar] [CrossRef]
- Castegnaro, M.; Canadas, D.; Vrabcheva, T.; Petkova-Bocharova, T.; Chernozemsky, I.N.; Pfohl-Leszkowicz, A. Balkan endemic nephropathy: Role of ochratoxins A through biomarkers. Mol. Nutr. Food Res. 2006, 50, 519–529. [Google Scholar] [CrossRef]
- Malir, F.; Louda, M.; Ostry, V.; Toman, J.; Ali, N.; Grosse, Y.; Malirova, E.; Pacovsky, J.; Pickova, D.; Brodak, M.; et al. Analyses of biomarkers of exposure to nephrotoxic mycotoxins in a cohort of patients with renal tumours. Mycotoxin Res. 2019, 35, 391–403. [Google Scholar] [CrossRef]
- Khoi, C.-S.; Chen, J.-H.; Lin, T.-Y.; Chiang, C.-K.; Hung, K.-Y. Ochratoxin A-Induced Nephrotoxicity: Up-to-Date Evidence. Int. J. Mol. Sci. 2021, 22, 11237. [Google Scholar] [CrossRef] [PubMed]
- Marasas, W.F.O.; Riley, R.T.; Hendricks, K.A.; Stevens, V.L.; Sadler, T.W.; Gelineau-van Waes, J.; Missmer, S.A.; Cabrera, J.; Torres, O.; Gelderblom, W.C.A.; et al. Fumonisins Disrupt Sphingolipid Metabolism, Folate Transport, and Neural Tube Development in Embryo Culture and In Vivo: A Potential Risk Factor for Human Neural Tube Defects among Populations Consuming Fumonisin-Contaminated Maize. J. Nutr. 2004, 134, 711–716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stockmann-Juvala, H.; Savolainen, K. A review of the toxic effects and mechanisms of action of fumonisin B1. Hum. Exp. Toxicol. 2008, 27, 799–809. [Google Scholar] [CrossRef]
- Chilaka, C.A.; Obidiegwu, J.E.; Chilaka, A.C.; Atanda, O.O.; Mally, A. Mycotoxin Regulatory Status in Africa: A Decade of Weak Institutional Efforts. Toxins 2022, 14, 442. [Google Scholar] [CrossRef] [PubMed]
- Sudakin, D.L. Trichothecenes in the environment: Relevance to human health. Toxicol. Lett. 2003, 143, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Mokubedi, S.M.; Phoku, J.Z.; Changwa, R.N.; Gbashi, S.; Njobeh, P.B. Analysis of Mycotoxins Contamination in Poultry Feeds Manufactured in Selected Provinces of South Africa Using UHPLC-MS/MS. Toxins 2019, 11, 452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Jia, Z.; Yin, S.; Shan, A.; Gao, R.; Qu, Z.; Liu, M.; Nie, S. Toxic Effects of Maternal Zearalenone Exposure on Uterine Capacity and Fetal Development in Gestation Rats. Reprod. Sci. 2014, 21, 743–753. [Google Scholar] [CrossRef] [Green Version]
- Tatay, E.; Espín, S.; García-Fernández, A.-J.; Ruiz, M.-J. Oxidative damage and disturbance of antioxidant capacity by zearalenone and its metabolites in human cells. Toxicol. Vitr. 2017, 45, 334–339. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.-Q.; Zhao, A.-H.; Wang, J.-J.; Tian, Y.; Yan, Z.-H.; Dri, M.; Shen, W.; De Felici, M.; Li, L. Oxidative stress as a plausible mechanism for zearalenone to induce genome toxicity. Gene 2022, 829, 146511. [Google Scholar] [CrossRef] [PubMed]
- Pierron, A.; Alassane-Kpembi, I.; Oswald, I.P. Impact of mycotoxin on immune response and consequences for pig health. Anim. Nutr. 2016, 2, 63–68. [Google Scholar] [CrossRef]
- Wu, K.; Ren, C.; Gong, Y.; Gao, X.; Rajput, S.A.; Qi, D.; Wang, S. The insensitive mechanism of poultry to zearalenone: A review. Anim. Nutr. 2021, 7, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Jones, F.T.; Genter, M.B.; Hagler, W.M.; Hansen, J.A.; Mowrey, B.A.; Poore, M.H.; Whitlow, L.W. Understanding and Coping with Effects of Mycotoxins in Livestock Feed and Forage. North Carolina Cooperative Extension Service. 1994. Available online: https://projects.ncsu.edu/cals/an_sci/extension/animal/nutr/Understanding_mycotoxins.pdf (accessed on 6 January 2022).
- Mostrom, M.S.; Jacobsen, B.J. Ruminant Mycotoxicosis. Veter.-Clin. N. Am. Food Anim. Pract. 2011, 27, 315–344. [Google Scholar] [CrossRef] [PubMed]
- Gallo, A.; Giuberti, G.; Frisvad, J.C.; Bertuzzi, T.; Nielsen, K.F. Review on mycotoxin issues in ruminants: Occurrence in forages, effects of mycotoxin ingestion on health status and animal performance and practical strategies to counteract their negative effects. Toxins 2015, 7, 3057–3111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kemboi, D.C.; Ochieng, P.E.; Antonissen, G.; Croubels, S.; Scippo, M.-L.; Okoth, S.; Kangethe, E.K.; Faas, J.; Doupovec, B.; Lindahl, J.F.; et al. Multi-Mycotoxin Occurrence in Dairy Cattle and Poultry Feeds and Feed Ingredients from Machakos Town, Kenya. Toxins 2020, 12, 762. [Google Scholar] [CrossRef]
- Hof, H. Mycotoxins in milk for human nutrition: Cow, sheep and human breast milk. GMS Infect. Dis. 2016, 4, Doc03. [Google Scholar] [CrossRef]
- Jouany, J.P.; Yiannikouris, A.; Bertin, G. Risk assessment of mycotoxins in ruminants and ruminant products. Options Mediterr. A 2009, 85, 205–224. Available online: http://om.ciheam.org/article.php?IDPDF=801009 (accessed on 6 January 2022).
- Upadhaya, S.D.; Park, M.A.; Ha, J.K. Mycotoxins and Their Biotransformation in the Rumen: A Review. Asian-Australas. J. Anim. Sci. 2010, 23, 1250–1260. [Google Scholar] [CrossRef]
- Adams, R.S.; Kephart, K.B.; Ishler, V.A.; Hutchinson, L.J.; Roth, G.W. Mold and Mycotoxin Problems in Livestock Feeding. Penn State Extension. 2016. Available online: https://extension.psu.edu/mold-and-mycotoxin-problems-in-livestock-feedingAntonissen (accessed on 6 January 2022).
- Zhang, K.; Wong, J.W.; Hayward, D.G.; Vaclavikova, M.; Liao, C.-D.; Trucksess, M.W. Determination of Mycotoxins in Milk-Based Products and Infant Formula Using Stable Isotope Dilution Assay and Liquid Chromatography Tandem Mass Spectrometry. J. Agric. Food Chem. 2013, 61, 6265–6273. [Google Scholar] [CrossRef] [PubMed]
- Serraino, A.; Bonilauri, P.; Kerekes, K.; Farkas, Z.; Giacometti, F.; Canever, A.; Zambrini, A.V.; Ambrus, Á. Occurrence of Aflatoxin M1 in Raw Milk Marketed in Italy: Exposure Assessment and Risk Characterization. Front. Microbiol. 2019, 10, 2516. [Google Scholar] [CrossRef]
- Mobashar, M.; Hummel, J.; Blank, R.; Südekum, K.-H. Ochratoxin A in Ruminants–A Review on Its Degradation by Gut Microbes and Effects on Animals. Toxins 2010, 2, 809–839. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Fan, Z.; Nie, D.; Zhao, Z.; Han, Z. Analysis of the Carry-Over of Ochratoxin A from Feed to Milk, Blood, Urine, and Different Tissues of Dairy Cows Based on the Establishment of a Reliable LC-MS/MS Method. Molecules 2019, 24, 2823. [Google Scholar] [CrossRef] [Green Version]
- Hashimoto, Y.; Katsunuma, Y.; Nunokawa, M.; Minato, H.; Yonemochi, C. Influence of repeated ochratoxin A ingestion on milk production and its carry-over into the milk, blood and tissues of lactating cows. Anim. Sci. J. 2016, 87, 541–546. [Google Scholar] [CrossRef]
- Turkoglu, C.; Keyvan, E. Determination of Aflatoxin M1 and Ochratoxin A in Raw, Pasteurized and UHT Milk in Turkey. Acta Sci. Vet. 2019, 47, 89667. [Google Scholar] [CrossRef] [Green Version]
- Eriksen, G.S.; Pettersson, H. Toxicological evaluation of trichothecenes in animal feed. Anim. Feed. Sci. Technol. 2004, 114, 205–239. [Google Scholar] [CrossRef]
- Gallo, A.; Mosconi, M.; Trevisi, E.; Santos, R.R. Adverse Effects of Fusarium Toxins in Ruminants: A Review of in Vivo and in Vitro Studies. Dairy 2022, 3, 474–499. [Google Scholar] [CrossRef]
- Popescu, R.G.; Rădulescu, A.L.; Georgescu, S.E.; Dinischiotu, A. Aflatoxins in Feed: Types, Metabolism, Health Consequences in Swine and Mitigation Strategies. Toxins 2022, 14, 853. [Google Scholar] [CrossRef] [PubMed]
- NGFA, National Grain and Feed Association. FDA Regulatory Guidance for Mycotoxins—A Guide for Grain Elevators, Feed Manufacturers, Grain Processors and Exporters; NGFA, National Grain and Feed Association: Washington, DC, USA, 2011. [Google Scholar]
- Pu, J.; Yuan, Q.; Yan, H.; Tian, G.; Chen, D.; He, J.; Zheng, P.; Yu, J.; Mao, X.; Huang, Z.; et al. Effects of Chronic Exposure to Low Levels of Dietary Aflatoxin B1 on Growth Performance, Apparent Total Tract Digestibility and Intestinal Health in Pigs. Animals 2021, 11, 336. [Google Scholar] [CrossRef]
- Marin, D.; Motiu, M.; Pistol, G.; Gras, M.; Israel-Roming, F.; Calin, L.; Stancu, M.; Taranu, I. Diet contaminated with ochratoxin A at the highest level allowed by EU recommendation disturbs liver metabolism in weaned piglets. World Mycotoxin J. 2016, 9, 587–596. [Google Scholar] [CrossRef]
- Marin, D.E.; Pistol, G.C.; Gras, M.A.; Palade, M.L.; Taranu, I. Comparative effect of ochratoxin A on inflammation and oxidative stress parameters in gut and kidney of piglets. Regul. Toxicol. Pharmacol. 2017, 89, 224–231. [Google Scholar] [CrossRef]
- Gan, F.; Zhang, Z.; Hu, Z.; Hesketh, J.; Xue, H.; Chen, X.; Hao, S.; Huang, Y.; Ezea, P.C.; Parveen, F.; et al. Ochratoxin A promotes porcine circovirus type 2 replication in vitro and in vivo. Free Radic. Biol. Med. 2015, 80, 33–47. [Google Scholar] [CrossRef]
- Gan, F.; Zhou, Y.; Hou, L.; Qian, G.; Chen, X.; Huang, K. Ochratoxin A induces nephrotoxicity and immunotoxicity through different MAPK signaling pathways in PK15 cells and porcine primary splenocytes. Chemosphere 2017, 182, 630–637. [Google Scholar] [CrossRef]
- Burel, C.; Tanguy, M.; Guerre, P.; Boilletot, E.; Cariolet, R.; Queguiner, M.; Postollec, G.; Pinton, P.; Salvat, G.; Oswald, I.P.; et al. Effect of Low Dose of Fumonisins on Pig Health: Immune Status, Intestinal Microbiota and Sensitivity to Salmonella. Toxins 2013, 5, 841–864. [Google Scholar] [CrossRef] [Green Version]
- Rao, Z.-X.; Tokach, M.D.; Woodworth, J.C.; DeRouchey, J.M.; Goodband, R.D.; Calderón, H.I.; Dritz, S.S. Effects of Fumonisin-Contaminated Corn on Growth Performance of 9 to 28 kg Nursery Pigs. Toxins 2020, 12, 604. [Google Scholar] [CrossRef]
- Ensley, S.M.; Radke, S.L. Mycotoxins in Grains and Feeds. In Diseases of Swine, 11th ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2019; pp. 1055–1071. [Google Scholar] [CrossRef]
- Pierron, A.; Alassane-Kpembi, I.; Oswald, I.P. Impact of two mycotoxins deoxynivalenol and fumonisin on pig intestinal health. Porc. Health Manag. 2016, 2, 21. [Google Scholar] [CrossRef] [Green Version]
- Döll, S.; Dänicke, S. The Fusarium toxins deoxynivalenol (DON) and zearalenone (ZON) in animal feeding. Prev. Vet. Med. 2011, 102, 132–145. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.-H.; Weng, C.-F.; Chen, B.-J.; Chang, M.-H. Toxicity of different Fusarium mycotoxins on growth performance, immune responses and efficacy of a mycotoxin degrading enzyme in pigs. Anim. Res. 2006, 55, 579–590. [Google Scholar] [CrossRef] [Green Version]
- Ogunade, I.; Martinez-Tuppia, C.; Queiroz, O.; Jiang, Y.; Drouin, P.; Wu, F.; Vyas, D.; Adesogan, A. Silage review: Mycotoxins in silage: Occurrence, effects, prevention, and mitigation. J. Dairy Sci. 2018, 101, 4034–4059. [Google Scholar] [CrossRef]
- Coppock, R.W.; Jacobsen, B.J. Mycotoxins in animal and human patients. Toxicol. Ind. Health 2009, 25, 637–655. [Google Scholar] [CrossRef]
- Tiemann, U.; Brüssow, K.-P.; Jonas, L.; Pöhland, R.; Schneider, F.; Dänicke, S. Effects of diets with cereal grains contaminated by graded levels of two Fusarium toxins on selected immunological and histological measurements in the spleen of gilts1,2. J. Anim. Sci. 2006, 84, 236–245. [Google Scholar] [CrossRef] [Green Version]
- Tiemann, U.; Brüssow, K.-P.; Dänicke, S.; Vanselow, J. Feeding of pregnant sows with mycotoxin-contaminated diets and their non-effect on foetal and maternal hepatic transcription of genes of the insulin-like growth factor system. Food Addit. Contam. Part A 2008, 25, 1365–1373. [Google Scholar] [CrossRef]
- Reddy, K.E.; Song, J.; Lee, H.-J.; Kim, M.; Kim, D.-W.; Jung, H.J.; Kim, B.; Lee, Y.; Yu, D.; Kim, D.-W.; et al. Effects of High Levels of Deoxynivalenol and Zearalenone on Growth Performance, and Hematological and Immunological Parameters in Pigs. Toxins 2018, 10, 114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reddy, K.E.; Lee, W.; Jeong, J.Y.; Lee, Y.; Lee, H.-J.; Kim, M.S.; Kim, D.-W.; Yu, D.; Cho, A.; Oh, Y.K.; et al. Effects of deoxynivalenol- and zearalenone-contaminated feed on the gene expression profiles in the kidneys of piglets. Asian-Australas. J. Anim. Sci. 2018, 31, 138–148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reddy, K.E.; Jeong, J.Y.; Lee, Y.; Lee, H.-J.; Kim, M.S.; Kim, D.-W.; Jung, H.J.; Choe, C.; Oh, Y.K.; Lee, S.D. Deoxynivalenol- and zearalenone-contaminated feeds alter gene expression profiles in the livers of piglets. Asian-Australas. J. Anim. Sci. 2018, 31, 595–606. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patil, R.D.; Sharma, R.; Asrani, R.K. Mycotoxicosis and its control in poultry: A review. J. Poult. Sci. Technol. 2014, 2, 1–10. [Google Scholar]
- Filazi, A.; Yurdakok-Dikmen, B.; Kuzukiran, O.; Sireli, U.T. Mycotoxins in Poultry. In Poultry Science; Manafi, M., Ed.; IntechOpen: London, UK, 2017; pp. 73–92. [Google Scholar] [CrossRef] [Green Version]
- Swamy, H.; Smith, T.; Cotter, P.; Boermans, H.; Sefton, A. Effects of feeding blends of grains naturally contaminated with Fusarium mycotoxins on production and metabolism in broilers. Poult. Sci. 2002, 81, 966–975. [Google Scholar] [CrossRef] [PubMed]
- Swamy, H.V.L.N.; Smith, T.K.; Karrow, N.A.; Boermans, H.J. Effects of feeding blends of grains naturally contaminated with Fusarium mycotoxins on growth and immunological parameters of broiler chickens. Poult. Sci. 2004, 83, 533–543. [Google Scholar] [CrossRef]
- Girish, C.; Smith, T.; Boermans, H.; Karrow, N. Effects of Feeding Blends of Grains Naturally Contaminated with Fusarium Mycotoxins on Performance, Hematology, Metabolism, and Immunocompetence of Turkeys. Poult. Sci. 2008, 87, 421–432. [Google Scholar] [CrossRef]
- Yunus, A.W.; Blajet-Kosicka, A.; Kosicki, R.; Khan, M.Z.; Rehman, H.; Böhm, J. Deoxynivalenol as a contaminant of broiler feed: Intestinal development, absorptive functionality, and metabolism of the mycotoxin. Poult. Sci. 2012, 91, 852–861. [Google Scholar] [CrossRef]
- Lee, J.T.; Jessen, K.A.; Beltran, R.; Starkl, V.; Schatzmayr, G.; Borutova, R.; Caldwell, D.J. Mycotoxin-contaminated diets and deactivating compound in laying hens: 1. Effects on performance characteristics and relative organ weight. Poult. Sci. 2012, 91, 2089–2095. [Google Scholar] [CrossRef]
- Antonissen, G.; Van Immerseel, F.; Pasmans, F.; Ducatelle, R.; Janssens, G.P.J.; De Baere, S.; Mountzouris, K.; Su, S.; Wong, E.A.; De Meulenaer, B.; et al. Mycotoxins Deoxynivalenol and Fumonisins Alter the Extrinsic Component of Intestinal Barrier in Broiler Chickens. J. Agric. Food Chem. 2015, 63, 10846–10855. [Google Scholar] [CrossRef]
- Antonissen, G.; Croubels, S.; Pasmans, F.; Ducatelle, R.; Eeckhaut, V.; Devreese, M.; Verlinden, M.; Haesebrouck, F.; Eeckhout, M.; De Saeger, S.; et al. Fumonisins affect the intestinal microbial homeostasis in broiler chickens, predisposing to necrotic enteritis. Veter.-Res. 2015, 46, 98. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Quan, H.; Li, X.; Li, Q.; Haque, A.; Shi, Q.; Fu, Q.; He, C. Contamination with Fumonisin B and Deoxynivalenol Is a Threat to Egg Safety and Contributes to Gizzard Ulcerations of Newborn Chickens. Front. Microbiol. 2021, 12, 676671. [Google Scholar] [CrossRef]
- Chen, S.S.; Li, Y.-H.; Lin, M.-F. Chronic Exposure to the Fusarium Mycotoxin Deoxynivalenol: Impact on Performance, Immune Organ, and Intestinal Integrity of Slow-Growing Chickens. Toxins 2017, 9, 334. [Google Scholar] [CrossRef] [Green Version]
- Metayer, J.-P.; Travel, A.; Mika, A.; Bailly, J.-D.; Cleva, D.; Boissieu, C.; Le Guennec, J.; Froment, P.; Albaric, O.; Labrut, S.; et al. Lack of Toxic Interaction between Fusariotoxins in Broiler Chickens Fed throughout Their Life at the Highest Level Tolerated in the European Union. Toxins 2019, 11, 455. [Google Scholar] [CrossRef] [Green Version]
- Travel, A.; Metayer, J.-P.; Mika, A.; Bailly, J.-D.; Cleva, D.; Boissieu, C.; Le Guennec, J.; Albaric, O.; Labrut, S.; Lepivert, G.; et al. Toxicity of Fumonisins, Deoxynivalenol, and Zearalenone Alone and in Combination in Turkeys Fed with the Maximum European Union–Tolerated Level. Avian Dis. 2019, 63, 703–712. [Google Scholar] [CrossRef]
- Kolawole, O.; Graham, A.; Donaldson, C.; Owens, B.; Abia, W.A.; Meneely, J.; Alcorn, M.J.; Connolly, L.; Elliott, C.T. Low Doses of Mycotoxin Mixtures below EU Regulatory Limits Can Negatively Affect the Performance of Broiler Chickens: A Longitudinal Study. Toxins 2020, 12, 433. [Google Scholar] [CrossRef]
- Dwivedi, P.; Burns, R.B. Pathology of ochratoxin A in young broiler chicks. Res. Vet. Sci. 1984, 36, 92–103. [Google Scholar] [CrossRef]
- Dwivedi, P.; Burns, R.; Maxwell, M. Ultrastructural study of the liver and kidney in ochratoxicosis A in young broiler chicks. Res. Veter.-Sci. 1984, 36, 104–116. [Google Scholar] [CrossRef]
- Dwivedi, P.; Burns, R. Effect of ochratoxin A on immunoglobulins in broiler chicks. Res. Veter.-Sci. 1984, 36, 117–121. [Google Scholar] [CrossRef]
- Dwivedi, P.; Burns, R. Immunosuppressive effects of Ochratoxin a in young Turkeys. Avian Pathol. 1985, 14, 213–225. [Google Scholar] [CrossRef] [Green Version]
- Tamilmani, T.; Biswas, A.; Mandal, A. Performance, Immune Response and Blood Biochemical Traits of Broiler Chickens Fed Graded Levels of Dietary Aflatoxin and Ochratoxin Combination. Indian J. Anim. Res. 2020, 55, 704–709. [Google Scholar] [CrossRef]
- Zhai, S.; Zhu, Y.; Feng, P.; Li, M.; Wang, W.; Yang, L.; Yang, Y. Ochratoxin A: Its impact on poultry gut health and microbiota, an overview. Poult. Sci. 2021, 100, 101037. [Google Scholar] [CrossRef] [PubMed]
- Weibking, T.S.; Ledoux, D.R.; Bermudez, A.J.; Turk, J.R.; Rottinghaus, G.E.; Wang, E.; Merrill, J.A.H. Effects of Feeding Fusarium moniliforme Culture Material, Containing Known Levels of Fumonisin B1, on the Young Broiler Chick. Poult. Sci. 1993, 72, 456–466. [Google Scholar] [CrossRef]
- Weibking, T.; LeDoux, D.R.; Bermudez, A.J.; Turk, J.R.; Rottinghaus, G.E. Effects on Turkey Poults of Feeding Fusarium moniliforme M-1325 Culture Material Grown under Different Environmental Conditions. Avian Dis. 1995, 39, 32. [Google Scholar] [CrossRef] [PubMed]
- Bermudez, A.J.; LeDoux, D.R.; Rottinghaus, G.E. Effects of Fusarium moniliforme Culture Material Containing Known Levels of Fumonisin B 1 in Ducklings. Avian Dis. 1995, 39, 879–886. [Google Scholar] [CrossRef] [PubMed]
- Danicke, S.; Ueberschar, K.; Halle, I.; Matthes, S.; Valenta, H.; Flachowsky, G. Effect of addition of a detoxifying agent to laying hen diets containing uncontaminated or Fusarium toxin-contaminated maize on performance of hens and on carryover of zearalenone. Poult. Sci. 2002, 81, 1671–1680. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.; Norred, W.P.; Bacon, C.W.; Riley, R.T.; Merrill, A.H., Jr. Inhibition of sphingolipid biosynthesis by fumonisins. Implications for diseases associated with Fusarium moniliforme. J. Biol. Chem. 1991, 266, 14486–14490. [Google Scholar] [CrossRef] [PubMed]
- Murugesan, G.R.; Ledoux, D.R.; Naehrer, K.; Berthiller, F.; Applegate, T.J.; Grenier, B.; Phillips, T.D.; Schatzmayr, G. Prevalence and effects of mycotoxins on poultry health and performance, and recent development in mycotoxin counteracting strategies. Poult. Sci. 2015, 94, 1298–1315. [Google Scholar] [CrossRef]
- Lumsangkul, C.; Chiang, H.-I.; Lo, N.-W.; Fan, Y.-K.; Ju, J.-C. Developmental Toxicity of Mycotoxin Fumonisin B1 in Animal Embryogenesis: An Overview. Toxins 2019, 11, 114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alaboudi, A.R.; Osaili, T.M.; Otoum, G. Quantification of mycotoxin residues in domestic and imported chicken muscle, liver and kidney in Jordan. Food Control 2022, 132, 108511. [Google Scholar] [CrossRef]
- Antonissen, G.; De Baere, S.; Devreese, M.; Van Immerseel, F.; Martel, A.; Croubels, S. Feed contamination with Fusarium mycotoxins induces a corticosterone stress response in broiler chickens. Poult. Sci. 2017, 96, 14–17. [Google Scholar] [CrossRef] [PubMed]
- Boermans, H.J.; Leung, M. Mycotoxins and the pet food industry: Toxicological evidence and risk assessment. Int. J. Food Microbiol. 2007, 119, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Tegzes, J.H.; Oakley, B.B.; Brennan, G. Comparison of mycotoxin concentrations in grain versus grain-free dry and wet commercial dog foods. Toxicol. Commun. 2019, 3, 61–66. [Google Scholar] [CrossRef] [Green Version]
- Thompson, A. Ingredients: Where Pet Food Starts. Top. Companion Anim. Med. 2008, 23, 127–132. [Google Scholar] [CrossRef] [PubMed]
- El-Tawaab, A.A.A.; El-Hofy, F.I.; Mahmoud, A.H.; Rashed, D.M. Mycotoxin residues in different chicken products by HPLC and their inactivation using Gamma radiation. Int. J. Pharm. Res. Allied Sci. 2019, 8, 71–81. [Google Scholar]
- Castaldo, L.; Graziani, G.; Gaspari, A.; Izzo, L.; Tolosa, J.; Rodríguez-Carrasco, Y.; Ritieni, A. Target Analysis and Retrospective Screening of Multiple Mycotoxins in Pet Food Using UHPLC-Q-Orbitrap HRMS. Toxins 2019, 11, 434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shao, M.; Li, L.; Gu, Z.; Yao, M.; Xu, D.; Fan, W.; Yan, L.; Song, S. Mycotoxins in commercial dry pet food in China. Food Addit. Contam. Part B 2018, 11, 237–245. [Google Scholar] [CrossRef]
- Leung, M.C.K.; Díaz-Llano, G.; Smith, T.K. Mycotoxins in Pet Food: A Review on Worldwide Prevalence and Preventative Strategies. J. Agric. Food Chem. 2006, 54, 9623–9635. [Google Scholar] [CrossRef]
- Rumbeiha, W.; Morrison, J. A Review of Class I and Class II Pet Food Recalls Involving Chemical Contaminants from 1996 to 2008. J. Med. Toxicol. 2011, 7, 60–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wouters, A.T.B.; Casagrande, R.A.; Wouters, F.; Watanabe, T.T.N.; Boabaid, F.M.; Cruz, C.E.F.; Driemeier, D. An outbreak of aflatoxin poisoning in dogs associated with aflatoxin B1–contaminated maize products. J. Veter.-Diagn. Investig. 2013, 25, 282–287. [Google Scholar] [CrossRef] [Green Version]
- FDA. Recalls & Withdrawals. The U.S. Food and Drug Administration. 2022. Available online: https://www.fda.gov/animal-veterinary/safety-health/recalls-withdrawals (accessed on 27 November 2022).
- Glanemann, B.; Humm, K.; Pegram, C.; Chan, D.L. An investigation into an outbreak of pancytopenia in cats in the United Kingdom. J. Veter.-Intern. Med. 2023, 37, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Zadravec, M.; Markov, K.; Lešić, T.; Frece, J.; Petrović, D.; Pleadin, J. Biocontrol Methods in Avoidance and Downsizing of Mycotoxin Contamination of Food Crops. Process 2022, 10, 655. [Google Scholar] [CrossRef]
- Leslie, J.F.; Moretti, A.; Mesterházy, Á.; Ameye, M.; Audenaert, K.; Singh, P.K.; Richard-Forget, F.; Chulze, S.N.; Del Ponte, E.M.; Chala, A.; et al. Key Global Actions for Mycotoxin Management in Wheat and Other Small Grains. Toxins 2021, 13, 725. [Google Scholar] [CrossRef] [PubMed]
- Nada, S.; Nikola, T.; Bozidar, U.; Ilija, D.; Andreja, R. Prevention and practical strategies to control mycotoxins in the wheat and maize chain. Food Control 2022, 136, 108855. [Google Scholar] [CrossRef]
- Dorner, J.W. Biological Control of Aflatoxin Contamination in Corn Using a Nontoxigenic Strain of Aspergillus flavus. J. Food Prot. 2009, 72, 801–804. [Google Scholar] [CrossRef]
- Pitt, J.; Manthong, C.; Siriacha, P.; Chotechaunmanirat, S.; Markwell, P. Studies on the biocontrol of aflatoxin in maize in Thailand. Biocontrol Sci. Technol. 2015, 25, 1070–1091. [Google Scholar] [CrossRef]
- Karunakaran, C.; Muir, W.; Jayas, D.; White, N.; Abramson, D. Safe storage time of high moisture wheat. J. Stored Prod. Res. 2001, 37, 303–312. [Google Scholar] [CrossRef]
- Jayas, D.S.; White, N.D. Storage and drying of grain in Canada: Low cost approaches. Food Control 2003, 14, 255–261. [Google Scholar] [CrossRef]
- Akhila, P.P.; Sunooj, K.V.; Navaf, M.; Aaliya, B.; Sudheesh, C.; Sasidharan, A.; Sabu, S.; Mir, S.A.; George, J.; Khaneghah, A.M. Application of innovative packaging technologies to manage fungi and mycotoxin contamination in agricultural products: Current status, challenges, and perspectives. Toxicon 2022, 214, 18–29. [Google Scholar] [CrossRef]
- FAO. Food and Agricultural Organization-Worldwide Regulations for Mycotoxins in Food and Feed in 2003. FAO Food and Nutrition Paper No. 81. Rome, Italy. 2004. Available online: https://www.fao.org/3/y5499e/y5499e0j.htm#bm19.1.8 (accessed on 4 December 2022).
- CXS 193-1995; CAC. The Codex Alimentarius Commission. General Standard for Contaminants and Toxins in Food and Feed, amended in 2019. FAO/WHO of the United Nations: New York, NY, USA.
- EC. European Commission 2006. Commission Regulation (EC) No 1881/2006 of 19 December as amended, on Setting Maximum Levels of Certain Contaminants in Foodstuffs. 2006. Available online: https://leap.unep.org/countries/eu/national-legislation/commission-regulation-ec-no-18812006-setting-maximum-levels (accessed on 4 December 2022).
- ANVISA (Agência Nacional de Vigilância Sanitária). Dispõe Sobre Limites Máximos Tolerados (LMT) Para Micotoxinas em Alimentos; Resolução RDC n. 7, 18/02/2011; Diário Oficial da União: Brasília, Brazil, 2011.
- USDA-FAS. USDA Foreign Agricultural Service, GAIN Report Number:CH18026. China Releases Standard for Maximum Levels of Mycotoxins in Foods. 2018. Available online: https://www.fas.usda.gov/data/china-china-releases-standard-maximum-levels-mycotoxins-foods (accessed on 4 June 2022).
- FASA India, Food Safety and Standards Authority of India. First Amendment Regulation Related to Limit of Metal Contaminant, Aflatoxin and Mycotoxin. 2020. Available online: https://www.fssai.gov.in/upload/notifications/2020/08/5f3d09f97b78aGazette_Notification_Limit_Metal_19_08_2020.pdf (accessed on 13 October 2022).
- Li, X.; Zhao, L.; Fan, Y.; Jia, Y.; Sun, L.; Ma, S.; Ji, C.; Ma, Q.; Zhang, J. Occurrence of mycotoxins in feed ingredients and complete feeds obtained from the Beijing region of China. J. Anim. Sci. Biotechnol. 2014, 5, 37. [Google Scholar] [CrossRef] [Green Version]
- South African Government. Farm Feeds Regulations: Amendment. Department of Agriculture, Forestry and Fisheries No.R.70. 2020. Available online: https://www.gov.za/sites/default/files/gcis_document/201409/3293570.pdf (accessed on 12 February 2022).




| Region | Major Mycotoxins | Number of Samples Tested | Positive Rate (%) | Median (µg/kg) | Average Level (µg/kg) | Maximum Level (µg/kg) |
|---|---|---|---|---|---|---|
| Europe | Aflatoxin (total) | 3711 | 7 | 2 | 6 | 92 |
| ZEA | 6185 | 47 | 28 | 100 | 57,147 | |
| DON | 6565 | 60 | 263 | 351 | 11,875 | |
| T-2 | 4156 | 32 | 15 | 3 | 1387 | |
| FUM | 4187 | 46 | 170 | 645 | 16,241 | |
| OTA | 3666 | 15 | 3 | 9 | 560 | |
| North America | Aflatoxin (total) | 1655 | 4 | 4 | 26 | 482 (corn) |
| ZEA | 1661 | 39 | 120 | 293 | 26,466 | |
| DON | 1604 | 75 | 474 | 789 | 43,517 | |
| T-2 | 1486 | 2 | 80 | 185 | 3153 | |
| FUM | 1655 | 48 | 827 | 2738 | 66,588 | |
| OTA | 1655 | 3 | 3 | 27 | 750 | |
| South and Central America | Aflatoxin (total) | 7258 | 19 | 3 | 5 | 179 |
| ZEA | 6724 | 46 | 61 | 149 | 43,852 | |
| DON | 6134 | 61 | 440 | 736 | 26,320 | |
| T-2 | 3474 | 20 | 33 | 41 | 321 | |
| OTA | 6759 | 71 | 3 | 1832 | 56,000 | |
| FUM | 2095 | 9 | 1120 | 7 | 86 | |
| Asia | Aflatoxin (total) | 3350 | 25 | 9 | 47 | 2495 |
| ZEA | 3247 | 62 | 44 | 145 | 11,786 | |
| DON | 3360 | 71 | 365 | 546 | 17,550 | |
| T-2 | 2873 | 7 | 22 | 31 | 169 | |
| OTA | 3225 | 81 | 501 | 1316 | 35,445 | |
| FUM | 2892 | 25 | 3 | 12 | 571 | |
| Middle East and North Africa | Aflatoxin (total) | 116 | 7 | 2 | 2 | 5 |
| ZEA | 119 | 68 | 34 | 134 | 1928 | |
| DON | 119 | 78 | 225 | 497 | 5170 | |
| T-2 | 109 | 17 | 10 | 11 | 30 | |
| OTA | 119 | 76 | 307 | 769 | 8586 | |
| FUM | 111 | 12 | 2 | 3 | 7 | |
| Africa (without North Africa) | Aflatoxin (total) | 1059 | 7 | 4 | 28 | 1032 |
| ZEA | 1071 | 44 | 32 | 78 | 3091 | |
| DON | 1071 | 76 | 331 | 592 | 7254 | |
| T-2 | 1071 | 0 | 37 | 43 | 74 | |
| OTA | 1071 | 59 | 142 | 452 | 10,368 | |
| Aflatoxin (total) | 1059 | 5 | 3 | 9 | 84 |
| Country/Region | Mycotoxins Identified | Above EU Limits (%) | References |
|---|---|---|---|
| Africa (corn, n = 20) | 86% of maize and peanut samples contained four mycotoxins including AFB1, FB1, ZEA and OTA. | AFB1: 30% of positive samples (>4 µg/kg) | [63] |
| USA (corn, n = 1828) | 7.6% of samples contain AFs (mean concentration 15.2 ppb), 75.7% contain DON (1.6%: >5000 ppb), and 59.7% have FB (10,000 ppb), 43% contain OTA (4.9 ppb), and 3.4% contain ZEA. | AFs: mean = 15.2 µg/kg, Max = 606 µg/kg DON: 1.6% FB: 59.7% ZEA: 25% | [64] |
| USA (maize samples, n = 90) | Maize samples collected from 10 locations in Michigan state for 2 years. Every sample was contaminated with at least four and six mycotoxins in 2017 and 2018, respectively. Incidence and severity of each mycotoxin varied by year and across locations. | DON: 1.6% of samples exceeded 5000 µg/kg FUMs: 9.6% of grains exceeded 10,000 µg/kg | [65] |
| Brazil (n = 230, processed rice) | Total of 55.1% of samples contain more than 1 mycotoxin. 17% had AFs and ZEA, 24.2% had AFs and OTA, 6.2% had AFs and citreoviridin (CTV), 4.6% had OTA and CTV, and 3.1% had ZON and CTV, respectively. | AFs: 10–20 µg/kg in 4%, >20 to 30 µg/kg in 2%, and >30.00 µg/kg in 3%. | [66] |
| Canada (corn, n = 750) | Aflatoxin in 1.1% of samples (4.4 ppb), DON in 41% of samples (282 ppb), FB in 14% (280 ppb), OTA in 2.7% (34 ppb) and ZEA in 4.5%. | N/A | [67] |
| China (corn n = 520) | 93% contains FBs (mean 2528 ppb). 1.0%, 2.7%, 14%, 22%, 44% and 6.0% of the samples were detected with 7, 6, 5, 4, 3 and 2 kinds of mycotoxins, respectively. | N/A | [68,69] |
| China (72 barley samples and 83 wheat samples) | 40 barley (56%) and 35 wheat (42%) samples were mycotoxin positive. Among the positive samples, at least two mycotoxins were detected in 70% of barley samples and 54% of wheat samples. | DON: 6% barley and 6% wheat samples T-2 toxin: 7% barley and 5% wheat samples | [70] |
| China (338 unprocessed wheat samples) | 40 (11.8%), 77 (22.8%), 49 (14.5%) and 41 (12.1%) samples were contaminated with two, three, four and five mycotoxins, respectively. The rate of co-occurrence of fumonisins with other Fusarium toxins was 37.6%. | DON: 44.7–52.4% ZEA: 13.9 AFB1: 0.6–2.1% | [71] |
| Lebanon (durum wheat from two warehouses, n = 300) | 23.3–25.3% of samples had AFB1 levels >2 μg/kg, respectively. 52.0% and 44.6% of samples had OTA levels of 0.51–9.71 μg/kg, respectively. | AFB1: 23.3–25.3% (>2 µg/kg) OTA: 25.33–28.67% (>3 µg/kg) | [72] |
| Korea (brown rice, millet, sorghum, maize and mixed cereal, n = 5) | FUMs, DON, nivalenol and ZEA were more frequently and simultaneously detected in all cereal grains, and 54% of wheat samples had at least two mycotoxins. AFB1 was detected in 1% (brown rice)–9% (millet) of each grain group with mean levels 1.1–5.2 ng/g. | AFB1: 4% (>10 µg/kg) | [73] |
| Nigeria (rice, n = 21) | AFs in all samples at 28–372 μg/kg. OTA, ZEA, DON, FB1 and FB2 in 66.7, 53.4, 23.8, 14.3 and 4.8% of the samples. Co-occurrence of AFs, OTA and ZEA was very common, and up to five mycotoxins were detected in one sample. | AFB1: 100% (28–372 µg/kg) OTA: 66.7% (134–341 μg/kg) | [74] |
| India (n = 150, maize) | 150 freshly harvested maize samples during 2010–2011 and 2011–2012. 28, 20, 58, 23 and 11 were positive for AFB1, OTA FB1, DON and T-2 toxin, respectively. | AFB1: 18.7% (48–58 µg/kg) | [75] |
| Pakistan (rice, n = 208) | 35% of samples were AFs positive and 19% were OTA positive, respectively. | AFB1: 19% of positive Total AFs: 24% positive OTA: 14% | [76] |
| Pakistan (corn, n = 7) | 100% of samples were AFs positive and a higher level of AFG1 in all maize varieties. OTA was detected in 71% of maize samples at 2.14–214 μg/kg. | Total AFs: 100% (>20 μg/kg) OTA: 52.2% (>5 μg/kg) | [77] |
| Africa (corn, n = 444) | AFs and FUM co-contamination occurred in 35% of the samples. | AFs: 31.7% FUMs: 1.3% DON: 8.9% ZEA: 3.8% OTA: 4.2% | [78] |
| Ghana (maize, n = 180) | 72.2% of samples were AFT positive with a total AFT 4.27–441.02 µg/kg, in the order of AFB1 > AFB2 > AFG1 > AFG2, 57.2% of samples were OTA positive: 4.00–97.51 µg/kg. | AFs: 70.50% > EU limits, 64.44% > Ghana limits OTA: 54.1% > EU limit, 49.9% > Ghana limit | [79] |
| Kenya (maize, n = 350) | 55% of 350 maize samples collected following the 2004 aflatoxicosis had AFs at levels higher than 20 µg/kg. 35% had levels > 100 ppb. | AFs: 55% (>20 µg/kg) | [80] |
| Kenya (maize, n = 350) | AFB1 and FUM were found in 80 and 85% of the samples, respectively. AFB1 in 25% and FUM in 48% of samples exceeded EU limits. | AFB1: 25% (>5 μg/kg) FUMs: 48% (>2 mg/kg) | [81] |
| Kenya (milled rice, n = 204) | Sterigmatocystin aflatoxin, citrinin, OTA, fumonisin, diacetoxyscirpenol, HT2, T2 and DON were identified. 3.5% of samples had six toxins in different combinations. | AFs: 13.5%, OTA: 6%; HT2 + T2: 0.5% | [82] |
| Uganda (n = 105, different grains) | AFs and OTA were detected in 8.3–100% of samples, and co-occurrence of AFs, OTA and DON ranged from 8.3–35.3%, with the highest incidence in sorghum. | N/A | [83] |
| Country or Region | Type of Foods | Sample Number | Mycotoxins Detected | Concentration Range (μg/kg) | >EU Limits (%) | Method of Detection | Reference |
|---|---|---|---|---|---|---|---|
| Canada | Cereal-based infant foods | 627 | OTA positive in 41% of samples, 114 exceeded Canadian regulation | Up to 4.85 with a mean of 0.59 μg/L | 23% | LC-MS | [86] |
| USA | Infant cereals | 155 | OTA positive: 47 (30%) of 155 infant cereals | 0.6–22.1 | 100% of the positive samples | LC-MS/MS | [87] |
| USA | Breakfast cereals | 489 | OTA in 205 samples (42%) | 0.1–9.3 | 3.3% | HPLC-FLD | [88] |
| Spain | Corn-based foods | 25 | The incidence of DON, ZEA ZOL and T-2 was 68, 44, 24% and 0.4%, respectively | DON: 29–195 ZEA: 34–216 ZOL: 36–71 T-2: <50 | N/A | GC-FID and HPLC | [89] |
| Spain | Infant foods | 60 | Aflatoxins in 12 samples (20%) | N/A | 10% | HPLC-FLD | [90] |
| China | Infant foods in the market | 820 | Low levels of 12 mycotoxins in the following order: DON (55.7%) > ZEA (8.2%) > FB1 (3.7%) > OTA (1.1%) > FB2 (0.7%) | AFs: not detected ZEA: 0.2–8.8 DON: 1.1–912.3 FB1: 242.3 FB2: 252.4 OTA: 0.2–3.0 | FBs: 0.87% | LC-MS/MS | [91] |
| Portugal | Breakfast cereals | 26 | 96% of samples containing multiple mycotoxins including AFs, OTA, FUMs, DON, ZEA | AFB1: 0–0.13 OTA: 0–0.1 FB1 + FB2: 0–84 DON: 59–207.8 ZEA: 0.4–5.6 | 0% | UPLC-FLD GC-MS UPLC-MS/MS | [92] |
| Spain | Breakfast cereals | 72 | AFs in 1 sample | Total AFs: 0.5 | 0% | HPLC-FLD | [93] |
| Spain | Corn snacks | 72 | AFs in 1 sample | 0.8 | 0% | HPLC-FLD | [93] |
| Romania | Wheat-based foods: flour, bread, pasta and biscuit (n = 181) | 181 | DON and 15Ac-DON were detected in 63% (114) and 5% (9) of all samples | DON: 1.9–1947 15 Ac-DON: 14.2–32.6 | DON: 5% | GC-QqQ-MS/MS | [94] |
| Brazil | Maize snacks | 18 | FUM: 100% DON: 8% ZEA: 8% | FUMs: 2.9–303.5 DON: 0–40.4 ZEA: 0–54 | N/A | LC/MS/MS | [95] |
| Breakfast cereals | 10 | FUMs: 100% DON: 10% | FUMs: 2.7–551.8 DON: 0–120.8 | ||||
| Wheat pasta | 30 | FUM: 13.3% DON: 100% ZEA: 73.3% | FUMs: 0–130 DON: 83.9–860.8 ZEA: 0–205.6 | ||||
| Crackers | 14 | DON: 100% ZEA: 100% | DON: 139–916 ZEA: 26.9–117.6 | ||||
| Tanzania | Maize-based flour used for feeding children | 41 | AFs: 32% DON: 44% Fumonisins: 83% | Total AFs: 0.11–386 DON: 57–825 Fumonisins: 63–2284 | N/A | validated HPLC-FLD | [96] |
| Ghana | Infants and young children’s foods derived from cereal | 35 | 71% of samples contained AFB1 | AFB1: 0.18 ± 0.01 to 36.10 ± 0.32 | 71% (>0.1 μg/kg) | HPLC-FLD | [97] |
| Ghana | Rice brands | 27 | Aflatoxins B1, B2, G1 and G2 | AFB1: 65.77–ND AFB2: 19.27–0.01 AFG1: 1.05–ND AFG2: 0.12–ND | 29.6% (>10 µg/kg) | HPLC-FLD | [97] |
| Ghana | Cereal-based food brands | 20 | AFB1, B2, G1 and G2 | AFB1: 35.46–0.96 AFB2: 4.92–0.51 AFG1: 6.95–0.27 AFG2: 0.82–0.1 | 33% (>4 µg/kg) | HPLC-FLD | [98] |
| Ghana | Pasta brands | 6 | AFB1 and B2 | AFB1: 0.930–0.935 AFB2: 0.85–0.853 | 0% | HPLC-FLD | [98] |
| Iran | Baby food | 40 | Aflatoxins B1, B2 and G2 in 20% to 60% of samples | AFB1: 0.04–0.84 AFB2: 0.01–0.08 AFG2: 0.007–008 | AFB1: 30% | HPLC-FLD | [99] |
| Iran | Rice-based baby food | 30 | AFB1 was detected in 68.7% (33/48) of samples | AFB1: 0–15.15 | 39.6% | HPLC-FLD | [100] |
| Morocco | Pasta | 106 | ZEA, DON, HT-2 and T-2 toxins were present in 51.8%, 43.5%, 34.9% and 16% of samples, AFB1 in 2 samples | AFB1: 0–0.25 ZEA: 0.5–3.0 DON: 16–900 HT-2: 4–419 T-2: 4–50 | DON: 21% | LC/MS/MS | [101] |
| South Africa | Corn-based opaque beers | 32 | 94% of samples had 2–5 mycotoxins AFB1: 6%, FB1: 53%, FB2: 32%, FB3: 6%, DON: 84% | AFB1: 5.8–7.0 Total FBs: 36–182 DON: up to 72 | AFB1: 6% | LC-MS | [102] |
| Namibian | Sorghum malt omalodu | 45 | AFB1, AFB2 and AFG1 in 14%, 5% and 3% of otombo malts. FB1, FB2 and FB3, in 42%, 22% and 3% of otombo malts, respectively | AFB1: 0.61–28.3 AFB2: 0.14–2.35 AFG1: 0.39–6.95 FB1: 12–500.2 FB2: 7.55–79.46 FB3: 21.6–136.6 | AFB1: 20% (>5 µg/kg) | LC/MS/MS | [102] |
| Namibian | Sorghum malt otombo | 36 | Aflatoxin B1, B2 and G1 in 14%, 5% and 3% of otombo malts Fumonisin B1, B2 and B3 in 42%, 22% and 3% of otombo malts, respectively | AFB1: 0.56–54.2 AFB2: 0.5–4.48 AFG1: 0.4 FB1: 8.17–88.3 FB2: 5.92–46.8 FB3: 22 | AFB1: 40% (>5 µg/kg) | LC/MS/MS | [103] |
| Pakistan | Processed foods | 125 | 38% of 125 samples were contaminated with four types of aflatoxins | AFB1: 0.02–1.24 AFB2: 0.02–0.37 AFG1: 0.25–2.7 AFG2: 0.21–1.3 | HPLC-FLD | [104] | |
| Pakistan | Breakfast cereal | 237 | 41%: AFs OTA: 48% ZEA: 53% | AFB1: LOD ‡—6.90 Total AFs: LOD—7.45 OTA: LOD—8.45 ZEA: LOD—118.10 | AFB1: 16% Total AFs: 8% OTA: 30% ZEA: 8% | HPLC-FLD | [105] |
| Pakistan | Wheat products Spaghetti Noodles Macaroni Lasagne Bucatini | 25 34 29 37 22 | 36, 24, 34, 24 and 36% of spaghetti, noodles, macaroni, lasagne and bucatini were AFs positive, and 28, 18, 34, 32 and 50% were ZEA positive, respectively | AFs: LOD—55.6 ZEA: LOD—69.8 | AFs: 18–28% ZEA: 15–36% | HPLC-FLD | [106] |
| Poultry Species, Age and Feeding Period | Sample Size (n) | Mycotoxins in Diet and Concentrations | Health Effects | References |
|---|---|---|---|---|
| Broiler chicken, 1 day old, fed for 56 days | 360 | Diet contaminated with fusarium mycotoxins: 0.14–9.7 mg/kg DON, 18–21.6 mg/kg fusaric acid (FA), 0.1–0.8 mg/kg ZEA | Body weight gain and feed intake of chickens decreased quadratically; blood erythrocyte count and serum uric acid concentration increased linearly and the serum lipase activity decreased linearly; a significant quadratic effect on serum albumin and γ-glutamyltransferase activity; blood hemoglobin and biliary IgA concentrations responded in significant linear and quadratic patterns. Efficiency of feed utilization was not affected. | [204] |
| Broiler chicken, 1 day old, fed for 42 days | 360 | Fusarium mycotoxin, 5.9–9.5 mg/kg DON, 19.1–21.4 mg/kg fusaric acid (FA), 0.4–0.7 mg/kg ZEA and 0.3–0.5 mg/kg 15AC-DON | Body weight gains and feed intake of chickens decreased linearly while peripheral blood monocytes decreased linearly with increasing toxin levels during the grower stage (21–42 days). Reduced B-cell count linearly but increased the T-cell count on day 28. | [205] |
| Turkey, 1 day old (n = 300), fed for 12 weeks | 300 | Blends of grains naturally contaminated with fusarium mycotoxins: DON, 15Ac-DON ZEA and FA | Turkey’s performance and some blood and immunological parameters were adversely affected by feedborne fusarium mycotoxins, and polymeric glucomannan mycotoxin adsorbent (GMA) prevented most of the adverse effects. | [206] |
| Male broilers at 7 d of age, fed for 5 weeks | 75 | Diets contain 0.265, 1.68 and 12.2 mg of DON/kg; 0.013, 0.145 and 1.094 mg ZEA/kg | The weekly weight gain decreased linearly (P ≤ 0.041) with increasing DON levels during the first 3 weeks of exposure; the weight gain was not influenced thereafter. As the levels of DON increased, the titers against Newcastle disease virus increased linearly during week 2 and week 4 of exposure, but decreased linearly (P = 0.006) during week 5 of exposure. | [207] |
| 25-wk-old laying hens (n = 384) | 384 | Birds were fed diets contaminated with AFB and DON for a 6-wk phase followed by a 4-week recovery phase | Elevated relative liver and kidney weights (P < 0.05), reduced feed intake, egg production and egg weights (P < 0.05) at the medium and high toxin levels following the toxin phase, but the deactivation compound reduced (P < 0.05) relative liver and kidney weights following the recovery period. | [208] |
| One-day-old broiler chicks (n= 308), fed for 16 days | 308 | Diet contaminated by FBs (18.6 mg FB1 + FB2/kg feed) | A significant increase in the plasma sphinganine/sphingosine ratio. Villus height and crypt depth of ileum were significantly reduced. Changed the microbiota composition in the ileum. A higher percentage of chickens fed an FB-contaminated diet developed subclinical necrotic enteritis following C. perfringens challenge. | [209] |
| One-day-old broiler chicks, fed for 15 days | 308 | Diet 1: contaminated with 4.6 mg DON/kg. Diet 2: contaminated with 25.4 mg FB1 + FB2/kg. Diet 3: containing 4.3 mg DON and 22.9 mg FB1 + FB2/kg. | Changed the intestinal mucus layer and several intestinal epithelial antioxidative mechanisms. Both mycotoxins decreased gene expression of the intestinal zinc transporter (ZnT)-1 and regulated intracellular methionine homeostasis, which are both important for preserving the cell’s critical antioxidant activity. | [210] |
| SPF embryonated eggs aged 11 days | 221 | Inoculated into albumen with different doses of FB1, FB2 or DON, or their combinations. | Reduced hatching rate, caused gizzard ulcers and hemorrhagic lungs. Resulted in higher mortality of the progeny of breeder hens and higher mycotoxin residues in the gizzards and the lungs of the progenies. | [211] |
| 5-day-old chickens, fed for 16 weeks | 280 | Diets containing 0, 2, 5 and 10 mg/kg of DON. | The diets contaminated with DON at 5 mg/kg caused heavier spleens, increase DON-induced cellular proliferation, apoptosis and DNA damage signals in the spleen. Expression of gatekeeper protein claudin-5 was increased in jejunum of female birds but decreased in that of male birds. | [212] |
| Broiler chickens | 70 | Five groups of chickens fed control diets (mycotoxin free), the DON diets (5 mg/kg), the FB diet (20 mg FB1 + FB2/kg), ZEA diet (0.5 mg/kg) and diets contained 5, 20, and 0.5 mg/kg of DON, FB1 + FB2, and ZEA. | No difference in performances between groups that could be attributed to FBs, the relative weight of organs, biochemistry, histopathology, intestinal morphometry, indicators of oxidative damage and markers of testicle toxicity. Significantly increased sphinganine and SA to SO ratio in broilers fed FB-added diets. | [213] |
| Turkeys of 55-day old fed experimental diet for 14 days | 70 | Five groups of turkeys fed with control diets, the DON diets (5 mg/kg), the FB diet (20 mg FB1 + FB2/kg), ZEA diet (0.5 mg/kg) and diets with DON, FB1 + FB2, and ZEA (5, 20, and 0.5 mg/kg of), respectively. | Increased the SA to SO ratio in the liver of turkeys fed diets containing FB, but had no apparent toxication symptoms. No interactions/synergies among DON, FB, and ZON. | [214] |
| One-day-old chicks used for each trial (n = 2200) | 2200 | Feed contaminated with low levels of mycotoxins (below EU’s regulatory limits). | A strong positive relationship was observed between broilers’ feed efficiency and DON (R2 = 0.85), FBs (R2 = 0.53), DAS (R2 = 0.86), ZEA (R2 = 0.92), ENNs (R2 = 0.60) and BEV (R2 = 0.73). The mixture of ZEA, DON and FBs (p = 0.01, R2 = 0.84), and the mixture of ZEA, DON and DAS (p = 0.001, R2 = 0.91) had significant interactive effect on the birds’ feed efficiency. | [215] |
| Day-old male cobb chicks (1600 birds, 64 pens, 25 birds/pen) | 1600 | Broilers were fed control diet with minimal mycotoxins and formulated diet containing moderate levels of fusarium mycotoxins (MT). | Compared with control, the diet containing moderate fusarium mycotoxins reduced body weight (BW), increased feed efficiency on days 35 and 42 with increased duodenal crypt depth and reduced goblet cells, and poultry production efficiency. | [64] |
| Date of Recall | Brand Name | Product Description | Recall Reason | Company Name |
|---|---|---|---|---|
| July 29, 2021 | Triumph, Evolve, Nature Farms, Elm and others | Dog Food | High levels of aflatoxin | Sunshine Mills, Inc., Halifax, VA, USA |
| January 11, 2021 | Sportmix, Nunn Better, ProPac and others | Dog and Cat Pet Food | Aflatoxin exceeds acceptable levels | Midwest Pet Food, Inc., Evansville, IN, USA |
| December 30, 2020 | Sportmix | Dog and Cat Food | High levels of aflatoxin | Midwest Pet Food, Inc., Evansville, IN, USA |
| October 8, 2020 | Champ, Field Trial, Good Dog and others | Pet Food | Possibly aflatoxin | Sunshine Mills, Inc., Halifax, VA, USA |
| September 2, 2020 | Family Pet, Heartland Farms and Paws Happy Life | Dog Food | Elevated levels of aflatoxin | Sunshine Mills, Inc., Halifax, VA, USA |
| September 23, 2019 | Gramco | Hog Grower Pellets | High levels of vomitoxin (DON) | Gramco, Inc., Springville, NY, USA |
| May 6, 2019 | Southern States | Various Animal Feed | Elevated aflatoxin levels | Cargill, Inc., Cleveland, NC, USA |
| European Union (µg/kg) | CAC (µg/kg) | US (µg/kg) | Brazil (µg/kg) | China (µg/kg) | India (µg/kg) | South Africa (µg/kg) | ||
|---|---|---|---|---|---|---|---|---|
| Total Aflatoxins | Total | 4–10 | 15 | 20 | 50 | 20 | 15 | 10 |
| Aflatoxin B1 | B1 | 2–5 | 10 | 20 | 30 | 5–20 | 10 | 5 |
| Deoxynivalenol (DON) | 1250–1750 | 2000 | 1000 | 200–3000 | 1000 | 1000 | NR | |
| Fumonisins (B1 + B2) | B1 + B2 | 1000–4000 * | 4000 | 2000–4000 * | 1000–5000 * | NR | NR | NR |
| Ochratoxin A | 3–5 | 5 | NR | 50 | 5 | NR | NR | |
| Zearalenone (ZEA) | 100–350 * | 100 | NR | 100–600 * | 60 | NR | NR | |
| Reference | [253] | [252] | [251] | [254] | [255] | [256] | [251] |
| European Union (µg/kg) | US (µg/kg) | Brazil (µg/kg) | China (µg/kg) | India (µg/kg) | South Africa (µg/kg) | ||
|---|---|---|---|---|---|---|---|
| Total Aflatoxins | Total | 50 | 20–300 * | 50 | NR | ||
| Aflatoxin B1 | B1 | 20 | NR | 10–50 * | NR | 50 | |
| Deoxynivalenol (DON) | 8000 | 5000–10,000 * | NR | 1000–5000 * | NR | 5000 | |
| Fumonisins (B1 + B2) | 60,000 | 5000–100,000 * | NR | 5000–50,000 * | NR | 50,000 | |
| Ochratoxin A | 250 | NR | NR | 100 | NR | 200 | |
| Zearalenone (ZEA) | 2000–3000 | NR | NR | 100–500 * | NR | 5000 | |
| Reference | [253] | [251] | [253,254] | [257] | [256] | [253,258] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, J.; Pedroso, I.R. Mycotoxins in Cereal-Based Products and Their Impacts on the Health of Humans, Livestock Animals and Pets. Toxins 2023, 15, 480. https://doi.org/10.3390/toxins15080480
Yu J, Pedroso IR. Mycotoxins in Cereal-Based Products and Their Impacts on the Health of Humans, Livestock Animals and Pets. Toxins. 2023; 15(8):480. https://doi.org/10.3390/toxins15080480
Chicago/Turabian StyleYu, Jianmei, and Ivana Ramos Pedroso. 2023. "Mycotoxins in Cereal-Based Products and Their Impacts on the Health of Humans, Livestock Animals and Pets" Toxins 15, no. 8: 480. https://doi.org/10.3390/toxins15080480
APA StyleYu, J., & Pedroso, I. R. (2023). Mycotoxins in Cereal-Based Products and Their Impacts on the Health of Humans, Livestock Animals and Pets. Toxins, 15(8), 480. https://doi.org/10.3390/toxins15080480




