Prognostic Value of Metabolic, Volumetric and Textural Parameters of Baseline [18F]FDG PET/CT in Early Triple-Negative Breast Cancer
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Clinical and Histological Parameters
2.3. [18F]FDG PET/CT Examination
2.4. [18F]FDG PET/CT Imaging Analysis
- -
- SUV: calculated using the following equation:SUV = (tissue radioactivity [Bq]/tissue weight [g])/(injected activity [Bq]/body weight [g]).
- ○
- Max: highest value of a voxel in the VOI
- ○
- Mean: mean value of all voxels in the VOI
- ○
- Peak: mean value of all voxels in 1 cm3 centered around the highest voxel value
- -
- MTV: considered as the volume of all voxels included in the VOI
- -
- TLG: defined by the formula SUVmean × MTV
- -
- Homogeneity: measures the local homogeneity of a pixel pair—homogeneity is expected to be large if each pixel pair’s gray levels are similar.
- -
- Entropy: measures the randomness of a gray-level distribution—the entropy is expected to be high if the gray levels are distributed randomly throughout the tumor region.
- -
- Short-Run Emphasis (SRE): measures the distribution of short series—the value is expected to be large if the number of short series is high.
- -
- Long-Run Emphasis (LRE): measures the distribution of long series—the value is expected to be large if the number of long series is high.
- -
- Low-Gray-level Zone Emphasis (LGZE): measures the distribution of low-gray-level zones.
- -
- High-Gray-level Zone Emphasis (HGZE): measures the distribution of high-gray-level zones.
2.5. Statistical Analysis
3. Results
3.1. Patient Inclusion
3.2. Comparison of Raw and Harmonized Data
3.3. Patient Characteristics and PET/CT Baseline Parameters
3.4. PET/CT Features with Clinical and Histological Factors
3.5. Tumor Characteristics, Therapeutic Management and PET/CT Features with Prognostic Factors
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, caac.21660. [Google Scholar] [CrossRef]
- Morris, G.J.; Naidu, S.; Topham, A.K.; Guiles, F.; Xu, Y.; McCue, P.; Schwartz, G.F.; Park, P.K.; Rosenberg, A.L.; Brill, K.; et al. Differences in Breast Carcinoma Characteristics in Newly Diagnosed African-American and Caucasian Patients: A Single-Institution Compilation Compared with the National Cancer Institute’s Surveillance, Epidemiology, and End Results Database. Cancer 2007, 110, 876–884. [Google Scholar] [CrossRef] [PubMed]
- Hanrahan, E.O.; Gonzalez-Angulo, A.M.; Giordano, S.H.; Rouzier, R.; Broglio, K.R.; Hortobagyi, G.N.; Valero, V. Overall Survival and Cause-Specific Mortality of Patients With Stage T1a, BN0M0 Breast Carcinoma. J. Clin. Oncol. 2007, 25, 4952–4960. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haffty, B.G.; Yang, Q.; Reiss, M.; Kearney, T.; Higgins, S.A.; Weidhaas, J.; Harris, L.; Hait, W.; Toppmeyer, D. Locoregional Relapse and Distant Metastasis in Conservatively Managed Triple Negative Early-Stage Breast Cancer. J. Clin. Oncol. 2006, 24, 5652–5657. [Google Scholar] [CrossRef] [PubMed]
- Cortazar, P.; Zhang, L.; Untch, M.; Mehta, K.; Costantino, J.P.; Wolmark, N.; Bonnefoi, H.; Cameron, D.; Gianni, L.; Valagussa, P.; et al. Pathological Complete Response and Long-Term Clinical Benefit in Breast Cancer: The CTNeoBC Pooled Analysis. Lancet 2014, 384, 164–172. [Google Scholar] [CrossRef] [Green Version]
- Dent, R.; Trudeau, M.; Pritchard, K.I.; Hanna, W.M.; Kahn, H.K.; Sawka, C.A.; Lickley, L.A.; Rawlinson, E.; Sun, P.; Narod, S.A. Triple-Negative Breast Cancer: Clinical Features and Patterns of Recurrence. Clin. Cancer Res. 2007, 13, 4429–4434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liedtke, C.; Mazouni, C.; Hess, K.R.; André, F.; Tordai, A.; Mejia, J.A.; Symmans, W.F.; Gonzalez-Angulo, A.M.; Hennessy, B.; Green, M.; et al. Response to Neoadjuvant Therapy and Long-Term Survival in Patients with Triple-Negative Breast Cancer. J. Clin. Oncol. 2008, 26, 1275–1281. [Google Scholar] [CrossRef]
- Carey, L.A.; Dees, E.C.; Sawyer, L.; Gatti, L.; Moore, D.T.; Collichio, F.; Ollila, D.W.; Sartor, C.I.; Graham, M.L.; Perou, C.M. The Triple Negative Paradox: Primary Tumor Chemosensitivity of Breast Cancer Subtypes. Clin. Cancer Res. 2007, 13, 2329–2334. [Google Scholar] [CrossRef] [Green Version]
- Prat, A.; Perou, C.M. Deconstructing the Molecular Portraits of Breast Cancer. Mol. Oncol. 2011, 5, 5–23. [Google Scholar] [CrossRef]
- Lehmann, B.D.; Bauer, J.A.; Chen, X.; Sanders, M.E.; Chakravarthy, A.B.; Shyr, Y.; Pietenpol, J.A. Identification of Human Triple-Negative Breast Cancer Subtypes and Preclinical Models for Selection of Targeted Therapies. J. Clin. Investig. 2011, 121, 2750–2767. [Google Scholar] [CrossRef] [Green Version]
- Lehmann, B.D.; Jovanović, B.; Chen, X.; Estrada, M.V.; Johnson, K.N.; Shyr, Y.; Moses, H.L.; Sanders, M.E.; Pietenpol, J.A. Refinement of Triple-Negative Breast Cancer Molecular Subtypes: Implications for Neoadjuvant Chemotherapy Selection. PLoS ONE 2016, 11, e0157368. [Google Scholar] [CrossRef] [PubMed]
- Jézéquel, P.; Kerdraon, O.; Hondermarck, H.; Guérin-Charbonnel, C.; Lasla, H.; Gouraud, W.; Canon, J.-L.; Gombos, A.; Dalenc, F.; Delaloge, S.; et al. Identification of Three Subtypes of Triple-Negative Breast Cancer with Potential Therapeutic Implications. Breast Cancer Res. 2019, 21, 65. [Google Scholar] [CrossRef] [PubMed]
- Jézéquel, P.; Loussouarn, D.; Guérin-Charbonnel, C.; Campion, L.; Vanier, A.; Gouraud, W.; Lasla, H.; Guette, C.; Valo, I.; Verrièle, V.; et al. Gene-Expression Molecular Subtyping of Triple-Negative Breast Cancer Tumours: Importance of Immune Response. Breast Cancer Res. 2015, 17, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jézéquel, P.; Guette, C.; Lasla, H.; Gouraud, W.; Boissard, A.; Guérin-Charbonnel, C.; Campone, M. ITRAQ-Based Quantitative Proteomic Analysis Strengthens Transcriptomic Subtyping of Triple-Negative Breast Cancer Tumors. Proteomics 2019, 19, 1800484. [Google Scholar] [CrossRef] [PubMed]
- Urru, S.A.M.; Gallus, S.; Bosetti, C.; Moi, T.; Medda, R.; Sollai, E.; Murgia, A.; Sanges, F.; Pira, G.; Manca, A.; et al. Clinical and Pathological Factors Influencing Survival in a Large Cohort of Triple-Negative Breast Cancer Patients. BMC Cancer 2018, 18, 56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goldhirsch, A.; Winer, E.P.; Coates, A.S.; Gelber, R.D.; Piccart-Gebhart, M.; Thürlimann, B.; Senn, H.-J.; Albain, K.S.; André, F.; Bergh, J.; et al. Personalizing the Treatment of Women with Early Breast Cancer: Highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann. Oncol. 2013, 24, 2206–2223. [Google Scholar] [CrossRef] [PubMed]
- Loi, S.; Michiels, S.; Salgado, R.; Sirtaine, N.; Jose, V.; Fumagalli, D.; Kellokumpu-Lehtinen, P.-L.; Bono, P.; Kataja, V.; Desmedt, C.; et al. Tumor Infiltrating Lymphocytes Are Prognostic in Triple Negative Breast Cancer and Predictive for Trastuzumab Benefit in Early Breast Cancer: Results from the FinHER Trial. Ann. Oncol. 2014, 25, 1544–1550. [Google Scholar] [CrossRef]
- Pruneri, G.; Gray, K.P.; Vingiani, A.; Viale, G.; Curigliano, G.; Criscitiello, C.; Láng, I.; Ruhstaller, T.; Gianni, L.; Goldhirsch, A.; et al. Tumor-Infiltrating Lymphocytes (TILs) Are a Powerful Prognostic Marker in Patients with Triple-Negative Breast Cancer Enrolled in the IBCSG Phase III Randomized Clinical Trial 22-00. Breast Cancer Res. Treat. 2016, 158, 323–331. [Google Scholar] [CrossRef]
- von Minckwitz, G.; Untch, M.; Blohmer, J.-U.; Costa, S.D.; Eidtmann, H.; Fasching, P.A.; Gerber, B.; Eiermann, W.; Hilfrich, J.; Huober, J.; et al. Definition and Impact of Pathologic Complete Response on Prognosis After Neoadjuvant Chemotherapy in Various Intrinsic Breast Cancer Subtypes. J. Clin. Oncol. 2012, 30, 1796–1804. [Google Scholar] [CrossRef] [Green Version]
- Masuda, N.; Lee, S.-J.; Ohtani, S.; Im, Y.-H.; Lee, E.-S.; Yokota, I.; Kuroi, K.; Im, S.-A.; Park, B.-W.; Kim, S.-B.; et al. Adjuvant Capecitabine for Breast Cancer after Preoperative Chemotherapy. N. Engl. J. Med. 2017, 376, 2147–2159. [Google Scholar] [CrossRef]
- Salaün, P.-Y.; Abgral, R.; Malard, O.; Querellou-Lefranc, S.; Quere, G.; Wartski, M.; Coriat, R.; Hindie, E.; Taieb, D.; Tabarin, A.; et al. Actualisation des recommandations de bonne pratique clinique pour l’utilisation de la TEP en cancérologie. Bull. Cancer 2019, 106, 262–274. [Google Scholar] [CrossRef] [PubMed]
- Ohara, M.; Shigematsu, H.; Tsutani, Y.; Emi, A.; Masumoto, N.; Ozaki, S.; Kadoya, T.; Okada, M. Role of FDG-PET/CT in Evaluating Surgical Outcomes of Operable Breast Cancer—Usefulness for Malignant Grade of Triple-Negative Breast Cancer. Breast 2013, 22, 958–963. [Google Scholar] [CrossRef] [PubMed]
- Kajáry, K.; Tőkés, T.; Dank, M.; Kulka, J.; Szakáll, S.; Lengyel, Z. Correlation of the Value of 18F-FDG Uptake, Described by SUVmax, SUVavg, Metabolic Tumour Volume and Total Lesion Glycolysis, to Clinicopathological Prognostic Factors and Biological Subtypes in Breast Cancer. Nucl. Med. Commun. 2015, 36, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Arslan, E.; Çermik, T.F.; Trabulus, F.D.C.; Talu, E.C.K.; Başaran, Ş. Role of 18F-FDG PET/CT in Evaluating Molecular Subtypes and Clinicopathological Features of Primary Breast Cancer. Nucl. Med. Commun. 2018, 39, 680–690. [Google Scholar] [CrossRef]
- Abubakar, Z.; Reddy Akepati, N.; Bikkina, P. Correlation of Maximum Standardized Uptake Values in 18F-Fluorodeoxyglucose Positron Emission Tomography-Computed Tomography Scan with Immunohistochemistry and Other Prognostic Factors in Breast Cancer. Indian J. Nucl. Med. 2019, 34, 10–13. [Google Scholar] [CrossRef]
- Ege Aktas, G.; Taştekin, E.; Sarikaya, A. Assessment of Biological and Clinical Aggressiveness of Invasive Ductal Breast Cancer Using Baseline 18F-FDG PET/CT-Derived Volumetric Parameters. Nucl. Med. Commun. 2018, 39, 83–93. [Google Scholar] [CrossRef]
- Koo, H.R.; Park, J.S.; Kang, K.W.; Cho, N.; Chang, J.M.; Bae, M.S.; Kim, W.H.; Lee, S.H.; Kim, M.Y.; Kim, J.Y.; et al. 18F-FDG Uptake in Breast Cancer Correlates with Immunohistochemically Defined Subtypes. Eur. Radiol. 2014, 24, 610–618. [Google Scholar] [CrossRef]
- Lee, J.; Lee, E.J.; Moon, S.H.; Kim, S.; Hyun, S.H.; Cho, Y.S.; Choi, J.Y.; Kim, B.-T.; Lee, K.-H. Strong Association of Epidermal Growth Factor Receptor Status with Breast Cancer FDG Uptake. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1438–1447. [Google Scholar] [CrossRef]
- Heudel, P.; Cimarelli, S.; Montella, A.; Bouteille, C.; Mognetti, T. Value of PET-FDG in Primary Breast Cancer Based on Histopathological and Immunohistochemical Prognostic Factors. Int. J. Clin. Oncol. 2010, 15, 588–593. [Google Scholar] [CrossRef]
- Tural, D.; Kivrak Salim, D.; Mutlu, H.; Erkilic, M.; Gunduz, S.; Karakurt, M.; Musri, F.; Tuna, S.; Boz, A.; Aydin, F.; et al. Is There Any Relation between PET-CT SUVmax Value and Prognostic Factors in Locally Advanced Breast Cancer? J. BUON 2015, 20, 1282–1286. [Google Scholar]
- Kitajima, K.; Fukushima, K.; Miyoshi, Y.; Nishimukai, A.; Hirota, S.; Igarashi, Y.; Katsuura, T.; Maruyama, K.; Hirota, S. Association between 18F-FDG Uptake and Molecular Subtype of Breast Cancer. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 1371–1377. [Google Scholar] [CrossRef] [PubMed]
- Koolen, B.B.; Vrancken Peeters, M.J.T.F.D.; Wesseling, J.; Lips, E.H.; Vogel, W.V.; Aukema, T.S.; van Werkhoven, E.; Gilhuijs, K.G.A.; Rodenhuis, S.; Rutgers, E.J.T.; et al. Association of Primary Tumour FDG Uptake with Clinical, Histopathological and Molecular Characteristics in Breast Cancer Patients Scheduled for Neoadjuvant Chemotherapy. Eur. J. Nucl. Med. Mol. Imaging 2012, 39, 1830–1838. [Google Scholar] [CrossRef] [PubMed]
- Sasada, S.; Masumoto, N.; Suzuki, E.; Sueoka, S.; Goda, N.; Kajitani, K.; Emi, A.; Kadoya, T.; Okada, M. Prediction of Biological Characteristics of Breast Cancer Using Dual-Phase FDG PET/CT. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 831–837. [Google Scholar] [CrossRef]
- Jena, A.; Taneja, S.; Singh, A.; Negi, P.; Sarin, R.; Das, P.K.; Singhal, M. Reliability of 18F-FDG PET Metabolic Parameters Derived Using Simultaneous PET/MRI and Correlation With Prognostic Factors of Invasive Ductal Carcinoma: A Feasibility Study. Am. J. Roentgenol. 2017, 209, 662–670. [Google Scholar] [CrossRef] [PubMed]
- Koo, H.R.; Park, J.S.; Kang, K.W.; Han, W.; Park, I.A.; Moon, W.K. Correlation between 18F-FDG Uptake on PET/CT and Prognostic Factors in Triple-Negative Breast Cancer. Eur. Radiol. 2015, 25, 3314–3321. [Google Scholar] [CrossRef] [PubMed]
- Pak, K.; Seok, J.W.; Kim, H.Y.; Nguyen, T.L.; Kim, K.; Kim, S.J.; Kim, I.-J.; Hopper, J. Prognostic Value of Metabolic Tumor Volume and Total Lesion Glycolysis in Breast Cancer: A Meta-Analysis. Nucl. Med. Commun. 2020, 41, 824–829. [Google Scholar] [CrossRef] [PubMed]
- Wen, W.; Xuan, D.; Hu, Y.; Li, X.; Liu, L.; Xu, D. Prognostic Value of Maximum Standard Uptake Value, Metabolic Tumor Volume, and Total Lesion Glycolysis of Positron Emission Tomography/Computed Tomography in Patients with Breast Cancer: A Systematic Review and Meta-Analysis. PLoS ONE 2019, 14, e0225959. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, Y.J.; Paeng, J.C.; Cheon, G.J.; Lee, D.S.; Chung, J.-K.; Kang, K.W. Prediction of Breast Cancer Recurrence Using Lymph Node Metabolic and Volumetric Parameters from 18F-FDG PET/CT in Operable Triple-Negative Breast Cancer. Eur. J. Nucl. Med. Mol. Imaging. 2017, 44, 1787–1795. [Google Scholar] [CrossRef]
- Lambin, P.; Rios-Velazquez, E.; Leijenaar, R.; Carvalho, S.; van Stiphout, R.G.P.M.; Granton, P.; Zegers, C.M.L.; Gillies, R.; Boellard, R.; Dekker, A.; et al. Radiomics: Extracting More Information from Medical Images Using Advanced Feature Analysis. Eur. J. Cancer 2012, 48, 441–446. [Google Scholar] [CrossRef] [Green Version]
- Hatt, M.; Tixier, F.; Visvikis, D.; Cheze Le Rest, C. Radiomics in PET/CT: More Than Meets the Eye? J. Nucl. Med. 2017, 58, 365–366. [Google Scholar] [CrossRef]
- Orlhac, F.; Soussan, M.; Maisonobe, J.-A.; Garcia, C.A.; Vanderlinden, B.; Buvat, I. Tumor Texture Analysis in 18F-FDG PET: Relationships Between Texture Parameters, Histogram Indices, Standardized Uptake Values, Metabolic Volumes, and Total Lesion Glycolysis. J. Nucl. Med. 2014, 55, 414–422. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soussan, M.; Orlhac, F.; Boubaya, M.; Zelek, L.; Ziol, M.; Eder, V.; Buvat, I. Relationship between Tumor Heterogeneity Measured on FDG-PET/CT and Pathological Prognostic Factors in Invasive Breast Cancer. PLoS ONE 2014, 9, e94017. [Google Scholar] [CrossRef] [PubMed]
- Groheux, D.; Martineau, A.; Teixeira, L.; Espié, M.; de Cremoux, P.; Bertheau, P.; Merlet, P.; Lemarignier, C. 18FDG-PET/CT for Predicting the Outcome in ER+/HER2- Breast Cancer Patients: Comparison of Clinicopathological Parameters and PET Image-Derived Indices Including Tumor Texture Analysis. Breast Cancer Res. 2017, 19, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moscoso, A.; Ruibal, Á.; Domínguez-Prado, I.; Fernández-Ferreiro, A.; Herranz, M.; Albaina, L.; Argibay, S.; Silva-Rodríguez, J.; Pardo-Montero, J.; Aguiar, P. Texture Analysis of High-Resolution Dedicated Breast 18 F-FDG PET Images Correlates with Immunohistochemical Factors and Subtype of Breast Cancer. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Zhang, J.; Wang, Y.; Xu, X.; Zhang, Y.; Zhang, Y.; Liu, G.; Cheng, J. Textural Features of 18F-FDG PET after Two Cycles of Neoadjuvant Chemotherapy Can Predict PCR in Patients with Locally Advanced Breast Cancer. Ann. Nucl. Med. 2017, 31, 544–552. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-C.; Chen, C.-J.; Hsu, W.-L.; Chang, S.-M.; Huang, Y.-F.; Tyan, Y.-C. Prognostic Significance of Metabolic Parameters and Textural Features on 18 F-FDG PET/CT in Invasive Ductal Carcinoma of Breast. Sci. Rep. 2019, 9, 10946. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.; Lee, D.-E.; Park, S.; Kim, T.S.; Jung, S.-Y.; Lee, S.; Kang, H.S.; Lee, E.S.; Sim, S.H.; Park, I.H.; et al. Predicting Response to Neoadjuvant Chemotherapy in Patients With Breast Cancer: Combined Statistical Modeling Using Clinicopathological Factors and FDG PET/CT Texture Parameters. Clin. Nucl. Med. 2019, 44, 21–29. [Google Scholar] [CrossRef]
- Groheux, D.; Majdoub, M.; Tixier, F.; Le Rest, C.C.; Martineau, A.; Merlet, P.; Espié, M.; de Roquancourt, A.; Hindié, E.; Hatt, M.; et al. Do Clinical, Histological or Immunohistochemical Primary Tumour Characteristics Translate into Different 18F-FDG PET/CT Volumetric and Heterogeneity Features in Stage II/III Breast Cancer? Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 1682–1691. [Google Scholar] [CrossRef] [Green Version]
- Molina-García, D.; García-Vicente, A.M.; Pérez-Beteta, J.; Amo-Salas, M.; Martínez-González, A.; Tello-Galán, M.J.; Soriano-Castrejón, Á.; Pérez-García, V.M. Intratumoral Heterogeneity in 18F-FDG PET/CT by Textural Analysis in Breast Cancer as a Predictive and Prognostic Subrogate. Ann. Nucl. Med. 2018, 32, 379–388. [Google Scholar] [CrossRef]
- Lee, J.W.; Lee, S.M. Radiomics in Oncological PET/CT: Clinical Applications. Nucl. Med. Mol. Imaging 2018, 52, 170–189. [Google Scholar] [CrossRef]
- Guezennec, C.; Robin, P.; Orlhac, F.; Bourhis, D.; Delcroix, O.; Gobel, Y.; Rousset, J.; Schick, U.; Salaün, P.-Y.; Abgral, R. Prognostic Value of Textural Indices Extracted from Pretherapeutic 18-F FDG-PET/CT in Head and Neck Squamous Cell Carcinoma. Head Neck 2019, 41, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.-C.; Chang, K.-P.; Fang, Y.-H.D.; Tsang, N.-M.; Ng, S.-H.; Hsu, C.-L.; Liao, C.-T.; Yen, T.-C. Tumor Heterogeneity Measured on F-18 Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography Combined with Plasma Epstein-Barr Virus Load Predicts Prognosis in Patients with Primary Nasopharyngeal Carcinoma: Heterogeneity on PET Predicts NPC Prognosis. Laryngoscope 2017, 127, E22–E28. [Google Scholar] [CrossRef] [PubMed]
- Folkert, M.R.; Setton, J.; Apte, A.P.; Grkovski, M.; Young, R.J.; Schöder, H.; Thorstad, W.L.; Lee, N.Y.; Deasy, J.O.; Oh, J.H. Predictive Modeling of Outcomes Following Definitive Chemoradiotherapy for Oropharyngeal Cancer Based on FDG-PET Image Characteristics. Phys. Med. Biol. 2017, 62, 5327–5343. [Google Scholar] [CrossRef] [PubMed]
- Cheng, N.-M.; Fang, Y.-H.D.; Lee, L.; Chang, J.T.-C.; Tsan, D.-L.; Ng, S.-H.; Wang, H.-M.; Liao, C.-T.; Yang, L.-Y.; Hsu, C.-H.; et al. Zone-Size Nonuniformity of 18F-FDG PET Regional Textural Features Predicts Survival in Patients with Oropharyngeal Cancer. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 419–428. [Google Scholar] [CrossRef]
- Fried, D.V.; Mawlawi, O.; Zhang, L.; Fave, X.; Zhou, S.; Ibbott, G.; Liao, Z.; Court, L.E. Stage III Non–Small Cell Lung Cancer: Prognostic Value of FDG PET Quantitative Imaging Features Combined with Clinical Prognostic Factors. Radiology 2016, 278, 214–222. [Google Scholar] [CrossRef] [Green Version]
- Lovinfosse, P.; Janvary, Z.L.; Coucke, P.; Jodogne, S.; Bernard, C.; Hatt, M.; Visvikis, D.; Jansen, N.; Duysinx, B.; Hustinx, R. FDG PET/CT Texture Analysis for Predicting the Outcome of Lung Cancer Treated by Stereotactic Body Radiation Therapy. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 1453–1460. [Google Scholar] [CrossRef]
- Yue, Y.; Cui, X.; Bose, S.; Audeh, W.; Zhang, X.; Fraass, B. Stratifying Triple-Negative Breast Cancer Prognosis Using 18F-FDG-PET/CT Imaging. Breast Cancer Res. Treat. 2015, 153, 607–616. [Google Scholar] [CrossRef] [Green Version]
- Johnson, W.E.; Li, C.; Rabinovic, A. Adjusting Batch Effects in Microarray Expression Data Using Empirical Bayes Methods. Biostatistics 2007, 8, 118–127. [Google Scholar] [CrossRef]
- Shabalin, A.A.; Tjelmeland, H.; Fan, C.; Perou, C.M.; Nobel, A.B. Merging Two Gene-Expression Studies via Cross-Platform Normalization. Bioinformatics 2008, 24, 1154–1160. [Google Scholar] [CrossRef] [Green Version]
- Smyth, G.K. Limma: Linear Models for Microarray Data. In Bioinformatics and Computational Biology Solutions Using R and Bioconductor; Gentleman, R., Carey, V.J., Huber, W., Irizarry, R.A., Dudoit, S., Eds.; Statistics for Biology and Health; Springer: New York, NY, USA, 2005; pp. 397–420. ISBN 978-0-387-25146-2. [Google Scholar]
- Reese, S.E.; Archer, K.J.; Therneau, T.M.; Atkinson, E.J.; Vachon, C.M.; de Andrade, M.; Kocher, J.-P.A.; Eckel-Passow, J.E. A New Statistic for Identifying Batch Effects in High-Throughput Genomic Data That Uses Guided Principal Component Analysis. Bioinformatics 2013, 29, 2877–2883. [Google Scholar] [CrossRef] [Green Version]
- Jiménez-Ballvé, A.; García García-Esquinas, M.; Salsidua-Arroyo, O.; Serrano-Palacio, A.; García-Sáenz, J.A.; Ortega Candil, A.; Fuentes Ferrer, M.E.; Rodríguez Rey, C.; Román-Santamaría, J.M.; Moreno, F.; et al. Papel pronóstico del volumen metabólico tumoral y de la glucólisis tumoral total en los estudios 18F-FDG PET/TC de estadificación del cáncer localmente avanzado de mama. Rev. Española Med. Nucl. Imagen Mol. 2016, 35, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, N.; Kataoka, M.; Sugawara, Y.; Ochi, T.; Kiyoto, S.; Ohsumi, S.; Mochizuki, T. Volume-Based Parameters of 18F-Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography Improve Disease Recurrence Prediction in Postmastectomy Breast Cancer Patients With 1 to 3 Positive Axillary Lymph Nodes. Int. J. Radiat. Oncol. Biol. Phys. 2013, 87, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Buvat, I. Les limites du SUV. Médecine Nucléaire 2007, 31, 165–172. [Google Scholar] [CrossRef]
- Nestle, U.; Kremp, S.; Schaefer-Schuler, A.; Sebastian-Welsch, C.; Hellwig, D.; Rübe, C.; Kirsch, C.-M. Comparison of Different Methods for Delineation of 18F-FDG PET-Positive Tissue for Target Volume Definition in Radiotherapy of Patients with Non-Small Cell Lung Cancer. J. Nucl. Med. 2005, 46, 1342–1348. [Google Scholar] [PubMed]
- Orlhac, F.; Theze, B.; Soussan, M.; Boisgard, R.; Buvat, I. Multiscale Texture Analysis: From 18F-FDG PET Images to Histologic Images. J. Nuc. Med. 2016, 57, 1823–1828. [Google Scholar] [CrossRef] [Green Version]
- Umutlu, L.; Kirchner, J.; Bruckmann, N.M.; Morawitz, J.; Antoch, G.; Ingenwerth, M.; Bittner, A.-K.; Hoffmann, O.; Haubold, J.; Grueneisen, J.; et al. Multiparametric Integrated 18F-FDG PET/MRI-Based Radiomics for Breast Cancer Phenotyping and Tumor Decoding. Cancers 2021, 13, 2928. [Google Scholar] [CrossRef]
- Krajnc, D.; Papp, L.; Nakuz, T.S.; Magometschnigg, H.F.; Grahovac, M.; Spielvogel, C.P.; Ecsedi, B.; Bago-Horvath, Z.; Haug, A.; Karanikas, G.; et al. Breast Tumor Characterization Using [18F]FDG-PET/CT Imaging Combined with Data Preprocessing and Radiomics. Cancers 2021, 13, 1249. [Google Scholar] [CrossRef]
- Antunovic, L.; De Sanctis, R.; Cozzi, L.; Kirienko, M.; Sagona, A.; Torrisi, R.; Tinterri, C.; Santoro, A.; Chiti, A.; Zelic, R.; et al. PET/CT Radiomics in Breast Cancer: Promising Tool for Prediction of Pathological Response to Neoadjuvant Chemotherapy. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1468–1477. [Google Scholar] [CrossRef]
- Li, P.; Wang, X.; Xu, C.; Liu, C.; Zheng, C.; Fulham, M.J.; Feng, D.; Wang, L.; Song, S.; Huang, G. 18F-FDG PET/CT Radiomic Predictors of Pathologic Complete Response (PCR) to Neoadjuvant Chemotherapy in Breast Cancer Patients. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 1116–1126. [Google Scholar] [CrossRef]
- Roy, S.; Whitehead, T.D.; Li, S.; Ademuyiwa, F.O.; Wahl, R.L.; Dehdashti, F.; Shoghi, K.I. Co-Clinical FDG-PET Radiomic Signature in Predicting Response to Neoadjuvant Chemotherapy in Triple-Negative Breast Cancer. Eur. J. Nucl. Med. Mol. Imaging 2021. [Google Scholar] [CrossRef]
- Nakajo, M.; Jinguji, M.; Nakabeppu, Y.; Nakajo, M.; Higashi, R.; Fukukura, Y.; Sasaki, K.; Uchikado, Y.; Natsugoe, S.; Yoshiura, T. Texture Analysis of 18F-FDG PET/CT to Predict Tumour Response and Prognosis of Patients with Esophageal Cancer Treated by Chemoradiotherapy. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Nakajo, M.; Jinguji, M.; Shinaji, T.; Aoki, M.; Tani, A.; Nakabeppu, Y.; Nakajo, M.; Sato, M.; Yoshiura, T. A Pilot Study of Texture Analysis of Primary Tumor [18F]FDG Uptake to Predict Recurrence in Surgically Treated Patients with Non-Small Cell Lung Cancer. Mol. Imaging Biol. 2019, 21, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Krarup, M.M.K.; Nygård, L.; Vogelius, I.R.; Andersen, F.L.; Cook, G.; Goh, V.; Fischer, B.M. Heterogeneity in Tumours: Validating the Use of Radiomic Features on 18F-FDG PET/CT Scans of Lung Cancer Patients as a Prognostic Tool. Radiother. Oncol. 2020, 144, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-W.; Shen, W.-C.; Hsieh, T.-C.; Liang, J.-A.; Hung, Y.-C.; Yeh, L.-S.; Chang, W.-C.; Lin, W.-C.; Yen, K.-Y.; Kao, C.-H. Textural Features of Cervical Cancers on FDG-PET/CT Associate with Survival and Local Relapse in Patients Treated with Definitive Chemoradiotherapy. Sci. Rep. 2018, 8, 11859. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bundschuh, R.A.; Dinges, J.; Neumann, L.; Seyfried, M.; Zsoter, N.; Papp, L.; Rosenberg, R.; Becker, K.; Astner, S.T.; Henninger, M.; et al. Textural Parameters of Tumor Heterogeneity in 18F-FDG PET/CT for Therapy Response Assessment and Prognosis in Patients with Locally Advanced Rectal Cancer. J. Nucl. Med. 2014, 55, 891–897. [Google Scholar] [CrossRef] [Green Version]
- Dittrich, D.; Pyka, T.; Scheidhauer, K.; Lütje, S.; Essler, M.; Bundschuh, R.A. Textural features in FDG-PET/CT can predict outcome in melanoma patients to treatment with Vemurafenib and Ipililumab. Nuklearmedizin 2020, 59, 228–234. [Google Scholar] [CrossRef]
- Burstein, M.D.; Tsimelzon, A.; Poage, G.M.; Covington, K.R.; Contreras, A.; Fuqua, S.A.W.; Savage, M.I.; Osborne, C.K.; Hilsenbeck, S.G.; Chang, J.C.; et al. Comprehensive Genomic Analysis Identifies Novel Subtypes and Targets of Triple-Negative Breast Cancer. Clin. Cancer Res. 2015, 21, 1688–1698. [Google Scholar] [CrossRef] [Green Version]
- Orlhac, F.; Nioche, C.; Soussan, M.; Buvat, I. Understanding Changes in Tumor Texture Indices in PET: A Comparison Between Visual Assessment and Index Values in Simulated and Patient Data. J. Nucl. Med. 2017, 58, 387–392. [Google Scholar] [CrossRef]
- Zhuang, M.; García, D.V.; Kramer, G.M.; Frings, V.; Smit, E.F.; Dierckx, R.; Hoekstra, O.S.; Boellaard, R. Variability and Repeatability of Quantitative Uptake Metrics in 18F-FDG PET/CT of Non–Small Cell Lung Cancer: Impact of Segmentation Method, Uptake Interval, and Reconstruction Protocol. J. Nucl. Med. 2019, 60, 600–607. [Google Scholar] [CrossRef] [Green Version]
- Kaalep, A.; Sera, T.; Oyen, W.; Krause, B.J.; Chiti, A.; Liu, Y.; Boellaard, R. EANM/EARL FDG-PET/CT Accreditation—Summary Results from the First 200 Accredited Imaging Systems. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 412–422. [Google Scholar] [CrossRef] [Green Version]








| Patient Information | Mean [min–max] | SD |
|---|---|---|
| Age | 52.6 [25–90] | 14.7 |
| Tumor characteristics | n = 111 | % |
| Inflammatory tumor | 12 | 10.9 |
| T stage | ||
| T1–T2 | 80 | 71.8 |
| T3–T4 | 31 | 28.2 |
| N stage | ||
| cN0 | 70 | 62.7 |
| cN+ | 41 | 37.3 |
| Ductal carcinoma | 104 | 93.7 |
| mSBR grade | ||
| 2 | 8 | 7.2 |
| 3 | 103 | 92.8 |
| Mitoses (n = 106) | ||
| 1 | 5 | 4.7 |
| 2 | 17 | 16.0 |
| 3 | 84 | 79.2 |
| Associated in situ carcinoma | 25 | 27.5 |
| Ki67 ≥ 20% | 94 | 91.3 |
| Unifocal tumor Multifocal tumor | 86 25 | 77.5 22.5 |
| Lymphovascular emboli | 22 | 21.6 |
| Therapy | n = 111 | % |
| Chemotherapy (CT) | ||
| Adjuvant | 29 | 27.1 |
| Neoadjuvant | 78 | 72.9 |
| Type of CT | ||
| Anthracyclines + taxanes | 81 | 76.4 |
| Without anthracyclines | 3 | 2.8 |
| Platinum salts | 22 | 20.8 |
| Tumor surgery | ||
| Conservative | 73 | 65.8 |
| Radical | 38 | 34.2 |
| Lymph node surgery | ||
| Dissection | 66 | 59.5 |
| Sentinel | 45 | 40.5 |
| Radiotherapy | ||
| Yes | 104 | 94.5 |
| No | 7 | 5.5 |
| (pCR) (n = 74) | ||
| Yes | 22 | 29.7 |
| No | 52 | 70.3 |
| PET/CT imaging | n = 111 | % |
| NPET stage | ||
| N0PET | 61 | 55.0 |
| N+PET | 50 | 45.0 |
| PET Parameters | Mean (SD) | Median (IQR) |
|---|---|---|
| SUVmax | 14.6 (7.6) | 12.8 (9.6–18.3) |
| SUVmean | 5.8 (2.1) | 5.3 (4.5–6.65) |
| SUVpeak | 12.1 (5.9) | 10.9 (7.9–14.2) |
| MTV | 22.57 (46.99) | 6.8 (2.9–18.1) |
| TLG | 191.33 (578.25) | 37.5 (13.65–110.1) |
| Variables | Homogeneity p-Value | MTV p-Value | Entropy p-Value | SRE p-Value |
|---|---|---|---|---|
| T3–T4 vs. T1–T2 | 0.614 | <0.001 | <0.001 | 0.545 |
| cN+ vs. cN0 | 0.403 | <0.001 | 0.027 | 0.713 |
| N+PET vs. N0PET | 0.037 | <0.001 | 0.001 | 0.164 |
| Inflammatory tumor | 0.117 | <0.001 | <0.001 | 0.511 |
| mSBR 3 vs. 2 | 0.004 | 0.321 | 0.020 | 0.044 |
| Unifocal vs. multifocal | 0.522 | 0.094 | 0.661 | 0.666 |
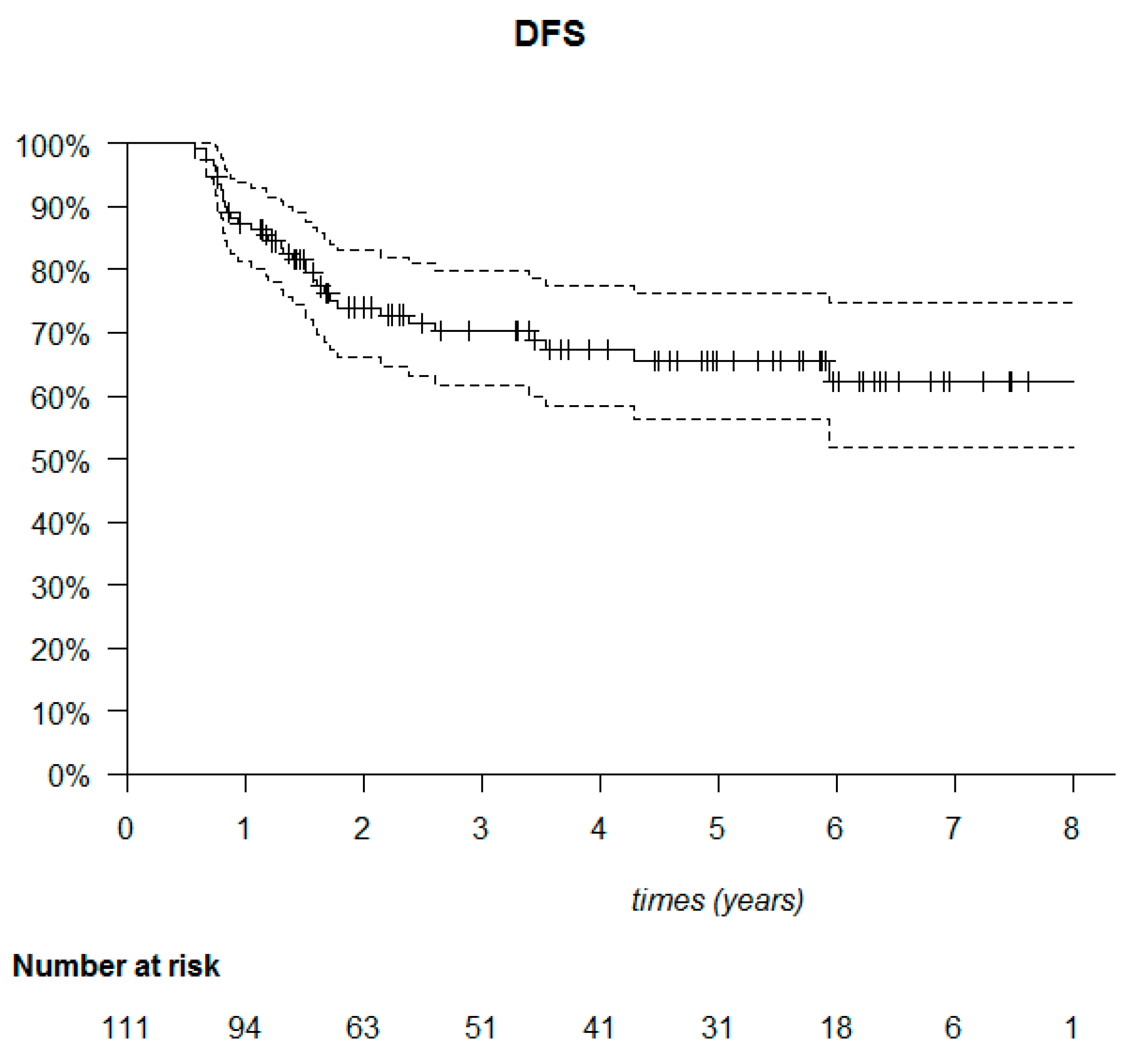
| Variables | DFS | OS | ||
|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | |
| Tumor characteristics | ||||
| Inflammatory: yes | 2.79 (1.14; 6.81) | 0.025 | 4.53 (1.61; 12.78) | 0.004 |
| T3–T4 vs. T1–T2 | 1.56 (0.77; 3.17) | 0.220 | 2.78 (1.16; 6.69) | 0.022 |
| cN+ vs. cN0 | 3.3 (1.62; 6.71) | 0.001 | 2.58 (1.06; 6.32) | 0.038 |
| mSBR 3 vs. 2 | 1.47 (0.35; 6.14) | 0.599 | 1.85 (0.25; 13.84) | 0.549 |
| Associated in situ carcinoma: yes | 1.51 (0.64; 3.53) | 0.343 | 2.27 (0.86; 6.02) | 0.099 |
| Ki67 ≥ 20% vs. Ki 67 < 20% | 0.63 (0.19; 2.1) | 0.452 | 0.69 (0.16; 3.01) | 0.624 |
| Unifocal vs. multifocal tumor | 0.51 (0.25; 1.04) | 0.066 | 0.21 (0.08; 0.5) | <0.001 |
| Therapy | ||||
| NAC vs. adjuvant CT | 1.8 (0.69; 4.69) | 0.226 | 2.78 (0.64; 11.98) | 0.171 |
| Without anthracyclines vs. anthracyclines | 16.38 (4.6; 58.37) | <0.001 | 36.36 (8.28; 159.64) | <0.001 |
| Platin salts vs. anthracyclines | 3.76 (1.73; 8.16) | 0.001 | 8.2 (3.07; 21.91) | <0.001 |
| Radical surgery/conservative surgery | 1.56 (0.79; 3.09) | 0.202 | 4.04 (1.61; 10.14) | 0.003 |
| pCR vs. absence of pCR | 0.17 (0.04; 0.73) | 0.017 | 0.33 (0.07; 1.45) | 0.141 |
| PET/CT Parameters | DFS | OS | ||
|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | |
| N+PET vs. N0PET | 2.91 (1.42; 5.97) | 0.004 | 3.85 (1.4; 10.59) | 0.009 |
| SUVmax | 1.03 (0.99; 1.07) | 0.131 | 1.04 (0.99; 1.09) | 0.153 |
| SUVmean | 1.07 (0.92; 1.24) | 0.361 | 1.11 (0.92; 1.34) | 0.267 |
| SUVpeak | 1.03 (0.98; 1.09) | 0.260 | 1.04 (0.97; 1.12) | 0.232 |
| MTV | 1.39 (1.1; 1.76) | 0.006 | 1.7 (1.24; 2.33) | 0.001 |
| TLG | 1.31 (1.07; 1.6) | 0.008 | 1.53 (1.17; 2.01) | 0.002 |
| Entropy | 2.08 (1.1; 3.96) | 0.025 | 2.46 (1.02; 5.97) | 0.046 |
| Homogeneity | 0.19 (0; 10.58) | 0.421 | 0.13 (0; 30.74) | 0.469 |
| SRE | 7.49 (0; 88,438,904.54) | 0.809 | 6.79 (0; 24,884,680,339.1) | 0.865 |
| LRE | 1.38 (0.03; 57.66) | 0.864 | 2.33 (0.02; 277.48) | 0.729 |
| LGZE | 0.47 (0.18; 1.21) | 0.118 | 0.34 (0.09; 1.25) | 0.105 |
| HGZE | 1.48 (0.96; 2.3) | 0.078 | 1.61 (0.91; 2.86) | 0.101 |
| Parameters | DFS | OS | ||
|---|---|---|---|---|
| p-Value | HR (95% CI) | p-Value | HR (95% CI) | |
| MTV | 0.125 | 0.014 | 1.52 (1.09; 2.12) | |
| TLG | 0.158 | - | ||
| Entropy | 0.192 | - | ||
| SUVmax | - | - | ||
| N+PET vs. N0PET | 0.036 | 2.36 (1.06; 5.24) | 0.074 | |
| Inflam | - | - | ||
| T3–T4 vs. T1–T2 | - | - | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouron, C.; Mathie, C.; Seegers, V.; Morel, O.; Jézéquel, P.; Lasla, H.; Guillerminet, C.; Girault, S.; Lacombe, M.; Sher, A.; et al. Prognostic Value of Metabolic, Volumetric and Textural Parameters of Baseline [18F]FDG PET/CT in Early Triple-Negative Breast Cancer. Cancers 2022, 14, 637. https://doi.org/10.3390/cancers14030637
Bouron C, Mathie C, Seegers V, Morel O, Jézéquel P, Lasla H, Guillerminet C, Girault S, Lacombe M, Sher A, et al. Prognostic Value of Metabolic, Volumetric and Textural Parameters of Baseline [18F]FDG PET/CT in Early Triple-Negative Breast Cancer. Cancers. 2022; 14(3):637. https://doi.org/10.3390/cancers14030637
Chicago/Turabian StyleBouron, Clément, Clara Mathie, Valérie Seegers, Olivier Morel, Pascal Jézéquel, Hamza Lasla, Camille Guillerminet, Sylvie Girault, Marie Lacombe, Avigaelle Sher, and et al. 2022. "Prognostic Value of Metabolic, Volumetric and Textural Parameters of Baseline [18F]FDG PET/CT in Early Triple-Negative Breast Cancer" Cancers 14, no. 3: 637. https://doi.org/10.3390/cancers14030637






