Differences in the Active Endometrial Microbiota across Body Weight and Cancer in Humans and Mice
Abstract
:Simple Summary
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
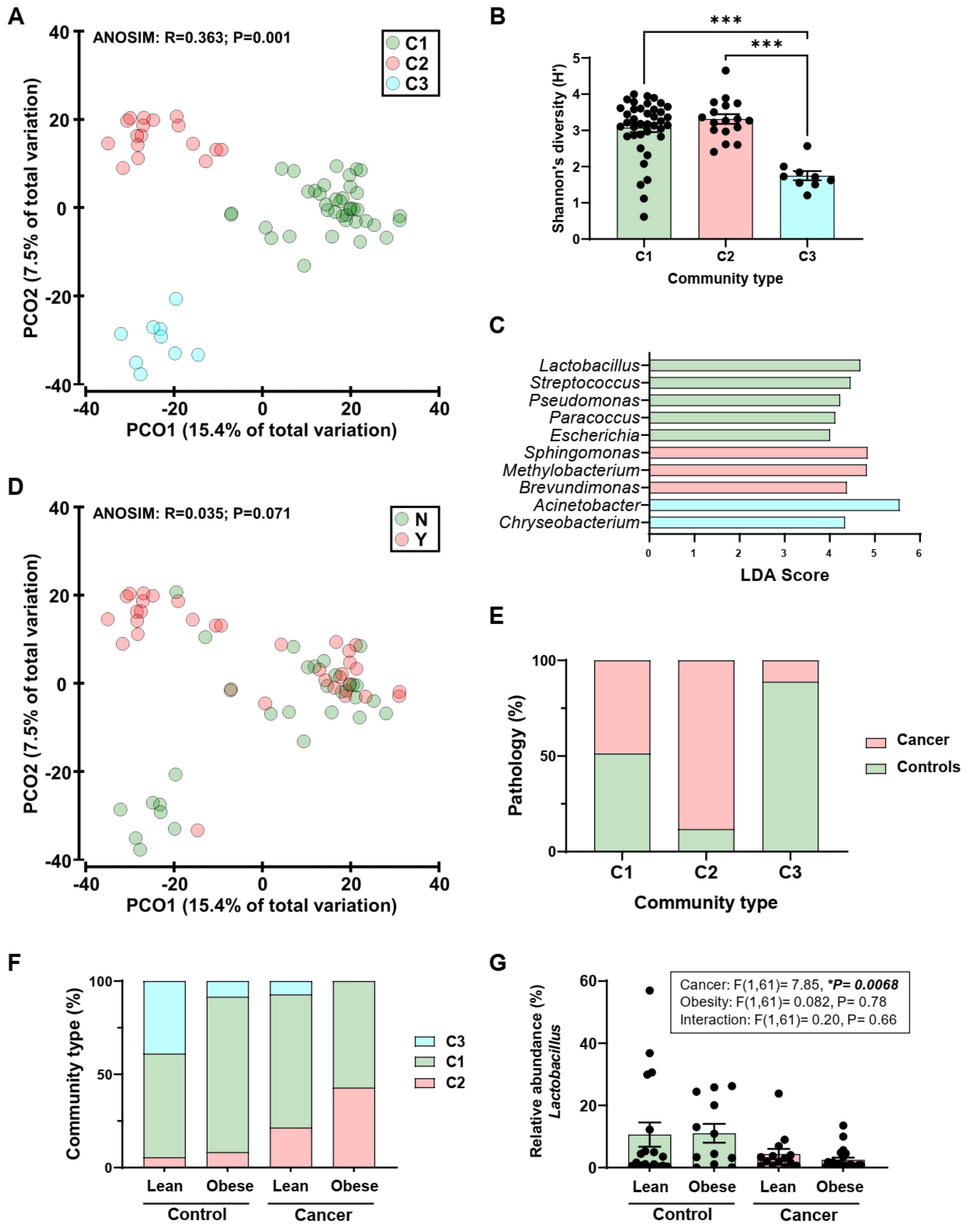
2.2. Human Endometrial Microbiota Is Altered in Cancer
2.3. Obesity Does Not Determine Lactobacillus Abundance in the Uterus
2.4. Endometrial Tumors and Adjacent Normal Endometrium Share Similar Microbiota
2.5. Western Diet-Induced Mouse Model of Obesity
2.6. Mouse Endometrial Microbiota
3. Discussion
4. Methods
4.1. Patient Cohort and Ethics
4.2. Collection of Human Blood and Endometrial Tissues
4.3. Mouse Diet Study
4.4. Mouse Genotyping
4.5. Mouse Metabolic Analyses
4.6. Mouse Uterine Pathology
4.7. 16S rRNA Transcript Amplicon Sequencing and Analysis
4.8. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Dossus, L.; Lukanova, A.; Rinaldi, S.; Allen, N.; Cust, A.E.; Becker, S.; Tjonneland, A.; Hansen, L.; Overvad, K.; Chabbert-Buffet, N. Hormonal, metabolic, and inflammatory profiles and endometrial cancer risk within the EPIC cohort—A factor analysis. Am. J. Epidemiol. 2013, 177, 787–799. [Google Scholar] [CrossRef]
- MacMahon, B. Risk factors for endometrial cancer. Gynecol. Oncol. 1974, 2, 122–129. [Google Scholar] [CrossRef]
- McGonigle, K.F.; Karlan, B.Y.; Barbuto, D.A.; Leuchter, R.S.; Lagasse, L.D.; Judd, H.L. Development of endometrial cancer in women on estrogen and progestin hormone replacement therapy. Gynecol. Oncol. 1994, 55, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Rose, P.G. Endometrial carcinoma. N. Engl. J. Med. 1996, 335, 640–649. [Google Scholar] [CrossRef]
- Bhaskaran, K.; Douglas, I.; Forbes, H.; dos-Santos-Silva, I.; Leon, D.A.; Smeeth, L. Body-mass index and risk of 22 specific cancers: A population-based cohort study of 5.24 million UK adults. Lancet 2014, 384, 755–765. [Google Scholar] [CrossRef] [Green Version]
- Calle, E.E.; Rodriguez, C.; Walker-Thurmond, K.; Thun, M.J. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of US adults. N. Engl. J. Med. 2003, 348, 1625–1638. [Google Scholar] [CrossRef] [Green Version]
- Painter, J.N.; O’Mara, T.A.; Marquart, L.; Webb, P.M.; Attia, J.; Medland, S.E.; Cheng, T.; Dennis, J.; Holliday, E.G.; McEvoy, M.; et al. Genetic Risk Score Mendelian Randomization Shows that Obesity Measured as Body Mass Index, but not Waist:Hip Ratio, Is Causal for Endometrial Cancer. Cancer Epidemiol. Biomark. Prev. 2016, 25, 1503–1510. [Google Scholar] [CrossRef] [Green Version]
- O‘Mara, T.A.; Glubb, D.M.; Kho, P.F.; Thompson, D.J.; Spurdle, A.B. Genome-Wide Association Studies of Endometrial Cancer: Latest Developments and Future Directions. Cancer Epidemiol. Biomark. Amp. Prev. 2019, 28, 1095–1102. [Google Scholar] [CrossRef]
- Avgerinos, K.I.; Spyrou, N.; Mantzoros, C.S.; Dalamaga, M. Obesity and cancer risk: Emerging biological mechanisms and perspectives. Metabolism 2019, 92, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Vucenik, I.; Stains, J.P. Obesity and cancer risk: Evidence, mechanisms, and recommendations. Ann. N. Y. Acad. Sci. 2012, 1271, 37–43. [Google Scholar] [CrossRef]
- Onstad, M.A.; Schmandt, R.E.; Lu, K.H. Addressing the Role of Obesity in Endometrial Cancer Risk, Prevention, and Treatment. J. Clin. Oncol. 2016, 34, 4225–4230. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wang, J. The Role of Metabolic Syndrome in Endometrial Cancer: A Review. Front. Oncol. 2019, 9, 744. [Google Scholar] [CrossRef] [Green Version]
- Łaniewski, P.; Ilhan, Z.E.; Herbst-Kralovetz, M.M. The microbiome and gynaecological cancer development, prevention and therapy. Nat. Rev. Urol. 2020, 17, 232–250. [Google Scholar] [CrossRef]
- Sharma, V.R.; Singh, M.; Kumar, V.; Yadav, M.; Sehrawat, N.; Sharma, D.K.; Sharma, A.K. Microbiome dysbiosis in cancer: Exploring therapeutic strategies to counter the disease. Semin. Cancer Biol. 2021, 70, 61–70. [Google Scholar] [CrossRef]
- Hawkins, G.M.; Burkett, W.C.; McCoy, A.N.; Nichols, H.B.; Olshan, A.F.; Broaddus, R.; Merker, J.D.; Weissman, B.; Brewster, W.R.; Roach, J.; et al. Differences in the microbial profiles of early stage endometrial cancers between Black and White women. Gynecol. Oncol. 2022. [Google Scholar] [CrossRef]
- Walsh, D.M.; Hokenstad, A.N.; Chen, J.; Sung, J.; Jenkins, G.D.; Chia, N.; Nelson, H.; Mariani, A.; Walther-Antonio, M.R.S. Postmenopause as a key factor in the composition of the Endometrial Cancer Microbiome (ECbiome). Sci. Rep. 2019, 9, 19213. [Google Scholar] [CrossRef] [PubMed]
- Castaño-Rodríguez, N.; Goh, K.L.; Fock, K.M.; Mitchell, H.M.; Kaakoush, N.O. Dysbiosis of the microbiome in gastric carcinogenesis. Sci. Rep. 2017, 7, 15957. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, R.; Tun, H.M.; Jahan, M.; Zhang, Z.; Kumar, A.; Dilantha Fernando, W.G.; Farenhorst, A.; Khafipour, E. Comparison of DNA-, PMA-, and RNA-based 16S rRNA Illumina sequencing for detection of live bacteria in water. Sci. Rep. 2017, 7, 5752. [Google Scholar] [CrossRef] [Green Version]
- Carini, P.; Marsden, P.J.; Leff, J.W.; Morgan, E.E.; Strickland, M.S.; Fierer, N. Relic DNA is abundant in soil and obscures estimates of soil microbial diversity. Nat. Microbiol. 2016, 2, 16242. [Google Scholar] [CrossRef]
- Paramsothy, S.; Nielsen, S.; Kamm, M.A.; Deshpande, N.P.; Faith, J.J.; Clemente, J.C.; Paramsothy, R.; Walsh, A.J.; van den Bogaerde, J.; Samuel, D.; et al. Specific Bacteria and Metabolites Associated With Response to Fecal Microbiota Transplantation in Patients With Ulcerative Colitis. Gastroenterology 2019, 156, 1440–1454.e1442. [Google Scholar] [CrossRef] [Green Version]
- Contreras, C.M.; Gurumurthy, S.; Haynie, J.M.; Shirley, L.J.; Akbay, E.A.; Wingo, S.N.; Schorge, J.O.; Broaddus, R.R.; Wong, K.-K.; Bardeesy, N.; et al. Loss of Lkb1 Provokes Highly Invasive Endometrial Adenocarcinomas. Cancer Res. 2008, 68, 759–766. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Contreras, C.M.; Akbay, E.A.; Gallardo, T.D.; Haynie, J.M.; Sharma, S.; Tagao, O.; Bardeesy, N.; Takahashi, M.; Settleman, J.; Wong, K.K.; et al. Lkb1 inactivation is sufficient to drive endometrial cancers that are aggressive yet highly responsive to mTOR inhibitor monotherapy. Dis. Model. Mech. 2010, 3, 181–193. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, K.H.; Wu, W.; Dave, B.; Slomovitz, B.M.; Burke, T.W.; Munsell, M.F.; Broaddus, R.R.; Walker, C.L. Loss of Tuberous Sclerosis Complex-2 Function and Activation of Mammalian Target of Rapamycin Signaling in Endometrial Carcinoma. Clin. Cancer Res. 2008, 14, 2543–2550. [Google Scholar] [CrossRef] [Green Version]
- Slaughter, K.N.; Okon, I.I.; McMeekin, S.; Benbrook, D.; Scott, B.; Moore, K. Expression Profiles of LKB1/AMPK in Endometrial Cancer Specimens as a Potential Biomarker for Targeted Metabolic Drug Therapy. Brachytherapy 2014, 14, 100–101. [Google Scholar] [CrossRef]
- Devlin, M.J.; Robbins, A.; Cosman, M.N.; Moursi, C.A.; Cloutier, A.M.; Louis, L.; Van Vliet, M.; Conlon, C.; Bouxsein, M.L. Differential effects of high fat diet and diet-induced obesity on skeletal acquisition in female C57BL/6J vs. FVB/NJ Mice. Bone Rep. 2018, 8, 204–214. [Google Scholar] [CrossRef]
- Hu, C.C.; Qing, K.; Chen, Y. Diet-Induced Changes in Stearoyl-CoA Desaturase 1 Expression in Obesity-Prone and -Resistant Mice. Obes. Res. 2004, 12, 1264–1270. [Google Scholar] [CrossRef]
- Dutta, S.; Sengupta, P. Men and mice: Relating their ages. Life Sci. 2016, 152, 244–248. [Google Scholar] [CrossRef]
- Passarello, K.; Kurian, S.; Villanueva, V. Endometrial Cancer: An Overview of Pathophysiology, Management, and Care. Semin. Oncol. Nurs. 2019, 35, 157–165. [Google Scholar] [CrossRef]
- Matthews, K.J.; Brock, E.; Cohen, S.A.; Chelmow, D. Hysterectomy in obese patients: Special considerations. Clin. Obs. Gynecol. 2014, 57, 106–114. [Google Scholar] [CrossRef]
- Clark, L.H.; Keku, T.O.; McCoy, N.A.; Hawkins, G.; Bae-Jump, V.L.; Brewster, W.R. Alterations in the uterine microbiome in patients with early endometrial cancer: Variations by ethnicity and obesity. J. Clin. Oncol. 2017, 35, e17114. [Google Scholar] [CrossRef]
- Amabebe, E.; Anumba, D.O.C. The Vaginal Microenvironment: The Physiologic Role of Lactobacilli. Front. Med. 2018, 5, 181. [Google Scholar] [CrossRef] [Green Version]
- Peric, A.; Weiss, J.; Vulliemoz, N.; Baud, D.; Stojanov, M. Bacterial Colonization of the Female Upper Genital Tract. Int. J. Mol. Sci. 2019, 20, 3405. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Molina, N.M.; Sola-Leyva, A.; Saez-Lara, M.J.; Plaza-Diaz, J.; Tubić-Pavlović, A.; Romero, B.; Clavero, A.; Mozas-Moreno, J.; Fontes, J.; Altmäe, S. New Opportunities for Endometrial Health by Modifying Uterine Microbial Composition: Present or Future? Biomolecules 2020, 10, 593. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Winters, A.D.; Romero, R.; Gervasi, M.T.; Gomez-Lopez, N.; Tran, M.R.; Garcia-Flores, V.; Pacora, P.; Jung, E.; Hassan, S.S.; Hsu, C.-D.; et al. Does the endometrial cavity have a molecular microbial signature? Sci. Rep. 2019, 9, 9905. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Song, X.; Wei, W.; Zhong, H.; Dai, J.; Lan, Z.; Li, F.; Yu, X.; Feng, Q.; Wang, Z.; et al. The microbiota continuum along the female reproductive tract and its relation to uterine-related diseases. Nat. Commun. 2017, 8, 875. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, N.; Guo, R.; Wang, J.; Zhou, W.; Ling, Z. Contribution of Lactobacillus iners to Vaginal Health and Diseases: A Systematic Review. Front. Cell. Infect. Microbiol. 2021, 11, 792787. [Google Scholar] [CrossRef]
- Petrova, M.I.; Reid, G.; Vaneechoutte, M.; Lebeer, S. Lactobacillus iners: Friend or Foe? Trends Microbiol. 2017, 25, 182–191. [Google Scholar] [CrossRef]
- Healy, M.E.; Chow, J.D.; Byrne, F.L.; Breen, D.S.; Leitinger, N.; Li, C.; Lackner, C.; Caldwell, S.H.; Hoehn, K.L. Dietary effects on liver tumor burden in mice treated with the hepatocellular carcinogen diethylnitrosamine. J. Hepatol. 2015, 62, 599–606. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oh, H.Y.; Seo, S.-S.; Kong, J.-S.; Lee, J.-K.; Kim, M.K. Association between Obesity and Cervical Microflora Dominated by Lactobacillus iners in Korean Women. J. Clin. Microbiol. 2015, 53, 3304–3309. [Google Scholar] [CrossRef] [Green Version]
- Mitchell, C.M.; Haick, A.; Nkwopara, E.; Garcia, R.; Rendi, M.; Agnew, K.; Fredricks, D.N.; Eschenbach, D. Colonization of the upper genital tract by vaginal bacterial species in nonpregnant women. Am. J. Obstet. Gynecol. 2015, 212, 611.e1–611.e9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rizzatti, G.; Lopetuso, L.R.; Gibiino, G.; Binda, C.; Gasbarrini, A. Proteobacteria: A Common Factor in Human Diseases. Biomed. Res. Int. 2017, 2017, 9351507. [Google Scholar] [CrossRef] [Green Version]
- Walther-António, M.R.S.; Chen, J.; Multinu, F.; Hokenstad, A.; Distad, T.J.; Cheek, E.H.; Keeney, G.L.; Creedon, D.J.; Nelson, H.; Mariani, A.; et al. Potential contribution of the uterine microbiome in the development of endometrial cancer. Genome Med. 2016, 8, 122. [Google Scholar] [CrossRef] [Green Version]
- Li, C.; Gu, Y.; He, Q.; Huang, J.; Song, Y.; Wan, X.; Li, Y. Integrated Analysis of Microbiome and Transcriptome Data Reveals the Interplay Between Commensal Bacteria and Fibrin Degradation in Endometrial Cancer. Front. Cell. Infect. Microbiol. 2021, 11, 748558. [Google Scholar] [CrossRef] [PubMed]
- Medina-Bastidas, D.; Camacho-Arroyo, I.; García-Gómez, E. Current findings in endometrial microbiome: Impact on uterine diseases. Reproduction 2022, 163, R81–R96. [Google Scholar] [CrossRef]
- Zhou, B.; Sun, C.; Huang, J.; Xia, M.; Guo, E.; Li, N.; Lu, H.; Shan, W.; Wu, Y.; Li, Y.; et al. The biodiversity Composition of Microbiome in Ovarian Carcinoma Patients. Sci. Rep. 2019, 9, 1691. [Google Scholar] [CrossRef]
- Moreno, I.; Simon, C. Relevance of assessing the uterine microbiota in infertility. Fertil. Steril. 2018, 110, 337–343. [Google Scholar] [CrossRef] [Green Version]
- Alexopoulos, S.J.; Chen, S.Y.; Brandon, A.E.; Salamoun, J.M.; Byrne, F.L.; Garcia, C.J.; Beretta, M.; Olzomer, E.M.; Shah, D.P.; Philp, A.M.; et al. Mitochondrial uncoupler BAM15 reverses diet-induced obesity and insulin resistance in mice. Nat. Commun. 2020, 11, 2397. [Google Scholar] [CrossRef]
- Truett, G.E.; Heeger, P.; Mynatt, R.L.; Truett, A.A.; Walker, J.A.; Warman, M.L. Preparation of PCR-quality mouse genomic DNA with hot sodium hydroxide and tris (HotSHOT). Biotechniques 2000, 29, 52–54. [Google Scholar] [CrossRef] [PubMed]
- Haifer, C.; Paramsothy, S.; Borody, T.J.; Clancy, A.; Leong, R.W.; Kaakoush, N.O. Long-Term Bacterial and Fungal Dynamics following Oral Lyophilized Fecal Microbiota Transplantation in Clostridioides difficile Infection. mSystems 2021, 6, e00905-20. [Google Scholar] [CrossRef]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; et al. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kozich, J.J.; Westcott, S.L.; Baxter, N.T.; Highlander, S.K.; Schloss, P.D. Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the MiSeq Illumina sequencing platform. Appl. Environ. Microbiol. 2013, 79, 5112–5120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic biomarker discovery and explanation. Genome Biol. 2011, 12, R60. [Google Scholar] [CrossRef] [Green Version]



| Characteristics: Median or n (%) | Lean Cancer (n = 17) | Obese Cancer (n = 23) | Lean Control (n = 18) | Obese Control (n = 12) | p-Value m |
|---|---|---|---|---|---|
| Age (years) | 69.0 a | 68.0 | 60.5 a | 59.5 | 0.003 |
| BMI (kg/m2) | 23.74 b | 38.20 b | 23.65 b | 31.95 b | <0.0001 |
| Stage | |||||
| I A | 10 (58.8%) | 15 (65.2%) | N/A | N/A | |
| I B | 4 (23.5%) | 3 (13%) | |||
| II A | 0 (0.0%) | 1 (4.3%) | |||
| III A | 0 (0.0%) | 1 (4.3%) | |||
| III C | 1 (5.9%) | 2 (8.7%) | |||
| IV A | 1 (5.9%) | 0 (0.0%) | |||
| IV B | 1 (5.9%) | 1 (4.3%) | |||
| Grade | N/A | N/A | |||
| 1 | 13 (76.5%) | 14 (60.9%) | |||
| 2 | 3 (17.6%) | 8 (34.8%) | |||
| 3 | 1 (5.9%) | 1 (4.3%) | |||
| Parity | |||||
| 0 | 5 (29.4%) | 6 (26.1%) | 6 (33.3%) | 3 (25.0%) | |
| 1 or 2 | 9 (52.9%) | 6 (26.1%) | 8 (44.4%) | 6 (50.0%) | |
| 3 or more | 3 (17.6%) | 11 (47.8%) | 4 (22.2%) | 3 (25.0%) | |
| Gravidity | |||||
| 0 | 4 (23.5%) | 7 (30.4%) | 4 (22.2%) | 2 (16.7%) | |
| 1 or 2 | 8 (47.1%) | 4 (17.4%) | 11 (61.1%) | 6 (50.0%) | |
| 3 or 4 | 2 (11.8%) | 10 (43.5%) | 3 (16.7%) | 3 (25.0%) | |
| 5 or more | 3 (17.6%) | 2 (8.7%) | 0 (0.0%) | 1 (8.3%) | |
| Age at Menarche (years) | 13.5 (n = 16) | 13.0 | 13.0 (n = 17) | 13.0 | 0.567 |
| Age at Menopause (years) | 51.0 | 52.0 | 52.0 (n = 15) | 50.5 | 0.681 |
| Any systemic HRT history | (n = 15) | ||||
| 8 (53.3%) | 4 (17.4%) | 7 (38.9%) | 3 (25.0%) | ||
| Years on systemic HRT | 0.50 (n = 15) | 0.00 | 0.00 | 0.00 | 0.055 |
| No OCP History | (n = 14) | (n = 26) | (n = 18) | ||
| 6 (42.9%) | 9 (40.9%) | 7 (43.8%) | 6 (50.0%) | ||
| Years on OCP | 1.25 (n = 14) | 2.00 (n = 21) | 4.50 (n = 16) | 0.50 | 0.839 |
| Comorbidities | |||||
| Type 2 Diabetes | 0 (0.0%) | 5 (21.7%) | 0 (0.0%) | 4 (33.3%) | |
| Hyperlipidemia | 5 (29.4%) | 6 (26.1%) | 2 (11.1%) | 8 (66.7%) | |
| Hypertension | 6 (35.3%) | 18 (78.3%) | 1 (5.6%) | 6 (50.0%) | |
| Hypothyroidism | 4 (23.5%) | 3 (13.0%) | 4 (22.2%) | 2 (16.7%) | |
| Regular Medications | 12 (70.6%) | 19 (82.6%) | 11 (61.1%) | 9 (75.0%) | |
| Tobacco Use | (n = 21) | (n = 10) | |||
| Never | 10 (58.8%) | 11 (52.4%) | 16 (88.9%) | 8 (80.0%) | |
| Current | 2 (11.8%) | 2 (9.5%) | 0 (0.0%) | 1 (10.0%) | |
| Previous | 5 (29.4%) | 8 (38.1%) | 2 (11.1%) | 1 (10.0%) | |
| Pack Years | 0.00 | 0.00 (n = 21) | 0.00 | 0.00 (n = 10) | 0.171 |
| Glucose (mmol/L) | 4.70 c | 5.20 (n = 23) | 5.10 | 5.60 c | 0.013 |
| Insulin (μU/mL) | 4.30 d (n = 15) | 9.50 de (n = 22) | 4.80 e (n = 15) | 7.70 (n = 10) | 0.002 |
| HbA1c IFCC (mmol/mol) | 37.0 (n = 16) | 39.0 f | 34.5 fg | 40.0 g | 0.003 |
| HbA1c NGSP (%) | 5.55 (n = 16) | 5.80 h | 5.35 h | 5.80 | 0.005 |
| HDL (mmol/L) | 1.90 (n = 16) | 1.30 (n = 20) | 1.65 | 1.30 | 0.043 |
| LDL (mmol/L) | 2.80 (n = 16) | 2.85 (n = 20) | 2.45 | 1.90 | 0.233 |
| Triglycerides (mmol/L) | 1.45 (n = 8) | 1.35 (n = 20) | 1.30 (n = 15) | 1.60 (n = 11) | 0.554 |
| CRP (mg/L) | 1 i (n = 16) | 4 ij (n = 22) | 1.5 j | 3 | 0.003 |
| Estradiol (pmol/L) | 19.0 k (n = 13) | 39.5 kl (n = 20) | 16.0 l (n = 17) | 31.0 g (n = 11) | 0.005 |
| Pathology Results, n (%) | ||||||
|---|---|---|---|---|---|---|
| Genotype | Diet | Mice (n) | Normal | Hyperplasia | Atypical Hyperplasia | Cancer |
| Lkb1+/+ | Chow | 4 | 1 (25%) | 2 (50%) | 1 (25%) | |
| Lkb1+/+ | Western | 4 | 1 (25%) | 3 (75%) | ||
| Sprr2f-Cre; Lkb1L/+ | Chow | 24 | 3 (12.5%) | 6 (25%) | 14 (58.3%) | 1 (4.2%) |
| Sprr2f-Cre; Lkb1L/+ | Western | 29 | 8 (27.6%) | 7 (24.1%) | 13 (44.8%) | 1 (3.5%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaakoush, N.O.; Olzomer, E.M.; Kosasih, M.; Martin, A.R.; Fargah, F.; Lambie, N.; Susic, D.; Hoehn, K.L.; Farrell, R.; Byrne, F.L. Differences in the Active Endometrial Microbiota across Body Weight and Cancer in Humans and Mice. Cancers 2022, 14, 2141. https://doi.org/10.3390/cancers14092141
Kaakoush NO, Olzomer EM, Kosasih M, Martin AR, Fargah F, Lambie N, Susic D, Hoehn KL, Farrell R, Byrne FL. Differences in the Active Endometrial Microbiota across Body Weight and Cancer in Humans and Mice. Cancers. 2022; 14(9):2141. https://doi.org/10.3390/cancers14092141
Chicago/Turabian StyleKaakoush, Nadeem O., Ellen M. Olzomer, Melidya Kosasih, Amy R. Martin, Farokh Fargah, Neil Lambie, Daniella Susic, Kyle L. Hoehn, Rhonda Farrell, and Frances L. Byrne. 2022. "Differences in the Active Endometrial Microbiota across Body Weight and Cancer in Humans and Mice" Cancers 14, no. 9: 2141. https://doi.org/10.3390/cancers14092141







