PGC-1β and ERRα Promote Glutamine Metabolism and Colorectal Cancer Survival via Transcriptional Upregulation of PCK2
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Lentiviral Transduction
2.3. Epitope-Tagging of Endogenous PGC-1β
2.4. Immunoprecipitation of PGC-1β
2.5. Inducible PGC-1β Vector and Cell Line Generation
2.6. siRNA Transfections
2.7. RNA Sequencing and Analysis
2.8. Western Blot Analyses
2.9. L-Glutamine Utilization Assays
2.10. Intracellular Metabolite Analysis
2.11. PCK2-ALFA Plasmid for Stable Expression
2.12. Confocal Microscopy
2.13. Statistical Analysis
3. Results
3.1. Endogenous PGC-1β Interacts with ERRα, Promotes ERRα Expression, and Anchorage-Independent Growth
3.2. PGC-1β Requires Its LRELL Motif at Amino Acids 343–347 to Interact with ERRα
3.3. PGC-1β and ERRα Promote PCK2 Expression
3.4. PCK2 Promotes Anchorage-Independent Growth and Glutamine Utilization
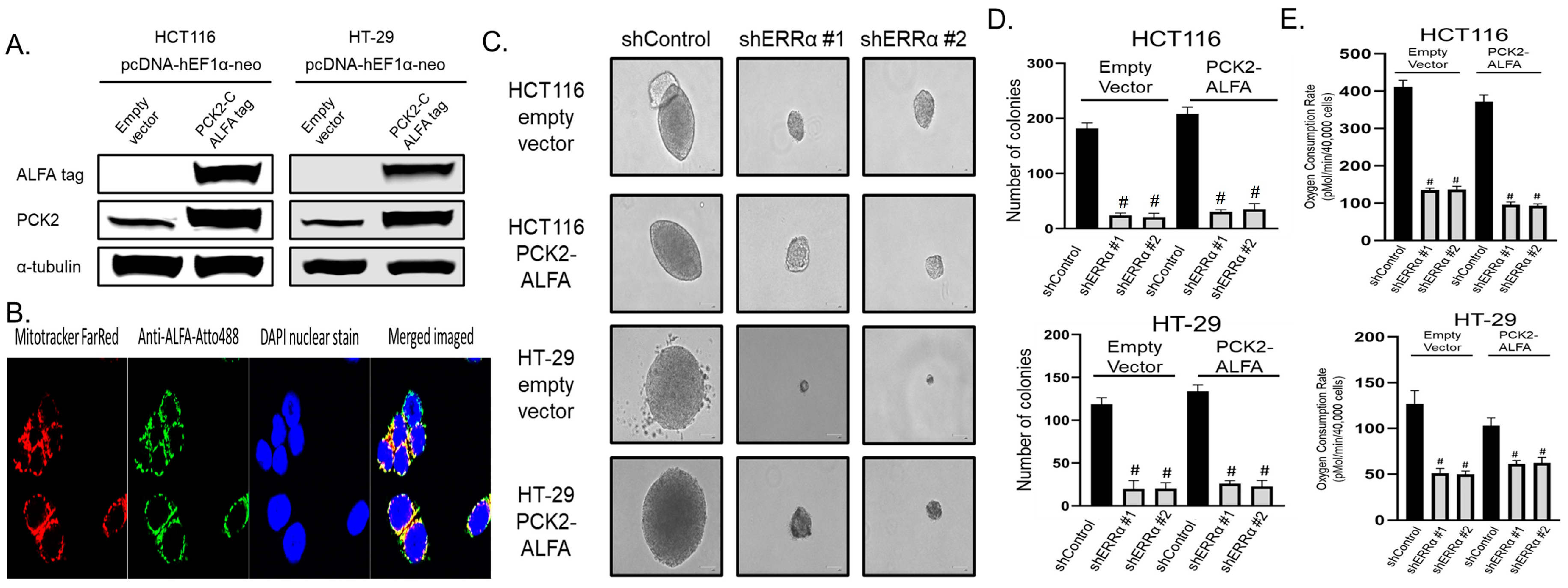
3.5. ERRα Depletion Causes Loss of Anchorage-Independent Growth and Glutamine Utilization That Is Not Rescued by Over-Expression of PCK2
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hock, M.B.; Kralli, A. Transcriptional Control of Mitochondrial Biogenesis and Function. Annu. Rev. Physiol. 2009, 71, 177–203. [Google Scholar] [CrossRef] [Green Version]
- Bellafante, E.; Morgano, A.; Salvatore, L.; Murzilli, S.; Di Tullio, G.; D’Orazio, A.; Latorre, D.; Villani, G.; Moschetta, A. PGC-1β Promotes Enterocyte Lifespan and Tumorigenesis in the Intestine. Proc. Natl. Acad. Sci. USA 2014, 111, E4523–E4531. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fisher, K.W.; Das, B.; Kim, H.S.; Clymer, B.K.; Gehring, D.; Smith, D.R.; Costanzo-Garvey, D.L.; Fernandez, M.R.; Brattain, M.G.; Kelly, D.L.; et al. AMPK Promotes Aberrant PGC1beta Expression To Support Human Colon Tumor Cell Survival. Mol. Cell Biol. 2015, 35, 3866–3879. [Google Scholar] [CrossRef] [Green Version]
- McCall, J.L.; Gehring, D.; Clymer, B.K.; Fisher, K.W.; Das, B.; Kelly, D.L.; Kim, H.; White, M.A.; Lewis, R.E. KSR1 and EPHB4 Regulate Myc and PGC1beta To Promote Survival of Human Colon Tumors. Mol. Cell Biol. 2016, 36, 2246–2261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berg, J.A.; Belyeu, J.R.; Morgan, J.T.; Ouyang, Y.; Bott, A.J.; Quinlan, A.R.; Gertz, J.; Rutter, J. XPRESSyourself: Enhancing, Standardizing, and Automating Ribosome Profiling Computational Analyses Yields Improved Insight into Data. PLOS Comput. Biol. 2020, 16, e1007625. [Google Scholar] [CrossRef] [Green Version]
- Love, M.I.; Huber, W.; Anders, S. Moderated Estimation of Fold Change and Dispersion for RNA-Seq Data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [Green Version]
- Trapnell, C.; Roberts, A.; Goff, L.; Pertea, G.; Kim, D.; Kelley, D.R.; Pimentel, H.; Salzberg, S.L.; Rinn, J.L.; Pachter, L. Differential Gene and Transcript Expression Analysis of RNA-Seq Experiments with TopHat and Cufflinks. Nat. Protoc. 2012, 7, 562–578. [Google Scholar] [CrossRef] [Green Version]
- Robinson, J.T.; Thorvaldsdóttir, H.; Winckler, W.; Guttman, M.; Lander, E.S.; Getz, G.; Mesirov, J.P. Integrative Genomics Viewer. Nat. Biotechnol. 2011, 29, 24–26. [Google Scholar] [CrossRef] [Green Version]
- Rha, G.B.; Wu, G.; Shoelson, S.E.; Chi, Y.-I. Multiple Binding Modes between HNF4alpha and the LXXLL Motifs of PGC-1alpha Lead to Full Activation. J. Biol. Chem. 2009, 284, 35165–35176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delerive, P.; Wu, Y.; Burris, T.P.; Chin, W.W.; Suen, C.S. PGC-1 Functions as a Transcriptional Coactivator for the Retinoid X Receptors. J. Biol. Chem. 2002, 277, 3913–3917. [Google Scholar] [CrossRef] [PubMed]
- Takacs, M.; Petoukhov, M.V.; Atkinson, R.A.; Roblin, P.; Ogi, F.-X.; Demeler, B.; Potier, N.; Chebaro, Y.; Dejaegere, A.; Svergun, D.I.; et al. The Asymmetric Binding of PGC-1α to the ERRα and ERRγ Nuclear Receptor Homodimers Involves a Similar Recognition Mechanism. PLoS ONE 2013, 8, e67810. [Google Scholar] [CrossRef]
- Greschik, H.; Althage, M.; Flaig, R.; Sato, Y.; Chavant, V.; Peluso-Iltis, C.; Choulier, L.; Cronet, P.; Rochel, N.; Schüle, R.; et al. Communication between the ERRalpha Homodimer Interface and the PGC-1alpha Binding Surface via the Helix 8–9 Loop. J. Biol. Chem. 2008, 283, 20220–20230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kallen, J.; Schlaeppi, J.-M.; Bitsch, F.; Filipuzzi, I.; Schilb, A.; Riou, V.; Graham, A.; Strauss, A.; Geiser, M.; Fournier, B. Evidence for Ligand-Independent Transcriptional Activation of the Human Estrogen-Related Receptor α (ERRα): Crystal structure of errα ligand binding domain in complex with peroxisome proliferator-activated receptor coactivator-1α. J. Biol. Chem. 2004, 279, 49330–49337. [Google Scholar] [CrossRef] [Green Version]
- Chang, C.; Kazmin, D.; Jasper, J.S.; Kunder, R.; Zuercher, W.J.; McDonnell, D.P. The Metabolic Regulator ERRα, a Downstream Target of HER2/IGF-1R, as a Therapeutic Target in Breast Cancer. Cancer Cell 2011, 20, 500–510. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kressler, D.; Schreiber, S.N.; Knutti, D.; Kralli, A. The PGC-1-Related Protein PERC Is a Selective Coactivator of Estrogen Receptor Alpha. J. Biol. Chem. 2002, 277, 13918–13925. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, S.; Chang, C.; Safi, R.; Liu, X.; Baldi, R.; Jasper, J.S.; Anderson, G.R.; Liu, T.; Rathmell, J.C.; Dewhirst, M.W.; et al. ERRα Regulated Lactate Metabolism Contributes to Resistance to Targeted Therapies in Breast Cancer. Cell Rep. 2016, 15, 323–335. [Google Scholar] [CrossRef] [Green Version]
- Lin, J.; Tarr, P.T.; Yang, R.; Rhee, J.; Puigserver, P.; Newgard, C.B.; Spiegelman, B.M. PGC-1beta in the Regulation of Hepatic Glucose and Energy Metabolism. J. Biol. Chem. 2003, 278, 30843–30848. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deblois, G.; Hall, J.A.; Perry, M.-C.; Laganière, J.; Ghahremani, M.; Park, M.; Hallett, M.; Giguère, V. Genome-Wide Identification of Direct Target Genes Implicates Estrogen-Related Receptor Alpha as a Determinant of Breast Cancer Heterogeneity. Cancer Res. 2009, 69, 6149–6157. [Google Scholar] [CrossRef] [Green Version]
- Deblois, G.; Chahrour, G.; Perry, M.-C.; Sylvain-Drolet, G.; Muller, W.J.; Giguère, V. Transcriptional Control of the ERBB2 Amplicon by ERRalpha and PGC-1beta Promotes Mammary Gland Tumorigenesis. Cancer Res. 2010, 70, 10277–10287. [Google Scholar] [CrossRef] [Green Version]
- Laganière, J.; Tremblay, G.B.; Dufour, C.R.; Giroux, S.; Rousseau, F.; Giguère, V. A Polymorphic Autoregulatory Hormone Response Element in the Human Estrogen-Related Receptor Alpha (ERRalpha) Promoter Dictates Peroxisome Proliferator-Activated Receptor Gamma Coactivator-1alpha Control of ERRalpha Expression. J. Biol. Chem. 2004, 279, 18504–18510. [Google Scholar] [CrossRef]
- Tremblay, A.M.; Wilson, B.J.; Yang, X.-J.; Giguère, V. Phosphorylation-Dependent Sumoylation Regulates Estrogen-Related Receptor-Alpha and -Gamma Transcriptional Activity through a Synergy Control Motif. Mol. Endocrinol. 2008, 22, 570–584. [Google Scholar] [CrossRef] [PubMed]
- Chun, S.Y.; Johnson, C.; Washburn, J.G.; Cruz-Correa, M.R.; Dang, D.T.; Dang, L.H. Oncogenic KRAS Modulates Mitochondrial Metabolism in Human Colon Cancer Cells by Inducing HIF-1α and HIF-2α Target Genes. Mol. Cancer 2010, 9, 293. [Google Scholar] [CrossRef] [Green Version]
- Götzke, H.; Kilisch, M.; Martínez-Carranza, M.; Sograte-Idrissi, S.; Rajavel, A.; Schlichthaerle, T.; Engels, N.; Jungmann, R.; Stenmark, P.; Opazo, F.; et al. The ALFA-Tag Is a Highly Versatile Tool for Nanobody-Based Bioscience Applications. Nat. Commun. 2019, 10, 4403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lelliott, C.J.; Medina-Gomez, G.; Petrovic, N.; Kis, A.; Feldmann, H.M.; Bjursell, M.; Parker, N.; Curtis, K.; Campbell, M.; Hu, P.; et al. Ablation of PGC-1beta Results in Defective Mitochondrial Activity, Thermogenesis, Hepatic Function, and Cardiac Performance. PLoS Biol. 2006, 4, e369. [Google Scholar] [CrossRef] [PubMed]
- Victorino, V.J.; Barroso, W.A.; Assunção, A.K.M.; Cury, V.; Jeremias, I.C.; Petroni, R.; Chausse, B.; Ariga, S.K.; Herrera, A.C.S.A.; Panis, C.; et al. PGC-1β Regulates HER2-Overexpressing Breast Cancer Cells Proliferation by Metabolic and Redox Pathways. Tumour Biol. J. Int. Soc. Oncodev. Biol. Med. 2016, 37, 6035–6044. [Google Scholar] [CrossRef]
- Hernandez, C.; Molusky, M.; Li, Y.; Li, S.; Lin, J.D. Regulation of Hepatic ApoC3 Expression by PGC-1β Mediates Hypolipidemic Effect of Nicotinic Acid. Cell Metab. 2010, 12, 411–419. [Google Scholar] [CrossRef] [Green Version]
- Bensard, C.L.; Wisidagama, D.R.; Olson, K.A.; Berg, J.A.; Krah, N.M.; Schell, J.C.; Nowinski, S.M.; Fogarty, S.; Bott, A.J.; Wei, P.; et al. Regulation of Tumor Initiation by the Mitochondrial Pyruvate Carrier. Cell Metab. 2020, 31, 284–300. [Google Scholar] [CrossRef]
- Schell, J.C.; Olson, K.A.; Jiang, L.; Hawkins, A.J.; Van Vranken, J.G.; Xie, J.; Egnatchik, R.A.; Earl, E.G.; Deberardinis, R.J.; Rutter, J. A Role for the Mitochondrial Pyruvate Carrier as a Repressor of the Warburg Effect and Colon Cancer Cell Growth. Mol. Cell 2014, 56, 400–413. [Google Scholar] [CrossRef] [Green Version]
- Barry, J.B.; Laganière, J.; Giguère, V. A Single Nucleotide in an Estrogen-Related Receptor Alpha Site Can Dictate Mode of Binding and Peroxisome Proliferator-Activated Receptor Gamma Coactivator 1alpha Activation of Target Promoters. Mol. Endocrinol. 2006, 20, 302–310. [Google Scholar] [CrossRef] [Green Version]
- Lin, J.; Puigserver, P.; Donovan, J.; Tarr, P.; Spiegelman, B.M. Peroxisome Proliferator-Activated Receptor Gamma Coactivator 1beta (PGC-1beta ), a Novel PGC-1-Related Transcription Coactivator Associated with Host Cell Factor. J. Biol. Chem. 2002, 277, 1645–1648. [Google Scholar] [CrossRef]
- Sammarco, G.; Gallo, G.; Vescio, G.; Picciariello, A.; De Paola, G.; Trompetto, M.; Currò, G.; Ammendola, M. Mast Cells, MicroRNAs and Others: The Role of Translational Research on Colorectal Cancer in the Forthcoming Era of Precision Medicine. J. Clin. Med. 2020, 9, 2852. [Google Scholar] [CrossRef]
- Denbo, J.W.; Yamashita, S.; Passot, G.; Egger, M.; Chun, Y.S.; Kopetz, S.E.; Maru, D.; Brudvik, K.W.; Wei, S.H.; Conrad, C.; et al. RAS Mutation Is Associated with Decreased Survival in Patients Undergoing Repeat Hepatectomy for Colorectal Liver Metastases. J. Gastrointest. Surg. 2017, 21, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Shady, W.; Petre, E.N.; Vakiani, E.; Ziv, E.; Gonen, M.; Brown, K.T.; Kemeny, N.E.; Solomon, S.B.; Solit, D.B.; Sofocleous, C.T. Kras Mutation Is a Marker of Worse Oncologic Outcomes after Percutaneous Radiofrequency Ablation of Colorectal Liver Metastases. Oncotarget 2017, 8, 66117–66127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goffredo, P.; Utria, A.F.; Beck, A.C.; Chun, Y.S.; Howe, J.R.; Weigel, R.J.; Vauthey, J.-N.; Hassan, I. The Prognostic Impact of KRAS Mutation in Patients Having Curative Resection of Synchronous Colorectal Liver Metastases. J. Gastrointest. Surg. Off. J. Soc. Surg. Aliment. Tract 2019, 23, 1957–1963. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Wang, T.; Li, R.; Du, Y.; Lv, H.; Zhang, L.; Chen, H.; Shi, X.; Li, Q.; Shen, J. The Discovery of a Novel Series of Potential ERRα Inverse Agonists Based on P-Nitrobenzenesulfonamide Template for Triple-Negative Breast Cancer in Vivo. J. Enzym. Inhib. Med. Chem. 2022, 37, 125–134. [Google Scholar] [CrossRef]
- Lynch, C.; Zhao, J.; Sakamuru, S.; Zhang, L.; Huang, R.; Witt, K.L.; Merrick, B.A.; Teng, C.T.; Xia, M. Identification of Compounds That Inhibit Estrogen-Related Receptor Alpha Signaling Using High-Throughput Screening Assays. Molecules 2019, 24, 841. [Google Scholar] [CrossRef] [Green Version]
- Duellman, S.J.; Calaoagan, J.M.; Sato, B.G.; Fine, R.; Klebansky, B.; Chao, W.-R.; Hobbs, P.; Collins, N.; Sambucetti, L.; Laderoute, K.R. A Novel Steroidal Inhibitor of Estrogen-Related Receptor α (ERRα). Biochem. Pharmacol. 2010, 80, 819–826. [Google Scholar] [CrossRef] [Green Version]
- Du, Y.; Song, L.; Zhang, L.; Ling, H.; Zhang, Y.; Chen, H.; Qi, H.; Shi, X.; Li, Q. The Discovery of Novel, Potent ERR-Alpha Inverse Agonists for the Treatment of Triple Negative Breast Cancer. Eur. J. Med. Chem. 2017, 136, 457–467. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, P.; Chen, H.; Li, Q.; Chen, L.; Qi, H.; Shi, X.; Du, Y. Characterization of a Selective Inverse Agonist for Estrogen Related Receptor α as a Potential Agent for Breast Cancer. Eur. J. Pharmacol. 2016, 789, 439–448. [Google Scholar] [CrossRef]
- Willy, P.J.; Murray, I.R.; Qian, J.; Busch, B.B.; Stevens, W.C.; Martin, R.; Mohan, R.; Zhou, S.; Ordentlich, P.; Wei, P.; et al. Regulation of PPARγ Coactivator 1α (PGC-1α) Signaling by an Estrogen-Related Receptor α (ERRα) Ligand. Proc. Natl. Acad. Sci. USA 2004, 101, 8912–8917. [Google Scholar] [CrossRef]
- Patch, R.J.; Searle, L.L.; Kim, A.J.; De, D.; Zhu, X.; Askari, H.B.; O’Neill, J.C.; Abad, M.C.; Rentzeperis, D.; Liu, J.; et al. Identification of Diaryl Ether-Based Ligands for Estrogen-Related Receptor α as Potential Antidiabetic Agents. J. Med. Chem. 2011, 54, 788–808. [Google Scholar] [CrossRef] [PubMed]
- Brearley, M.C.; Daniel, Z.C.T.R.; Loughna, P.T.; Parr, T.; Brameld, J.M. The Phosphoenolpyruvate Carboxykinase (PEPCK) Inhibitor, 3-Mercaptopicolinic Acid (3-MPA), Induces Myogenic Differentiation in C2C12 Cells. Sci. Rep. 2020, 10, 22177. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, J.; Latorre, P.; Carrodeguas, J.A.; Velázquez-Campoy, A.; Sancho, J.; López-Buesa, P. Inhibition of Pig Phosphoenolpyruvate Carboxykinase Isoenzymes by 3-Mercaptopicolinic Acid and Novel Inhibitors. PLoS ONE 2016, 11, e0159002. [Google Scholar] [CrossRef] [Green Version]
- Balan, M.D.; Mcleod, M.J.; Lotosky, W.R.; Ghaly, M.; Holyoak, T. Inhibition and Allosteric Regulation of Monomeric Phosphoenolpyruvate Carboxykinase by 3-Mercaptopicolinic Acid. Biochemistry 2015, 54, 5878–5887. [Google Scholar] [CrossRef]
- Mcleod, M.J.; Krismanich, A.P.; Assoud, A.; Dmitrienko, G.I.; Holyoak, T. Characterization of 3-[(Carboxymethyl)Thio]Picolinic Acid: A Novel Inhibitor of Phosphoenolpyruvate Carboxykinase. Biochemistry 2019, 58, 3918–3926. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frodyma, D.E.; Troia, T.C.; Rao, C.; Svoboda, R.A.; Berg, J.A.; Shinde, D.D.; Thomas, V.C.; Lewis, R.E.; Fisher, K.W. PGC-1β and ERRα Promote Glutamine Metabolism and Colorectal Cancer Survival via Transcriptional Upregulation of PCK2. Cancers 2022, 14, 4879. https://doi.org/10.3390/cancers14194879
Frodyma DE, Troia TC, Rao C, Svoboda RA, Berg JA, Shinde DD, Thomas VC, Lewis RE, Fisher KW. PGC-1β and ERRα Promote Glutamine Metabolism and Colorectal Cancer Survival via Transcriptional Upregulation of PCK2. Cancers. 2022; 14(19):4879. https://doi.org/10.3390/cancers14194879
Chicago/Turabian StyleFrodyma, Danielle E., Thomas C. Troia, Chaitra Rao, Robert A. Svoboda, Jordan A. Berg, Dhananjay D. Shinde, Vinai C. Thomas, Robert E. Lewis, and Kurt W. Fisher. 2022. "PGC-1β and ERRα Promote Glutamine Metabolism and Colorectal Cancer Survival via Transcriptional Upregulation of PCK2" Cancers 14, no. 19: 4879. https://doi.org/10.3390/cancers14194879





