SelectMDx and Multiparametric Magnetic Resonance Imaging of the Prostate for Men Undergoing Primary Prostate Biopsy: A Prospective Assessment in a Multi-Institutional Study
Abstract
:Simple Summary
Abstract
1. Introduction
2. Material and Methods
2.1. Study Population
2.2. mpMRI
2.3. SelectMDx Sampling
2.4. Prostatic Biopsy
2.5. Statistical Analysis
3. Results
3.1. SelectMDx Performance for PCa and csPCa at Biopsy
3.2. Impact of Implementation of SelectMDx Versus PSAD into the mpMRI Pathway to Select Patients Candidate for Prostate Biopsy
3.3. Impact of Different Screening Strategies to Select Patients Candidate for Prostate Biopsy
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mottet, N.; van den Bergh, R.C.N.; Briers, E.; Van den Broeck, T.; Cumberbatch, M.G.; De Santis, M.; Fanti, S.; Fossati, N.; Gandaglia, G.; Gillessen, S.; et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer—2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur. Urol. 2021, 79, 243–262. [Google Scholar] [CrossRef]
- Draisma, G.; Etzioni, R.; Tsodikov, A.; Mariotto, A.; Wever, E.; Gulati, R.; Feuer, E.; de Koning, H. Lead time and overdiagnosis in prostate-specific antigen screening: Importance of methods and context. J. Natl. Cancer Inst. 2009, 101, 374–383. [Google Scholar] [CrossRef] [Green Version]
- Logozzi, M.; Mizzoni, D.; Capasso, C.; Del Prete, S.; Di Raimo, R.; Falchi, M.; Angelini, D.F.; Sciarra, A.; Maggi, M.; Supuran, C.T.; et al. Plasmatic exosomes from prostate cancer patients show increased carbonic anhydrase IX expression and activity and low pH. J. Enzyme Inhib. Med. Chem. 2020, 35, 280–288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maggi, M.; Gentilucci, A.; Salciccia, S.; Gatto, A.; Gentile, V.; Colarieti, A.; Von Heland, M.; Busetto, G.M.; Del Giudice, F.; Sciarra, A. Psychological impact of different primary treatments for prostate cancer: A critical analysis. Andrologia 2019, 51, e13157. [Google Scholar] [CrossRef] [PubMed]
- Panebianco, V.; Barchetti, F.; Sciarra, A.; Ciardi, A.; Indino, E.L.; Papalia, R.; Gallucci, M.; Tombolini, V.; Gentile, V.; Catalano, C. Multiparametric magnetic resonance imaging vs. standard care in men being evaluated for prostate cancer: A randomized study. Urol. Oncol. 2015, 33, 17.e1–17.e7. [Google Scholar] [CrossRef]
- Kasivisvanathan, V.; Rannikko, A.S.; Borghi, M.; Panebianco, V.; Mynderse, L.A.; Vaarala, M.H.; Briganti, A.; Budäus, L.; Hellawell, G.; Hindley, R.G.; et al. MRI-Targeted or Standard Biopsy for Prostate-Cancer Diagnosis. N. Engl. J. Med. 2018, 378, 1767–1777. [Google Scholar] [CrossRef] [PubMed]
- Cooperberg, M.R.; Carroll, P.R.; Dall’Era, M.A.; Davies, B.J.; Davis, J.W.; Eggener, S.E.; Feng, F.Y.; Lin, D.W.; Morgan, T.M.; Morgans, A.K.; et al. The State of the Science on Prostate Cancer Biomarkers: The San Francisco Consensus Statement. Eur. Urol. 2019, 76, 268–272. [Google Scholar] [CrossRef]
- Sciarra, A.; Maggi, M.; Salciccia, S.; Nicolai, A.; Tortorella, E.; Giantulli, S.; Magliocca, F.M.; Silvestri, I.; Taglieri, L.; Cattarino, S.; et al. Tissue Expression of Androgen Receptor Splice Variant 7 at Radical Prostatectomy Predicts Risk of Progression in Untreated Nonmetastatic Prostate Cancer. Oncology 2021, 18, 1–5. [Google Scholar]
- Catalona, W.J.; Partin, A.W.; Sanda, M.G.; Wei, J.T.; Klee, G.G.; Bangma, C.H.; Slawin, K.M.; Marks, L.S.; Loeb, S.; Broyles, D.L.; et al. A multicenter study of [-2]pro-prostate specific antigen combined with prostate specific antigen and free prostate specific antigen for prostate cancer detection in the 2.0 to 10.0 ng/mL prostate specific antigen range. J. Urol. 2011, 185, 1650–1655. [Google Scholar] [CrossRef] [Green Version]
- Loeb, S.; Sanda, M.G.; Broyles, D.L.; Shin, S.S.; Bangma, C.H.; Wei, J.T.; Partin, A.W.; Klee, G.G.; Slawin, K.M.; Marks, L.S.; et al. The prostate health index selectively identifies clinically significant prostate cancer. J. Urol. 2015, 193, 1163–1169. [Google Scholar] [CrossRef] [Green Version]
- Ferro, M.; Bruzzese, D.; Perdonà, S.; Marino, A.; Mazzarella, C.; Perruolo, G.; D’Esposito, V.; Cosimato, V.; Buonerba, C.; Di Lorenzo, G.; et al. Prostate Health Index (Phi) and Prostate Cancer Antigen 3 (PCA3) significantly improve prostate cancer detection at initial biopsy in a total PSA range of 2–10 ng/mL. PLoS ONE 2013, 8, e67687. [Google Scholar] [CrossRef] [PubMed]
- Braun, K.; Sjoberg, D.D.; Vickers, A.J.; Lilja, H.; Bjartell, A.S. A Four-kallikrein Panel Predicts High-grade Cancer on Biopsy: Independent Validation in a Community Cohort. Eur. Urol. 2016, 69, 505–511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nordström, T.; Vickers, A.; Assel, M.; Lilja, H.; Grönberg, H.; Eklund, M. Comparison Between the Four-kallikrein Panel and Prostate Health Index for Predicting Prostate Cancer. Eur. Urol. 2015, 68, 139–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vickers, A.J.; Cronin, A.M.; Aus, G.; Pihl, C.; Becker, C.; Pettersson, K.; Scardino, P.T.; Hugosson, J.; Lilja, H. A panel of kallikrein markers can reduce unnecessary biopsy for prostate cancer: Data from the European Randomized Study of Prostate Cancer Screening in Göteborg, Sweden. BMC Med. 2008, 6, 19. [Google Scholar] [CrossRef] [Green Version]
- Leyten, G.H.; Hessels, D.; Jannink, S.A.; Smit, F.P.; de Jong, H.; Cornel, E.B.; de Reijke, T.M.; Vergunst, H.; Kil, P.; Knipscheer, B.C.; et al. Prospective multicentre evaluation of PCA3 and TMPRSS2-ERG gene fusions as diagnostic and prognostic urinary biomarkers for prostate cancer. Eur. Urol. 2014, 65, 534–542. [Google Scholar] [CrossRef]
- Ochiai, A.; Okihara, K.; Kamoi, K.; Oikawa, T.; Shimazui, T.; Murayama, S.; Tomita, K.; Umekawa, T.; Uemura, H.; Miki, T. Clinical utility of the prostate cancer gene 3 (PCA3) urine assay in Japanese men undergoing prostate biopsy. BJU Int. 2013, 111, 928–933. [Google Scholar] [CrossRef]
- Busetto, G.M.; Del Giudice, F.; Maggi, M.; De Marco, F.; Porreca, A.; Sperduti, I.; Magliocca, F.M.; Salciccia, S.; Chung, B.I.; De Berardinis, E.; et al. Prospective assessment of two-gene urinary test with multiparametric magnetic resonance imaging of the prostate for men undergoing primary prostate biopsy. World J. Urol. 2020. [Google Scholar] [CrossRef]
- De la Calle, C.M.; Fasulo, V.; Cowan, J.E.; Lonergan, P.E.; Maggi, M.; Gadzinski, A.J.; Yeung, R.A.; Saita, A.; Cooperberg, M.R.; Shinohara, K.; et al. Clinical Utility of 4Kscore®, ExosomeDx™ and Magnetic Resonance Imaging for the Early Detection of High Grade Prostate Cancer. J. Urol. 2021, 205, 452–460. [Google Scholar] [CrossRef]
- Leapman, M.S.; Nguyen, H.G.; Cooperberg, M.R. Clinical Utility of Biomarkers in Localized Prostate Cancer. Curr. Oncol. Rep. 2016, 18, 30. [Google Scholar] [CrossRef] [Green Version]
- Klein, E.A.; Cooperberg, M.R.; Magi-Galluzzi, C.; Simko, J.P.; Falzarano, S.M.; Maddala, T.; Chan, J.M.; Li, J.; Cowan, J.E.; Tsiatis, A.C.; et al. A 17-gene assay to predict prostate cancer aggressiveness in the context of Gleason grade heterogeneity, tumor multifocality, and biopsy under sampling. Eur. Urol. 2014, 66, 550–560. [Google Scholar] [CrossRef] [Green Version]
- Tosoian, J.J.; Chappidi, M.R.; Bishoff, J.T.; Freedland, S.J.; Reid, J.; Brawer, M.; Stone, S.; Schlomm, T.; Ross, A.E. Prognostic utility of biopsy-derived cell cycle progression score in patients with National Comprehensive Cancer Network low-risk prostate cancer undergoing radical prostatectomy: Implications for treatment guidance. BJU Int. 2017, 120, 808–814. [Google Scholar] [CrossRef]
- Roach, M., 3rd; Hanks, G.; Thames, H., Jr.; Schellhammer, P.; Shipley, W.U.; Sokol, G.H.; Sandler, H. Defining biochemical failure following radiotherapy with or without hormonal therapy in men with clinically localized prostate cancer: Recommendations of the RTOG-ASTRO Phoenix Consensus Conference. Int. J. Radiat. Oncol. Biol. Phys. 2006, 65, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Cooperberg, M.R.; Simko, J.P.; Cowan, J.E.; Reid, J.E.; Djalilvand, A.; Bhatnagar, S.; Gutin, A.; Lanchbury, J.S.; Swanson, G.P.; Stone, S.; et al. Validation of a cell-cycle progression gene panel to improve risk stratification in a contemporary prostatectomy cohort. J. Clin. Oncol. 2013, 31, 1428–1434. [Google Scholar] [CrossRef] [PubMed]
- Erho, N.; Crisan, A.; Vergara, I.A.; Mitra, A.P.; Ghadessi, M.; Buerki, C.; Bergstralh, E.J.; Kollmeyer, T.; Fink, S.; Haddad, Z.; et al. Discovery and validation of a prostate cancer genomic classifier that predicts early metastasis following radical prostatectomy. PLoS ONE 2013, 8, e66855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, S.G.; Chang, S.L.; Spratt, D.E.; Erho, N.; Yu, M.; Ashab, H.A.; Alshalalfa, M.; Speers, C.; Tomlins, S.A.; Davicioni, E.; et al. Development and validation of a 24-gene predictor of response to postoperative radiotherapy in prostate cancer: A matched, retrospective analysis. Lancet Oncol. 2016, 17, 1612–1620. [Google Scholar] [CrossRef]
- Scattoni, V.; Lazzeri, M.; Lughezzani, G.; De Luca, S.; Passera, R.; Bollito, E.; Randone, D.; Abdollah, F.; Capitanio, U.; Larcher, A.; et al. Head-to-head comparison of prostate health index and urinary PCA3 for predicting cancer at initial or repeat biopsy. J. Urol. 2013, 190, 496–501. [Google Scholar] [CrossRef]
- Boegemann, M.; Stephan, C.; Cammann, H.; Vincendeau, S.; Houlgatte, A.; Jung, K.; Blanchet, J.; Semjonow, A. The percentage of prostate-specific antigen (PSA) isoform [-2]proPSA and the Prostate Health Index improve the diagnostic accuracy for clinically relevant prostate cancer at initial and repeat biopsy compared with total PSA and percentage free PSA in men aged ≤65 years. BJU Int. 2016, 117, 72–79. [Google Scholar]
- Stephan, C.; Jung, K.; Semjonow, A.; Schulze-Forster, K.; Cammann, H.; Hu, X.; Meyer, H.; Bögemann, M.; Miller, K.; Friedersdorff, F. Comparative assessment of urinary prostate cancer antigen 3 and TMPRSS2:ERG gene fusion with the serum [-2]proprostate-specific antigen-based prostate health index for detection of prostate cancer. Clin. Chem. 2013, 59, 280–288. [Google Scholar] [CrossRef] [Green Version]
- Gupta, A.; Roobol, M.J.; Savage, C.J.; Peltola, M.; Pettersson, K.; Scardino, P.T.; Vickers, A.J.; Schröder, F.H.; Lilja, H. A four-kallikrein panel for the prediction of repeat prostate biopsy: Data from the European Randomized Study of Prostate Cancer screening in Rotterdam, Netherlands. Br. J. Cancer 2010, 103, 708–714. [Google Scholar] [CrossRef] [Green Version]
- Auprich, M.; Augustin, H.; Budäus, L.; Kluth, L.; Mannweiler, S.; Shariat, S.F.; Fisch, M.; Graefen, M.; Pummer, K.; Chun, F.K.H. A comparative performance analysis of total prostate-specific antigen, percentage free prostate-specific antigen, prostate-specific antigen velocity and urinary prostate cancer gene 3 in the first, second and third repeat prostate biopsy. BJU Int. 2012, 109, 1627–1635. [Google Scholar] [CrossRef]
- Haese, A.; de la Taille, A.; van Poppel, H.; Marberger, M.; Stenzl, A.; Mulders, P.F.A.; Huland, H.; Abbou, A.; Remzi, M.; Tinzl, M.; et al. Clinical utility of the PCA3 urine assay in European men scheduled for repeat biopsy. Eur. Urol. 2008, 54, 1081–1088. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, A.K.; Reese, A.C.; Cooperberg, M.R.; Sadetsky, N.; Shinohara, K. Utility of PCA3 in patients undergoing repeat biopsy for prostate cancer. Prostate Cancer Prostatic Dis. 2012, 15, 100–105. [Google Scholar] [CrossRef] [Green Version]
- Stewart, G.D.; Van Neste, L.; Delvenne, P.; Delrée, P.; Delga, A.; McNeill, S.A.; O’Donnell, M.; Clark, J.; Van Criekinge, W.; Bigley, J.; et al. Clinical utility of an epigenetic assay to detect occult prostate cancer in histopathologically negative biopsies: Results of the MATLOC study. J. Urol. 2013, 189, 1110–1116. [Google Scholar] [CrossRef] [PubMed]
- Uhr, A.; Glick, L.; Gomella, L.G. An overview of biomarkers in the diagnosis and management of prostate cancer. Can. J. Urol. 2020, 27, 24–27. [Google Scholar]
- Weinreb, J.C.; Barentsz, J.O.; Choyke, P.L.; Cornud, F.; Haider, M.A.; Macura, K.J.; Margolis, D.; Schnall, M.D.; Shtern, F.; Tempany, C.M.; et al. PI-RADS Prostate Imaging—Reporting and Data System: 2015, Version 2. Eur. Urol. 2016, 69, 16–40. [Google Scholar] [CrossRef]
- Groskopf, J.; Aubin, S.M.; Deras, I.L.; Blase, A.; Bodrug, S.; Clark, C.; Brentano, S.; Mathis, J.; Pham, J.; Meyer, T.; et al. APTIMA PCA3 molecular urine test: Development of a method to aid in the diagnosis of prostate cancer. Clin. Chem. 2006, 52, 1089–1095. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hendriks, R.J.; van der Leest, M.M.G.; Dijkstra, S.; Barentsz, J.O.; Criekinge, W.V.; Hulsbergen-van de Kaa, C.A.; Schalken, J.A.; Mulders, P.F.A.; van Oort, I.M. A urinary biomarker-based risk score correlates with multiparametric MRI for prostate cancer detection. Prostate 2017, 77, 1401–1407. [Google Scholar] [CrossRef]
- Van Neste, L.; Hendriks, R.J.; Dijkstra, S.; Trooskens, G.; Cornel, E.B.; Jannink, S.A.; de Jong, H.; Hessels, D.; Smit, F.P.; Melchers, W.J.; et al. Detection of High-grade Prostate Cancer Using a Urinary Molecular Biomarker-Based Risk Score. Eur. Urol. 2016, 70, 740–748. [Google Scholar] [CrossRef]
- Epstein, J.I.; Egevad, L.; Amin, M.B.; Delahunt, B.; Srigley, J.R.; Humphrey, P.A. The 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma: Definition of Grading Patterns and Proposal for a New Grading System. Am. J. Surg. Pathol. 2016, 40, 244–252. [Google Scholar] [CrossRef]
- Carlsson, S.V.; Roobol, M.J. Improving the evaluation and diagnosis of clinically significant prostate cancer in 2017. Curr. Opin. Urol. 2017, 27, 198–204. [Google Scholar] [CrossRef] [Green Version]
- Sciarra, A.; Gentilucci, A.; Silvestri, I.; Salciccia, S.; Cattarino, S.; Scarpa, S.; Gatto, A.; Frantellizzi, V.; Von Heland, M.; Ricciuti, G.P.; et al. Androgen receptor variant 7 (AR-V7) in sequencing therapeutic agents for castratrion resistant prostate cancer: A critical review. Medicine 2019, 98, e15608. [Google Scholar] [CrossRef] [PubMed]
- Busetto, G.M.; Giovannone, R.; Antonini, G.; Rossi, A.; Del Giudice, F.; Tricarico, S.; Ragonesi, G.; Gentile, V.; De Berardinis, E. Short-term pretreatment with a dual 5α-reductase inhibitor before bipolar transurethral resection of the prostate (B-TURP): Evaluation of prostate vascularity and decreased surgical blood loss in large prostates. BJU Int. 2015, 116, 117–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flammia, S.; Frisenda, M.; Maggi, M.; Magliocca, F.M.; Ciardi, A.; Panebianco, V.; De Berardinis, E.; Salciccia, S.; Di Pierro, G.B.; Gentilucci, A.; et al. Cribriform pattern does not have a significant impact in Gleason Score ≥7/ISUP Grade ≥2 prostate cancers submitted to radical prostatectomy. Medicine 2020, 99, e22156. [Google Scholar] [CrossRef] [PubMed]
- Ferro, M.; Musi, G.; Matei, D.V.; Mistretta, A.F.; Luzzago, S.; Cozzi, G.; Bianchi, R.; Di Trapani, E.; Cioffi, A.; Lucarelli, G.; et al. Assessment of PSIM (Prostatic Systemic Inflammatory Markers) Score in Predicting Pathologic Features at Robotic Radical Prostatectomy in Patients with Low-Risk Prostate Cancer Who Met the Inclusion Criteria for Active Surveillance. Diagnostics 2021, 11, 355. [Google Scholar] [CrossRef] [PubMed]
- Ferro, M.; Lucarelli, G.; de Cobelli, O.; Del Giudice, F.; Musi, G.; Mistretta, F.A.; Luzzago, S.; Busetto, G.M.; Buonerba, C.; Sciarra, A.; et al. The emerging landscape of tumor marker panels for the identification of aggressive prostate cancer: The perspective through bibliometric analysis of an Italian translational working group in uro-oncology. Minerva Urol. Nephrol. 2021. [Google Scholar] [CrossRef]
- Sciarra, A.; Maggi, M.; Del Proposto, A.; Magliocca, F.M.; Ciardi, A.; Panebianco, V.; De Berardinis, E.; Salciccia, S.; Di Pierro, G.B.; Gentilucci, A.; et al. Impact of uni- or multifocal perineural invasion in prostate cancer at radical prostatectomy. Transl. Androl. Urol. 2021, 10, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Cerrato, A.; Bedia, C.; Capriotti, A.L.; Cavaliere, C.; Gentile, V.; Maggi, M.; Montone, C.M.; Piovesana, S.; Sciarra, A.; Tauler, R.; et al. Untargeted metabolomics of prostate cancer zwitterionic and positively charged compounds in urine. Anal. Chim. Acta 2021, 1158, 338381. [Google Scholar] [CrossRef]
- Busetto, G.M.; Giovannone, R.; Ferro, M.; Tricarico, S.; Del Giudice, F.; Matei, D.V.; De Cobelli, O.; Gentile, V.; De Berardinis, E. Chronic bacterial prostatitis: Efficacy of short-lasting antibiotic therapy with prulifloxacin (Unidrox®) in association with saw palmetto extract, lactobacillus sporogens and arbutin (Lactorepens®). BMC Urol. 2014, 14, 53. [Google Scholar] [CrossRef] [Green Version]
- Porreca, A.; D’Agostino, D.; Romagnoli, D.; Del Giudice, F.; Maggi, M.; Palmer, K.; Falabella, R.; De Berardinis, E.; Sciarra, A.; Ferro, M.; et al. The Clinical Efficacy of Nitrofurantoin for Treating Uncomplicated Urinary Tract Infection in Adults: A Systematic Review of Randomized Control Trials. Urol. Int. 2021, 1–10. [Google Scholar] [CrossRef]
- Leyten, G.H.; Hessels, D.; Smit, F.P.; Jannink, S.A.; de Jong, H.; Melchers, W.J.; Cornel, E.B.; de Reijke, T.M.; Vergunst, H.; Kil, P.; et al. Identification of a Candidate Gene Panel for the Early Diagnosis of Prostate Cancer. Clin. Cancer Res. 2015, 21, 3061–3070. [Google Scholar] [CrossRef] [Green Version]
- Haese, A.; Trooskens, G.; Steyaert, S.; Hessels, D.; Brawer, M.; Vlaeminck-Guillem, V.; Ruffion, A.; Tilki, D.; Schalken, J.; Groskopf, J.; et al. Multicenter Optimization and Validation of a 2-Gene mRNA Urine Test for Detection of Clinically Significant Prostate Cancer before Initial Prostate Biopsy. J. Urol. 2019, 202, 256–263. [Google Scholar] [CrossRef]
- Rubio-Briones, J.; Borque-Fernando, A.; Esteban, L.M.; Mascarós, J.M.; Ramírez-Backhaus, M.; Casanova, J.; Collado, A.; Mir, C.; Gómez-Ferrer, A.; Wong, A.; et al. Validation of a 2-gene mRNA urine test for the detection of ≥GG2 prostate cancer in an opportunistic screening population. Prostate 2020, 80, 500–507. [Google Scholar] [CrossRef] [PubMed]
- Maggi, M.; Panebianco, V.; Mosca, A.; Salciccia, S.; Gentilucci, A.; Di Pierro, G.; Busetto, G.M.; Barchetti, G.; Campa, R.; Sperduti, I.; et al. Prostate Imaging Reporting and Data System 3 Category Cases at Multiparametric Magnetic Resonance for Prostate Cancer: A Systematic Review and Meta-analysis. Eur. Urol. Focus 2020, 6, 463–478. [Google Scholar] [CrossRef]
- Dijkstra, S.; Govers, T.M.; Hendriks, R.J.; Schalken, J.A.; Van Criekinge, W.; Van Neste, L.; Grutters, J.P.C.; Sedelaar, J.P.M.; van Oort, I.M. Cost-effectiveness of a new urinary biomarker-based risk score compared to standard of care in prostate cancer diagnostics—A decision analytical model. BJU Int. 2017, 120, 659–665. [Google Scholar] [CrossRef]
- Govers, T.M.; Hessels, D.; Vlaeminck-Guillem, V.; Schmitz-Dräger, B.J.; Stief, C.G.; Martinez-Ballesteros, C.; Ferro, M.; Borque-Fernando, A.; Rubio-Briones, J.; Sedelaar, J.P.M.; et al. Cost-effectiveness of SelectMDx for prostate cancer in four European countries: A comparative modeling study. Prostate Cancer Prostatic Dis. 2019, 22, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Sathianathen, N.J.; Kuntz, K.M.; Alarid-Escudero, F.; Lawrentschuk, N.L.; Bolton, D.M.; Murphy, D.G.; Weight, C.J.; Konety, B.R. Incorporating Biomarkers into the Primary Prostate Biopsy Setting: A Cost-Effectiveness Analysis. J. Urol. 2018, 200, 1215–1220. [Google Scholar] [CrossRef] [PubMed]
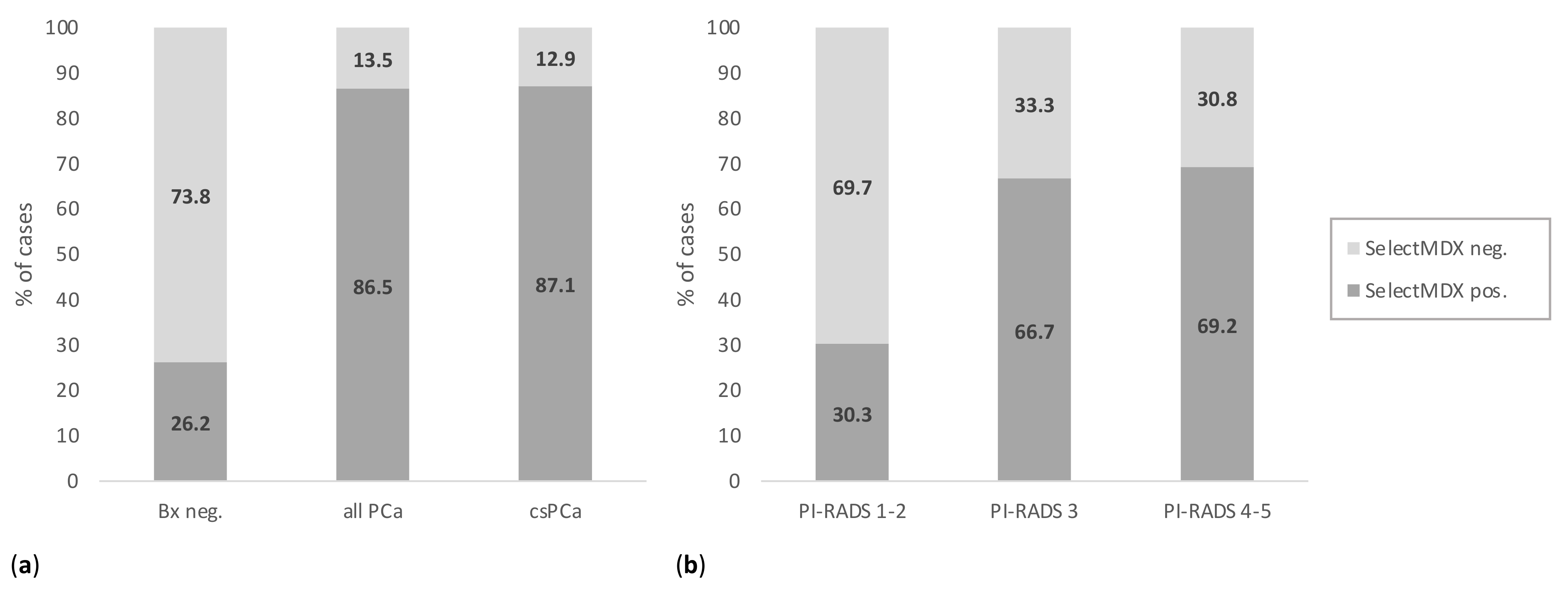



| Parameter | Negative PCa | All PCa | csPCa | p-Value * | p-Value ** |
|---|---|---|---|---|---|
| Number of cases n (%) | 206 (66.5) | 104 (33.5) | 62 (20.0) | ||
| Age (years) | |||||
| mean ± SD | 64 ± 7.7 | 64 ± 8.2 | 63 ± 9.0 | 0.691 | 0.518 |
| median | 65 | 65 | 64 | ||
| range | 44–79 | 45–79 | 45–79 | ||
| Total PSA (ng/mL) | |||||
| Mean ± SD | 6.9 ± 4.1 | 9.0 ± 4.3 | 9.5 ± 5.0 | <0.0001 | 0.0001 |
| median | 6.1 | 8 | 8.1 | ||
| range | 1.0–19.5 | 1.0–19.9 | 1.2–19.9 | ||
| PSAD (ng/mL/mL) | |||||
| Mean ± SD | 0.13 ± 0.08 | 0.18 ± 0.11 | 0.18 ± 0.12 | <0.0001 | 0.0001 |
| median | 0.1 | 0.15 | 0.16 | ||
| range | 0.02–0.36 | 0.02–0.53 | 0.02–0.53 | ||
| SelectMDx score, n (%) | |||||
| negative | 152 (73.8) | 14 (13.5) | 8 (12.9) | <0.0001 | <0.0001 |
| positive | 54 (26.2) | 90 (86.5) | 54 (87.1) | ||
| probability for csPCa (%) | |||||
| mean ± SD | 6.9 ± 13.9 | 25.9 ± 18.2 | 27.7 ± 18.1 | <0.0001 | <0.0001 |
| median (range) | 0(0–62) | 24.0(0–81) | 25.0(0–81) | ||
| mpMRI PI-RADS score, n (%) | |||||
| PI-RADS 1–2 | 160 (77.7) | 18 (17.3) | 10 (16.1) | <0.0001 | <0.0001 |
| PI-RADS 3 | 22 (10.7) | 32 (30.8) | 14 (22.6) | ||
| PI-RADS 4–5 | 24 (11.6) | 54 (51.9) | 38 (61.3) | ||
| SelectMDx score and | |||||
| mpMRI PI-RADS score, n (%) | |||||
| SelectMDx positive, mpMRI positive | 6 (2.9) | 48 (46.2) | 34 (54.8) | 0.001 | <0.0001 |
| SelectMDx negative, mpMRI negative | 134 (65.0) | 8 (7.7) | 4 (6.5) | ||
| SelectMDx positive, mpMRI negative | 48 (23.4) | 42 (40.4) | 20 (32.2) | 0.008 | <0.0001 |
| SelectMDx negative, mpMRI positive | 18 (8.7) | 6 (5.7) | 4 (6.5) |
| Parameter | All PCa | csPCa | ||||||
|---|---|---|---|---|---|---|---|---|
| Sensitivity | Specificity | NPV | PPV | Sensitivity | Specificity | NPV | PPV | |
| SelectMDx | 86.5 (78.5–91.9) | 73.8 (64.7–79.3) | 91.6 | 62.5 | 87.1 (76.2–93.5) | 63.7 (57.5–69.4) | 95.2 | 37.5 |
| mpMRI PI-RADS | 51.9 (42.4–61.3) | 88.3 (83.2–92.1) | 78.4 | 69.2 | 61.3 (48.8–72.4) | 83.9 (78.7–87.9) | 89.7 | 48.7 |
| mpMRI PI-RADS and SelectMDx | 46.2 (36.9–55.7) | 97.1 (93.6–98.8) | 78.1 | 88.9 | 54.8 (42.5–66.6) | 91.9 (87.8–94.8) | 89.1 | 63.0 |
| mpMRI PI-RADS and total PSA | 50.0 (40.6–59.4) | 90.3 (85.4–93.7) | 78.2 | 72.2 | 58.1 (45.7–69.5) | 85.5 (80.5–89.3) | 89.1 | 50.0 |
| mpMRI PI-RADS and total PSAD | 38.5 (29.7–48.1) | 95.1 (91.1–97.4) | 75.4 | 80.0 | 45.2 (33.4–57.5) | 91.1 (86.9–94.1) | 86.9 | 56.0 |
| Strategy | Avoided Biopsies, n (%) | PCa, n (%) | csPCa, n (%) | ||
|---|---|---|---|---|---|
| n= 310 | n = 104 | n = 62 | |||
| Detected | Missed | Detected | Missed | ||
| strategy 1 | 166 (53.5) | 90 (86.5) | 14 (13.5) | 54 (87.1) | 8 (12.9) |
| strategy 2 | 178 (57.4) | 86 (82.7) | 18 (17.3) | 52 (83.9) | 10 (16.1) |
| strategy 3 | 232 (74.8) | 54 (51.9) | 50 (48.1) | 38 (61.3) | 24 (38.7) |
| strategy 4 | 220 (71.0) | 74 (71.1) | 30 (28.9) | 46 (74.2) | 16 (25.8) |
| strategy 5 | 256 (82.6) | 48 (46.1) | 116 (53.9) | 34 (54.8) | 28 (45.2) |
| strategy 6 | 124 (40.0) | 102 (98.1) | 2 (1.9) | 60 (96.8) | 2 (3.2) |
| strategy 7 | 142 (45.8) | 96 (92.3) | 8 (7.7) | 58 (93.5) | 6 (6.5) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maggi, M.; Del Giudice, F.; Falagario, U.G.; Cocci, A.; Russo, G.I.; Di Mauro, M.; Sepe, G.S.; Galasso, F.; Leonardi, R.; Iacona, G.; et al. SelectMDx and Multiparametric Magnetic Resonance Imaging of the Prostate for Men Undergoing Primary Prostate Biopsy: A Prospective Assessment in a Multi-Institutional Study. Cancers 2021, 13, 2047. https://doi.org/10.3390/cancers13092047
Maggi M, Del Giudice F, Falagario UG, Cocci A, Russo GI, Di Mauro M, Sepe GS, Galasso F, Leonardi R, Iacona G, et al. SelectMDx and Multiparametric Magnetic Resonance Imaging of the Prostate for Men Undergoing Primary Prostate Biopsy: A Prospective Assessment in a Multi-Institutional Study. Cancers. 2021; 13(9):2047. https://doi.org/10.3390/cancers13092047
Chicago/Turabian StyleMaggi, Martina, Francesco Del Giudice, Ugo G. Falagario, Andrea Cocci, Giorgio Ivan Russo, Marina Di Mauro, Giuseppe Salvatore Sepe, Fabio Galasso, Rosario Leonardi, Gabriele Iacona, and et al. 2021. "SelectMDx and Multiparametric Magnetic Resonance Imaging of the Prostate for Men Undergoing Primary Prostate Biopsy: A Prospective Assessment in a Multi-Institutional Study" Cancers 13, no. 9: 2047. https://doi.org/10.3390/cancers13092047











